Preliminary Investigation of the Antioxidant, Anti-Diabetic, and Anti-Inflammatory Activity of Enteromorpha intestinalis Extracts
Abstract
1. Introduction
2. Results
2.1. Solvent Extracts of E. intestinalis Are Promising Source of Antioxidative Phytochemicals
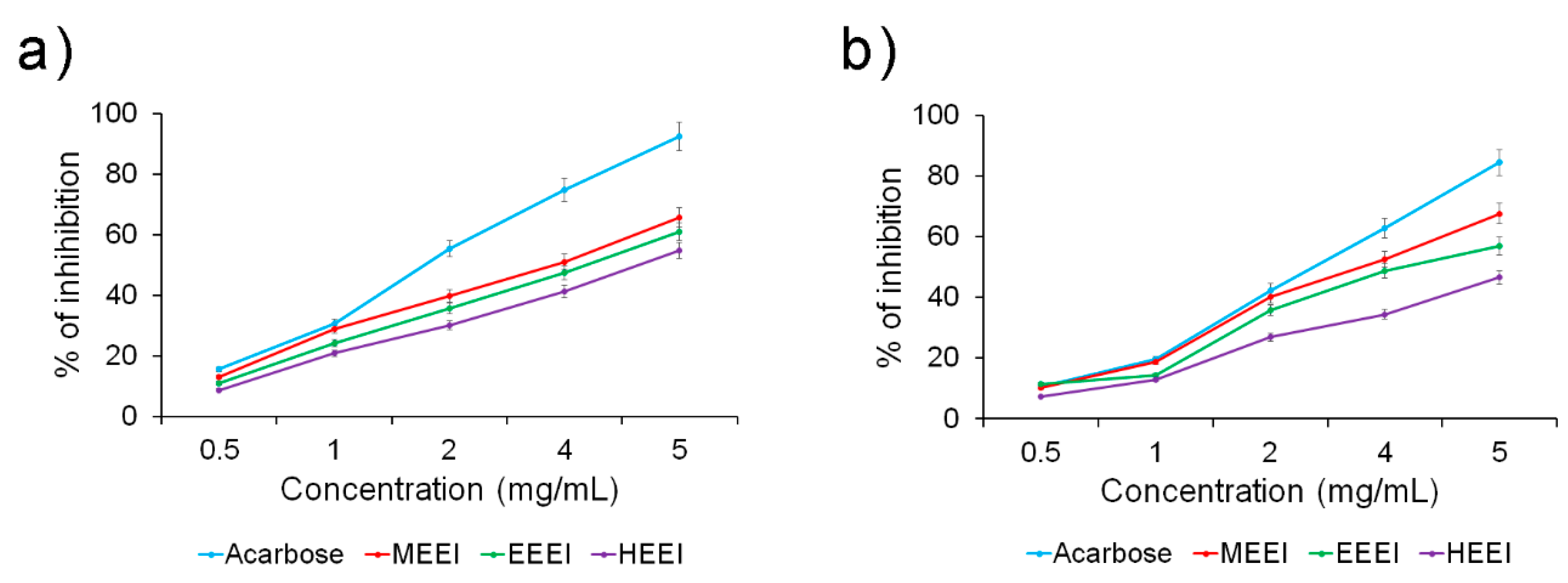
2.2. Solvent Extracts of E. intestinalis Exhibit Potential Antidiabetic Activity
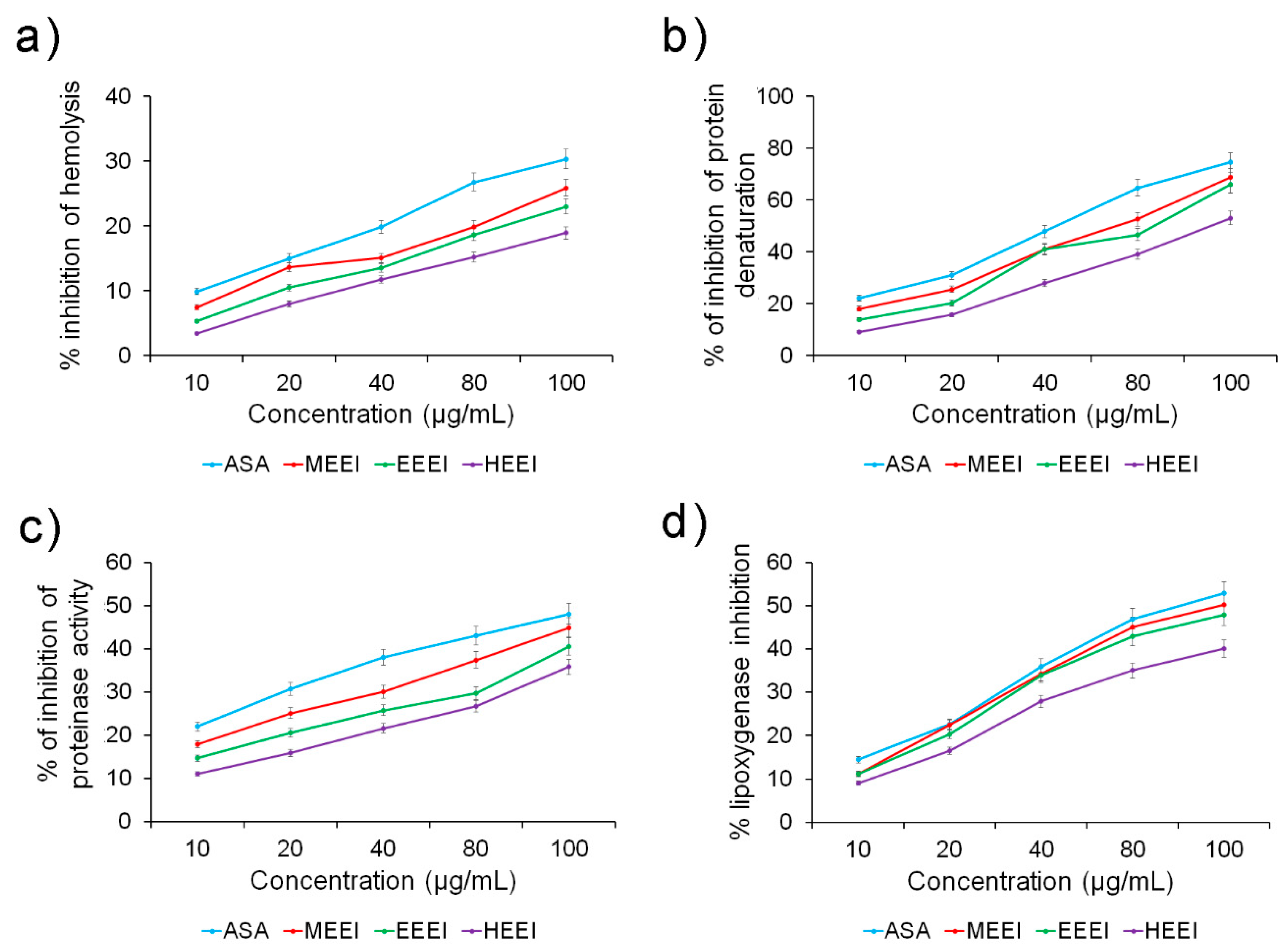
2.3. Solvent Extracts of E. intestinalis Are Promising Sources of Anti-Inflammatory Compounds
3. Discussion
4. Materials and Methods
4.1. Sampling
4.2. Extract Preparation
4.3. UV Visible and FT-IR Spectroscopy
4.4. Qualitative Phytochemical Screening
4.5. Estimation of Total Phenol Content and Total Flavonoids
4.6. Estimation of Total Flavonoid Content
4.7. Estimation of Ascorbic Acid Content
4.8. Determination of Free Radical Scavenging Activity
4.8.1. 1,1-Diphenyl-2-picrylhydrazyl Radical (DPPH•) Scavenging Activity
4.8.2. Hydroxyl Radical Scavenging Activity
4.8.3. Superoxide Anion Radical Scavenging Activity
4.8.4. Hydrogen Peroxide Radical Scavenging Activity
4.8.5. Total Antioxidant Activity by Phosphomolybdenum Assay
4.9. Antidiabetic Activity
4.9.1. Determination of α-Amylase Inhibition
4.9.2. Determination of α-Glucosidase Inhibition
4.10. Anti-Inflammatory Activity
4.10.1. Membrane Lysis Assay
Preparation of Erythrocyte Suspension
Heat-Induced Hemolysis
Protein Denaturation Assay
Proteinase Inhibitory Activity
Lipoxygenase Inhibition Assay
4.11. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Tian, C.; Hao, L.; Yi, W.; Ding, S.; Xu, F. Polyphenols, Oxidative Stress, and Metabolic Syndrome. Oxid. Med. Cell. Longev. 2020, 2020, 7398453. [Google Scholar] [CrossRef]
- Hussain, T.; Tan, B.; Yin, Y.; Blachier, F.; Tossou, M.C.; Rahu, N. Oxidative stress and inflammation: What polyphenols can do for us? Oxid. Med. Cell. Longev. 2016, 2016, 7432797. [Google Scholar] [CrossRef] [PubMed]
- Cotas, J.; Leandro, A.; Monteiro, P.; Pacheco, D.; Figueirinha, A.; Gonçalves, A.M.M.; da Silva, G.J.; Pereira, L. Seaweed Phenolics: From Extraction to Applications. Mar. Drugs 2020, 18, 384. [Google Scholar] [CrossRef]
- Zafar, M.S.; Quarta, A.; Marradi, M.; Ragusa, A. Recent developments in the reduction of oxidative stress through antioxidant polymeric formulations. Pharmaceutics 2019, 11, 505. [Google Scholar] [CrossRef]
- Zhang, H.; Tsao, R. Dietary polyphenols, oxidative stress and antioxidant and anti-inflammatory effects. Curr. Opin. Food Sci. 2016, 8, 33–42. [Google Scholar] [CrossRef]
- Mohanty, S.; Pradhan, B.; Patra, S.; Behera, C.; Nayak, R.; Jena, M. Screening for nutritive bioactive compounds in some algal strains isolated from coastal Odisha. J. Adv. Plant Sci. 2020, 10, 1–8. [Google Scholar]
- Farasat, M.; Khavari-Nejad, R.A.; Nabavi, S.M.; Namjooyan, F. Antioxidant activity, total phenolics and flavonoid contents of some edible green seaweeds from northern coasts of the Persian Gulf. Iran. J. Pharm. Res. 2014, 13, 163–170. [Google Scholar] [PubMed]
- Cho, M.; Kang, I.J.; Won, M.H.; Lee, H.S.; You, S. The antioxidant properties of ethanol extracts and their solvent-partitioned fractions from various green seaweeds. J. Med. Food 2010, 13, 1232–1239. [Google Scholar] [CrossRef]
- Pradhan, B.; Patra, S.; Behera, C.; Nayak, R.; Patil, S.; Bhutia, S.K.; Jena, M. Enteromorpha compressa extract induces anticancer activity through apoptosis and autophagy in oral cancer. Mol. Biol. Rep. 2020, 47, 9567–9578. [Google Scholar] [CrossRef]
- Wekre, M.E.; Kåsin, K.; Underhaug, J.; Holmelid, B.; Jordheim, M. Quantification of polyphenols in seaweeds: A case study of Ulva intestinalis. Antioxidants 2019, 8, 612. [Google Scholar] [CrossRef]
- Tang, H.; Inoue, M.; Uzawa, Y.; Kawamura, Y. Anti-tumorigenic components of a sea weed, Eeteromorpha clathrata. BioFactors 2004, 22, 107–110. [Google Scholar] [CrossRef]
- Khanavi, M.; Gheidarloo, R.; Sadati, N.; Ardekani, M.S.; Nabavi, S.M.; Tavajohi, S.; Ostad, S.N. Cytotoxicity of fucosterol containing fraction of marine algae against breast and colon carcinoma cell line. Pharmacogn. Mag. 2012, 8, 60–64. [Google Scholar] [PubMed]
- Lavoie, S.; Sweeney-Jones, A.M.; Mojib, N.; Dale, B.; Gagaring, K.; McNamara, C.W.; Quave, C.L.; Soapi, K.; Kubanek, J. Antibacterial oligomeric polyphenols from the green alga Cladophora socialis. J. Org. Chem. 2019, 84, 5035–5045. [Google Scholar] [CrossRef]
- Yan, X.; Yang, C.; Lin, G.; Chen, Y.; Miao, S.; Liu, B.; Zhao, C. Antidiabetic potential of green seaweed Enteromorpha prolifera flavonoids regulating insulin signaling pathway and gut microbiota in type 2 diabetic mice. J. Food Sci. 2019, 84, 165–173. [Google Scholar] [CrossRef]
- Lin, G.; Liu, X.; Yan, X.; Liu, D.; Yang, C.; Liu, B.; Huang, Y.; Zhao, C. Role of green macroalgae Enteromorpha prolifera polyphenols in the modulation of gene expression and intestinal microflora profiles in type 2 diabetic mice. Int. J. Mol. Sci. 2018, 20, 25. [Google Scholar] [CrossRef]
- Pradhan, B.; Patra, S.; Nayak, R.; Behera, C.; Dash, S.R.; Nayak, S.; Sahu, B.B.; Bhutia, S.K.; Jena, M. Multifunctional role of fucoidan, sulfated polysaccharides in human health and disease: A journey under the sea in pursuit of potent therapeutic agents. Int. J. Biol. Macromol. 2020, 164, 4263–4278. [Google Scholar] [CrossRef] [PubMed]
- Pradhan, B.; Nayak, R.; Patra, S.; Jit, B.P.; Ragusa, A.; Jena, M. Bioactive Metabolites from Marine Algae as Potent Pharmacophores against Oxidative Stress-Associated Human Diseases: A Comprehensive Review. Molecules 2021, 26, 37. [Google Scholar] [CrossRef] [PubMed]
- Ferrero-Miliani, L.; Nielsen, O.H.; Andersen, P.S.; Girardin, S.E. Chronic inflammation: Importance of NOD2 and NALP3 in interleukin-1β generation. Clin. Exp. Immunol. 2007, 147, 227–235. [Google Scholar] [CrossRef]
- Wallace, J.L.; Vong, L. NSAID-induced gastrointestinal damage and the design of GI-sparing NSAIDs. Curr. Opin. Investig. Drugs 2008, 9, 1151–1156. [Google Scholar]
- Maharana, S.; Pradhan, B.; Jena, M.; Misra, M.K. Diversity of Phytoplankton in Chilika Lagoon, Odisha, India. Environ. Ecol. 2019, 37, 737–746. [Google Scholar]
- Patra, S.; Pradhan, B.; Nayak, R.; Behera, C.; Rout, L.; Jena, M.; Efferth, T.; Bhutia, S.K. Chemotherapeutic efficacy of curcumin and resveratrol against cancer: Chemoprevention, chemoprotection, drug synergism and clinical pharmacokinetics. Semin. Cancer Biol. 2020, 30215–30217. [Google Scholar] [CrossRef]
- Pradhan, B.; Patra, S.; Maharana, S.; Behera, C.; Dash, S.R.; Jena, M. Demarcating antioxidant response against aluminum induced oxidative stress in Westiellopsis prolifica Janet 1941. Int. J. Phytoremediation 2020, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Ragusa, A.; Centonze, C.; Grasso, M.E.; Latronico, M.F.; Mastrangelo, P.F.; Fanizzi, F.P.; Maffia, M. Composition and Statistical Analysis of Biophenols in Apulian Italian EVOOs. Foods 2017, 6, 90. [Google Scholar] [CrossRef] [PubMed]
- Ragusa, A.; Centonze, C.; Grasso, M.E.; Latronico, M.F.; Mastrangelo, P.F.; Sparascio, F.; Fanizzi, F.P.; Maffia, M. A Comparative Study of Phenols in Apulian Italian Wines. Foods 2017, 6, 24. [Google Scholar] [CrossRef]
- Ragusa, A.; Centonze, C.; Grasso, M.E.; Latronico, M.F.; Mastrangelo, P.F.; Sparascio, F.; Maffia, M. HPLC Analysis of Phenols in Negroamaro and Primitivo Red Wines from Salento. Foods 2019, 8, 45. [Google Scholar] [CrossRef]
- Boulet, J.; Ducasse, M.-A.; Cheynier, V. Ultraviolet spectroscopy study of phenolic substances and other major compounds in red wines: Relationship between astringency and the concentration of phenolic substances: UV spectroscopy of red wine components. Aust. J. Grape Wine Res. 2017, 23, 193–199. [Google Scholar] [CrossRef]
- Sudha, G.; Balasundaram, A. Analysis of bioactive compounds in Padina pavonica using HPLC, UV-VIS and FTIR techniques. J. Pharmacogn. Phytochem. 2018, 7, 3192–3195. [Google Scholar]
- Rajeswari, R.; Jeyaprakash, K. Bioactive potential analysis of brown seaweed Sargassum wightii using UV-VIS and FT-IR. J. Drug Deliv. Ther. 2019, 9, 150–153. [Google Scholar] [CrossRef][Green Version]
- Pradhan, B.; Patra, S.; Dash, S.R.; Nayak, R.; Behera, C.; Jena, M. Evaluation of the anti-bacterial activity of methanolic extract of Chlorella vulgaris Beyerinck [Beijerinck] with special reference to antioxidant modulation. Futur. J. Pharm. Sci. 2021, 7, 17. [Google Scholar] [CrossRef]
- Pradhan, B.; Patra, S.; Dash, S.R.; Maharana, S.; Behera, C.; Jena, M. Antioxidant responses against aluminum metal stress in Geitlerinema amphibium. SN Appl. Sci. 2020, 2, 800. [Google Scholar] [CrossRef]
- Crujeiras, A.B.; Díaz-Lagares, A.; Carreira, M.C.; Amil, M.; Casanueva, F.F. Oxidative stress associated to dysfunctional adipose tissue: A potential link between obesity, type 2 diabetes mellitus and breast cancer. Free Radic. Res. 2013, 47, 243–256. [Google Scholar] [CrossRef] [PubMed]
- Darley-usmar, V.M.; Hogg, N.; O’leary, V.J.; Wilson, M.T.; Moncada, S. The simultaneous generation of superoxide and nitric oxide can initiate lipid peroxidation in human low density lipoprotein. Free Radic. Res. Commun. 1992, 17, 9–20. [Google Scholar] [CrossRef]
- Behera, S.K. Phytochemical screening and antioxidant properties of methanolic extract of root of Asparagus racemosus Linn. Int. J. Food Prop. 2018, 21, 2681–2688. [Google Scholar] [CrossRef]
- Aurand, L.W.; Boone, N.H.; Giddings, G.G. Superoxide and singlet oxygen in milk lipid peroxidation. J. Dairy Sci. 1977, 60, 363–369. [Google Scholar] [CrossRef]
- Pradhan, B.; Baral, S.; Patra, S.; Behera, C.; Nayak, R.; MubarakAli, D.; Jena, M. Delineation of gamma irradiation (60Co) induced oxidative stress by decrypting antioxidants and biochemical responses of microalga, Chlorella sp. Biocatal. Agric. Biotechnol. 2020, 25, 101595. [Google Scholar] [CrossRef]
- Kedare, S.B.; Singh, R.P. Genesis and development of DPPH method of antioxidant assay. J. Food Sci. Technol. 2011, 48, 412–422. [Google Scholar] [CrossRef]
- Olokoba, A.B.; Obateru, O.A.; Olokoba, L.B. Type 2 diabetes mellitus: A review of current trends. Oman Med. J. 2012, 27, 269. [Google Scholar] [CrossRef]
- Del Coco, L.; Vergara, D.; De Matteis, S.; Mensà, E.; Sabbatinelli, J.; Prattichizzo, F.; Bonfigli, A.R.; Storci, G.; Bravaccini, S.; Pirini, F.; et al. NMR-Based Metabolomic Approach Tracks Potential Serum Biomarkers of Disease Progression in Patients with Type 2 Diabetes Mellitus. J. Clin. Med. 2019, 8, 720. [Google Scholar] [CrossRef]
- Sudha, P.; Zinjarde, S.S.; Bhargava, S.Y.; Kumar, A.R. Potent α-amylase inhibitory activity of Indian Ayurvedic medicinal plants. BMC Complement. Altern. Med. 2011, 11, 5. [Google Scholar]
- Standl, E.; Baumgartl, H.J.; Füchtenbusch, M.; Stemplinger, J. Effect of acarbose on additional insulin therapy in type 2 diabetic patients with late failure of sulphonylurea therapy. Diabetes Obes. Metab. 1999, 1, 215–220. [Google Scholar] [CrossRef]
- Makinde, E.A.; Ovatlarnporn, C.; Sontimuang, C.; Herbette, G.; Olatunji, O.J. Chemical Constituents from the Aerial Part of Tiliacora triandra (Colebr.) Diels and Their α-Glucosidase and α-Amylase Inhibitory Activity. Nat. Prod. Commun. 2020, 15, 1934578X19899595. [Google Scholar] [CrossRef]
- Umapathy, E.; Ndebia, E.J.; Meeme, A.; Adam, B.; Menziwa, P.; Nkeh-Chungag, B.N.; Iputo, J.E. An experimental evaluation of Albuca setosa aqueous extract on membrane stabilization, protein denaturation and white blood cell migration during acute inflammation. J. Med. Plant Res. 2010, 4, 789–795. [Google Scholar]
- Okoli, C.O.; Akah, P.A.; Onuoha, N.J.; Okoye, T.C.; Nwoye, A.C.; Nworu, C.S. Acanthus montanus: An experimental evaluation of the antimicrobial, anti-inflammatory and immunological properties of a traditional remedy for furuncles. BMC Complement. Altern. Med. 2008, 8, 27. [Google Scholar] [CrossRef]
- Govindappa, M.; Poojashri, M.N. Antimicrobial, antioxidant and in vitro anti-inflammatory activity of ethanol extract and active phytochemical screening of Wedelia trilobata (L.) Hitchc. J. Pharmacogn. Phytother. 2011, 3, 43–51. [Google Scholar]
- Rackova, L.; Oblozinsky, M.; Kostalova, D.; Kettmann, V.; Bezakova, L. Free radical scavenging activity and lipoxygenase inhibition of Mahonia aquifolium extract and isoquinoline alkaloids. J. Inflamm. 2007, 4, 15. [Google Scholar] [CrossRef]
- Khasawneh, M.A.; Elwy, H.M.; Hamza, A.A.; Fawzi, N.M.; Hassan, A.H. Antioxidant, anti-lipoxygenase and cytotoxic activity of Leptadenia pyrotechnica (Forssk.) decne polyphenolic constituents. Molecules 2011, 16, 7510–7521. [Google Scholar] [CrossRef]
- Rath, J.; Adhikary, S.P. Algal Flora of Chilika Lake; Daya Publishing House: New Delhi, India, 2005. [Google Scholar]
- Patra, S.; Panda, P.K.; Naik, P.P.; Panigrahi, D.P.; Praharaj, P.P.; Bhol, C.S.; Mahapatra, K.K.; Padhi, P.; Jena, M.; Patil, S.; et al. Terminalia bellirica extract induces anticancer activity through modulation of apoptosis and autophagy in oral squamous cell carcinoma. Food Chem. Toxicol. 2020, 136, 111073. [Google Scholar] [CrossRef]
- Quettier-Deleu, C.; Gressier, B.; Vasseur, J.; Dine, T.; Brunet, C.; Luyckx, M.; Cazin, M.; Cazin, J.C.; Bailleul, F.; Trotin, F. Phenolic compounds and antioxidant activities of buckwheat (Fagopyrum esculentum Moench) hulls and flour. J. Ethnopharmacol. 2000, 72, 35–42. [Google Scholar] [CrossRef]
- Al-Ani, M.; Opara, L.U.; Al-Bahri, D.; Al-Rahbi, N. Spectrophotometric quantification of ascorbic acid contents of fruit and vegetables using the 2,4-dinitrophenylhydrazine method. J. Food Agric. Environ. 2007, 5, 165. [Google Scholar]
- Patra, S.; Bhol, C.S.; Panigrahi, D.P.; Praharaj, P.P.; Pradhan, B.; Jena, M.; Bhutia, S.K. Gamma irradiation promotes chemo-sensitization potential of gallic acid through attenuation of autophagic flux to trigger apoptosis in an NRF2 inactivation signalling pathway. Free Radic. Biol. Med. 2020, 160, 111–124. [Google Scholar] [CrossRef] [PubMed]
- Chakrabarti, R.; Singh, B.; Vanchhawng, L.; Thirumurugan, K. Screening of nine herbal plants for in vitro α-amylase inhibition. Screening 2014, 7, 84–89. [Google Scholar]
- Kumar, S.; Kumar, V.; Prakash, O. Enzymes inhibition and antidiabetic effect of isolated constituents from Dillenia indica. Biomed Res. Int. 2013, 2013, 382063. [Google Scholar] [CrossRef]
- Shinde, U.A.; Phadke, A.S.; Nair, A.M.; Mungantiwar, A.A.; Dikshit, V.J.; Saraf, M.N. Membrane stabilizing activity—A possible mechanism of action for the anti-inflammatory activity of Cedrus deodara wood oil. Fitoterapia 1999, 70, 251–257. [Google Scholar] [CrossRef]
- Gunathilake, K.D.P.P.; Ranaweera, K.K.D.S.; Rupasinghe, H.P.V. Influence of boiling, steaming and frying of selected leafy vegetables on the in vitro anti-inflammation associated biological activities. Plants 2018, 7, 22. [Google Scholar] [CrossRef]
- Gambhire, M.; Juvekar, A.; Wankhede, S. Evaluation of anti-inflammatory activity of methanol extract of Barleria cristata leaves by in vivo and in vitro methods. Int. J. Pharmacol. 2009, 7, 1–4. [Google Scholar]
- Sakat, S.; Juvekar, A.R.; Gambhire, M.N. In vitro antioxidant and anti-inflammatory activity of methanol extract of Oxalis corniculata Linn. Int. J. Pharm. Pharm. Sci. 2010, 2, 146–155. [Google Scholar]
- Wu, H. Affecting the activity of soybean lipoxygenase-1. J. Mol. Graph. 1996, 14, 331–337. [Google Scholar] [CrossRef]


| Bioactive Compounds | MEEI | EEEI | HEEI |
|---|---|---|---|
| Alkaloids | + | + | + |
| Glycosides | + | + | + |
| Reducing sugars | − | − | − |
| Proteins | + | + | + |
| Terpenoids | + | + | + |
| Phenols and Tannins | + | + | + |
| Steroids | − | − | − |
| Saponins | + | − | − |
| Anthocyanins | − | − | − |
| Coumarin | + | + | + |
| Extract | Peak Value (cm−1) | Functional Group | Bonding Pattern |
|---|---|---|---|
| MEEI | 3726.47 | Alcohol (free) | O-H stretching |
| 2920.23 | Alkane | C-H stretching | |
| 2850.79 | Alkane | C-H stretching | |
| 2358.94 | Carbon dioxide | O=C=O | |
| 1743.65 | Esters (6-membered lactone) | C=O stretching | |
| 1031.92 | Sulfoxide | S=O stretching | |
| 669.30 | Halo compound | C-Br | |
| EEEI | 3354.21 | Alcohol (intermolecular bond) | O-H stretching |
| 2922.16 | Alkane | C-H stretching | |
| 2850.79 | Alkane | C-H stretching | |
| 2358.94 | Carbon dioxide | O=C=O | |
| 1643.35 | Alkene (cis-disubstituted) | C=C stretching | |
| 1045.42 | Sulfoxide | S=O stretching | |
| 667.37 | Halo compound | C-Br | |
| HEEI | 3726.47 | Alcohol (free) | O-H stretching |
| 2922.16 | Alkane | C-H stretching | |
| 2852.72 | Alkane | C-H stretching | |
| 2358.94 | Carbon dioxide | O=C=O | |
| 1710.86 | Conjugated acid | C=O stretching | |
| 1462.04 | Sulfate | S=O stretching | |
| 1259.52 | Alkyl aryl ether | C-O stretching | |
| 750.31 | Monosubstituted | C-H bending | |
| 669.30 | Halo compound | C-Br |
| Assay | MEEI | EEEI | HEEI |
|---|---|---|---|
| Total phenolic content (mg of GAE 1/g) | 23.00 ± 0.05 * | 59.67 ± 0.01 **++ | 11.91 ± 0.06 *+ |
| Total flavonoid content (mg of RUE 2/g) | 416.00 ± 0.03 ## | 624.67 ± 0.05 ##++ | 152.00 ± 0.14 #+ |
| Ascorbic acid content (µg of AA/g) | 336.00 ± 0.36 $ | 446.00 ± 0.43 $$++ | 195.00 ± 0.33 $+ |
| Total antioxidant activity (mg of AAE 3/g) | 88.00 ± 0.02 & | 177.33 ± 0.02 &&++ | 45.00 ± 0.04 &+ |
| Assay | Ascorbic Acid | MEEI | EEEI | HEEI |
|---|---|---|---|---|
| DPPH• scavenging activity | 2.62 ± 0.23 | 0.50 ± 0.01 ** | 3.38 ± 0.57 * | 3.68 ± 0.98 * |
| H2O2 scavenging activity | 3.13 ± 0.87 | 4.72 ± 0.67 * | 3.22 ± 0.68 * | 18.41 ± 1.78 ** |
| Hydroxyl radical scavenging activity | 2.26 ± 0.22 | 0.32 ± 0.01 ** | 2.58 ± 0.11 * | 2.68 ± 0.87 * |
| Superoxide scavenging activity | 2.26 ± 0.67 | 2.49 ± 0.32 * | 2.73 ± 0.16 * | 3.69 ± 0.73 * |
| Assay | Acarbose | MEEI | EEEI | HEEI |
|---|---|---|---|---|
| α-Amylase activity | 2.81 ± 0.78 | 3.81 ± 0.45 * | 4.15 ± 0.56 * | 4.68 ± 0.33 * |
| α-Glucosidase activity | 3.32 ± 0.96 | 3.82 ± 0.56 * | 4.32 ± 0.78 * | 5.44 ± 0.51 * |
| Assay | Acetylsalicylic Acid | MEEI | EEEI | HEEI |
|---|---|---|---|---|
| Hemolysis inhibition activity | 8.62 ± 0.99 | 10.81 ± 1.12 * | 11.21 ± 1.76 * | 13.09 ±1.98 * |
| Protein denaturation activity | 3.14 ± 0.88 | 3.68 ± 0.81 * | 3.95 ± 0.19 * | 4.88 ± 0.49 * |
| Proteinase activity | 5.11 ± 0.97 | 5.84 ± 0.91 * | 6.89 ± 0.37 * | 7.60 ± 0.12 * |
| Lipoxygenase activity | 4.52 ± 0.58 | 4.72 ± 0.76 * | 4.95 ± 0.86 * | 6.01 ± 0.27 * |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pradhan, B.; Patra, S.; Behera, C.; Nayak, R.; Jit, B.P.; Ragusa, A.; Jena, M. Preliminary Investigation of the Antioxidant, Anti-Diabetic, and Anti-Inflammatory Activity of Enteromorpha intestinalis Extracts. Molecules 2021, 26, 1171. https://doi.org/10.3390/molecules26041171
Pradhan B, Patra S, Behera C, Nayak R, Jit BP, Ragusa A, Jena M. Preliminary Investigation of the Antioxidant, Anti-Diabetic, and Anti-Inflammatory Activity of Enteromorpha intestinalis Extracts. Molecules. 2021; 26(4):1171. https://doi.org/10.3390/molecules26041171
Chicago/Turabian StylePradhan, Biswajita, Srimanta Patra, Chhandashree Behera, Rabindra Nayak, Bimal Prasad Jit, Andrea Ragusa, and Mrutyunjay Jena. 2021. "Preliminary Investigation of the Antioxidant, Anti-Diabetic, and Anti-Inflammatory Activity of Enteromorpha intestinalis Extracts" Molecules 26, no. 4: 1171. https://doi.org/10.3390/molecules26041171
APA StylePradhan, B., Patra, S., Behera, C., Nayak, R., Jit, B. P., Ragusa, A., & Jena, M. (2021). Preliminary Investigation of the Antioxidant, Anti-Diabetic, and Anti-Inflammatory Activity of Enteromorpha intestinalis Extracts. Molecules, 26(4), 1171. https://doi.org/10.3390/molecules26041171










