Valorization of Apple Peels through the Study of the Effects on the Amyloid Aggregation Process of κ-Casein
Abstract
1. Introduction
2. Results
2.1. Total Phenolic Content (TPC), Antioxidant Properties, and Phenolic Characterization of Extracts from Apples Peels
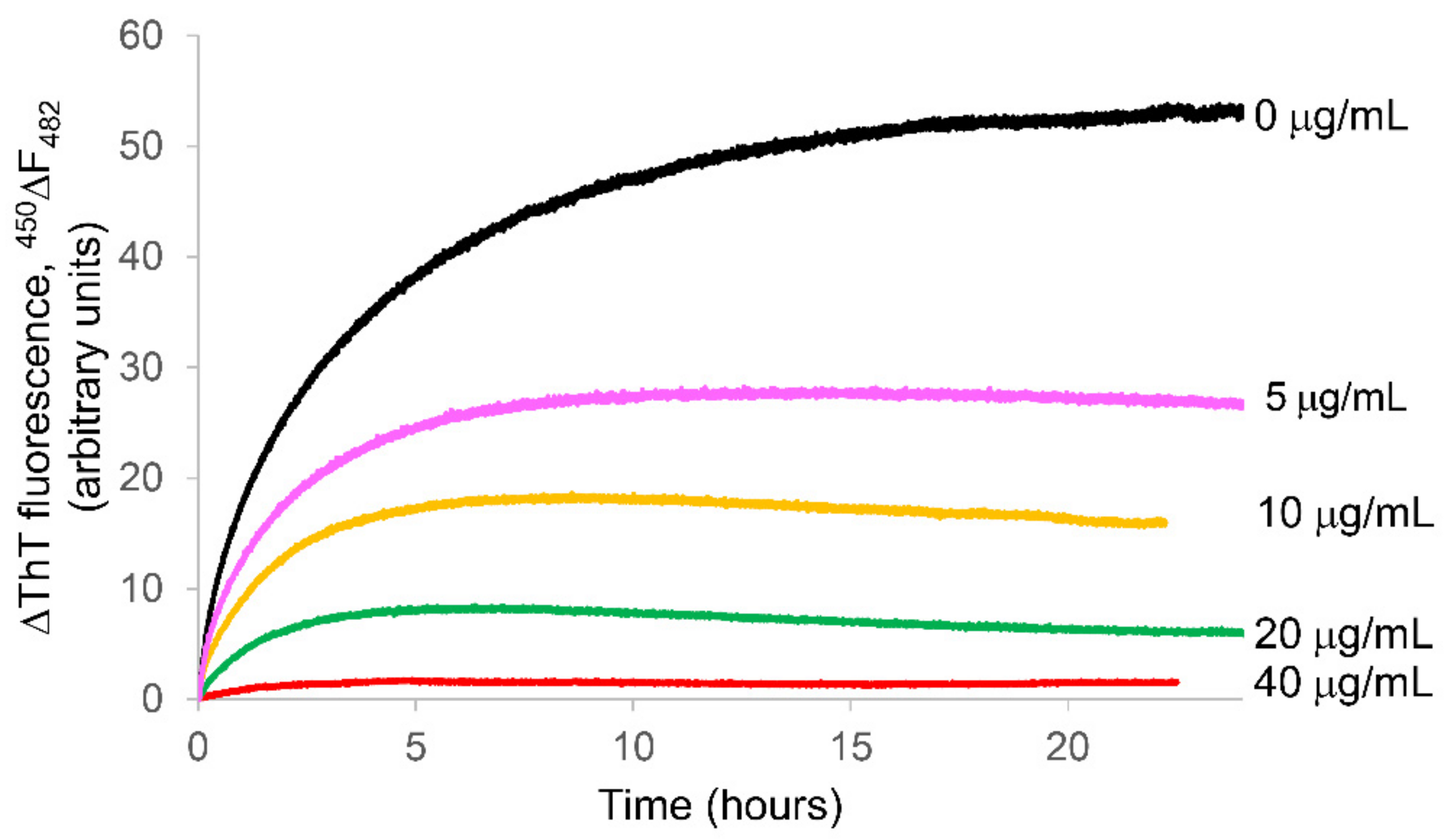
2.2. Ability of the APP Extract to Influence κ-Casein Fibrillogenesis and Disaggregate κ-casein Preformed Fibrils
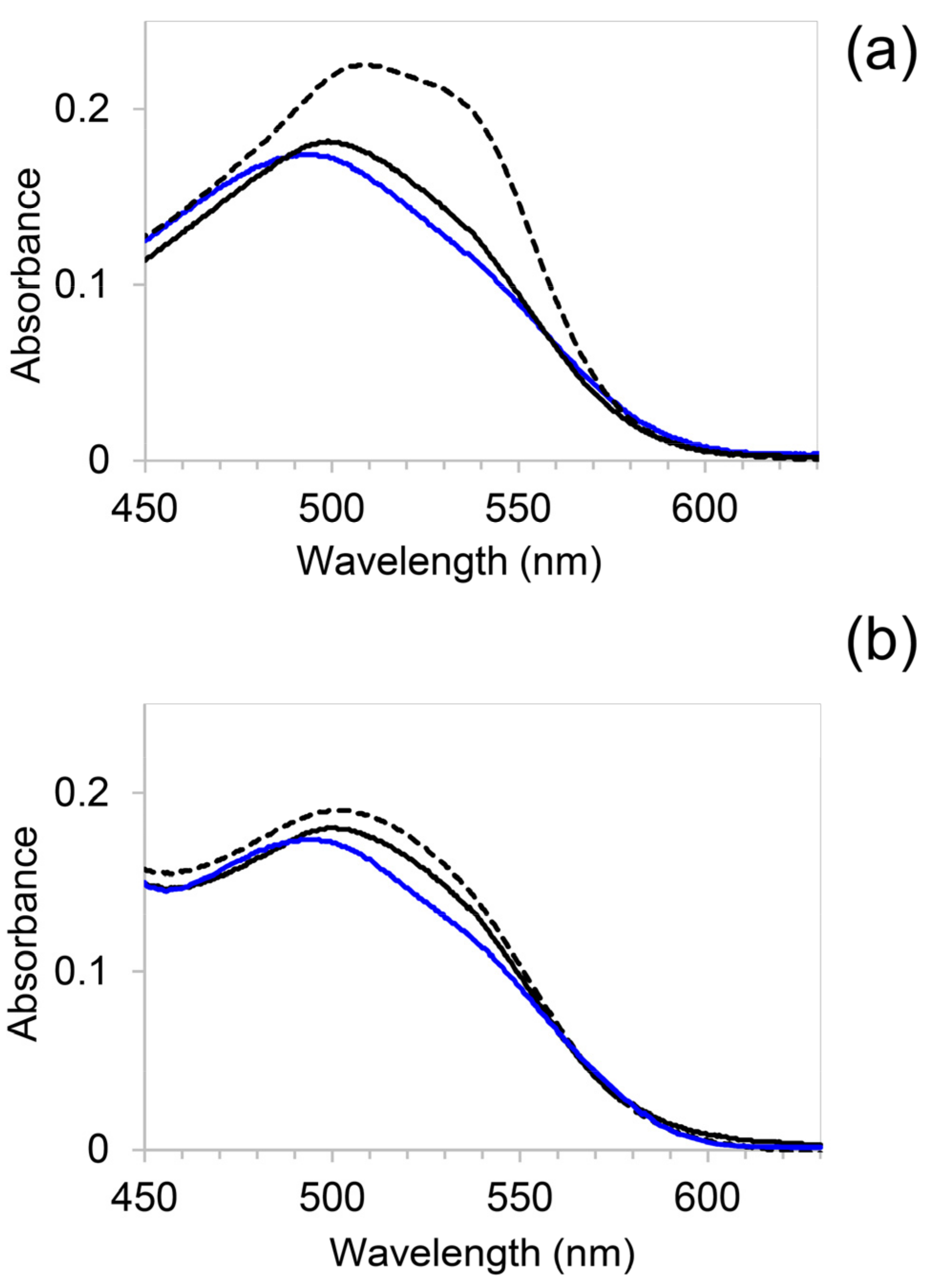
2.3. β-Sheet-Amyloid Formation Assessed by the Congo Red Binding Assay
2.4. Apple Peel Polyphenols Influence on κ-Casein Secondary Structure
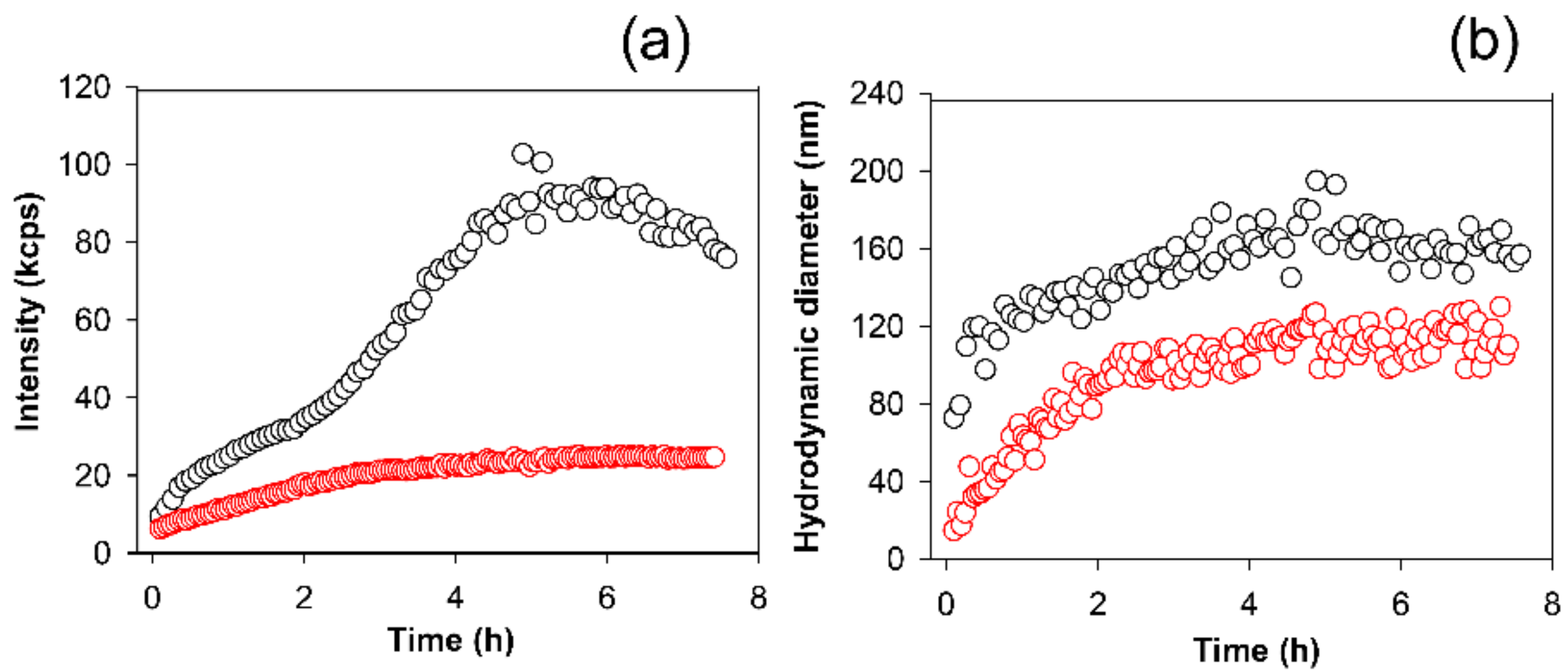
2.5. Apple Peel Polyphenols Influence on κ-Casein Aggregation Monitored by Light Scattering
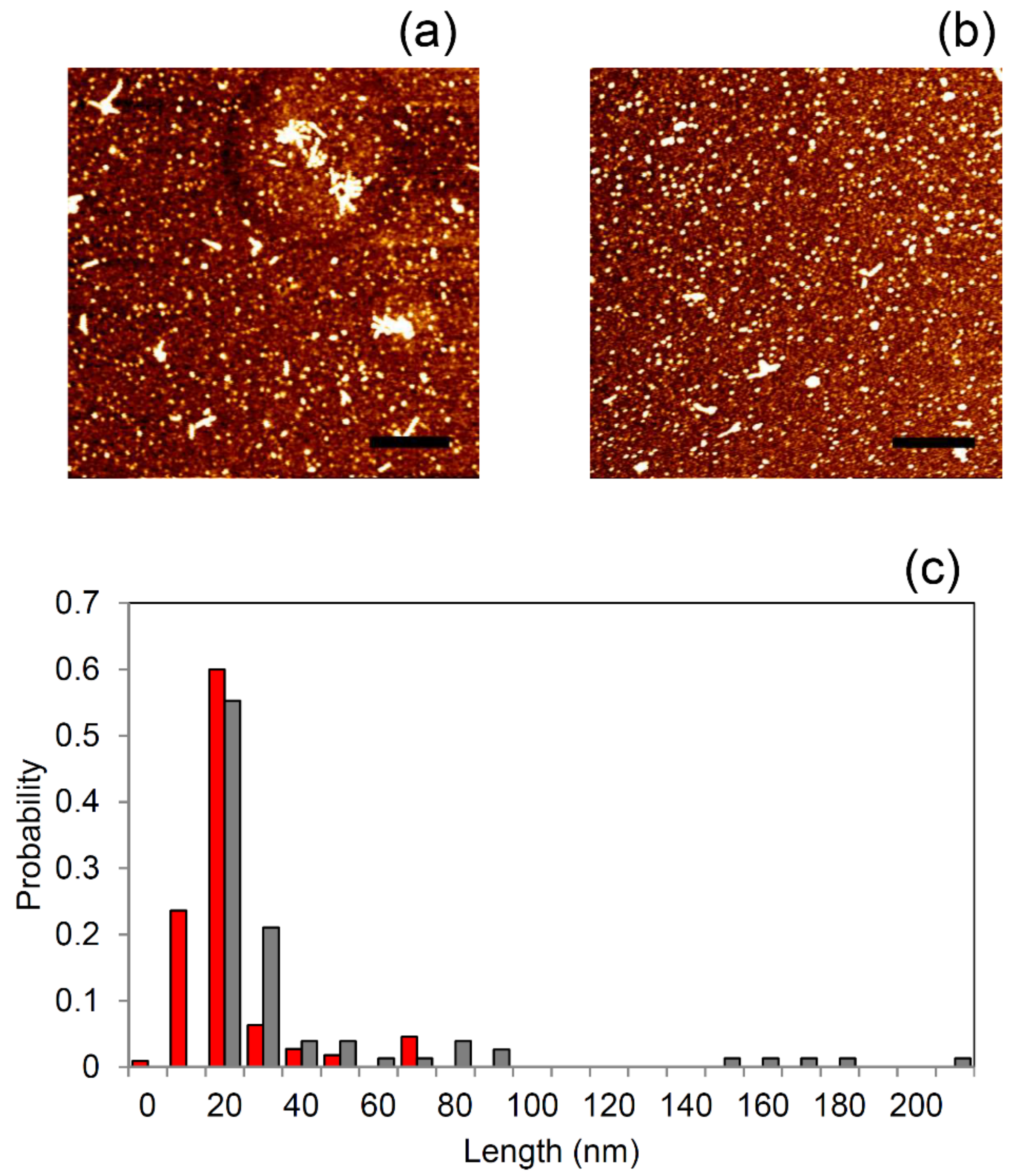
2.6. Morphology of κ-Casein Aggregates Incubated with Apple Peel Polyphenols
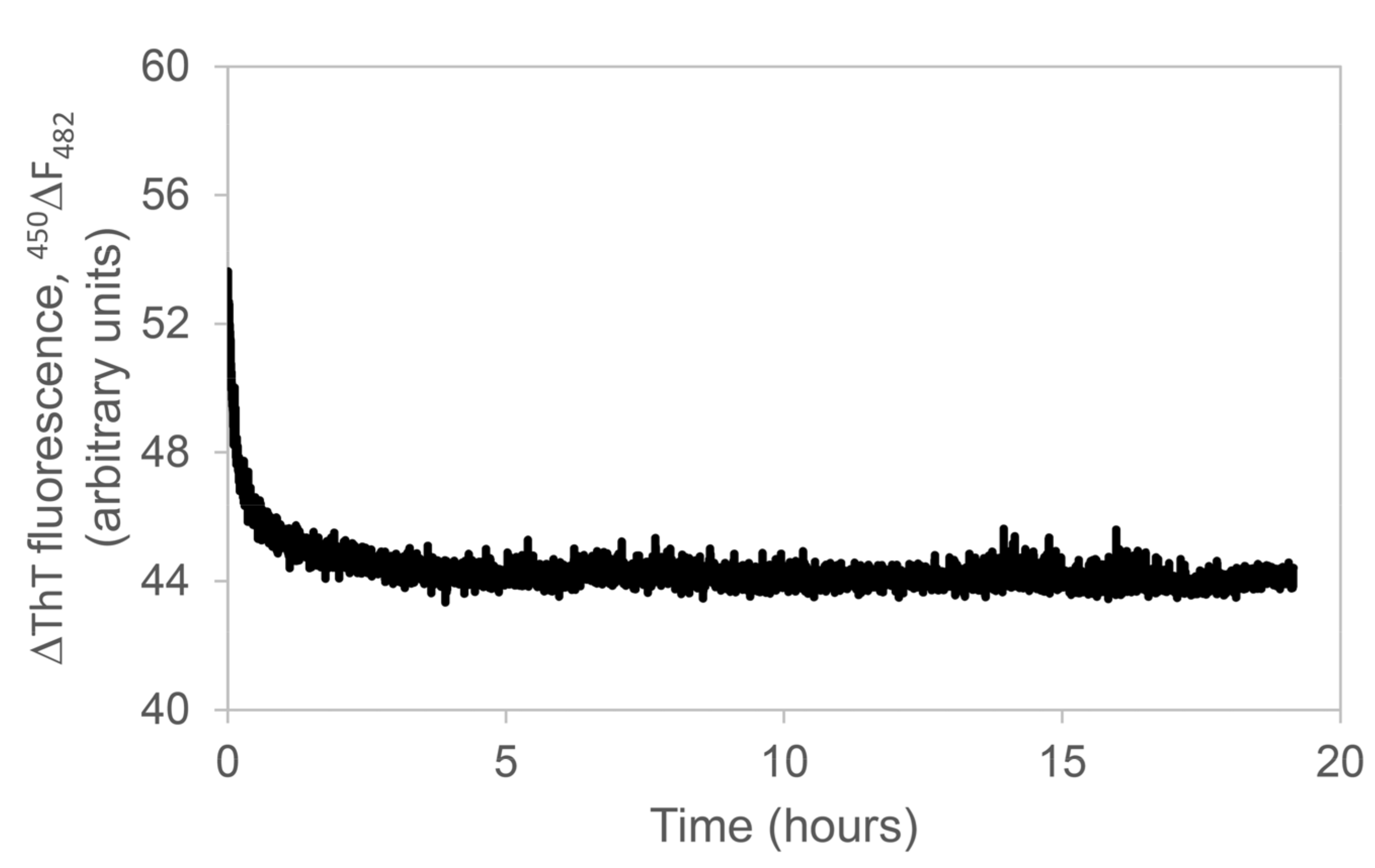
2.7. Apple Peel Polyphenols Effect on Aβ1–42 Peptide Aggregation Monitored by Light Scattering
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Plant Materials
4.3. APP Extraction, Phenolic Content Determination and Antioxidant Properties Evaluation
4.3.1. Polyphenol Extraction
4.3.2. Total Polyphenol Content (TPC) by Folin–Ciocalteu
4.3.3. Identification of Polyphenols by UHPLC–HESI-MS
4.3.4. Oxygen Radical Absorbance Capacity (ORAC) Assay
4.4. Cell Culture and Cytotoxicity Assay
4.5. κ-Casein Amyloid Formation
4.6. Atomic Force Microscopy (AFM) Measurements
4.7. Thioflavin T (ThT) Spectrofluorometric Measurements
4.8. Congo Red Binding Assay
4.9. Aβ1–42 Peptide Amyloid Formation
4.10. Static and Dynamic Light Scattering
4.11. Circular Dichroism
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
Abbreviations
| APP | Apple Peel Polyphenols |
| CD | Circular Dichroism |
| LS | Light Scattering |
| ThT | Thioflavin T |
| TE | Trolox Equivalents |
| CTE | Catechin Equivalents |
| FW | Fresh Weight |
| AFM | Atomic Force Microscopy |
| FAR-UV | Far-UltraViolet |
| ORAC | Oxygen Radical Antioxidant Capacity |
| TPC | Total Polyphenol Content |
| CR | Congo Red |
References
- Diacono, M.; Persiani, A.; Testani, E.; Montemurro, F.; Ciaccia, C. Recycling agricultural wastes and by-products in organic farming: Biofertilizer production, yield performance and carbon footprint analysis. Sustainability 2019, 11, 3824. [Google Scholar] [CrossRef]
- Gulsunoglu, Z.; Purves, R.; Karbancioglu-Guler, F.; Kilic-Akyilmaz, M. Enhancement of phenolic antioxidants in industrial apple waste by fermentation with Aspergillus spp. Biocatal. Agric. Biotechnol. 2020, 25, 101562. [Google Scholar] [CrossRef]
- Wolfe, K.L.; Liu, R.H. Apple peels as a value-added food ingredient. J. Agric. Food Chem. 2003, 51, 1676–1683. [Google Scholar] [CrossRef]
- Wandjou, J.G.N.; Lancioni, L.; Barbalace, M.C.; Hrelia, S.; Papa, F.; Sagratini, G.; Vittori, S.; Dall’Acqua, S.; Caprioli, G.; Beghelli, D.; et al. Comprehensive characterization of phytochemicals and biological activities of the Italian ancient apple ‘Mela Rosa dei Monti Sibillini’. Food Res. Int. 2020, 137, 109422. [Google Scholar] [CrossRef]
- Lončarić, A.; Matanović, K.; Ferrer, P.; Kovač, T.; Šarkanj, B.; Skendrović Babojelić, M.; Lores, M. Peel of traditional apple varieties as a great source of bioactive compounds: Extraction by micro-matrix solid-phase dispersion. Foods 2020, 9, 80. [Google Scholar] [CrossRef]
- Wolfe, K.; Wu, X.; Liu, R.H. Antioxidant activity of apple peels. J. Agric. Food Chem. 2003, 51, 609–614. [Google Scholar] [CrossRef] [PubMed]
- Miura, T.; Chiba, M.; Kasai, K.; Nozaka, H.; Nakamura, T.; Shoji, T.; Kanda, T.; Ohtake, Y.; Sato, T. Apple procyanidins induce tumor cell apoptosis through mitochondrial pathway activation of caspase-3. Carcinogenesis 2008, 29, 585–593. [Google Scholar] [CrossRef] [PubMed]
- Akiyama, H.; Sato, Y.; Watanabe, T.; Nagaoka, M.H.; Yoshioka, Y.; Shoji, T.; Kanda, T.; Yamada, K.; Totsuka, M.; Teshima, R.; et al. Dietary unripe apple polyphenol inhibits the development of food allergies in murine models. FEBS Lett. 2005, 579, 4485–4491. [Google Scholar] [CrossRef] [PubMed]
- Sunagawa, T.; Shimizu, T.; Kanda, T.; Tagashira, M.; Sami, M.; Shirasawa, T. Procyanidins from apples (Malus pumila Mill.) extend the lifespan of Caenorhabditis elegans. Planta Med. 2011, 77, 122–127. [Google Scholar] [CrossRef] [PubMed]
- Zhao, S.; Zhang, L.; Yang, C.; Li, Z.; Rong, S. Procyanidins and Alzheimer’s disease. Mol. Neurobiol. 2019, 56, 5556–5567. [Google Scholar] [CrossRef] [PubMed]
- Khan, H.; Ullah, H.; Aschner, M.; Cheang, W.S.; Akkol, E.K. Neuroprotective effects of quercetin in Alzheimer’s disease. Biomolecules 2020, 10, 59. [Google Scholar] [CrossRef]
- Boyina, H.K.; Geethakhrishnan, S.L.; Panuganti, S.; Gangarapu, K.; Devarakonda, K.P.; Bakshi, V.; Guggilla, S.R. In Silico and In Vivo Studies on Quercetin as Potential Anti-Parkinson Agent. In GeNeDis 2018: Advances in Experimental Medicine and Biology; Vlamos, P., Ed.; Springer: Cham, Switerlands, 2020; Volume 1195, pp. 1–11. [Google Scholar]
- Phan, H.T.; Samarat, K.; Takamura, Y.; Azo-Oussou, A.F.; Nakazono, Y.; Vestergaard, M.D.C. Polyphenols modulate alzheimer’s amyloid beta aggregation in a structure-dependent manner. Nutrients 2019, 11, 756. [Google Scholar] [CrossRef]
- Carrell, R.W.; Lomas, D.A. Conformational disease. Lancet 1997, 350, 134–138. [Google Scholar] [CrossRef]
- Makin, O.S.; Serpell, L.C. Examining the structure of the mature amyloid fibril. Biochem. Soc. Trans. 2002, 30, 521–525. [Google Scholar] [CrossRef] [PubMed]
- Serpell, L.C.; Sunde, M.; Benson, M.D.; Tennent, G.A.; Pepys, M.B.; Fraser, P.E. The protofilament substructure of amyloid fibrils. J. Mol. Biol. 2000, 300, 1033–1039. [Google Scholar] [CrossRef] [PubMed]
- Dobson, C.M. Protein folding and misfolding. Nature 2003, 426, 884–890. [Google Scholar] [CrossRef] [PubMed]
- Koike, H.; Katsuno, M. Ultrastructure in Transthyretin Amyloidosis: From Pathophysiology to Therapeutic Insights. Biomedicines 2019, 7, 11. [Google Scholar] [CrossRef]
- Koike, H.; Nishi, R.; Ikeda, S.; Kawagashira, Y.; Iijima, M.; Sakurai, T.; Shimohata, T.; Katsuno, M.; Sobue, G.J. The morphology of amyloid fibrils and their impact on tissue damage in hereditary transthyretin amyloidosis: An ultrastructural study. J. Neurol. Sci. 2018, 394, 99–106. [Google Scholar] [CrossRef]
- Canale, C.; Seghezza, S.; Vilasi, S.; Carrotta, R.; Bulone, D.; Diaspro, A.; San Biagio, P.L.; Dante, S. Different effects of Alzheimer’s peptide Aβ (1–40) oligomers and fibrils on supported lipid membranes. Biophys. Chem. 2013, 182, 23–29. [Google Scholar] [CrossRef]
- Walsh, D.M.; Klyubin, I.; Fadeeva, J.V.; Rowan, M.J.; Selkoe, D.J. Amyloid-β oligomers: Their production, toxicity and therapeutic inhibition. Biochem. Soc. Trans. 2002, 30, 552–557. [Google Scholar] [CrossRef]
- Vilasi, A.; Vilasi, S.; Romano, R.; Acernese, F.; Barone, F.; Balestrieri, M.L.; Maritato, R.; Irace, G.; Sirangelo, I. Unraveling amyloid toxicity pathway in NIH3T3 cells by a combined proteomic and 1 H-NMR metabonomic approach. J. Cell. Physiol. 2013, 228, 1359–1367. [Google Scholar] [CrossRef]
- Choi, D.Y.; Lee, Y.J.; Hong, J.T.; Lee, H.J. Antioxidant properties of natural polyphenols and their therapeutic potentials for Alzheimer’s disease. Brain Res. Bull. 2012, 87, 144–153. [Google Scholar] [CrossRef]
- Ngoungoure, V.L.N.; Schluesener, J.; Moundipa, P.F.; Schluesener, H. Natural polyphenols binding to amyloid: A broad class of compounds to treat different human amyloid diseases. Mol. Nutr. Food Res. 2015, 59, 8–20. [Google Scholar] [CrossRef]
- Stefani, M.; Rigacci, S. Beneficial properties of natural phenols: Highlight on protection against pathological conditions associated with amyloid aggregation. Biofactors 2014, 40, 482–493. [Google Scholar] [CrossRef]
- Francioso, A.; Punzi, P.; Boffi, A.; Lori, C.; Martire, S.; Giordano, C.; D’Erme, M.; Mosca, L. β-sheet interfering molecules acting against β-amyloid aggregation and fibrillogenesis. Bioorg. Med. Chem. 2015, 23, 1671–1683. [Google Scholar] [CrossRef]
- Porat, Y.; Abramowitz, A.; Gazit, E. Inhibition of amyloid fibril formation by polyphenols: Structural similarity and aromatic interactions as a common inhibition mechanism. Chem. Biol. Drug Des. 2006, 67, 27–37. [Google Scholar] [CrossRef]
- Hudson, S.A.; Ecroyd, H.; Dehle, F.C.; Musgrave, I.F.; Carver, J.A. (−)-Epigallocatechin-3-gallate (EGCG) maintains κ-casein in its pre-fibrillar state without redirecting its aggregation pathway. J. Mol. Biol. 2009, 392, 689–700. [Google Scholar] [CrossRef]
- Wang, J.B.; Wang, Y.M.; Zeng, C.M. Quercetin inhibits amyloid fibrillation of bovine insulin and destabilizes preformed fibrils. Biochem. Biophys. Res. Commun. 2011, 415, 675–679. [Google Scholar] [CrossRef]
- Ono, K.; Yoshiike, Y.; Takashima, A.; Hasegawa, K.; Naiki, H.; Yamada, M. Potent anti-amyloidogenic and fibril-destabilizing effects of polyphenols in vitro: Implications for the prevention and therapeutics of Alzheimer’s disease. J. Neurochem. 2003, 87, 172–181. [Google Scholar] [CrossRef]
- Toda, T.; Sunagawa, T.; Kanda, T.; Tagashira, M.; Shirasawa, T.; Shimizu, T. Apple Procyanidins Suppress Amyloid-Protein Aggregation. Biochem. Res. Int. 2011, 2011, 784698. [Google Scholar] [CrossRef]
- Cui, L.; Zhang, Y.; Cao, H.; Wang, Y.; Teng, T.; Ma, G.; Li, Y.; Li, K.; Zhang, Y. Ferulic acid inhibits the transition of amyloid-β42 monomers to oligomers but accelerates the transition from oligomers to fibrils. J. Alzheimers Dis. 2013, 37, 19–28. [Google Scholar] [CrossRef]
- Iannuzzi, C.; Borriello, M.; Irace, G.; Cammarota, M.; Di Maro, A.; Sirangelo, I. Vanillin Affects Amyloid Aggregation and Non-Enzymatic Glycation in Human Insulin. Sci. Rep. 2017, 7, 15086. [Google Scholar] [CrossRef]
- Ma, W.; Tribet, C.; Guyot, S.; Zanchi, D. Tannin-controlled micelles and fibrils of k-casein. Chem. Phys. 2019, 151, 245103. [Google Scholar] [CrossRef]
- Hudson, S.A.; Ecroyd, H.; Kee, T.W.; Carver, J.A. The thioflavin T fluorescence assay for amyloid fibril detection can be biased by the presence of exogenous compounds. FEBS J. 2009, 276, 5960–5972. [Google Scholar] [CrossRef] [PubMed]
- Holt, C.; Sawyer, L. Caseins as rheomorphic proteins: Interpretation of primary and secondary structures of the αS1-, β-and κ-caseins. J. Chem. Soc. Faraday Trans. 1993, 89, 2683–2692. [Google Scholar] [CrossRef]
- Thurn, A.; Burchard, W.; Niki, R. Structure of casein micelles I. Small angle neutron scattering and light scattering from β-and χ-casein. Colloid Polym. Sci 1987, 265, 653–666. [Google Scholar] [CrossRef]
- Vreeman, H.J.; Brinkhuis, J.A.; Van der Spek, C.A. Some association properties of bovine SH-k-casein. Biophys. Chem. 1981, 14, 185–193. [Google Scholar] [CrossRef]
- McSweeney, P.L.; Fox, P.F. Advanced Dairy Chemistry: Volume 1A: Proteins: Basic Aspects; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2013. [Google Scholar]
- Farrell, H.M.; Cooke, P.H.; Wickham, E.D.; Piotrowski, E.G.; Hoagland, P.D. Environmental influences on bovine κ-casein: Reduction and conversion to fibrillar (amyloid) structures. J. Protein Chem. 2003, 22, 259–273. [Google Scholar] [CrossRef]
- Ecroyd, H.; Koudelka, T.; Thorn, D.C.; Williams, D.M.; Devlin, G.; Hoffmann, P.; Carver, J.A. Dissociation from the oligomeric state is the rate-limiting step in fibril formation by κ-casein. J. Biol. Chem. 2008, 283, 9012–9022. [Google Scholar] [CrossRef]
- Ecroyd, H.; Thorn, D.C.; Liu, Y.; Carver, J.A. The dissociated form of κ-casein is the precursor to its amyloid fibril formation. Biochem. J. 2010, 429, 251–260. [Google Scholar] [CrossRef]
- Thorn, D.C.; Ecroyd, H.; Carver, J.A. The two-faced nature of milk casein proteins: Amyloid fibril formation and chaperone-like activity. Aust. J. Dairy Technol. 2009, 64, 34. [Google Scholar]
- Leonil, J.; Henry, G.; Jouanneau, D.; Delage, M.M.; Forge, V.; Putaux, J.L. Kinetics of fibril formation of bovine κ-casein indicate a conformational rearrangement as a critical step in the process. J. Mol. Biol. 2008, 381, 1267–1280. [Google Scholar] [CrossRef] [PubMed]
- Thorn, D.C.; Meehan, S.; Sunde, M.; Rekas, A.; Gras, S.L.; MacPhee, C.E.; Dobson, C.M.; Wilson, M.R.; Carver, J.A. Amyloid fibril formation by bovine milk κ-casein and its inhibition by the molecular chaperones αS-and β-casein. Biochemistry 2005, 44, 17027–17036. [Google Scholar] [CrossRef]
- Carrotta, R.; Canale, C.; Diaspro, A.; Trapani, A.; San Biagio, P.L.; Bulone, D. Inhibiting effect of α(s1)-casein on Aβ(1–40) fibrillogenesis. Biochim. Biophys. Acta Gen. Subj. 2012, 1820, 124–132. [Google Scholar] [CrossRef]
- Carrotta, R.; Vilasi, S.; Librizzi, F.; Martorana, V.; Bulone, D.; San Biagio, P.L. α-Casein inhibition mechanism in concanavalin A aggregation process. J. Phys. Chem. B 2012, 116, 14700–14707. [Google Scholar] [CrossRef]
- Librizzi, F.; Carrotta, R.; Spigolon, D.; Bulone, D.; San Biagio, P.L. α-Casein inhibits insulin amyloid formation by preventing the onset of secondary nucleation processes. J. Phys. Chem. Lett. 2014, 5, 3043–3048. [Google Scholar] [CrossRef]
- Feng, B.Y.; Toyama, B.H.; Wille, H.; Colby, D.W.; Collins, S.R.; May, B.C.; Prusiner, S.B.; Weissman, J.; Shoichet, B.K. Small-molecule aggregates inhibit amyloid polymerization. Nat. Chem. Biol. 2008, 4, 197–199. [Google Scholar] [CrossRef]
- Kumosinski, T.F.; Brown, E.M.; Farrell, H.M., Jr. Three-dimensional molecular modeling of bovine caseins: A refined, energy-minimized k-casein structure. J. Dairy Sci. 1993, 76, 2507–2520. [Google Scholar] [CrossRef]
- Jain, N.; Bhattacharya, M.; Mukhopadhyay, S. Chain collapse of an amyloidogenic intrinsically disordered protein. Biophys. J. 2011, 101, 1720–1729. [Google Scholar] [CrossRef]
- Sabaté, R.; Estelrich, J.J. Evidence of the existence of micelles in the fibrillogenesis of beta-amyloid peptide. Phys. Chem. B 2005, 109, 11027–11032. [Google Scholar] [CrossRef]
- Morel, B.; Hingant, E.; Fontes, P.; Alvarez-Martinez, M.T.; Arnaud, J.D.; Liautard, J.P.; Pujo-Menjouet, L.; Hingant, E.; Fontes, P.; Alvarez-Martinez, M.T.; et al. Dynamic micellar oligomers of amyloid beta peptides play a crucial role in their aggregation mechanisms. Phys. Chem. Chem. Phys. 2018, 20, 20597–20614. [Google Scholar] [CrossRef]
- Hingant, E.; Fontes, P.; Alvarez-Martinez, M.T.; Arnaud, J.D.; Liautard, J.P.; Pujo-Menjouet, L. A micellar on-pathway intermediate step explains the kinetics of prion amyloid formation. PLoS Comput. Biol. 2014, 10, e1003735. [Google Scholar] [CrossRef]
- Liu, R.H. Health benefits of fruit and vegetables are from additive and synergistic combinations of phytochemicals. Am. J. Clin. Nutr. 2003, 78, 517S–520S. [Google Scholar] [CrossRef]
- Prior, R.L. Oxygen radical absorbance capacity (ORAC): New horizons in relating dietary antioxidants/bioactives and health benefits. J. Funct. Foods 2015, 18, 797–810. [Google Scholar] [CrossRef]
- Jairath, G.; Chatli, M.K.; Biswas, K. Comparative Study on In vitro and In vivo Evaluation of Antioxidant Potential of Apple Peel Extract and Aloe Vera Gel. J. Food Process. Preserv. 2016, 40, 607–614. [Google Scholar] [CrossRef]
- Bitalebi, S.; Nikoo, M.; Rahmanifarah, K.; Noori, F.; Gavlighi, H.A. Effect of apple peel extract as natural antioxidant on lipid and protein oxidation of rainbow trout (Oncorhynchus mykiss) mince. Int. Aquat. Res. 2019, 11, 135–146. [Google Scholar] [CrossRef]
- He, R.R.; Wang, M.; Wang, C.Z.; Chen, B.T.; Lu, C.N.; Yao, X.S.; Chen, J.X.; Kurihara, H.J. Protective effect of apple polyphenols against stress-provoked influenza viral infection in restraint mice. Agric. Food Chem. 2011, 59, 3730–3737. [Google Scholar] [CrossRef]
- Xu, Z.R.; Li, J.Y.; Dong, X.W.; Tan, Z.J.; Wu, W.Z.; Xie, Q.M.; Yang, Y.M. Apple Polyphenols Decrease Atherosclerosis and Hepatic Steatosis in ApoE-/- Mice through the ROS/MAPK/NF-κB Pathway. Nutrients 2015, 7, 7085–7105. [Google Scholar] [CrossRef]
- Ogino, Y.; Osada, K.; Nakamura, S.; Ohta, Y.; Kanda, T.; Sugano, M. Absorption of dietary cholesterol oxidation products and their downstream metabolic effects are reduced by dietary apple polyphenols. Lipids 2007, 42, 151–161. [Google Scholar] [CrossRef]
- Levine, H., III. Thioflavine T interaction with synthetic Alzheimer’s disease β-amyloid peptides: Detection of amyloid aggregation in solution. Protein Sci. 1993, 2, 404–410. [Google Scholar] [CrossRef]
- Hawe, A.; Sutter, M.; Jiskoot, W. Extrinsic fluorescent dyes as tools for protein characterization. Pharm. Res. 2008, 25, 1487–1499. [Google Scholar] [CrossRef]
- Leibiger, C.; Kosyakova, N.; Mkrtchyan, H.; Glei, M.; Trifonov, V.; Liehr, T. First molecular cytogenetic high resolution characterization of the NIH 3T3 cell line by murine multicolor banding. J. Histochem. Cytochem. 2013, 61, 306–312. [Google Scholar] [CrossRef] [PubMed]
- Sirangelo, I.; Iannuzzi, C.; Vilasi, S.; Irace, G.; Giuberti, G.; Misso, G.; D’Alessandro, A.; Abbruzzese, A.; Caraglia, M. W7FW14F apomyoglobin amyloid aggregates-mediated apoptosis is due to oxidative stress and AKT inactivation caused by Ras and Rac. J. Cell. Physiol. 2009, 221, 412–423. [Google Scholar] [CrossRef] [PubMed]
- Iannuzzi, C.; Vilasi, S.; Portaccio, M.; Irace, G.; Sirangelo, I. Heme binding inhibits the fibrillization of amyloidogenic apomyoglobin and determines lack of aggregate cytotoxicity. Protein Sci. 2007, 16, 507–516. [Google Scholar] [CrossRef]
- Coelho-Cerqueira, E.; Pinheiro, A.S.; Follmer, C. Pitfalls associated with the use of Thioflavin-T to monitor anti-fibrillogenic activity. Bioorg. Med. Chem. Lett. 2014, 24, 3194–3198. [Google Scholar] [CrossRef]
- Frid, P.; Anisimov, S.V.; Popovic, N. Congo red and protein aggregation in neurodegenerative diseases. Brain Res. Rev. 2007, 53, 135–160. [Google Scholar]
- Vilasi, S.; Carrotta, R.; Ricci, C.; Rappa, G.C.; Librizzi, F.; Martorana, V.; Ortore, M.G.; Mangione, M.R. Inhibition of Aβ1-42 Fibrillation by Chaperonins: Human Hsp60 Is a Stronger Inhibitor than Its Bacterial Homologue GroEL. ACS Chem. Neurosci. 2019, 10, 3565–3574. [Google Scholar] [CrossRef]
- Petkova, A.T.; Ishii, Y.; Balbach, J.J.; Antzutkin, O.N.; Leapman, R.D.; Delaglio, F.; Tycko, R. A structural model for Alzheimer’s β-amyloid fibrils based on experimental constraints from solid state NMR. Proc. Natl. Acad. Sci. USA 2002, 99, 16742–16747. [Google Scholar] [CrossRef]
- Stsiapura, V.I.; Maskevich, A.A.; Kuzmitsky, V.A.; Turoverov, K.K.; Kuznetsova, I.M. Computational study of thioflavin T torsional relaxation in the excited state. J. Phys. Chem. A 2007, 111, 4829–4835. [Google Scholar] [CrossRef]
- Chu, G.C.L.; Wilson, K. Evaluating Maturity of Empire, Idared and Spartan Apples. Factsheet—Ontario. Ministry of Agriculture, Food and Rural Affairs. Order No. 00-027. March 2000. Available online: http://www.omafra.gov.on.ca/english/crops/facts/00-027.htm (accessed on 13 February 2021).
- Ceymann, M.; Arrigoni, E.; Scharer, H.; Bozzi Nising, A.; Hurrell, R.F. Identification of apples rich in health-promoting flavan-3-ols and phenolic acids by measuring the polyphenol profile. J. Food Compos. 2012, 26, 128–135. [Google Scholar] [CrossRef]
- Di Stefano, V.; Pitonzo, R.; Novara, M.E.; Bongiorno, D.; Indelicato, S.; Gentile, C.; Avellone, G.; Bognanni, R.; Scandurra, S.; Melilli, M.G. Antioxidant activity and phenolic composition in pomegranate (Punica granatum L.) genotypes from south Italy by UHPLC-Orbitrap-MS approach. J. Sci. Food Agric. 2019, 99, 1038–1045. [Google Scholar] [CrossRef] [PubMed]
- Di Stefano, V.; Melilli, M.G. Effect of storage on quality parameters and phenolic content of Italian extra-virgin olive oils. Nat. Prod. Res. 2020, 34, 78–86. [Google Scholar] [CrossRef] [PubMed]
- Cao, G.; Alessio, H.M.; Cutler, R.G. Oxygen-radical absorbance capacity assay for antioxidants. Free Radic. Biol. Med. 1993, 14, 303–311. [Google Scholar] [CrossRef]
- Higuchi, K.; Hosowaka, M.; Takeda, T. Fluorometric determination of amyloid fibrils in vitro using the fluorescent dye, thioflavin T1. Anal. Biochem. 1989, 177, 244–249. [Google Scholar]
- Fezoui, Y.; Hartley, D.M.; Harper, J.D.; Khurana, R.; Walsh, D.M.; Condron, M.M.; Selkoe, D.J.; Lansbury, P.T., Jr.; Fink, A.L.; Teplow, D.B. An improved method of preparing the amyloid beta-protein for fibrillogenesis and neurotoxicity experiments. Amyloid 2000, 7, 166–178. [Google Scholar] [CrossRef]
- El-Agnaf, O.M.; Mahil, D.S.; Patel, B.P.; Austen, B.M. Oligomerization and toxicity of beta-amyloid-42 implicated in Alzheimer’s disease. Biochem. Biophys. Res. Commun. 2000, 273, 273–1007. [Google Scholar] [CrossRef]
- Pusey, P.N. Introduction to scattering experiments. In Neutrons, X-rays, and Light: Scattering Methods Applied to Soft Condensed Matter; Zemb, T., Lindner, P., Eds.; Elsevier: Amsterdam, The Netherlands, 2002. [Google Scholar]
- Berne, B.J.; Pecora, R. Dynamic Light Scattering; John Wiley: New York, NY, USA, 1976. [Google Scholar]
- Stepanek, P. The method and some applications. In Dynamic Light Scattering; Brown, W., Ed.; Clarendon Press: Oxford, UK, 1993. [Google Scholar]
- Francaviglia, D.; Farina, V.; Avellone, G.; Lo Bianco, R. Fruit yield and quality responses of apple cvars Gala and Fuji to partial rootzone drying under Mediterranean conditions. J. Agric. Sci. 2013, 151, 556–569. [Google Scholar] [CrossRef]



| Tissue | ORAC Value (µmol TE/g FW) | TPC (mg CTE/100 g of FW) |
|---|---|---|
| Peel | 118 ± 5 | 315 ± 20 |
| Flesh | 13 ± 5 | 42 ± 5 |
| Whole fruit | 22 ± 7 | 54 ± 8 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guarrasi, V.; Rappa, G.C.; Costa, M.A.; Librizzi, F.; Raimondo, M.; Di Stefano, V.; Germanà, M.A.; Vilasi, S. Valorization of Apple Peels through the Study of the Effects on the Amyloid Aggregation Process of κ-Casein. Molecules 2021, 26, 2371. https://doi.org/10.3390/molecules26082371
Guarrasi V, Rappa GC, Costa MA, Librizzi F, Raimondo M, Di Stefano V, Germanà MA, Vilasi S. Valorization of Apple Peels through the Study of the Effects on the Amyloid Aggregation Process of κ-Casein. Molecules. 2021; 26(8):2371. https://doi.org/10.3390/molecules26082371
Chicago/Turabian StyleGuarrasi, Valeria, Giacoma Cinzia Rappa, Maria Assunta Costa, Fabio Librizzi, Marco Raimondo, Vita Di Stefano, Maria Antonietta Germanà, and Silvia Vilasi. 2021. "Valorization of Apple Peels through the Study of the Effects on the Amyloid Aggregation Process of κ-Casein" Molecules 26, no. 8: 2371. https://doi.org/10.3390/molecules26082371
APA StyleGuarrasi, V., Rappa, G. C., Costa, M. A., Librizzi, F., Raimondo, M., Di Stefano, V., Germanà, M. A., & Vilasi, S. (2021). Valorization of Apple Peels through the Study of the Effects on the Amyloid Aggregation Process of κ-Casein. Molecules, 26(8), 2371. https://doi.org/10.3390/molecules26082371








