Role of Gut Microbiota, Probiotics and Prebiotics in the Cardiovascular Diseases
Abstract
1. Introduction
2. Cardiovascular Disease: Risk Factors and the Mechanism of Development
3. Gut Microbiota and Its Effects on Human Health
4. Gut Microbiota, Its Diet-Derived Products, and Cardiovascular Diseases
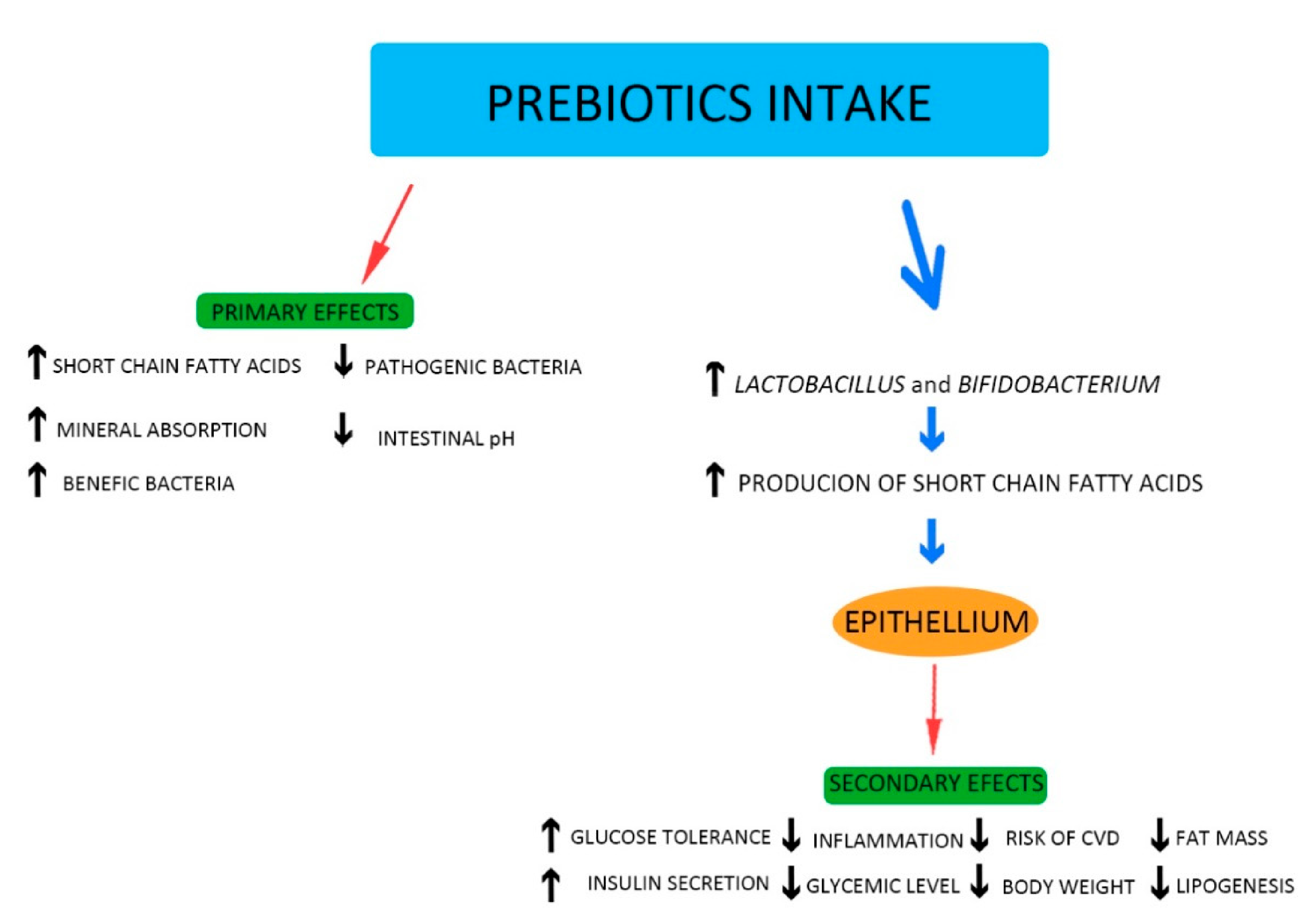
5. Probiotics and Prebiotics: General Information
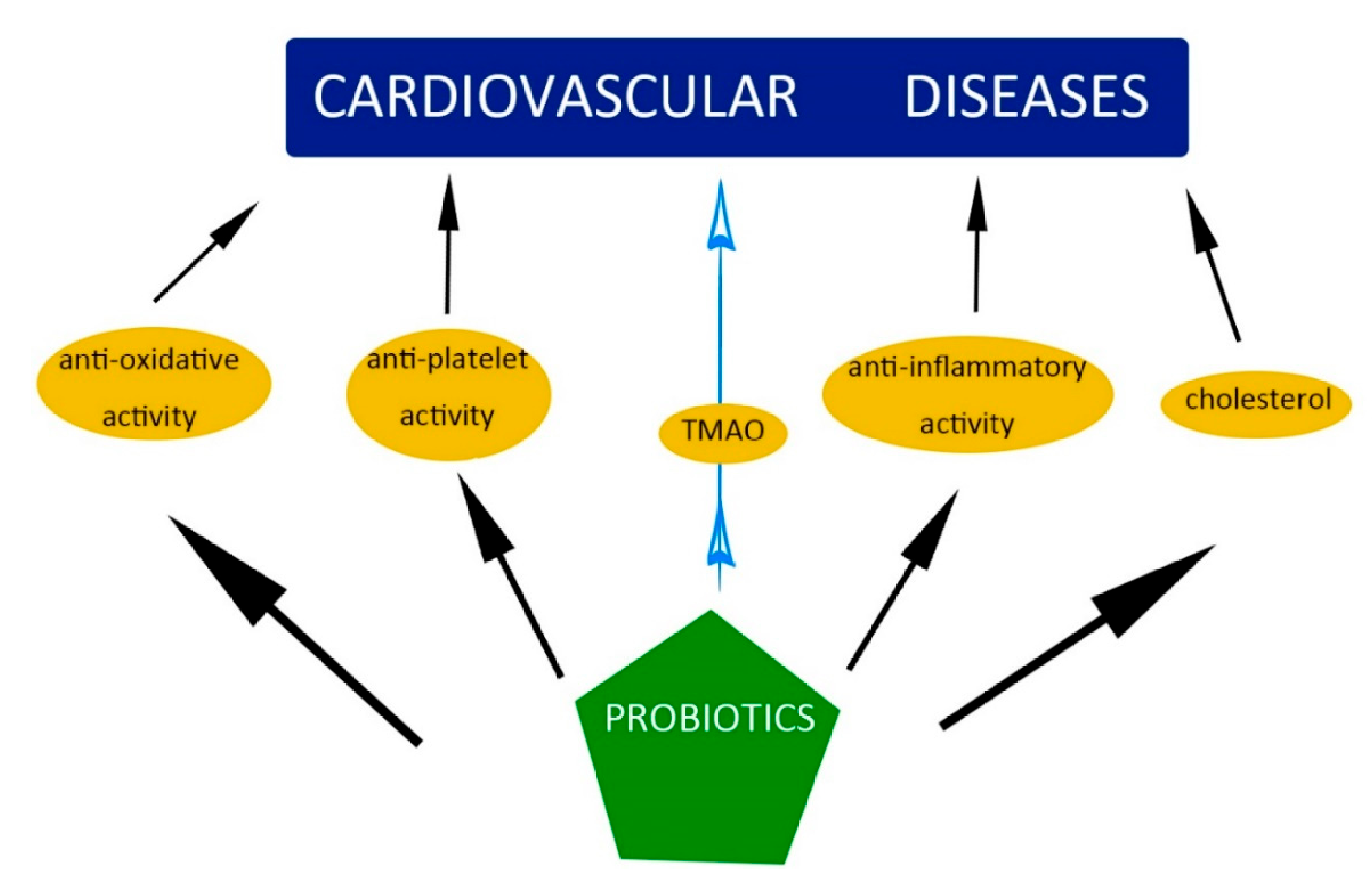
6. The Influence of Probiotics and Prebiotics on the Mechanisms and Factors Causing CVD
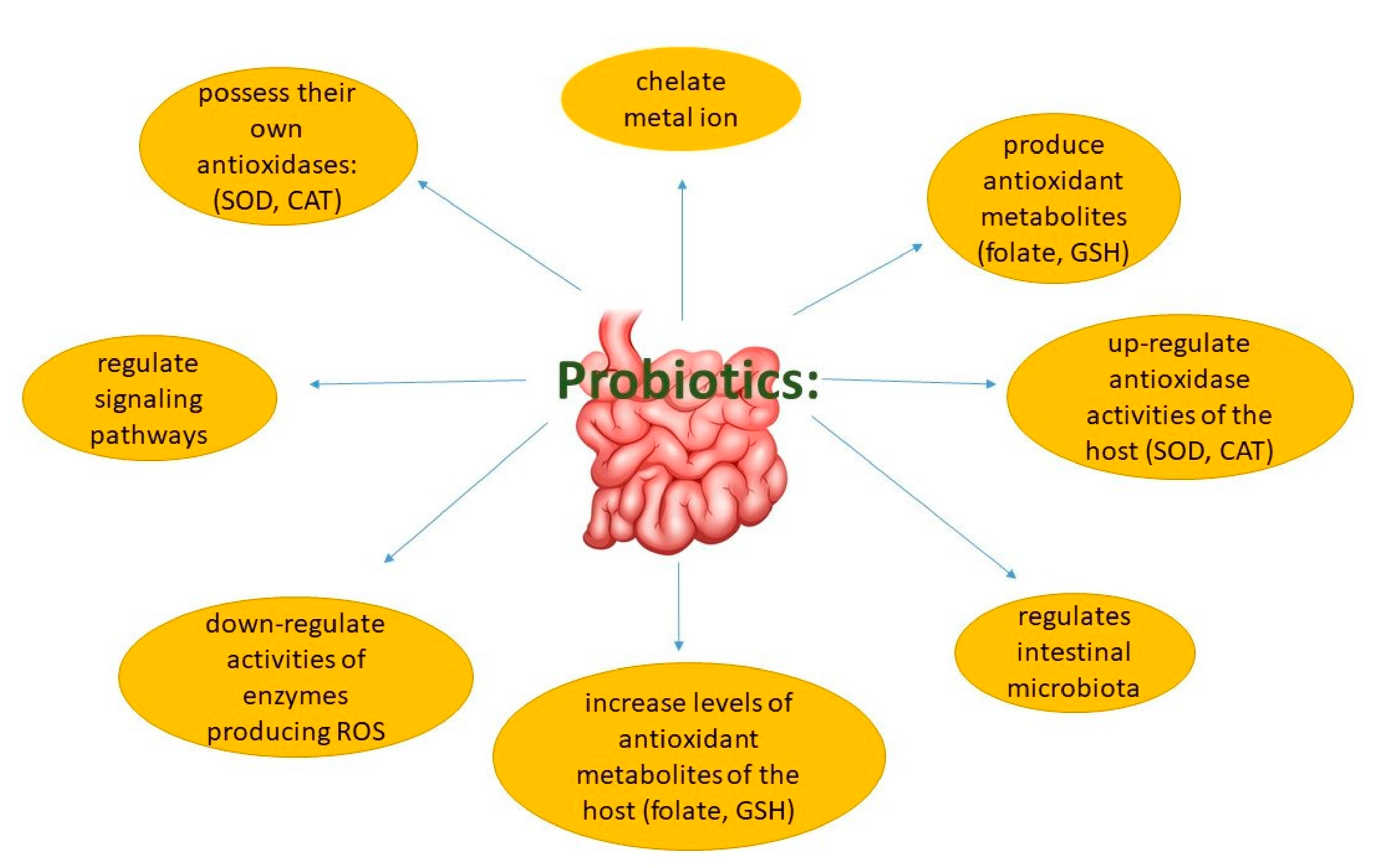
6.1. Oxidative Stress
6.2. Inflammation
6.3. Hypercholesterolemia and High Blood Pressure
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Katsimichas, T.; Antonopoulos, A.S.; Katsimichas, A.; Ohtani, T.; Sakata, Y.; Tousoulis, D. The intestinal microbiota and cardiovascular disease. Cardiovasc. Res. 2019, 115, 1471–1486. [Google Scholar] [CrossRef]
- Attaye, I.; Pinto-Sietsma, S.; Herrema, H.; Nieuwdorp, M. A crucial role for diet in the relationship between gut microbiota and cardiometabolic disease. Annu. Rev. Med. 2020, 71, 149–161. [Google Scholar] [CrossRef] [PubMed]
- Jonsson, A.L.; Bäckhed, F. Role of gut microbiota in atherosclerosis. Nat. Rev. Cardiol. 2017, 14, 79–87. [Google Scholar]
- Tang, W.H.W.; Kitai, T.; Hazen, S.L. Gut microbiota in cardiovascular health and disease. Circ. Res. 2017, 120, 1183–1196. [Google Scholar] [CrossRef] [PubMed]
- Tang, W.H.W.; Bäckhed, F.; Landmesser, U.; Hazen, S.L. Intestinal microbiota in cardiovascular health and disease: JACC state-of-the-art review. J. Am. Coll. Cardiol. 2019, 73, 2089–2105. [Google Scholar] [CrossRef] [PubMed]
- Gerdes, V.; Gueimonde, M.; Pajunen, L.; Nieuwdorp, M.; Laitinen, K. How strong is the evidence that gut microbiota composition can be influenced by lifestyle interventions in a cardio-protective way? Atherosclerosis 2020, 311, 124–142. [Google Scholar] [CrossRef]
- Shanthi, M.; Pekka, P.; Bo, N. Global Atlas on Cardiovascular Diseases Prevention and Control; World Health Organization in collaboration with the World Heart Federation and the World Stroke Organization: Geneva, Switzerland, 2011; pp. 3–18. [Google Scholar]
- Guarner, F.; Malagelada, J.R. Gut flora in health and disease. Lancet 2003, 361, 512–519. [Google Scholar] [CrossRef]
- Olas, B. Probiotics, prebiotics and synbiotics—A promising strategy in prevention and treatment of cardiovascular diseases? Int. J. Mol. Sci. 2020, 21, 9737. [Google Scholar] [CrossRef]
- Xu, C.; Weng, Z.; Zhang, L.; Xu, J.; Dahal, M.; Basnet, T.; Gu, A. HDL cholesterol: A potential mediator of the association between urinary cadmium concentration and cardiovascular disease risk. Ecotoxicol. Environ. Saf. 2021, 208, 111433. [Google Scholar] [CrossRef] [PubMed]
- Kurilenko, N.; Fatkhullina, A.R.; Mazitova, A.; Koltsova, E.K. Act Locally, Act Globally-Microbiota, Barriers, and Cytokines in Atherosclerosis. Cells 2021, 10, 348. [Google Scholar] [CrossRef]
- Paganelli, F.; Mottola, G.; Fromonot, J.; Marlinge, M.; Deharo, P.; Guieu, R.; Ruf, J. Hyperhomocysteinemia and cardiovascular disease: Is the adenosinergic system the missing link? Int. J. Mol. Sci. 2021, 22, 1690. [Google Scholar] [CrossRef]
- Krasi, G.; Precone, V.; Paolacci, S.; Stuppia, L.; Nodari, S.; Romeo, F.; Perrone, M.; Bushati, V.; Dautaj, A.; Bertelli, M. Genetics and pharmacogenetics in the diagnosis and therapy of cardiovascular diseases. Acta Biomed. 2019, 90, 7–19. [Google Scholar] [PubMed]
- Doughty, K.N.; Del Pilar, N.X.; Audette, A.; Katz, D.L. Lifestyle medicine and the management of cardiovascular disease. Curr. Cardiol. Rep. 2017, 19, 116. [Google Scholar] [CrossRef]
- Ma, W.; Zhang, B.; Yang, Y.; Qi, L.; Meng, L.; Zhang, Y.; Huo, Y. Correlating the relationship between interarm systolic bloodpressure and cardiovascular disease risk factors. J. Clin. Hypertens. 2017, 19, 466–471. [Google Scholar] [CrossRef]
- Eckburg, P.B.; Bik, E.M.; Bernstein, C.N.; Purdom, E.; Dethlefsen, L.; Sargent, M.; Gill, S.R.; Nelson, K.E.; Relman, D.A. Diversity of the human intestinal microbial flora. Science 2005, 308, 1635–1638. [Google Scholar] [CrossRef]
- Bäumler, A.J.; Sperandio, V. Interactions between the microbiota and pathogenic bacteria in the gut. Nature 2016, 535, 85–93. [Google Scholar] [CrossRef] [PubMed]
- De Filippo, C.; Cavalieri, D.; Di Paola, M.; Ramazzotti, M.; Poullet, J.B.; Massart, S.; Collini, S.; Pieraccini, G.; Lionetti, P. Impact of diet in shaping gut microbiota revealed by a comparative study in children from Europe and rural Africa. Proc. Natl. Acad. Sci. USA 2010, 107, 14691–14696. [Google Scholar] [CrossRef] [PubMed]
- Gensollen, T.; Iyer, S.S.; Kasper, D.L.; Blumberg, R.S. How colonization by microbiota in early life shapes the immune system. Science 2016, 352, 539–544. [Google Scholar] [CrossRef] [PubMed]
- Katsi, V.; Didagelos, M.; Skevofilax, S.; Armenis, I.; Kartalis, A.; Vlachopoulos, C.; Karvounis, H.; Tousoulis, D. Gut microbiota—Gut dysbiosis—Arterial hypertension: New horizons. Curr. Hyperten. Rev. 2019, 15, 40–46. [Google Scholar] [CrossRef] [PubMed]
- Suárez-Zamorano, N.; Fabbiano, S.; Chevalier, C.; Stojanović, O.; Colin, D.J.; Stevanović, A.; Veyrat-Durebex, C.; Tarallo, V.; Rigo, D.; Germain, S.; et al. Microbiota depletion promotes browning of white adipose tissue and reduces obesity. Nat. Med. 2015, 21, 1497–1501. [Google Scholar] [CrossRef]
- Suganya, K.; Son, T.; Kim, K.W.; Koo, B.S. Impact of gut microbiota: How it could play roles beyond the digestive system on development of cardiovascular and renal diseases. Microb. Pathog. 2020, 104583, in press. [Google Scholar] [CrossRef]
- Arora, T.; Bäckhed, F. The gut microbiota and metabolic disease: Current understanding and future perspectives. J. Intern. Med. 2016, 280, 339–349. [Google Scholar] [CrossRef]
- Wang, Z.; Koonen, D.; Hofker, M.; Fu, J. Gut microbiome and lipid metabolism: From associations to mechanisms. Curr. Opin. Lipidol. 2016, 27, 216–224. [Google Scholar] [CrossRef]
- Trautwein, E.K.; Peters, H.P.F.; Mela, D.J.; Edwards, C.; Herrema, H.; Fud, J.; Geldof, M.; Albers, R. Is gut microbiota a relevant and competitive dietary target for cardiometabolic health? Proceedings of an expert workshop. Trends Food Sci. Technol. 2018, 81, 146–154. [Google Scholar] [CrossRef]
- Levy, M.; Kolodziejczyk, A.A.; Thaiss, C.A.; Elinav, E. Dysbiosis and the immune system. Nat. Rev. Immunol. 2017, 17, 219–232. [Google Scholar] [CrossRef]
- Cui, X.; Ye, L.; Li, J.; Jin, L.; Wang, W.; Li, S.; Bao, M.; Wu, S.; Li, L.; Geng, B.; et al. Metagenomic and metabolomic analyses unveil dysbiosis of gut microbiota in chronic heart failure patients. Sci. Rep. 2018, 8, 635. [Google Scholar] [CrossRef] [PubMed]
- Kummen, M.; Mayerhofer, C.C.K.; Vestad, B.; Broch, K.; Awoyemi, A.; StormLarsen, C.; Ueland, T.; Yndestad, A.; Hov, J.R.; Trøseid, M. Gut microbiota signature in heart failure defined from profiling of 2 independent cohorts. J. Am. Coll. Cardiol. 2018, 71, 1184–1186. [Google Scholar] [CrossRef] [PubMed]
- Sandek, A.; Swidsinski, A.; Schroedl, W.; Watson, A.; Valentova, M.; Herrmann, R.; Scherbakov, N.; Cramer, L.; Rauchhaus, M.; Grosse-Herrenthey, A.; et al. Intestinal blood flow in patients with chronic heart failure: A link with bacterial growth, gastrointestinal symptoms, and cachexia. J. Am. Coll. Cardiol. 2014, 64, 1092–1102. [Google Scholar] [CrossRef]
- Pasini, E.; Aquilani, R.; Testa, C.; Baiardi, P.; Angioletti, S.; Boschi, F.; Verri, M.; Dioguardi, F. Pathogenic gut flora in patients with chronic heart failure. JACC Heart Fail. 2016, 4, 220–227. [Google Scholar] [CrossRef] [PubMed]
- Jie, Z.; Xia, H.; Zhong, S.L.; Feng, Q.; Li, S.; Liang, S.; Zhong, H.; Liu, Z.; Gao, Y.; Zhao, H.; et al. The gut microbiome in atherosclerotic cardiovascular disease. Nat. Commun. 2017, 8, 845. [Google Scholar] [CrossRef]
- Randrianarisoa, E.; Lehn-Stefan, A.; Wang, X.; Hoene, M.; Peter, A.; Heinzmann, S.S.; Zhao, X.; Konigsrainer, V.; Konigsrainer, A.; Balletshofer, B.; et al. Relationship of serum trimethylamine N-oxide (TMAO) levels with early atherosclerosis in humans. Sci. Rep. 2016, 6, 26745. [Google Scholar] [CrossRef]
- Senthong, V.; Wang, Z.; Li, X.S.; Fan, Y.; Wu, Y.; Tang, W.H.W.; Hazen, S.L. Intestinal microbiota-generated metabolite trimethylamine-N-oxide and 5-year mortality risk in stable coronary artery disease: The contributory role of intestinal microbiota in a COURAGE-like patient cohort. J Am. Heart. Assoc. 2016, 5, e002816. [Google Scholar] [CrossRef]
- Roncal, C.; Martínez-Aguilar, E.; Orbe, J.; Ravassa, S.; Fernandez-Montero, A.; Saenz-Pipaon, G.; Ugarte, A.; Estella-Hermoso de Mendoza, A.; Rodriguez, J.A.; Fernandez-Alonso, S.; et al. Trimethylamine-N oxide (TMAO) predicts cardiovascular mortality in peripheral artery disease. Sci. Rep. 2019, 9, 15580. [Google Scholar] [CrossRef] [PubMed]
- Miao-En, Y.; Peng-Da, L.; Xu-Jie, Z.; Lei, W. Trimethylamine-N-oxide has prognostic value in coronary heart disease: A meta-analysis and dose-response analysis. BMC Cardiovasc. Disord. 2020, 20, 6953212. [Google Scholar]
- Haghikia, A.; Li, X.S.; Liman, T.G.; Bledau, N.; Schmidt, D.; Zimmermann, F.; Kränkel, N.; Widera, C.; Sonnenschein, K.; Haghikia, A.; et al. Gut microbiota-dependent trimethylamine N-oxide predicts risk of cardiovascular events in patients with stroke and is related to proinflammatory monocytes. Arterioscler. Thromb. Vasc. Biol. 2018, 38, 2225–2235. [Google Scholar] [CrossRef] [PubMed]
- Marques, F.Z.; Nelson, E.; Chu, P.Y.; Horlock, D.; Fiedler, D.; Ziemann, M.; Tan, J.K.; Kuruppu, S.; Rajapakse, N.W.; El-Osta, A.; et al. High-Fiber diet and acetate supplementation change the gutmicrobiota and prevent the development of hypertension and heart failure in hypertensive mice. Circulation 2017, 135, 964–977. [Google Scholar] [CrossRef]
- Luedde, M.; Winkler, T.; Heinsen, F.A.; Rühlemann, M.C.; Spehlmann, M.E.; Bajrovic, A.; Lieb, W.; Franke, A.; Ott, S.J.; Frey, N. Heart failure is associated with depletion of core intestinal microbiota. ESC Heart Fail 2017, 4, 282–290. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; van Esch, B.C.A.M.; Wagenaar, G.T.M.; Garssen, J.; Folkerts, G.; Henricks, P.A.J. Pro- and anti-inflammatory effects of short chain fatty acids on immune and endothelial cells. Eur. J. Pharmacol. 2018, 831, 52–59. [Google Scholar] [CrossRef]
- Kuipers, F.; Bloks, V.W.; Groen, A.K. Beyond intestinal soap–bile acids in metabolic control. Nat. Rev. Endocrinol. 2014, 10, 488–498. [Google Scholar] [CrossRef]
- Döring, B.; Lütteke, T.; Geyer, J.; Petzinger, E. The SLC10 carrier family: Transport functions and molecular structure. Curr. Top. Membr. 2012, 70, 105–168. [Google Scholar]
- Katsimichas, T.; Ohtani, T.; Motooka, D.; Tsukamoto, Y.; Kioka, H.; Nakamoto, K.; Konishi, S.; Chimura, M.; Sengoku, K.; Miyawaki, H.; et al. Non-Ischemic Heart Failure with Reduced Ejection Fraction Is Associated with Altered Intestinal Microbiota. Circ. J. 2018, 82, 1640–1650. [Google Scholar] [CrossRef]
- Yan, Q.; Gu, Y.; Li, X.; Yang, W.; Jia, L.; Chen, C.; Han, X.; Huang, Y.; Zhao, L.; Li, P.; et al. Alterations of the gut microbiome in hypertension. Front. Cell. Infect. Microbiol. 2017, 7, 381. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Zhao, F.; Wang, Y.; Chen, J.; Tao, J.; Tian, G.; Wu, S.; Liu, W.; Cui, Q.; Geng, B.; et al. Gut microbiota dysbiosis contributes to the development of hypertension. Microbiome 2017, 5, 14. [Google Scholar] [CrossRef]
- Zhu, Q.; Gao, R.; Zhang, Y.; Pan, D.; Zhu, Y.; Zhang, X.; Yang, R.; Jiang, R.; Xu, Y.; Qin, H. Dysbiosis signatures of gut microbiota in coronary artery disease. Physiol. Genom. 2018, 50, 893–903. [Google Scholar] [CrossRef]
- Emoto, T.; Yamashita, T.; Kobayashi, T.; Sasaki, N.; Hirota, Y.; Hayashi, T.; So, A.; Kasahara, K.; Yodoi, K.; Matsumoto, T.; et al. Characterization of gut microbiota profiles in coronary artery disease patients using data mining analysis of terminal restriction fragment length polymorphism: Gut microbiota could be a diagnostic marker of coronary artery disease. Heart Ves. 2017, 32, 39–46. [Google Scholar] [CrossRef]
- Gerritsen, J.; Smidt, H.; Rijkers, G.T.; de Vos, W.M. Intestinal microbiota in human health and disease: The impact of probiotics. Genes Nutr. 2011, 6, 209–240. [Google Scholar] [CrossRef] [PubMed]
- Kothari, D.; Patel, P.; Kim, S.K. Probiotic supplements might not be universally-effective and safe: A review. Biomed. Pharmacoth. 2019, 11, 537–547. [Google Scholar] [CrossRef] [PubMed]
- Zucko, J.; Starcevic, A.; Diminic, J.; Oros, D.; Mortazavian, A.M.; Putnik, P. Probiotic-friend or foe? Curr. Opin. Food Sci. 2020, 32, 45–49. [Google Scholar] [CrossRef]
- Guildelines for the Evaluation of Probiotics in Food; Joint FAO/WHO Working Group on Drafting Guidelines for the Evaluation of Probiotics in Food: London, ON, Canada, 2002.
- Pandey, K.R.; Naik, S.R.; Vakil, B.V. Probiotics, prebiotics and synbiotics-A review. J. Food Sci. Technol. 2010, 52, 7577–7587. [Google Scholar] [CrossRef]
- Nowak, A.; Paliwoda, A.; Blasiak, J. Anti-proliferative, pro-apoptotic and anti-oxidative activity of Lactobacillus and Bifidobacterium strains: A review of mechanism and therapeutic perspectives. Crit. Rev. Food. Sci. Nutr. 2018, 1, 1–12. [Google Scholar] [CrossRef]
- Farias, D.P.; Araújo, F.F.; Neri-Numa, I.A.; Pastore, G.M. Prebiotics: Trends in food, health and technological applications. Trends Food Sci. Technol. 2019, 93, 23–35. [Google Scholar] [CrossRef]
- Mohanty, D.; Misra, S.; Mohapatra, S.; Sahu, P.S. Prebiotics and synbiotics: Recent concepts in nutrition. Food Biosci. 2018, 26, 152–160. [Google Scholar] [CrossRef]
- Quigley, E.M.M. Prebiotics and probiotics in digestive health. Clin. Gastroenterol. Hepatol. 2019, 17, 333–344. [Google Scholar] [CrossRef]
- Pokusaeva, K.; Fitzgerald, G.F.; van Sinderen, D. Carbohydrate metabolism in Bifidobacteria. Genes Nutr. 2011, 6, 285–306. [Google Scholar] [CrossRef] [PubMed]
- Colantonio, A.G.; Werner, S.L.; Brown, M. The effects of prebiotics and substances with prebiotic properties on metabolic and inflammatory biomarkers in individuals with type 2 diabetes mellitus: A systematic review. J. Acad. Nutr. Diet. 2020, 120, 587–607. [Google Scholar] [CrossRef]
- Khangwal, I.; Shukla, P. Potential prebiotics and their transmission mechanisms: Recent approaches. J. Food Drug Anal. 2019, 27, 649–656. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Zhou, B.; Zhou, X.; Wang, Y.; Wang, H.; Jia, S.; Zhang, Z.; Chu, C.; Mu, J. Combined lowering effects of rosuvastatin and L. acidophilus on cholesterol levels in rat. J. Microbiol. Biotechnol. 2019, 29, 473–481. [Google Scholar] [CrossRef]
- Lin, M.Y.; Chang, F.J. Antioxidative effect of intestinal bacteria Bifidobacterium longum ATCC 15708 and Lactobacillus acidophilus ATCC 4356. Dig. Dis. Sci. 2000, 45, 1617–1622. [Google Scholar] [CrossRef]
- Wang, Y.; Wu, Y.; Wang, Y.; Xu, H.; Mei, X.; Yu, D.; Wang, Y.; Li, W. Antioxidant properties of probiotic bacteria. Nutrients 2017, 9, 521. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Wu, Y.; Wang, Y.; Fu, A.; Gong, L.; Li, W.; Li, Y. Bacillus amyloliquefaciens SC06 alleviates the oxidative stress of IPEC-1 via modulating Nrf2/Keap1 signaling pathway and decreasing ROS production. Appl. Microbiol. Biotechnol. 2016, 101, 1–12. [Google Scholar] [CrossRef]
- Harrison, D.; Griendling, K.K.; Landmesser, U.; Hornig, B.; Drexler, H. Role of oxidative stress in atherosclerosis. Am. J. Cardiol. 2003, 91, 7–11. [Google Scholar] [CrossRef]
- Lee, I.T.; Yang, C.M. Inflammatory signalings involved in airway and pulmonary diseases. Mediat. Inflamm. 2013, 2013, 791231. [Google Scholar] [CrossRef]
- Gómez-Guzmán, M.; Toral, M.; Romero, M.; Jimenez, R.; Galindo, P.; Sanchez, M.; Zarzuelo, M.J.; Olivares, M.; Galvez, J.; Duarte, J. Antihypertensive effects of probiotics Lactobacillus strains in spontaneously hypertensive rats. Mol. Nutr. Food Res. 2015, 59, 2326–2336. [Google Scholar] [CrossRef]
- Belton, O.; Byrne, D.; Kearney, D.; Leahy, A.; Fitzgerald, D.J. Cyclooxygenase-1 and -2-dependent prostacyclin formation in patients with atherosclerosis. Circulation 2000, 102, 840–845. [Google Scholar] [CrossRef] [PubMed]
- Patel, B.; Kumar, P.; Banerjee, R.; Basu, M.; Pal, A.; Samanta, M.; Das, S. Lactobacillus acidophilus attenuates Aeromonas hydrophila induced cytotoxicity in catla thymus macrophages by modulating oxidative stress and inflammation. Mol. Immunol. 2016, 75, 69–83. [Google Scholar] [CrossRef] [PubMed]
- Asemi, Z.; Zare, Z.; Shakeri, H.; Sabihi, S.S.; Esmaillzadeh, A. Effect of multispecies probiotic supplements on metabolic profiles, hs-CRP, and oxidative stress in patients with type 2 diabetes. Ann. Nutr. Metab. 2013, 63, 1–9. [Google Scholar] [CrossRef]
- Bao, Y.; Wang, Z.; Zhang, Y.; Zhang, J.; Wang, L.; Dong, X.; Su, F.; Yao, G.; Wang, S.; Zhang, H. Effect of Lactobacillus plantarum P-8 on lipid metabolism in hyperlipidemic rat model. Eur. J. Lipid Sci. Technol. 2012, 114, 1230–1236. [Google Scholar] [CrossRef]
- Guarino, M.P.C.; Altomare, A.; Emerenziani, S.; Di Rosa, C.; Ribolsi, M.; Balestrieri, P.; Iovino, P.; Rocchi, G.; Cicala, M. Mechanisms of Action of Prebiotics and Their Efects on Gastro-Intestinal Disorders in Adults. Nutrients 2020, 12, 1037. [Google Scholar] [CrossRef] [PubMed]
- Formes, H.; Reinhardt, C. The gut microbiota-a modulator of endothelial cell function and a contributing environmental factor to arterial thrombosis. Expert Rev. Hematol. 2019, 12, 541–549. [Google Scholar]
- Tenorio-Jimenez, C.; Martinez-Rmirez, M.J.; Tercero-Lozano, M.; Arraiza-Irigoyen, C.; Del Castillo-Codes, I.; Olza, J.; Plaza-Diaz, J.; Fontana, L.; Migueles, J.H.; Olivares, M.; et al. Evaluation of the effect of Lactobacillus reuteri V3401 on biomarkers of inflammation, cardiovascular risk and liver steatosis in obese adults with metabolic syndrome: A randomized clinical trial (PROSIR). BMC Compl. Alter. Med. 2018, 18, 1–8. [Google Scholar] [CrossRef]
- Kanner, J.; Lapidot, T. The stomach as a bioreactor: Dietary lipid peroxidation in the gastric fluid and the efects of plant-derived antioxidants. Free Radic. Biol. Med. 2001, 31, 1388–1395. [Google Scholar] [CrossRef]
- Goderska, K. The antioxidant and prebiotic properties of lactobionic acid. Appl. Microbiol. Biotechnol. 2019, 103, 3737–3751. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, R.; Yun, J.W. Lactobionic acid reduces body weight gain in diet induced obese rats by targeted inhibition of galectin-1. Biochem. Biophys. Res. Commun. 2015, 463, 1311–1316. [Google Scholar] [CrossRef] [PubMed]
- Kaulpiboon, J.; Rudeekulthamrong, P.; Watanasatitarpa, S.; Ito, K.; Pongsawasdi, P. Synthesis of long-chain isomaltooligosaccharides from tapioca starch and an in vitro investigation of their prebiotic properties. J. Mol. Catal. B 2015, 120, 127–135. [Google Scholar] [CrossRef]
- Singh, D.P.; Singh, J.; Boparai, R.K.; Zhu, J.H.; Mantri, S.; Khare, P.; Khardori, R.; Kondepudi, K.K.; Chopra, K.; Bishnoi, M. Isomalto-oligosaccharides, a prebiotic, functionally augment green tea e_ects against high fat diet-induced metabolic alterations via preventing gut dysbacteriosis in mice. Pharmacol. Res. 2017, 123, 103–113. [Google Scholar] [CrossRef]
- Vulevic, J.; Juric, A.; Walton, G.E.; Claus, S.P.; Tzortzis, G.; Toward, R.E.; Gibson, G.R. Influence of galactooligosaccharide mixture (B-GOS) on gut microbiota, immune parameters and metabolomics in elderly persons. Br. J. Nutr. 2015, 114, 586–595. [Google Scholar] [CrossRef]
- Jiang, J.; Feng, N.; Zhang, C.; Liu, F.; Zhao, J.; Zhang, H.; Chen, W. Lactobacillus reuteri A9 and Lactobacillus mucosae A13 isolated from Chinese superlongevity people modulate lipid metabolism in a hypercholesterolemia rat model. FEMS Microbiol. Lett. 2019, 366. [Google Scholar] [CrossRef]
- Jones, M.L.; Martoni, C.J.; Prakash, S. Oral supplementation with probiotic L. reuteri NCIMB 30242 increases mean circulating 25-hydroxyvitamin D: A post hoc analysis of a randomized controlled trial. J. Clin. Endocrinol. Metab. 2013, 98, 2944–2951. [Google Scholar] [CrossRef]
- Jung, S.; Lee, Y.J.; Kim, M.; Kim, M.; Kwak, J.H.; Lee, J.W.; Lee, J.H. Supplementation with two probiotic strains, Lactobacillus curvatus HY7601 and Lactobacillus plantarum KY1032, reduced body adiposity and Lp-PLA 2 activity in overweight subjects. J. Func. Foods 2015, 19, 744–752. [Google Scholar] [CrossRef]
- Asemi, Z.; Samimi, M.; Tabasi, Z.; Talebian, P.; Azarbad, Z.; Hydarzadeh, Z.; Esmaillzadeh, A. Effect of daily consumption of probiotic yoghurt on lipid profiles in pregnant women: A randomized controlled clinical trial. J. Matern. Fetal. Neonatal. Med. 2012, 25, 1552–1556. [Google Scholar] [CrossRef] [PubMed]
- Hoppu, U.; Isolauri, E.; Koskinen, P.; Laitinen, K. Maternal dietary counseling reduces total and LDL cholesterol postpartum. Nutrition 2014, 30, 159–164. [Google Scholar] [CrossRef] [PubMed]
- de Brito Alves, J.L.; de Oliveira, Y.; Carvalho, N.N.C.; Cavalcante, R.G.S.; Lira, M.M.P.; do Nascimento, L.C.P.; Magnani, M.; Vidal, H.; de Andrade Bragad, V.; de Souza, E.L. Gut microbiota and probiotic intervention as a promising therapeutic for pregnant women with cardiometabolic disorders: Present and future directions. Pharmacol. Res. 2019, 145, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Jiang, J.; Wu, C.; Zhang, C.; Zhao, J.; Yu, L.; Zhang, H.; Narbad, A.; Chen, W.; Zhai, Q. Effects of probiotic supplementation on cardiovascular risk factors in hypercholesterolemia: A systematic review and meta-analysis of randomized clinical trial. J. Funct. Foods 2020, 74, 104–177. [Google Scholar] [CrossRef]
- Lew, L.C.; Choi, S.B.; Khoo, B.Y.; Sreenivasan, S.; Ong, K.L.; Liong, M.T. Lactobacillus plantarum DR7 reduces cholesterol via phosphorylation of AMPK that down-regulated the mRNA expression of HMG-CoA reductase. Kor. J. Food Sci. Animal Res. 2018, 38, 350–361. [Google Scholar]
- Lew Nguyen, T.D.T.; Kang, J.H.; Lee, M.S. Characterization of Lactobacillus plantarum PH04, a potential probiotic bacterium with cholesterol-lowering effects. Inter. J. Food Microbiol. 2007, 113, 358–361. [Google Scholar] [CrossRef] [PubMed]
- Mizushima, S.; Ohshige, K.; Watanabe, J.; Kimura, M.; Kodowski, T.; Nakamurs, Y.; Tochikubo, O.; Ueshima, H. Randomized controlled trial of sour milk on blood pressure in bordeline hypertensive men. Am. J. Hypertens 2004, 17, 701–706. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, T. Potential benefits of garlic and other dietary supplements for the management of hypertension. Rev. Exp. Ther. Med. 2020, 19, 1479–1484. [Google Scholar]
- Schreiber, O.; Petersson, J.; Phillipson, M.; Perry, M.; Ross, S.; Holm, L. Lactobacillus reuteri prevents colitis by reducing P-selectin-associated leukocyte- and platelet-endothelial cell interactions. Am. J. Physiol. Gastrointest. Liver Physiol. 2009, 296, 534–542. [Google Scholar] [CrossRef] [PubMed]
- Haro, C.; Medina, M. Lactobacillus casei CRL 431 improves endothelial and platelet functionality in a pneumococcal infection model. Benef. Microbes 2019, 10, 533–541. [Google Scholar] [CrossRef]
- Parnell, J.A.; Reimer, R.A. Effect of prebiotic fibre supplementation on hepatic gene expression and serum lipids: A dose-response study in JCR:LA-cp rats. Br. J. Nutr. 2010, 103, 1177–1184. [Google Scholar] [CrossRef]
- Hume, M.P.; Nicolucci, A.C.; Reimer, R.A. Prebiotic supplementation improves appetite control in children with overweight and obesity: A randomized controlled trial. Am. J. Clin. Nutr. 2017, 105, 790–799. [Google Scholar] [CrossRef]
- Nicolucci, A.C.; Hume, M.P.; Martinez, I.; Mayengbam, S.; Walter, J.; Reimer, R.A. Prebiotics reduce body fat and alter intestinal microbiota in children who are overweight or with obesity. Gastroenterology 2017, 153, 711–722. [Google Scholar] [CrossRef]
- Kim, B.; Choi, H.N.; Yim, J.E. Effect of diet on the gut microbiota associated with obesity. J. Obesity Metab. Synd. 2019, 28, 216–224. [Google Scholar] [CrossRef]
- Mofid, V.; Izadi, A.; Majtehedi, S.Y.; Khemoat, L. Therapeutic and nutritional effects of synbiotic yogurts in children and adults: A clinical review. Probiotics Antimicrob. Proteins 2019, 10, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Liong, M.T.; Dunshea, F.R.; Shah, N.P. Effects of a symbiotic containing Lactobacillus acidophilus ATCC 4962 on plasma lipid profiles and morphology of erythrocytes in hypercholesterolemic pigs on high- and low-fat diets. Br. J. Nutr. 2007, 98, 736–744. [Google Scholar] [CrossRef]
- Haghighat, N.; Mohammadshahi, M.; Shayanpour, S.; Haghighizadeh, M.H. Effect of symbiotic and probiotic supplementation on serum levels of endothelial cell adhesion molecules in hemodialysis patients: A randomized control study. Probiotics Antimicro. Proteins 2018, 1, 1–9. [Google Scholar]
| Study Groups | Microbiota Results | References |
|---|---|---|
| Non ischemic heart failure with reduced ejection fraction; n = 28 (vs. 19 controls) | ↑ Streptococcus, Veillonella, Eggerthela ↓ Prevotella, SMB53 (Clostridiaceae) | [42] |
| Patients with ischemic or dilated cardiomyopathy; n = 84 (vs. 266 controls) | ↑ Prevotella, Hungatella (Lacnospiraceae), Succiniclasticum ↓ Blautia, Anaerostipes, Faecalibacterium, Lachnospiraceae, Bifidobacterium, Eubacterium, Coprococcus | [28] |
| Stable systolic heart failure; n = 20 (vs. 20 controls) | ↑ Escherichia-Shigella ↓ Blautia, Collinsella, Ruminococcaceae, Erysipelotrichaceaem Faecalibacterium | [38] |
| Patients with ischemic or dilated cardiomyopathy; n = 53 (vs. 40 controls) | ↑ Ruminococcus, Acinetobacter, Veillonella ↓ Faecalibacterium, Alistipes, Oscilibacter | [27] |
| Patients with hypertension (≥140/90 mmHg); n = 60 (vs. 60 controls) | ↑ Klebsiella, Salmonella, Streptococcus, Clostridium, Parabacteroides, Eggerthella ↓ Faecalibacterium, Roseburia, Synergistetes | [43] |
| Patients with hypertension (≥140/90 mmHg) and pre-hypertensive patients (125/80–139/90 mmHg); n = 155 (vs. 41 controls) | ↑ Prevotella, Klebsiella, Porphyromonas ↓ Faecalibacterium, Roseburia, Bifidobacterium, Oscillibacter, Coprococcus, Butyrivibrio | [44] |
| Patients with coronary artery disease; n = 70 (vs. 98 controls) | ↑ Escherichia-Shigella, Lactobacillus, Enterococcus, Streptococcus ↓ Faecalibacterium, Roseburia, Eubacterium, Subdoligranulum | [45] |
| Patients with stable angina and old myocardial infarction who underwent percutaneous coronary intervention or bypass; n = 39 (vs. 30 controls) | ↑ Lactobacillales ↓ Bacteroides, Clostridium | [46] |
| Patients with atherosclerotic plaques with clinical presentations of stable or unstable angina or acute myocardial infarction; n = 218 (vs. 187 controls) | ↑ Enterobacteriaceae, Streptococcus, Lactobacillus salivarius, Atopobium parvulum, Ruminococcus gnavus, Eggerthella lenta ↓ Roseburia, Faecalibacterium | [31] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Oniszczuk, A.; Oniszczuk, T.; Gancarz, M.; Szymańska, J. Role of Gut Microbiota, Probiotics and Prebiotics in the Cardiovascular Diseases. Molecules 2021, 26, 1172. https://doi.org/10.3390/molecules26041172
Oniszczuk A, Oniszczuk T, Gancarz M, Szymańska J. Role of Gut Microbiota, Probiotics and Prebiotics in the Cardiovascular Diseases. Molecules. 2021; 26(4):1172. https://doi.org/10.3390/molecules26041172
Chicago/Turabian StyleOniszczuk, Anna, Tomasz Oniszczuk, Marek Gancarz, and Jolanta Szymańska. 2021. "Role of Gut Microbiota, Probiotics and Prebiotics in the Cardiovascular Diseases" Molecules 26, no. 4: 1172. https://doi.org/10.3390/molecules26041172
APA StyleOniszczuk, A., Oniszczuk, T., Gancarz, M., & Szymańska, J. (2021). Role of Gut Microbiota, Probiotics and Prebiotics in the Cardiovascular Diseases. Molecules, 26(4), 1172. https://doi.org/10.3390/molecules26041172








