Formulation and Evaluation of a Clove Oil-Encapsulated Nanofiber Formulation for Effective Wound-Healing
Abstract
1. Introduction
2. Results and Discussion
2.1. Constituents of Clove Essential Oil
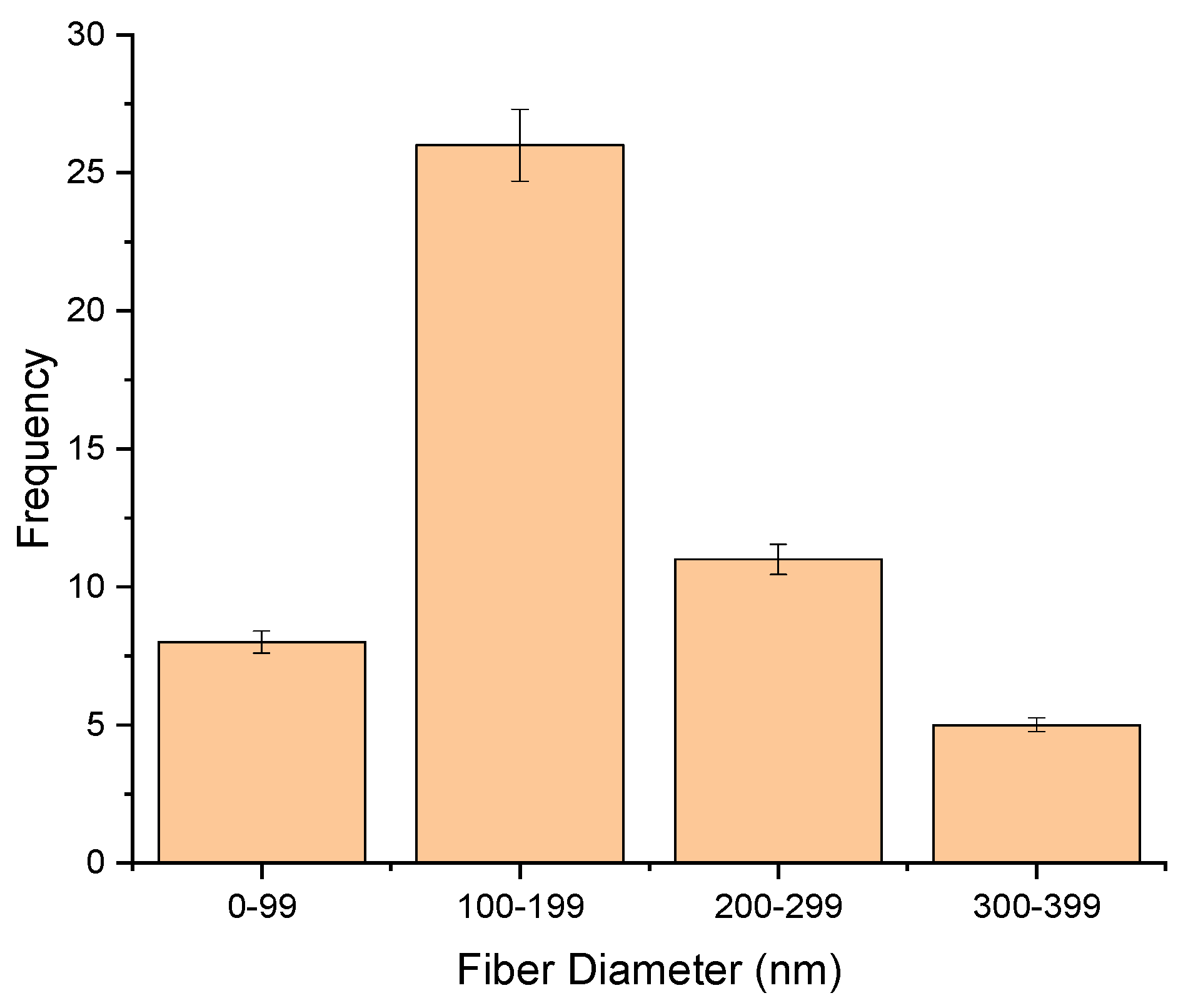
2.2. Morphology and Fiber Diameter
2.3. FTIR Analysis
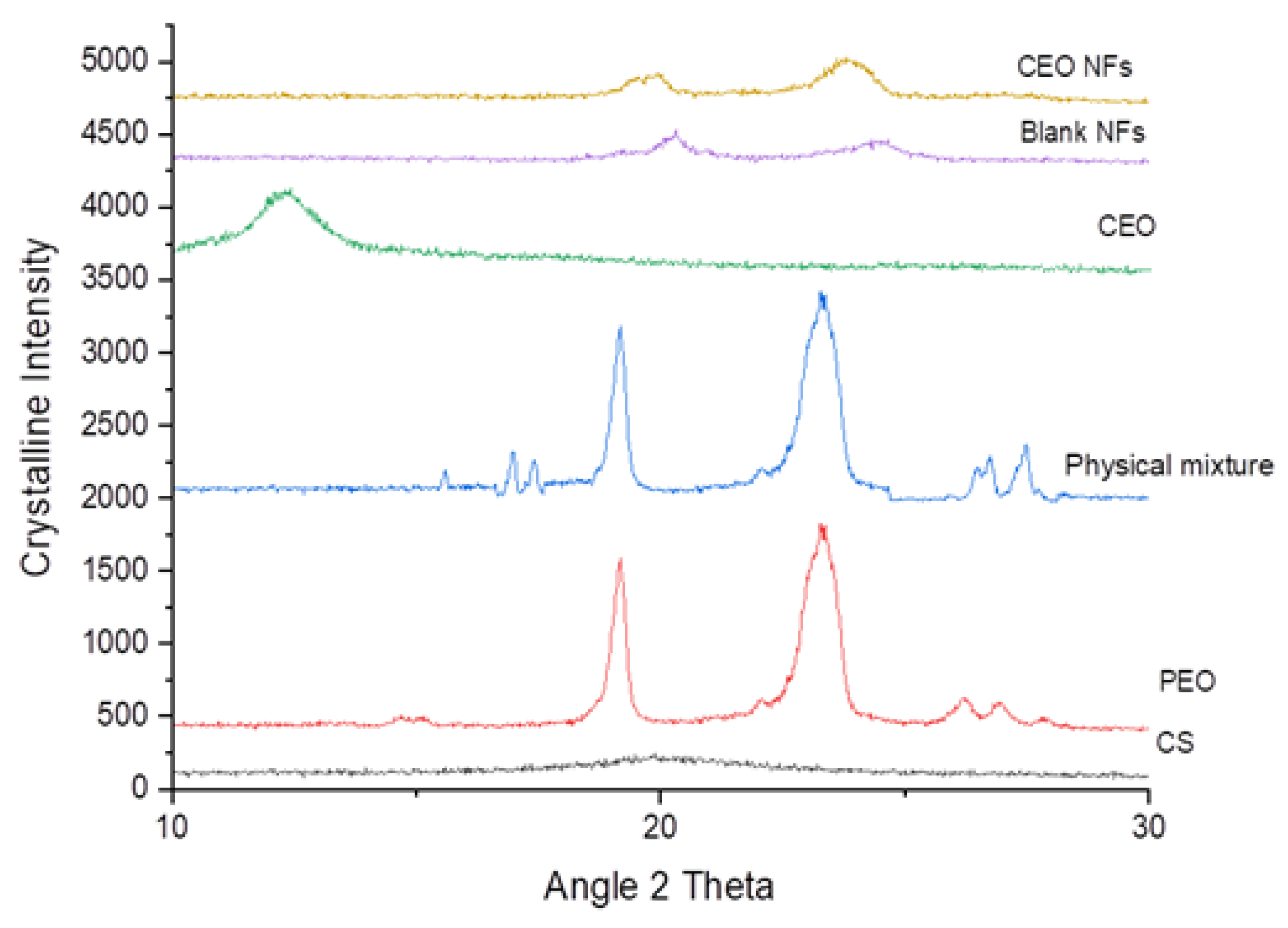
2.4. XRD Analysis
2.5. Percentage Oil Content, Drug-Loading Efficiency (DLE%), and Percentage Yield
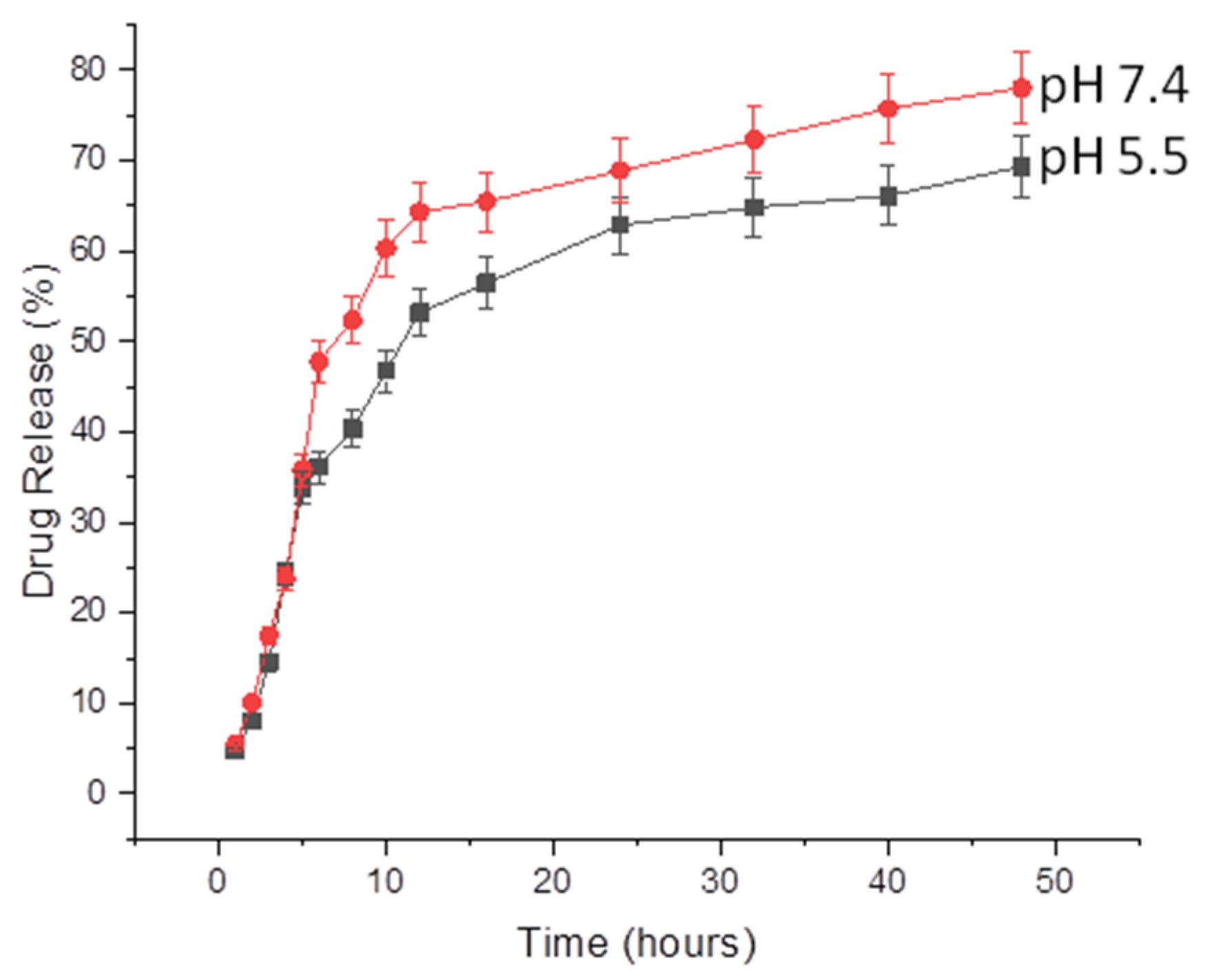
2.6. Release of CEO from NFs
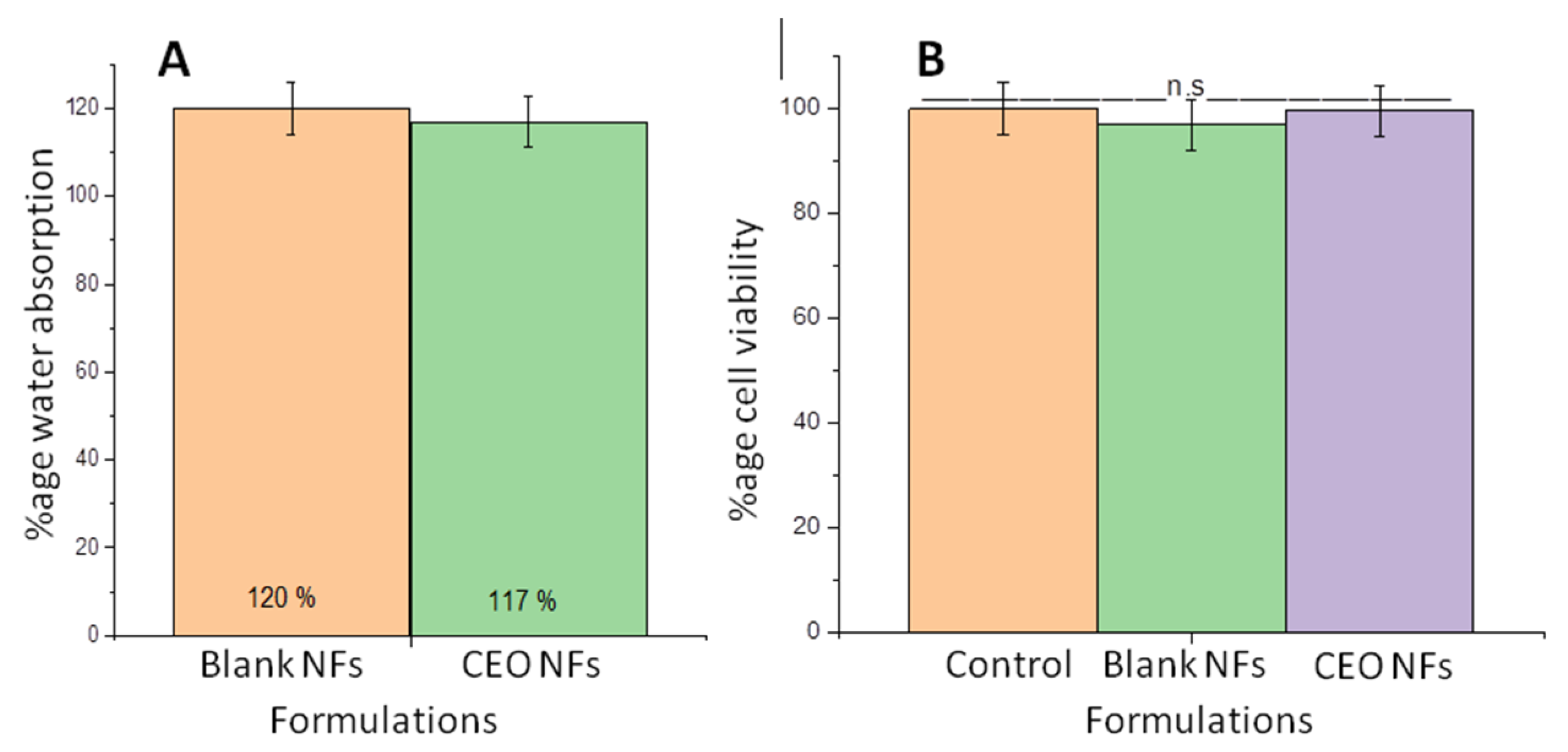
2.7. Water Absorption Capacity
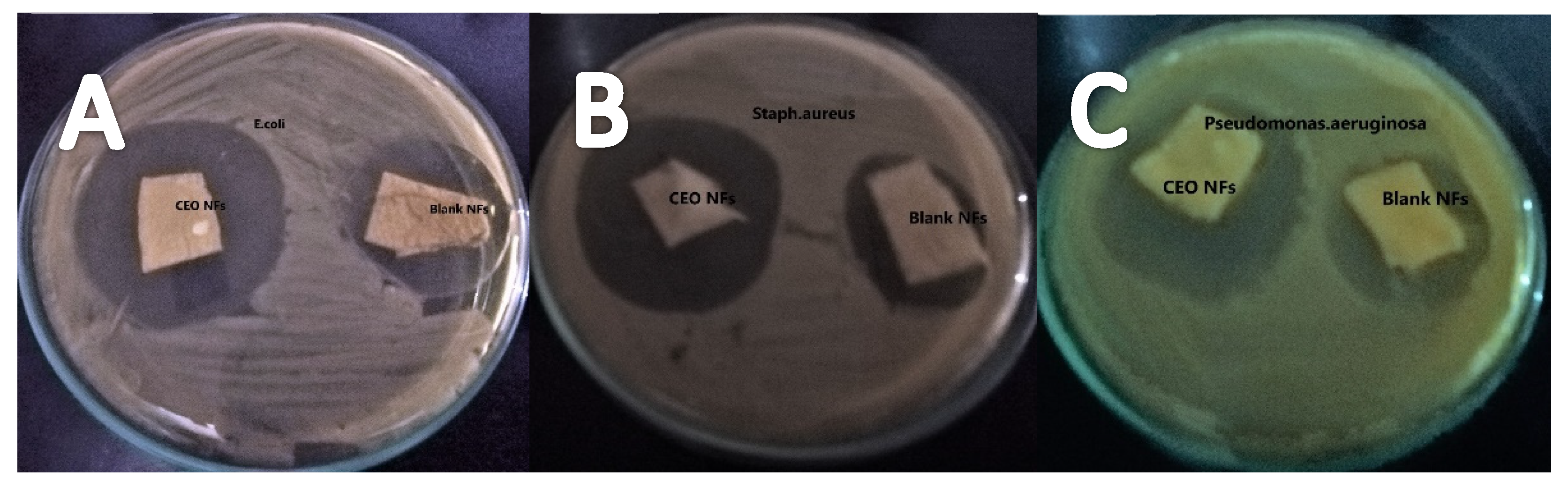
2.8. Antibacterial Activity
2.9. Cytotoxicity Evaluation
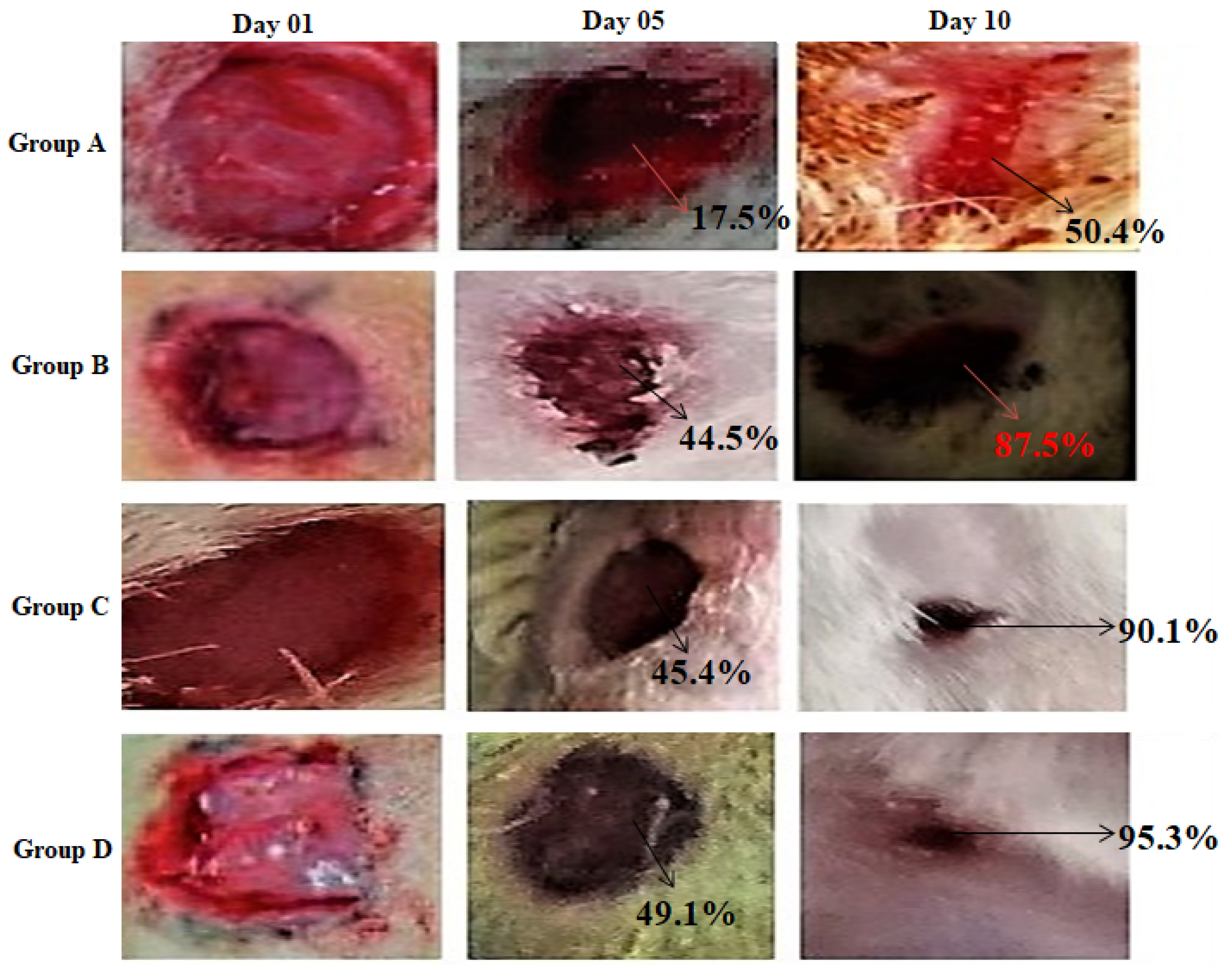
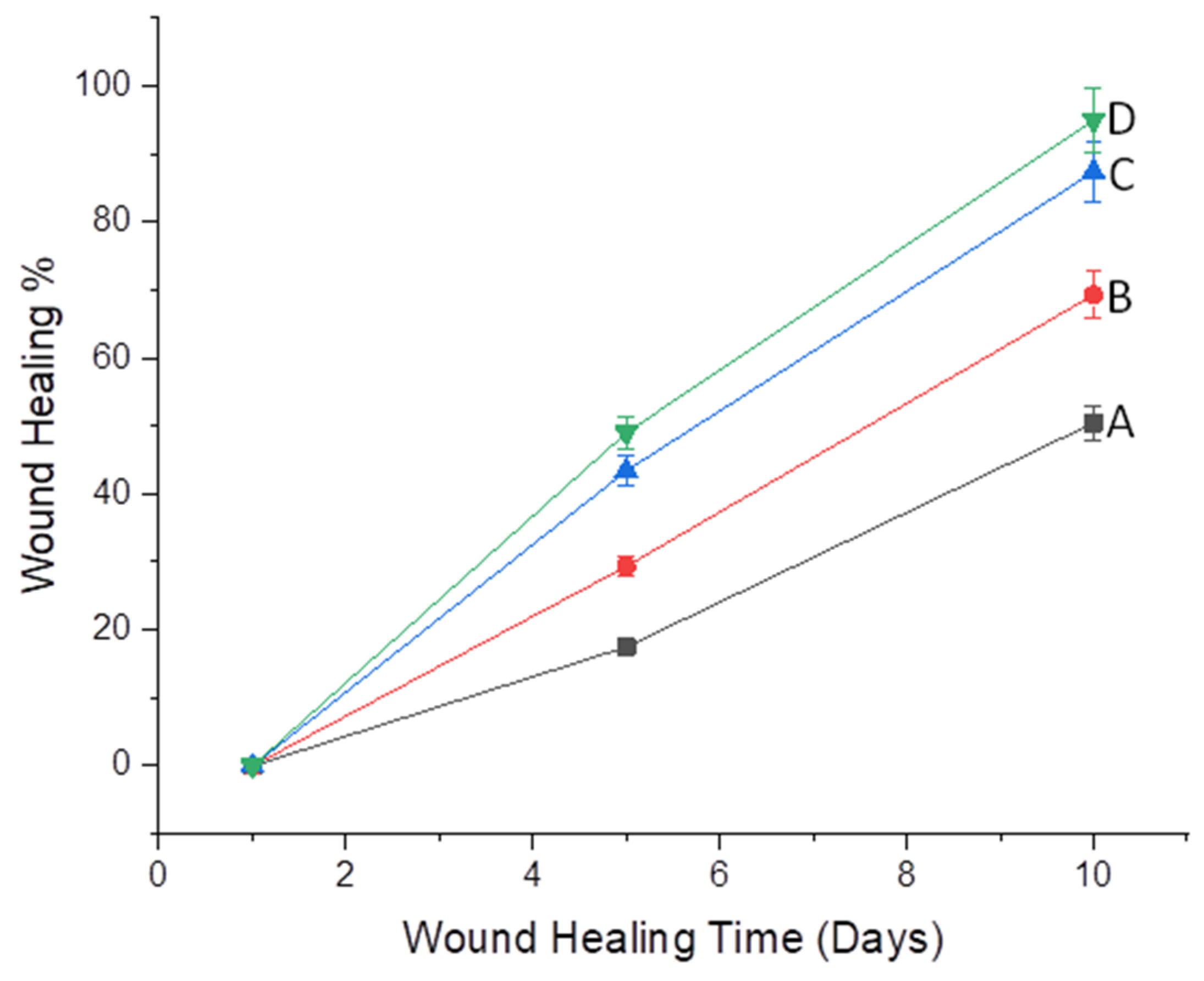
2.10. Wound-Healing Potential
3. Materials and Methods
3.1. Materials
3.2. Methodology
3.3. Electrospinning Solution
3.4. Fiber Formation
3.5. Solution and Process Variables
3.6. Surface Morphology and Average Fiber Diameter
3.7. Fourier Transform Infra-Red Spectroscopy
3.8. X-ray Diffraction
3.9. Degree of Swelling and Weight Loss
3.10. Percentage Oil Content, Drug-Loading Efficiency (DLE%), and Percentage Yield
3.11. Drug-Release Studies
3.12. In Vitro Cytotoxicity
3.13. Antibacterial Activity
3.14. In Vivo Wound-Healing
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Edwards, R.; Harding, K.G. Bacteria and wound healing. Curr. Opin. Infect. Dis. 2004, 17, 91–96. [Google Scholar] [CrossRef]
- MacNeil, S. Biomaterials for tissue engineering of skin. Mater. Today 2008, 11, 26–35. [Google Scholar] [CrossRef]
- Silva, N.C.C.; Fernandes Júnior, A. Biological properties of medicinal plants: A review of their antimicrobial activity. J. Venom. Anim. Toxins Incl. Trop. Dis. 2010, 16, 402–413. [Google Scholar] [CrossRef]
- Nuñez, L.; D’Aquino, M. Microbicide activity of clove essential oil (Eugenia caryophyllata). Braz. J. Microbiol. 2012, 43, 1255–1260. [Google Scholar] [CrossRef] [PubMed]
- Pramod, K.; Ansari, S.H.; Ali, J. Eugenol: A natural compound with versatile pharmacological actions. Nat. Prod. Commun. 2010, 5, 1934578X1000501236. [Google Scholar] [CrossRef]
- Milind, P.; Deepa, K. Clove: A champion spice. Int. J. Res. Ayurveda Pharm. 2011, 2, 47–54. [Google Scholar]
- Jun-xia, X.; Hai-yan, Y.; Jian, Y. Microencapsulation of sweet orange oil by complex coacervation with soybean protein isolate/gum Arabic. Food Chem. 2011, 125, 1267–1272. [Google Scholar] [CrossRef]
- Chaieb, K.; Hajlaoui, H.; Zmantar, T.; Kahla-Nakbi, A.B.; Rouabhia, M.; Mahdouani, K.; Bakhrouf, A. The chemical composition and biological activity of clove essential oil, Eugenia caryophyllata (Syzigium aromaticum L. Myrtaceae): A short review. Phytother. Res. Int. J. Devoted Pharmacol. Toxicol. Eval. Nat. Prod. Deriv. 2007, 21, 501–506. [Google Scholar] [CrossRef] [PubMed]
- Rafiq, M.; Hussain, T.; Abid, S.; Nazir, A.; Masood, R. Development of sodium alginate/PVA antibacterial nanofibers by the incorporation of essential oils. Mater. Res. Express 2018, 5, 035007. [Google Scholar] [CrossRef]
- Unalan, I.; Slavik, B.; Buettner, A.; Goldmann, W.H.; Frank, G.; Boccaccini, A.R. Physical and antibacterial properties of peppermint essential oil loaded poly (ε-caprolactone)(PCL) electrospun fiber mats for wound healing. Front. Bioeng. Biotechnol. 2019, 7, 346. [Google Scholar] [CrossRef]
- Alam, P.; Ansari, M.J.; Anwer, M.K.; Raish, M.; Kamal, Y.K.; Shakeel, F. Wound healing effects of nanoemulsion containing clove essential oil. Artif. Cellsnanomed. Biotechnol. 2017, 45, 591–597. [Google Scholar] [CrossRef]
- Khunkitti, W.; Veerapan, P.; Hahnvajanawong, C. In vitro bioactivities of clove buds oil (Eugenia caryophyllata) and its effect on dermal fibroblast. Int. J. Pharm. Pharm. Sci. 2012, 4, 556–560. [Google Scholar]
- Tonglairoum, P.; Ngawhirunpat, T.; Rojanarata, T.; Kaomongkolgit, R.; Opanasopit, P. Fabrication and evaluation of nanostructured herbal oil/hydroxypropyl-β-cyclodextrin/polyvinylpyrrolidone mats for denture stomatitis prevention and treatment. AAPS Pharmscitech 2016, 17, 1441–1449. [Google Scholar] [CrossRef]
- Bakar, L.; Abdullah, M.; Doolaanea, A.; Ichwan, S. PLGA-chitosan nanoparticle-mediated gene delivery for oral cancer treatment: A brief review. J. Phys. 2017, 884, 012117. [Google Scholar]
- Yuan, Y.; Chesnutt, B.M.; Haggard, W.O.; Bumgardner, J.D. Deacetylation of chitosan: Material characterization and in vitro evaluation via albumin adsorption and pre-osteoblastic cell cultures. Materials 2011, 4, 1399–1416. [Google Scholar] [CrossRef]
- Rabea, E.I.; Badawy, M.E.-T.; Stevens, C.V.; Smagghe, G.; Steurbaut, W. Chitosan as antimicrobial agent: Applications and mode of action. Biomacromolecules 2003, 4, 1457–1465. [Google Scholar] [CrossRef]
- Gonçalves, R.P.; Ferreira, W.H.; Gouvêa, R.F.; Andrade, C.T. Effect of chitosan on the properties of electrospun fibers from mixed poly (vinyl alcohol)/chitosan solutions. Mater. Res. 2017, 20, 984–993. [Google Scholar] [CrossRef]
- Qasim, S.B.; Zafar, M.S.; Najeeb, S.; Khurshid, Z.; Shah, A.H.; Husain, S.; Rehman, I.U. Electrospinning of chitosan-based solutions for tissue engineering and regenerative medicine. Int. J. Mol. Sci. 2018, 19, 407. [Google Scholar] [CrossRef]
- Alavarse, A.C.; de Oliveira Silva, F.W.; Colque, J.T.; da Silva, V.M.; Prieto, T.; Venancio, E.C.; Bonvent, J.-J. Tetracycline hydrochloride-loaded electrospun nanofibers mats based on PVA and chitosan for wound dressing. Mater. Sci. Eng. C 2017, 77, 271–281. [Google Scholar] [CrossRef]
- Veleirinho, B.; Berti, F.V.; Dias, P.F.; Maraschin, M.; Ribeiro-do-Valle, R.M.; Lopes-da-Silva, J.A. Manipulation of chemical composition and architecture of non-biodegradable poly (ethylene terephthalate)/chitosan fibrous scaffolds and their effects on L929 cell behavior. Mater. Sci. Eng. C 2013, 33, 37–46. [Google Scholar] [CrossRef]
- Cramariuc, B.; Cramariuc, R.; Scarlet, R.; Manea, L.R.; Lupu, I.G.; Cramariuc, O. Fiber diameter in electrospinning process. J. Electrost. 2013, 71, 189–198. [Google Scholar] [CrossRef]
- García-Moreno, P.J.; Stephansen, K.; van der Kruijs, J.; Guadix, A.; Guadix, E.M.; Chronakis, I.S.; Jacobsen, C. Encapsulation of fish oil in nanofibers by emulsion electrospinning: Physical characterization and oxidative stability. J. Food Eng. 2016, 183, 39–49. [Google Scholar] [CrossRef]
- Mori, C.L.; dos Passos, N.A.; Oliveira, J.E.; Altoé, T.F.; Mori, F.A.; Mattoso, L.H.C.; Scolforo, J.R.; Tonoli, G.H.D. Nanostructured polylactic acid/candeia essential oil mats obtained by electrospinning. J. Nanomater. 2015, 2015. [Google Scholar] [CrossRef]
- Semwal, A.; Ramandeep, S. Preparation, characterization and biological evaluation of chitosanmoxifloxacin prodrugs for pharmaceuticals. Marmara Pharm. J. 2014, 18, 36–42. [Google Scholar] [CrossRef]
- Ramesh, S.; Yuen, T.F.; Shen, C.J. Conductivity and FTIR studies on PEO–LiX [X: CF3SO3−, SO42−] polymer electrolytes. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2008, 69, 670–675. [Google Scholar] [CrossRef] [PubMed]
- Senak, L.; Davies, M.A.; Mendelsohn, R. A quantitative IR study of hydrocarbon chain conformation in alkanes and phospholipids: CH2 wagging modes in disordered bilayer and HII phases. J. Phys. Chem. 1991, 95, 2565–2571. [Google Scholar] [CrossRef]
- Hosseini, S.F.; Zandi, M.; Rezaei, M.; Farahmandghavi, F. Two-step method for encapsulation of oregano essential oil in chitosan nanoparticles: Preparation, characterization and in vitro release study. Carbohydr. Polym. 2013, 95, 50–56. [Google Scholar] [CrossRef] [PubMed]
- Shetta, A.; Kegere, J.; Mamdouh, W. Comparative study of encapsulated peppermint and green tea essential oils in chitosan nanoparticles: Encapsulation, thermal stability, in-vitro release, antioxidant and antibacterial activities. Int. J. Biol. Macromol. 2019, 126, 731–742. [Google Scholar] [CrossRef] [PubMed]
- Tønnesen, H.H.; Karlsen, J. Alginate in drug delivery systems. Drug Dev. Ind. Pharm. 2002, 28, 621–630. [Google Scholar] [CrossRef]
- Marques, H.M.C. A review on cyclodextrin encapsulation of essential oils and volatiles. Flavour Fragr. J. 2010, 25, 313–326. [Google Scholar] [CrossRef]
- Huerta, R.R.; Silva, E.K.; El-Bialy, T.; Saldaña, M.D. Clove essential oil emulsion-filled cellulose nanofiber hydrogel produced by high-intensity ultrasound technology for tissue engineering applications. Ultrason. Sonochem. 2020, 64, 104845. [Google Scholar] [CrossRef]
- Aman, R.M.; Hashim, I.I.A.; Meshali, M.M. Novel Clove Essential Oil Nanoemulgel Tailored by Taguchi’s Model and Scaffold-Based Nanofibers: Phytopharmaceuticals with Promising Potential as Cyclooxygenase-2 Inhibitors in External Inflammation. Int. J. Nanomed. 2020, 15, 2171. [Google Scholar] [CrossRef]
- Unalan, I.; Endlein, S.J.; Slavik, B.; Buettner, A.; Goldmann, W.H.; Detsch, R.; Boccaccini, A.R. Evaluation of electrospun poly (ε-caprolactone)/gelatin nanofiber mats containing clove essential oil for antibacterial wound dressing. Pharmaceutics 2019, 11, 570. [Google Scholar] [CrossRef]
- Cui, H.; Bai, M.; Rashed, M.M.; Lin, L. The antibacterial activity of clove oil/chitosan nanoparticles embedded gelatin nanofibers against Escherichia coli O157: H7 biofilms on cucumber. Int. J. Food Microbiol. 2018, 266, 69–78. [Google Scholar] [CrossRef]
- Ghorbani, F.M.; Kaffashi, B.; Shokrollahi, P.; Seyedjafari, E.; Ardeshirylajimi, A. PCL/chitosan/Zn-doped nHA electrospun nanocomposite scaffold promotes adipose derived stem cells adhesion and proliferation. Carbohydr. Polym. 2015, 118, 133–142. [Google Scholar] [CrossRef] [PubMed]
- Lambers, H.; Piessens, S.; Bloem, A.; Pronk, H.; Finkel, P. Natural skin surface pH is on average below 5, which is beneficial for its resident flora. Int. J. Cosmet. Sci. 2006, 28, 359–370. [Google Scholar] [CrossRef] [PubMed]
- Abid, S.; Hussain, T.; Nazir, A.; Zahir, A.; Ramakrishna, S.; Hameed, M.; Khenoussi, N. Enhanced antibacterial activity of PEO-chitosan nanofibers with potential application in burn infection management. Int. J. Biol. Macromol. 2019, 135, 1222–1236. [Google Scholar] [CrossRef]
- Ayoola, G.; Lawore, F.; Adelowotan, T.; Aibinu, I.; Adenipekun, E.; Coker, H.; Odugbemi, T. Chemical analysis and antimicrobial activity of the essential oil of Syzigium aromaticum (clove). Afr. J. Microbiol. Res. 2008, 2, 162–166. [Google Scholar]
- Liu, Y.; Wang, S.; Zhang, R.; Lan, W.; Qin, W. Development of poly (lactic acid)/chitosan fibers loaded with essential oil for antimicrobial applications. Nanomaterials 2017, 7, 194. [Google Scholar] [CrossRef] [PubMed]
- Agnes Mary, S.; Giri Dev, V. Electrospun herbal nanofibrous wound dressings for skin tissue engineering. J. Text. Inst. 2015, 106, 886–895. [Google Scholar] [CrossRef]
- Tang, Y.; Zhou, Y.; Lan, X.; Huang, D.; Luo, T.; Ji, J.; Mafang, Z.; Miao, X.; Wang, H.; Wang, W. Electrospun gelatin nanofibers encapsulated with peppermint and chamomile essential oils as potential edible packaging. J. Agric. Food Chem. 2019, 67, 2227–2234. [Google Scholar] [CrossRef]
- Schiffman, J.D.; Schauer, C.L. A review: Electrospinning of biopolymer nanofibers and their applications. Polym. Rev. 2008, 48, 317–352. [Google Scholar] [CrossRef]
- Pillay, V.; Dott, C.; Choonara, Y.E.; Tyagi, C.; Tomar, L.; Kumar, P.; du Toit, L.C.; Ndesendo, V.M. A review of the effect of processing variables on the fabrication of electrospun nanofibers for drug delivery applications. J. Nanomater. 2013. [Google Scholar] [CrossRef]
- Kesici Güler, H.; Cengiz Çallıoğlu, F.; Sesli Çetin, E. Antibacterial PVP/cinnamon essential oil nanofibers by emulsion electrospinning. J. Text. Inst. 2019, 110, 302–310. [Google Scholar] [CrossRef]
- da Silva Campelo, M.; Melo, E.O.; Arrais, S.P.; do Nascimento, F.B.S.A.; Gramosa, N.V.; de Aguiar Soares, S.; Ribeiro, M.E.N.P.; da Silva, C.R.; Júnior, H.V.N.; Ricardo, N.M.P.S. Clove essential oil encapsulated on nanocarrier based on polysaccharide: A strategy for the treatment of vaginal candidiasis. Coll. Surf. A Physicochem. Eng. Asp. 2021, 610, 125732. [Google Scholar] [CrossRef]
- Vasile, B.S.; Birca, A.C.; Musat, M.C.; Holban, A.M. Wound dressings coated with silver nanoparticles and essential oils for the management of wound infections. Materials 2020, 13, 1682. [Google Scholar] [CrossRef]
- Shakeel, F.; Alam, P.; Anwer, M.K.; Alanazi, S.A.; Alsarra, I.A.; Alqarni, M.H. Wound healing evaluation of self-nanoemulsifying drug delivery system containing Piper cubeba essential oil. 3 Biotech 2019, 9, 1–9. [Google Scholar] [CrossRef]
- Sharma, S.; Barkauskaite, S.; Duffy, B.; Jaiswal, A.K.; Jaiswal, S. Characterization and antimicrobial activity of biodegradable active packaging enriched with clove and thyme essential oil for food packaging application. Foods 2020, 9, 1117. [Google Scholar] [CrossRef] [PubMed]
- Hadidi, M.; Pouramin, S.; Adinepour, F.; Haghani, S.; Jafari, S.M. Chitosan nanoparticles loaded with clove essential oil: Characterization, antioxidant and antibacterial activities. Carbohydr. Polym. 2020, 236, 116075. [Google Scholar] [CrossRef] [PubMed]
- Bilal, M.; Zhao, Y.; Iqbal, H.M. Development and characterization of essential oils incorporated chitosan-based cues with antibacterial and antifungal potentialities. J. Radiat. Res. Appl. Sci. 2020, 13, 174–179. [Google Scholar] [CrossRef]
- Bagheri, R.; Ariaii, P.; Motamedzadegan, A. Characterization, antioxidant and antibacterial activities of chitosan nanoparticles loaded with nettle essential oil. J. Food Meas. Charact. 2020, 15, 1395–1402. [Google Scholar] [CrossRef]


| S. No. | Constituents | Retention Time (min) | Percentage Composition | S. No. | Constituents | Retention Time (min) | Percentage Composition | S. No. | Constituents | Retention Time (min) | Percentage Composition |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 2-Heptanol acetate | 9.28 | 0.016 | 12 | β-Gurjunene | 19.2 | 0.32 | 23 | α-Farnese | 21.32 | 0.057 |
| 2 | 2-Nonanone | 10.23 | 0.012 | 13 | α-Ylangene | 19.42 | 0.015 | 24 | Eugenyl acetate | 21.47 | 8.32 |
| 3 | Acetic acid, phenylmethyl ester | 11.2 | 0.06 | 25 | Cadina-1,4-diene | 21.53 | 0.16 | ||||
| 4 | Methyl salicylate | 14.19 | 0.055 | 14 | α-Humulene | 19.81 | 1.37 | 26 | α-Calacorene | 21.77 | 0.037 |
| 5 | Chavicol | 14.52 | 0.175 | 15 | Alloaromadendrene | 20.13 | 0.034 | 27 | 1-Vinyl-2,6,6-Trimethylcyclohex-1- | ||
| 6 | α-Cubebene | 15.35 | 0.034 | 16 | δ-Cadinene | 20.43 | 0.26 | Ene | 21.83 | 0.57 | |
| 7 | Eugenol | 16.48 | 71.43 | 17 | (E)-5-Acetyl-2,2-dimethyl-1-(3’-methyl-1’,3’-yl)bicyclo[2.1.0]pentan | 20.67 | 0.059 | 28 | Caryophyllenyl alcohol | 22.04 | 0.12 |
| 8 | α-Copaene | 17.58 | 0.53 | 18 | γ-Muurolene | 20.89 | 0.073 | 29 | (−)-Caryophyllene oxide | 22.15 | 0.43 |
| 9 | Cis-isoeugenol | 18.13 | 0.018 | 19 | α-Amorphene | 20.94 | 0.037 | 30 | Caryophylla-4(12),8(13)-dien-5.beta.ol | 22.28 | 0.027 |
| 10 | β-Elemene | 18.32 | 0.043 | 20 | α-Muurolene | 21.06 | 0.034 | 31 | Vulgarol B 43 2,3,4- | 22.42 | 0.85 |
| 21 | β-Selinene | 21.18 | 0.056 | 32 | Trimethoxyacetophenone | 23.13 | 0.46 | ||||
| 11 | Caryophyllene | 18.86 | 10.32 | 22 | β –Cadinene | 21.27 | 0.063 | 33 | Benzyl benzoate | 24.75 | 0.49 |
Sample | Polymer Ratio CS:PEO | Concentration of Oil% | Viscosity | Conductivity | Fiber Morphology |
|---|---|---|---|---|---|
| 01 02 03 04 05 06 | 50:50 50:50 60:40 60:40 70:30 70:30 | 0.5 1 0.5 1 0.5 1 | 1852 1871 1878 1883 1894 1912 | 2247 2228 2221 2215 2217 2206 | Smooth beadles Smooth beadles Smooth with some beads Beaded Beaded Beaded |
| Formulation Code | CEO NFs |
|---|---|
| CEO Content (%) | 87.6 ± 13.1 |
| CEO loading (%) | 8.9 ± 0.98 |
| Total yield (%) | 79 ± 9.35 |
| Days | Group A (Untreated) | Group B (Treated with Blank) | Group C (Treated with CEONFs) | Group D (Treated with Commercial Product) |
|---|---|---|---|---|
| 01 | 0 ± 0 ns | 0 ± 0 ns | 0 ± 0 ns | 0 ± 0 ns |
| 05 | 17.56 ± 3.4 | 29.37 ± 2.18 ns | 45.41 ± 3.5 ** | 49.1 ± 3.4 ** |
| 10 | 50.44 ± 4.12 | 69. 3 ± 2.5 * | 90.13 ± 1.5 ** | 95.3 ± 3.2 *** |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hameed, M.; Rasul, A.; Waqas, M.K.; Saadullah, M.; Aslam, N.; Abbas, G.; Latif, S.; Afzal, H.; Inam, S.; Akhtar Shah, P. Formulation and Evaluation of a Clove Oil-Encapsulated Nanofiber Formulation for Effective Wound-Healing. Molecules 2021, 26, 2491. https://doi.org/10.3390/molecules26092491
Hameed M, Rasul A, Waqas MK, Saadullah M, Aslam N, Abbas G, Latif S, Afzal H, Inam S, Akhtar Shah P. Formulation and Evaluation of a Clove Oil-Encapsulated Nanofiber Formulation for Effective Wound-Healing. Molecules. 2021; 26(9):2491. https://doi.org/10.3390/molecules26092491
Chicago/Turabian StyleHameed, Misbah, Akhtar Rasul, Muhammad Khurram Waqas, Malik Saadullah, Nosheen Aslam, Ghulam Abbas, Sumera Latif, Hafsa Afzal, Sana Inam, and Pervaiz Akhtar Shah. 2021. "Formulation and Evaluation of a Clove Oil-Encapsulated Nanofiber Formulation for Effective Wound-Healing" Molecules 26, no. 9: 2491. https://doi.org/10.3390/molecules26092491
APA StyleHameed, M., Rasul, A., Waqas, M. K., Saadullah, M., Aslam, N., Abbas, G., Latif, S., Afzal, H., Inam, S., & Akhtar Shah, P. (2021). Formulation and Evaluation of a Clove Oil-Encapsulated Nanofiber Formulation for Effective Wound-Healing. Molecules, 26(9), 2491. https://doi.org/10.3390/molecules26092491






