An Overview of the Antimicrobial Properties of Lignocellulosic Materials
Abstract
1. Introduction
2. Lignocellulosic Materials and Main Compounds
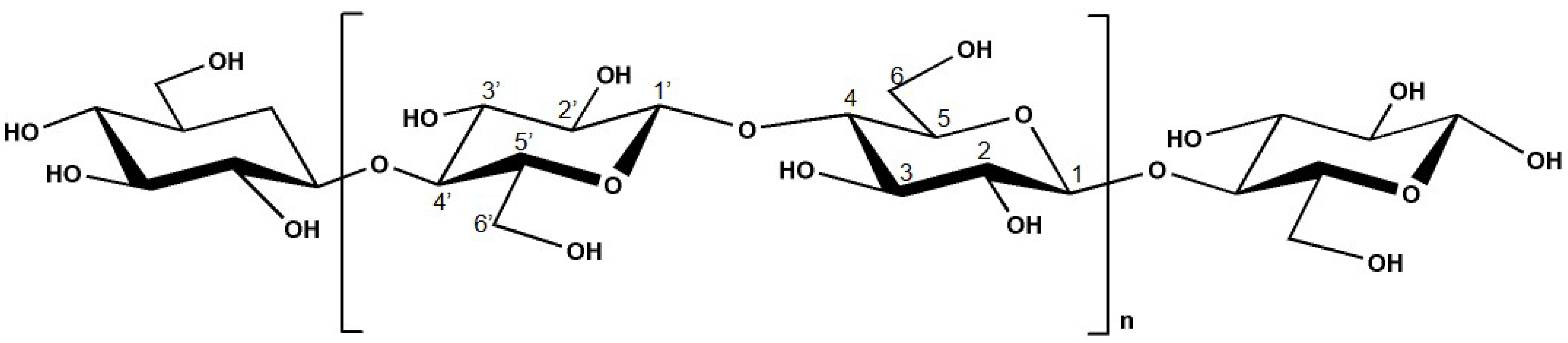
2.1. Cellulose
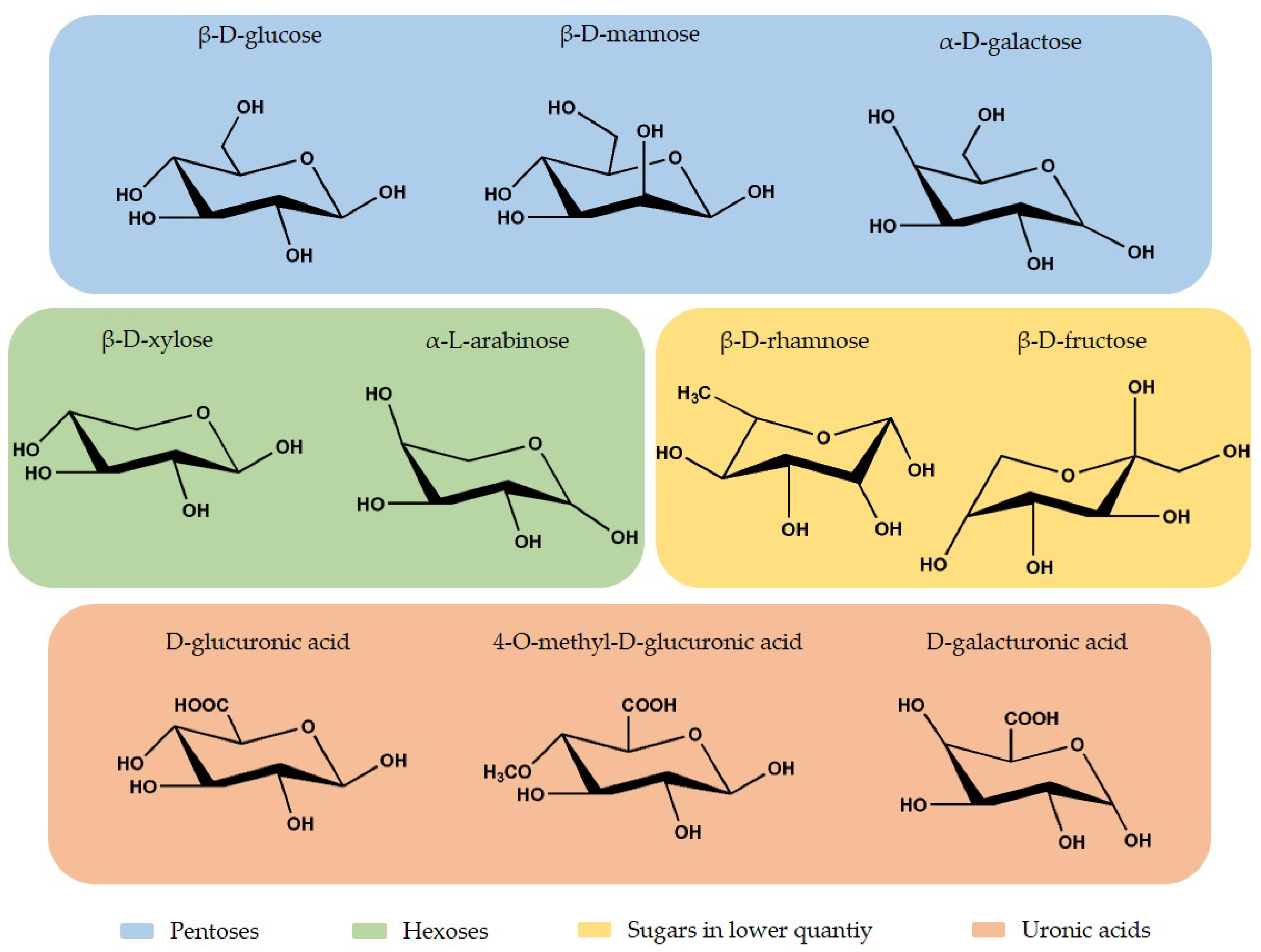
2.2. Hemicellulose
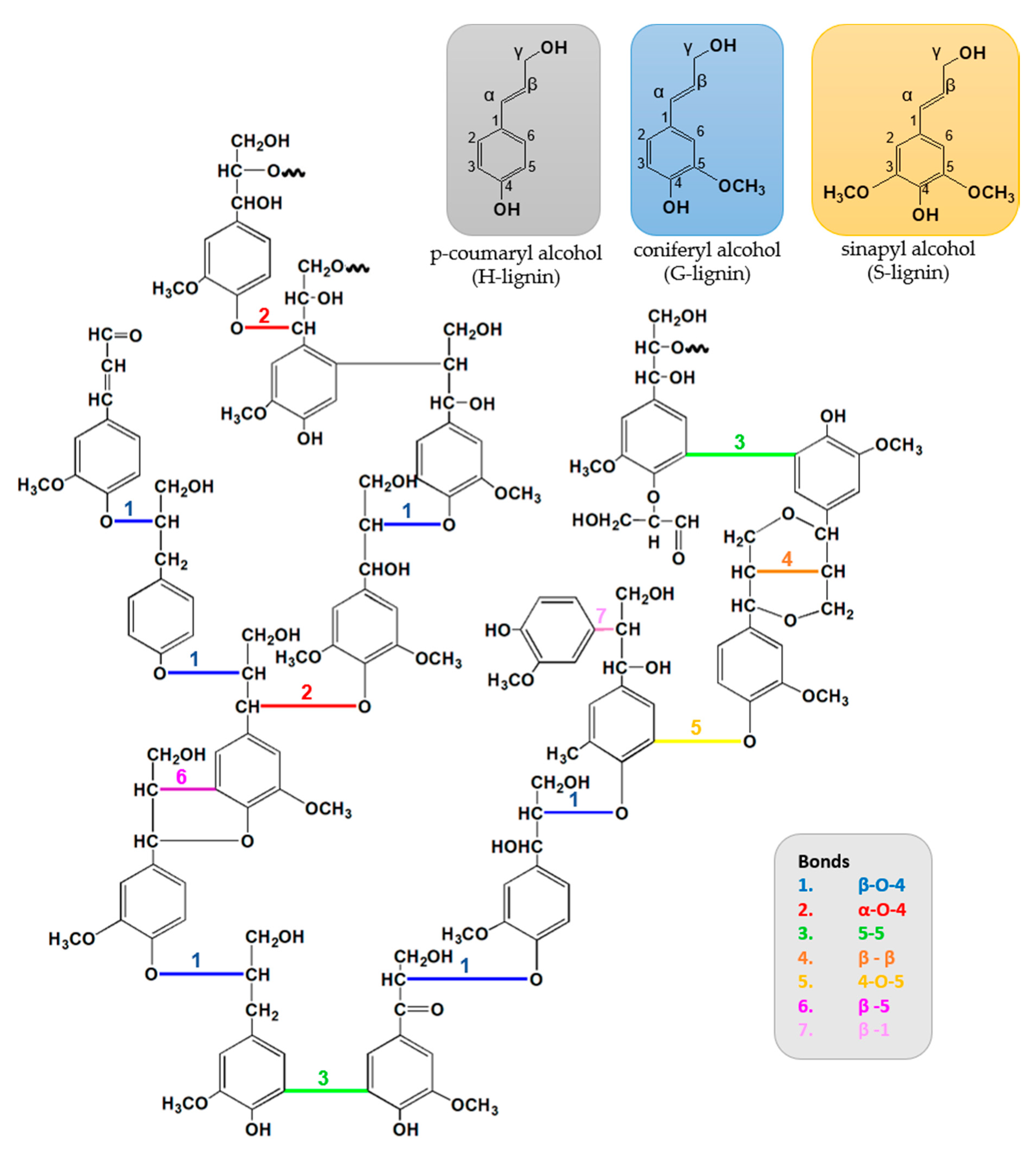
2.3. Lignin
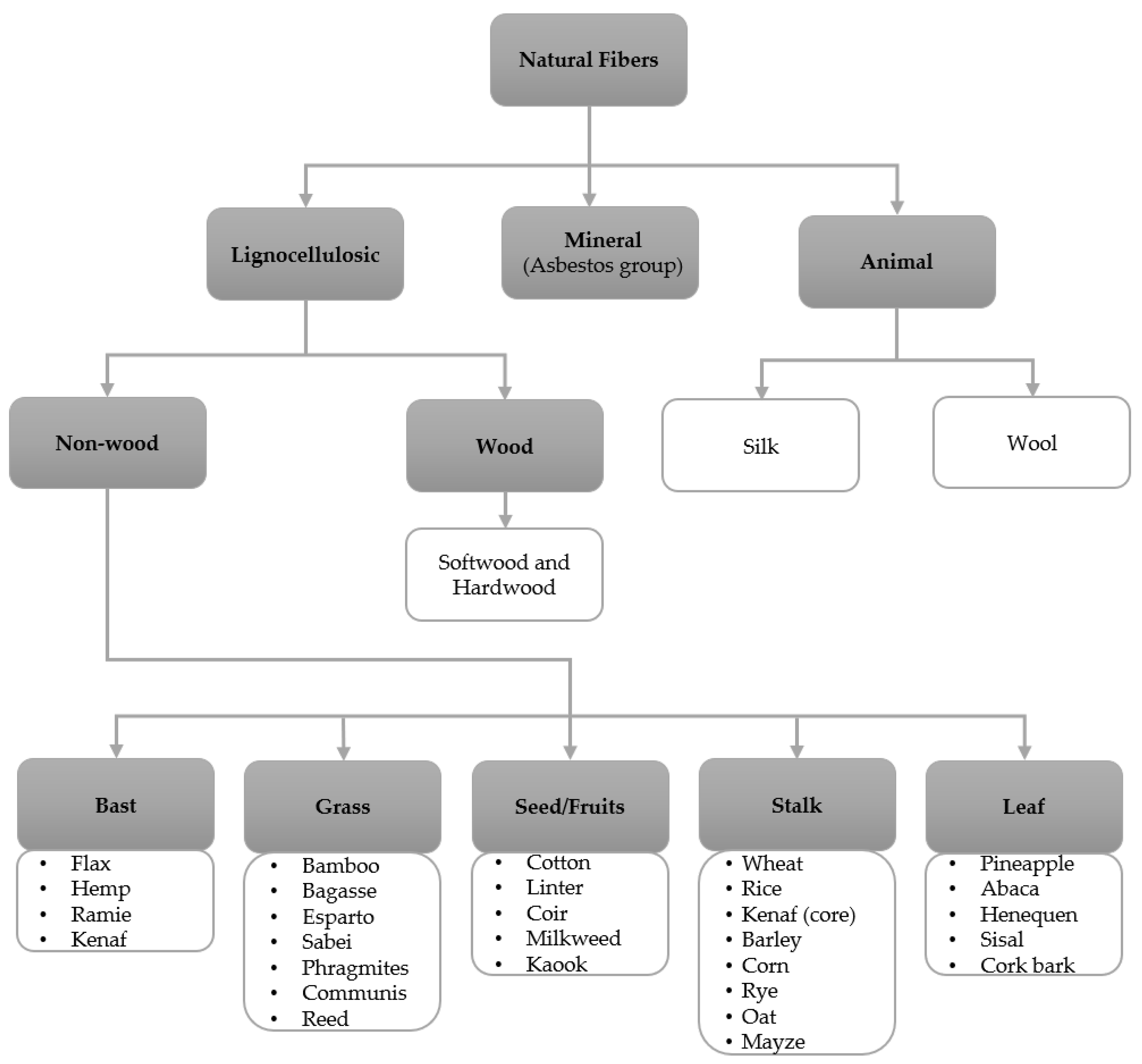
3. Lignocellulosic Fibers
3.1. Wood Fibers
3.2. Non-Wood Fibers
4. Intellectual Property
5. Conclusions and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Franco, A.R.; Fernandes, E.M.; Rodrigues, M.T.; Rodrigues, F.J.; Gomes, M.E.; Leonor, I.B.; Kaplan, D.L.; Reis, R.L. Antimicrobial coating of spider silk to prevent bacterial attachment on silk surgical sutures. Acta Biomater. 2019, 99, 236–246. [Google Scholar] [CrossRef]
- Song, B.; Zhang, E.; Han, X.; Zhu, H.; Shi, Y.; Cao, Z. Engineering and Application Perspectives on Designing an Antimicrobial Surface. ACS Appl. Mater. Interfaces 2020, 12, 21330–21341. [Google Scholar] [CrossRef] [PubMed]
- Roy, R.; Tiwari, M.; Donelli, G.; Tiwari, V. Strategies for combating bacterial biofilms: A focus on anti-biofilm agents and their mechanisms of action. Virulence 2018, 9, 522–554. [Google Scholar] [CrossRef]
- Simões, M.; Simões, L.C.; Vieira, M.J. A review of current and emergent biofilm control strategies. LWT Food Sci. Technol. 2010, 43, 573–583. [Google Scholar] [CrossRef]
- Yin, W.; Wang, Y.; Liu, L.; He, J. Biofilms: The Microbial “Protective Clothing” in Extreme Environments. Int. J. Mol. Sci. 2019, 20, 3423. [Google Scholar] [CrossRef] [PubMed]
- Srey, S.; Jahid, I.K.; Ha, S.-D. Biofilm formation in food industries: A food safety concern. Food Control 2013, 31, 572–585. [Google Scholar] [CrossRef]
- Xu, D.; Jia, R.; Li, Y.; Gu, T. Advances in the treatment of problematic industrial biofilms. World J. Microbiol. Biotechnol. 2017, 33. [Google Scholar] [CrossRef]
- Khameneh, B.; Iranshahy, M.; Soheili, V.; Fazly Bazzaz, B.S. Review on plant antimicrobials: A mechanistic viewpoint. Antimicrob. Resist. Infect. Control 2019, 8, 118. [Google Scholar] [CrossRef]
- Choudhury, R.R.; Gohil, J.M.; Mohanty, S.; Nayak, S.K. Antifouling, fouling release and antimicrobial materials for surface modification of reverse osmosis and nanofiltration membranes. J. Mater. Chem. A 2018, 6, 313–333. [Google Scholar] [CrossRef]
- Sharma, R.; Jafari, S.M.; Sharma, S. Antimicrobial bio-nanocomposites and their potential applications in food packaging. Food Control 2020, 112. [Google Scholar] [CrossRef]
- Al-Tayyar, N.A.; Youssef, A.M.; Al-Hindi, R. Antimicrobial food packaging based on sustainable Bio-based materials for reducing foodborne Pathogens: A review. Food Chem. 2020, 310, 125915. [Google Scholar] [CrossRef]
- Topuz, F.; Uyar, T. Antioxidant, antibacterial and antifungal electrospun nanofibers for food packaging applications. Food Res. Int. 2020, 130, 108927. [Google Scholar] [CrossRef] [PubMed]
- Agbor, V.B.; Cicek, N.; Sparling, R.; Berlin, A.; Levin, D.B. Biomass pretreatment: Fundamentals toward application. Biotechnol. Adv. 2011, 29, 675–685. [Google Scholar] [CrossRef] [PubMed]
- Ten, E.; Vermerris, W. Functionalized Polymers from Lignocellulosic Biomass: State of the Art. Polymers 2013, 5, 600–642. [Google Scholar] [CrossRef]
- Pauly, M.; Keegstra, K. Plant cell wall polymers as precursors for biofuels. Curr. Opin. Plant Biol. 2010, 13, 305–312. [Google Scholar] [CrossRef]
- Zheng, Y.; Zhao, J.; Xu, F.; Li, Y. Pretreatment of lignocellulosic biomass for enhanced biogas production. Prog. Energy Combust. Sci. 2014, 42, 35–53. [Google Scholar] [CrossRef]
- Siqueira, G.; Bras, J.; Dufresne, A. Cellulosic Bionanocomposites: A Review of Preparation, Properties and Applications. Polymers 2010, 2, 728–765. [Google Scholar] [CrossRef]
- Singhvi, M.S.; Chaudhari, S.; Gokhale, D.V. Lignocellulose processing: A current challenge. RSC Adv. 2014, 4, 8271–8277. [Google Scholar] [CrossRef]
- Fernandes, E.M.; Pires, R.A.; Mano, J.F.; Reis, R.L. Bionanocomposites from lignocellulosic resources: Properties, applications and future trends for their use in the biomedical field. Prog. Polym. Sci. 2013, 38, 1415–1441. [Google Scholar] [CrossRef]
- Thakur, V.K.; Thakur, M.K. Processing and characterization of natural cellulose fibers/thermoset polymer composites. Carbohydr. Polym. 2014, 109, 102–117. [Google Scholar] [CrossRef] [PubMed]
- Miao, C.; Hamad, W.Y. Cellulose reinforced polymer composites and nanocomposites: A critical review. Cellulose 2013, 20, 2221–2262. [Google Scholar] [CrossRef]
- Eichhorn, S.J.; Dufresne, A.; Aranguren, M.; Marcovich, N.E.; Capadona, J.R.; Rowan, S.J.; Weder, C.; Thielemans, W.; Roman, M.; Renneckar, S.; et al. Review: Current international research into cellulose nanofibres and nanocomposites. J. Mater. Sci. 2010, 45, 1–33. [Google Scholar] [CrossRef]
- Gupta, V.K.; Carrott, P.J.; Singh, R.; Chaudhary, M.; Kushwaha, S. Cellulose: A review as natural, modified and activated carbon adsorbent. Bioresour. Technol. 2016, 216, 1066–1076. [Google Scholar] [CrossRef]
- Wang, H.; Gurau, G.; Rogers, R.D. Ionic liquid processing of cellulose. Chem. Soc. Rev. 2012, 41, 1519–1537. [Google Scholar] [CrossRef]
- Wang, S.; Lu, A.; Zhang, L. Recent advances in regenerated cellulose materials. Prog. Polym. Sci. 2016, 53, 169–206. [Google Scholar] [CrossRef]
- Oliva, C.; Huang, W.; El Badri, S.; Lee, M.A.L.; Ronholm, J.; Chen, L.; Wang, Y. Concentrated sulfuric acid aqueous solution enables rapid recycling of cellulose from waste paper into antimicrobial packaging. Carbohydr. Polym. 2020, 241. [Google Scholar] [CrossRef]
- Sun, L.; Yang, S.; Qian, X.; An, X. High-efficacy and long term antibacterial cellulose material: Anchored guanidine polymer via double “click chemistry”. Cellulose 2020, 27, 8799–8812. [Google Scholar] [CrossRef]
- Guna, V.; Ilangovan, M.; Hu, C.; Nagananda, G.S.; Ananthaprasad, M.G.; Venkatesh, K.; Reddy, N. Antimicrobial Natural Cellulose Fibers from Hyptis suaveolens for Potential Biomedical and Textiles Applications. J. Natl. Fibers 2019, 1–10. [Google Scholar] [CrossRef]
- Onofrei, M.D.; Dobos, A.M.; Dunca, S.; Ioanid, E.G.; Ioan, S. Biocidal activity of cellulose materials for medical implants. J. Appl. Polym. Sci. 2015, 132. [Google Scholar] [CrossRef]
- Li, S.M.; Dong, Y.Y.; Ma, M.G.; Fu, L.H.; Sun, R.C.; Xu, F. Hydrothermal synthesis, characterization, and bactericidal activities of hybrid from cellulose and TiO(2). Carbohydr. Polym. 2013, 96, 15–20. [Google Scholar] [CrossRef]
- Yadav, C.; Maji, P.K. Synergistic effect of cellulose nanofibres and bio- extracts for fabricating high strength sodium alginate based composite bio-sponges with antibacterial properties. Carbohydr. Polym. 2019, 203, 396–408. [Google Scholar] [CrossRef]
- Ilangovan, M.; Guna, V.; Hu, C.; Nagananda, G.S.; Reddy, N. Curcuma longa L. plant residue as a source for natural cellulose fibers with antimicrobial activity. Ind. Crops Prod. 2018, 112, 556–560. [Google Scholar] [CrossRef]
- Jacob, J.; Haponiuk, J.; Thomas, S.; Peter, G.; Gopi, S. Use of Ginger Nanofibers for the Preparation of Cellulose Nanocomposites and Their Antimicrobial Activities. Fibers 2018, 6, 79. [Google Scholar] [CrossRef]
- Gogoi, B.; Barua, S.; Sarmah, J.K.; Karak, N. In situ synthesis of a microbial fouling resistant, nanofibrillar cellulose-hyperbranched epoxy composite for advanced coating applications. Prog. Organ. Coat. 2018, 124, 224–231. [Google Scholar] [CrossRef]
- Gabov, K.; Oja, T.; Deguchi, T.; Fallarero, A.; Fardim, P. Preparation, characterization and antimicrobial application of hybrid cellulose-lignin beads. Cellulose 2016, 24, 641–658. [Google Scholar] [CrossRef]
- George, D.; Maheswari, P.U.; Sheriffa Begum, K.M.M.; Arthanareeswaran, G. Biomass-Derived Dialdehyde Cellulose Cross-linked Chitosan-Based Nanocomposite Hydrogel with Phytosynthesized Zinc Oxide Nanoparticles for Enhanced Curcumin Delivery and Bioactivity. J. Agric. Food Chem. 2019, 67, 10880–10890. [Google Scholar] [CrossRef]
- Anagha, B.; George, D.; Maheswari, P.U.; Begum, K.M.M.S. Biomass Derived Antimicrobial Hybrid Cellulose Hydrogel with Green ZnO Nanoparticles for Curcumin Delivery and its Kinetic Modelling. J. Polym. Environ. 2019, 27, 2054–2067. [Google Scholar] [CrossRef]
- Ahmad, N.; Tayyeb, D.; Ali, I.; KAlruwaili, N.; Ahmad, W.; Khan, A.H.; Iqbal, M.S. Development and Characterization of Hemicellulose-Based Films for Antibacterial Wound-Dressing Application. Polymers 2020, 12, 548. [Google Scholar] [CrossRef] [PubMed]
- Bouaziz, F.; Koubaa, M.; Ellouz Ghorbel, R.; Ellouz Chaabouni, S. Biological properties of water-soluble polysaccharides and hemicelluloses from almond gum. Int. J. Biol. Macromol. 2017, 95, 667–674. [Google Scholar] [CrossRef] [PubMed]
- Dominguez-Robles, J.; Larraneta, E.; Fong, M.L.; Martin, N.K.; Irwin, N.J.; Mutje, P.; Tarres, Q.; Delgado-Aguilar, M. Lignin/poly(butylene succinate) composites with antioxidant and antibacterial properties for potential biomedical applications. Int. J. Biol. Macromol. 2020, 145, 92–99. [Google Scholar] [CrossRef]
- Gordobil, O.; Herrera, R.; Yahyaoui, M.; İlk, S.; Kaya, M.; Labidi, J. Potential use of kraft and organosolv lignins as a natural additive for healthcare products. RSC Adv. 2018, 8, 24525–24533. [Google Scholar] [CrossRef]
- Shen, X.; Berton, P.; Shamshina, J.L.; Rogers, R.D. Preparation and comparison of bulk and membrane hydrogels based on Kraft- and ionic-liquid-isolated lignins. Green Chem. 2016, 18, 5607–5620. [Google Scholar] [CrossRef]
- Aadil, K.R.; Prajapati, D.; Jha, H. Improvement of physcio-chemical and functional properties of alginate film by Acacia lignin. Food Packag. Shelf Life 2016, 10, 25–33. [Google Scholar] [CrossRef]
- García, A.; Spigno, G.; Labidi, J. Antioxidant and biocide behaviour of lignin fractions from apple tree pruning residues. Ind. Crops Prod. 2017, 104, 242–252. [Google Scholar] [CrossRef]
- Sunthornvarabhas, J.; Rungthaworn, P.; Sukatta, U.; Juntratip, N.; Sriroth, K. Antimicrobial Tendency of Bagasse Lignin Extracts by Raman Peak Intensity. Sugar Tech 2020, 22, 697–705. [Google Scholar] [CrossRef]
- Kaur, R.; Uppal, S.K.; Sharma, P. Antioxidant and Antibacterial Activities of Sugarcane Bagasse Lignin and Chemically Modified Lignins. Sugar Tech 2017, 19, 675–680. [Google Scholar] [CrossRef]
- Sunthornvarabhas, J.; Liengprayoon, S.; Suwonsichon, T. Antimicrobial kinetic activities of lignin from sugarcane bagasse for textile product. Ind. Crops Prod. 2017, 109, 857–861. [Google Scholar] [CrossRef]
- Dong, X.; Dong, M.; Lu, Y.; Turley, A.; Jin, T.; Wu, C. Antimicrobial and antioxidant activities of lignin from residue of corn stover to ethanol production. Ind. Crops Prod. 2011, 34, 1629–1634. [Google Scholar] [CrossRef]
- Wang, G.; Xia, Y.; Liang, B.; Sui, W.; Si, C. Successive ethanol-water fractionation of enzymatic hydrolysis lignin to concentrate its antimicrobial activity. J. Chem. Technol. Biotechnol. 2018, 93, 2977–2987. [Google Scholar] [CrossRef]
- Juikar, S.J.; Nadanathangam, V. Microbial Production of Nanolignin from Cotton Stalks and Its Application onto Cotton and Linen Fabrics for Multifunctional Properties. Waste Biomass Valorization 2019, 11, 6073–6083. [Google Scholar] [CrossRef]
- Wang, G.; Pang, T.; Xia, Y.; Liu, X.; Li, S.; Parvez, A.M.; Kong, F.; Si, C. Subdivision of bamboo kraft lignin by one-step ethanol fractionation to enhance its water-solubility and antibacterial performance. Int. J. Biol. Macromol. 2019, 133, 156–164. [Google Scholar] [CrossRef] [PubMed]
- Lefatshe, K.; Muiva, C.M.; Kebaabetswe, L.P. Extraction of nanocellulose and in-situ casting of ZnO/cellulose nanocomposite with enhanced photocatalytic and antibacterial activity. Carbohydr. Polym. 2017, 164, 301–308. [Google Scholar] [CrossRef]
- Li, S.-M.; Fu, L.-H.; Ma, M.-G.; Zhu, J.-F.; Sun, R.-C.; Xu, F. Simultaneous microwave-assisted synthesis, characterization, thermal stability, and antimicrobial activity of cellulose/AgCl nanocomposites. Biomass Bioenergy 2012, 47, 516–521. [Google Scholar] [CrossRef]
- Fan, L.; Zhang, H.; Gao, M.; Zhang, M.; Liu, P.; Liu, X. Cellulose nanocrystals/silver nanoparticles: In-situ preparation and application in PVA films. Holzforschung 2020, 74, 523–528. [Google Scholar] [CrossRef]
- Liu, H.; Song, J.; Shang, S.; Song, Z.; Wang, D. Cellulose nanocrystal/silver nanoparticle composites as bifunctional nanofillers within waterborne polyurethane. ACS Appl. Mater. Interfaces 2012, 4, 2413–2419. [Google Scholar] [CrossRef]
- Girio, F.M.; Fonseca, C.; Carvalheiro, F.; Duarte, L.C.; Marques, S.; Bogel-Lukasik, R. Hemicelluloses for fuel ethanol: A review. Bioresour. Technol. 2010, 101, 4775–4800. [Google Scholar] [CrossRef] [PubMed]
- Scheller, H.V.; Ulvskov, P. Hemicelluloses. Annu. Rev. Plant Biol. 2010, 61, 263–289. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Dai, G.; Yang, H.; Luo, Z. Lignocellulosic biomass pyrolysis mechanism: A state-of-the-art review. Prog. Energy Combust. Sci. 2017, 62, 33–86. [Google Scholar] [CrossRef]
- Zhou, X.; Li, W.; Mabon, R.; Broadbelt, L.J. A Critical Review on Hemicellulose Pyrolysis. Energy Technol. 2017, 5, 52–79. [Google Scholar] [CrossRef]
- Sella Kapu, N.; Trajano, H.L. Review of hemicellulose hydrolysis in softwoods and bamboo. Biofuels Bioprod. Biorefining 2014, 8, 857–870. [Google Scholar] [CrossRef]
- Fu, G.-Q.; Zhang, S.-C.; Chen, G.-G.; Hao, X.; Bian, J.; Peng, F. Xylan-based hydrogels for potential skin care application. Int. J. Biol. Macromol. 2020, 158, 244–250. [Google Scholar] [CrossRef] [PubMed]
- Arellano-Sandoval, L.; Delgado, E.; Camacho-Villegas, T.A.; Bravo-Madrigal, J.; Manríquez-González, R.; Lugo-Fabres, P.H.; Toriz, G.; García-Uriostegui, L. Development of thermosensitive hybrid hydrogels based on xylan-type hemicellulose from agave bagasse: Characterization and antibacterial activity. MRS Commun. 2020, 10, 147–154. [Google Scholar] [CrossRef]
- Adler, E. Lignin Chemistry-Past, Present and Future. Wood Sci. Technol. 1977, 11, 169–218. [Google Scholar] [CrossRef]
- Erfani Jazi, M.; Narayanan, G.; Aghabozorgi, F.; Farajidizaji, B.; Aghaei, A.; Kamyabi, M.A.; Navarathna, C.M.; Mlsna, T.E. Structure, chemistry and physicochemistry of lignin for material functionalization. SN Appl. Sci. 2019, 1, 1–19. [Google Scholar] [CrossRef]
- Laurichesse, S.; Avérous, L. Chemical modification of lignins: Towards biobased polymers. Prog. Polym. Sci. 2014, 39, 1266–1290. [Google Scholar] [CrossRef]
- Schutyser, W.; Renders, T.; Van den Bosch, S.; Koelewijn, S.F.; Beckham, G.T.; Sels, B.F. Chemicals from lignin: An interplay of lignocellulose fractionation, depolymerisation, and upgrading. Chem. Soc. Rev. 2018, 47, 852–908. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Pu, Y.; Ragauskas, A.; Yang, B. From lignin to valuable products-strategies, challenges, and prospects. Bioresour. Technol. 2019, 271, 449–461. [Google Scholar] [CrossRef]
- Tobimatsu, Y.; Schuetz, M. Lignin polymerization: How do plants manage the chemistry so well? Curr. Opin. Biotechnol. 2019, 56, 75–81. [Google Scholar] [CrossRef] [PubMed]
- Thakur, V.K.; Thakur, M.K.; Raghavan, P.; Kessler, M.R. Progress in Green Polymer Composites from Lignin for Multifunctional Applications: A Review. ACS Sustain. Chem. Eng. 2014, 2, 1072–1092. [Google Scholar] [CrossRef]
- Liu, Q.; Luo, L.; Zheng, L. Lignins: Biosynthesis and Biological Functions in Plants. Int. J. Mol. Sci. 2018, 19, 335. [Google Scholar] [CrossRef]
- Chio, C.; Sain, M.; Qin, W. Lignin utilization: A review of lignin depolymerization from various aspects. Renew. Sustain. Energy Rev. 2019, 107, 232–249. [Google Scholar] [CrossRef]
- Windeisen, E.; Wegener, G. Lignin as Building Unit for Polymers. In Polymer Science: A Comprehensive Reference; Elsevier: Amsterdam, The Netherlands, 2012; Volume 10, pp. 255–265. [Google Scholar]
- Bhatia, S.K.; Jagtap, S.S.; Bedekar, A.A.; Bhatia, R.K.; Patel, A.K.; Pant, D.; Rajesh Banu, J.; Rao, C.V.; Kim, Y.G.; Yang, Y.H. Recent developments in pretreatment technologies on lignocellulosic biomass: Effect of key parameters, technological improvements, and challenges. Bioresour. Technol. 2020, 300. [Google Scholar] [CrossRef] [PubMed]
- Collins, M.N.; Nechifor, M.; Tanasa, F.; Zanoaga, M.; McLoughlin, A.; Strozyk, M.A.; Culebras, M.; Teaca, C.A. Valorization of lignin in polymer and composite systems for advanced engineering applications—A review. Int. J. Biol. Macromol. 2019, 131, 828–849. [Google Scholar] [CrossRef]
- Aadil, K.R.; Pandey, N.; Mussatto, S.I.; Jha, H. Green synthesis of silver nanoparticles using acacia lignin, their cytotoxicity, catalytic, metal ion sensing capability and antibacterial activity. J. Environ. Chem. Eng. 2019, 7, 103296. [Google Scholar] [CrossRef]
- Chandna, S.; Thakur, N.S.; Reddy, Y.N.; Kaur, R.; Bhaumik, J. Engineering Lignin Stabilized Bimetallic Nanocomplexes: Structure, Mechanistic Elucidation, Antioxidant, and Antimicrobial Potential. ACS Biomater. Sci. Eng. 2019, 5, 3212–3227. [Google Scholar] [CrossRef]
- Lee, E.; Song, Y.; Lee, S. Crosslinking of lignin/poly(vinyl alcohol) nanocomposite fiber webs and their antimicrobial and ultraviolet-protective properties. Text. Res. J. 2017, 89, 3–12. [Google Scholar] [CrossRef]
- Mehta, M.J.; Kumar, A. Ionic Liquid Stabilized Gelatin-Lignin Films: A Potential UV-Shielding Material with Excellent Mechanical and Antimicrobial Properties. Chemistry 2019, 25, 1269–1274. [Google Scholar] [CrossRef]
- Hao, L.C.; Sapuan, S.M.; Hassan, M.R.; Sheltami, R.M. Natural fiber reinforced vinyl polymer composites. In Natural Fibre Reinforced Vinyl Ester and Vinyl Polymer Composites; Woodhead Publishing: Cambridge, UK, 2018; pp. 27–70. [Google Scholar] [CrossRef]
- Sanjay, M.R.; Siengchin, S.; Parameswaranpillai, J.; Jawaid, M.; Pruncu, C.I.; Khan, A. A comprehensive review of techniques for natural fibers as reinforcement in composites: Preparation, processing and characterization. Carbohydr. Polym. 2019, 207, 108–121. [Google Scholar] [CrossRef]
- Gurunathan, T.; Mohanty, S.; Nayak, S.K. A review of the recent developments in biocomposites based on natural fibres and their application perspectives. Compos. Part A Appl. Sci. Manuf. 2015, 77, 1–25. [Google Scholar] [CrossRef]
- Yang, J.; Ching, Y.C.; Chuah, C.H. Applications of Lignocellulosic Fibers and Lignin in Bioplastics: A Review. Polymers 2019, 11, 751. [Google Scholar] [CrossRef]
- Ramesh, M.; Palanikumar, K.; Reddy, K.H. Plant fibre based bio-composites: Sustainable and renewable green materials. Renew. Sustain. Energy Rev. 2017, 79, 558–584. [Google Scholar] [CrossRef]
- Faruk, O.; Bledzki, A.K.; Fink, H.-P.; Sain, M. Progress Report on Natural Fiber Reinforced Composites. Macromol. Mater. Eng. 2014, 299, 9–26. [Google Scholar] [CrossRef]
- Mochane, M.J.; Mokhena, T.C.; Mokhothu, T.H.; Mtibe, A.; Sadiku, E.R.; Ray, S.S.; Ibrahim, I.D.; Daramola, O.O. Recent progress on natural fiber hybrid composites for advanced applications: A review. Express Polym. Lett. 2019, 13, 159–198. [Google Scholar] [CrossRef]
- Cai, J.; He, Y.; Yu, X.; Banks, S.W.; Yang, Y.; Zhang, X.; Yu, Y.; Liu, R.; Bridgwater, A.V. Review of physicochemical properties and analytical characterization of lignocellulosic biomass. Renew. Sustain. Energy Rev. 2017, 76, 309–322. [Google Scholar] [CrossRef]
- Silva, S.P.; Sabino, M.A.; Fernandes, E.M.; Correlo, V.M.; Boesel, L.F.; Reis, R.L. Cork: Properties, capabilities and applications. Int. Mater. Rev. 2013, 50, 345–365. [Google Scholar] [CrossRef]
- Sarasini, F.; Fiore, V. A systematic literature review on less common natural fibres and their biocomposites. J. Clean. Prod. 2018, 195, 240–267. [Google Scholar] [CrossRef]
- Guna, V.; Ilangovan, M.; Adithya, K.; Srinivas, C.V.; Yogesh, S.; Nagananda, G.S.; Venkatesh, K.; Reddy, N. Biofibers and biocomposites from sabai grass: A unique renewable resource. Carbohydr. Polym. 2019, 218, 243–249. [Google Scholar] [CrossRef]
- Kaur, R.; Uppal, S.K. Structural characterization and antioxidant activity of lignin from sugarcane bagasse. Colloid Polym. Sci. 2015, 293, 2585–2592. [Google Scholar] [CrossRef]
- Munir, M.T.; Aviat, F.; Pailhories, H.; Eveillard, M.; Irle, M.; Federighi, M.; Belloncle, C. Direct screening method to assess antimicrobial behavior of untreated wood. Eur. J. Wood Wood Prod. 2019, 77, 319–322. [Google Scholar] [CrossRef]
- Feng, J.; Dong, P.; Li, R.; Li, C.; Xie, X.; Shi, Q. Effects of wood fiber properties on mold resistance of wood polypropylene composites. Int. Biodeterior. Biodegrad. 2019, 140, 152–159. [Google Scholar] [CrossRef]
- Jamili, F.; Mirjalili, M.; Zamani, H.A. Antibacterial wood-plastic composite produced from treated and natural dyed wood fibers. Polym. Polym. Compos. 2019, 27, 347–355. [Google Scholar] [CrossRef]
- Wu, C.-S.; Liao, H.-T. Fabrication, characterization, and application of polyester/wood flour composites. J. Polym. Eng. 2017, 37, 689–698. [Google Scholar] [CrossRef]
- Musiol, M.; Jurczyk, S.; Sobota, M.; Klim, M.; Sikorska, W.; Zieba, M.; Janeczek, H.; Rydz, J.; Kurcok, P.; Johnston, B.; et al. (Bio)Degradable Polymeric Materials for Sustainable Future-Part 3: Degradation Studies of the PHA/Wood Flour-Based Composites and Preliminary Tests of Antimicrobial Activity. Materials 2020, 13, 2200. [Google Scholar] [CrossRef] [PubMed]
- Prapruddivongs, C.; Sombatsompop, N. Roles and evidence of wood flour as an antibacterial promoter for triclosan-filled poly(lactic acid). Compos. Part B Eng. 2012, 43, 2730–2737. [Google Scholar] [CrossRef]
- Varghese, S.A.; Pulikkalparambil, H.; Rangappa, S.M.; Siengchin, S.; Parameswaranpillai, J. Novel biodegradable polymer films based on poly(3-hydroxybutyrate-co-3-hydroxyvalerate) and Ceiba pentandra natural fibers for packaging applications. Food Packag. Shelf Life 2020, 25, 100538. [Google Scholar] [CrossRef]
- Treinyte, J.; Bridziuviene, D.; Fataraite-Urboniene, E.; Rainosalo, E.; Rajan, R.; Cesoniene, L.; Grazuleviciene, V. Forestry wastes filled polymer composites for agricultural use. J. Clean. Prod. 2018, 205, 388–406. [Google Scholar] [CrossRef]
- Kalinoski, R.M.; Shi, J. Hydrogels derived from lignocellulosic compounds: Evaluation of the compositional, structural, mechanical and antimicrobial properties. Ind. Crops Prod. 2019, 128, 323–330. [Google Scholar] [CrossRef]
- Gonçalves, F.; Correia, P.; Silva, S.P.; Almeida-Aguiar, C. Evaluation of antimicrobial properties of cork. FEMS Microbiol. Lett. 2016, 363, fnv231. [Google Scholar] [CrossRef]
- Francesko, A.; Blandón, L.; Vázquez, M.; Petkova, P.; Morato, J.; Pfeifer, A.; Heinze, T.; Mendoza, E.; Tzanov, T. Enzymatic Functionalization of Cork Surface with Antimicrobial Hybrid Biopolymer/Silver Nanoparticles. ACS Appl. Mater. Interfaces 2015, 7, 9792–9799. [Google Scholar] [CrossRef]
- Ketema, A.; Worku, A. Antibacterial Finishing of Cotton Fabric Using Stinging Nettle (Urtica dioica L.) Plant Leaf Extract. J. Chem. 2020, 2020, 4049273. [Google Scholar] [CrossRef]
- Li, Q.; Ibrahim, L.; Zhou, W.; Zhang, M.; Fernando, G.F.; Wang, L.; Yuan, Z. Holistic solution to natural fiber deterioration in cement composite using hybrid treatments. Cellulose 2019, 27, 981–989. [Google Scholar] [CrossRef]
- Thakur, K.; Kalia, S.; Pathania, D.; Kumar, A.; Sharma, N.; Schauer, C.L. Surface functionalization of lignin constituent of coconut fibers via laccase-catalyzed biografting for development of antibacterial and hydrophobic properties. J. Clean. Prod. 2016, 113, 176–182. [Google Scholar] [CrossRef]
- Lazić, B.D.; Pejić, B.M.; Kramar, A.D.; Vukčević, M.M.; Mihajlovski, K.R.; Rusmirović, J.D.; Kostić, M.M. Influence of hemicelluloses and lignin content on structure and sorption properties of flax fibers (Linum usitatissimum L.). Cellulose 2017, 25, 697–709. [Google Scholar] [CrossRef]
- Spiridon, I.; Anghel, N.C.; Darie-Nita, R.N.; Iwanczuk, A.; Ursu, R.G.; Spiridon, I.A. New composites based on starch/Ecoflex(R)/biomass wastes: Mechanical, thermal, morphological and antimicrobial properties. Int. J. Biol. Macromol. 2020, 156, 1435–1444. [Google Scholar] [CrossRef] [PubMed]
- Torres-Giner, S.; Hilliou, L.; Melendez-Rodriguez, B.; Figueroa-Lopez, K.J.; Madalena, D.; Cabedo, L.; Covas, J.A.; Vicente, A.A.; Lagaron, J.M. Melt processability, characterization, and antibacterial activity of compression-molded green composite sheets made of poly(3-hydroxybutyrate-co-3-hydroxyvalerate) reinforced with coconut fibers impregnated with oregano essential oil. Food Packag. Shelf Life 2018, 17, 39–49. [Google Scholar] [CrossRef]
- Batista, P.H.D.; Byrski, D.; Lamping, M.; Romandini, R. IP-Based Incentives against Antimicrobial Crisis: A European Perspective. IIC Int. Rev. Intellect. Prop. Compet. Law 2019, 50, 30–76. [Google Scholar] [CrossRef]
- Jin, F.; Jing, F.; Xiaomo, H.; Qingshan, S. Antibacterial Wood-Plastic Composite and Preparation Method Thereof. CN Patent CN104893331A, 2015. [Google Scholar]
- Mingzhi, H.; Jiao, L.; Dan, W.; Shiyong, W.; Ming, Y.; Yueying, Z. Novel Antibacterial Wood-Plastic Board and Preparation Method Thereof. CN Patent CN106752049A, 2016. [Google Scholar]
- Wang, G.; Tian, G.; Yu, Y.; Song, Y.; Jiang, Z. Natural Lignocellulose Material with Modified Zinc Oxide and Preparation Method Thereof. CN Patent CN101659751A, 2009. [Google Scholar]
- Junhua, Z.; Xingli, Z. Wood-Plastic Composite Material for Enhancing Heat Conductivity with Carbon Nanofiber and Preparation Method Thereof. CN Patent CN108841188A, 2018. [Google Scholar]
- Fuming, J. Wood-Plastic Flooring and Manufacturing Method Thereof. CN Patent CN105350741A, 2015. [Google Scholar]
- Xiaozhong, F.; Hongying, J.; Fengkui, L.; Xiaoxiang, W.; Maofang, W.; Lei, X. Wood-Plastic Floor. CN Patent CN106183293A, 2016. [Google Scholar]
- Jianhua, D.; Anxiang, H.; Ning, J.; Dan, L.; Lifen, L.; Xun, L.; Jian, L.; Nengming, L.; Ming, L.; Jianlin, L.; et al. Preparation Method for Anticorrosive Antibacterial Wood Fiber Composite. CN Patent CN109731747A, 2018. [Google Scholar]
- Preparation Technology for Antibacterial Environmental-Friendly Wood-Plastic Composite Material. CN Patent CN108789762A, 2018.
- Carol, H.; Andreas, K.; Petri, W. Lignocellulosic substrates with enhanced antibacterial properties and method for obtaining those. EP Patent EP2199046A1, 2008. [Google Scholar]
- Guangkui, J.; Deying, L.; Dengyun, T.; Shouliang, W.; Zhibing, Z.; Decheng, Z. Antibacterial Impregnation Treatment Equipment for Wooden Floor and Process Thereof. CN Patent CN108724381A, 2018. [Google Scholar]
- Ding, L.; Yilin, T.; Shuangcheng, W.; Ripeng, X.; Jinzhu, Z. Functional Regenerated Cellulose Fiber and Preparation Method and Application Thereof. CN Patent CN105506765A, 2015. [Google Scholar]
- Michel, D.; Armand, L. Process for the Preparation of Lignin Based Polyurethane Products. CN Patent CN105637036A, 2013. [Google Scholar]
- Qing, C.; Hang, Z. Lignocellulose-Ellagic Acid Bio-Plastic Food Packaging Film and Preparation Method. CN Patent CN107934198A, 2017. [Google Scholar]
- Zhang, Y.S.; Khademhosseini, A. Advances in engineering hydrogels. Science 2017, 356. [Google Scholar] [CrossRef]
- Junqui, C.; Jinhao, H.; Weifeng, L.; Xueqing, Q.; Dongjie, Y.; Xiao, Z.; Junqi, C.; Jinhao, H.; Weifeng, L.; Xueqing, Q.; et al. High-Strength Lignin/Polyvinyl Alcohol Composite Antibacterial Aquagel and Preparation Method. CN Patent CN110240774A, 2019. [Google Scholar]
- Melita, D.; Burkhard, F.; Michael, M.; Fugmann, B.; Dietze, M.; Mager, M. Biomedical Foam Articles. AU Patent AU2007236166A1, 2007. [Google Scholar]
- Liuyun, J.; Ye, L.; Shuting, T.; Chengdong, X.; Shengpei, S.; Yuzhu, J. Bamboo fiber/hydroxyapatite/polylactic Acid Ternary Combined Degradable Porous Material and Preparing Method Thereof. CN Patent CN106075601A, 2016. [Google Scholar]
- Haojie, D.; Liuyun, J.; Bingli, M. Bamboo Fiber/Nanometer Apatite Composite Material and Preparation Method Thereof. CN Patent CN108607116A, 2018. [Google Scholar]
| Compound | Origin | Antimicrobial Activity Tested Against | Application | Ref |
|---|---|---|---|---|
| Cellulose | Wood | E. coli, S. aureus | Packaging | [27] |
| E. coli, S. aureus | [35] | |||
| E. coli, P. aeruginosa, B. subtilis | Tissue engineering, wound dressing | [31] | ||
| Sugarcane Bagasse | S.aureus, T. rubrum | Skin infective | [36] | |
| Wastewater purification | [37] | |||
| Tulsi | E. coli, S. aureus, B. cereus, Ser. marcescens | Biomedical | [28] | |
| Ginger | E. coli, S. aureus, B.cereus, Sal. thyphimirium | Packaging, wound dressing, surgical material | [33] | |
| Hemicellulose | Plantago Ovata seed husk | E. coli, S. aureus, P. aeruginosa | Wound dressing, drug delivery | [38] |
| Almond gum | Actinomycetes sp, Sal. thyphimirium, K. pneumonia, L. monocytogenes, S. aureus, Sal. enterica, P. aeruginosa, B. thuringiensis, B. subtilis | Food and non-food | [39] | |
| Lignin | Softwood | S. aureus | Biomedical | [40] |
| Eucalyptus | A. niger E. coli, S. aureus, Pr. microbilis, Pr. vulgaris, P. aeruginosa, Entero. aerogenes, B. thuringiensis, Sal. enterica serotype typhmurium and Strept. mutans | Antimicrobial additive or agent in food, textile, or chemical industry | [41] | |
| Spruce | [41] | |||
| Poplar | E. coli | Drug delivery, food packaging, wound dressing, | [42] | |
| Acacia | E. coli, S. aureus | Active packaging | [43] | |
| Apple tree pruning residues | A. niger, Sacch. cerevisiae | Food antioxidant | [44] | |
| Sugarcane Bagasse | E. coli, S. aureus, P. aeruginosa, S. epidermidis | [45] | ||
| B. aryabhattai, Klebsiella sp. | Natural antibacterial agent | [46] | ||
| S. epidermidis | Antimicrobial textile | [47] | ||
| Corn | L. monocytogenes, S. aureus, E. coli, Sal. enteritidis, C. lipolytica | Antioxidant and antimicrobial | [48] | |
| E. coli, S. aureus, B. subtilis, Sal. enterica | Natural antibacterial agent | [49] | ||
| Cotton stalks | S. aureus, K. pneumoniae | Medical and technical textiles | [50] | |
| Bamboo | E. coli, S. aureus, B. subtilis, Sal. enterica | Natural antibacterial agent | [51] |
| Fiber | Cellulose (%) | Hemicellulose (%) | Lignin (%) | Ref | |
|---|---|---|---|---|---|
| Wood | Softwood (Pine) | 45.0–50.0 | 25.0–35.0 | 25.0–35.0 | [86] |
| Hardwood (Poplar) | 50.8–53.3 | 26.2–28.7 | 15.5–16.3 | [86] | |
| Non-wood | Apple tree pruning | 75.81 | 7.84 | 4.03 | [44] |
| Bamboo | 30.60 | 17.00 | 3.41 | [51] | |
| Cork | 6–25 | 13–26 | [87] | ||
| Cotton | 82.7–92 | 5.7–6 | 0 | [88] | |
| Flax | 71–81 | 18.6–20.6 | 2.2–3 | [88] | |
| Hemp | 70.2–74.4 | 17.9–22.4 | 3.7–5.7 | [88] | |
| Pineapple | 70–82 | 15–19 | 5–12 | [88] | |
| Sabai Grass | 42.9 | 21.1 | 18.5 | [89] | |
| Sisal | 56.5–78 | 5.6–16.5 | 8–14 | [88] | |
| Sugarcane Bagasse | 42.11 | 28.42 | 19.29 | [90] | |
| Publication Number | Title | Priority Year | Ref |
|---|---|---|---|
| Wood-polymer composites (WPC) | |||
| CN104893331A | Antibacterial wood–plastic composite and preparation method thereof | 2015 | [109] |
| CN106752049A | Novel antibacterial wood–plastic board and preparation method thereof | 2016 | [110] |
| CN101659751A | Natural lignocellulose material with modified zinc oxide and preparation method thereof | 2009 | [111] |
| CN108841188A | Wood–plastic composite material for enhancing heat conductivity with carbon nanofiber and preparation method thereof | 2018 | [112] |
| CN105350741A | Wood–plastic flooring and manufacturing method therefor | 2015 | [113] |
| CN106183293A | Wood–plastic floor | 2016 | [114] |
| CN109731747A | Preparation method for anticorrosive antibacterial wood fiber composite | 2018 | [115] |
| CN108789762A | Preparation technology for antibacterial environmentally friendly wood–plastic composite material | 2018 | [116] |
| Other uses claiming antibacterial property | |||
| EP2199046A1 | Lignocellulosic substrates with enhanced antibacterial properties and method for obtaining those | 2008 | [117] |
| CN108724381A | Antibacterial impregnation treatment equipment for wooden floor and process thereof | 2018 | [118] |
| CN105506765A | Functional regenerated cellulose fiber and preparation method and application thereof | 2015 | [119] |
| CN105637036A | Process for the preparation of lignin based polyurethane products | 2013 | [120] |
| CN107934198A | Lignocellulose-ellagic acid bio-plastic food packaging film and preparation method | 2017 | [121] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lobo, F.C.M.; Franco, A.R.; Fernandes, E.M.; Reis, R.L. An Overview of the Antimicrobial Properties of Lignocellulosic Materials. Molecules 2021, 26, 1749. https://doi.org/10.3390/molecules26061749
Lobo FCM, Franco AR, Fernandes EM, Reis RL. An Overview of the Antimicrobial Properties of Lignocellulosic Materials. Molecules. 2021; 26(6):1749. https://doi.org/10.3390/molecules26061749
Chicago/Turabian StyleLobo, Flávia C. M., Albina R. Franco, Emanuel M. Fernandes, and Rui L. Reis. 2021. "An Overview of the Antimicrobial Properties of Lignocellulosic Materials" Molecules 26, no. 6: 1749. https://doi.org/10.3390/molecules26061749
APA StyleLobo, F. C. M., Franco, A. R., Fernandes, E. M., & Reis, R. L. (2021). An Overview of the Antimicrobial Properties of Lignocellulosic Materials. Molecules, 26(6), 1749. https://doi.org/10.3390/molecules26061749