Synthesis of Piperidine Nucleosides as Conformationally Restricted Immucillin Mimics
Abstract
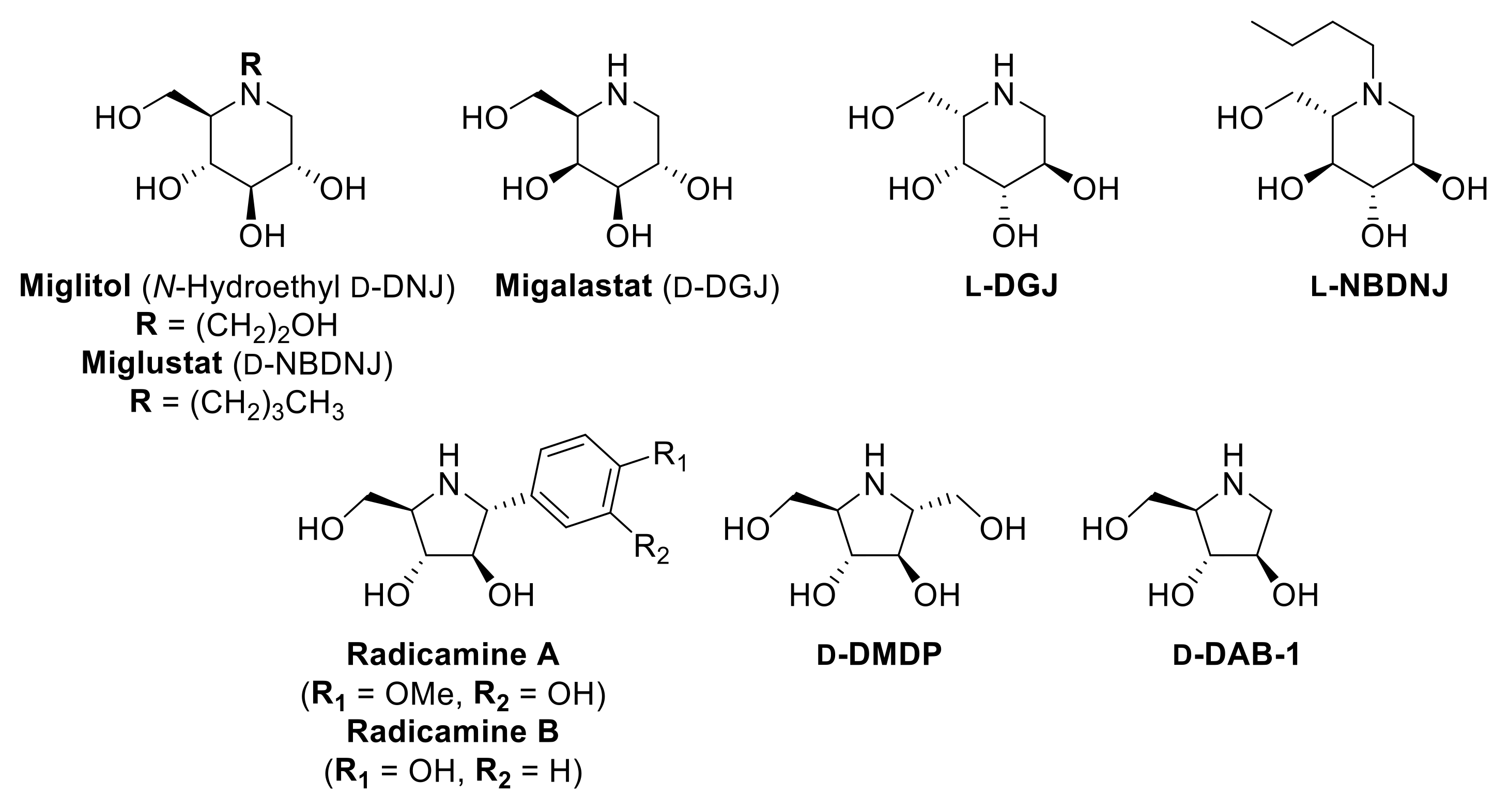
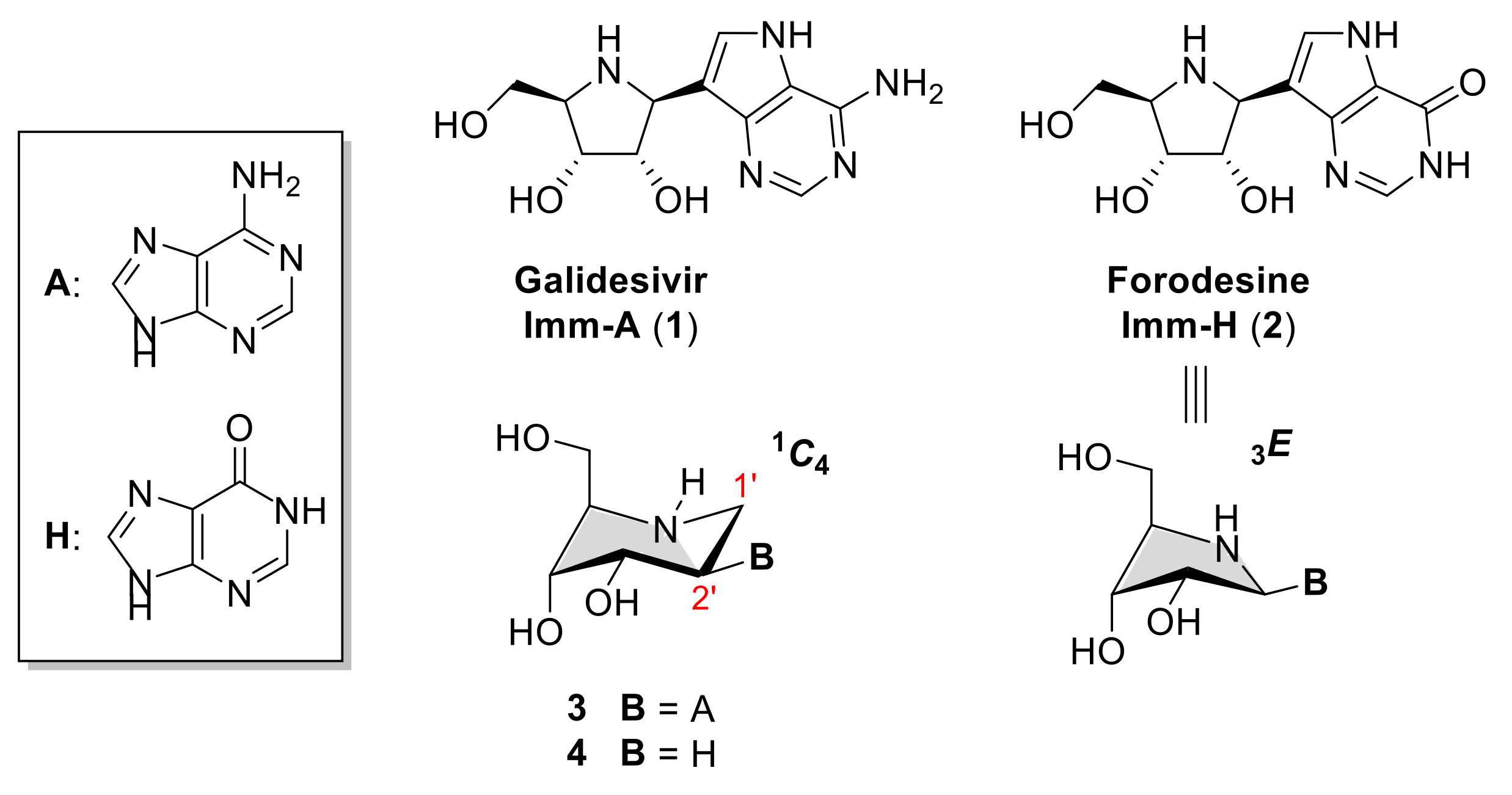
1. Introduction
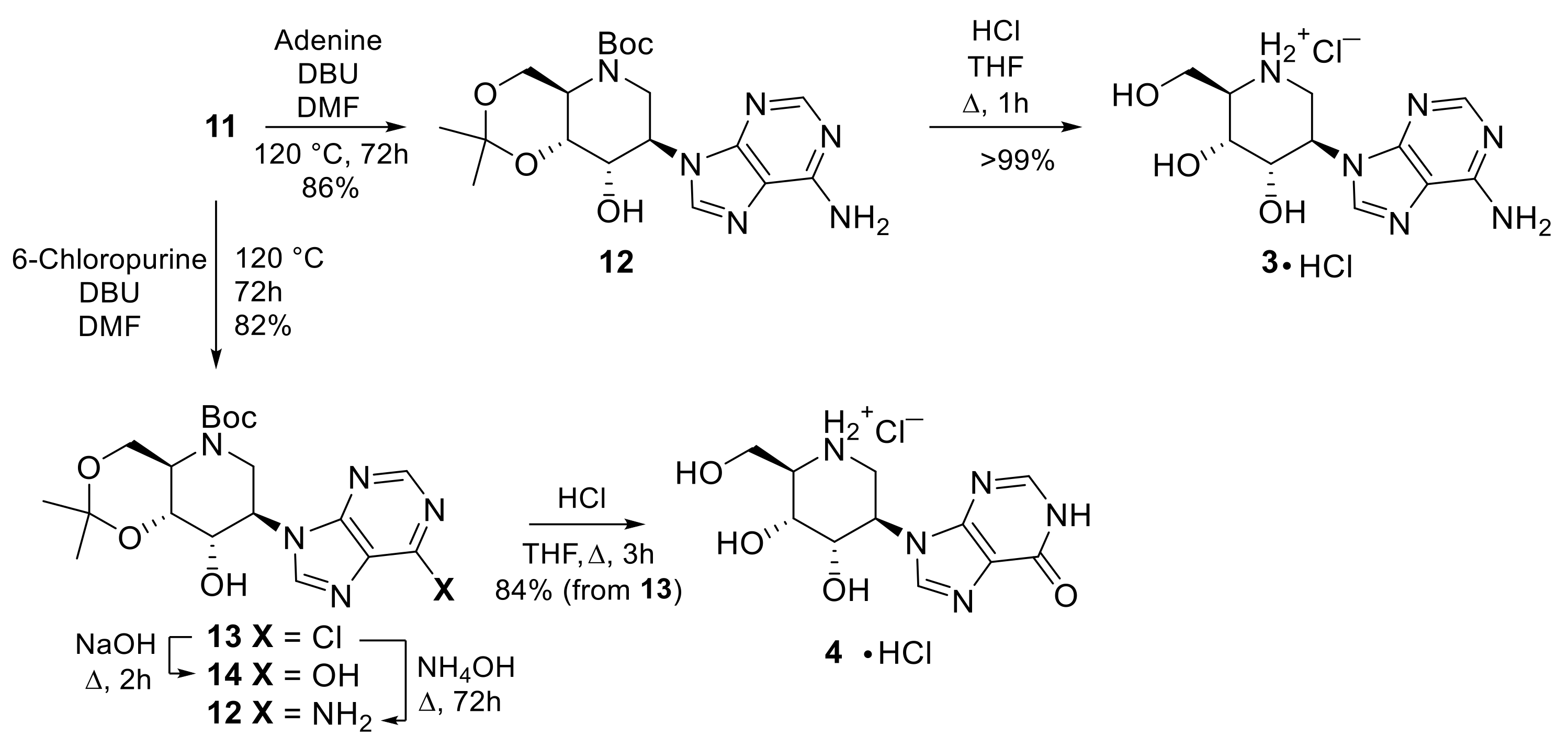
2. Results and Discussion
3. Materials and Methods
3.1. Chemistry
3.2. Procedures for the Synthesis of 3–13
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Varki, A.; Cummings, R.D.; Esko, J.D.; Stanley, P.; Hart, G.W.; Aebi, M.; Darvill, A.G.; Kinoshita, T.; Packer, N.H.; Prestegard, J.H.; et al. Essentials of Glycobiology, 3rd ed.; Cold Spring Harbor Laboratory Press: Cold Spring Harbor, NY, USA, 2009. [Google Scholar]
- Gloster, T.M.; Vocadlo, D.J. Developing inhibitors of glycan processing enzymes as tools for enabling glycobiology. Nat. Chem. Biol. 2012, 8, 683–694. [Google Scholar] [CrossRef] [PubMed]
- Stütz, A.E.; Wrodnigg, T.M. Carbohydrate-processing enzymes of the lysosome: Diseases caused by misfolded mutants and sugar mimetics as correcting pharmacological chaperones. Adv. Carbohydr. Chem. Biochem. 2016, 73, 225–302. [Google Scholar] [PubMed]
- Compain, P.; Martin, O.R. Iminosugars: From Synthesis to Therapeutic Applications, 1st ed.; John Wiley & Sons, Ltd: Chichester, UK, 2007. [Google Scholar]
- Horne, G.; Wilson, F.X.; Tinsley, J.; Williams, D.H.; Storer, R. Iminosugars past, present and future: Medicines for tomorrow. Drug Discov. Today 2011, 16, 107–118. [Google Scholar] [CrossRef]
- Chao, L.; Jongkees, S. High-Throughput approaches in carbohydrate-active enzymology: Glycosidase and glycosyl transferase inhibitors, evolution, and discovery. Angew. Chem. Int. Ed. 2019, 58, 12750–12760. [Google Scholar] [CrossRef] [PubMed]
- Nash, R.J.; Kato, A.; Yu, C.-Y.; Fleet, G.W. Iminosugars as therapeutic agents: Recent advances and promising trends. Future Med. Chem. 2011, 3, 1513–1521. [Google Scholar] [CrossRef]
- Alonzi, D.S.; Scott, K.A.; Dwek, R.A.; Zitzmann, N. Iminosugar antivirals: The therapeutic sweet spot. Biochem. Soc. Trans. 2017, 45, 571–582. [Google Scholar] [CrossRef]
- Evans DeWald, L.; Starr, C.; Butters, T.; Treston, A.; Warfield, K.L. Iminosugars: A host-targeted approach to combat Flaviviridae infections. Antiviral Res. 2020, 184, 104881. [Google Scholar] [CrossRef] [PubMed]
- Wrodnigg, T.; Steiner, A.; Ueberbacher, B. Natural and synthetic iminosugars as carbohydrate processing enzyme inhibitors for cancer therapy. Anticancer. Agents Med. Chem. 2008, 8, 77–85. [Google Scholar] [CrossRef] [PubMed]
- Derosa, G.; Maffioli, P. Mini-special issue paper management of diabetic patients with hypoglycemic agents α-glucosidase inhibitors and their use in clinical practice. Arch. Med. Sci. 2012, 5, 899–906. [Google Scholar] [CrossRef]
- Platt, F.M.; D’Azzo, A.; Davidson, B.L.; Neufeld, E.F.; Tifft, C.J. Lysosomal storage diseases. Nat. Rev. Dis. Prim. 2018, 4, 27. [Google Scholar] [CrossRef]
- Cheviet, T.; Lefebvre-Tournier, I.; Wein, S.; Peyrottes, S. Plasmodium purine metabolism and its inhibition by nucleoside and nucleotide analogues. J. Med. Chem. 2019, 62, 8365–8391. [Google Scholar] [CrossRef]
- D’Alonzo, D.; Guaragna, A.; Palumbo, G. Glycomimetics at the mirror: Medicinal chemistry of l-iminosugars. Curr. Med. Chem. 2009, 16, 473–505. [Google Scholar] [CrossRef]
- Gloster, T.M.; Davies, G.J. Glycosidase inhibition: Assessing mimicry of the transition state. Org. Biomol. Chem. 2010, 8, 305–320. [Google Scholar] [CrossRef]
- Stütz, A.E.; Paulsen, H. Iminosugars as Glycosidase Inhibitors; Wiley-VCH Verlag GmbH: Hoboken, NJ, USA, 1998. [Google Scholar]
- Kumar Thakur, A.; Kumar, Y.; K Goyal, K. Pharmacotherapeutics of miglitol: An α-glucosidase inhibitor. J. Anal. Pharm. Res. 2018, 7, 617–619. [Google Scholar] [CrossRef]
- Hollak, C.E.M.; Hughes, D.; van Schaik, I.N.; Schwierin, B.; Bembi, B. Miglustat (Zavesca®) in type 1 Gaucher disease: 5-year results of a post-authorisation safety surveillance programme. Pharmacoepidemiol. Drug Saf. 2009, 18, 770–777. [Google Scholar] [CrossRef]
- Patterson, M.C.; Mengel, E.; Vanier, M.T.; Moneuse, P.; Rosenberg, D.; Pineda, M. Treatment outcomes following continuous miglustat therapy in patients with Niemann-Pick disease Type C: A final report of the NPC Registry. Orphanet J. Rare Dis. 2020, 15, 104. [Google Scholar] [CrossRef] [PubMed]
- Riccio, E.; Zanfardino, M.; Ferreri, L.; Santoro, C.; Cocozza, S.; Capuano, I.; Imbriaco, M.; Feriozzi, S.; Pisani, A. Switch from enzyme replacement therapy to oral chaperone migalastat for treating Fabry disease: Real-life data. Eur. J. Hum. Genet. 2020, 28, 1662–1668. [Google Scholar] [CrossRef] [PubMed]
- Jenkinson, S.F.; Fleet, G.W.J.; Nash, R.J.; Koike, Y.; Adachi, I.; Yoshihara, A.; Morimoto, K.; Izumori, K.; Kato, A. Looking-glass synergistic pharmacological chaperones: DGJ and L-DGJ from the enantiomers of tagatose. Org. Lett. 2011, 13, 4064–4067. [Google Scholar] [CrossRef] [PubMed]
- D’Alonzo, D.; De Fenza, M.; Porto, C.; Iacono, R.; Huebecker, M.; Cobucci-Ponzano, B.; Priestman, D.A.; Platt, F.; Parenti, G.; Moracci, M.; et al. N-Butyl-l-deoxynojirimycin (l-NBDNJ): Synthesis of an allosteric enhancer of α-glucosidase activity for the treatment of Pompe disease. J. Med. Chem. 2017, 60. [Google Scholar] [CrossRef] [PubMed]
- De Fenza, M.; D’Alonzo, D.; Esposito, A.; Munari, S.; Loberto, N.; Santangelo, A.; Lampronti, I.; Tamanini, A.; Rossi, A.; Ranucci, S.; et al. Exploring the effect of chirality on the therapeutic potential of N-alkyl-deoxyiminosugars: Anti-inflammatory response to Pseudomonas aeruginosa infections for application in CF lung disease. Eur. J. Med. Chem. 2019, 175, 63–71. [Google Scholar] [CrossRef] [PubMed]
- Esposito, A.; D’Alonzo, D.; De Fenza, M.; De Gregorio, E.; Tamanini, A.; Lippi, G.; Dechecchi, M.C.; Guaragna, A. Synthesis and therapeutic applications of iminosugars in cystic fibrosis. Int. J. Mol. Sci. 2020, 21, 3353. [Google Scholar] [CrossRef] [PubMed]
- Shibano, M.; Tsukamoto, D.; Masuda, A.; Tanka, Y.; Kusano, G. Two new pyrrolidine alkaloids, radicamines A and B, as inhibitors of alpha-glucosidase from lobelia chinensis lour. Chem. Pharm. Bull. 2001, 49, 1362–1365. [Google Scholar] [CrossRef] [PubMed]
- Haarr, M.B.; Lopéz, Ó.; Pejov, L.; Fernández-Bolaños, J.G.; Lindbäck, E.; Sydnes, M.O. 1,4-Dideoxy-1,4-imino-d-arabinitol (DAB) analogues possessing a hydrazide imide moiety as potent and selective α-mannosidase inhibitors. ACS Omega 2020, 5, 18507–18514. [Google Scholar] [CrossRef] [PubMed]
- Asano, N.; Oseki, K.; Kizu, H.; Matsui, K. Nitrogen-in-the-ring pyranoses and furanoses: Structural basis of inhibition of mammalian glycosidases. J. Med. Chem. 1994, 37, 3701–3706. [Google Scholar] [CrossRef]
- Evans, G.; Schramm, V.; Tyler, P. The immucillins: Design, synthesis and application of transition- state analogues. Curr. Med. Chem. 2015, 22. [Google Scholar] [CrossRef]
- Evans, G.B.; Tyler, P.C.; Schramm, V.L. Immucillins in infectious diseases. ACS Infect. Dis. 2018, 4, 107–117. [Google Scholar] [CrossRef]
- Lim, S.-Y.; Osuna, C.E.; Best, K.; Taylor, R.; Chen, E.; Yoon, G.; Kublin, J.L.; Schalk, D.; Schultz-Darken, N.; Capuano, S.; et al. A direct-acting antiviral drug abrogates viremia in Zika virus-infected rhesus macaques. Sci. Transl. Med. 2020, 12, eaau9135. [Google Scholar] [CrossRef]
- Warren, T.K.; Wells, J.; Panchal, R.G.; Stuthman, K.S.; Garza, N.L.; Van Tongeren, S.A.; Dong, L.; Retterer, C.J.; Eaton, B.P.; Pegoraro, G.; et al. Protection against filovirus diseases by a novel broad-spectrum nucleoside analogue BCX4430. Nature 2014, 508, 402–405. [Google Scholar] [CrossRef]
- Elfiky, A.A. Ribavirin, Remdesivir, Sofosbuvir, Galidesivir, and Tenofovir against SARS-CoV-2 RNA dependent RNA polymerase (RdRp): A molecular docking study. Life Sci. 2020, 253, 117592. [Google Scholar] [CrossRef]
- A Study to Evaluate the Safety, Pharmacokinetics and Antiviral Effects of Galidesivir in Yellow Fever or COVID-19; last update: July 23, 2020. Start Date: April 2020. Available online: https://clinicaltrials.gov/ct2/show/NCT03891420 (accessed on 17 November 2020).
- Kicska, G.A.; Long, L.; Horig, H.; Fairchild, C.; Tyler, P.C.; Furneaux, R.H.; Schramm, V.L.; Kaufman, H.L. Immucillin H, a powerful transition-state analog inhibitor of purine nucleoside phosphorylase, selectively inhibits human T lymphocytes. Proc. Natl. Acad. Sci. USA 2001, 98, 4593–4598. [Google Scholar] [CrossRef] [PubMed]
- Evans, G.B.; Schramm, V.L.; Tyler, P.C. The transition to magic bullets—transition state analogue drug design. Medchemcomm 2018, 9, 1983–1993. [Google Scholar] [CrossRef]
- Gandhi, V.; Kilpatrick, J.M.; Plunkett, W.; Ayres, M.; Harman, L.; Du, M.; Bantia, S.; Davisson, J.; Wierda, W.G.; Faderl, S.; et al. A proof-of-principle pharmacokinetic, pharmacodynamic, and clinical study with purine nucleoside phosphorylase inhibitor immucillin-H (BCX-1777, forodesine). Blood 2005, 106, 4253–4260. [Google Scholar] [CrossRef]
- Ho, M.-C.; Shi, W.; Rinaldo-Matthis, A.; Tyler, P.C.; Evans, G.B.; Clinch, K.; Almo, S.C.; Schramm, V.L. Four generations of transition-state analogues for human purine nucleoside phosphorylase. Proc. Natl. Acad. Sci. USA 2010, 107, 4805–4812. [Google Scholar] [CrossRef] [PubMed]
- Le, B.T.; Chen, S.; Abramov, M.; Herdewijn, P.; Veedu, R.N. Evaluation of anhydrohexitol nucleic acid, cyclohexenyl nucleic acid and D-altritol nucleic acid-modified 2′-O-methyl RNA mixmer antisense oligonucleotides for exon skipping in vitro. Chem. Commun. 2016, 52, 13467–13470. [Google Scholar] [CrossRef]
- De Fenza, M.; Eremeeva, E.; Troisi, R.; Yang, H.; Esposito, A.; Sica, F.; Herdewijn, P.; D’Alonzo, D.; Guaragna, A. Structure—activity relationship study of a potent α-thrombin binding aptamer incorporating hexitol nucleotides. Chem. A Eur. J. 2020, 26, 9589–9597. [Google Scholar] [CrossRef]
- Herdewijn, P. Nucleic acids with a six-membered ‘carbohydrate’ mimic in the backbone. Chem. Biodivers. 2010, 7, 1–59. [Google Scholar] [CrossRef] [PubMed]
- D’Alonzo, D.; Amato, J.; Schepers, G.; Froeyen, M.; Van Aerschot, A.; Herdewijn, P.; Guaragna, A. Enantiomeric selection properties of β-homoDNA: Enhanced pairing for heterochiral complexes. Angew. Chem. Int. Ed. 2013, 52, 6662–6665. [Google Scholar] [CrossRef]
- Paolella, C.; D’Alonzo, D.; Schepers, G.; Van Aerschot, A.; Di Fabio, G.; Palumbo, G.; Herdewijn, P.; Guaragna, A. Oligonucleotides containing a ribo-configured cyclohexanyl nucleoside: Probing the role of sugar conformation in base pairing selectivity. Org. Biomol. Chem. 2015, 13, 10041–10049. [Google Scholar] [CrossRef] [PubMed]
- D’Alonzo, D.; Froeyen, M.; Schepers, G.; Di Fabio, G.; Van Aerschot, A.; Herdewijn, P.; Palumbo, G.; Guaragna, A. 1′,5′-Anhydro-l-ribo-hexitol adenine nucleic acids (α-L-HNA-A): Synthesis and chiral selection properties in the mirror image world. J. Org. Chem. 2015, 80, 5014–5022. [Google Scholar] [CrossRef]
- Esposito, A.; Giovanni, C.; De Fenza, M.; Talarico, G.; Chino, M.; Palumbo, G.; Guaragna, A.; D’Alonzo, D. A stereoconvergent Tsuji—Trost reaction in the synthesis of cyclohexenyl nucleosides. Chem. -Eur. J. 2020, 26, 2597–2601. [Google Scholar] [CrossRef]
- Oliviero, G.; Amato, J.; Borbone, N.; D’Errico, S.; Piccialli, G.; Mayol, L. Synthesis of N-1 and ribose modified inosine analogues on solid support. Tetrahedron Lett. 2007, 48, 397–400. [Google Scholar] [CrossRef]
- Oliviero, G.; Amato, J.; Borbone, N.; D’Errico, S.; Piccialli, G.; Bucci, E.; Piccialli, V.; Mayol, L. Synthesis of 4-N-alkyl and ribose-modified AICAR analogues on solid support. Tetrahedron 2008, 64, 6475–6481. [Google Scholar] [CrossRef]
- Guaragna, A.; D’Errico, S.; D’Alonzo, D.; Pedatella, S.; Palumbo, G. A general approach to the synthesis of 1-deoxy-l-iminosugars. Org. Lett. 2007, 9, 3473–3476. [Google Scholar] [CrossRef]
- Böshagen, H.; Heiker, F.-R.; Matthias Schüller, A. The chemistry of the 1-deoxynojirimycin system. Synthesis of 2-acetamido-1,2-dideoxynojirimycin from 1-deoxynojirimycin. Carbohydr. Res. 1987, 164, 141–148. [Google Scholar] [CrossRef]
- Pedatella, S.; Guaragna, A.; D’Alonzo, D.; De Nisco, M.; Palumbo, G. Triphenylphosphine polymer-bound/iodine complex: A suitable reagent for the preparation of O-isopropylidene sugar derivatives. Synthesis 2006, 305–308. [Google Scholar] [CrossRef]
- Guaragna, A.; Amoresano, A.; Pinto, V.; Monti, G.; Mastrobuoni, G.; Marino, G. Synthesis and proteomic activity evaluation of a new isotope-coded affinity tagging (ICAT) reagent. Bioconjug. Chem. 2008, 19, 1095–1104. [Google Scholar] [CrossRef]
- Esposito, A.; De Gregorio, E.; De Fenza, M.; D’Alonzo, D.; Satawani, A.; Guaragna, A. Expeditious synthesis and preliminary antimicrobial activity of deflazacort and its precursors. RSC Adv. 2019, 9, 21519–21524. [Google Scholar] [CrossRef]
- De Gregorio, E.; Esposito, A.; Vollaro, A.; De Fenza, M.; D’Alonzo, D.; Migliaccio, A.; Iula, V.D.; Zarrilli, R.; Guaragna, A. N-Nonyloxypentyl-l-deoxynojirimycin inhibits growth, biofilm formation and virulence factors expression of Staphylococcus aureus. Antibiotics 2020, 9, 362. [Google Scholar] [CrossRef] [PubMed]
- Esposito, A.; D’Alonzo, D.; D’Errico, S.; De Gregorio, E.; Guaragna, A. Toward the identification of novel antimicrobial agents: One-pot synthesis of lipophilic conjugates of N-alkyl d- and l-iminosugars. Mar. Drugs 2020, 18, 572. [Google Scholar] [CrossRef]
- Moussa, Z.; Judeh, Z.M.A.; Ahmed, S.A. Polymer-supported triphenylphosphine: Application in organic synthesis and organometallic reactions. RSC Adv. 2019, 9, 35217–35272. [Google Scholar] [CrossRef]
- D’Alonzo, D.; Guaragna, A.; Palumbo, G. Exploring the role of chirality in nucleic acid recognition. Chem. Biodivers. 2011, 8, 373–413. [Google Scholar] [CrossRef] [PubMed]
- Maurinsh, Y.; Rosemeyer, H.; Esnouf, R.; Medvedovici, A.; Wang, J.; Ceulemans, G.; Lescrinier, E.; Hendrix, C.; Busson, R.; Sandra, P.; et al. Synthesis and pairing properties of oligonucleotides containing 3-hydroxy-4-hydroxymethyl-1-cyclohexanyl nucleosides. Chem. Eur. J. 1999, 5, 2139–2150. [Google Scholar] [CrossRef]
- Rengasamy, R.H.; Curtis-Long, M.J.; Ryu, H.W.; Oh, K.Y.; Park, K.H. Stereoselective synthesis of l-deoxyaltronojirimycin from l-serine. Bull. Korean Chem. Soc. 2009, 30, 1531–1534. [Google Scholar] [CrossRef][Green Version]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
De Fenza, M.; Esposito, A.; D’Alonzo, D.; Guaragna, A. Synthesis of Piperidine Nucleosides as Conformationally Restricted Immucillin Mimics. Molecules 2021, 26, 1652. https://doi.org/10.3390/molecules26061652
De Fenza M, Esposito A, D’Alonzo D, Guaragna A. Synthesis of Piperidine Nucleosides as Conformationally Restricted Immucillin Mimics. Molecules. 2021; 26(6):1652. https://doi.org/10.3390/molecules26061652
Chicago/Turabian StyleDe Fenza, Maria, Anna Esposito, Daniele D’Alonzo, and Annalisa Guaragna. 2021. "Synthesis of Piperidine Nucleosides as Conformationally Restricted Immucillin Mimics" Molecules 26, no. 6: 1652. https://doi.org/10.3390/molecules26061652
APA StyleDe Fenza, M., Esposito, A., D’Alonzo, D., & Guaragna, A. (2021). Synthesis of Piperidine Nucleosides as Conformationally Restricted Immucillin Mimics. Molecules, 26(6), 1652. https://doi.org/10.3390/molecules26061652








