LC-HRMS Profiling and Antidiabetic, Anticholinergic, and Antioxidant Activities of Aerial Parts of Kınkor (Ferulago stellata)
Abstract
1. Introduction
2. Results and Discussion
3. Materials and Methods
3.1. Chemicals and Plant Materials
3.2. Preparation of the Water and Ethanol Extracts
3.3. Reducing Ability Assays
3.4. Radical Scavenging Activities
3.5. Anticholinergic Assay
3.6. Antidiabetic Assay
3.7. Determination of Inhibition Parameters
3.8. Total Phenolic and Flavonoid Contents
3.9. Preparation of Samples for LC-HRMS Analysis
3.10. Instruments and Chromatographic Conditions of LC-HRMS
3.11. Optimization of LC-HRMS Procedure
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Rummun, N.; Rondeau, P.; Bourdon, E.; Pires, E.; McCullagh, J.; Claridge, T.D.W.; Bahorun, T.; Li, W.W.; Neergheen, V.S. Terminalia bentzoe, a mascarene endemic plant, inhibits human hepatocellular carcinoma cells growth in vitro via G0/G1 phase cell cycle arrest. Pharmaceuticals 2020, 13, 303. [Google Scholar] [CrossRef] [PubMed]
- Choi, E.M.; Suh, K.S.; Park, S.Y.; Yun, S.; Chin, S.O.; Rhee, S.Y.; Chon, S. Orientin reduces the inhibitory effects of 2,3,7,8-tetrachlorodibenzo-p-dioxin on adipogenic differentiation and insulin signaling pathway inmurine 3T3-L1 adipocytes. Chem. Biol. Interact. 2020, 318, 108978. [Google Scholar] [CrossRef] [PubMed]
- Zubay, P.; Kunzelmann, J.; Ittzes, A.; Zamborine, E.N.; Szabo, K. Allelopathic effects of leachates of Juglans regia L., Populus tremula L. and juglone on germination of temperate zone cultivated medicinal and aromatic plants. Agroforest. Syst. 2021, 95, 431–442. [Google Scholar] [CrossRef]
- Chukwuma, C.I.; Matsabisa, M.G.; Ibrahim, M.A.; Erukainure, O.L.; Chabalala, M.H.; Islam, M.S. Medicinal plants with concomitant anti-diabetic and anti-hypertensive effects as potential sources of dual acting therapies against diabetes and hypertension: A review. J. Ethnopharmacol. 2019, 10, 329–360. [Google Scholar] [CrossRef] [PubMed]
- Topal, M.; Gocer, H.; Topal, F.; Kalin, P.; Polat Köse, P.; Gülçin, İ.; Çakmak, K.C.; Küçük, M.; Durmaz, L.; Gören, A.C.; et al. Antioxidant, antiradical and anticholinergic properties of cynarin purified from the illyrian thistle (Onopordum illyricum L.). J. Enzyme Inhib. Med. Chem. 2016, 31, 266–275. [Google Scholar] [CrossRef]
- Gulcin, I.; Topal, F.; Oztürk Sarikaya, S.B.; Bursal, E.; Gören, A.C.; Bilsel, M. Polyphenol contents and antioxidant properties of medlar (Mespilus germanica L.). Rec. Nat. Prod. 2011, 5, 158–175. [Google Scholar]
- Elmastas, M.; Türkekul, İ.; Öztürk, L.; Gulcin, I.; Işıldak, Ö.; Aboul-Enein, H.Y. The antioxidant activity of two wild edible mushrooms (Morchella vulgaris and Morchella esculanta). Comb. Chem. High. Throughput Screen. 2006, 9, 443–448. [Google Scholar] [CrossRef] [PubMed]
- Gulcin, I.; Tel, A.Z.; Kirecci, E. Antioxidant, antimicrobial, antifungal and antiradical activities of Cyclotrichium niveum (Boiss.) Manden and Scheng. Int. J. Food Prop. 2008, 11, 450–471. [Google Scholar] [CrossRef]
- Ak, T.; Gulcin, I. Antioxidant and radical scavenging properties of curcumin. Chem. Biol. Interact. 2008, 174, 27–37. [Google Scholar] [CrossRef] [PubMed]
- Talaz, O.; Gulcin, I.; Göksu, S.; Saracoglu, N. Antioxidant activity of 5,10-dihydroindeno[1,2-b]indoles containing substituents on dihydroindeno part. Bioorg. Med. Chem. 2009, 17, 6583–6589. [Google Scholar] [CrossRef] [PubMed]
- Şerbetçi Tohma, H.; Gulcin, I. Antioxidant and radical scavenging activity of aerial parts and roots of Turkish liquorice (Glycyrrhiza glabra L.). Int. J. Food Prop. 2010, 13, 657–671. [Google Scholar] [CrossRef]
- Gulcin, I. Antioxidant activity of eugenol-a structure and activity relationship study. J. Med. Food 2011, 14, 975–985. [Google Scholar] [CrossRef]
- Elmastas, M.; Celik, S.M.; Genc, N.; Aksit, H.; Erenler, R.; Gulcin, I. Antioxidant activity of an Anatolian herbal tea-Origanum minutiflorum: Isolation and characterization of its secondary metabolites. Int. J. Food Prop. 2018, 21, 374–384. [Google Scholar] [CrossRef]
- Gulcin, I.; Oktay, M.; Kireçci, E.; Küfrevioğlu, Ö.İ. Screening of antioxidant and antimicrobial activities of anise (Pimpinella anisum L.) seed extracts. Food Chem. 2003, 83, 371–382. [Google Scholar] [CrossRef]
- Köksal, E.; Bursal, E.; Dikici, E.; Tozoğlu, F.; Gulcin, I. Antioxidant activity of Melissa officinalis leaves. J. Med. Plants Res. 2011, 5, 217–222. [Google Scholar]
- Gulcin, I.; Büyükokuroğlu, M.E.; Oktay, M.; Küfrevioğlu, Ö.İ. Antioxidant and analgesic activities of turpentine of Pinus nigra Arn. Subsp. pallsiana (Lamb.) Holmboe. J. Ethnopharmacol. 2003, 86, 51–58. [Google Scholar] [CrossRef]
- Dalar, A.; Mukemre, M.; Unal, M.; Ozgokce, F. Traditional medicinal plants of Agri province, Turkey. J. Ethnopharmacol. 2018, 226, 56–72. [Google Scholar] [CrossRef] [PubMed]
- Oktay, M.; Gulcin, I.; Küfrevioğlu, Ö.İ. Determination of in vitro antioxidant activity of fennel (Foeniculum vulgare) seed extracts. Lebensm. Wissen. Technol. 2003, 36, 263–271. [Google Scholar] [CrossRef]
- Santos, P.H.; Kammers, J.C.; Silva, A.P.; Oliveira, J.V.; Hense, H. Antioxidant and antibacterial compounds from feijoa leaf extracts obtained by pressurized liquid extraction and supercritical fluid extraction. Food Chem. 2021, 344, 128620. [Google Scholar] [CrossRef]
- Villaverde, J.J.; Sandín-españa, P.; Sevilla-morán, B.; López-goti, C.; Alonso-prados, J.L. Biopesticides from natural products: Current development, legislative framework, and future trends. BioResource 2016, 11, 5618–5640. [Google Scholar] [CrossRef]
- Gulcin, I.; Küfrevioğlu, Ö.İ.; Oktay, M. Purification and characterization of polyphenol oxidase from nettle (Urtica dioica L.) and inhibition effects of some chemicals on the enzyme activity. J. Enzyme Inhib. Med. Chem. 2005, 20, 297–302. [Google Scholar] [CrossRef]
- Gulcin, I.; Mshvildadze, V.; Gepdiremen, A.; Elias, R. Antioxidant activity of a triterpenoid glycoside isolated from the berries of Hedera colchica: 3-O-(β-D-glucopyranosyl)-hederagenin. Phytother. Res. 2006, 20, 130–134. [Google Scholar] [CrossRef]
- Gulcin, I.; Küfrevioğlu, Ö.İ.; Oktay, M.; Büyükokuroğlu, M.E. Antioxidant, antimicrobial, antiulcer and analgesic activities of nettle (Urtica dioica L.). J. Ethnopharmacol. 2004, 90, 205–215. [Google Scholar] [CrossRef]
- Gulcin, I.; Alici, H.A.; Cesur, M. Determination of in vitro antioxidant and radical scavenging activities of propofol. Chem. Pharm. Bull. 2005, 53, 281–285. [Google Scholar] [CrossRef]
- Zengin, G.; Aumeeruddy-Elalfi, Z.; Mollica, A.; Yilmaz, M.A.; Mahomoodally, M.F. In vitro and in silico perspectives on biological and phytochemical profile of three halophyte species-A source of innovative phytopharmaceuticals from nature. Phytomedicine 2018, 38, 35–44. [Google Scholar] [CrossRef]
- Gulcin, I.; Şat, İ.G.; Beydemir, Ş.; Elmastaş, M.; Küfrevioğlu, Ö.İ. Comparison of antioxidant activity of clove (Eugenia caryophylata Thunb) buds and lavender (Lavandula stoechas L.). Food Chem. 2004, 87, 393–400. [Google Scholar] [CrossRef]
- Gulcin, I.; Mshvildadze, V.; Gepdiremen, A.; Elias, R. Antioxidant activity of saponins isolated from ivy: α-Hederin, hederasaponin-C, hederacolchiside-E and hederacolchiside F. Planta Med. 2004, 70, 561–563. [Google Scholar] [CrossRef]
- Gulcin, I.; Beydemir, Ş.; Şat, İ.G.; Küfrevioğlu, Ö.İ. Evaluation of antioxidant activity of cornelian cherry (Cornus mas L.). Acta Aliment. Hung. 2005, 34, 193–202. [Google Scholar] [CrossRef]
- Gocer, H.; Topal, F.; Topal, M.; Küçük, M.; Teke, D.; Gulcin, I.; Alwasel, S.H.; Supuran, C.T. Acetylcholinesterase and carbonic anhydrase isoenzymes I and II inhibition profiles of taxifolin. J. Enzyme Inhib. Med. Chem. 2016, 31, 441–447. [Google Scholar] [CrossRef]
- Akter, K.; Lanza, E.A.; Martin, S.A.; Myronyuk, N.; Rua, M.; Raffa, R.B. Diabetes mellitus and Alzheimer’s disease: Shared pathology and treatment? Br. J. Clin. Pharmacol. 2010, 71, 365–376. [Google Scholar] [CrossRef]
- Taslimi, P.; Gulcin, I. Antioxidant and anticholinergic properties of olivetol. J. Food Biochem. 2018, 42, e12516. [Google Scholar] [CrossRef]
- Yiğit, B.; Kaya, R.; Taslimi, P.; Işık, Y.; Karaman, M.; Yiğit, M.; Özdemir, İ.; Gulcin, I. Imidazolinium chloride salts bearing wing tip groups: Synthesis, molecular docking and metabolic enzymes inhibition. J. Mol. Struct. 2019, 1179, 709–718. [Google Scholar] [CrossRef]
- Bal, S.; Demirci, O.; Aktaş, A.; Şen, B.; Taslimi, P.; Aktaş, A.; Gök, Y.; Aygün, M.; Gulcin, I. PEPPSI type Pd(II)NHC complexes bearing Chloro-/Fluorobenzyl group: Synthesis, characterization, crystal structures, α-glycosidase and acetylcholinesterase ınhibitory properties. Polyhedron 2021, 198, 115060. [Google Scholar] [CrossRef]
- Bursal, E.; Taslimi, P.; Gören, A.; Gulcin, I. Assessments of anticholinergic, antidiabetic, antioxidant activities and phenolic content of Stachys annua. Biocat. Agric. Biotechnol. 2020, 28, 101711. [Google Scholar] [CrossRef]
- Polat Köse, L.; Bingöl, Z.; Kaya, R.; Gören, A.C.; Akincioğlu, H.; Durmaz, L.; Koksal, E.; Alwasel, S.; Gulcin, I. Anticholinergic and antioxidant activities of avocado (Folium perseae) leaves - Phytochemical content by LC-MS/MS Analysis. Int. J. Food Prop. 2020, 23, 878–893. [Google Scholar] [CrossRef]
- Gulcin, I.; Tel, A.Z.; Gören, A.C.; Taslimi, P.; Alwasel, S. Sage (Salvia pilifera): Determination its polyphenol contents, anticholinergic, antidiabetic and antioxidant activities. J. Food Measure. Charac. 2019, 13, 2062–2074. [Google Scholar] [CrossRef]
- Fatah, N.H.A.; Amen, Y.; Abdel Bar, F.M.; Halim, A.F.; Saad, H.E.A. Antioxidants and α-glucosidase Inhibitors from Lactuca serriola L. Rec. Nat. Prod. 2020, 14, 410–415. [Google Scholar] [CrossRef]
- Gulcin, I.; Kaya, R.; Gören, A.C.; Akıncıoğlu, H.; Topal, M.; Bingöl, Z.; Çetin Çakmak, K.; Ozturk Sarikaya, S.B.; Durmaz, L.; Alwasel, S. Anticholinergic, antidiabetic and antioxidant activities of Cinnamon (Cinnamomum verum) bark extracts: Polyphenol contents analysis by LC-MS/MS. Int. J. Food Prop. 2019, 22, 1511–1526. [Google Scholar] [CrossRef]
- Artunç, T.; Menzek, A.; Taslimi, P.; Gulcin, I.; Kazaz, C.; Şahin, E. Synthesis and antioxidant activities of phenol derivatives from 1,6-bis(dimethoxyphenyl)hexane-1,6-dione. Bioorg. Chem. 2020, 100, 103884. [Google Scholar] [CrossRef]
- Uysal, A.; Zengin, G.; Mahomoodally, M.F.; Picot-Allain, C.; Jeko, J.; Cziaky, Z.; Rodrigues, M.J.; Ak, G.; Polat, R.; Urusan, Z.; et al. A comparative study on biological properties and chemical profiles of different solvent extracts from Centaurea bingoelensis, an endemic plant of Turkey. Process. Biochem. 2021, 102, 315–324. [Google Scholar] [CrossRef]
- Gürağaç Dereli, F.T.; Ilhan, M.; Küpeli Akkol, E. Identification of the main active antidepressant constituents in a traditional Turkish medicinal plant, Centaurea kurdica Reichardt. J. Ethnopharmacol. 2020, 249, 112373. [Google Scholar] [CrossRef] [PubMed]
- Miller, D.D. Minerals. In Food Chemistry; Fennem, O.R., Ed.; Marcel Dekker: New York, NY, USA, 1996; pp. 61–649. [Google Scholar]
- Cakmakçı, S.; Topdaş, E.F.; Kalın, P.; Han, H.; Şekerci, P.; Polat Kose, L.; Gulcin, I. Antioxidant capacity and functionality of oleaster (Elaeagnus angustifolia L.) flour and crust in a new kind of fruity ice cream. Int. J. Food Sci. Technol. 2015, 50, 472–481. [Google Scholar] [CrossRef]
- Köksal, E.; Gulcin, I.; Öztürk Sarıkaya, S.B.; Bursal, E. On the in vitro antioxidant activity of silymarin. J. Enzyme Inhib. Med. Chem. 2009, 24, 395–405. [Google Scholar] [CrossRef] [PubMed]
- Topal, F.; Topal, M.; Gocer, H.; Kalın, P.; Koçyiğit, U.M.; Gulcin, I.; Alwasel, S.H. Antioxidant activity of taxifolin: An activity-structure relationship. J. Enzyme Inhib. Med. Chem. 2016, 31, 674–683. [Google Scholar] [CrossRef]
- Gulcin, I.; Elmastaş, M.; Aboul-Enein, H.Y. Antioxidant activity of clove oil-A powerful antioxidant source. Arab. J. Chem. 2012, 5, 489–499. [Google Scholar] [CrossRef]
- Gulcin, I. Antioxidants and antioxidant methods-An updated overview. Arch. Toxicol. 2020, 94, 651–715. [Google Scholar] [CrossRef]
- Baran, A.; Karakılıc, E.; Faiz, Ö.; Ozen, F. Synthesis of chalcone-containing zinc and cobalt metallophthalocyanines; investigation of their photochemical, DPPH radical scavenging and metal chelating characters. Org. Commun. 2020, 13, 65–78. [Google Scholar] [CrossRef]
- Gulcin, I. Antioxidant activity of food constituents: An overview. Arch. Toxicol. 2012, 86, 345–391. [Google Scholar] [CrossRef]
- Gulcin, I. Antioxidant activity of L-Adrenaline: An activity-structure insight. Chem. Biol. Interact. 2009, 179, 71–80. [Google Scholar] [CrossRef]
- Gulcin, I.; Huyut, Z.; Elmastaş, M.; Aboul-Enein, H.Y. Radical scavenging and antioxidant activity of tannic acid. Arab. J. Chem. 2010, 3, 43–53. [Google Scholar] [CrossRef]
- Tohma, H.; Köksal, E.; Kılıç, Ö.; Alan, Y.; Yılmaz, M.A.; Gulcin, I.; Bursal, E.; Alwasel, S.H. RP-HPLC/MS/MS analysis of the phenolic compounds, antioxidant and antimicrobial activities of Salvia L. species. Antioxidants 2016, 5, 38. [Google Scholar] [CrossRef]
- Gulcin, I.; Beydemir, Ş.; Alici, H.A.; Elmastas, M.; Büyükokuroğlu, M.E. In vitro antioxidant properties of morphine. Pharmacol. Res. 2004, 49, 59–66. [Google Scholar] [CrossRef]
- Gulcin, I. Antioxidant properties of resveratrol: A structure-activity insight. Innov. Food Sci. Emerg. 2010, 11, 210–218. [Google Scholar] [CrossRef]
- Gulcin, I. Comparison of in vitro antioxidant and antiradical activities of L-tyrosine and L-Dopa. Amino Acids 2007, 32, 431–438. [Google Scholar] [CrossRef]
- Ibrahim, S.R.M.; Mohamed, A.M.; Zayed, G.F.; Ross, S.A. 8-Hydroxyirilone 5-methyl ether and 8-hydroxyirilone, new antioxidant and α-amylase inhibitors isoflavonoids from Iris germanica rhizomes. Bioorg. Chem. 2017, 70, 192–198. [Google Scholar] [CrossRef] [PubMed]
- Zengin, G.; Uysal, A.; Diuzheva, A.; Gunes, E.; Jeko, J.; Cziaky, Z.; Picot-Allain, C.M.N.; Mahomoodally, M.F. Characterization of phytochemical components of Ferula halophile extracts using HPLC-MS/MS and their pharmacological potentials: Amulti-functional insight. J. Pharm. Biomed. Anal. 2018, 160, 374–382. [Google Scholar] [CrossRef]
- Wang, S.S.; Wang, D.M.; Liu, Z.H. Synergistic, additive and antagonistic effects of Potentilla fruticose combined with EGb761 on antioxidant capacities and the possible mechanism. Ind. Crops Prod. 2015, 67, 227–238. [Google Scholar] [CrossRef]
- Cipriani, A.; de Sousa, A.L.; Tenfen, A.; Siebert, D.A.; de Gasper, A.L.; Vitali, L.; Micke, G.A.; Alberton, M.D. Phenolic compounds of Eugenia involucrata (Myrtaceae) extracts and associated antioxidant and inhibitory effects on acetylcholinesterase and α-glucosidase. Nat. Prod. Res. 2020, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Dasgin, S.; Gok, Y.; Barut Celepci, D.; Taslimi, P.; İzmirli, M.; Aktaş, A.; Gulcin, I. Synthesis, characterization, crystal structure and bioactivity properties of the benzimidazole-functionalized PEPPSI type of Pd(II)NHC complexes. J. Mol. Struct. 2021, 1228, 129442. [Google Scholar] [CrossRef]
- Aktas, A.; Barut Celepci, D.; Gök, Y.; Taslimi, P.; Akıncıoğlu, H.; Gulcin, I. A novel Ag-N-heterocyclic carbene complex bearing the hydroxyethyl ligand: Synthesis, characterization, crystal and spectral structures and bioactivity properties. Crystals 2020, 10, 171. [Google Scholar] [CrossRef]
- Hashmi, S.; Khan, S.; Shafiq, Z.; Taslimi, P.; Ishaq, M.; Sadeghian, N.; Karaman, S.H.; Akhtar, N.; Islam, M.; Asari, A.; et al. Probing 4-(diethylamino)-salicylaldehyde-based thiosemicarbazones as multi-target directed ligands against cholinesterases, carbonic anhydrases and α-glycosidase enzymes. Bioorg. Chem. 2021, 107, 104554. [Google Scholar] [CrossRef]
- Kazancı, A.; Gok, Y.; Kaya, R.; Aktaş, A.; Taslimi, P.; Gulcin, I. Synthesis, characterization and bioactivities of dative donor ligand N-heterocyclic carbene (NHC) precursors and their Ag(I)NHC coordination compounds. Polyhedron 2021, 193, 114866. [Google Scholar] [CrossRef]
- Gulcin, I.; Şat, I.G.; Beydemir, Ş.; Kufrevioglu, Ö.İ. Evaluation of the in vitro antioxidant properties of extracts of broccoli (Brassica oleracea L.). Ital. J. Food Sci. 2004, 16, 17–30. [Google Scholar]
- Göçer, H.; Gulcin, I. Caffeic acid phenethyl ester (CAPE): Correlation of structure and antioxidant properties. Int. J. Food Sci. Nutr. 2011, 62, 821–825. [Google Scholar] [CrossRef] [PubMed]
- Gulcin, I.; Topal, F.; Çakmakçı, R.; Gören, A.C.; Bilsel, M.; Erdoğan, U. Pomological features, nutritional quality, polyphenol content analysis and antioxidant properties of domesticated and three wild ecotype forms of raspberries (Rubus idaeus L.). J. Food Sci. 2011, 76, C585–C593. [Google Scholar] [CrossRef] [PubMed]
- Taslimi, P.; Koksal, E.; Gören, A.C.; Bursal, E.; Aras, A.; Kılıç, O.; Alwasel, S.; Gulcin, I. Anti-Alzheimer, antidiabetic and antioxidant potential of Satureja cuneifolia and analysis of ıts phenolic contents by LC-MS/MS. Arab. J. Chem. 2020, 13, 4528–4537. [Google Scholar] [CrossRef]
- Jiang, Y.; Fang, Z.; Leonard, W.; Zhang, P. Phenolic compounds in Lycium berry: Composition, health benefits and industrial applications. J. Func. Foods 2021, 77, 194340. [Google Scholar] [CrossRef]
- Buldurun, K.; Turan, N.; Bursal, E.; Mantarcı, A.; Turkan, F.; Taslimi, P.; Gulcin, I. Synthesis, spectroscopic properties, crystal structures, antioxidant activities and enzyme inhibition determination of Co(II) and Fe(II) complexes of Schiff base. Res. Chem. Intermed. 2020, 46, 283–297. [Google Scholar] [CrossRef]
- Gulcin, I.; Bursal, E.; Şehitoğlu, H.M.; Bilsel, M.; Gören, A.C. Polyphenol contents and antioxidant activity of lyophilized aqueous extract of propolis from Erzurum, Turkey. Food Chem. Toxicol. 2010, 48, 2227–2238. [Google Scholar] [CrossRef]
- Gulcin, I. The antioxidant and radical scavenging activities of black pepper (Piper nigrum) seeds. Int. J. Food Sci. Nutr. 2005, 56, 491–499. [Google Scholar] [CrossRef]
- Gulcin, I.; Elmastas, M.; Aboul-Enein, H.Y. Determination of antioxidant and radical scavenging activity of basil (Ocimum basilicum) assayed by different methodologies. Phytother. Res. 2007, 21, 354–361. [Google Scholar] [CrossRef]
- Elmastas, M.; Gulcin, I.; Işıldak, Ö.; Küfrevioğlu, Ö.İ.; İbaoğlu, K.; Aboul-Enein, H.Y. Antioxidant capacity of bay (Laurus nobilis L.) leaves extracts. J. Iran. Chem. Soc. 2006, 3, 258–266. [Google Scholar] [CrossRef]
- Oyaizu, M. Studies on product of browning reaction prepared from glucose amine. Jpn. J. Nutr. 1986, 44, 307–315. [Google Scholar] [CrossRef]
- Gulcin, I. Antioxidant activity of caffeic acid (3,4-dihydroxycinnamic acid). Toxicology 2006, 217, 213–220. [Google Scholar] [CrossRef] [PubMed]
- Gulcin, I. Antioxidant and antiradical activities of L-Carnitine. Life Sci. 2006, 78, 803–811. [Google Scholar] [CrossRef]
- Çetin Çakmak, K.; Gulcin, I. Anticholinergic and antioxidant activities of usnic acid-An activity-structure insight. Toxicol. Rep. 2019, 6, 1273–1280. [Google Scholar] [CrossRef] [PubMed]
- Bursal, E.; Gulcin, I. Polyphenol contents and in vitro antioxidant activities of lyophilized aqueous extract of kiwifruit (Actinidia deliciosa). Food Res. Int. 2011, 44, 1482–1489. [Google Scholar] [CrossRef]
- Blois, M.S. Antioxidant determinations by the use of a stable free radical. Nature 1958, 26, 1199–1200. [Google Scholar] [CrossRef]
- Gulcin, I.; Berashvili, D.; Gepdiremen, A. Antiradical and antioxidant activity of total anthocyanins from Perilla pankinensis decne. J. Ethnopharmacol. 2005, 101, 287–293. [Google Scholar] [CrossRef]
- Romanucci, V.; Giordano, M.; Davinelli, S.; Di Marino, C.; Ladhari, A.; De Marco, A. Antimicrobial Effect and Antioxidant Activity of Triterpenes Isolated from Gymnema sylvestre R. Br. Rec. Nat. Prod. 2020, 14, 210–213. [Google Scholar] [CrossRef]
- Gulcin, I.; Mshvildadze, V.; Gepdiremen, A.; Elias, R. Screening of antioxidant and antiradical activity of monodesmosides and crude extract from Leontice smirnowii Tuber. Phytomedicine 2006, 13, 343–351. [Google Scholar] [CrossRef] [PubMed]
- Gulcin, I.; Elias, R.; Gepdiremen, A.; Boyer, L. Antioxidant activity of lignans from fringe tree (Chionanthus virginicus L.). Eur. Food Res. Technol. 2006, 223, 759–767. [Google Scholar] [CrossRef]
- Gulcin, I.; Elias, R.; Gepdiremen, A.; Boyer, L.; Köksal, E. A comparative study on the antioxidant activity of fringe tree (Chionanthus virginicus L.) extracts. Afr. J. Biotechnol. 2007, 6, 410–418. [Google Scholar]
- Gulcin, I.; Elias, R.; Gepdiremen, A.; Taoubi, K.; Köksal, E. Antioxidant secoiridoids from fringe tree (Chionanthus virginicus L.). Wood Sci. Technol. 2009, 43, 195–212. [Google Scholar] [CrossRef]
- Bursal, E.; Aras, A.; Kılıç, Ö.; Taslimi, P.; Gören, A.C.; Gulcin, I. Phytochemical content, antioxidant activity and enzyme inhibition effect of Salvia eriophora Boiss. & Kotschy against acetylcholinesterase, α-amylase, butyrylcholinesterase and α-glycosidase enzymes. J. Food Biochem. 2019, 43, e12776. [Google Scholar]
- Ellman, G.L.; Courtney, K.D.; Andres, V., Jr.; Featherstone, R.M. A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem. Pharmacol. 1961, 7, 88–95. [Google Scholar] [CrossRef]
- Akıncıoğlu, A.; Akıncıoğlu, H.; Gulcin, I.; Durdağı, S.; Supuran, C.T.; Göksu, S. Discovery of potent carbonic anhydrase and acetylcholine esterase inhibitors: Novel sulfamoylcarbamates and sulfamides derived from acetophenones. Bioorg. Med. Chem. 2015, 23, 3592–3602. [Google Scholar] [CrossRef]
- Gulcin, I.; Dastan, A. Synthesis of dimeric phenol derivatives and determination of in vitro antioxidant and radical scavenging activities. J. Enzyme Inhib. Med. Chem. 2007, 22, 685–695. [Google Scholar] [CrossRef]
- Karimov, A.; Orujova, A.; Taslimi, P.; Sadeghian, N.; Mammadov, B.; Karaman, H.S.; Farzaliyev, V.; Sujayev, A.; Taş, R.; Alwasel, S.; et al. Novel functionally substituted esters based on sodium diethyldithiocarbamate derivatives: Synthesis, characterization, biological activity and molecular docking studies. Bioorg. Chem. 2020, 99, 103762. [Google Scholar] [CrossRef]
- Tao, Y.; Zhang, Y.; Cheng, Y.; Wang, Y. Rapid screening and identification of α-glucosidase inhibitors from mulberry leaves using enzyme-immobilized magnetic beads coupled with HPLC/MS and NMR. Biomed. Chromatogr. 2013, 27, 148–155. [Google Scholar] [CrossRef]
- Pedrood, K.; Sherefati, M.; Taslimi, P.; Mohammadi-Khanaposhtani, M.; Asgari, M.S.; Hosseini, S.; Rastegar, H.; Larijani, B.; Mahdavi, M.; Taslimi, P.; et al. Design, synthesis, characterization, enzymatic inhibition evaluations, and docking study of novel quinazolinone derivatives. Int. J. Biol. Macromol. 2021, 170, 1–12. [Google Scholar] [CrossRef]
- Xiao, Z.; Storms, R.; Tsang, A. A quantitative starch–iodine method for measuring alpha-amylase and glucoamylase activities. Anal. Biochem. 2006, 351, 146–148. [Google Scholar] [CrossRef]
- Lineweaver, H.; Burk, D. The determination of enzyme dissociation constants. J. Am. Chem. Soc. 1934, 56, 658–666. [Google Scholar] [CrossRef]
- Gulcin, I.; Beydemir, S. Phenolic compounds as antioxidants: Carbonic anhydrase isoenzymes inhibitors. Mini Rev. Med. Chem. 2013, 13, 408–430. [Google Scholar] [PubMed]
- Folin, O.; Ciocalteu, V. On tyrosine and tryptophane determinations in proteins. J. Biol. Chem. 1927, 73, 627–650. [Google Scholar] [CrossRef]
- Sehitoglu, M.H.; Han, H.; Kalin, P.; Gulcin, I.; Ozkan, A.; Aboul-Enein, H.Y. Pistachio (Pistacia vera L.) gum: A potent inhibitor of reactive oxygen species. J. Enzyme Inhib. Med. Chem. 2015, 30, 264–269. [Google Scholar] [CrossRef] [PubMed]
- Gulcin, I.; Kirecci, E.; Akkemik, E.; Topal, F.; Hisar, O. Antioxidant and antimicrobial activities of an aquatic plant: Duckweed (Lemna minor L.). Turk. J. Biol. 2010, 34, 175–188. [Google Scholar]
- Kalın, P.; Gulcin, I.; Gören, A.C. Antioxidant activity and polyphenol content of cranberries (Vaccinium macrocarpon). Rec. Nat. Prod. 2015, 9, 496–502. [Google Scholar]
- Hamad, H.O.; Alma, M.H.; Gulcin, I.; Yılmaz, M.A.; Karaoğul, E. Evaluation of phenolic contents and bioactivity of root and nutgall extracts from Iraqian Quercus infectoria Olivier. Rec. Nat. Prod. 2017, 11, 205–210. [Google Scholar]
- Han, H.; Yılmaz, H.; Gulcin, I. Antioxidant activity of flaxseed (Linum usitatissimum L.) and analysis of its polyphenol contents by LC-MS/MS. Rec. Nat. Prod. 2018, 12, 397–402. [Google Scholar] [CrossRef]
- Sarikahya, N.B.; Goren, A.C.; Kirmizigul, S. Simultaneous determination of several flavonoids and phenolic compounds in nineteen different Cephalaria species by HPLC-MS/MS. J. Pharm. Biomed. 2019, 173, 120–125. [Google Scholar] [CrossRef] [PubMed]
- Ozer, Z.; Carıkcı, S.; Yılmaz, H.; Kılıc, T.; Dirmenci, T.; Goren, A.C. Determination of secondary metabolites of Origanum vulgare subsp. hirtum and O. vulgare subsp. vulgare by LC-MS/MS. J. Chem. Metrol. 2020, 14, 25–34. [Google Scholar] [CrossRef]
- Topal, M. Secondary metabolites of ethanol extracts of Pinus sylvestris cones from Eastern Anatolia and their antioxidant, cholinesterase and α-glucosidase activities. Rec. Nat. Prod. 2019, 14, 129–138. [Google Scholar] [CrossRef]
- Halfon, B.; Çetin, O.; Kokdil, G.; Topçu, G. Chemical investigation and bioactivity screening of Salvia cassia extracts. Rec. Nat. Prod. 2018, 13, 156–166. [Google Scholar] [CrossRef]
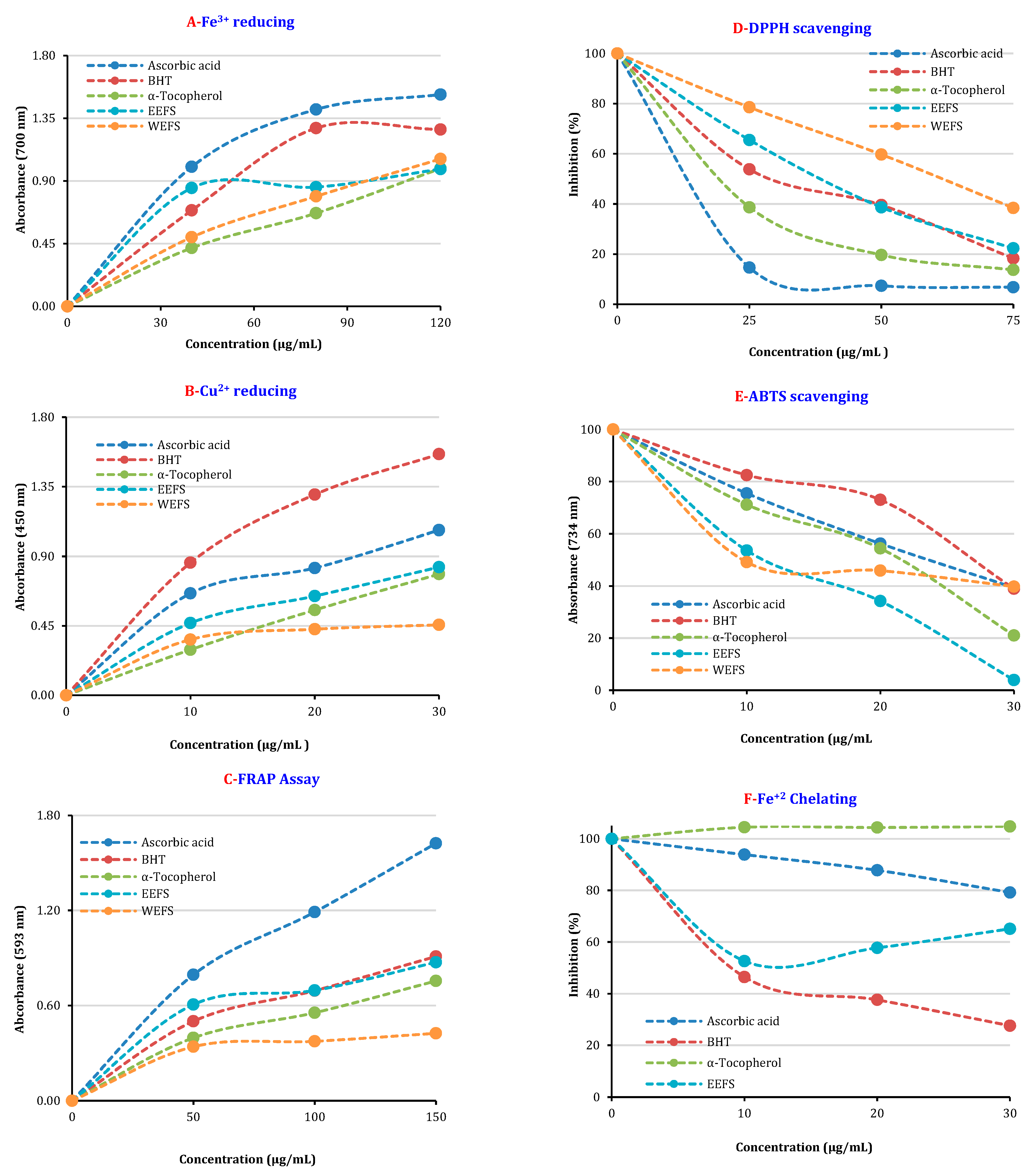
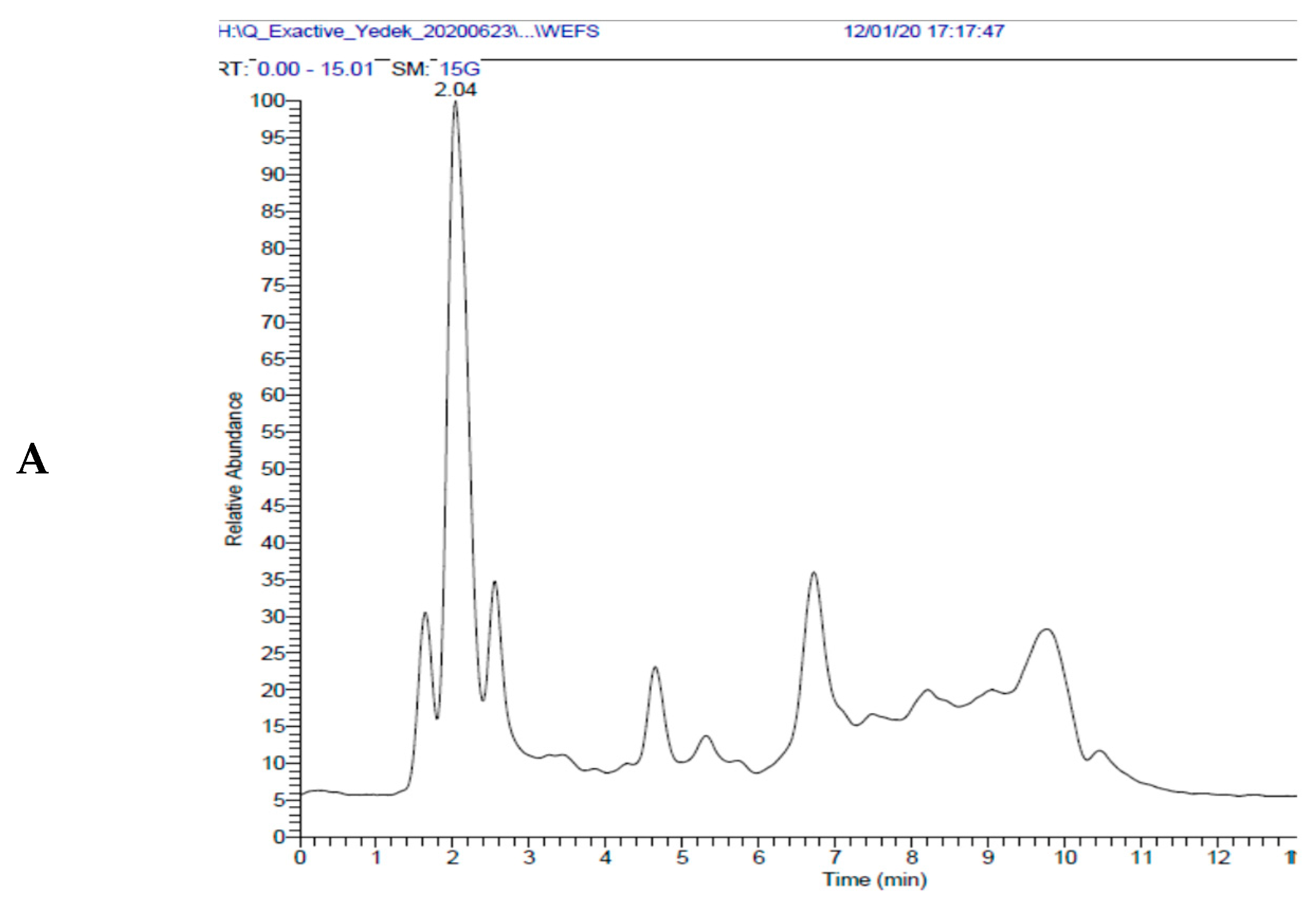
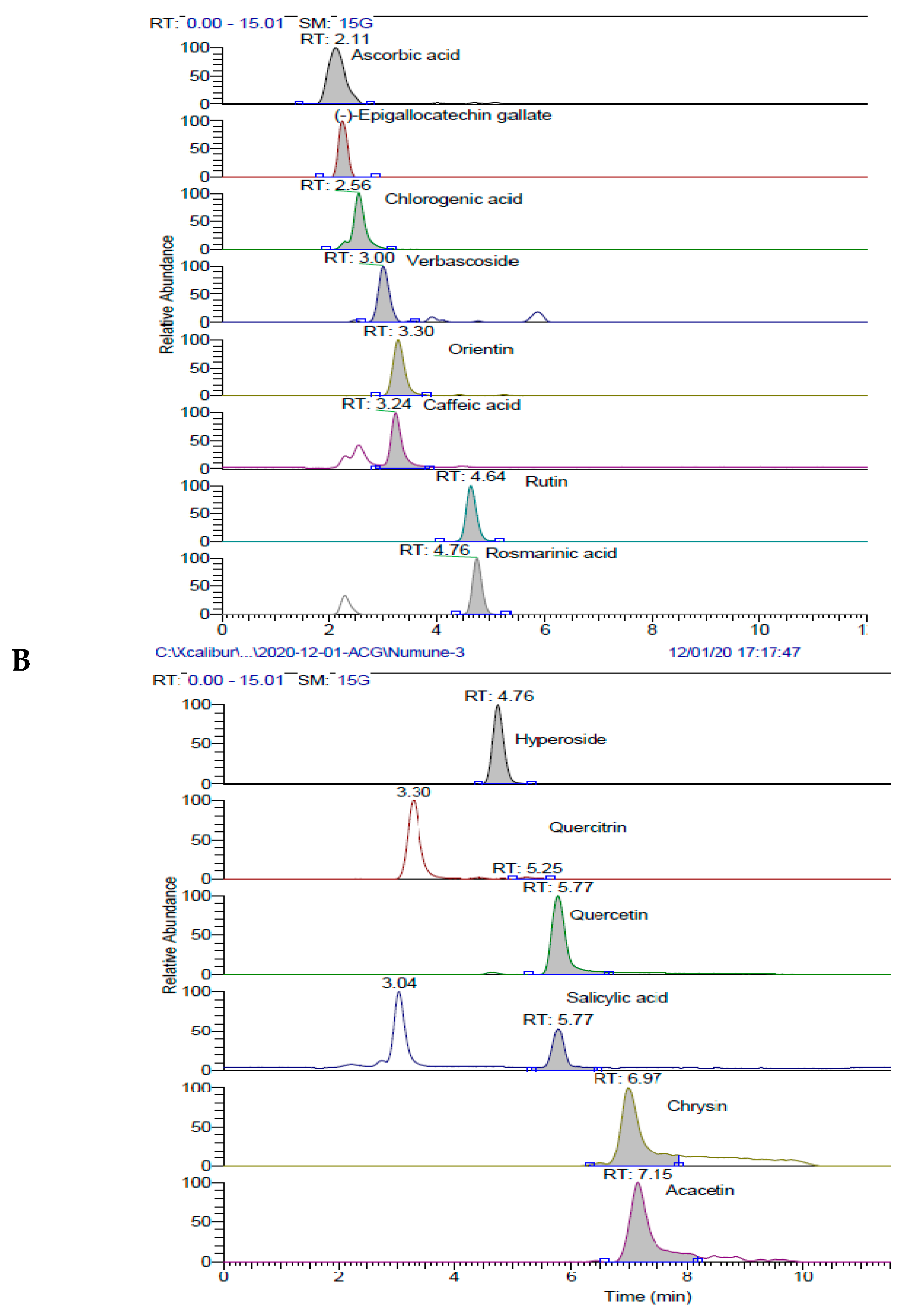
| Antioxidants | Fe3+ Reducing | Cu2+ Reducing | Fe3+-TPTZ Reducing | |||
|---|---|---|---|---|---|---|
| λ700 | r2 | λ450 | r2 | λ593 | r2 | |
| α-tocopherol | 0.990 ± 0.007 | 0.9942 | 0.785 ± 0.061 | 0.9986 | 0.755 ± 0.075 | 0.9867 |
| Ascorbic acid | 1.520 ± 0.028 | 0.9970 | 1.069 ± 0.007 | 0.9722 | 1.624 ± 0.015 | 0.9930 |
| BHT | 1.269 ± 0.005 | 0.9880 | 1.561 ± 0.089 | 0.9978 | 0.909 ± 0.006 | 0.9874 |
| EEFS | 0.985 ± 0.013 | 0.9199 | 0.830 ± 0.022 | 0.9869 | 0.873 ± 0.012 | 0.9553 |
| WEFS | 1.058 ± 0.021 | 0.9973 | 0.456 ± 0.034 | 0.9742 | 0.424 ± 0.016 | 0.9510 |
| Compounds | DPPH• Scavenging | ABTS•+ Scavenging | Fe2+ Chelating | |||
|---|---|---|---|---|---|---|
| IC50 * | r2 | IC50 * | r2 | IC50 * | r2 | |
| α-tocopherol | 23.1 ± 0.032 | 0.9825 | 15.4 ± 0.03 | 0.9866 | 33.0 ± 0.17 | 0.9109 |
| Ascorbic acid | 16.1 ± 0.03 | 0.9566 | 23.1 ± 0.01 | 0.9998 | 99.0 ± 0.36 | 0.9985 |
| BHT | 31.5 ± 0.01 | 0.9754 | 26.7 ± 0.08 | 0.9717 | 14.8 ± 0.56 | 0.9646 |
| EEFS | 34.7 ± 0.22 | 0.9965 | 7.8 ± 0.01 | 0.9844 | 31.5 ± 0.13 | 0.903 |
| WEFS | 57.8 ± 0.07 | 0.9993 | 19.3 ± 0.04 | 0.9419 | - * | - * |
| No | Compounds | WEFS | EEFS | U (%) |
|---|---|---|---|---|
| 1 | Ascorbic acid | 47.41 | 172.44 | 3.94 |
| 2 | (−)-Epigallocatechin | <LOD | <LOD | 3.09 |
| 3 | (−)-Epigallocatechin gallate | 1.59 | <LOD | 3.76 |
| 4 | Chlorogenic acid | 10103.18 | 44642.39 | 3.58 |
| 5 | Fumaric acid | <LOD | 3109.11 | 2.88 |
| 6 | Verbascoside | 6.59 | 225.72 | 2.93 |
| 7 | Orientin | 491.59 | 15329.03 | 3.67 |
| 8 | Caffeic acid | 24.41 | 126.39 | 3.74 |
| 9 | (+)-trans taxifolin | <LOD | 2.10 | 3.35 |
| 10 | Luteolin-7-rutinoside | <LOD | <LOD | 3.06 |
| 11 | Naringin | <LOD | <LOD | 4.20 |
| 12 | Luteolin 7-glucoside | <LOD | <LOD | 4.14 |
| 13 | Rutin | 14013.35 | 156907.40 | 3.07 |
| 14 | Rosmarinic acid | 26.88 | 134.22 | 3.77 |
| 15 | Hyperoside | 105.94 | 2633.75 | 3.46 |
| 16 | Dihydrokaempferol | <LOD | 2.33 | 2.86 |
| 17 | Quercitrin | 3.82 | 105.78 | 3.78 |
| 18 | Myricetin | <LOD | 0.47 | 4.18 |
| 19 | Quercetin | 30.82 | 197.18 | 2.95 |
| 20 | Salicylic acid | 27.53 | 130.76 | 1.89 |
| 21 | Naringenin | <LOD | 23.12 | 4.20 |
| 22 | Luteolin | <LOD | 11.06 | 3.42 |
| 23 | Nepetin | <LOD | <LOD | 2.19 |
| 24 | Apigenin | <LOD | 8.60 | 2.87 |
| 25 | Hispidulin | <LOD | 66.12 | 3.41 |
| 26 | Isosakuranetin | <LOD | <LOD | 3.98 |
| 27 | Caffeic acid phenethyl ester | <LOD | 0.23 | 3.13 |
| 28 | Chrysin | 6.47 | 1.38 | 3.24 |
| 29 | Acacetin | 5.53 | 9.92 | 3.98 |
| 30 | Emodin | <LOD | 1.56 | 4.27 |
| Compounds | RT | m/z | δ ppm | Ionization Mode | Linear Range | Linear Regression Equation | LOD/LOQ | R2 | Recovery |
|---|---|---|---|---|---|---|---|---|---|
| Ascorbic acid | 1.99 | 175.0248 | −0.81 | Negative | 0.5–10 | y = 0.00347x − 0.00137 | 0.39/1.29 | 0.9988 | 96.20 |
| (−)-Epigallocatechin | 2.15 | 307.0812 | −1.07 | Positive | 0.3–5 | y = 0.00317x + 0.000443 | 0.17/0.57 | 0.9947 | 102.22 |
| Chlorogenic acid | 2.21 | 353.0878 | −0.91 | Negative | 0.05–10 | y = 0.00817x + 0.000163 | 0.02/0.06 | 0.9994 | 96.68 |
| Verbascoside | 2.43 | 623.1981 | −0.61 | Negative | 0.1–10 | y = 0.00758x + 0.000563 | 0.03/0.1 | 0.9995 | 96.19 |
| Orientin | 2.45 | 447.0933 | −0.45 | Negative | 0.1–10 | y = 0.00757x + 0.000347 | 0.01/0.03 | 0.9993 | 96.22 |
| Caffeic acid | 2.89 | 179.0350 | 1.72 | Negative | 0.3–10 | y = 0.0304x + 0.00366 | 0.08/0.27 | 0.9993 | 94.51 |
| Luteolin-7-rutinoside | 3.09 | 593.1512 | −0.26 | Negative | 0.1–10 | y = 0.00879x + 0.000739 | 0.01/0.03 | 0.9988 | 93.05 |
| Naringin | 3.17 | 579.1719 | −0.07 | Negative | 0.05–10 | y = 0.00576x − 0.000284 | 0.01/0.03 | 0.9991 | 101.91 |
| Luteolin 7-glucoside | 3.85 | 447.0933 | −0.32 | Negative | 0.1–7 | y = 0.0162x + 0.00226 | 0.01/0.03 | 0.9961 | 96.31 |
| Hesperidin | 3.85 | 609.1825 | 0.29 | Negative | 0.05–10 | y = 0.00423x + 0.0000138 | 0.01/0.03 | 0.9994 | 96.14 |
| Rutin | 4.12 | 609.1461 | 0.12 | Negative | 0.05–10 | y = 0.00329x − 0.00005576 | 0.01/0.03 | 0.999 | 96.97 |
| Syringic acid | 4.24 | 197.0456 | −0.26 | Negative | 0.5–10 | y = 0.0000831x + 0.000024 | 0.1/0.3 | 0.9991 | 97.29 |
| Rosmarinic acid | 4.48 | 359.0772 | 0.01 | Negative | 0.05–10 | y = 0.00717x − 0.0003067 | 0.01/0.03 | 0.9992 | 99.85 |
| Hyperoside | 4.66 | 463.0882 | −0.17 | Negative | 0.05–10 | y = 0.0072x − 0.00003096 | 0.01/0.03 | 0.9995 | 96.62 |
| Apigenin 7-glucoside | 4.58 | 431.0984 | −0.06 | Negative | 0.3–7 | y = 0.0246x + 0.00306 | 0.01/0.03 | 0.9962 | 96.07 |
| Quercitrin | 4.88 | 447.0933 | −0.18 | Negative | 0.05–10 | y = 0.0179 + 0.0003331 | 0.01/0.03 | 0.999 | 97.00 |
| Quercetin | 5.13 | 301.0354 | −0.32 | Negative | 0.1–10 | y = 0.0509x + 0.00467 | 0.01/0.03 | 0.9978 | 96.41 |
| Salicylic acid | 5.15 | 137.0244 | −0.44 | Negative | 0.3–10 | y= 0.0361x + 0.00245 | 0.01/0.03 | 0.9982 | 92.88 |
| Naringenin | 5.68 | 271.0612 | −0.12 | Negative | 0.1–10 | y = 0.0281x + 0.00182 | 0.01/0.03 | 0.9995 | 86.65 |
| Luteolin | 5.72 | 285.0405 | 0.46 | Negative | 0.1–10 | y = 0.117x + 0.00848 | 0.01/0.03 | 0.9981 | 96.98 |
| Apigenin | 5.74 | 269.0456 | −0.25 | Negative | 0.3–10 | y = 0.104x + 0.0199 | 0.01/0.03 | 0.9998 | 81.55 |
| Hispidulin | 5.84 | 301.0707 | −0.18 | Positive | 0.05–10 | y = 0.02614x + 0.0003114 | 0.01/0.03 | 0.9993 | 98.36 |
| Isosakuranetin | 5.86 | 285.0769 | −0.21 | Negative | 0.05–10 | y = 0.0235x + 0.000561 | 0.01/0.03 | 0.9992 | 96.56 |
| Chrysin | 6.20 | 253.0506 | −0.29 | Negative | 0.05–7 | y = 0.0964x − 0.0002622 | 0.01/0.03 | 0.999 | 87.92 |
| Acacetin | 6.24 | 283.0612 | −1.08 | Negative | 0.05–7 | y = 0.046x + 0.0001875 | 0.01/0.03 | 0.9995 | 87.52 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kızıltaş, H.; Bingol, Z.; Gören, A.C.; Kose, L.P.; Durmaz, L.; Topal, F.; Alwasel, S.H.; Gulcin, İ. LC-HRMS Profiling and Antidiabetic, Anticholinergic, and Antioxidant Activities of Aerial Parts of Kınkor (Ferulago stellata). Molecules 2021, 26, 2469. https://doi.org/10.3390/molecules26092469
Kızıltaş H, Bingol Z, Gören AC, Kose LP, Durmaz L, Topal F, Alwasel SH, Gulcin İ. LC-HRMS Profiling and Antidiabetic, Anticholinergic, and Antioxidant Activities of Aerial Parts of Kınkor (Ferulago stellata). Molecules. 2021; 26(9):2469. https://doi.org/10.3390/molecules26092469
Chicago/Turabian StyleKızıltaş, Hatice, Zeynebe Bingol, Ahmet Ceyhan Gören, Leyla Polat Kose, Lokman Durmaz, Fevzi Topal, Saleh H. Alwasel, and İlhami Gulcin. 2021. "LC-HRMS Profiling and Antidiabetic, Anticholinergic, and Antioxidant Activities of Aerial Parts of Kınkor (Ferulago stellata)" Molecules 26, no. 9: 2469. https://doi.org/10.3390/molecules26092469
APA StyleKızıltaş, H., Bingol, Z., Gören, A. C., Kose, L. P., Durmaz, L., Topal, F., Alwasel, S. H., & Gulcin, İ. (2021). LC-HRMS Profiling and Antidiabetic, Anticholinergic, and Antioxidant Activities of Aerial Parts of Kınkor (Ferulago stellata). Molecules, 26(9), 2469. https://doi.org/10.3390/molecules26092469







