Synthesis of 4-Arylselanyl-1H-1,2,3-triazoles from Selenium-Containing Carbinols
Abstract
1. Introduction
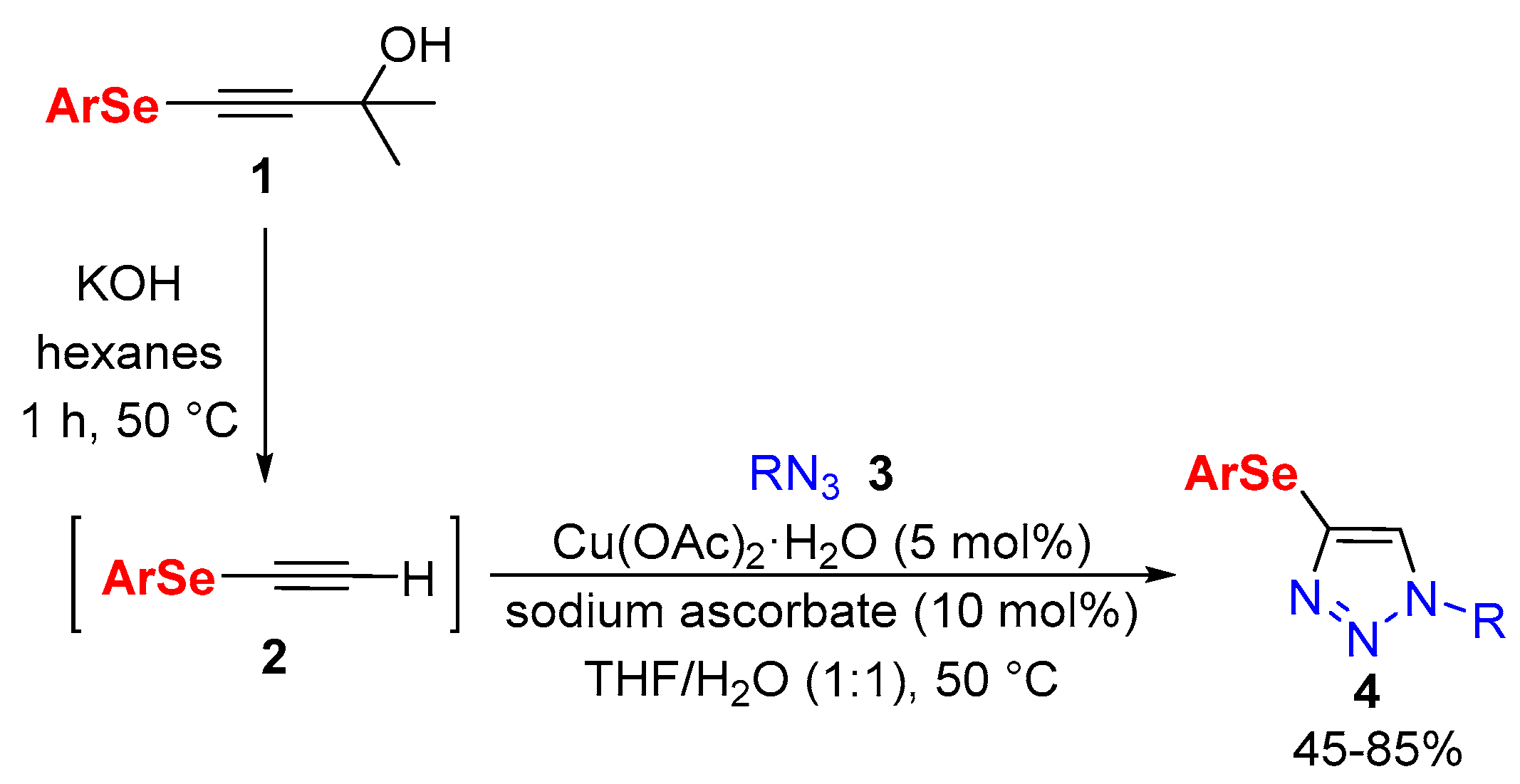
2. Results and Discussion
3. Materials and Methods
General Procedure for the Synthesis of 4-Arylselanyl-1H-1,2,3-triazoles 4
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Dehaen, W.; Bakulev, V.A. (Eds.) Chemistry of 1,2,3-Triazoles; Topics in Heterocyclic Chemistry; Springer International Publishing: Cham, Switzerland, 2015; Volume 40, ISBN 9783319079615. [Google Scholar]
- Themed collection: Click chemistry: Function follows form. Chem. Soc. Rev. 2010, 39, 1221–1407.
- Bertozzi, C.R. A Decade of Bioorthogonal Chemistry, Themed collection: Bioorthogonal Chemistry in Biology. Acc. Chem. Res. 2011, 44, 651–840. [Google Scholar] [CrossRef]
- Costa, G.P.; Baldinotti, R.S.M.; Fronza, M.G.; Nascimento, J.E.R.; Dias, I.F.C.; Sonego, M.S.; Seixas, F.K.; Collares, T.; Perin, G.; Jacob, R.G.; et al. Synthesis, Molecular Docking, and Preliminary Evaluation of 2-(1,2,3-Triazoyl)benzaldehydes as Multifunctional Agents for the Treatment of Alzheimer’s Disease. ChemMedChem 2020, 15, 610–622. [Google Scholar] [CrossRef] [PubMed]
- Begnini, K.R.; Duarte, W.R.; da Silva, L.P.; Buss, J.H.; Goldani, B.S.; Fronza, M.; Segatto, N.V.; Alves, D.; Savegnago, L.; Seixas, F.K.; et al. Apoptosis Induction by 7-Chloroquinoline-1,2,3-Triazoyl Carboxamides in Triple Negative Breast Cancer Cells. Biomed. Pharmacother. 2017, 91, 510–516. [Google Scholar] [CrossRef]
- Xie, J.; Seto, C.T. A Two Stage Click-Based Library of Protein Tyrosine Phosphatase Inhibitors. Bioorg. Med. Chem. 2007, 15, 458–473. [Google Scholar] [CrossRef]
- Lee, T.; Cho, M.; Ko, S.-Y.; Youn, H.-J.; Baek, D.J.; Cho, W.-J.; Kang, C.-Y.; Kim, S. Synthesis and Evaluation of 1,2,3-Triazole Containing Analogues of the Immunostimulant α-GalCer. J. Med. Chem. 2007, 50, 585–589. [Google Scholar] [CrossRef] [PubMed]
- Parrish, B.; Emrick, T. Soluble Camptothecin Derivatives Prepared by Click Cycloaddition Chemistry on Functional Aliphatic Polyesters. Bioconjugate Chem. 2007, 18, 263–267. [Google Scholar] [CrossRef]
- Pokhodylo, N.; Shyyka, O.; Matiychuk, V. Synthesis and Anticancer Activity Evaluation of New 1,2,3-Triazole-4-Carboxamide Derivatives. Med. Chem Res. 2014, 23, 2426–2438. [Google Scholar] [CrossRef]
- Wilhelm, E.A.; Machado, N.C.; Pedroso, A.B.; Goldani, B.S.; Seus, N.; Moura, S.; Savegnago, L.; Jacob, R.G.; Alves, D. Organocatalytic Synthesis and Evaluation of 7-Chloroquinoline-1,2,3-Triazoyl Carboxamides as Potential Antinociceptive, Anti-Inflammatory and Anticonvulsant Agent. RSC Adv. 2014, 4, 41437–41445. [Google Scholar] [CrossRef]
- Chavan, S.R.; Gavale, K.S.; Khan, A.; Joshi, R.; Kumbhar, N.; Chakravarty, D.; Dhavale, D.D. Iminosugars Spiro-Linked with Morpholine-Fused 1,2,3-Triazole: Synthesis, Conformational Analysis, Glycosidase Inhibitory Activity, Antifungal Assay, and Docking Studies. ACS Omega 2017, 2, 7203–7218. [Google Scholar] [CrossRef]
- Hein, C.D.; Liu, X.-M.; Wang, D. Click Chemistry, a Powerful Tool for Pharmaceutical Sciences. Pharm Res. 2008, 25, 2216–2230. [Google Scholar] [CrossRef] [PubMed]
- Tron, G.C.; Pirali, T.; Billington, R.A.; Canonico, P.L.; Sorba, G.; Genazzani, A.A. Click Chemistry Reactions in Medicinal Chemistry: Applications of the 1,3-Dipolar Cycloaddition between Azides and Alkynes. Med. Res. Rev. 2008, 28, 278–308. [Google Scholar] [CrossRef] [PubMed]
- Angell, Y.L.; Burgess, K. Peptidomimetics via Copper-Catalyzed Azide–Alkyne Cycloadditions. Chem. Soc. Rev. 2007, 36, 1674. [Google Scholar] [CrossRef] [PubMed]
- Oueis, E.; Jaspars, M.; Westwood, N.J.; Naismith, J.H. Enzymatic Macrocyclization of 1,2,3-Triazole Peptide Mimetics. Angew. Chem. Int. Ed. 2016, 55, 5842–5845. [Google Scholar] [CrossRef] [PubMed]
- Huisgen, R. 1,3-Dipolar Cycloadditions. Past and Future. Angew. Chem. Int. Ed. Engl. 1963, 2, 565–598. [Google Scholar] [CrossRef]
- Rostovtsev, V.V.; Green, L.G.; Fokin, V.V.; Sharpless, K.B. A Stepwise Huisgen Cycloaddition Process: Copper(I)-Catalyzed Regioselective “Ligation” of Azides and Terminal Alkynes. Angew. Chem. Int. Ed. 2002, 41, 2596–2599. [Google Scholar] [CrossRef]
- Tornøe, C.W.; Christensen, C.; Meldal, M. Peptidotriazoles on Solid Phase: [1,2,3]-Triazoles by Regiospecific Copper(I)-Catalyzed 1,3-Dipolar Cycloadditions of Terminal Alkynes to Azides. J. Org. Chem. 2002, 67, 3057–3064. [Google Scholar] [CrossRef]
- Krasiński, A.; Radić, Z.; Manetsch, R.; Raushel, J.; Taylor, P.; Sharpless, K.B.; Kolb, H.C. In Situ Selection of Lead Compounds by Click Chemistry: Target-Guided Optimization of Acetylcholinesterase Inhibitors. J. Am. Chem. Soc. 2005, 127, 6686–6692. [Google Scholar] [CrossRef]
- Lee, L.V.; Mitchell, M.L.; Huang, S.-J.; Fokin, V.V.; Sharpless, K.B.; Wong, C.-H. A Potent and Highly Selective Inhibitor of Human α-1,3-Fucosyltransferase via Click Chemistry. J. Am. Chem. Soc. 2003, 125, 9588–9589. [Google Scholar] [CrossRef]
- Hein, J.E.; Tripp, J.C.; Krasnova, L.B.; Sharpless, K.B.; Fokin, V.V. Copper(I)-Catalyzed Cycloaddition of Organic Azides and 1-Iodoalkynes. Angew. Chem. Int. Ed. 2009, 48, 8018–8021. [Google Scholar] [CrossRef]
- Zhang, L.; Chen, X.; Xue, P.; Sun, H.H.Y.; Williams, I.D.; Sharpless, K.B.; Fokin, V.V.; Jia, G. Ruthenium-Catalyzed Cycloaddition of Alkynes and Organic Azides. J. Am. Chem. Soc. 2005, 127, 15998–15999. [Google Scholar] [CrossRef] [PubMed]
- Boren, B.C.; Narayan, S.; Rasmussen, L.K.; Zhang, L.; Zhao, H.; Lin, Z.; Jia, G.; Fokin, V.V. Ruthenium-Catalyzed Azide−Alkyne Cycloaddition: Scope and Mechanism. J. Am. Chem. Soc. 2008, 130, 8923–8930. [Google Scholar] [CrossRef] [PubMed]
- McNulty, J.; Keskar, K.; Vemula, R. The First Well-Defined Silver(I)-Complex-Catalyzed Cycloaddition of Azides onto Terminal Alkynes at Room Temperature. Chem. Eur. J. 2011, 17, 14727–14730. [Google Scholar] [CrossRef] [PubMed]
- McNulty, J.; Keskar, K. Discovery of a Robust and Efficient Homogeneous Silver(I) Catalyst for the Cycloaddition of Azides onto Terminal Alkynes. Eur. J. Org. Chem. 2012, 2012, 5462–5470. [Google Scholar] [CrossRef]
- Ding, S.; Jia, G.; Sun, J. Iridium-Catalyzed Intermolecular Azide-Alkyne Cycloaddition of Internal Thioalkynes under Mild Conditions. Angew. Chem. Int. Ed. 2014, 53, 1877–1880. [Google Scholar] [CrossRef]
- Jalani, H.; Karagöz, A.; Tsogoeva, S. Synthesis of Substituted 1,2,3-Triazoles via Metal-Free Click Cycloaddition Reactions and Alternative Cyclization Methods. Synthesis 2016, 49, 29–41. [Google Scholar] [CrossRef]
- Lima, C.G.S.; Ali, A.; van Berkel, S.S.; Westermann, B.; Paixão, M.W. Emerging Approaches for the Synthesis of Triazoles: Beyond Metal-Catalyzed and Strain-Promoted Azide–Alkyne Cycloaddition. Chem. Commun. 2015, 51, 10784–10796. [Google Scholar] [CrossRef] [PubMed]
- Ramasastry, S.S.V. Enamine/Enolate-Mediated Organocatalytic Azide-Carbonyl [3+2] Cycloaddition Reactions for the Synthesis of Densely Functionalized 1,2,3-Triazoles. Angew. Chem. Int. Ed. 2014, 53, 14310–14312. [Google Scholar] [CrossRef] [PubMed]
- John, J.; Thomas, J.; Dehaen, W. Organocatalytic Routes toward Substituted 1,2,3-Triazoles. Chem. Commun. 2015, 51, 10797–10806. [Google Scholar] [CrossRef]
- Ramachary, D.B.; Ramakumar, K.; Narayana, V.V. Amino Acid-Catalyzed Cascade [3+2]-Cycloaddition/Hydrolysis Reactions Based on the Push-Pull Dienamine Platform: Synthesis of Highly Functionalized N H-1,2,3-Triazoles. Chem. Eur. J. 2008, 14, 9143–9147. [Google Scholar] [CrossRef]
- Danence, L.J.T.; Gao, Y.; Li, M.; Huang, Y.; Wang, J. Organocatalytic Enamide–Azide Cycloaddition Reactions: Regiospecific Synthesis of 1,4,5-Trisubstituted-1,2,3-Triazoles. Chem. Eur. J. 2011, 17, 3584–3587. [Google Scholar] [CrossRef] [PubMed]
- Belkheira, M.; El Abed, D.; Pons, J.-M.; Bressy, C. Organocatalytic Synthesis of 1,2,3-Triazoles from Unactivated Ketones and Arylazides. Chem. Eur. J. 2011, 17, 12917–12921. [Google Scholar] [CrossRef] [PubMed]
- Sangwan, R.; Javed; Dubey, A.; Mandal, P.K. Organocatalytic [3+2] Cycloadditions: Toward Facile Synthesis of Sulfonyl-1,2,3-Triazolyl and Fully Substituted 1,2,3-Triazolyl Glycoconjugates. ChemistrySelect 2017, 2, 4733–4743. [Google Scholar] [CrossRef]
- Nogueira, C.W.; Rocha, J.B.T. Organoselenium and organotellurium compounds: Toxicology and pharmacology. In PATAI’S Chemistry of Functional Groups; Rappoport, Z., Ed.; John Wiley & Sons, Ltd.: Chichester, UK, 2011; ISBN 9780470682531. [Google Scholar]
- Alberto, E.E.; Braga, A.L. Selenium and Tellurium Chemistry: From Small Molecules to Biomolecules and Materials; Derek, W.J., Risto, L., Eds.; Springer: Berlin/Heidelberg, Germany, 2011; ISBN 978-3-642-20699-3. [Google Scholar]
- Iwaoka, M. Antioxidant Organoselenium Molecules. In Organoselenium Chemistry: Between Synthesis and Biochemistry; Santi, C., Ed.; Bentham Science, e-book: Sharjah, United Arab Emirates, 2014; ISBN 978-1-60805-838-9. [Google Scholar]
- Menezes, P.H.; Zeni, G. Vinyl Selenides. In PATAI’S Chemistry of Functional Groups; Rappoport, Z., Ed.; John Wiley & Sons, Ltd.: Chichester, UK, 2011; ISBN 9780470682531. [Google Scholar]
- Ursini, F.; Bindoli, A. The Role of Selenium Peroxidases in the Protection against Oxidative Damage of Membranes. Chem. Phys. Lipids 1987, 44, 255–276. [Google Scholar] [CrossRef]
- Stazi, A.V.; Trinti, B. Selenium deficiency in celiac disease: Risk of autoimmune thyroid diseases. Minerva Med. 2008, 99, 643–653. [Google Scholar] [PubMed]
- Petronilho, F.; Michels, M.; Danielski, L.G.; Goldim, M.P.; Florentino, D.; Vieira, A.; Mendonça, M.G.; Tournier, M.; Piacentini, B.; Giustina, A.D.; et al. Diphenyl Diselenide Attenuates Oxidative Stress and Inflammatory Parameters in Ulcerative Colitis: A Comparison with Ebselen. Pathol. Res. Pract. 2016, 212, 755–760. [Google Scholar] [CrossRef] [PubMed]
- Rosa, S.G.; Quines, C.B.; Stangherlin, E.C.; Nogueira, C.W. Diphenyl Diselenide Ameliorates Monosodium Glutamate Induced Anxiety-like Behavior in Rats by Modulating Hippocampal BDNF-Akt Pathway and Uptake of GABA and Serotonin Neurotransmitters. Physiol. Behav. 2016, 155, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, C.E.S.; Sari, M.H.M.; Zborowski, V.A.; Araujo, P.C.O.; Nogueira, C.W.; Zeni, G. P,P′-Methoxyl-Diphenyl Diselenide Elicits an Antidepressant-like Effect in Mice without Discontinuation Anxiety Phenotype. Pharmacol. Biochem. Behav. 2017, 154, 31–38. [Google Scholar] [CrossRef] [PubMed]
- Reis, A.S.; Pinz, M.; Duarte, L.F.B.; Roehrs, J.A.; Alves, D.; Luchese, C.; Wilhelm, E.A. 4-Phenylselenyl-7-Chloroquinoline, a Novel Multitarget Compound with Anxiolytic Activity: Contribution of the Glutamatergic System. J. Psychiatr. Res. 2017, 84, 191–199. [Google Scholar] [CrossRef]
- Salgueiro, W.G.; Goldani, B.S.; Peres, T.V.; Miranda-Vizuete, A.; Aschner, M.; da Rocha, J.B.T.; Alves, D.; Ávila, D.S. Insights into the Differential Toxicological and Antioxidant Effects of 4-Phenylchalcogenil-7-Chloroquinolines in Caenorhabditis Elegans. Free Rad. Biol. Med. 2017, 110, 133–141. [Google Scholar] [CrossRef]
- Lopes, E.F.; Penteado, F.; Thurow, S.; Pinz, M.; Reis, A.S.; Wilhelm, E.A.; Luchese, C.; Barcellos, T.; Dalberto, B.; Alves, D.; et al. Synthesis of Isoxazolines by the Electrophilic Chalcogenation of β,γ-Unsaturated Oximes: Fishing Novel Anti-Inflammatory Agents. J. Org. Chem. 2019, 84, 12452–12462. [Google Scholar] [CrossRef]
- Wirth, T. (Ed.) Organoselenium Chemistry: Synthesis and Reactions; Wiley-VCH-Verl: Weinheim, Germany, 2012; ISBN 9783527641956. [Google Scholar]
- Perin, G.; Alves, D.; Jacob, R.G.; Barcellos, A.M.; Soares, L.K.; Lenardão, E.J. Synthesis of Organochalcogen Compounds Using Non-Conventional Reaction Media. ChemistrySelect 2016, 1, 205–258. [Google Scholar] [CrossRef]
- Lenardão, E.J.; Santi, C.; Sancineto, L. New Frontiers in Organoselenium Compounds; Springer International Publishing: Cham, Switzerland, 2018; ISBN 9783319924045. [Google Scholar]
- Alves, D.; Goldani, B.; Lenardão, E.J.; Perin, G.; Schumacher, R.F.; Paixão, M.W. Copper Catalysis and Organocatalysis Showing the Way: Synthesis of Selenium-Containing Highly Functionalized 1,2,3-Triazoles. Chem. Rec. 2018, 18, 527–542. [Google Scholar] [CrossRef] [PubMed]
- Cruz, E.H.G.; Silvers, M.A.; Jardim, G.A.M.; Resende, J.M.; Cavalcanti, B.C.; Bomfim, I.S.; Pessoa, C.; de Simone, C.A.; Bottesele, G.V.; Braga, A.L.; et al. Synthesis and Antitumor Activity of Selenium-Containing Quinone-Based Triazoles Possessing two Redox Centres, and their Mechanistic Insights. Eur. J. Med. Chem. 2016, 122, 1–16. [Google Scholar] [CrossRef]
- Jardim, G.A.M.; Lima, D.J.B.; Valença, W.O.; Lima, D.J.B.; Cavalcanti, B.C.; Pessoa, C.; Rafique, J.; Braga, A.L.; Jacob, C.; da Silva Júnior, E.N.; et al. Synthesis of Selenium-Quinone Hybrid Compounds with Potential Antitumor Activity via Rh-Catalyzed C-H Bond Activation and Click Reactions. Molecules 2018, 23, 83. [Google Scholar] [CrossRef]
- Cui, F.; Chen, J.; Mo, Z.; Su, S.; Chen, Y.; Ma, X.; Tang, H.; Wang, H.; Pan, Y.; Xu, Y. Copper-Catalyzed Decarboxylative/Click Cascade Reaction: Regioselective Assembly of 5-Selenotriazole Anticancer Agents. Org. Lett. 2018, 20, 925–929. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Peng, X.; Wei, F.; Tung, C.-H.; Xu, Z. Copper(I)-Catalyzed Interrupted Click Reaction: Synthesis of Diverse 5-Hetero-Functionalized Triazoles. Angew. Chem. Int. Ed. 2016, 55, 649–653. [Google Scholar] [CrossRef] [PubMed]
- Stefani, H.A.; Leal, D.M.; Manarin, F. 4-Organochalcogenoyl-1H-1,2,3-Triazoles: Synthesis and Functionalization by a Nickel-Catalyzed Negishi Cross-Coupling Reaction. Tetrahedron Lett. 2012, 53, 6495–6499. [Google Scholar] [CrossRef]
- Saraiva, M.; Seus, N.; de Souza, D.; Rodrigues, O.; Paixão, M.; Jacob, R.; Lenardão, E.; Perin, G.; Alves, D. Synthesis of [(Arylselanyl)Alkyl]-1,2,3-Triazoles by Copper-Catalyzed 1,3-Dipolar Cycloaddition of (Arylselanyl)Alkynes with Benzyl Azides. Synthesis 2012, 44, 1997–2004. [Google Scholar] [CrossRef]
- Braga, A.L.; Reckziegel, A.; Silveira, C.C.; Comasseto, J.V. Vicinal Difunctionalization of Alkynyl Selenides with Lithium Butylcyano Cuprate and Electrophiles. Synth. Commun. 1994, 24, 1165–1170. [Google Scholar] [CrossRef]
- Lopes, E.F.; Dalberto, B.T.; Perin, G.; Alves, D.; Barcellos, T.; Lenardão, E.J. Synthesis of Terminal Ethynyl Aryl Selenides and Sulfides Based on the Retro-Favorskii Reaction of Hydroxypropargyl Precursors. Chem. Eur. J. 2017, 23, 13760–13765. [Google Scholar] [CrossRef] [PubMed]
- Ivasiv, V.; Albertini, C.; Gonçalves, A.E.; Rossi, M.; Bolognesi, M.L. Molecular Hybridization as a Tool for Designing Multitarget Drug Candidates for Complex Diseases. Curr. Top. Med. Chem. 2019, 19, 1694–1711. [Google Scholar] [CrossRef] [PubMed]
- Viegas-Junior, C.; Barreiro, E.J.; Fraga, C.A.M. Molecular Hybridization: A Useful Tool in the Design of New Drug Prototypes. Curr. Med. Chem. 2007, 14, 1829–1852. [Google Scholar] [CrossRef] [PubMed]
- Li, L.-J.; Zhang, Y.-Q.; Zhang, Y.; Zhu, A.-L.; Zhang, G.-S. Synthesis of 5-Functionalized-1,2,3-Triazoles via a One-Pot Aerobic Oxidative Coupling Reaction of Alkynes and Azides. Chin. Chem. Lett. 2014, 25, 1161–1164. [Google Scholar] [CrossRef]
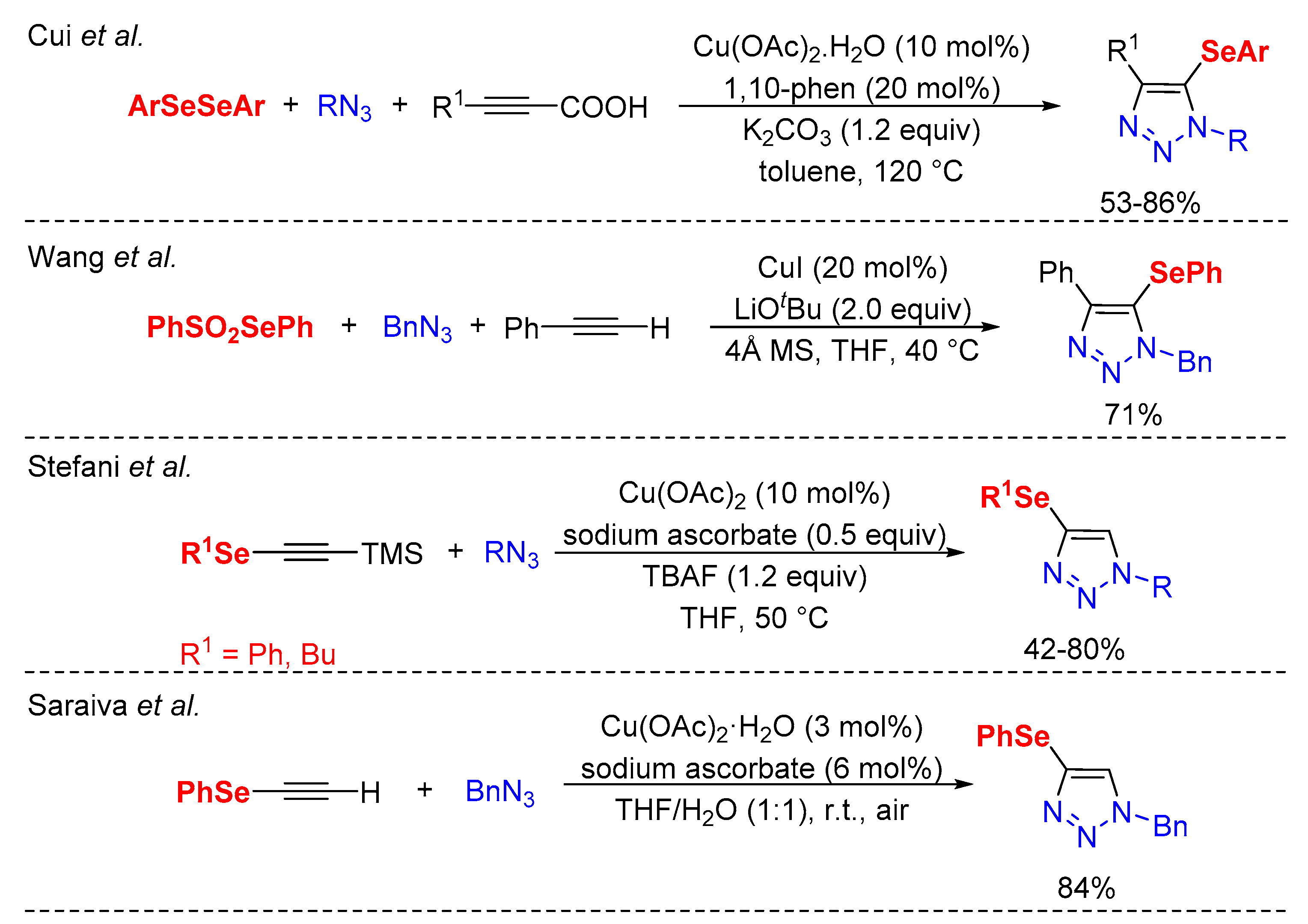



 | |||
| Entry | Copper Salt | Solvent | Yield (%) b |
| 1 | Cu(OAc)2·H2O | THF/H2O | 85 |
| 2 | CuI | THF/H2O | 60 |
| 3 c | CuI | DMSO | 32 |
| 4 | CuOnps | THF/H2O | traces |
| 5 | CuCl2 | THF/H2O | 75 |
| 6 | Cu(OAc)2·H2O | THF | - |
| 7 d | Cu(OAc)2·H2O | THF/H2O | 40 |
| 8 e | Cu(OAc)2·H2O | THF/H2O | 65 |
| 9 f | Cu(OAc)2·H2O | THF/H2O | 40 |
| 10 g | Cu(OAc)2·H2O | THF/H2O | 50 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Begini, F.; Balaguez, R.A.; Larroza, A.; Lopes, E.F.; Lenardão, E.J.; Santi, C.; Alves, D. Synthesis of 4-Arylselanyl-1H-1,2,3-triazoles from Selenium-Containing Carbinols. Molecules 2021, 26, 2224. https://doi.org/10.3390/molecules26082224
Begini F, Balaguez RA, Larroza A, Lopes EF, Lenardão EJ, Santi C, Alves D. Synthesis of 4-Arylselanyl-1H-1,2,3-triazoles from Selenium-Containing Carbinols. Molecules. 2021; 26(8):2224. https://doi.org/10.3390/molecules26082224
Chicago/Turabian StyleBegini, Francesca, Renata A. Balaguez, Allya Larroza, Eric F. Lopes, Eder João Lenardão, Claudio Santi, and Diego Alves. 2021. "Synthesis of 4-Arylselanyl-1H-1,2,3-triazoles from Selenium-Containing Carbinols" Molecules 26, no. 8: 2224. https://doi.org/10.3390/molecules26082224
APA StyleBegini, F., Balaguez, R. A., Larroza, A., Lopes, E. F., Lenardão, E. J., Santi, C., & Alves, D. (2021). Synthesis of 4-Arylselanyl-1H-1,2,3-triazoles from Selenium-Containing Carbinols. Molecules, 26(8), 2224. https://doi.org/10.3390/molecules26082224










