Okra (Abelmoschus Esculentus) as a Potential Dietary Medicine with Nutraceutical Importance for Sustainable Health Applications
Abstract
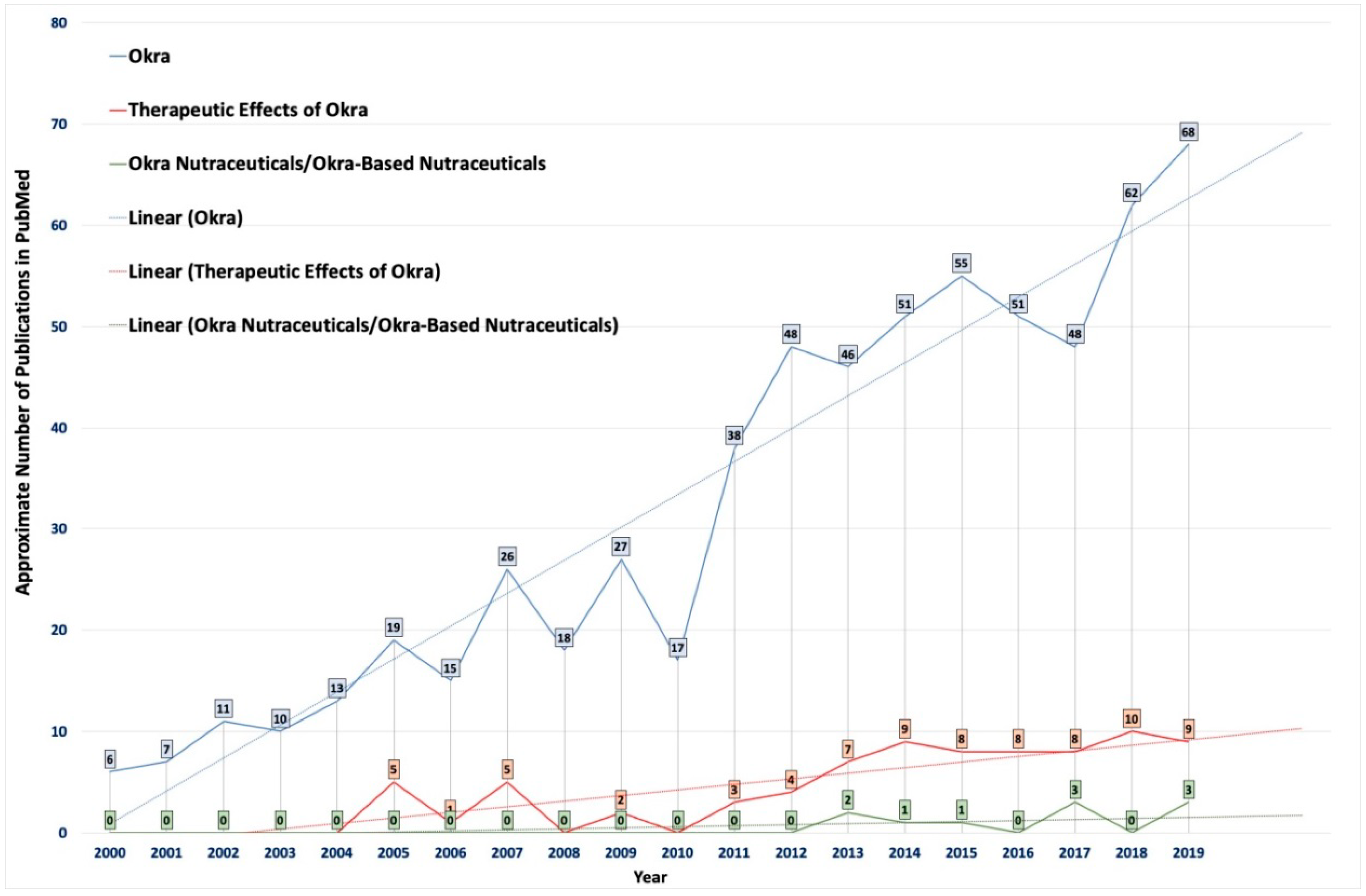
1. Introduction
2. Nutritional and Bioactive Constituents in Okra
3. The Pharmacological and Potential Applications of Okra-Derived Biomolecules
3.1. Antidiabetic Efficacy
3.2. Antioxidant Efficacy
3.3. Anticancer Effect
3.4. Immunomodulatory Potential
3.5. Microbicidal Action
4. In Vivo Studies on the Health Benefits of Okra and Its Components
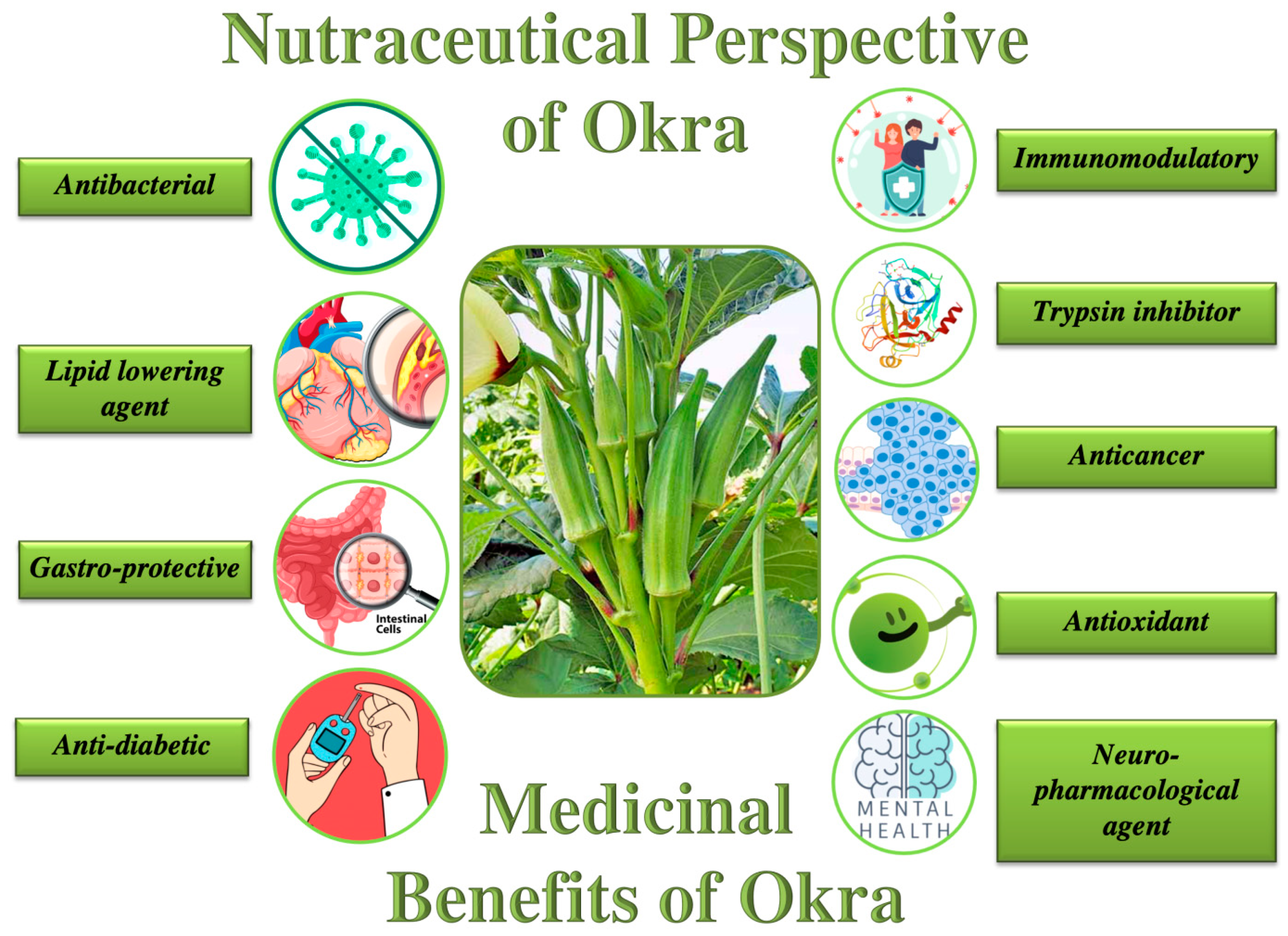
5. Therapeutic Prospects of Okra as Dietary Medicine/Nutraceuticals
6. Formulation and Development of Okra-Based Nutraceuticals
7. Global Okra Production and Possible Nutraceutical Market
8. Safety and Efficacy
9. Future Perspectives and Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Jain, N.; Jain, R.; Jain, V.; Jain, S. A review on: Abelmoschus esculentus. Pharmacia 2012, 1, 84–89. [Google Scholar]
- Lamont, W.J. Okra—A versatile vegetable crop. Hort Technol. 1999, 9, 179–184. [Google Scholar] [CrossRef]
- Tindall, H.D. Vegetables in the Tropics; Macmillan Publishers Limited: London, UK, 1983. [Google Scholar] [CrossRef]
- Dhaliwal, M.S. Okra (Abelmoschus esculentus) L. (Moench); Kalyani Publishers: New Delhi, India, 2010. [Google Scholar]
- Kumar, A.; Kumar, P.; Nadendla, R. A review on: Abelmoschus esculentus (Okra). Int. Res. J. Pharm. Appl. Sci. 2013, 3, 129–132. [Google Scholar]
- Archana, G.; Babu, P.A.S.; Sudharsan, K.; Sabina, K.; Raja, R.P.; Sivarajan, M.; Sukumar, M. Evaluation of Fat Uptake of Polysaccharide Coatings on Deep-Fat Fried Potato Chips by Confocal Laser Scanning Microscopy. Int. J. Food Prop. 2015, 19, 1583–1592. [Google Scholar] [CrossRef]
- Costantino, A.; Romanchik-Cerpovicz, J. Physical and sensory measures indicate moderate fat replacement in frozen dairy dessert is feasible using okra gum as a milk-fat ingredient substitute. J. Am. Diet. Assoc. 2004, 104, 44. [Google Scholar] [CrossRef]
- Yuennan, P.; Sajjaanantakul, T.; Kung, B. Effect of Okra Cell Wall and Polysaccharide on Physical Properties and Stability of Ice Cream. J. Food Sci. 2014, 79, E1522–E1527. [Google Scholar] [CrossRef]
- Islam, M.T. Phytochemical information and pharmacological activities of Okra (Abelmoschus esculentus): A literature-based review. Phytother. Res. 2019, 33, 72–80. [Google Scholar] [CrossRef]
- Durazzo, A.; Lucarini, M.; Novellino, E.; Souto, E.B.; Daliu, P.; Santini, A. Abelmoschus esculentus (L.): Bioactive Components’ Beneficial Properties-Focused on Antidiabetic Role-For Sustainable Health Applications. Molecules 2018, 24, 38. [Google Scholar] [CrossRef]
- S.E.P. Limited. 2021. Available online: https://www.sbepl.com/ (accessed on 18 January 2021).
- Hunan, H. QiyiXinyeCulture Media, China. 2021. Available online: https://www.globalsources.com/si/AS/Hunan-Qiyi/6008850060925/Showroom/3000000149681/ALL.htm (accessed on 19 January 2021).
- Vermerris, W.; Nicholson, R. Families of Phenolic Compounds and Means of Classification. Phenolic Compd. Biochem. 2006. [Google Scholar] [CrossRef]
- Savello, P.A.; Martin, F.W.; Hill, J.M. Nutritional composition of okra seed meal. J. Agric. Food Chem. 1980, 28, 1163–1166. [Google Scholar] [CrossRef]
- Cieślik, E.; Gębusia, A.; Filipiak-Florkiewicz, A.; Mickowska, B. The content of protein and of amino acids in Jerusalem artichoke tubers (Helianthus tuberosus L.) of red variety Rote Zonenkugel. Acta Sci. Pol. Technol. Aliment. 2012, 10, 433–441. [Google Scholar]
- Alghamdi, A.A. Nutritional Assessment of Different Date Fruits (Phoenix Dactylifera L.) Varieties Cultivated in Hail Province, Saudi Arabia. Biosci. Biotechnol. Res. Commun. 2018, 11, 263–269. [Google Scholar] [CrossRef]
- Gemede, H.F.; Haki, G.D.; Beyene, F.; Woldegiorgis, A.Z.; Rakshit, S.K. Proximate, mineral, and antinutrient compositions of indigenous Okra (Abelmoschus esculentus) pod accessions: Implications for mineral bioavailability. Food Sci. Nutr. 2015, 4, 223–233. [Google Scholar] [CrossRef] [PubMed]
- Aykroyd, W.R.; Gopalan, C.; Balasubramanian, S.C. The Nutritive Value of Indian Foods and the Planning of Satisfactory Diets. Spec. Rep. Ser. Indian Counc. Med. Res. 1963, 42, 1–255. [Google Scholar] [PubMed]
- Akintoye, H.; Adebayo, A.; Aina, O. Growth and Yield Response of Okra Intercropped with Live Mulches. Asian J. Agric. Res. 2011, 5, 146–153. [Google Scholar] [CrossRef]
- Available online: https://nutritiondata.self.com/facts/vegetables-and-vegetable-products/2497/2 (accessed on 10 November 2020).
- Sharma, K.; Gupta, A.; Kumar, M.; Singh, M.K.; Malik, S.; Singh, B.; Chaudhary, V. Effect of Integrated Nutrient Management and Foliar Spray of Bioregulators on Growth and Yield of Okra. Int. J. Curr. Microbiol. Appl. Sci. 2020, 9, 344–354. [Google Scholar]
- Kumar, R.; Patil, M.; Patil, S.; Paschapur, M. Evaluation of Abelmoschus esculentus mucilage as suspending agent in paracetamol suspension. Int. J. Pharm. Tech. Res. 2009, 1, 658–665. [Google Scholar]
- Avallone, S.; Tiemtore, T.-W.E.; Mouquet-Rivier, C.; Trèche, S. Nutritional value of six multi-ingredient sauces from Burkina Faso. J. Food Compos. Anal. 2008, 21, 553–558. [Google Scholar] [CrossRef]
- Kendall, C.; Jenkins, D.J.A. A Dietary portfolio: Maximal reduction of low-density lipoprotein cholesterol with diet. Curr. Atheroscler. Rep. 2004, 6, 492–498. [Google Scholar] [CrossRef]
- Agbo, A.E.; Gnakri, D.; Beugrz, G.M.; Fondio, L.; Kouamz, C.M. Maturity degree of four Okra fruits varieties and their nutrients composition. Electoron. J. Food Plants Chem. 2008, 5, 1–4. [Google Scholar]
- Langenberg, C.; Lotta, L.A. Genomic insights into the causes of type 2 diabetes. Lancet 2018, 391, 2463–2474. [Google Scholar] [CrossRef]
- Ripon, S.S.; Mahmood, A.; Chowdhury, M.M.; Islam, M.T. A Possible Anti-Atherothrombosis Activity via Cytoprotective Trait of the Clerodenrumviscosum Leaf Methanol Extract. Insights Biomed. 2016, 1, 1–6. [Google Scholar] [CrossRef]
- Tomoda, M.; Shimizu, N.; Gonda, R.; Kanari, M.; Yamada, H.; Hikino, H. Anticomplementary and hypoglycemic activity of Okra and Hibiscus mucilages. Carbohydr. Res. 1989, 190, 323–328. [Google Scholar] [CrossRef]
- Saha, D.; Jain, B.; Jain, V.K. Phytochemical evaluation and characterization of hypogylcemic activity of various extracts of Abelmoschus esculentus Linn. Fruit. Int. J. Pharm. Pharm. Sci. 2011, 3, 183–185. [Google Scholar]
- Khatun, H.; Rahman, A.; Biswas, M.; Islam, A.U. Water-soluble Fraction of Abelmoschus esculentus L. Interacts with Glucose and Metformin Hydrochloride and Alters Their Absorption Kinetics after Coadministration in Rats. ISRN Pharm. 2011, 2011, 260537. [Google Scholar] [CrossRef]
- Balakumar, P.; Kadian, S.; Mahadevan, N. Are PPAR alpha agonists a rational therapeutic strategy for preventing abnormalities of the diabetic kidney? Pharmacol. Res. 2012, 65, 430–436. [Google Scholar] [CrossRef]
- Lv, M.; Chen, Z.; Hu, G.-Y.; Li, Q. Therapeutic strategies of diabetic nephropathy: Recent progress and future perspectives. Drug Discov. Today 2015, 20, 332–346. [Google Scholar] [CrossRef]
- Ge, J.; Miao, J.-J.; Sun, X.-Y.; Yu, J.-Y. Huangkui capsule, an extract from Abelmoschus manihot (L.) medic, improves diabetic nephropathy via activating peroxisome proliferator–activated receptor (PPAR)-α/γ and attenuating endoplasmic reticulum stress in rats. J. Ethnopharmacol. 2016, 189, 238–249. [Google Scholar] [CrossRef]
- Zhang, L.; Li, P.; Xing, C.-Y.; Zhao, J.-Y.; He, Y.; Wang, J.-Q.; Wu, X.-F.; Liu, Z.; Zhang, A.; Lin, H.; et al. Efficacy and Safety of Abelmoschus manihot for Primary Glomerular Disease: A Prospective, Multicenter Randomized Controlled Clinical Trial. Am. J. Kidney Dis. 2014, 64, 57–65. [Google Scholar] [CrossRef]
- Du, L.; Tao, J.; Jiang, S.; Qian, D.; Guo, J.; Duan, J. Metabolic profiles of the Flos Abelmoschus manihot extract by intestinal bacteria from the normal and CKD model rats based on UPLC-Q-TOF/MS. Biomed. Chromatogr. 2016, 31. [Google Scholar] [CrossRef]
- Li, P.; Chen, Y.; Lin, H.; Ni, Z.; Zhan, Y.; Wang, R.; Yang, H.; Fang, J.-A.; Wang, N.; Li, W.-G.; et al. Abelmoschus manihot—A traditional Chinese medicine versus losartan potassium for treating IgA nephropathy: Study protocol for a randomized controlled trial. Trials 2017, 18, 170. [Google Scholar] [CrossRef] [PubMed]
- Tu, Y.; Sun, W.; Wan, Y.-G.; Che, X.-Y.; Pu, H.-P.; Yin, X.-J.; Chen, H.-L.; Meng, X.-J.; Huang, Y.-R.; Shi, X.-M. Huangkui capsule, an extract from Abelmoschus manihot (L.) medic, ameliorates adriamycin-induced renal inflammation and glomerular injury via inhibiting p38MAPK signaling pathway activity in rats. J. Ethnopharmacol. 2013, 147, 311–320. [Google Scholar] [CrossRef]
- Zhao, Q.; Wan, Y.G.; Sun, W.; Wang, C.J.; Wei, Q.X.; Chen, H.L.; Meng, X.J. Effects of Huangkui Capsule on renal inflammatory injury by intervening p38MAPK signaling pathway in rats with adriamycin-induced nephropathy. China J. Chin. Mater. Med. 2012, 37, 2926–2934. [Google Scholar]
- Mao, Z.-M.; Shen, S.-M.; Wan, Y.; Sun, W.; Chen, H.-L.; Huang, M.-M.; Yang, J.-J.; Wu, W.; Tang, H.-T.; Tang, R.-M. Huangkui capsule attenuates renal fibrosis in diabetic nephropathy rats through regulating oxidative stress and p38MAPK/Akt pathways, compared to α-lipoic acid. J. Ethnopharmacol. 2015, 173, 256–265. [Google Scholar] [CrossRef] [PubMed]
- Cai, H.; Su, S.; Qian, D.-W.; Guo, S.; Tao, W.; Cong, X.D.; Tang, R.; Duan, J. Renal protective effect and action mechanism of Huangkui capsule and its main five flavonoids. J. Ethnopharmacol. 2017, 206, 152–159. [Google Scholar] [CrossRef] [PubMed]
- Panneerselvam, K.; Ramachandran, S.; Sabitha, V.; Naveen, K.R. Antidiabetic and antihyperlipidemic potential of Abelmoschus esculentus (L.) Moench. in streptozotocin-induced diabetic rats. J. Pharm. Bioallied Sci. 2011, 3, 397–402. [Google Scholar] [CrossRef] [PubMed]
- Sabitha, V.; Panneerselvam, K.; Ramachandran, S. In vitroαglucosidase andα-amylase enzyme inhibitory effects in aqueous extracts of Abelmoscus esculentus (L.) Moench. Asian Pac. J.Trop. Biomed. 2012, 2, S162–S164. [Google Scholar] [CrossRef]
- Lu, Y.; Demleitner, M.F.; Song, L.; Rychlik, M.; Huang, D. Oligomeric proanthocyanidins are the active compounds in Abelmoschus esculentus Moench for its α-amylase and α-glucosidase inhibition activity. J. Funct. Foods 2016, 20, 463–471. [Google Scholar] [CrossRef]
- Zhang, T.; Xiang, J.; Zheng, G.; Yan, R.; Min, X. Preliminary characterization and anti-hyperglycemic activity of a pectic polysaccharide from okra (Abelmoschus esculentus (L.) Moench.). J. Funct. Foods 2018, 41, 19–24. [Google Scholar] [CrossRef]
- Gemede, H.F.; Haki, G.D.; Beyene, F.; Rakshit, S.K.; Woldegiorgis, A.Z. Indigenous Ethiopian okra (Abelmoschus esculentus) mucilage: A novel ingredient with functional and antioxidant properties. Food Sci. Nutr. 2018, 6, 563–571. [Google Scholar] [CrossRef]
- Wang, K.; Li, M.; Wen, X.; Chen, X.; He, Z.; Ni, Y. Optimization of ultrasound-assisted extraction of okra (Abelmoschus esculentus (L.) Moench.) polysaccharides based on response surface methodology and antioxidant activity. Int. J. Biol. Macromol. 2018, 114, 1056–1063. [Google Scholar] [CrossRef] [PubMed]
- Williamson, G.; Kay, C.D.; Crozier, A. The Bioavailability, Transport, and Bioactivity of Dietary Flavonoids: A Review from a Historical Perspective. Compr. Rev. Food Sci. Food Saf. 2018, 17, 1054–1112. [Google Scholar] [CrossRef] [PubMed]
- Shen, D.-D.; Li, X.; Qin, Y.-L.; Li, M.-T.; Han, Q.-H.; Zhou, J.; Lin, S.; Zhao, L.; Zhang, Q.; Qin, W.; et al. Physicochemical properties, phenolic profiles, antioxidant capacities, and inhibitory effects on digestive enzymes of okra (Abelmoschus esculentus) fruit at different maturation stages. J. Food Sci. Technol. 2019, 56, 1275–1286. [Google Scholar] [CrossRef]
- Gemede, H.F.; Ratta, N.; Haki, G.D.; Woldegiorgis, A.Z.; Beyene, F. Nutritional Quality and Health Benefits of Okra (Abelmoschus esculentus): A Review. J. Food Process. Technol. 2015, 6. [Google Scholar] [CrossRef]
- Gonzales, G.B.; Smagghe, G.; Grootaert, C.; Zotti, M.J.; Raes, K.; Van Camp, J. Flavonoid interactions during digestion, absorption, distribution and metabolism: A sequential structure–activity/property relationship-based approach in the study of bioavailability and bioactivity. Drug Metab. Rev. 2015, 47, 175–190. [Google Scholar] [CrossRef]
- Xia, F.; Zhong, Y.; Li, M.; Chang, Q.; Liao, Y.; Shi, Z.; Pan, R.-L. Antioxidant and Anti-Fatigue Constituents of Okra. Nutrients 2015, 7, 8846–8858. [Google Scholar] [CrossRef]
- Arapitsas, P. Identification and quantification of polyphenolic compounds from okra seeds and skins. Food Chem. 2008, 110, 1041–1045. [Google Scholar] [CrossRef]
- Ansari, N.M.; Houlihan, L.; Hussain, B.; Pieroni, A. Antioxidant activity of five vegetables traditionally consumed by South-Asian migrants in Bradford, Yorkshire, UK. Phytother. Res. 2005, 19, 907–911. [Google Scholar] [CrossRef]
- Khomsug, P.; Thongjaroenbuangam, W.; Pakdeenarong, N.; Suttajit, M.; Chantiratikul, P. Antioxidative activities and phenolic content of extracts from okra (Abelmoschus esculentus L.). Res. J. Biol. Sci. 2010, 5, 310–313. [Google Scholar] [CrossRef]
- Yuan, K.; Liao, H.; Dong, W.; Shi, X.; Liu, H. Analysis and comparison of the active components and antioxidant activities of extracts from Abelmoschus esculentus L. Pharmacogn. Mag. 2012, 8, 156–161. [Google Scholar] [CrossRef]
- Nguyen, N.H.; Ta, Q.T.H.; Pham, Q.T.; Luong, T.N.H.; Phung, V.T.; Duong, T.-H.; Van Giau, V. Anticancer Activity of Novel Plant Extracts and Compounds from Adenosma bracteosum (Bonati) in Human Lung and Liver Cancer Cells. Molecules 2020, 25, 2912. [Google Scholar] [CrossRef] [PubMed]
- Tavares-Carreón, F.; De la Torre-Zavala, S.; Arocha-Garza, H.F.; Souza, V.; Galán-Wong, L.J.; Avilés-Arnaut, H. In vitro anticancer activity of methanolic extract of Granulocystopsis sp., a microalgae from an oligotrophic oasis in the Chihuahuan desert. PeerJ 2020, 8, e8686. [Google Scholar] [CrossRef] [PubMed]
- National Cancer Institute. Understanding Cancer. 2020. Available online: https://www.cancer.gov/about-cancer/understanding (accessed on 12 November 2020).
- Vo, T.T.L.; Jang, W.-J.; Jeong, C.-H. Leukotriene A4 hydrolase: An emerging target of natural products for cancer chemoprevention and chemotherapy. Ann. N. Y. Acad. Sci. 2018, 1431, 3–13. [Google Scholar] [CrossRef] [PubMed]
- Reddy, M.N.; Adnan, M.; Alreshidi, M.M.; Saeed, M.; Patel, M. Evaluation of Anticancer, Antibacterial and Antioxidant Properties of a Medicinally Treasured Fern Tectariacoadunata with its Phytoconstituents Analysis by HR-LCMS. Anti-Cancer Agents Med. Chem. 2020, 20, 1845–1856. [Google Scholar] [CrossRef]
- Bhatt, M.; Patel, M.; Adnan, M.; Reddy, M.N. Anti-Metastatic Effects of Lupeol via the Inhibition of MAPK/ERK Pathway in Lung Cancer. Anti-Cancer Agents Med. Chem. 2020, 21, 201–206. [Google Scholar] [CrossRef]
- Adetuyi, F.; Ibrahim, T. Effect of Fermentation Time on the Phenolic, Flavonoid and Vitamin C Contents and Antioxidant Activities of Okra (Abelmoschus esculentus) Seeds. Niger. Food J. 2014, 32, 128–137. [Google Scholar] [CrossRef]
- Lin, Y.; Lu, M.-F.; Liao, H.-B.; Li, Y.-X.; Han, W.; Yuan, K. Content determination of the flavonoids in the different parts and different species of Abelmoschus esculentus L. by reversed phase-high performance liquid chromatograph and colorimetric method. Pharmacogn. Mag. 2014, 10, 278–284. [Google Scholar] [CrossRef]
- Deng, Y.; Li, S.; Wang, M.; Chen, X.; Tian, L.; Wang, L.; Yang, W.; Chen, L.; He, F.; Yin, W. Flavonoid-rich extracts from okra flowers exert antitumor activity in colorectal cancer through induction of mitochondrial dysfunction-associated apoptosis, senescence and autophagy. Food Funct. 2020, 11, 10448–10466. [Google Scholar] [CrossRef]
- Chaemsawang, W.; Prasongchean, W.; Papadopoulos, K.I.; Ritthidej, G.; Sukrong, S.; Wattanaarsakit, P. The Effect of Okra (Abelmoschus esculentus (L.) Moench) Seed Extract on Human Cancer Cell Lines Delivered in Its Native Form and Loaded in Polymeric Micelles. Int. J. Biomater. 2019, 2019, 9404383. [Google Scholar] [CrossRef]
- Ping, M.-H. Hyperin Controls the Development and Therapy of Gastric Cancer via Regulating Wnt/β-Catenin Signaling. Cancer Manag. Res. 2020, 12, 11773–11782. [Google Scholar] [CrossRef]
- Hong, C.-E.; Park, A.-K.; Lyu, S.-Y. Synergistic anticancer effects of lectin and doxorubicin in breast cancer cells. Mol. Cell. Biochem. 2014, 394, 225–235. [Google Scholar] [CrossRef] [PubMed]
- Yau, T.; Dan, X.; Ng, C.C.W.; Ng, T.B. Lectins with Potential for Anti-Cancer Therapy. Molecules 2015, 20, 3791–3810. [Google Scholar] [CrossRef] [PubMed]
- Figueiroa, E.D.O.; Da Cunha, C.R.A.; Albuquerque, P.B.; De Paula, R.A.; Aranda-Souza, M.; Da Silva, L.C.N.; Zagmignan, A.; Carneiro-Da-Cunha, M.G.; Da Silva, L.C.N.; Correia, M.T.D.S. Lectin-Carbohydrate Interactions: Implications for the Development of New Anticancer Agents. Curr. Med. Chem. 2017, 24, 3667–3680. [Google Scholar] [CrossRef]
- Monte, L.G.; Santi-Gadelha, T.; Reis, L.B.; Braganhol, E.; Prietsch, R.F.; Dellagostin, O.A.; Lacerda, E.R.R.; Gadelha, C.A.A.; Conceição, F.R.; Pinto, L.S. Lectin of Abelmoschus esculentus (okra) promotes selective antitumor effects in human breast cancer cells. Biotechnol. Lett. 2013, 36, 461–469. [Google Scholar] [CrossRef]
- Sharma, N.; Sabyasachi, S. Immunomodulatory Potential of Phytochemicals: Recent Updates; Springer Nature: Singapore, 2019. [Google Scholar]
- Hosseinzade, A.; Sadeghi, O.; Biregani, A.N.; Soukhtehzari, S.; Brandt, G.S.; Esmaillzadeh, A. Immunomodulatory Effects of Flavonoids: Possible Induction of T CD4+ Regulatory Cells Through Suppression of mTOR Pathway Signaling Activity. Front. Immunol. 2019, 10, 51. [Google Scholar] [CrossRef]
- Soares, G.S.F.; Assreuy, A.M.S.; Gadelha, C.A.A.; Gomes, V.M.; Delatorre, P.; Simões, R.C.; Cavada, B.S.; Leite, J.F.; Nagano, C.S.; Pinto, N.V.; et al. Purification and Biological Activities of Abelmoschus esculentus Seed Lectin. Protein J. 2012, 31, 674–680. [Google Scholar] [CrossRef]
- Ortaç, D.; Cemek, M.; Karaca, T.; Büyükokuroğlu, M.E.; Özdemir, Z.; Kocaman, A.T.; Göneş, S. In vivo anti-ulcerogenic effect of okra (Abelmoschus esculentus) on ethanol-induced acute gastric mucosal lesions. Pharm. Biol. 2018, 56, 165–175. [Google Scholar] [CrossRef]
- Sheu, S.-C.; Lai, M.-H. Composition analysis and immuno-modulatory effect of okra (Abelmoschus esculentus L.) extract. Food Chem. 2012, 134, 1906–1911. [Google Scholar] [CrossRef]
- Chen, H.; Jiao, H.; Cheng, Y.; Xu, K.; Jia, X.; Shi, Q.; Guo, S.; Wang, M.; Du, L.; Wang, F. In Vitro and In Vivo Immunomodulatory Activity of Okra (Abelmoschus esculentus L.) Polysaccharides. J. Med. Food 2016, 19, 253–265. [Google Scholar] [CrossRef]
- De Carvalho, C.C.C.R.; Cruz, P.A.; Da Fonseca, M.M.R.; Xavier-Filho, L. Antibacterial properties of the extract of Abelmoschus esculentus. Biotechnol. Bioprocess Eng. 2011, 16, 971–977. [Google Scholar] [CrossRef]
- Lengsfeld, C.; Titgemeyer, F.; Faller, G.; Hensel, A. Glycosylated compounds from okra inhibit adhesion of Helicobacter pylori to human gastric mucosa. J. Agric. Food Chem. 2004, 52, 1495–1503. [Google Scholar] [CrossRef] [PubMed]
- Fan, S.; Zhang, Y.; Sun, Q.; Yu, L.; Li, M.; Zheng, B.; Wu, X.; Yang, B.; Li, Y.; Huang, C. Extract of okra lowers blood glucose and serum lipids in high-fat diet-induced obese C57BL/6 mice. J. Nutr. Biochem. 2014, 25, 702–709. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Zhao, Y.; Wu, Q.; John, A.; Jiang, Y.; Yang, J.; Liu, H.; Yang, B. Structure characterisation of polysaccharides in vegetable “okra” and evaluation of hypoglycemic activity. Food Chem. 2018, 242, 211–216. [Google Scholar] [CrossRef]
- Damodaran, D.; Jeyakani, J.; Chauhan, A.; Kumar, N.; Chandra, N.; Surolia, A. CancerLectinDB: A database of lectins relevant to cancer. Glycoconj. J. 2007, 25, 191–198. [Google Scholar] [CrossRef] [PubMed]
- Pienta, K.J.; Nailk, H.; Akhtar, A.; Yamazaki, K.; Replogle, T.S.; Lehr, J.; Donat, T.L.; Tait, L.; Hogan, V.; Raz, A. Inhibition of Spontaneous Metastasis in a Rat Prostate Cancer Model by Oral Administration of Modified Citrus Pectin. JNCIJ. Natl. Cancer Inst. 1995, 87, 348–353. [Google Scholar] [CrossRef]
- Nangia-Makker, P.; Nakahara, S.; Hogan, V.; Raz, A. Galectin-3 in apoptosis, a novel therapeutic target. J. Bioenerg. Biomembr. 2007, 39, 79–84. [Google Scholar] [CrossRef]
- Olano-Martin, E.; Rimbach, G.H.; Gibson, G.R.; Rastall, R.A. Pectin and pectic-oligosaccharides induce apoptosis in in vitro human colonic adenocarcinoma cells. Anticancer Res. 2003, 23, 341–346. [Google Scholar]
- Vayssade, M.; Sengkhamparn, N.; Verhoef, R.; Delaigue, C.; Goundiam, O.; Vigneron, P.; Voragen, A.G.; Schols, H.A.; Nagel, M.-D. Antiproliferative and proapoptotic actions of okra pectin on B16F10 melanoma cells. Phytother. Res. 2009, 24, 982–989. [Google Scholar] [CrossRef]
- Soma Das, G.N.; Ghosh, L.K. Okra and its various applications in Drug Delivery, Food Technology, Health Care and Pharmacological Aspects—A Review. J. Pharm. Sci. Res. 2019, 11, 2139–2147. [Google Scholar]
- Geng, S.; Liu, Y.; Ma, H.; Chen, C. Extraction and Antioxidant Activity of Phenolic Compounds from Okra Flowers. Trop. J. Pharm. Res. 2015, 14, 807. [Google Scholar] [CrossRef]
- Hu, L.; Yu, W.; Li, Y.; Prasad, N.; Tang, Z. Antioxidant Activity of Extract and Its Major Constituents from Okra Seed on Rat Hepatocytes Injured by Carbon Tetrachloride. BioMed Res. Int. 2014, 2014, 341291. [Google Scholar] [CrossRef] [PubMed]
- Hurrell, R.F. Influence of Vegetable Protein Sources on Trace Element and Mineral Bioavailability. J. Nutr. 2003, 133, 2973S–2977S. [Google Scholar] [CrossRef] [PubMed]
- Faruque, A.; Ashraf, S.A.; Azad, A.A.; Prasad, P.B. Production and development of nutraceuticals using Bacillus subtilis NCIM 2708 under solid statefermentation by response surface methodology. Eur. Sci. J. 2013, 9. [Google Scholar] [CrossRef]
- Subrahmanyam, G.V.; Sushma, M.; Alekya, A.; Neeraja, C.; Harsha, H.S.S.; Ravindra, J. Antidiabetic Activity of Abelmoschus esculentus Fruit Extract. Int. J. Res. Pharm. Chem. 2011, 1, 17–20. [Google Scholar]
- Thöle, C.; Brandt, S.; Ahmed, N.; Hensel, A. Acetylated Rhamnogalacturonans from Immature Fruits of Abelmoschus esculentus Inhibit the Adhesion of Helicobacter pylori to Human Gastric Cells by Interaction with Outer Membrane Proteins. Molecules 2015, 20, 16770–16787. [Google Scholar] [CrossRef]
- Messing, J.; Thöle, C.; Niehues, M.; Shevtsova, A.; Glocker, E.; Borén, T.; Hensel, A. Antiadhesive Properties of Abelmoschus esculentus (Okra) Immature Fruit Extract against Helicobacter pylori Adhesion. PLoS ONE 2014, 9, e84836. [Google Scholar] [CrossRef]
- Dubey, P.; Mishra, S. A Review on: Diabetes and Okra (Abelmoschus esculentus). J. Med. Plants Stud. 2017, 5, 23–26. [Google Scholar]
- Abi, I.; Abi, L.; Ladan, M.J. Hypoglycaemic Effect of Abelmoschus Esculentus Extracts in Alloxan-Induced Diabetic Wistar Rats. Endocrinol. Diabetes Res. 2017, 3. [Google Scholar] [CrossRef]
- Yaradua, I.; Ibrahim, M.; Matazu, K.I.; Nasir, A.; Matazu, N.U.; Zainab, A.S.; Abdul Rahman, M.B.; Bilbis, L.; Abbas, A.Y. Antidiabetic Activity of Abelmoschus esculentus (Ex-Maradi Okra) Fruit in Alloxan-induced Diabetic Rats. Niger. J. Biochem. Mol. Biol. 2017, 32, 44–52. [Google Scholar]
- Abbas, A.; Muhammad, I.; Abdulrahman, M.B.; Bilbis, L.S.; Saidu, Y.; Onu, A. Possible Antidiabetic Mechanism of Action of Ex-maradi Okra Fruit Variety (Abelmoscus esculentus) on Alloxan Induced Diabetic Rats. Niger. J. Basic Appl. Sci. 2018, 25, 101. [Google Scholar] [CrossRef]
- Majd, N.E.; Tabandeh, M.R.; Shahriari, A.; Soleimani, Z. Okra (Abelmoscus esculentus) Improved Islets Structure, and Down-Regulated PPARs Gene Expression in Pancreas of High-Fat Diet and Streptozotocin-Induced Diabetic Rats. Cell J. 2018, 20, 31–40. [Google Scholar] [CrossRef]
- Nguekouo, P.T.; Mbaveng, A.T.; Kengne, A.P.N.; Woumbo, C.Y.; Tekou, F.A.; Oben, J.E. Effect of boiling and roasting on the antidiabetic activity of Abelmoschus esculentus (Okra) fruits and seeds in type 2 diabetic rats. J. Food Biochem. 2018, 42, e12669. [Google Scholar] [CrossRef]
- Hossen, M.A.; Jahan, I.; Mamun, M.A.A.; Sakir, J.A.M.S.; Shamimuzzaman, M.; Uddin, M.J.; Haque, M.E. CNS depressant and analgesic activities of Okra (Abelmoschus esculentus Linn.). Mol. Clin. Pharmacol. 2013, 4, 44–52. [Google Scholar]
- Wang, H.; Chen, G.; Ren, D.; Yang, S.-T. Hypolipidemic Activity of Okra is Mediated through Inhibition of Lipogenesis and Upregulation of Cholesterol Degradation. Phytother. Res. 2013, 28, 268–273. [Google Scholar] [CrossRef]
- Medina-Remón, A.; Casas, R.; Tressserra-Rimbau, A.; Ros, E.; González, M.M.; Fitó, M.; Corella, D.; Salas-Salvadó, J.; Raventós, R.M.L.; Estruch, R.; et al. Polyphenol intake from a Mediterranean diet decreases inflammatory biomarkers related to atherosclerosis: Asubstudy of the PREDIMED trial. Br. J. Clin. Pharmacol. 2016, 83, 114–128. [Google Scholar] [CrossRef]
- Doreddula, S.K.; Bonam, S.R.; Gaddam, D.P.; Desu, B.S.R.; RamaRao, N.; Pandy, V. Phytochemical Analysis, Antioxidant, Antistress, and Nootropic Activities of Aqueous and Methanolic Seed Extracts of Ladies Finger (Abelmoschus esculentus L.) in Mice. Sci. World J. 2014, 2014, 519848. [Google Scholar] [CrossRef]
- Adnan, M.; Patel, M.; Deshpande, S.; Alreshidi, M.; Siddiqui, A.J.; Reddy, M.N.; Emira, N.; De Feo, V. Effect of Adiantumphilippense Extract on Biofilm Formation, Adhesion with Its Antibacterial Activities Against Foodborne Pathogens, and Characterization of Bioactive Metabolites: An in vitro-in silico Approach. Front. Microbiol. 2020, 11, 823. [Google Scholar] [CrossRef]
- Ashraf, S.A.; Adnan, M.; Patel, M.; Siddiqui, A.J.; Sachidanandan, M.; Snoussi, M.; Hadi, S.S. Fish-Based Bioactives as Potent Nutraceuticals: Exploring the Therapeutic Perspective of Sustainable Food from the Sea. Mar. Drugs 2020, 18, 265. [Google Scholar] [CrossRef]
- Ahmad, F.; Ashraf, S.A.; Ahmad, F.A.; Ansari, J.A.; Siddiquee, R.A. Nutraceutical Market and its Regulation. Am. J. Food Technol. 2011, 6, 342–347. [Google Scholar] [CrossRef]
- Ashraf, S.A.; ElKhalifa, A.E.O.; Siddiqui, A.J.; Patel, M.; AwadElkareem, A.M.; Snoussi, M.; Ashraf, M.S.; Adnan, M.; Hadi, S.S. Cordycepin for Health and Wellbeing: A Potent Bioactive Metabolite of an Entomopathogenic Cordyceps Medicinal Fungus and Its Nutraceutical and Therapeutic Potential. Molecules 2020, 25, 2735. [Google Scholar] [CrossRef]
- Roy, A.; Shrivastava, S.L.; Mandal, S.M. Functional properties of Okra Abelmoschus esculentus L. (Moench): Traditional claims and scientific evidences. Plant Sci. Today 2014, 1, 121–130. [Google Scholar] [CrossRef]
- Zaharuddin, N.D.; Noordin, M.I.; Kadivar, A. The Use of Hibiscus esculentus (Okra) Gum in Sustaining the Release of Propranolol Hydrochloride in a Solid Oral Dosage Form. BioMed Res. Int. 2014, 2014, 735891. [Google Scholar] [CrossRef] [PubMed]
- Dwyer, J.T.; Coates, P.M.; Smith, M.J. Dietary Supplements: Regulatory Challenges and Research Resources. Nutrients 2018, 10, 41. [Google Scholar] [CrossRef] [PubMed]
- Petropoulos, S.A.; Fernandes, A.; Barros, L.; Ferreira, I.C.F.R. Chemical composition, nutritional value and antioxidant properties of Mediterranean okra genotypes in relation to harvest stage. Food Chem. 2018, 242, 466–474. [Google Scholar] [CrossRef] [PubMed]
- Institute of Medicine Standing (US) Committee on the Scientific Evaluation of Dietary Reference Intakes and its Panel on Folate, Other B Vitamins, and Choline. The National Academies Collection: Reports funded by National Institutes of Health. In Dietary Reference Intakes for Thiamin, Riboflavin, Niacin, Vitamin B(6), Folate, Vitamin B(12), Pantothenic Acid, Biotin, and Choline; National Academies Press: Washington, DC, USA, 1998. [Google Scholar]
- Gasperi, V.; Sibilano, M.; Savini, I.; Catani, M.V. Niacin in the Central Nervous System: An Update of Biological Aspects and Clinical Applications. Int. J. Mol. Sci. 2019, 20, 974. [Google Scholar] [CrossRef] [PubMed]
- Bakre, L.G.; Jaiyeoba, K.T. Effects of drying methods on the physicochemical and compressional characteristics of Okra powder and the release properties of its metronidazole tablet formulation. Arch. Pharmacal Res. 2009, 32, 259–267. [Google Scholar] [CrossRef] [PubMed]
- Jeong, W.-S.; Kong, A.-N.T. Biological Properties of Monomeric and Polymeric Catechins: Green Tea Catechins and Procyanidins. Pharm. Biol. 2004, 42 (Suppl. S1), 84–93. [Google Scholar] [CrossRef]
- Adelakun, O.; Oyelade, O.; Ade-Omowaye, B.; Adeyemi, I.; Van De Venter, M. Chemical composition and the antioxidative properties of Nigerian Okra Seed (Abelmoschus esculentus Moench) Flour. Food Chem. Toxicol. 2009, 47, 1123–1126. [Google Scholar] [CrossRef]
- Diwan, A.; Ninawe, A.; Harke, S. Gene editing (CRISPR-Cas) technology and fisheries sector. Can. J. Biotechnol. 2017, 1, 65–72. [Google Scholar] [CrossRef]
- Adelakun, O.E.; Oyelade, O.J. Chemical and Antioxidant Properties of Okra (Abelmoschus esculentus Moench) Seed. Nuts Seeds Health Dis. Prev. 2011, 841–846. [Google Scholar] [CrossRef]
- Adetuyi, F.; Ajala, L.; Ibrahim, T. Effect of the addition of defatted okra seed (Abelmoschus esculentus) flour on the chemical composition, functional properties and Zn bioavailability of plantain (Musa paradisiacal Linn) flour. J. Microbiol. Biotechnol. Food Sci. 2012, 2, 69–82. [Google Scholar]
- Jarret, R.L.; Wang, M.L.; Levy, I.J. Seed Oil and Fatty Acid Content in Okra (Abelmoschus esculentus) and Related Species. J. Agric. Food Chem. 2011, 59, 4019–4024. [Google Scholar] [CrossRef] [PubMed]
- Agostoni, C.; Moreno, L.; Shamir, R. Palmitic Acid and Health: Introduction. Crit. Rev. Food Sci. Nutr. 2015, 56, 1941–1942. [Google Scholar] [CrossRef] [PubMed]
- Dong, Z.; Zhang, J.; Tian, K.-W.; Pan, W.-J.; Wei, Z.-J. The Fatty Oil from Okra Seed: Supercritical Carbon Dioxide Extraction, Composition and Antioxidant Activity. Curr. Top. Nutraceutical Res. 2014, 12, 75–84. [Google Scholar]
- López-Huertas, E. Health effects of oleic acid and long chain omega-3 fatty acids (EPA and DHA) enriched milks. A review of intervention studies. Pharmacol. Res. 2010, 61, 200–207. [Google Scholar] [CrossRef]
- Jandacek, R.J. Linoleic Acid: A Nutritional Quandary. Healthcare 2017, 5, 25. [Google Scholar] [CrossRef]
- Steyn, N.P.; Mchiza, Z.; Hill, J.; Davids, Y.D.; Venter, I.; Hinrichsen, E.; Opperman, M.; Rumbelow, J.; Jacobs, P. Nutritional contribution of street foods to the diet of people in developing countries: A systematic review. Public Health Nutr. 2013, 17, 1363–1374. [Google Scholar] [CrossRef]
- Slavin, J.; Carlson, J. Carbohydrates. Adv. Nutr. 2014, 5, 760–761. [Google Scholar] [CrossRef]
- Xiao, J.B.; Capanoglu, E.; Jassbi, A.R.; Miron, A. Advance on the FlavonoidC-glycosides and Health Benefits. Crit. Rev. Food Sci. Nutr. 2015, 56, S29–S45. [Google Scholar] [CrossRef]
- Sunilson, J.J.; Jayaraj, P.; Mohan, M.; Kumari, A.G.; Varatharajan, R. Antioxidant and hepatoprotective effect of the roots of Hibiscus esculentus Linn. Int. J. Green Pharm. 2008, 2, 200. [Google Scholar] [CrossRef]
- Shenkin, A. Micronutrients in health and disease. Postgrad. Med. J. 2006, 82, 559–567. [Google Scholar] [CrossRef] [PubMed]
- Idris, S.; Yisa, J.; Itodo, A. Proximate and mineral composition of the leaves of Abelmoschus esculentus. Int. J. Trop. Agric. Food Syst. 2010, 3. [Google Scholar] [CrossRef]
- Caluête, M.; Souza, L.D.; Ferreira, E.D.S.; França, A.; Gadelha, C.D.A.; Aquino, J.S.; Santi-Gadelha, T. Nutritional, antinutritional and phytochemical status of okra leaves (Abelmoschus esculentus) subjected to different processes. Afr. J. Biotechnol. 2015, 14, 683–687. [Google Scholar]
- McCutcheon, A.; Ellis, S.; Hancock, R.; Towers, G. Antifungal screening of medicinal plants of British Columbian native peoples. J. Ethnopharmacol. 1994, 44, 157–169. [Google Scholar] [CrossRef]
- Kumar, S. Physicochemical, Phytochemical and toxicity studies on gum and mucilage from plant Abelmoschus esculentus. J. Phytopharm. 2014, 3, 200–203. [Google Scholar]
- Muhammad, I.; Matazu, I.K.; Yaradua, I.A.; Yau, S.; Nasir, A.; Bilbis, S.L.; Abbas, Y.A. Development of Okra-Based Antidiabetic Nutraceutical Formulation from Abelmoschus esculentus (L.) Moench (Ex-maradi Variety). Trop. J. Nat. Prod. Res. 2018, 2, 80–86. [Google Scholar] [CrossRef]
- Reddy, M.T.; Kadiyala, H.; Mutyala, G.; Hameedunnisa, B. Heterosis for Yield and Yield Components in Okra (Abelmoschus esculentus (L.) Moench). Chil. J. Agric. Res. 2012, 72, 316–325. [Google Scholar] [CrossRef]
- Dan-Dan, R.; Gu, C. Inhibition Effect of Okra Polysaccharides on Proliferation of Human Cancer Cell Lines. Food Sci. 2010, 31, 353–356. [Google Scholar] [CrossRef]
- M.R. Future, Seeds Market Research Report. 2020. Available online: https://www.marketresearchfuture.com/reports/okra-seeds-market-7715 (accessed on 11 October 2020).
- Wire, G.N. Okra Seeds Market Report Insights and Industry Analysis by Category (Conventional and Organic) and Region, Competitive Market Size, Share, Trends, and Forecast, 2018–2023. 2020. Available online: https://www.globenewswire.com/news-release/2019/07/25/1887743/0/en/Okra-Seeds-Market-Size-to-Reach-USD-352-7-Million-by-2023-at-9-8-CAGR-Predicts-Market-Research-Future.html (accessed on 15 October 2020).
- WBOC. Okra Seeds Market—Global Countries Data, 2020. Top Leading Countries, Companies, Consumption, Drivers, Trends, Forces Analysis, Revenue, Market Size & Growth, Global Forecast 2025. 2020. Available online: https://www.wboc.com/story/42977060/okra-seeds-market-2020-regional-analysis-with-top-countries-data-trends-definition-share-market-size-and-forecast-report-by-2026 (accessed on 2 December 2020).
- Uebelhack, R.; Bongartz, U.; Seibt, S.; Bothe, G.; Chong, P.-W.; De Costa, P.; Wszelaki, N. Double-Blind, Randomized, Three-Armed, Placebo-Controlled, Clinical Investigation to Evaluate the Benefit and Tolerability of Two Dosages of IQP-AE-103 in Reducing Body Weight in Overweight and Moderately Obese Subjects. J. Obes. 2019, 2019, 3412952. [Google Scholar] [CrossRef]
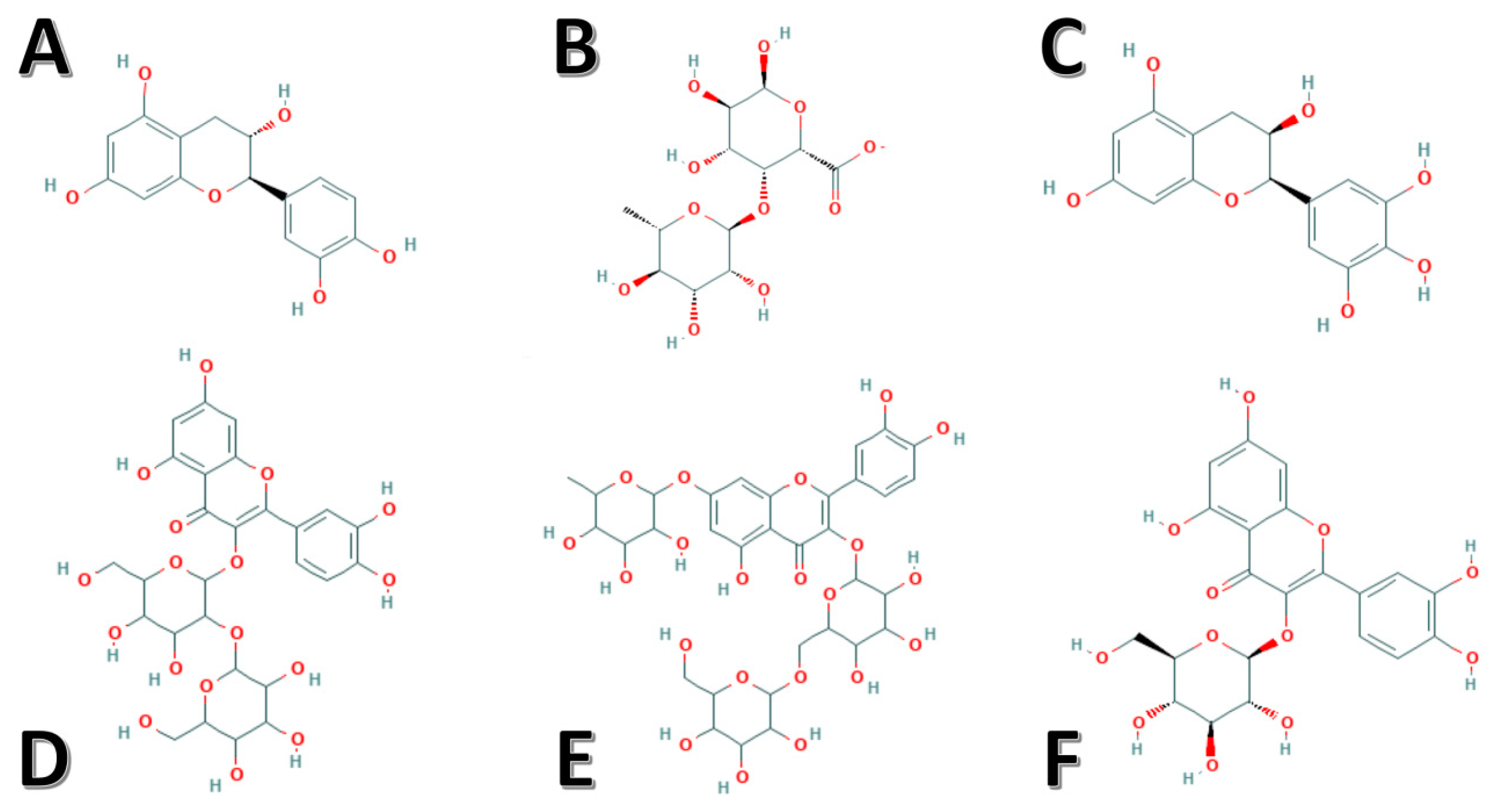
| S. No. | Dietary Constituents | Amount Per Serving | %DV * |
|---|---|---|---|
| 1. | Total calories | 130 kJ | 2 |
| 2. | Total carbohydrates | 7 g | 2 |
| 3. | Total protein | 2.0 g | 4 |
| 4. | Dietary fiber | 3.2 g | 13 |
| 5. | Starch | 0.3 g | - |
| 6. | Sugar | 1.2 g | - |
| 7. | Total fat | 0.1 g | - |
| 8. | Trans-fat | - | - |
| 9. | Saturated fat | 0.0 g | 0 |
| 10. | Cholesterol | 0.0 mg | 0 |
| 11. | Total omega-3 fatty acids | 0.001 g | - |
| 12. | Total omega-6 fatty acids | 0.026 g | - |
| 13. | Phytosterols | 0.024 g | - |
| S. No. | Minerals | Amount Per Serving | %DV * |
|---|---|---|---|
| 1. | Potassium | 303 mg | 9 |
| 2. | Calcium | 81.0 mg | 8 |
| 3. | Phosphorus | 63.0 mg | 6 |
| 4. | Magnesium | 57.0 mg | 14 |
| 5. | Copper | 0.1 mg | 5 |
| 6. | Selenium | 0.7 µg | 1 |
| 7. | Manganese | 1.0 mg | 50 |
| 8 | Zinc | 0.6 mg | 4 |
| 9. | Sodium | 8.0 mg | 0 |
| 10. | Iron | 0.8 mg | 4 |
| Bioactive Components | Therapeutic Benefits | Mechanisms of Action | Reference | |
|---|---|---|---|---|
| Polysaccharide | Antidiabetic | It helps to lower body weight and glucose levels, improve glucose tolerance, and decrease the total serum cholesterol levels in high-fat diet-fed C57BL/6 mice. | [79] | |
| Rhamnogalacturonan | Antidiabetic | Hypoglycemic effect, | [80] | |
| Lectins | Anticancer | Arrest the cell cycle and activate the caspase cascades. | [81] | |
| Inhibit cellular proliferation in human breast cancer in vitro. | [70] | |||
| Pectin | Anticancer | Involved in cell adhesion, growth, and survival, as well as tumor development and cancer prevention therapy. | [82,83,84] | |
| Antiproliferative and proapoptotic | Induce apoptosis and inhibit cellular proliferation. | [85] | ||
| Pectin | Lower bad cholesterol | Okra promotes cholesterol degradation and inhibits the production of fat in the body. It lowers bad cholesterol by altering the bile production in the intestines. This helps in eliminating the clots and deposited cholesterol. | [86] | |
| Polyphenolic compounds | Antioxidant | Extract exhibits a strong DPPH radical scavenging activity and reducing power. | [87] | |
| Quercetin 3-O-glucosyl (1→6) glucoside (QDG) and quercetin 3-O-glucoside (QG) | Antioxidant | Excellent reducing power and free radical scavenging capabilities, including DPPH, superoxide anions, and hydroxyl radicals. | [88] | |
| Vitamin C, calcium, iron, manganese, and magnesium | Antioxidant | Eliminating free radicals. | [86] | |
| Quercetin derivatives and epigallocatechin | Antioxidant | Inhibitory effects on the generation of reactive oxygen species (ROS). | [54] | |
| Polysaccharide | Metabolic disorders | Inhibition of LXR and PPAR signaling. | [79] | |
| Polyphenolic compounds, | Antioxidant | Perform the function of capturing free radicals and stopping the chain reactions. | [89] | |
| Vitamin A; B vitamins (B1, B2, B6); and vitamin C and traces of zinc, calcium, folic acid, and fiber | Pregnancy benefits | Folates prevent miscarriages. They are also beneficial in the formation of the neural tube of the fetus and protect these tubes, preventing defects. This helps prevent birth defects like spina bifida and can even stop constipation during pregnancy. | [86] | |
| Polyphenols like catechin and flavonoids like quercetin possess | Antifatigue effects | Decreased the levels of blood lactic acid (BLA) and BUN in the blood; MDA in the liver; and increased the levels of HG, SOD, and GSH in the liver during fatigue recovery, which proved that OSD could alleviate physical fatigue and promote recovery. | [51] | |
| Probiotics | Gut bacteria-friendly | Biosynthesis of the vitamin B complex. | [86] | |
| Glutathione | Detoxify liver, antioxidant | The slimy substance in okra contains substances that bind bile acid and cholesterol to detoxify the liver. | [86] | |
| Mucilaginous | Ulcer treatment | The slimy stuff in okra is alkaline. This helps in neutralizing the acid. Additionally, it provides a protective coating within the digestive tract, which speeds up the healing process of peptic ulcers. | [86] | |
| Mucilaginous with fiber | Relieves and prevents constipation | Bind toxins and lubricates the large intestines. This ensures effortless and normal bowel movement due to its natural laxative property. | [86] | |
| Vitamin K and C | Bone health and essential for the blood-clotting process. It also helps restore bone density and prevent osteoporosis. | Several mechanisms are suggested by which vitamin K can modulate bone metabolism. Besides the gamma-carboxylation of osteocalcin, a protein believed to be involved in bone mineralization, there is increasing evidence that vitamin K also positively affects the calcium balance, a key mineral in bone metabolism. | [86,90] | |
| Vitamin A, along with antioxidant contents such as lutein, xanthein, and carotenes | Improves vision | Okra contains beta-carotenes (precursor of vitamin A), xanthin, and lutein, all with antioxidant properties preventing eye problems like cataract and glaucoma. | [86] | |
| Glycosylated compounds | Antibacterial activity | Inhibit the adhesion of Helicobacter pylori to the human gastric mucosa. | [78] | |
| Rhamnogalacturonan Polysaccharides | Antiadhesive properties | Interrupt the adhesion of H. pylori to human stomach tissues via interfering with the outer membrane proteins. | [78,91,92,93] | |
| Polyphenols and flavonoids (okra seeds) | Antifatigue | Reduce the levels of BLA and BUN, enhancing hepatic glycogen storage and the promoting antioxidant ability by lowering the MDA level and increasing the SOD and GSH-PX levels. | [51] |
| Bioactive Components | Part | Health Benefits | Reference |
|---|---|---|---|
| Polyphenols Carotene | Pod | Important for eyesight, along with healthy skin. | [78,108] |
| Folic acid | Beneficial for fetus development. | [1,109] | |
| Thiamine | Improves the nervous system, brain, heart, stomach, muscles, and intestine functions. | [8,110] | |
| Riboflavin | Needed for growth and overall good health. | [111,112] | |
| Niacin | Keeps our nervous system, digestive system, and skin healthy. | [108,113] | |
| Vitamin C | Helps in the overall growth of the body and tissue repair. | [1,114] | |
| Oligomeric catechin | Seed | Prevents and is used for treating chronic ailments, e.g., cardiovascular diseases and cancer. | [10,115] |
| Flavonol derivatives | Improves vascular health, leading to a reduced risk of diseases. | [116,117] | |
| Lysine | Improves calcium absorption and retention. | [118,119] | |
| Palmitic acid | An important constituent of the cell membrane, with a critical role in protein palmitoylation and palmitoylated signal molecules. | [120,121] | |
| Oleic acid | Decreases the cholesterol levels and prevents heart diseases. | [122,123] | |
| Linoleic acid | Improves cardiovascular health. | [88,124] | |
| Carbohydrate | Roots | Prime energy source and fuel for the brain, kidney, heart, and muscles. | [125,126] |
| Flavonoids | Exhibits substantial anticancer, antioxidant, anti-inflammatory, and hepatoprotective activities. | [10,127] | |
| Minerals | Leaves | Helps in overall growth and body development. | [128,129] |
| Tannins | Accelerate blood clotting, reduces the serum lipid and blood pressure, and modulates the immune responses. | [130,131,132] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Elkhalifa, A.E.O.; Alshammari, E.; Adnan, M.; Alcantara, J.C.; Awadelkareem, A.M.; Eltoum, N.E.; Mehmood, K.; Panda, B.P.; Ashraf, S.A. Okra (Abelmoschus Esculentus) as a Potential Dietary Medicine with Nutraceutical Importance for Sustainable Health Applications. Molecules 2021, 26, 696. https://doi.org/10.3390/molecules26030696
Elkhalifa AEO, Alshammari E, Adnan M, Alcantara JC, Awadelkareem AM, Eltoum NE, Mehmood K, Panda BP, Ashraf SA. Okra (Abelmoschus Esculentus) as a Potential Dietary Medicine with Nutraceutical Importance for Sustainable Health Applications. Molecules. 2021; 26(3):696. https://doi.org/10.3390/molecules26030696
Chicago/Turabian StyleElkhalifa, Abd Elmoneim O., Eyad Alshammari, Mohd Adnan, Jerold C. Alcantara, Amir Mahgoub Awadelkareem, Nagat Elzein Eltoum, Khalid Mehmood, Bibhu Prasad Panda, and Syed Amir Ashraf. 2021. "Okra (Abelmoschus Esculentus) as a Potential Dietary Medicine with Nutraceutical Importance for Sustainable Health Applications" Molecules 26, no. 3: 696. https://doi.org/10.3390/molecules26030696
APA StyleElkhalifa, A. E. O., Alshammari, E., Adnan, M., Alcantara, J. C., Awadelkareem, A. M., Eltoum, N. E., Mehmood, K., Panda, B. P., & Ashraf, S. A. (2021). Okra (Abelmoschus Esculentus) as a Potential Dietary Medicine with Nutraceutical Importance for Sustainable Health Applications. Molecules, 26(3), 696. https://doi.org/10.3390/molecules26030696











