Plant Occurring Flavonoids as Modulators of the Aryl Hydrocarbon Receptor
Abstract
1. Introduction
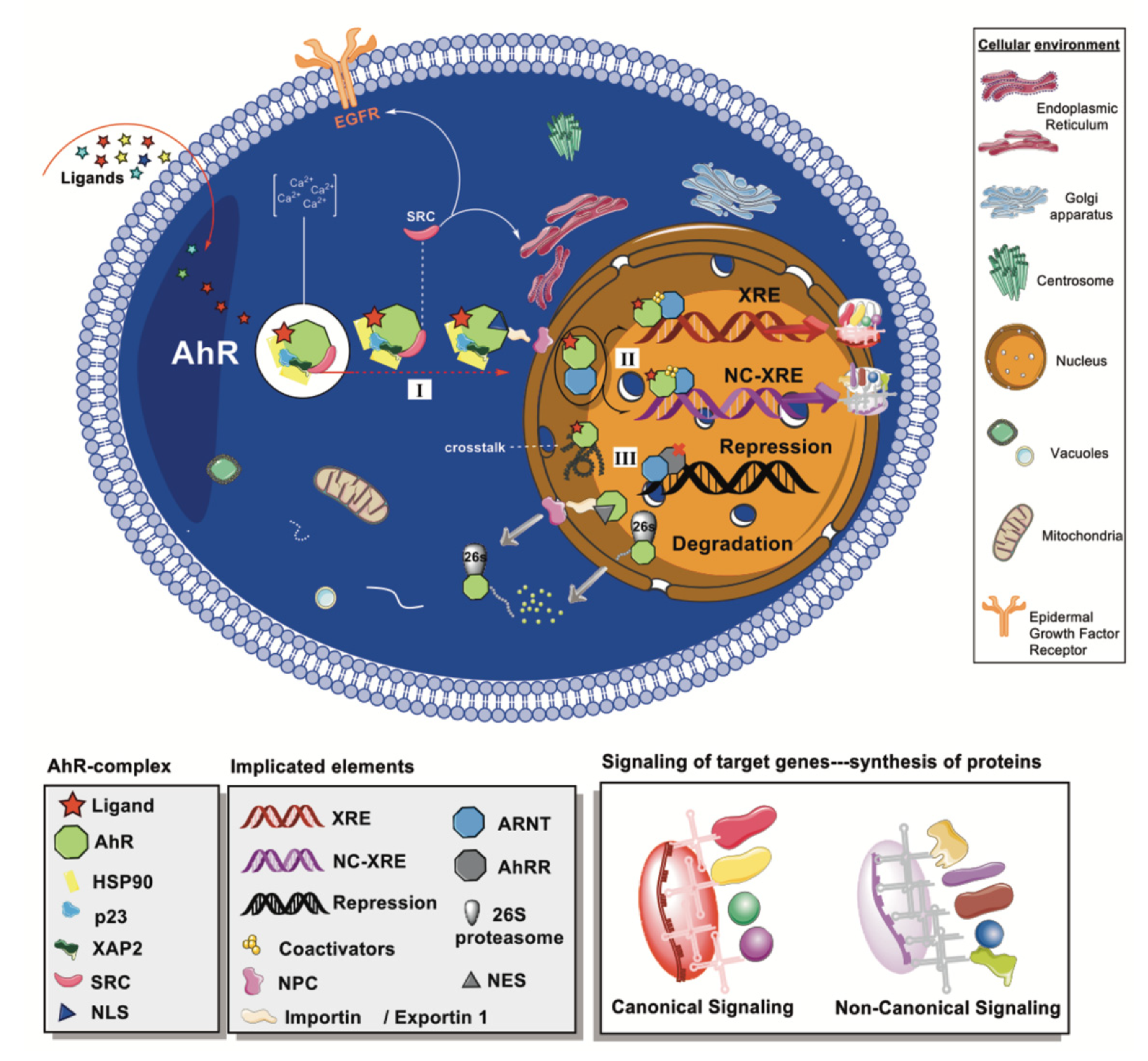
2. Aryl Hydrocarbon Receptor
2.1. Functional Domains
2.2. Cytoplasmic Complex and Signaling Pathways
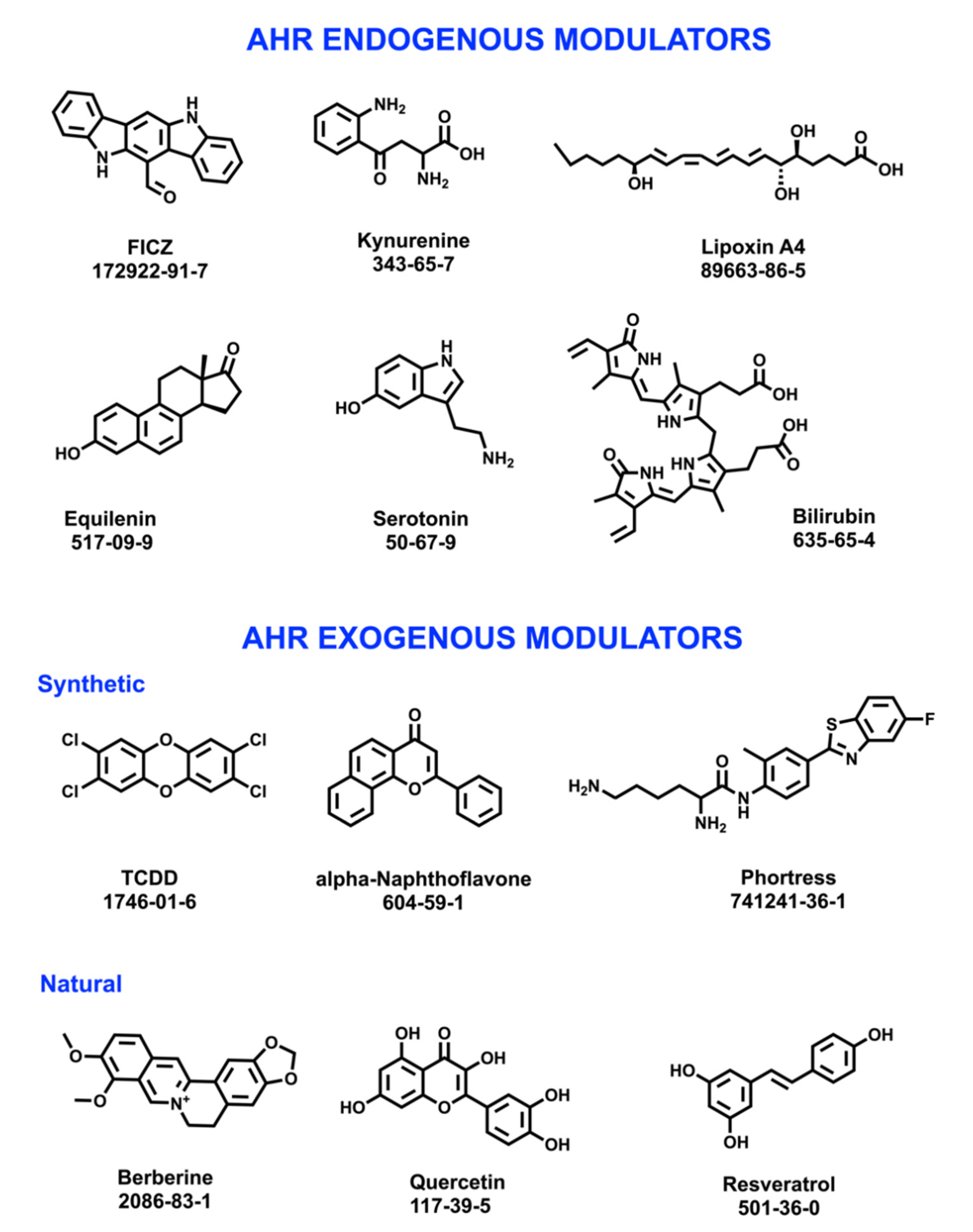
2.3. AhR Effects and Modulators
3. Flavonoids: Generalities and Modulating Effects of AhR
- The modulation of AhR by aglycones and glycosides analogous does not seem to differ significantly, noticeable when compare flavone luteolin vs. luteolin 7,3′-diglucoside (Table 2), that both display blockage effect on AhR transcriptional activation, although luteolin also exhibited agonist effects. In flavonols, the prototypical AhR modulator quercetin is reported as an agonist and as an antagonist of the receptor (Table 3), while its glycoside analogous guaijaverin is described as agonist, quercitrin as antagonist, and rutin has shown both agonist and antagonist effects. The flavanone aglycone hesperetin is reported as AhR antagonist, while exhibited also AhR agonist effects as the glycoside hesperidin. Naringenin differs from naringin in the agonist capacity, reported only for the glycoside derivative (Table 4).
- Comparing the flavone apigenin vs. the isoflavone analogous genistein, the modification from position 2 to position 3 appears to lead to the loss of agonist effects (Table 2 and Table 5). Similarly, a loss of agonism is observed when compared to unsaturated analogous (apigenin and kaempferol) vs. the saturated flavanone naringenin (Table 2, Table 3 and Table 4).
- In flavone derivatives, the 7-methoxy substitution causes a loss of agonist capacity noticeable when compared genkwanin vs. the tri-hydroxy substituted apigenin (Table 2). In flavanones (naringenin vs. sakuranetin) (Table 4) and isoflavones (genistein vs. prunetin) (Table 5), 7-methoxy derivatives and 7-hydroxy derivatives are all reported as non-agonist of AhR. The 7-hydroxy flavanone naringenin and the 7-hydroxy isoflavone genistein have shown AhR antagonism.
- The 4-methoxy substitution does not seem to affect the AhR agonist potential in flavones (acacetin vs. apigenin) (Table 2).
- In isoflavones, 4′-methoxy substitution confers AhR agonist activity (daidzein vs. formononetin and biochanin A vs. genistein) (Table 5).
4. Conclusions
5. Search Methodology
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Scarano, A.; Chieppa, M.; Santino, A. Looking at Flavonoid Biodiversity in Horticultural Crops: A Colored Mine with Nutritional Benefits. Plants 2018, 7, 98. [Google Scholar] [CrossRef] [PubMed]
- Durazzo, A.; Lucarini, M.; Souto, E.B.; Cicala, C.; Caiazzo, E.; Izzo, A.A.; Novellino, E.; Santini, A. Polyphenols: A Concise Overview on the Chemistry, Occurrence, and Human Health. Phytother. Res. 2019, 33, 2221–2243. [Google Scholar] [CrossRef] [PubMed]
- Panche, A.N.; Diwan, A.D.; Chandra, S.R. Flavonoids: An Overview. J. Nutr. Sci. 2016, 5, e47. [Google Scholar] [CrossRef] [PubMed]
- Gonzales, G.B. In Vitro Bioavailability and Cellular Bioactivity Studies of Flavonoids and Flavonoid-Rich Plant Extracts: Questions, Considerations and Future Perspectives. Proc. Nutr. Soc. 2017, 76, 175–181. [Google Scholar] [CrossRef] [PubMed]
- Ginwala, R.; Bhavsar, R.; Chigbu, D.I.; Jain, P.; Khan, Z.K. Potential Role of Flavonoids in Treating Chronic Inflammatory Diseases with a Special Focus on the Anti-Inflammatory Activity of Apigenin. Antioxidants 2019, 8, 35. [Google Scholar] [CrossRef]
- Perez-Vizcaino, F.; Fraga, C.G. Research Trends in Flavonoids and Health. Arch. Biochem. Biophys. 2018, 646, 107–112. [Google Scholar] [CrossRef]
- Procházková, D.; Boušová, I.; Wilhelmová, N. Antioxidant and Prooxidant Properties of Flavonoids. Fitoterapia 2011, 82, 513–523. [Google Scholar] [CrossRef]
- Nebert, D.W. Aryl Hydrocarbon Receptor (AHR): “Pioneer Member” of the Basic-Helix/Loop/Helix per-Arnt-Sim (BHLH/PAS) Family of “Sensors” of Foreign and Endogenous Signals. Prog. Lipid Res. 2017, 67, 38–57. [Google Scholar] [CrossRef]
- Bock, K.W. From TCDD-Mediated Toxicity to Searches of Physiologic AHR Functions. Biochem. Pharmacol. 2018, 155, 419–424. [Google Scholar] [CrossRef]
- Okey, A.B. An Aryl Hydrocarbon Receptor Odyssey to the Shores of Toxicology: The Deichmann Lecture, International Congress of Toxicology-XI. Toxicol. Sci. 2007, 98, 5–38. [Google Scholar] [CrossRef]
- Yang, T.; Feng, Y.; Chen, L.; Vaziri, N.D.; Zhao, Y.; Yang, T.; Feng, Y.; Chen, L.; Vaziri, N.D.; Zhao, Y. Dietary Natural Flavonoids Treating Cancer by Targeting Aryl Hydrocarbon Receptor. Crit. Rev. Toxicol. 2019, 49, 445–460. [Google Scholar] [CrossRef]
- Esser, C.; Lawrence, B.P.; Sherr, D.H.; Perdew, G.H.; Puga, A.; Barouki, R.; Coumoul, X. Old Receptor, New Tricks—The Ever-Expanding Universe of Aryl Hydrocarbon Receptor Functions. Report from the 4th AHR Meeting, 29–31 August 2018 in Paris, France. Int. J. Mol. Sci. 2018, 19, 3603. [Google Scholar] [CrossRef] [PubMed]
- Goya-Jorge, E.; Abdmouleh, F.; Carpio, L.E.; Giner, R.M.; Sylla-Iyarreta Veitía, M. Discovery of 2-Aryl and 2-Pyridinylbenzothiazoles Endowed with Antimicrobial and Aryl Hydrocarbon Receptor Agonistic Activities. Eur. J. Pharm. Sci. 2020, 151, 105386. [Google Scholar] [CrossRef] [PubMed]
- Goya-Jorge, E.; Doan, T.Q.; Scippo, M.L.; Muller, M.; Giner, R.M.; Barigye, S.J.; Gozalbes, R. Elucidating the Aryl Hydrocarbon Receptor Antagonism from a Chemical-Structural Perspective. SAR QSAR Environ. Res. 2020, 31. [Google Scholar] [CrossRef] [PubMed]
- Goya-Jorge, E.; Rampal, C.; Loones, N.; Barigye, S.J.; Carpio, L.E.; Gozalbes, R.; Ferroud, C.; Veitía, M.S.-I.; Giner, R.M. Targeting the Aryl Hydrocarbon Receptor with a Novel Set of Triarylmethanes. Eur. J. Med. Chem. 2020, 207, 112777. [Google Scholar] [CrossRef] [PubMed]
- Amakura, Y.; Tsutsumi, T.; Nakamura, M.; Handa, H.; Yoshimura, M.; Matsuda, R.; Yoshida, T. Aryl Hydrocarbon Receptor Ligand Activity of Commercial Health Foods. Food Chem. 2011, 126, 1515–1520. [Google Scholar] [CrossRef]
- Xue, Z.; Li, D.; Yu, W.; Zhang, Q.; Hou, X.; He, Y.; Kou, X. Mechanisms and Therapeutic Prospects of Polyphenols as Modulators of the Aryl Hydrocarbon Receptor. Food Funct. 2017, 8, 1414–1437. [Google Scholar] [CrossRef] [PubMed]
- Ciolino, H.P.; Daschner, P.J.; Yeh, G.C. Dietary Flavonols Quercetin and Kaempferol Are Ligands of the Aryl Hydrocarbon Receptor That Affect CYP1A1 Transcription Differentially. Biochem. J. 1999, 340, 715–722. [Google Scholar] [CrossRef]
- Safe, S.; Jin, U.H.; Park, H.; Chapkin, R.S.; Jayaraman, A. Aryl Hydrocarbon Receptor (AHR) Ligands as Selective Ahr Modulators (SAHRMS). Int. J. Mol. Sci. 2020, 21, 6654. [Google Scholar] [CrossRef] [PubMed]
- Carver, L.A.; Hogenesch, J.B.; Bradfield, C.A. Tissue Specific Expression of the Rat Ah-Receptor and ARNT MRNAs. Nucleic Acids Res. 1994, 22, 3038–3044. [Google Scholar] [CrossRef]
- Murray, I.A.; Patterson, A.D.; Perdew, G.H. Aryl Hydrocarbon Receptor Ligands in Cancer: Friend and Foe. Nat. Rev. Cancer 2014, 14, 801–814. [Google Scholar] [CrossRef] [PubMed]
- Hogenesch, J.B.; Chan, W.K.; Jackiw, V.H.; Brown, R.C.; Gu, Y.Z.; Pray-Grant, M.; Perdew, G.H.; Bradfield, C.A. Characterization of a Subset of the Basic-Helix-Loop-Helix-PAS Superfamily That Interacts with Components of the Dioxin Signaling Pathway. J. Biol. Chem. 1997, 272, 8581–8593. [Google Scholar] [CrossRef] [PubMed]
- Larigot, L.; Juricek, L.; Dairou, J.; Coumoul, X. AhR Signaling Pathways and Regulatory Functions. Biochim. Open 2018, 7, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Kewley, R.J.; Whitelaw, M.L.; Chapman-Smith, A. The Mammalian Basic Helix-Loop-Helix/PAS Family of Transcriptional Regulators. Int. J. Biochem. Cell Biol. 2004, 36, 189–204. [Google Scholar] [CrossRef]
- Schulte, K.W.; Green, E.; Wilz, A.; Platten, M.; Daumke, O. Structural Basis for Aryl Hydrocarbon Receptor-Mediated Gene Activation. Structure 2017, 25, 1025–1033.e3. [Google Scholar] [CrossRef] [PubMed]
- Kumar, M.B.; Ramadoss, P.; Reen, R.K.; vanden Heuvel, J.P.; Perdew, G.H. The Q-Rich Subdomain of the Human Ah Receptor Transactivation Domain Is Required for Dioxin-Mediated Transcriptional Activity. J. Biol. Chem. 2001, 276, 42302–42310. [Google Scholar] [CrossRef]
- Hankinson, O. Role of Coactivators in Transcriptional Activation by the Aryl Hydrocarbon Receptor. Arch. Biochem. Biophys. 2005, 433, 379–386. [Google Scholar] [CrossRef] [PubMed]
- Pratt, W.B.; Galigniana, M.D.; Harrell, J.M.; DeFranco, D.B. Role of Hsp90 and the Hsp90-Binding Immunophilins in Signalling Protein Movement. Cell. Signal. 2004, 16, 857–872. [Google Scholar] [CrossRef]
- Bell, D.R.; Poland, A. Binding of Aryl Hydrocarbon Receptor (AhR) to AhR-Interacting Protein: The Role of Hsp90. J. Biol. Chem. 2000, 275, 36407–36414. [Google Scholar] [CrossRef] [PubMed]
- Young, J.C.; Moarefi, I.; Ulrich Hartl, F. Hsp90: A Specialized but Essential Protein-Folding Tool. J. Cell Biol. 2001, 154, 267–273. [Google Scholar] [CrossRef]
- Cox, M.B.; Miller, C.A., III. The P23 Co-Chaperone Facilitates Dioxin Receptor Signaling in a Yeast Model System. Toxicol. Lett. 2002, 129, 13–21. [Google Scholar] [CrossRef]
- Shetty, P.V.; Bhagwat, B.Y.; Chan, W.K. P23 Enhances the Formation of the Aryl Hydrocarbon Receptor-DNA Complex. Biochem. Pharmacol. 2003, 65, 941–948. [Google Scholar] [CrossRef]
- Pappas, B.; Yang, Y.; Wang, Y.; Kim, K.; Chung, H.J.; Cheung, M.; Ngo, K.; Shinn, A.; Chan, W.K. P23 Protects the Human Aryl Hydrocarbon Receptor from Degradation via a Heat Shock Protein 90-Independent Mechanism. Biochem. Pharmacol. 2018, 152, 34–44. [Google Scholar] [CrossRef] [PubMed]
- Meyer, B.K.; Perdew, G.H. Characterization of the AhR-Hsp90-XAP2 Core Complex and the Role of the Immunophilin-Related Protein XAP2 in AhR Stabilization. Biochemistry 1999, 38, 8907–8917. [Google Scholar] [CrossRef] [PubMed]
- Carver, L.A.; Lapres, J.J.; Jain, S.; Dunham, E.E.; Bradfield, C.A. Characterization of the Ah Receptor-Associated Protein, ARA9. J. Biol. Chem. 1998, 273, 33580–33587. [Google Scholar] [CrossRef] [PubMed]
- Meyer, B.K.; Petrulis, J.R.; Perdew, G.H. Aryl Hydrocarbon (Ah) Receptor Levels Are Selectively Modulated by Hsp90-Associated Immunophilin Homolog XAP2. Cell Stress Chaperones 2000, 5, 243–254. [Google Scholar] [CrossRef]
- Dong, B.; Cheng, W.; Li, W.; Zheng, J.; Wu, D.; Matsumura, F.; Vogel, C.F.A. FRET Analysis of Protein Tyrosine Kinase C-Src Activation Mediated via Aryl Hydrocarbon Receptor. Biochim. Et Biophys. Acta (BBA) Gen. Subj. 2011, 1810, 427–431. [Google Scholar] [CrossRef]
- Ikuta, T.; Eguchi, H.; Tachibana, T.; Yoneda, Y.; Kawajiri, K. Nuclear Localization and Export Signals of the Human Aryl Hydrocarbon Receptor. J. Biol. Chem. 1998, 273, 2895–2904. [Google Scholar] [CrossRef]
- Ikuta, T.; Kobayashi, Y.; Kawajiri, K. Phosphorylation of Nuclear Localization Signal Inhibits the Ligand-Dependent Nuclear Import of Aryl Hydrocarbon Receptor. Biochem. Biophys. Res. Commun. 2004, 317, 545–550. [Google Scholar] [CrossRef]
- Enan, E.; Matsumura, F. Identification of C-Src as the Integral Component of the Cytosolic Ah Receptor Complex, Transducing the Signal of 2,3,7,8-Tetrachlorodibenzo-p-Dioxin (TCDD) through the Protein Phosphorylation Pathway. Biochem. Pharmacol. 1996, 52, 1599–1612. [Google Scholar] [CrossRef]
- Petrulis, J.R.; Kusnadi, A.; Ramadoss, P.; Hollingshead, B.; Perdew, G.H. The Hsp90 Co-Chaperone XAP2 Alters Importin β Recognition of the Bipartite Nuclear Localization Signal of the Ah Receptor and Represses Transcriptional Activity. J. Biol. Chem. 2003, 278, 2677–2685. [Google Scholar] [CrossRef]
- Hoffman, E.C.; Reyes, H.; Chu, F.F.; Sander, F.; Conley, L.H.; Brooks, B.A.; Hankinson, O. Cloning of a Factor Required for Activity of the Ah (Dioxin) Receptor. Science 1991, 252, 954–958. [Google Scholar] [CrossRef]
- Swanson, H.I.; Tullis, K.; Denison, M.S. Binding of Transformed Ah Receptor Complex to a Dioxin Responsive Transcriptional Enhancer: Evidence for Two Distinct Heteromeric DNA-Binding Forms. Biochemistry 1993, 32, 12841–12849. [Google Scholar] [CrossRef]
- Wright, E.J.; de Castro, K.P.; Joshi, A.D.; Elferink, C.J. Canonical and Non-Canonical Aryl Hydrocarbon Receptor Signaling Pathways Toxicology. Curr. Opin. Toxicol. 2017, 2, 87–92. [Google Scholar] [CrossRef] [PubMed]
- Yao, E.F.; Denison, M.S. DNA Sequence Determinants for Binding of Transformed Ah Receptor to a Dioxin-Responsive Enhancer. Biochemistry 1992, 31, 5060–5067. [Google Scholar] [CrossRef]
- Sakurai, S.; Shimizu, T.; Ohto, U. The Crystal Structure of the AhRR-ARNT Heterodimer Reveals the Structural Basis of the Repression of AhR-Mediated Transcription. J. Biol. Chem. 2017, 292, 17609–17616. [Google Scholar] [CrossRef] [PubMed]
- Hahn, M.E.; Allan, L.L.; Sherr, D.H. Regulation of Constitutive and Inducible AHR Signaling: Complex Interactions Involving the AHR Repressor. Biochem. Pharmacol. 2009, 77, 485–497. [Google Scholar] [CrossRef]
- Vogel, C.F.A.; Ishihara, Y.; Campbell, C.E.; Kado, S.Y.; Nguyen-Chi, A.; Sweeney, C.; Pollet, M.; Stemmann, T.H.; Tuscano, J.M. A Protective Role of Aryl Hydrocarbon Receptor Repressor in Inflammation and Tumor Growth. Cancers 2019, 11, 589. [Google Scholar] [CrossRef]
- Mimura, J.; Ema, M.; Sogawa, K.; Fujii-Kuriyama, Y. Identification of a Novel Mechanism of Regulation of Ah (Dioxin) Receptor Function. Genes Dev. 1999, 13, 20–25. [Google Scholar] [CrossRef]
- Kress, S.; Reichert, J.; Schwarz, M. Functional Analysis of the Human Cytochrome P4501A1 (CYP1A1) Gene Enhancer. Eur. J. Biochem. 1998, 258, 803–812. [Google Scholar] [CrossRef] [PubMed]
- Quattrochi, L.C.; Tukey, R.H. Nuclear Uptake of the Ah (Dioxin) Receptor in Response to Omeprazole: Transcriptional Activation of the Human CYP1A1 Gene. Mol. Pharmacol. 1993, 43, 504–508. [Google Scholar]
- Ye, W.; Chen, R.; Chen, X.; Huang, B.; Lin, R.; Xie, X.; Chen, J.; Jiang, J.; Deng, Y.; Wen, J. AhR Regulates the Expression of Human Cytochrome P450 1A1 (CYP1A1) by Recruiting Sp1. FEBS J. 2019, 286, 4215–4231. [Google Scholar] [CrossRef] [PubMed]
- Auyeung, D.J.; Kessler, F.K.; Ritter, J.K. Mechanism of Rat UDP-Glucuronosyltransferase 1A6 Induction by Oltipraz: Evidence for a Contribution of the AryL Hydrocarbon Receptor Pathway. Mol. Pharmacol. 2003, 63, 119–127. [Google Scholar] [CrossRef]
- Hankinson, O. The Role of AHR-Inducible Cytochrome P450s in Metabolism of Polyunsaturated Fatty Acids. Drug Metab. Rev. 2016, 48, 342–350. [Google Scholar] [CrossRef] [PubMed]
- Denison, M.S.; Soshilov, A.A.; He, G.; Degroot, D.E.; Zhao, B. Exactly the Same but Different: Promiscuity and Diversity in the Molecular Mechanisms of Action of the Aryl Hydrocarbon (Dioxin) Receptor. Toxicol. Sci. 2011, 124, 1–22. [Google Scholar] [CrossRef]
- Wilson, S.R.; Joshi, A.D.; Elferink, C.J. The Tumor Suppressor Kruppel-like Factor 6 Is a Novel Aryl Hydrocarbon Receptor DNA Binding Partner. J. Pharmacol. Exp. Ther. 2013, 345, 419–429. [Google Scholar] [CrossRef]
- Biscardi, J.S.; Maa, M.C.; Tice, D.A.; Cox, M.E.; Leu, T.H.; Parsons, S.J. C-Src-Mediated Phosphorylation of the Epidermal Growth Factor Receptor on Tyr845 and Tyr1101 Is Associated with Modulation of Receptor Function. J. Biol. Chem. 1999, 274, 8335–8343. [Google Scholar] [CrossRef] [PubMed]
- Haarmann-Stemmann, T.; Bothe, H.; Abel, J. Growth Factors, Cytokines and Their Receptors as Downstream Targets of Arylhydrocarbon Receptor (AhR) Signaling Pathways. Biochem. Pharmacol. 2009, 77, 508–520. [Google Scholar] [CrossRef]
- Yu, A.R.; Jeong, Y.J.; Hwang, C.Y.; Yoon, K.S.; Choe, W.; Ha, J.; Kim, S.S.; Pak, Y.K.; Yeo, E.J.; Kang, I. Alpha-Naphthoflavone Induces Apoptosis through Endoplasmic Reticulum Stress via c-Src-, ROS-, MAPKs-, and Arylhydrocarbon Receptor-Dependent Pathways in HT22 Hippocampal Neuronal Cells. Neurotoxicology 2019, 71, 39–51. [Google Scholar] [CrossRef] [PubMed]
- Domínguez-Acosta, O.; Vega, L.; Estrada-Muñiz, E.; Rodríguez, M.S.; Gonzalez, F.J.; Elizondo, G. Activation of Aryl Hydrocarbon Receptor Regulates the LPS/IFN$γ$-Induced Inflammatory Response by Inducing Ubiquitin-Proteosomal and Lysosomal Degradation of RelA/P65. Biochem. Pharmacol. 2018, 155, 141–149. [Google Scholar] [CrossRef]
- Mejía-García, A.; González-Barbosa, E.; Martínez-Guzmán, C.; Torres-Ramos, M.A.; Rodríguez, M.S.; Guzmán-León, S.; Elizondo, G. Activation of AHR Mediates the Ubiquitination and Proteasome Degradation of C-Fos through the Induction of Ubcm4 Gene Expression. Toxicology 2015, 337, 47–57. [Google Scholar] [CrossRef] [PubMed]
- Ohtake, F.; Fujii-Kuriyama, Y.; Kato, S. AhR Acts as an E3 Ubiquitin Ligase to Modulate Steroid Receptor Functions. Biochem. Pharmacol. 2009, 77, 474–484. [Google Scholar] [CrossRef] [PubMed]
- Ohtake, F.; Baba, A.; Takada, I.; Okada, M.; Iwasaki, K.; Miki, H.; Takahashi, S.; Kouzmenko, A.; Nohara, K.; Chiba, T.; et al. Dioxin Receptor Is a Ligand-Dependent E3 Ubiquitin Ligase. Nature 2007, 446, 562–566. [Google Scholar] [CrossRef] [PubMed]
- Matsumura, F. The Significance of the Nongenomic Pathway in Mediating Inflammatory Signaling of the Dioxin-Activated Ah Receptor to Cause Toxic Effects. Biochem. Pharmacol. 2009, 77, 608–626. [Google Scholar] [CrossRef]
- Puga, A.; Ma, C.; Marlowe, J.L. The Aryl Hydrocarbon Receptor Cross-Talks with Multiple Signal Transduction Pathways. Biochem. Pharmacol. 2009, 77, 713–722. [Google Scholar] [CrossRef]
- Roman, Á.C.; Carvajal-Gonzalez, J.M.; Merino, J.M.; Mulero-Navarro, S.; Fernández-Salguero, P.M. The Aryl Hydrocarbon Receptor in the Crossroad of Signalling Networks with Therapeutic Value. Pharmacol. Ther. 2018, 185, 50–63. [Google Scholar] [CrossRef]
- Ohtake, F.; Takeyama, K.-I.; Matsumoto, T.; Kitagawa, H.; Yamamoto, Y.; Nohara, K.; Tohyama, C.; Krust, A.; Mimura, J.; Chambon, P.; et al. Modulation of Oestrogen Receptor Signalling by Association with the Activated Dioxin Receptor. Nature 2003, 423, 545–550. [Google Scholar] [CrossRef]
- Mathew, L.K.; Sengupta, S.S.; LaDu, J.; Andreasen, E.A.; Tanguay, R.L. Crosstalk between AHR and Wnt Signaling through R-Spondin1 Impairs Tissue Regeneration in Zebrafish. FASEB J. 2008, 22, 3087–3096. [Google Scholar] [CrossRef]
- Chassot, A.A.; Bradford, S.T.; Auguste, A.; Gregoire, E.P.; Pailhoux, E.; de Rooij, D.G.; Schedl, A.; Chaboissier, M.C. WNT4 and RSPO1 Together Are Required for Cell Proliferation in the Early Mouse Gonad. Development 2012, 139, 4461–4472. [Google Scholar] [CrossRef]
- Tomaselli, S.; Megiorni, F.; Lin, L.; Mazzilli, M.C.; Gerrelli, D.; Majore, S.; Grammatico, P.; Achermann, J.C. Human RSPO1/R-Spondin1 Is Expressed during Early Ovary Development and Augments β-Catenin Signaling. PLoS ONE 2011, 6, e16366. [Google Scholar] [CrossRef]
- Komiya, Y.; Habas, R. Wnt Signal Transduction Pathways. Organogenesis 2008, 4, 68–75. [Google Scholar] [CrossRef]
- Neuzillet, C.; Tijeras-Raballand, A.; Cohen, R.; Cros, J.; Faivre, S.; Raymond, E.; De Gramont, A. Targeting the TGFβ Pathway for Cancer Therapy. Pharmacol. Ther. 2015, 147, 22–31. [Google Scholar] [CrossRef] [PubMed]
- Ishihara, Y.; Kado, S.Y.; Hoeper, C.; Harel, S.; Vogel, C.F.A. Role of NF-KB RelB in Aryl Hydrocarbon Receptor-Mediated Ligand Specific Effects. Int. J. Mol. Sci. 2019, 20, 2652. [Google Scholar] [CrossRef]
- Vogel, C.F.A.; Sciullo, E.; Li, W.; Wong, P.; Lazennec, G.; Matsumura, F. RelB, a New Partner of Aryl Hydrocarbon Receptor-Mediated Transcription. Mol. Endocrinol. 2007, 21, 2941–2955. [Google Scholar] [CrossRef] [PubMed]
- Pollenz, R.S. The Mechanism of AH Receptor Protein Down-Regulation (Degradation) and Its Impact on AH Receptor-Mediated Gene Regulation. Chem. Biol. Interact. 2002, 141, 41–61. [Google Scholar] [CrossRef]
- Pollenz, R.S.; Barbour, E.R. Analysis of the Complex Relationship between Nuclear Export and Aryl Hydrocarbon Receptor-Mediated Gene Regulation. Mol. Cell. Biol. 2000, 20, 6095–6104. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Yang, Y.; Russu, W.A.; Chan, W.K. The Aryl Hydrocarbon Receptor Undergoes Chaperone-Mediated Autophagy in Triple-Negative Breast Cancer Cells. Int. J. Mol. Sci. 2021, 22, 1654. [Google Scholar] [CrossRef] [PubMed]
- Rothhammer, V.; Quintana, F.J. The Aryl Hydrocarbon Receptor: An Environmental Sensor Integrating Immune Responses in Health and Disease. Nat. Rev. Immunol. 2019, 19, 184–197. [Google Scholar] [CrossRef]
- Lamas, B.; Natividad, J.M.; Sokol, H. Aryl Hydrocarbon Receptor and Intestinal Immunity. Mucosal Immunol. 2018, 11, 1024–1038. [Google Scholar] [CrossRef]
- Gao, J.; Xu, K.; Liu, H.; Liu, G.; Bai, M.; Peng, C.; Li, T.; Yin, Y. Impact of the Gut Microbiota on Intestinal Immunity Mediated by Tryptophan Metabolism. Front. Cell. Infect. Microbiol. 2018, 8, 1–22. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, K.A.; Elferink, C.J. Timing Is Everything: Consequences of Transient and Sustained AhR Activity. Biochem. Pharmacol. 2009, 77, 947–956. [Google Scholar] [CrossRef]
- Mescher, M.; Haarmann-Stemmann, T. Modulation of CYP1A1 Metabolism: From Adverse Health Effects to Chemoprevention and Therapeutic Options. Pharmacol. Ther. 2018, 187, 71–87. [Google Scholar] [CrossRef]
- Guerrina, N.; Traboulsi, H.; Eidelman, D.H.; Baglole, C.J. The Aryl Hydrocarbon Receptor and the Maintenance of Lung Health. Int. J. Mol. Sci. 2018, 19, 3882. [Google Scholar] [CrossRef]
- Bock, K.W. Human AHR Functions in Vascular Tissue: Pro- and Anti-Inflammatory Responses of AHR Agonists in Atherosclerosis. Biochem. Pharmacol. 2019, 159, 116–120. [Google Scholar] [CrossRef]
- Duval, C.; Blanc, E.; Coumoul, X. Aryl Hydrocarbon Receptor and Liver Fibrosis. Curr. Opin. Toxicol. 2018, 8, 8–13. [Google Scholar] [CrossRef]
- Puccetti, M.; Paolicelli, G.; Oikonomou, V.; de Luca, A.; Renga, G.; Borghi, M.; Pariano, M.; Stincardini, C.; Scaringi, L.; Giovagnoli, S.; et al. Towards Targeting the Aryl Hydrocarbon Receptor in Cystic Fibrosis. Mediat. Inflamm. 2018, 2018, 1601486. [Google Scholar] [CrossRef]
- Dietrich, C. Antioxidant Functions of the Aryl Hydrocarbon Receptor. Stem Cells Int. 2016, 2016. [Google Scholar] [CrossRef] [PubMed]
- Juricek, L.; Coumoul, X. The Aryl Hydrocarbon Receptor and the Nervous System. Int. J. Mol. Sci. 2018, 19, 2504. [Google Scholar] [CrossRef]
- Stejskalova, L.; Dvorak, Z.; Pavek, P. Endogenous and Exogenous Ligands of Aryl Hydrocarbon Receptor: Current State of Art. Curr. Drug Metab. 2011, 12, 198–212. [Google Scholar] [CrossRef] [PubMed]
- Rannug, A.; Rannug, U.; Rosenkranz, H.S.; Winqvist, L.; Westerholm, R.; Agurell, E.; Grafström, A.K. Certain Photooxidized Derivatives of Tryptophan Bind with Very High Affinity to the Ah Receptor and Are Likely to Be Endogenous Signal Substances. J. Biol. Chem. 1987, 262, 15422–15427. [Google Scholar] [CrossRef]
- Manzella, C.; Singhal, M.; Alrefai, W.A.; Saksena, S.; Dudeja, P.K.; Gill, R.K. Serotonin Is an Endogenous Regulator of Intestinal CYP1A1 via AhR. Sci. Rep. 2018, 8, 6103. [Google Scholar] [CrossRef]
- Seok, S.-H.; Lee, W.; Jiang, L.; Molugu, K.; Zheng, A.; Li, Y.; Park, S.; Bradfield, C.A.; Xing, Y. Structural Hierarchy Controlling Dimerization and Target DNA Recognition in the AHR Transcriptional Complex. Proc. Natl. Acad. Sci. USA 2017, 114, 5431–5436. [Google Scholar] [CrossRef]
- Zhou, H.; Wu, H.; Liao, C.; Diao, X.; Zhen, J.; Chen, L.; Xue, Q. Toxicology Mechanism of the Persistent Organic Pollutants (POPs) in Fish through AhR Pathway. Toxicol. Mech. Methods 2010, 20, 279–286. [Google Scholar] [CrossRef]
- Esser, C.; Rannug, A.; Stockinger, B. The Aryl Hydrocarbon Receptor in Immunity. Trends Immunol. 2009, 30, 447–454. [Google Scholar] [CrossRef] [PubMed]
- Marafini, I.; Di Fusco, D.; Dinallo, V.; Franzè, E.; Stolfi, C.; Sica, G.; Monteleone, G.; Monteleone, I. NPD-0414-2 and NPD-0414-24, Two Chemical Entities Designed as Aryl Hydrocarbon Receptor (AHR) Ligands, Inhibit Gut Inflammatory Signals. Front. Pharmacol. 2019, 10, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Bradshaw, T.; Westwell, A. The Development of the Antitumour Benzothiazole Prodrug, Phortress, as a Clinical Candidate. Curr. Med. Chem. 2005, 11, 1009–1021. [Google Scholar] [CrossRef]
- Loaiza-Pérez, A.I.; Kenney, S.; Boswell, J.; Hollingshead, M.; Alley, M.C.; Hose, C.; Ciolino, H.P.; Yeh, G.C.; Trepel, J.B.; Vistica, D.T.; et al. Aryl Hydrocarbon Receptor Activation of an Antitumor Aminoflavone: Basis of Selective Toxicity for MCF-7 Breast Tumor Cells. Mol. Cancer Ther. 2004, 3, 715–725. [Google Scholar]
- Mozaffarian, D.; Wu, J.H.Y. Flavonoids, Dairy Foods, and Cardiovascular and Metabolic Health: A Review of Emerging Biologic Pathways. Circ. Res. 2018, 122, 369–384. [Google Scholar] [CrossRef]
- Scarmeas, N.; Anastasiou, C.A.; Yannakoulia, M. Nutrition and Prevention of Cognitive Impairment. Lancet Neurol. 2018, 17, 1006–1015. [Google Scholar] [CrossRef]
- Agati, G.; Tattini, M. Multiple Functional Roles of Flavonoids in Photoprotection. New Phytol. 2010, 186, 786–793. [Google Scholar] [CrossRef] [PubMed]
- Lattanzio, V. Phenolic Compounds: Introduction. In Natural Products: Phytochemistry, Botany and Metabolism of Alkaloids, Phenolics and Terpenes; Ramawat, K.G., Mérillon, J.-M., Eds.; Springer: Berlin/Heidelberg, Germany, 2013; pp. 1543–1580. ISBN 978-3-642-22144-6. [Google Scholar]
- Abbas, M.; Saeed, F.; Anjum, F.M.; Afzaal, M.; Tufail, T.; Bashir, M.S.; Ishtiaq, A.; Hussain, S.; Suleria, H.A.R. Natural Polyphenols: An Overview. Int. J. Food Prop. 2017, 20, 1689–1699. [Google Scholar] [CrossRef]
- Pandey, R.P.; Sohng, J.K. Genetics of Flavonoids. In Natural Products: Phytochemistry, Botany and Metabolism of Alkaloids, Phenolics and Terpenes; Ramawat, K.G., Mérillon, J.-M., Eds.; Springer: Berlin/Heidelberg, Germany, 2013; pp. 1617–1645. ISBN 978-3-642-22144-6. [Google Scholar]
- Abotaleb, M.; Samuel, S.M.; Varghese, E.; Varghese, S.; Kubatka, P.; Liskova, A.; Büsselberg, D. Flavonoids in Cancer and Apoptosis. Cancers 2019, 11, 28. [Google Scholar] [CrossRef] [PubMed]
- Forkmann, G.; Heller, W. 1.26 Biosynthesis of Flavonoids. In Comprehensive Natural Products Chemistry; Barton, S.D., Nakanishi, K., Meth-Cohn, O., Eds.; Pergamon: Oxford, UK, 1999; pp. 713–748. ISBN 978-0-08-091283-7. [Google Scholar]
- Teng, H.; Chen, L. Polyphenols and Bioavailability: An Update. Crit. Rev. Food Sci. Nutr. 2019, 59, 2040–2051. [Google Scholar] [CrossRef] [PubMed]
- Cermak, R.; Durazzo, A.; Maiani, G.; Böhm, V.; Kammerer, D.R.; Carle, R.; Wiczkowski, W.; Piskula, M.K.; Galensa, R. The Influence of Postharvest Processing and Storage of Foodstuffs on the Bioavailability of Flavonoids and Phenolic Acids. Mol. Nutr. Food Res. 2009, 53, S184–S193. [Google Scholar] [CrossRef] [PubMed]
- Tungmunnithum, D.; Thongboonyou, A.; Pholboon, A.; Yangsabai, A. Flavonoids and Other Phenolic Compounds from Medicinal Plants for Pharmaceutical and Medical Aspects: An Overview. Medicines 2018, 5, 93. [Google Scholar] [CrossRef]
- Quideau, S.; Deffieux, D.; Douat-Casassus, C.; Pouységu, L. Plant Polyphenols: Chemical Properties, Biological Activities, and Synthesis. Angew. Chem. Int. Ed. 2011, 50, 586–621. [Google Scholar] [CrossRef]
- Xie, Y.; Huang, S.; Su, Y. Dietary Flavonols Intake and Risk of Esophageal and Gastric Cancer: A Meta-Analysis of Epidemiological Studies. Nutrients 2016, 8, 91. [Google Scholar] [CrossRef]
- Romagnolo, D.F.; Selmin, O.I. Flavonoids and Cancer Prevention: A Review of the Evidence. J. Nutr. Gerontol. Geriatr. 2012, 31, 206–238. [Google Scholar] [CrossRef]
- Martinez-Perez, C.; Ward, C.; Cook, G.; Mullen, P.; McPhail, D.; Harrison, D.J.; Langdon, S.P. Novel Flavonoids as Anti-Cancer Agents: Mechanisms of Action and Promise for Their Potential Application in Breast Cancer. Biochem. Soc. Trans. 2014, 42, 1017–1023. [Google Scholar] [CrossRef]
- Hooper, L.; Kay, C.; Abdelhamid, A.; Kroon, P.A.; Cohn, J.S.; Rimm, E.B.; Cassidy, A. Effects of Chocolate, Cocoa, and Flavan-3-Ols on Cardiovascular Health: A Systematic Review and Meta-Analysis of Randomized Trials. Am. J. Clin. Nutr. 2012, 95, 740–751. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Ouyang, Y.Y.; Liu, J.; Zhao, G. Flavonoid Intake and Risk of CVD: A Systematic Review and Meta-Analysis of Prospective Cohort Studies. Br. J. Nutr. 2014, 111, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Rees, A.; Dodd, G.F.; Spencer, J.P.E. The Effects of Flavonoids on Cardiovascular Health: A Review of Human Intervention Trials and Implications for Cerebrovascular Function. Nutrients 2018, 10, 1852. [Google Scholar] [CrossRef] [PubMed]
- Cheng, J.; Zhou, L.; Liu, Q.; Tickner, J.; Tan, Z.; Li, X.; Liu, M.; Lin, X.; Wang, T.; Pavlos, N.J.; et al. Cyanidin Chloride Inhibits Ovariectomy-Induced Osteoporosis by Suppressing RANKL-Mediated Osteoclastogenesis and Associated Signaling Pathways. J. Cell. Physiol. 2018, 233, 2502–2512. [Google Scholar] [CrossRef]
- Beking, K.; Vieira, A. Flavonoid Intake and Disability-Adjusted Life Years Due to Alzheimer’s and Related Dementias: A Population-Based Study Involving Twenty-Three Developed Countries. Public Health Nutr. 2010, 13, 1403–1409. [Google Scholar] [CrossRef]
- Ninfali, P.; Antonini, E.; Frati, A.; Scarpa, E.-S. C-Glycosyl Flavonoids from Beta Vulgaris Cicla and Betalains from Beta Vulgaris Rubra: Antioxidant, Anticancer and Antiinflammatory Activities—A Review. Phytother. Res. 2017, 31, 871–884. [Google Scholar] [CrossRef]
- Farzaei, M.H.; Singh, A.K.; Kumar, R.; Croley, C.R.; Pandey, A.K.; Coy-Barrera, E.; Patra, J.K.; Das, G.; Kerry, R.G.; Annunziata, G.; et al. Targeting Inflammation by Flavonoids: Novel Therapeutic Strategy for Metabolic Disorders. Int. J. Mol. Sci. 2019, 20, 4957. [Google Scholar] [CrossRef] [PubMed]
- Farhat, G.; Drummond, S.; Al-Dujaili, E.A.S. Polyphenols and Their Role in Obesity Management: A Systematic Review of Randomized Clinical Trials. Phytother. Res. 2017, 31, 1005–1018. [Google Scholar] [CrossRef] [PubMed]
- Al-Dosary, D.I.; Alhomida, A.S.; Ola, M.S. Protective Effects of Dietary Flavonoids in Diabetic Induced Retinal Neurodegeneration. Curr. Drug Targets 2017, 18, 1468–1476. [Google Scholar] [CrossRef]
- Belwal, T.; Nabavi, S.F.; Nabavi, S.M.; Habtemariam, S. Dietary Anthocyanins and Insulin Resistance: When Food Becomes a Medicine. Nutrients 2017, 9, 1111. [Google Scholar] [CrossRef]
- Chirumbolo, S.; Bjørklund, G.; Lysiuk, R.; Vella, A.; Lenchyk, L.; Upyr, T. Targeting Cancer with Phytochemicals via Their Fine Tuning of the Cell Survival Signaling Pathways. Int. J. Mol. Sci. 2018, 19, 3568. [Google Scholar] [CrossRef]
- Dong, J.; Zhang, Q.; Cui, Q.; Huang, G.; Pan, X.; Li, S. Flavonoids and Naphthoflavonoids: Wider Roles in the Modulation of Cytochrome P450 Family 1 Enzymes. ChemMedChem 2016, 2102–2118. [Google Scholar] [CrossRef]
- van der Heiden, E.; Bechoux, N.; Muller, M.; Sergent, T.; Schneider, Y.J.; Larondelle, Y.; Maghuin-Rogister, G.; Scippo, M.L. Food Flavonoid Aryl Hydrocarbon Receptor-Mediated Agonistic/Antagonistic/Synergic Activities in Human and Rat Reporter Gene Assays. Anal. Chim. Acta 2009, 637, 337–345. [Google Scholar] [CrossRef] [PubMed]
- Doan, T.Q.; Connolly, L.; Igout, A.; Muller, M.; Scippo, M.L. In Vitro Differential Responses of Rat and Human Aryl Hydrocarbon Receptor to Two Distinct Ligands and to Different Polyphenols. Environ. Pollut. 2020, 265, 114966. [Google Scholar] [CrossRef]
- Zhang, S.; Qin, C.; Safe, S.H. Flavonoids as Aryl Hydrocarbon Receptor Agonists/Antagonists: Effects of Structure and Cell Context. Environ. Health Perspect. 2003, 111, 1877–1882. [Google Scholar] [CrossRef]
- Ashida, H.; Fukuda, I.; Yamashita, T.; Kanazawa, K. Flavones and Flavonols at Dietary Levels Inhibit a Transformation of Aryl Hydrocarbon Receptor Induced by Dioxin. FEBS Lett. 2000, 476, 213–217. [Google Scholar] [CrossRef]
- Ashida, H. Suppressive Effects of Flavonoids on Dioxin Toxicity. BioFactors 2000, 12, 201–206. [Google Scholar] [CrossRef]
- Xue, Y.; Shui, X.; Su, W.; He, Y.; Lu, X.; Zhang, Y.; Yan, G.; Huang, S.; Lei, W.; Chen, C. Baicalin Inhibits Inflammation and Attenuates Myocardial Ischaemic Injury by Aryl Hydrocarbon Receptor. J. Pharm. Pharmacol. 2015, 67, 1756–1764. [Google Scholar] [CrossRef] [PubMed]
- Zhu, W.; Chen, X.; Yu, J.; Xiao, Y.; Li, Y.; Wan, S.; Su, W.; Liang, D. Baicalin Modulates the Treg/Teff Balance to Alleviate Uveitis by Activating the Aryl Hydrocarbon Receptor. Biochem. Pharmacol. 2018, 154, 18–27. [Google Scholar] [CrossRef]
- Arivazhagan, L.; Subramanian, S.P. Tangeretin, a Citrus Flavonoid Attenuates Oxidative Stress and Protects Hepatocellular Architecture in Rats with 7, 12 Dimethylbenz(a)Anthracene Induced Experimental Mammary Carcinoma. J. Funct. Foods 2015, 15, 339–353. [Google Scholar] [CrossRef]
- Ronnekleiv-Kelly, S.M.; Nukaya, M.; Díaz-Díaz, C.J.; Megna, B.W.; Carney, P.R.; Geiger, P.G.; Kennedy, G.D. Aryl Hydrocarbon Receptor-Dependent Apoptotic Cell Death Induced by the Flavonoid Chrysin in Human Colorectal Cancer Cells. Cancer Lett. 2016, 370, 91–99. [Google Scholar] [CrossRef]
- Feng, J.; Zheng, T.; Hou, Z.; Lv, C.; Xue, A.; Han, T.; Han, B.; Sun, X.; Wei, Y. Luteolin, an Aryl Hydrocarbon Receptor Ligand, Suppresses Tumor Metastasis in Vitro and in Vivo. Oncol Rep 2020, 44, 2231–2240. [Google Scholar] [CrossRef]
- Jin, U.H.; Park, H.; Li, X.; Davidson, L.A.; Allred, C.; Patil, B.; Jayaprakasha, G.; Orr, A.A.; Mao, L.; Chapkin, R.S.; et al. Structure-Dependent Modulation of Aryl Hydrocarbon Receptor-Mediated Activities by Flavonoids. Toxicol. Sci. 2018, 164, 205–217. [Google Scholar] [CrossRef]
- Goya-Jorge, E.; Giner, R.M.; Sylla-Iyarreta Veitía, M.; Gozalbes, R.; Barigye, S.J. Predictive Modeling of Aryl Hydrocarbon Receptor (AhR) Agonism. Chemosphere 2020, 127068. [Google Scholar] [CrossRef]
- Park, H.; Jin, U.H.; Orr, A.A.; Echegaray, S.P.; Davidson, L.A.; Allred, C.D.; Chapkin, R.S.; Jayaraman, A.; Lee, K.; Tamamis, P.; et al. Isoflavones as Ah Receptor Agonists in Colon-Derived Cell Lines: Structure-Activity Relationships. Chem. Res. Toxicol. 2019, 32, 2353–2364. [Google Scholar] [CrossRef] [PubMed]
- Sun, F.; Indran, I.R.; Zhang, Z.W.; Tan, M.H.E.; Li, Y.; Lim, Z.L.R.; Hua, R.; Yang, C.; Soon, F.F.; Li, J.; et al. A Novel Prostate Cancer Therapeutic Strategy Using Icaritin-Activated Arylhydrocarbon-Receptor to Co-Target Androgen Receptor and Its Splice Variants. Carcinogenesis 2015, 36, 757–768. [Google Scholar] [CrossRef] [PubMed]
- Kaur, M.; Badhan, R.K.S. Phytochemical Mediated-Modulation of the Expression and Transporter Function of Breast Cancer Resistance Protein at the Blood-Brain Barrier: An in-Vitro Study. Brain Res. 2017, 1654, 9–23. [Google Scholar] [CrossRef] [PubMed]
- Bostikova, Z.; Moserova, M.; Pavek, P.; Stiborova, M.; Hodek, P. Role of Dihydromyricetin in Cytochrome P450-Mediated Metabolism and Carcinogen Activation. Neuroendocrinol. Lett. 2015, 36, 46–52. [Google Scholar] [PubMed]
- Wang, S.; Dunlap, T.L.; Howell, C.E.; Mbachu, O.C.; Rue, E.A.; Phansalkar, R.; Chen, S.N.; Pauli, G.F.; Dietz, B.M.; Bolton, J.L. Hop (Humulus Lupulus L.) Extract and 6-Prenylnaringenin Induce P450 1A1 Catalyzed Estrogen 2-Hydroxylation. Chem. Res. Toxicol. 2016, 29, 1142–1150. [Google Scholar] [CrossRef] [PubMed]
- Tan, Y.Q.; Chiu-Leung, L.C.; Mei Lin, S.; Leung, L.K. The Citrus Flavonone Hesperetin Attenuates the Nuclear Translocation of Aryl Hydrocarbon Receptor. Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2018, 210, 57–64. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Lv, Q.; Meng Miao, Y.; Miao Qiao, S.; Dai, Y.; Feng Wei, Z. Cardamonin, a Natural Flavone, Alleviates Inflammatory Bowel Disease by the Inhibition of NLRP3 Inflammasome Activation via an AhR/Nrf2/NQO1 Pathway. Biochem. Pharmacol. 2018, 155, 494–509. [Google Scholar] [CrossRef] [PubMed]
- Dunlap, T.L.; Howell, C.E.; Mukand, N.; Chen, S.N.; Pauli, G.F.; Dietz, B.M.; Bolton, J.L. Red Clover Aryl Hydrocarbon Receptor (AhR) and Estrogen Receptor (ER) Agonists Enhance Genotoxic Estrogen Metabolism. Chem. Res. Toxicol. 2017, 30, 2084–2092. [Google Scholar] [CrossRef] [PubMed]
- Bialesova, L.; Novotna, A.; Macejova, D.; Brtko, J.; Dvorak, Z. Agonistic Effect of Selected Isoflavones on Arylhydrocarbon Receptor in a Novel AZ-AhR Transgenic Gene Reporter Human Cell Line. Gen. Physiol. Biophys. 2015, 34, 331–334. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Froyen, E.B.; Steinberg, F.M. Genistein Decreases Basal Hepatic Cytochrome P450 1A1 Protein Expression and Activity in Swiss Webster Mice. Nutr. Res. 2016, 36, 430–439. [Google Scholar] [CrossRef] [PubMed]
- Ashida, H.; Harada, K.; Mishima, S.; Mitani, T.; Yamashita, Y.; Matsumura, F. Luteolin Suppresses TCDD-Induced Wasting Syndrome in a Cultured Adipocyte Model. Pestic. Biochem. Physiol. 2015, 120, 14–20. [Google Scholar] [CrossRef] [PubMed]
- Miao, Y.; Lv, Q.; Qiao, S.; Yang, L.; Tao, Y.; Yan, W.; Wang, P.; Cao, N.; Dai, Y.; Wei, Z. Alpinetin Improves Intestinal Barrier Homeostasis via Regulating AhR/Suv39h1/TSC2/MTORC1/Autophagy Pathway. Toxicol. Appl. Pharmacol. 2019, 384, 1–15. [Google Scholar] [CrossRef]
- Lv, Q.; Shi, C.; Qiao, S.; Cao, N.; Guan, C.; Dai, Y.; Wei, Z. Alpinetin Exerts Anti-Colitis Efficacy by Activating AhR, Regulating MiR-302/DNMT- 1/CREB Signals, and Therefore Promoting Treg Differentiation. Cell Death Dis. 2018, 9, 1–25. [Google Scholar] [CrossRef]
- Fukuda, I.; Mukai, R.; Kawase, M.; Yoshida, K.; Ashida, H. Interaction between the Aryl Hydrocarbon Receptor and Its Antagonists, Flavonoids. Biochem. Biophys. Res. Commun. 2007, 359, 822–827. [Google Scholar] [CrossRef]
- Medjakovic, S.; Jungbauer, A. Red Clover Isoflavones Biochanin A and Formononetin Are Potent Ligands of the Human Aryl Hydrocarbon Receptor. J. Steroid Biochem. Mol. Biol. 2008, 108, 171–177. [Google Scholar] [CrossRef]


| Flavonoids | Bioassays a | Cell lines & Tissues b | [μM] | Reference |
|---|---|---|---|---|
| Flavones Acacetin, Apigenin, Baicalin, Chrysin, Flavone, Genkwanin, Luteolin, Luteolin 7,3′-diglucoside, Scutellarein, Tangeretin, Tricetin, 4′,7-dimethoxy-5-hydroxyflavone, 4′,5,7-trimethoxyflavone, 3′,4′,5,7-tetramethoxyflavone. | RT-qPCR, PCR, LucRGA, ChIP, WB, IF, IHC | Caco2, YAMC, DLN, HepG2 AhR-Lucia, H1L6.1c2, 3T3-L1 (AhR) HCT116, MDA-MB-231, Hepatic tissue (rats), Myocardial tissue (mice) | 10–100 | [128,129,130,131,132,133,134,135,136,137] |
| Flavonols Fisetin, Flavonol, Galangin, Gossypetin, Guaijaverin, Icaritin, Isorhamnetin, Kaempferol, Morin, Myricetin, Quercetin, Quercitrin, Robinetin, Rutin. Tamarixetin, 3,6,2′,3′-Tetrahydroxyflavone, 3,6,2′,4′-Tetrahydroxyflavone. | RT-qPCR, ChIP, WB, LucRGA | LNCaP, CWR22Rv1, Caco2, H1L6.1c2, 3T3-L1 (AhR), PBMEC/C1-2, HepG2 AhR-Lucia | 10–100 | [128,129,135,136,138,139] |
| Dihydroflavonols Taxifolin, Dihydromyricetin | RT-qPCR, ChIP, WB, LucRGA | Caco2, HepG2 | 0.1–100 | [135,140] |
| Flavolignane Silymarin | LucRGA | H1L6.1c2 | 25 | [139] |
| Flavanones Alpinetin, Eriodictyol, Flavanone, Hesperetin, Hesperidin, Isoxanthohumol, Naringenin, Naringin, Naringenin Trimethyl Ether, Sakuranetin, 6-Prenylnaringenin, 8-Prenylnaringenin | LucRGA, RT-qPCR, WB, ChIP, Co-IP, EROD | HepG2, MCF-7, HepG2 AhR-Lucia, H1L6.1c2, Caco2, YAMC, PBMEC/C1-2, EL-4 | 0.1–100 | [136,137,139,141,142] |
| Flavan 3-ol Epigallocatechin gallate | WB, LucRGA | 3T3-L1 (AhR), HepG2 AhR-Lucia | 30–100 | [128,136] |
| Chalcone Cardamonin | qPCR, WB | THP-1 | 3.0–30 | [143] |
| Isoflavones Biochanin A, Daidzein, Formononetin, Genistein, Puerarin, Prunetin, 4′,7-dimethoxy-5-hydroxyisoflavone, 4′,5,7-trimethoxyisoflavone, 3′,4′,5,7-tetramethoxyisoflavone | RT-qPCR, LucRGA, WB | Caco2, YAMC, H1L6.1c2, MCF-7, HC-04, HepG2 (AZ-AhR), Hepa-1c1c7, HepG2 AhR-Lucia | 0.1–1000 | [136,137,139,144,145,146] |
| Flavones | |||
|---|---|---|---|
Flavone 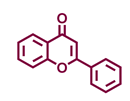 | Chrysin 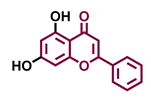 | Apigenin  | Luteolin 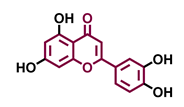 |
| Agonist [139] Antagonist [129] | Agonist [133,139] Antagonist [129] | Agonist [135,136,139] Antagonist [129] | Agonist [135] Antagonist [147] |
Tricetin 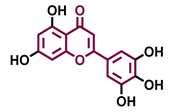 | Scutellarein 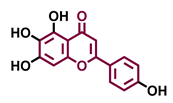 | Acacetin 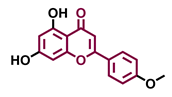 | Genkwanin 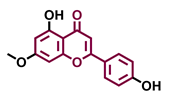 |
| Agonist [135] | Agonist [136] | Agonist [137] | Non-agonist [137] |
4′,7-Dimethoxy-5-hydroxyflavone  | 4′,5,7-Trimethoxyflavone 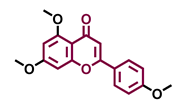 | 3′,4′,5,7-tetramethoxyflavone 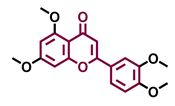 | Tangeretin  |
| Agonist [137] | Non-agonist [137] | Agonist [137] | Antagonist [132] |
Baicalin  | Luteolin 7,3′-diglucoside 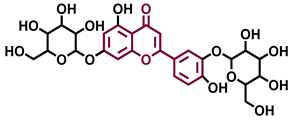 | ||
| Agonist [131,139] Antagonist [130] | Antagonist [128] | ||
| Flavonols | |||
|---|---|---|---|
Flavonol  | Galangin 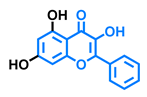 | Kaempferol 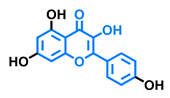 | Fisetin 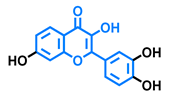 |
| Agonist [128] Antagonist [128] | Antagonist [128] | Agonist [135] Antagonist [128,135] | Agonist [135,139] Antagonist [128,135] |
3,6,2′,3′-Tetrahydroxyflavone 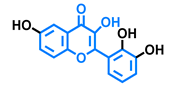 | 3,6,2′,4′-Tetrahydroxyflavone 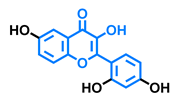 | Morin 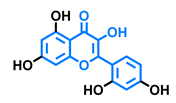 | Quercetin 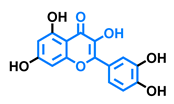 |
| Agonist [135] | Agonist [135] Antagonist [135] | Agonist [135] Antagonist [129] | Agonist [135,139] Antagonist [128] |
Robinetin 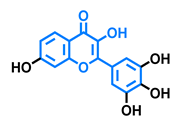 | Gossypetin  | Myricetin 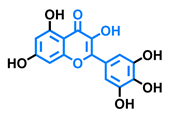 | Isorhamnetin 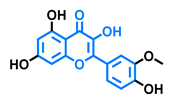 |
| Agonist [135] | Agonist [135] | Agonist [135] Antagonist [129] | Antagonist [129] |
Tamarixetin 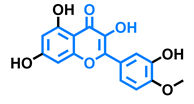 | Icaritin 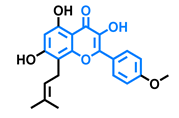 | Guaijaverin 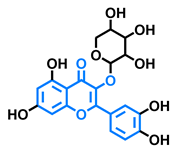 | Quercitrin  |
| Agonist [128], Antagonist [129] | Agonist [138] | Agonist [136] | Antagonist [128] |
Rutin  Agonist [139] Antagonist [128] | |||
| Flavanones | ||
|---|---|---|
Flavanone  | Naringenin 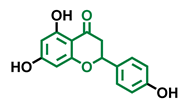 | Eriodictyol  |
| Antagonist [129] Agonist [128] | Antagonist [129] Non-agonist [137] | Antagonist [128] |
Alpinetin  | Sakuranetin 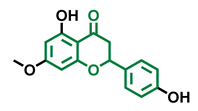 | Hesperetin 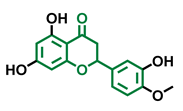 |
| Agonist [148,149] | Non-agonist [136] | Agonist [139] Antagonist [142] |
Naringenin Trimethyl Ether 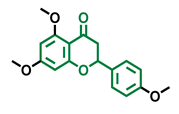 | Hesperidin 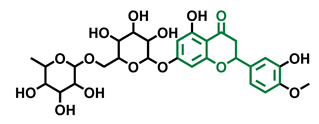 | Naringin  |
| Non-agonist [137] | Agonist [136,139] | Agonist [139] Antagonist [128] |
6-Prenylnaringenin 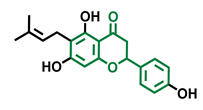 | 8-Prenylnaringenin  | Isoxanthohumol  |
| Agonist [141] | Agonist [141] | Non-agonist [141] |
| Isoflavones | |||
|---|---|---|---|
Daidzein  | Genistein 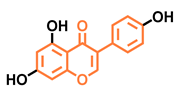 | Formononetin 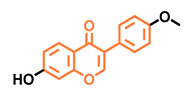 | Biochanin A  |
| Antagonist [144] Non-agonist [145] | Antagonist [144,146] Non-agonist [136,137] | Agonist [144,145] | Agonist [139,144,145] |
Prunetin 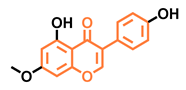 | 4′,7-dimethoxy-5-hydroxyisoflavone 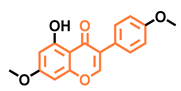 | 4′,5,7-trimethoxyisoflavone 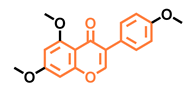 | 3′,4′,5,7-tetramethoxyisoflavone 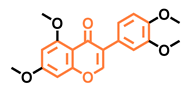 |
| Non-agonist [137] | Non-agonist [137] | Agonist [137] | Non-agonist [137] |
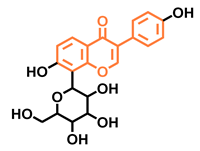 | Puerarin | ||
| Non-antagonist [128] | |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Goya-Jorge, E.; Jorge Rodríguez, M.E.; Veitía, M.S.-I.; Giner, R.M. Plant Occurring Flavonoids as Modulators of the Aryl Hydrocarbon Receptor. Molecules 2021, 26, 2315. https://doi.org/10.3390/molecules26082315
Goya-Jorge E, Jorge Rodríguez ME, Veitía MS-I, Giner RM. Plant Occurring Flavonoids as Modulators of the Aryl Hydrocarbon Receptor. Molecules. 2021; 26(8):2315. https://doi.org/10.3390/molecules26082315
Chicago/Turabian StyleGoya-Jorge, Elizabeth, María Elisa Jorge Rodríguez, Maité Sylla-Iyarreta Veitía, and Rosa M. Giner. 2021. "Plant Occurring Flavonoids as Modulators of the Aryl Hydrocarbon Receptor" Molecules 26, no. 8: 2315. https://doi.org/10.3390/molecules26082315
APA StyleGoya-Jorge, E., Jorge Rodríguez, M. E., Veitía, M. S.-I., & Giner, R. M. (2021). Plant Occurring Flavonoids as Modulators of the Aryl Hydrocarbon Receptor. Molecules, 26(8), 2315. https://doi.org/10.3390/molecules26082315










