Abstract
Polycyclic endoperoxides are rare natural metabolites found and isolated in plants, fungi, and marine invertebrates. The purpose of this review is a comparative analysis of the pharmacological potential of these natural products. According to PASS (Prediction of Activity Spectra for Substances) estimates, they are more likely to exhibit antiprotozoal and antitumor properties. Some of them are now widely used in clinical medicine. All polycyclic endoperoxides presented in this article demonstrate antiprotozoal activity and can be divided into three groups. The third group includes endoperoxides, which show weak antiprotozoal activity with a reliability of up to 70%, and this group includes only 1.1% of metabolites. The second group includes the largest number of endoperoxides, which are 65% and show average antiprotozoal activity with a confidence level of 70 to 90%. Lastly, the third group includes endoperoxides, which are 33.9% and show strong antiprotozoal activity with a confidence level of 90 to 99.6%. Interestingly, artemisinin and its analogs show strong antiprotozoal activity with 79 to 99.6% confidence against obligate intracellular parasites which belong to the genera Plasmodium, Toxoplasma, Leishmania, and Coccidia. In addition to antiprotozoal activities, polycyclic endoperoxides show antitumor activity in the proportion: 4.6% show weak activity with a reliability of up to 70%, 65.6% show an average activity with a reliability of 70 to 90%, and 29.8% show strong activity with a reliability of 90 to 98.3%. It should also be noted that some polycyclic endoperoxides, in addition to antiprotozoal and antitumor properties, show other strong activities with a confidence level of 90 to 97%. These include antifungal activity against the genera Aspergillus, Candida, and Cryptococcus, as well as anti-inflammatory activity. This review provides insights on further utilization of polycyclic endoperoxides by medicinal chemists, pharmacologists, and the pharmaceutical industry.
1. Introduction
Polycyclic endoperoxides are a rare group of naturally occurring metabolites found in various parts of plants such as leaves, roots, bark, stems, seeds, fruits, and flowers [1,2,3,4,5,6,7,8,9,10,11,12,13,14,15,16,17]. In addition, they have been found in extracts of various types of marine invertebrates and algae, and endoperoxides are synthesized by various types of fungi, fungal endophytes, and other microorganisms [8,9,13,14,15,18,19,20].
As shown in recent years, many polycyclic endoperoxides, both natural and synthetic, have antimalarial effects [21,22]. It is known that malaria or “swamp fever” refers to a group of transmissible infectious diseases transmitted to humans by bites of female mosquitoes belonging to the genus Anopheles, caused by parasitic protists of the genus Plasmodium, mainly P. falciparum [23,24,25]. According to the WHO World Malaria Report, at the beginning of the 21st century, the incidence ranged from 350 to 500 million cases per year, of which 1 to 3 million ended in death [26,27]. In connection with these ominous data, any new sources of natural antimalarial agents are of great interest to medicine and pharmacology, as well as to the pharmaceutical industry [28,29,30].
In this review, we will look at rare and unusual polycyclic endoperoxides isolated from different terrestrial and marine sources. The biological activity of many polycyclic endoperoxides has not been determined, and we present the pharmacological activities detected experimentally and predicted based on the structure-activity relationships using the PASS (Prediction of Activity Spectra for Substances) software [31,32,33]. PASS estimates the probabilities of several thousand biological activities with an average accuracy of about 96%. Probability of belonging to the class of “actives” Pa is calculated for each activity, providing the assessment of the hidden pharmacological potential of the investigated natural polycyclic endoperoxides [2,7,13,14,17,31,32,33].
2. Polycyclic Endoperoxides Derived from Marine Sources
Marine algae (both microalgae and macrophytes) and invertebrates are the main source of biologically active secondary metabolites, which include hydrocarbons, terpenoids, lipids, steroids, carotenoids, aromatic compounds, and alkaloids, as well as mixed compounds containing heteroatoms and polycyclic endoperoxides [2,4,5,6,7,8,17,18,19,20,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57,58,59,60,61,62,63,64].
A series of polycyclic peroxides such as contrunculin B (1) as well as the trunuclin peroxides (2–7) were discovered in the extracts of Australian marine sponge Latrunculia conulosa [65], Latrunculia sp. [66] and found in an Okinawan sponge Sigmosceptrella sp. [67]. Structures (1–16) can be seen in Figure 1, and their biological activity is presented in Table 1. Two unusual endoperoxide diterpenoids (8 and 9) were isolated from the brown seaweed Taonia atomaria [68]. Cytotoxic 8,11-epidioxy-7-hydroxy-3,12,15(17)-cembratrien-16,2-olide called cembranolide C (or denticulatolide, 10) known as icthyotoxin was found in soft corals Lobophytum denticulatum, Sinularia mayi, and Sarcophyton crassocaule and its acetate (11) was also found in L. denticulatum extract [69,70,71,72].
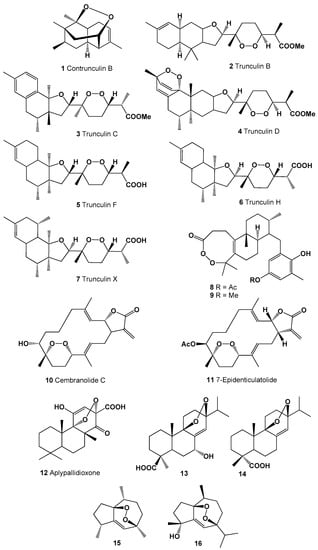
Figure 1.
Bioactive polycyclic endoperoxides derived from marine sources.

Table 1.
Biological activity of natural polycyclic peroxides derived from marine sources.
Norditerpenoid, aplypallidioxone (12) was detected in Australian encrusting sponge Aplysilla pallida [73], and two abietic acids (13 and 14) that were previously found in plants have also been found in green algae Elodea canadensis [74].
A guaiane-type sesquiterpene, 1,7-epidioxy-5-guaiene (15) was found and later isolated from Axinyssa sponge [75], and an oxygenated sesquiterpenoid, 1,7-epidioxy-5-guaien-4-ol called peroxygibberol (16), was isolated from a Formosan soft coral, Sinularia gibberosa, which demonstrated moderate cytotoxicity toward a human liver carcinoma cell line [76]. Structures (16–35) can be seen in Figure 2, and their biological activity is presented in Table 2.
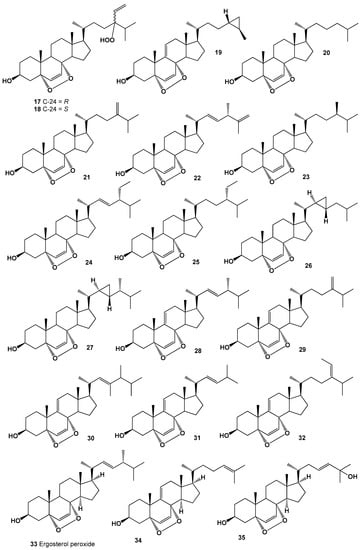
Figure 2.
Bioactive polycyclic endoperoxides derived from marine sources.

Table 2.
Biological activity of natural polycyclic peroxides derived from marine sources.
An extract of a marine sponge, Lendenfeldia chondrodes has led to the isolation and identification of two C-24 stereoisomers (17 and 18) of steroid, 5R,8R-epidioxy-24-hydroperoxy-cholesta-6,28(29)-dien-3α-ol. Obtained data with the molecular formula of steroid indicated that a hydroperoxy group and a vinyl group are attached at position-24 in both the R- and S- configurations [77], and cytotoxic steroid, (3β,5α,8α,24R,25R)-epidioxy-24,26-cyclocholesta-6,9(11)-dien-3-ol (19) was identified from Tethya sp. [78].
Interestingly, steroid, (3β,5α,8α)-epidioxycholest-6-en-3-ol (20) was found in three cone snail species, Conus ebraeus, C. leopardus, and C. tessulatus (family Conidae) [79], and was also present in the extract of polychaete worm Perinersis aibuhitensis [80], it was also isolated from the steroid fraction of sponges Axinella cannabina, Luffariella cf. variabilis [81,82], the tunicate Cynthia savignyi [83], and in long-spined sea urchin Diadema setosum [84]. Isolated steroid showed antibacterial, antifungal, and cytotoxic activities [81,82,83,84]. Detection of this steroid in various species of marine invertebrates could indicate that they all share a food chain, and the source of this steroid may be algae.
(3β,5α,8α)-Epidioxy-24-methylenecholest-6-en-3-ol (21) has been isolated from the several marine invertebrates, tunicate Ascidia nigra, pillar coral Dendrogyra cylindrus, marine sponge Thalysias juniperina, and sea hare Aplysia dactylomela [85]; in addition, this steroid was found in the tunicates Dendrodoa grossularia and Ascidiella aspersa, the gastropoda Aplysia depilans and Aplysia punctata [86], the sea anenome Metridium senile [87], and the sponge Tethya aurantia [88].
(3β,5α,8α,22E,24S)-Epidioxy-24-methylcholesta-6,22,25-trien-3-ol called axinysterol (22), (3β,5α,8α,24R)-Epidioxy-24-methylcholest-6-en-3-ol (23) and (3β,5α,8α,22E,24R)-Epidioxystigmasta-6,22-dien-3-ol (24) were detected in MeOH extract of the marine sponge Luffariella cf. variabilis [85].
22,23-Dihydro-5,8-epidioxystigmast-6-en-3-ol (25) was surrounded by Luffariella cf. variabilis, Tethya sp., and sea squirt Dendrodoa grossularia [82,85,86,87,88]. (3β,5α,8α)-Epidioxy-22,23-cyclopropacholest-6-en-3-ol (26) and (3β,5α,8α)-endoperoxy-23-demethylgorgost-6-en-3-ol (27) were discovered in soft corals Sinularia maxima, S. gibberosa and Sinularia sp. [89,90].
(3β,5α,8α,22E,24S)-Epidioxyergosta-6,9(11),22-trien-3-ol (28) was found in two tunicates Ascidia nigra and Dendrogyra cylindrus and sponge Thalysias juniperina [82,85,91], and (3β,5α,8α)-epidioxy-24-methylcholesta-6,9(11),24(28)-trien-3-ol (29) was detected in Ascidia nigra [85,88].
(3β,5α,8α,22E,24R)-Epidioxy-23,24-dimethylcholesta-6,22-dien-3-ol (30) was isolated from MeOH extract of the single-celled algae Odontella aurita [92], and it was also found in edible mushrooms Lentinus edodes, which are also known as shiitake [93].
(3β,5α,8α,22E)-Epidioxy-24-norcholesta-6,22-dien-3-ol (31) was detected in the sea pen, opisthobranch mollusk Virgularia sp. [94], and in A. nigra, D. cylindrus, and T. juniperina [85]. (3β,5α,8α,24(28E))-Epidioxy-24-ethylcholesta-6,24(28)-dien-3-ol (32) has been isolated and structure elucidated from several tunicates, namely Ascidia nigra and Dendrogyra cylindrus, and (3β,5α,8α,24(28)Z)-form was detected in Dendrodoa grossularia [85,86].
Cytotoxic (3β,5α,8α,22E,24R)-epidioxyergosta-6,22-dien-3-ol (33), well-known as 5α,8α-peroxyergosterol, is the most widely distributed steroid in the plant kingdom, lichens and fungi [5,6], and is also found in marine sponges Axinella cannabina, Halichondria sp., Suberites carnosus, Spirastrella abata, Thalysias juniperina [85,95,96,97], the sea lily Gymnocrinus richeri [98], and tunicates Ascidia nigra, Dendrogyra cylindrus [88].
Two cytotoxic steroids, 5α,8α-epidioxy-cholesta-6,9(11),24-trien-3β-ol (34) and 5α,8α-epidioxy-cholesta-6,23-dien-3β,25-diol (35) were isolated from a marine sponge Monanchora sp. [99]. Series 5α,8α-epidioxysteroids: 20, 21, 23, 32, and 36–41 were isolated from the MeOH extracts of the Gorgonian Eunicella cavolini and the tunicate Trididemnum inarmatum. Compound (36), bearing a cyclopropyl moiety in the side chain, exhibited the highest antiproliferative activity [100]. Structures (36–41) can be seen in Figure 3, and their biological activity are presented in Table 3.
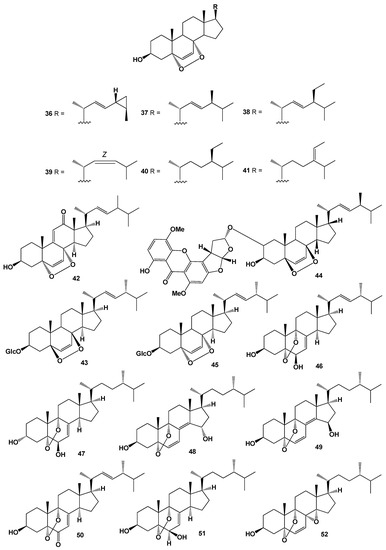
Figure 3.
Bioactive polycyclic endoperoxides derived from marine sources and fungi.

Table 3.
Biological activity of natural polycyclic peroxides derived from marine sources and fungi.
3. Polycyclic Endoperoxides Derived from Fungi and Fungal Endophytes
Fungi, fungal endophytes, myxomycetes, and the lichenized Ascomycetes are of great interest to pharmacologists and chemists, since they produce many biologically active substances, such as aromatic and phenolic compounds, tannins, hydrocarbons, lipids, unusual steroids, triterpenoids, heterocyclic compounds, peptides, and polycyclic endoperoxides [101,102,103,104,105,106,107,108,109,110,111,112,113,114].
In fungi, both cultivated and wild, polycyclic endoperoxides are found in small quantities, but ergosterol peroxide (33) is the most abundant [5,6]. Below, we present data on the distribution of this steroid and other polycyclic endoperoxides in fungi, fungal endophytes and lichens.
Trung and co-workers [115], using a modernized quantitative high-performance liquid chromatography method, found that ergosterol peroxide is present in wild mushrooms such as Fomitopsis dochmius, F. carneus, Daldinia concentrica, Ganoderma applanatum, G. lobatum, G. multiplicder G. lucidum, Phellinus igniarius, and Trametes gibbosa. In addition, this steroid has been detected in other species of wild fungi, fungal endophytes and lichens: Claviceps purpurea, Ganoderma lucidum, G. tsugae, G. sichuanense, Daedalea quercina, Piptoporus betulinus, Cryptoporus volvatus, Guignardia laricina, Lampteromyces japonicus, Botrytis cinerea, Lactarius uvidus, L. volemus, Cryptoporus volvatus, Dictyonema glabratum, Lasiosphaera nipponica, Gloeophyllum odoratum, Gymnopilus spectabilis, Hericium erinaceus, Hypsizigus marmoreus, Inonotus obliquus, I. radiatus, Lenzites betulina, Meripilus giganteus, Microporus flabelliformis, Naematoloma fasciculare, Phellinus pini, P. ribis, P. torulosus, Roseoformes subflexibilis, Pyropolyporus fomentarius, Pisolithus tinctorius, Polyporus tuberaster, Pseudephebe pubescens [5,6,17,116], and from the edible mushroom Volvariella volvacea [117]. In addition, ergosterol peroxide has been found in some Ascomycetes, Aspergillus sp., A. niger, A. oryzae, A. flavus, A. terreus, and A. fumigatus, Fusurium monilforme, F. osysporum, Penicillium rubrum, and P. sclerotigenum [5]. Ragasa [118] researched Philippine mushrooms and found ergosterol peroxide in Auricularia auricula-judae, Coprinopsis lagopus, Pleurotus florida, and Phellinus gilvus.
It is known that ergosterol peroxide isolated from edible or medicinal mushrooms demonstrates antitumor activity against colorectal cancer, hepatocellular carcinoma, prostate cancer, myeloma, and leukemia [119,120,121,122,123], and it also possesses antioxidant, anti-inflammatory, and antiviral activities, as well as induce the apoptosis of cancer cells [124,125,126,127,128].
Endoperoxide (42), bearing a keto group at the 12 position, has been isolated from the fungus Fusarium monilforme [129]. Endoperoxy glycoside (43) was detected in ethanol extract of the fungus Lactarius volemus, which demonstrated anticancer activity [130,131]. Ergosterol peroxide (33) and unusual steroid called asperversin A (44) have been isolated from endophytic fungus of Aspergillus versicolor that was isolated from the seaweed Sargassum thunbergii. Both steroid antibiotics showed antibacterial activity against Escherichia coli and Staphylococcus aureus [132], and another steroid named fuscoporianol D (45) was found in a MeOH extract of in field-grown mycelia of Inonotus obliquus [133].
Several steroids containing a 5,9-position peroxide moiety have been isolated from some mushroom extracts. For example, endoperoxide (46) was found in Boletus calopus white mushroom [134], and steroid (47) produces by two fungi Panellus serotinus and Lepista nuda [135]. Two steroids named nigerasterols A and B (48 and 49) were isolated from the extracts of an endophytic fungus of Aspergillus niger MA-132, which was isolated from the mangrove plant Avicennia marina [136], and steroids (49–52) were found in Buna shimeji and Pleurotus ostreatus [137]. A rare chamigrane-type sesquiterpenes called steperoxides A (53), B (54), C (55), and D (56) have been isolated from the hydnoid fungus Steccherinum ochraceum [Phanerochaetaceae]. Compound (53) demonstrated anticancer properties, and compounds (54 and 57) showed significant antimicrobial activity against Staphylococcus aureus [138,139,140,141]. Structures (53–68) can be seen in Figure 3, and their biological activity are presented in Table 4.

Table 4.
Bioactivity of natural polycyclic peroxides derived from fungi and fungal endophytes.
Anti-tumor nor-sesquiterpene endoperoxides called talaperoxide A (57), B (58), C (59), and D (60) were isolated from culture of fungi Talaromyces species HN21-3C, and from a mangrove endophytic fungus, Talaromyces flavus isolated from the mangrove plant Sonneratia apetala [142]. Isolated fungal metabolites demonstrated antineoplastic activity against MCF-7, MDA-MB-435, HepG2, HeLa, and PC-3 cancer cell lines [143,144]. Semi-synthetic derivative (61) of the fungal derived natural product showed the antiparasitic and cytotoxic activity against Trypanosoma brucei and Hela cells, respectively [145]. Chamigrane endoperoxide named merulin C (62), were isolated from the culture broth extract of an endophytic fungus of Xylocarpus granatum [146].
Caryophyllene-derived meroterpenoids, called cytosporolides A (63), B (64), and C (65), which have a unique peroxylactone skeleton, were isolated from cultures of the fungus Cytospora sp. Obtained metabolites demonstrated significant antimicrobial activity against the Gram-positive bacteria Staphylococcus aureus and S. pneumonia [147].
Two unprecedented spiroketal endoperoxides named chloropupukeanolides A (66) and B (67) were isolated from an endophytic fungus Pestalotiopsis fici. Compound (66) showed significant anti-HIV-1 and cytotoxic effects [148].
It is known that natural hypocrellin is a dark red dye with photodynamic activity against several microorganisms was isolated from the fungus Hypocrella bambusae, and its photooxidation product called peroxyhypocrellin (68) has an anthracene endoperoxide arrangement within the perylene quinone structure [149]. Structures (42–68) can be seen in Figure 3 and Figure 4, and their biological activity is presented in Table 3 and Table 4.
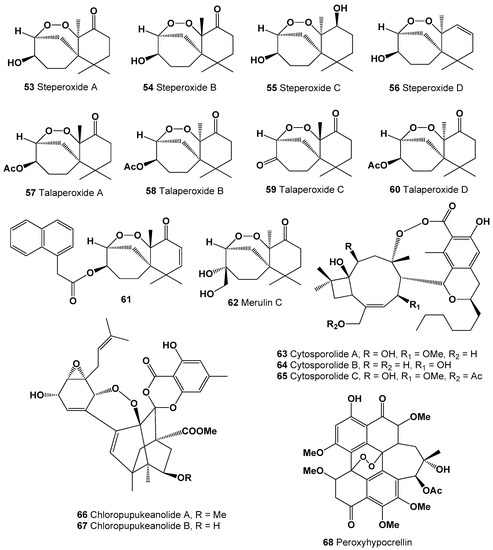
Figure 4.
Bioactive polycyclic endoperoxides derived from fungi and fungal endophytes.
4. Polycyclic Endoperoxides Derived from Plants and Liverworts
The largest amount of endoperoxides has been found, isolated and partially biological activity determined in plants and liverworts [1,2,5,6,8,9,14,19,150,151,152,153,154].
A peroxide-sesquetepene, called nardosaldehyde (69) was isolated from the roots of Nardostachys chinensis, and biological activity was not determined [155]. Structures (69–89) can be seen in Figure 5, and their biological activity is presented in Table 5 and Table 6. Peroxygibberol (16) is marine peroxide (5.9%) was also found in Agarwood oil obtained from highly infected Aquilaria malaccensis wood [156].
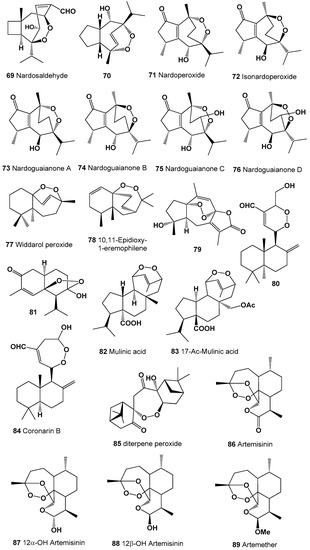
Figure 5.
Bioactive polycyclic endoperoxides derived from plants.

Table 5.
Biological activity of the natural polycyclic peroxides derived from plants.

Table 6.
Biological activity of the natural polycyclic peroxides derived from plants.
An antimalarial guaiane-type sesquiterpenoids (70), nardoperoxide (71), and isonardoperoxide (72) were isolated from Nardostachys chinensis roots [157,158,159], and in addition to this, nardoguaianone A (73), B (74), C (75), and D (76) were also highlighted from the same plant [160].
Widdarol peroxide (77) and its analogue (78) were found in hexane extract from the fruits of Schisandra grandiflora, which showed anti-proliferative activity against Hela (cervical cancer), A549 (lung cancer), DU-145 (prostate cancer), and MCF-7 (breast cancer) cancer cell lines [161].
Polycyclic sesquetepene, 1α,8α-epidioxy-4α-hydroxy-5αH-guai-7(11),9-dien-12,8-olide (79), which has anti-influenza viral properties, were isolated from the plant Curcuma wenyujin, which is mainly in the Wenzhou region of China [162] and was recently found in the flowering plant Acorus calamus [163]. Diterpenoid, (E,E)-15-hydroxylabda-8(17),11,13-trien-16-al (80) was detected in an extract of Alpinia chinensis [164]. Cadinane sesquiterpene, (−)-(5S,6S,7S,9R,10S)-7-hydroxy-5,7-epidioxycadinan-3-ene-2-one (81) was isolated and identified from the aerial part of the invasive plant Eupatorium adenophorum [165]. Diterpenoids called mulinic acid (82) and 17-acetoxymulinic acid (83) have been isolated from the aerial parts of Mulinum crassifolium (Umbelliferae) [166,167], and semi-synthetic derivatives (94, 95 and 96) were obtained from mulinic acid [168,169].
A cytotoxic seven-membered endoperoxide hemiacetal called coronarin B (84) was isolated from the flowers of Alpinia chinensis and Hedychium coronarium [164,170,171]. Unusual diterpene peroxide (85), with potent activity against Plasmodium falciparum, has been isolated from Amomum krervanh [172].
Endoperoxide called artemisinin (86) was found in 1979 in the extract of the Chinese herb qinghaosu (Artemisia annua) [173]. Currently, artemisinin and its derivatives (87–93) are widely used throughout the world as antimalarial drugs against the protozoan parasites [174,175,176,177]. An interesting mechanism of action for these compounds appears to involve heme-mediated degradation of the endoperoxide bridge to form carbon-centered free radicals, and these free radicals are selectively toxic to malaria parasites [178,179,180]. Artemisinin and its derivatives exhibit antitumor, antifungal, and other activities [181,182,183]. Structures (90–114) can be seen in Figure 6, and their biological activity is presented in Table 6 and Table 7.
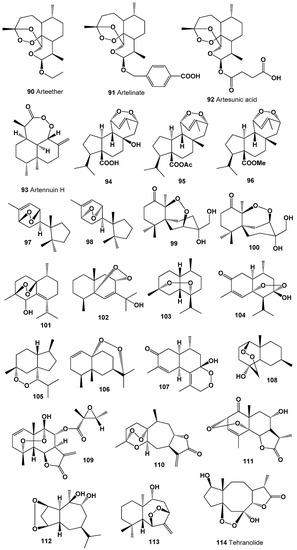
Figure 6.
Bioactive polycyclic endoperoxides derived from plants and liverworts.

Table 7.
Biological activity of natural polycyclic peroxides derived from plants.
Endoperoxy cuparene-type sesquiterpenoids (97 and 98, structures are shown in Figure 6, and activity is shown in Table 7) were identified from the Japanese liverwort Jungermannia infusca [184,185]. The chamigranes called merulin B (99) and C (100) have been found in an extract of the culture broth of a Thai mangrove-derived fungus [186,187].
Muurolane sesquiterpene endoperoxide, 1,4-peroxy-5-hydroxy-muurol-6-ene (101) has been obtained from plant Illicium tsangii (family Schisandraceae) [188,189,190]. The peroxide called schisansphene A (102) was isolated from the plant Schisandra sphenanthera, also known as the magnolia berry [191].
Highly oxygenated sesquiterpene (+)-muurolan-4,7-peroxide (103) was found in the essential oil of the liverwort Plagiochila asplenioides [192], and two sesquiterpene endoperoxides (104 and 105) were isolated from the aerial parts of the invasive plant Eupatorium adenophorum [193,194]. Unusual endoperoxide (106) was detected in the Ligularia veitchiana [195], compound (107) was isolated from the leaves of Eupatorium adenophorum [196], and metabolite (108) was found in extracts of the Xylopia emarginata [197]. The aerial parts of Montanoa hibiscifolia afforded rare endoperoxide (109) [198].
The xanthane-type sesquiterpenoid 4β,5β-epoxyxanthatin-1α,4α-endoperoxide (110) was found in the aerial parts of Xanthium strumarium [199], and 2α,5α-endoperoxide (111), which possess the 6α,12-eudesmanolide structure, was detected in areal parts of the Artemisia herba-alba [200]. The sesquiterpene peroxide (112) has been found from the aerial parts of Croton arboreous [201].
Allohimachalane peroxide (113) has been obtained from Illicium tsangii [188,189,190], and an unusual sesquiterpene lactone with endoperoxide group, called tehranolide (114) with strong antimalarial activity has been discovered in many Iranian Artemisia species: A. aucheri, A. austriaca, A. biennis, A. campestris, A. deserti, A. diffusa, A. gypsacea, A. haussknechtii, A. kermanensis, A. kopetdaghensis, A. kulbadica, A. oliveriana, A. persica, A. santolina, A. sieberi, A. tschernieviana, A. ciniformis, A. incana, A. turanica, and A. tournefortiana [202].
The hemiacetal of tricycloperoxyhumulone A (115) was detected in hops (Humulus lupulus) [203]. Structures (115–128) can be seen in Figure 7, and their biological activity is presented in Table 8. Highly oxygenated limonoid featuring an unprecedented 3,4-peroxide-bridged A-seco skeleton called walsuronoid A (116) was isolated from Walsura robusta (family Meliaceae). The isolated peroxide showed weak antimalarial activity [204].
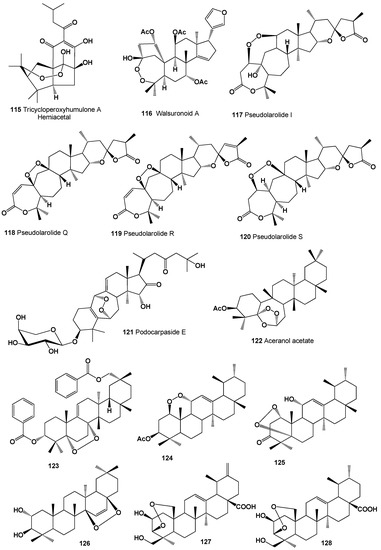
Figure 7.
Bioactive polycyclic endoperoxides derived from plants.

Table 8.
Biological activity of natural polycyclic peroxides derived from plants.
A cytotoxic peroxytriterpene dilactone called pseudolarolide I (117) has been isolated from the seeds of Pseudolarix kaempferi [205], and the leaves of P. kaemferi contains three triterpene peroxides, pseudolarolides Q (118), R (119), and S (120) [206]. An unusual glycoside, 3β,15α,25-trihydroxy-16,23-dioxo-6α,19α-epidioxy-9,10-seco-9,19-cyclolanost-5 (10),9(11)-diene 3-O-α-1-arabinopyranoside called podocarpaside E (121), was isolated from the roots of Actaea podocarpa [207].
A triterpene, 5α,6α-epidioxy-5β,6β-epoxy-9,13-dimethyl-25,26-dinoroleanan-3β-ol acetate, called aceranol acetate (122), which shows anti-inflammatory activity, was isolated from the stems and leaves of Acer mandshuricum [208]. The isolated compound also exhibited moderate activity against four human cancer cell lines (HL-60, SK-OV-3, A549, and HT-29).
A peroxy-multiflorane triterpene ester, (3α,5α,8α,20α)-5,8-epidioxymultiflora-6,9(11)-diene-3,29-diol 3,29-dibenzoate (123), was isolated from the processed seeds of Trichosanthes kirilowii. The obtained compound showed in vitro cytotoxicity against human-tumor cell lines (Hela, HL-60, and MCF-7) [209]. A peroxy triterpene, 3β-acetoxy-1β,11α-epidioxy-12-ursene (124), was isolated from the aerial roots of Ficus microcarpa [210]. An antimicrobial triterpenoid, 1α,5α-dioxy-11α-hydroxyurs-12-en-3-one (125), was found and obtained from the rhizome of Vladimiria muliensis [211].
The benzene extract of the bark of Sapium baccatum contained the nor-triterpene peroxide baccatin (126), which has been isolated and studied [212]. Two highly oxygenated ursane-type triterpenoids, (2β,3β)-3,25-epidioxy-2,24-dihydroxyursa-12,20(30)-dien-28-oic acid (127) and (2β,3β)-3,25-epidioxy-2,24-dihydroxyurs-12-en-28-oic acid (128), were detected in the EtOH extract of Gentiana aristata [213].
Highly oxygenated steroidal metabolites called physalin K (129) and Q (130) were found in extracts of the areal parts of Physalis alkekengi var. franchetii [214]. Structures (129–143) can be seen in Figure 8, and their biological activity is presented in Table 9. Plant withanolide called jaborosalactone 15 (131) was isolated from the flowering plant Jaborosa odonelliana, which was collected during autumn in Argentina [215]. Physangulidine G (132) was isolated from the aerial parts of Deprea bitteriana, D. cuyacensis, and D. zamorae [216].
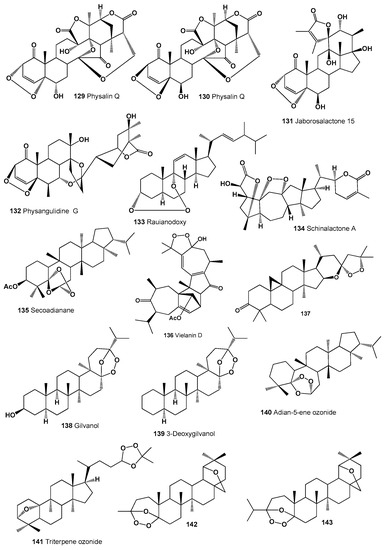
Figure 8.
Bioactive polycyclic endoperoxides derived from plants.

Table 9.
Biological activity of natural polycyclic peroxides derived from plants.
A unique compound, a 3,9-(1,2,3-trioxocine)-type steroid called rauianodoxy (133), and an ergosterol peroxide (33) were isolated from the Australian plant Rauia nodosa (family Rutaceae) [217]. An unusual endoperoxide called schinalactone A (134), which has a compressed ring A and shows anticancer activity against PANC-1 cell lines, was detected in the stems and roots of the magnolia vine, Schisandra sphenanthera [218].
A secoadianane-type steroid (135) was found and identified in the herbaceous plant Dorstenia brasiliensis (Moraceae) [219,220]. A polycyclic peroxide called vielanin D (136), which showed anti-plasmodial activity, was extracted from fresh and dry leaves of the plant Senecio selloi [221]. The peroxy steroid (16S,23R)-16,23-epoxy-23,25-epidioxycycloartan-3-one (137) was found in the Texas yellow-star, Lindheimera texana (Asteraceae) [222].
Two triterpenes, called gilvanol (138) and 3-deoxydilvanol (139), have been detected in the extracts of the red-bark oak, Quercus gilva [170,223]. An interesting endoperoxide, adian-5-ene ozonide (140) was found in the fern leaves of Adiantum monochlamys (Pteridaceae) and Oleandra wallichii (Davalliaceae), and another peroxide, a triterpene ozonide (141), was detected in the root extract of Senecio selloi [224,225].
Interesting and rare 1,2,4-trioxolanes (142 and 143) were derived from natural two allobetulin derivatives; however, biological activity has not been determined [226]. Two 9,13-diepoxy labdane diterpenoids called amoenolide K (144) and its 19-acetate (145) were detected in the areal parts from Amphiachyris amoena [227], and ent-8β,12α-epidioxy-12β-hydroxylabda-9(11),13-dien-15-oic acid γ-lactone (146) was obtained from the aerial parts of Premna oligotricha [228]. Structures (144–154) can be seen in Figure 9, and their biological activity is presented in Table 10.
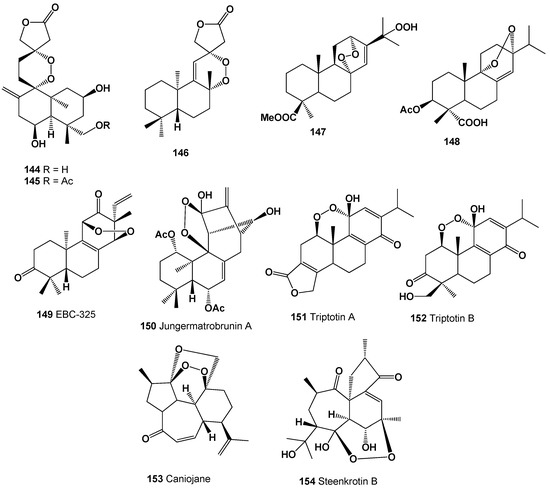
Figure 9.
Bioactive polycyclic endoperoxides derived from plants.

Table 10.
Biological activity of natural polycyclic peroxides derived from plants.
Diterpenic acids (13, 14, 147 and 148) have been identified from lipid extract of the different species. The diterpenic acid methyl ester (147) was isolated from the leaves of Moroccan Juniperus thurifera and J. phoenicea [229], compound (148) was detected in MeOH extract of Safvia oxyodon [230], and two abietic acids (13 and 14) were obtained from areal parts from the Abies marocana, Lepechinia caulescens, and Caryopteris nepetaefolia [231,232,233].
The diterpenoid endoperoxide called EBC-325 (149) was obtained from an extract of Croton insularis [234,235]. The diterpenoid endoperoxide called jungermatrobrunin A (150), detected in the liverwort Jungermannia atrobrunnea, has an unusual rearrangement-kaurene skeleton with a peroxide bridge [236], and two similar oxygenated diterpenes called triptotins A (151) and B (152) were found in extracts of the Tripterygium wilfordii [237]. The roots of Jatropha curcas contained peroxide caniojane (153) [238], and another peroxide called steenkrotin B (154) was found in ethanol extract of the leaves of Croton steenkampianus (Euphorbiaceae), which displayed mild anti-plasmodial activity [239].
Several adamantane type polycyclic polyprenylated acylphloroglucinols (155–164) possessing an unprecedented seco-adamantane architecture combined with a peroxide ring have been isolated and identified from extracts of some plants [240]. Thus, one compound called hypersubone B (155) was isolated from the leaves of Hypericum subsessile and exhibited significant cytotoxicity against four human cancer lines in vitro, HepG2, Eca109, HeLa, and A549 [241], and hyperisampsins N (157) and O (158), which exhibited significant cytotoxic activities toward HL-60 cells, were found in the aerial parts of H. sampsonii [242]. Structures (155–164) can be seen in Figure 10, and their biological activity is presented in Table 11.
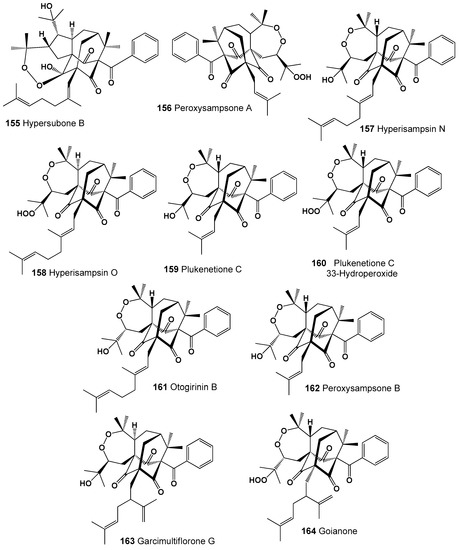
Figure 10.
Bioactive adamantane type polycyclic endoperoxides derived from plants.

Table 11.
Biological activity of natural polycyclic peroxides derived from plants.
Peroxysampsones A and B (156 and 162) were isolated from the roots of the Chinese medicinal plant H. sampsonii, and compound (156) showed comparable activity with norfloxacin against a NorA over-expressing multidrug-resistant strain of Staphylococcus aureus SA-1199B [243]. Two prenylated benzophenone derivatives, plukenetiones C (159) and hydroperoxide (160), have been isolated from the fruits of the Barbadian plant Clusia plukenetii [244], and otogirinin B (98) was detected in Hypericum erectum [245]. Garcimultiflorone G (163), which shows anti-inflammatory activity, was isolated from the fruits of Garcinia multiflora [246], and another polycyclic peroxide called goianone (164) was found in fruits extracts of Clusia rosea [247].
Unusual polycyclic endoperoxides pregnane glycosides named periplocosides A (165), B (166) C (167), D (168), K (169), F (170), and E (171) have been isolated from the antitumor fraction, which was obtained from the CHCl3 extract of Periploca sepium [248,249,250]. Structures (165–171) can be seen in Figure 11, and their biological activity is presented in Table 11.
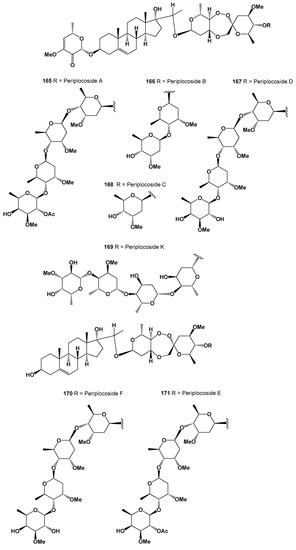
Figure 11.
Bioactive steroidal glycosides derived from plants.
5. Comparison of Biological Activities of Natural Polycyclic Endoperoxides
It is currently accepted that the biological activity of both natural and synthetic compounds depends on their structure [33,251,252]. Despite the activity cliffs observed for some drug-like compounds [253], which can be considered as a violation of this rule, structure-activity relationships (SAR) are widely used in medicinal chemistry for finding and optimization new pharmacological agents [254].
PASS is the first software for in silico estimation of biological activity profiles [33,255], of which the development has been started more than 30 years ago [256]. Its current implementation predicts about 8000 pharmacological effects, molecular mechanisms of action, pharmacological effects, toxicity, side effects, anti-targets, transporters-related interactions, gene expression regulation, and metabolic terms [31]. Due to the utilization of chemical descriptors that reflect the essential features of ligand-target interactions and a robust mathematical approach for analysis of structure-activity relationships, the average accuracy of PASS predictions was 96% [31,252,257,258]. Based on the PASS predictions provided by the appropriate web-service [259], over 29,000 researchers from 104 countries selected the most promising virtually designed molecules for synthesis and determined the optimal directions for testing their biological activity [260,261,262,263,264].
In this study, PASS predictions were used to estimate the general pharmacological potential for the analyzed natural polycyclic endoperoxides. For about 8000 pharmacological effects and molecular mechanisms of action, probabilities of belonging to the class of “actives” Pa, varied from zero to one, were estimated. The higher the Pa value is, the higher the probability of confirming the predicted activity in the experiment. On the other hand, estimated Pa values might be relatively small for some activities if the analyzed molecule is not like the active compounds from the PASS training set. Thus, PASS prediction interpretation requires considering two contradictory issues high probability of activity vs. high structural novelty. The researcher decides which issue is more critical, depending on the task or the project [18,31,35,257,258].
5.1. Antiprotozoal Activity of Natural Polycyclic Endoperoxides
Currently, about 120,000 articles have been published that are devoted to antiprotozoal and antiparasitic activities of both natural and synthetic compounds [265,266,267,268,269,270,271].
Analyzing the data obtained with PASS of natural polycyclic endoperoxides and artemisinin and its analogs currently used in medicine, it can be stated that for all polycyclic endoperoxides, antiprotozoal activity is estimated with a Pa from 70 to 99.6%. For some compounds, antiparasitic activity is also estimated, with a Pa from 50 to 88.3%. The antiprotozoal and antiparasitic activities predicted using the PASS are shown in Table 1, Table 2, Table 3, Table 4, Table 5, Table 6, Table 7, Table 8, Table 9, Table 10 and Table 11, and the chemical structures are shown in Figure 1, Figure 2, Figure 3, Figure 4, Figure 5, Figure 6, Figure 7, Figure 8, Figure 9, Figure 10 and Figure 11. A 3D graph of the predicted pharmacological activities of artemisinin (86) and its analogs is shown in Figure 12.
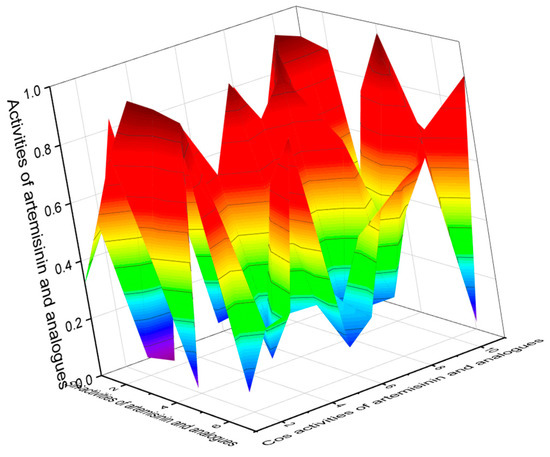
Figure 12.
The 3D graph shows the predicted and calculated pharmacological activities of artemisinin (86) and its analogs, such as 12α-OH-artemisinin (87), 12β-OH- artemisinin (88), artemether (89), arteether (90), artelinate (91), and artesunic acid (92). According to the PASS data, artemisinin and its analogs (86–92) show selective activity against obligate intracellular protozoan parasites belonging to the genera Plasmodium, Toxoplasma, Leishmania, and Coccidia, which is the main pharmacological activity with a confidence level of more than 90%. In addition, all these endoperoxides show antifungal activity against the opportunistic pathogenic yeasts Candida and Cryptococcus, as well as anticancer activity for some compounds; the confidence level exceeds 90 percent.
Artemisinin and its analogs (both natural and synthetic) are widely used in medical practice and are essential antimalarial treatment components. Figure 12 shows the predicted pharmacological activities of artemisinin and its analogs using PASS, and Figure 13 demonstrates the predicted pharmacological activities of artemisinin.
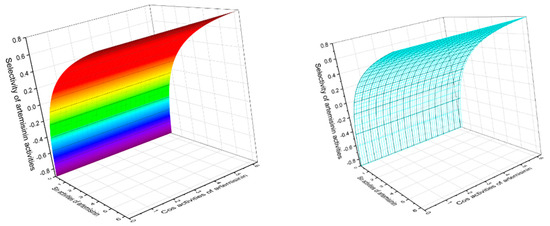
Figure 13.
The 3D graph shows the predicted and calculated pharmacological activities of artemisinin (86), which was found in 1979 in the extract of the Chinese herb Qinghaosu (Artemisia annua). According to PASS data, this endoperoxide demonstrated 16 different activities, with 5 activities having a found confidence of more than 90 percent. Antiprotozoal selective activity of artemisinin against obligate intracellular protozoan parasites belonging to the genera Plasmodium (99.5%), Toxoplasma (93%), Leishmania (92.3%), and Coccidia (78%) is the main pharmacological activity. In addition, artemisinin demonstrated strong anti-schistosomal activity (91.1%) against Schistosoma mansoni, a human blood fluke parasite. Additionally, artemisinin shows antifungal activity against an opportunistic pathogenic yeast Candida (91.5%) and Cryptococcus (85.3%), although anticancer activity is found at 80%.
5.2. Antitumor and Other Activities of Natural Polycyclic Endoperoxides
Many natural products exhibit antitumor and related activities and belong to different classes of chemical compounds, such as alkaloids, aromatic and phenolic metabolites, lipids, glycosides, and compounds containing acetylene or epoxy moieties [272,273,274,275,276,277,278]. These compounds also refer to various types of terpenoids, including steroids, triterpenoids, carotenoids, and polycyclic endoperoxides.
More than one million articles and reviews have been devoted to various antitumor and related activities of both natural and synthetic compounds. In an earlier section, we presented and discussed the antitumor activity of polycyclic endoperoxides isolated from various terrestrial and aquatic organisms computed using PASS.
According to the PASS estimates presented in Table 1, Table 2, Table 3, Table 4, Table 5, Table 6, Table 7, Table 8, Table 9, Table 10 and Table 11, many endoperoxides demonstrate antitumor and related activities to varying degrees. However, we are interested in compounds for which such activity is estimated with more than 95% probability. Figure 14 demonstrates natural compounds and their predicted antitumor activity with Pa > 95%.
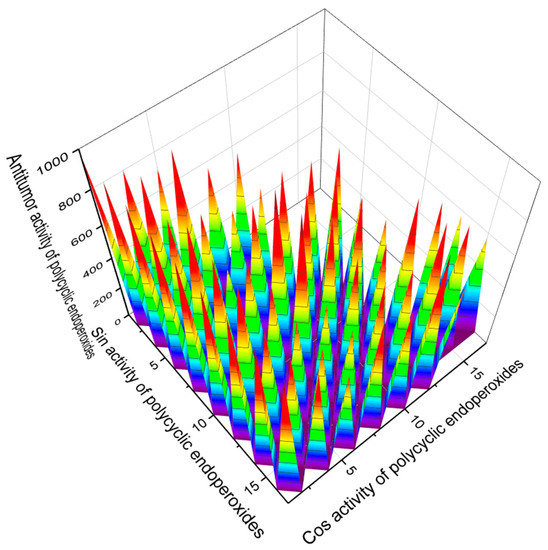
Figure 14.
The 3D graph shows the predicted and calculated antitumor activity of selected polycyclic endoperoxides (compound numbers: 11, 17, 30, 33, 142, 143, 164, and 165) showing the highest degree of confidence, more than 95%. These polycyclic endoperoxides can be used in clinical medicine as agents with strong antitumor activity.
Some polycyclic endoperoxides, in addition to antiparasitic, antiprotozoal, and antitumor activities, demonstrate other activities with Pa > 90%, which should also be mentioned in this article. This is primarily anti-inflammatory activity. Figure 15 demonstrates such compounds as well as their predicted anti-inflammatory activity. It should also be noted that endoperoxide artemisinin (86) and its analogs, and some other compounds, show antifungal activity. Figure 16 demonstrates predicted antifungal activity with Pa> 90%.
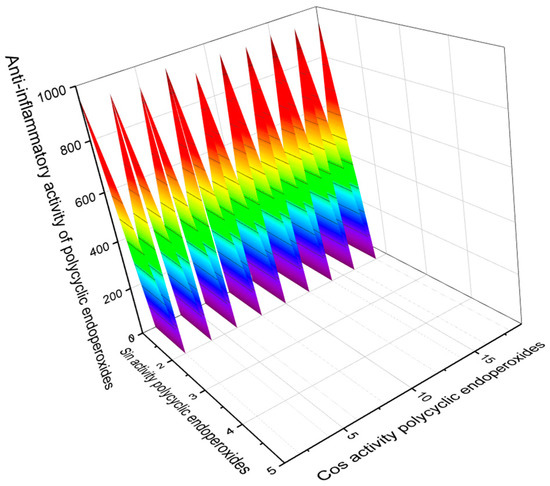
Figure 15.
The 3D graph shows the predicted and calculated anti-inflammatory activity of selected polycyclic endoperoxides (compound numbers: 1, 8, 9, 68, 94, 95, 96, 97, 98, 100, and 113) showing the highest degree of confidence, i.e., more than 95%. These polycyclic endoperoxides can be used as potential agents with strong anti-inflammatory activity.
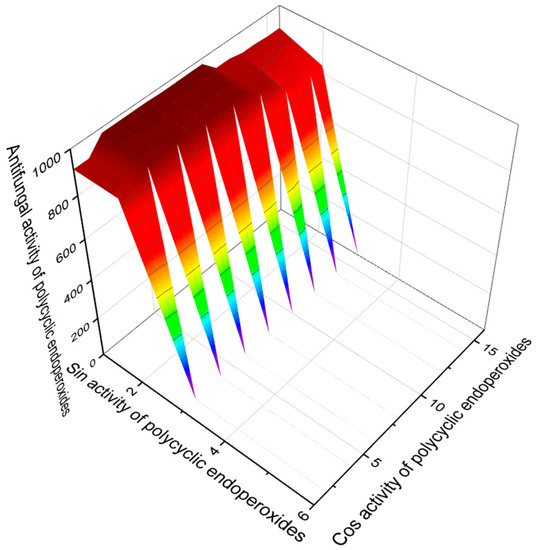
Figure 16.
The 3D graph shows the predicted and calculated antifungal activity of selected polycyclic endoperoxides (compound numbers: 86, 87, 88, 89, 90, 91, 92, 138, and 139) polycyclic endoperoxides showing the highest degree of confidence, more than 95%.
6. Conclusions
In this review, we presented more than 170 polycyclic endoperoxides isolated from various sources and showed that all endoperoxides demonstrate antiprotozoal activity with varying degrees of reliability, and among them, the artemisinin group and some other compounds are significantly distinguished from of all endoperoxides presented and have a strong antiprotozoal activity. Our data only confirm that the artemisinin group has unique properties, which is why it has been used in medical practice for more than 50 years in the fight against malaria parasites. In addition, the artemisinin group has a high antifungal activity, while some other endoperoxides have a strictly strong anti-inflammatory activity.
Compounds such as (19), (23), and (25) exhibited anti-hypercholesterolemic action, and compounds (166) and (168) have a strong stimulating effect on the respiratory and vasomotor centers of the brain. However, to confirm the conclusions regarding the in silico estimations, more research is required.
Author Contributions
Conceptualization, V.M.D. and V.V.P.; methodology, V.M.D.; software, T.A.G.; validation, V.M.D., N.S. and E.E.; formal analysis, V.V.P.; investigation, V.M.D.; data curation, V.V.P.; writing—original draft preparation, V.M.D.; writing—review and editing, V.M.D. and N.S.; visualization, V.V.P.; supervision, V.M.D.; project administration, V.M.D.; funding acquisition, V.V.P. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Data Availability Statement
Authors agree with the Publication Ethics Statement.
Acknowledgments
The work (GTA and PVV) was done in the framework of the Russian Federation fundamental research program for the long-term period for 2021–2030.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Dembitsky, V.M. Bioactive peroxides as potential therapeutic agents. Eur. J. Med. Chem. 2008, 43, 223–251. [Google Scholar] [CrossRef] [PubMed]
- Dembitsky, V.M.; Gloriozova, T.A.; Poroikov, V.V. Natural peroxy anticancer agents. Mini Rev. Med. Chem. 2007, 7, 571–589. [Google Scholar] [CrossRef] [PubMed]
- Dembitsky, V.M. Oxidation, epoxidation and sulfoxidation reactions catalysed by haloperoxidases. Tetrahedron 2003, 26, 4701–4720. [Google Scholar] [CrossRef]
- Dembitsky, V.M. Chemistry and biodiversity of the biologically active natural glycosides. Chem. Biodivers 2004, 1, 673–781. [Google Scholar] [CrossRef] [PubMed]
- Casteel, D.A. Peroxy natural products. Nat. Prod. Rep. 1992, 9, 289–312. [Google Scholar] [CrossRef]
- Casteel, D.A. Peroxy natural products. Nat. Prod. Rep. 1999, 16, 55–73. [Google Scholar] [CrossRef]
- Siddiq, A.; Yaremenko, I.; Terent’v, A.O.; Gloriozova, T.A.; Dzhemileva, L.U.; D’yakonov, A.V.; Vil, V.; Dembitsky, V.M. Phytomedicinal aspects of sesquiterpenoid peroxides: Origin, structures and biological activity. Frontiers Drug Chem. Clin. Res. 2019, 2, 1–10. [Google Scholar]
- Sikorsky, T.V.; Ermolenko, E.V.; Gloriozova, T.A.; Dembitsky, V.M. Mini Review: Anticancer activity of diterpenoid peroxides. Vietnam J. Chem. 2020, 58, 273–280. [Google Scholar] [CrossRef]
- Dembitsky, V.M.; Yaremenko, I.A. Stable and unstable 1,2-dioxolanes: Origin, synthesis, and biological activities. Sci. Synth. Knowl. Updates 2020, 38, 277–321. [Google Scholar]
- Dembitsky, V.M.; Vil, V.A. Medicinal chemistry of stable and unstable 1,2-dioxetanes: Origin, formation, and biological activities. Sci. Synth. Knowl. Updates 2019, 38, 333–377. [Google Scholar]
- Noronha, M.; Pawar, V.; Prajapati, A.; Subramanian, R.B. A literature review on traditional herbal medicines for malaria. S. Afr. J. Bot. 2020, 128, 292–303. [Google Scholar] [CrossRef]
- Bu, M.; Yang, B.B.; Hu, L. Natural endoperoxides as drug lead compounds. Curr. Med. Chem. 2016, 23, 383–405. [Google Scholar] [CrossRef] [PubMed]
- Vil, V.A.; Gloriozova, T.A.; Poroikov, V.V.; Terent’ev, A.O.; Savidov, N.; Dembitsky, V.M. Peroxy steroids derived from plant and fungi and their biological activities. Appl. Microbiol. Biotechnol. 2018, 102, 7657–7667. [Google Scholar] [CrossRef] [PubMed]
- Vil, V.A.; Terent’ev, A.O.; Savidov, N.; Gloriozova, T.A.; Poroikov, V.V.; Pounina, T.A.; Dembitsky, V.M. Hydroperoxy steroids and triterpenoids derived from plant and fungi: Origin, structures and biological activities. J. Steroid Biochem. Mol. Biol. 2019, 190, 76–87. [Google Scholar] [CrossRef] [PubMed]
- Dembitsky, V.M.; Poovarodom, S.; Leontowicz, H.; Leontowicz, M.; Vearasilp, S.; Trakhtenberg, S.; Gorinstein, S. The multiple nutrition properties of some exotic fruits: Biological activity and active metabolites. Food Res. Intern. 2011, 44, 1671–1701. [Google Scholar] [CrossRef]
- Dembitsky, V.; Shkrob, I.; Hanus, L.O. Ascaridole and related peroxides from the genus Chenopodium. Biomed. Pap. Med. Fac. Univ. Palacky Olomouc Czech Repub. 2008, 152, 209–215. [Google Scholar] [CrossRef]
- Dembitsky, V.M. Astonishing diversity of natural peroxides as potential therapeutic agents. J. Mol. Genet. Med. 2015, 9, 1000163. [Google Scholar]
- Ermolenko, E.V.; Imbs, A.B.; Gloriozova, T.A.; Poroikov, V.V.; Sikorskaya, T.V.; Dembitsky, V.M. Chemical diversity of soft coral steroids and their pharmacological activities. Mar. Drugs 2020, 18, 613. [Google Scholar] [CrossRef]
- Dembitsky, V.M. Antitumor and hepatoprotective activity of natural and synthetic neo steroids. Prog. Lipid Res. 2020, 79, 101048. [Google Scholar] [CrossRef]
- Mayer, A.M.S.; Guerrero, A.J.; Rodríguez, A.D.; Taglialatela-Scafati, O.; Nakamura, F.; Fusetani, N. Marine pharmacology in 2014–2015: Marine compounds with antibacterial, antidiabetic, antifungal, anti-Inflammatory, antiprotozoal, antituberculosis, antiviral, and anthelmintic activities; Affecting the immune and nervous systems, and other miscellaneous mechanisms of action. Mar. Drugs 2020, 18, 5. [Google Scholar]
- Budde, S.; Goerdeler, F.; Floß, J.; Kreitmeier, P.; Hicks, E.F.; Moscovitz, O.; Seeberger, P.H.; Davies, H.M.L.; Reiser, O. Visible light mediated oxidative ring expansion of anellated cyclopropanes to fused endoperoxides with antimalarial activity. Org. Chem. Front. 2020, 7, 1789–1795. [Google Scholar] [CrossRef]
- Yang, J.; He, Y.; Li, Y.; Zhang, X.; Wong, Y.K.; Shen, S.; Zhong, T.; Zhang, J.; Liu, Q.; Wang, J. Advances in the research on the targets of anti-malaria actions of artemisinin. Pharmacol. Ther. 2020, 216, 107697. [Google Scholar] [CrossRef] [PubMed]
- Tajuddeen, N.; Van Heerden, F.R. Anti-plasmodial natural products: An update. Malar. J. 2019, 18, 404. [Google Scholar] [CrossRef] [PubMed]
- Rukunga, G.; Simons, A.J. The Potential of Plants as a Source of Antimalarial Agents: A Review; Planta Phile Publications: Berlin, Germany, 2006; 72p. [Google Scholar]
- Tiwari, M.K.; Chaudhary, S. Artemisinin-derived antimalarial endoperoxides from bench-side to bed-side: Chronological advancements and future challenges. Med. Res. Rev. 2020, 40, 1220–1275. [Google Scholar] [CrossRef] [PubMed]
- Kotepui, M.; Kotepui, K.U.; Milanez, G.D. Global prevalence and mortality of severe Plasmodium malariae infection: A systematic review and meta-analysis. Malar. J. 2020, 19, 274. [Google Scholar] [CrossRef] [PubMed]
- Sharp, P.M.; Plenderleith, L.J.; Hahn, B.H. Ape Origins of human malaria. Annu. Rev. Microbiol. 2020, 74, 39–63. [Google Scholar] [CrossRef] [PubMed]
- Enserink, M. Malaria researchers wait for industry to join fight. Science 2000, 287, 1956–1958. [Google Scholar] [CrossRef] [PubMed]
- Chima, R.I.; Goodman, C.A.; Mills, A. The economic impact of malaria in Africa: A critical review of the evidence. Health Policy 2003, 63, 17–36. [Google Scholar] [CrossRef]
- Cock, I.E.; Selesho, M.I.; van Vuuren, S.F. A review of the traditional use of southern African medicinal plants for the treatment of malaria. J. Ethnopharm. 2019, 245, 112176. [Google Scholar] [CrossRef]
- Poroikov, V.V. Computer-aided drug design: From discovery of novel pharmaceutical agents to systems pharmacology. Biochem. (Moscow) Suppl. Ser. B Biomed. Chem. 2020, 14, 216–227. [Google Scholar] [CrossRef]
- Poroikov, V.V.; Filimonov, D.A.; Gloriozova, T.A.; Lagunin, A.A.; Druzhilovskiy, D.S.; Rudik, A.V.; Stolbov, L.A.; Dmitriev, A.V.; Tarasova, O.A.; Ivanov, S.M.; et al. Computer-aided prediction of biological activity spectra for organic compounds: The possibilities and limitations. Russ. Chem. Bull. 2019, 68, 2143–2154. [Google Scholar] [CrossRef]
- Muratov, E.N.; Bajorath, J.; Sheridan, R.P.; Tetko, I.; Filimonov, D.; Poroikov, V.; Oprea, T.; Baskin, I.I.; Varnek, A.; Roitberg, A.; et al. QSAR Without Borders. Chem. Soc. Rev. 2020, 49, 3525–3564. [Google Scholar] [CrossRef] [PubMed]
- Dembitsky, V.M.; Gloriozova, T.A.; Poroikov, V.V. Pharmacological activities of epithio steroids. J. Pharm. Res. Intern. 2017, 18, 1–19. [Google Scholar] [CrossRef]
- Dembitsky, V.M.; Dzhemileva, L.; Gloriozova, T.; D’yakonov, V. Natural and synthetic drugs used for the treatment of the dementia. Biochem. Biophys. Res. Commun. 2020, 524, 772–783. [Google Scholar] [CrossRef] [PubMed]
- Dembitsky, V.M.; Gloriozova, T.A.; Poroikov, V.V. Pharmacological profile of natural and synthetic compounds with rigid adamantane-based scaffolds as potential agents for the treatment of neurodegenerative diseases. Biochem. Biophys. Res. Commun. 2020, 529, 1225–1241. [Google Scholar] [CrossRef] [PubMed]
- Dembitsky, V.M. Bioactive cyclobutane-containing alkaloids. J. Nat. Med. 2008, 62, 1–33. [Google Scholar] [CrossRef]
- Ismail, F.M.D.; Levitsky, D.O.; Dembitsky, V.M. Aziridine alkaloids as potential therapeutic agents. Eur. J. Med. Chem. 2009, 44, 3373–3387. [Google Scholar] [CrossRef]
- Dembitsky, V.M. Naturally occurring bioactive cyclobutane-containing (CBC) alkaloids in fungi, fungal endophytes, and plants. Phytomedicine 2014, 21, 1559–1581. [Google Scholar] [CrossRef]
- Dembitsky, V.M. Astonishing diversity of natural surfactants: 4. Fatty acid amide glycosides, their analogs and derivatives. Lipids 2005, 40, 641–660. [Google Scholar] [CrossRef]
- Dembitsky, V.M. Astonishing diversity of natural surfactants: 5. Biologically active glycosides of aromatic metabolites. Lipids 2005, 40, 869–900. [Google Scholar] [CrossRef]
- Dembitsky, V.M. Astonishing diversity of natural surfactants: 6. Biologically active marine and terrestrial alkaloid glycosides. Lipids 2005, 40, 1081–1114. [Google Scholar] [CrossRef] [PubMed]
- Dembitsky, V.M. Astonishing diversity of natural surfactants: 7. Biologically active hemi-and monoterpenoid glycosides. Lipids 2006, 41, 1–27. [Google Scholar] [CrossRef] [PubMed]
- Dembitsky, V.M. Astonishing diversity of natural surfactants: 3. Carotenoid glycosides and isoprenoid glycolipids. Lipids 2005, 40, 535–557. [Google Scholar] [CrossRef] [PubMed]
- Dembitsky, V.M. Astonishing diversity of natural surfactants: 1. Glycosides of fatty acids and alcohols. Lipids 2004, 39, 933–953. [Google Scholar] [CrossRef] [PubMed]
- Dembitsky, V.M. Astonishing diversity of natural surfactants: 2. Polyether glycosidic ionophores and macrocyclic glycosides. Lipids 2005, 40, 219–248. [Google Scholar] [CrossRef] [PubMed]
- Řezanka, T.; Temina, M.; Tolstikov, A.G.; Dembitsky, V.M. Natural microbial UV radiation filters - mycosporine-like amino acids. Folia Microbiol. 2004, 49, 339–352. [Google Scholar] [CrossRef]
- Kuklev, D.V.; Domb, A.J.; Dembitsky, V.M. Bioactive acetylenic metabolites. Phytomedicine 2013, 20, 1145–1159. [Google Scholar] [CrossRef]
- Kuklev, D.V.; Dembitsky, V.M. Chemistry, origin, antitumor and other activities of fungal homo-dimeric alkaloids. Mathews J. Pharmaceut. Sci. 2016, 1, 004. [Google Scholar]
- Kilimnik, A.; Kuklev, D.V.; Dembitsky, V.M. Antitumor Acetylenic Lipids. Mathews J. Pharmaceut. Sci. 2016, 1, 005. [Google Scholar]
- Dembitsky, V.M.; Gloriozova, T.A.; Poroikov, V.V. Novel antitumor agents: Marine sponge alkaloids, their synthetic analogs and derivatives. Mini Rev. Med. Chem. 2005, 5, 319–336. [Google Scholar] [CrossRef]
- Dembitsky, V.M. Betaine ether-linked glycerolipids: Chemistry and biology. Prog. Lipid Res. 1996, 35, 1–51. [Google Scholar] [CrossRef]
- Dembitsky, V.M.; Maoka, T. Allenic and cumulenic lipids. Prog. Lipid Res. 2007, 46, 328–375. [Google Scholar] [CrossRef] [PubMed]
- Dembitsky, V.M.; Smoum, R.; Al-Quntar, A.A.; Ali, H.A.; Pergament, I.; Srebnik, M. Natural occurrence of boron-containing compounds in plants, algae and microorganisms. Plant Sci. 2002, 163, 931–942. [Google Scholar] [CrossRef]
- Dembitsky, V.M.; Levitsky, D.O. Arsenolipids. Prog. Lipid Res. 2004, 43, 403–448. [Google Scholar] [CrossRef]
- Dembitsky, V.M.; Rozentsvet, O.A.; Pechenkina, E.E. Glycolipids, phospholipids and fatty acids of brown algae species. Phytochemistry 1990, 29, 3417–3421. [Google Scholar] [CrossRef]
- Dembitsky, V.M.; Pechenkina-Shubina, E.E.; Rozentsvet, O.A. Glycolipids and fatty acids of some seaweeds and marine grasses from the Black Sea. Phytochemistry 1991, 30, 2279–2283. [Google Scholar] [CrossRef]
- Dembitsky, V.M.; Řezanka, T. Metabolites produced by nitrogen fixing Nostoc species. Folia Microbiol. 2005, 50, 363–391. [Google Scholar] [CrossRef]
- Dembitsky, V.M. Bromo- and iodo-containing alkaloids from marine microorganisms and sponges. Russ. J. Bioorg. Chem. 2002, 28, 170–182. [Google Scholar] [CrossRef]
- Dembitsky, V.M. Plasmalogens in phospholipids of marine invertebrates. Russ. J. Mar. Biol. 1979, 5, 86–90. [Google Scholar]
- Hanuš, L.O.; Levitsky, D.O.; Shkrob, I.; Dembitsky, V.M. Plasmalogens, fatty acids and alkyl glyceryl ethers of marine and freshwater clams and mussels. Food Chem. 2009, 116, 491–498. [Google Scholar] [CrossRef]
- Dembitsky, V.M.; Rozentsvet, O.A. Diacylglyceryltrimethylhomoserines and phospholipids of some green marine macrophytes. Phytochemistry 1989, 28, 3341–3343. [Google Scholar] [CrossRef]
- Dembitsky, V.M. Natural neo acids and neo alkanes: Their analogs and derivatives. Lipids 2006, 41, 309–340. [Google Scholar] [CrossRef] [PubMed]
- Dembitsky, V.M.; Rozentsvet, O.A. Phospholipid composition of some marine red algae. Phytochemistry 1990, 29, 3149–3152. [Google Scholar] [CrossRef]
- Butler, M.S.; Capon, R.J. Trunculin-F and contrunculin-A and -B: Novel oxygenated norterpenes from a southern Australian marine sponge, Latrunculia conulosa. Aust. J. Chem. 1993, 46, 1363–1374. [Google Scholar] [CrossRef]
- Ovenden, S.P.; Capon, R.J. Trunculins G–I: New norsesterterpene cyclic peroxides from a southern Australian marine sponge, Latrunculia sp. Aust. J. Chem. 1998, 51, 573–580. [Google Scholar] [CrossRef]
- Hirade, H.; de Voogd, N.J.; Suzuka, T.; Tanaka, J. Trunculins X and Y from an Okinawan sponge Sigmosceptrella sp. Tetrahedron 2019, 75, 4620–4625. [Google Scholar] [CrossRef]
- Gonzalez, A.G.; Martin, J.D.; Perez, C.; Rovirosa, J.; Tagle, B.; Clardy, J. Isolation and X-ray structural determination of three new diterpenoids from the marine alga Taonia atomaria. Chem. Lett. 1984, 13, 1649–1652. [Google Scholar] [CrossRef]
- Kusumi, T.; Ohtani, I.; Inouye, Y.; Kakisawa, I. Absolute configurations of cytotoxic marine cembranolides; Consideration of mosher’s method. Tetrahedron Lett. 1988, 29, 4731–4734. [Google Scholar] [CrossRef]
- Uchio, Y.; Eguchi, S.; Kuramoto, J.; Nakayama, M.; Hase, T. Denticulatolide, an ichthyotoxic peroxidecontaining cembranolide from the soft coral Lobophytum denticulatum. Tetrahedron Lett. 1985, 26, 4487–4490. [Google Scholar] [CrossRef]
- Fukazawa, Y. Conformational study of the cembranolide diterpene denticulatolide by molecular mechanics method. Tetrahedron Lett. 1986, 27, 1825–1828. [Google Scholar] [CrossRef]
- Uchio, Y.; Shizuko, E.; Yoshimasa, F.; Mitsuaki, K. 7-Epidenticulatolide, a new cembranolide with a cyclic peroxide function from the soft coral Lobophytum denticulatum. Bull. Chem. Soc. Japan 1992, 65, 1182–1185. [Google Scholar] [CrossRef]
- Hambley, T.W.; Taylor, W.C.; Toth, S. The constituents of marine sponges. IX New norditerpenoids from Aplysilla pallida. Aust. J. Chem. 1997, 50, 903–910. [Google Scholar] [CrossRef]
- Monaco, P.; Parrilli, M.; Previtera, L. Two endoperoxide diterpenes from Elodea Canadensis. Tetrahedron Lett. 1987, 28, 4609–4612. [Google Scholar] [CrossRef]
- Hirota, H.; Okino, T.; Yoshimura, E.; Fusetani, N. Five new antifouling sesquiterpenes from two marine sponges of the genus Axinyssa and the nudibranch Phyllidia pustulosa. Tetrahedron 1998, 54, 13971–13980. [Google Scholar] [CrossRef]
- Ahmed, A.F.; Kuo, Y.-H.; Dai, C.-F.; Sheu, J.H. Oxygenated terpenoids from a Formosan Soft coral Sinularia gibberosa. J. Nat. Prod. 2005, 68, 1208–1212. [Google Scholar] [CrossRef]
- Sera, Y.; Adachi, K.; Shizuri, Y. A new epidioxy sterol as an antifouling substance from a Palauan marine sponge, Lendenfeldia chondrodes. J. Nat. Prod. 1999, 62, 152–154. [Google Scholar] [CrossRef]
- Seo, Y.W.; Rho, J.R.; Cho, K.W.; Sim, C.J.; Shin, J.H. Isolation of epidioxysteroids from a sponge of the genus Tethya. Bull. Korean Chem. Soc. 1997, 18, 631–635. [Google Scholar]
- Aknin, M.; Viracaoundin, I.; Faure, R.; Gaydou, E.M. 5α,8α-Epidioxycholest-6-en-3-β-ol from three cone snails of the Indian ocean. J. Am. Oil Chem. Soc. 1998, 75, 1679–1681. [Google Scholar] [CrossRef]
- Seo, Y.; Rho, J.R.; Shin, J. Isolation of two steroids from the marine polychaete worm Perinersis aibuhitensis. Ocean Res. 1996, 18, 83–87. [Google Scholar]
- Fattorusso, E.; Magno, S.; Santacroce, C.; Sica, D. Sterol peroxides from the sponge Axinella cannabina. Gazz. Chim. Gazz. Ital. 1974, 104, 409–413. [Google Scholar]
- Gauvin, A.; Smadja, J.; Aknin, M.; Faure, R.; Gaydou, E.M. Isolation of bioactive 5α,8α-epidioxy sterols from the marine sponge Luffariella cf. variabilis. Can. J. Chem. 2000, 78, 986–992. [Google Scholar] [CrossRef]
- Abourriche, A.; Charrouf, M.; Chaib, N.; Bennamara, A.; Bontemps, N.; Francisco, C. Isolation and bioactivities of epidioxysterol from the tunicate Cynthia savignyi. Farmaco 2000, 55, 492–494. [Google Scholar] [CrossRef]
- Minh, C.V.; Van Kiem, P.; Kim, Y.H. Cytotoxic constituents of Diadema setosum. Arch. Pharmacal. Res. 2004, 27, 734–737. [Google Scholar] [CrossRef] [PubMed]
- Gunatilaka, A.A.L.; Gopichand, Y.; Schmitz, F.J.; Djerassi, C. Minor and trace sterols in marine invertebrates. 26. Isolation and structure elucidation of nine new 5α,8α-epidoxy sterols from four marine organisms. J. Org. Chem. 1981, 46, 3860–3866. [Google Scholar] [CrossRef]
- Jimenez, C.; Quiño, A.E.; Do Caste, L.; Riguera, R. Epidioxy sterols from the tunicates Dendrodoa grossularia and Ascidiella aspersa and the gastropoda Aplysia depilans and Aplysia punctate. J. Nat. Prod. 1986, 49, 905–909. [Google Scholar] [CrossRef]
- Findlay, J.A.; Patil, A.D. A novel sterol peroxide from the sea anenome Metridium senile. Steroids 1984, 44, 261–265. [Google Scholar] [CrossRef]
- Sheikh, Y.M.; Djerassi, C. Steroids from sponges. Tetrahedron 1974, 30, 4095–4103. [Google Scholar] [CrossRef]
- Anjaneyulu, A.S.R.; Sagar, K.S.; Venugopal, M.J.R.V. Terpenoid and steroid constituents of the Indian ocean soft coral Sinularia maxima. Tetrahedron 1995, 51, 10997–11010. [Google Scholar] [CrossRef]
- Faulkner, D.J. Marine natural products. Nat. Prod. Rep. 1997, 14, 257–302. [Google Scholar] [CrossRef]
- Faulkner, D.J. Marine natural products. Nat. Prod. Rep. 1991, 8, 97–114. [Google Scholar] [CrossRef]
- Toume, K.; Ishibashi, M. 5α,8α-Epidioxysterol sulfate from a diatom Odontella aurita. Phytochemistry 2002, 61, 359–360. [Google Scholar] [CrossRef]
- Yaoita, Y.; Amemiya, K.; Ohnuma, H.; Furumura, K.; Masaki, A.; Matsuki, T.; Kikuchi, M. Sterol constituents from five edible mushrooms. Chem. Pharm. Bull. 1998, 46, 944–950. [Google Scholar] [CrossRef]
- Stonard, R.J.; Petrovich, J.C.; Andersen, R.J. A new C26 sterol peroxide from the opisthobranch mollusk Adalaria sp. and the sea pen Virgularia sp. Steroids 1980, 36, 81–86. [Google Scholar] [CrossRef]
- Prawat, H.; Mahidol, C.; Wittayalai, S.; Intachote, P.; Kanchanapoom, T.; Ruchirawat, S. Nitrogenous sesquiterpenes from the Thai marine sponge Halichondria sp. Tetrahedron 2011, 67, 5651–5655. [Google Scholar] [CrossRef]
- Mishra, P.D.; Wahidullah, S.; De Souza, L.; Kamat, S.Y. Lipid constituents of marine sponge Suberites carnosus. Indian J. Chem. 1996, 35, 806–809. [Google Scholar]
- Im, K.S.; Nam, K.I.; Sim, C.J.; Jung, J.H. Sterol peroxide derivatives from the marine sponge Spirastrella abata. Korean J. Pharmacog. 2000, 31, 401–406. [Google Scholar]
- Feng, Y.; Khokhara, S.; Davis, R.A. Crinoids: Ancient organisms, modern chemistry. Nat. Prod. Rep. 2017, 34, 571–584. [Google Scholar] [CrossRef]
- Mun, B.; Wang, W.; Kim, H. Cytotoxic 5α,8α-epidioxy sterols from the marine sponge Monanchora sp. Arch. Pharm. Res. 2015, 38, 18–25. [Google Scholar] [CrossRef]
- Ioannou, E.; Aazika, A.F.A.; Dimitrios, M.Z.; Xanthippi, C.; Constantinos, A.; Alexisd, V.M.N.; Roussisa, V. 5α,8α-Epidioxysterols from the gorgonian Eunicella cavolini and the ascidian Trididemnum inarmatum: Isolation and evaluation of their antiproliferative activity. Steroids 2009, 74, 73–80. [Google Scholar] [CrossRef]
- Dembitsky, V.M. The multiple properties of some of the lichenized Ascomycetes: Biological activity and active metabolites. In Plant Adaptation Strategies in Changing Environment; Shukla, V., Kumar, S., Kumar, N., Eds.; Springer: Singapore, 2017. [Google Scholar]
- Torres, A.; Hochberg, M.; Pergament, I.; Smoum, R.; Niddam, V.; Dembitsky, V.M. A new UV-B absorbing mycosporine with photo protective activity from the lichenized ascomycete Collema cristatum. Eur. J. Biochem. 2004, 271, 780–784. [Google Scholar] [CrossRef]
- Dembitsky, V.M.; Řezanka, T.; Spížek, J.; Hanuš, L.O. Secondary metabolites of slime molds (myxomycetes). Phytochemistry 2005, 66, 747–769. [Google Scholar] [CrossRef]
- Dembitsky, V.M. Lipids of lichens. Prog. Lipid Res. 1992, 31, 373–397. [Google Scholar] [CrossRef]
- Dembitsky, V.M.; Rezanka, T.; Bychek, I.A.; Shustov, M.V. Identification of fatty acids from Cladonia lichens. Phytochemistry 1991, 30, 4015–4018. [Google Scholar] [CrossRef]
- Dembitsky, V.M.; Rezanka, T.; Bychek, I.A.; Shustov, M.V. Fatty acid composition of Parmelia lichens. Phytochemistry 1992, 31, 841–843. [Google Scholar] [CrossRef]
- Dembitsky, V.M.; Rezanka, T.; Bychek, I.A. Fatty acids and phospholipids from lichens of the order Lecanorales. Phytochemistry 1992, 31, 851–853. [Google Scholar] [CrossRef]
- Dembitsky, V.M.; Shubina, E.E.; Kashin, A.G. Phospholipid and fatty acid composition of some Basidiomycetes. Phytochemistry 1992, 31, 845–849. [Google Scholar] [CrossRef]
- Dembitsky, V.M.; Terent’ev, A.O.; Levitsky, D.O. Amino and fatty acids of wild edible mushrooms of the genus Boletus. Records Nat. Prod. 2010, 4, 218–225. [Google Scholar]
- Rustamova, N.; Bozorov, K.; Efferth, T. Novel secondary metabolites from endophytic fungi: Synthesis and biological properties. PhytoChem. Rev. 2020, 19, 425–448. [Google Scholar] [CrossRef]
- Vil, V.; Gloriozova, T.A.; Poroikov, V.V.; Terent’ev, A.O.; Savidov, N.; Dembitsky, V.M. Naturally occurring of α,β-diepoxy-containing compounds: Origin, structures, and biological activities. Appl. Microbiol. Biotech. 2019, 103, 3249–3264. [Google Scholar] [CrossRef]
- Zhang, Y.; Han, T.; Ming, Q.; Wu, L.; Rahman, K.; Qin, L. Alkaloids produced by endophytic fungi: A review. Nat. Prod. Commun. 2012, 7, 963–968. [Google Scholar] [CrossRef]
- Zhao, M.; Gödecke, T.; Gunn, J.; Duan, J.A.; Che, C.T. Protostane and fusidane triterpenes: A mini review. Molecules 2013, 18, 4054–4080. [Google Scholar] [CrossRef] [PubMed]
- Savidov, N.; Gloriozova, T.A.; Poroikov, V.V.; Dembitsky, V.M. Highly oxygenated isoprenoid lipids derived from fungi and fungal endophytes: Origin and biological activities. Steroids 2018, 140, 114–124. [Google Scholar] [CrossRef] [PubMed]
- Trung, H.V.; Tuan, N.N.; Thanh, N.T.; Giang, T.T.B.; Giang, D.T.T.; Ogunwande, I.; Thang, T.D. Determination of ergosterol and ergosterol peroxide in higher fungi species by high-performance liquid chromatography. J. Pharm. Phytochem. 2018, 7, 2376–2379. [Google Scholar]
- Dembitsky, V.M.; Rezanka, T.; Shubina, E.E. Unusual hydroxy fatty acids from some higher fungi. Phytochemistry 1993, 34, 1057–1059. [Google Scholar] [CrossRef]
- Mallavadhani, U.U.; Sudhakar, A.V.S.; Satyanarayana, K.V.S.; Mahapatraa, A.; Li, W. Chemical and analytical screening of some edible mushrooms. Food. Chem. 2006, 95, 58–64. [Google Scholar] [CrossRef]
- Ragasa, C.Y. Anticancer compounds from nine commercially grown and wild Philippine mushrooms. Manila J. Sci. 2018, 11, 42–57. [Google Scholar]
- Kahlos, K.; Kangas, L.; Hiltunen, R. Ergosterol peroxide, an active compound from Inonotus radiatus. Planta Med. 1989, 55, 389–390. [Google Scholar] [CrossRef]
- Chen, Y.K.; Kuo, Y.H.; Chiang, B.H.; Lo, J.M.; Sheen, L.Y. Cytotoxic activities of 9,11-dehydroergosterol peroxide and ergosterol peroxide from the fermentation mycelia of Ganoderma lucidum cultivated in the medium containing leguminous plants on Hep 3B cells. J. Agric. Food Chem. 2009, 57, 5713–5719. [Google Scholar] [CrossRef]
- Kobori, M.; Yoshida, M.; Ohnishi-Kameyama, M.; Shinmoto, H. Ergosterol peroxide from an edible mushroom suppresses inflammatory responses in RAW264.7 macrophages and growth of HT29 colon adenocarcinoma cells. Br. J. Pharmacol. 2007, 150, 209–219. [Google Scholar] [CrossRef]
- Russo, A.; Cardile, V.; Piovano, M.; Caggia, S.; Espinoza, C.L.; Garbarino, J.A. Pro-apoptotic activity of ergosterol peroxide and (22E)-ergosta-7,22-dien-5α-hydroxy-3,6-dione in human prostate cancer cells. Chem. Biol. Interact. 2010, 184, 352–358. [Google Scholar] [CrossRef]
- Li, X.; Wu, Q.; Bu, M.; Hu, L.; Du, W.W.; Jiao, C.; Pan, H.; Sdiri, M.; Wu, N.; Xie, Y. Ergosterol peroxide activates Foxo3-mediated cell death signaling by inhibiting AKT and c-Myc in human hepatocellular carcinoma cells. Oncotarget 2016, 7, 33948–33959. [Google Scholar] [CrossRef] [PubMed]
- Yasukawa, K.; Aoki, T.; Takido, M.; Ikekawa, T.; Saito, H.; Matsuzawa, T. Inhibitory effects of ergosterol isolated from the edible mushroom Hypsizigus marmoreus on TPA-induced inflammatory ear oedema and tumour promotion in mice. Phytother. Res. 1994, 8, 10–13. [Google Scholar] [CrossRef]
- Bu, M.; Cao, T.; Li, H.; Guo, M.; Yang, B.B.; Zeng, C.; Hu, L. Synthesis of 5α,8α-ergosterol peroxide 3-carbamate derivatives and a fluorescent mitochondria-targeting conjugate for enhanced anticancer activities. ChemMedChem 2017, 12, 466–474. [Google Scholar] [CrossRef] [PubMed]
- Bu, M.; Cao, T.; Li, H.; Guo, M.; Yang, B.B.; Zeng, C.; Zhou, Y.; Zhang, N.; Hu, L. Synthesis and biological evaluation of novel steroidal 5α,8α-epidioxyandrost-6-ene-3β-ol-17-(O-phenylacetamide)oxime derivatives as potential anticancer agents. Bioorg. Med. Chem. Lett. 2017, 27, 3856–3861. [Google Scholar] [CrossRef]
- Bok, J.W.; Lermer, L.; Chilton, J.; Klingeman, H.G.; Towers, G.H. Antitumor sterols from the mycelia of Cordyceps sinensis. Phytochemistry 1999, 51, 891–898. [Google Scholar] [CrossRef]
- Govindharaj, M.; Arumugam, S.; Nirmala, G.; Bharadwaj, M.; Murugiyan, K. Effect of marine basidiomycetes fulvifomes sp.-derived ergosterol peroxide on cytotoxicity and apoptosis induction in MCF-7 Cell Line. J. Fungi 2019, 5, 16. [Google Scholar] [CrossRef]
- Serebryakov, E.P.; Simolin, A.V.; Kucherov, V.F.; Rosynov, B.V. New metabolites of Fusarium moniliforme sheld. Tetrahedron 1970, 26, 5215–5219. [Google Scholar] [CrossRef]
- Zang, M.; Ying, J.Z. Economic fungi in the South West of China; Scientific Press: Beijing, China, 1994. [Google Scholar]
- Yue, J.M.; Chen, S.N.; Lin, Z.W.; Sun, H.D. Sterols from the fungus Lactarium volemus. Phytochemistry 2001, 56, 801–806. [Google Scholar] [CrossRef]
- Miao, F.P.; Li, X.D.; Liu, X.H.; Cichewicz, R.H.; Ji, N.Y. Secondary metabolites from an algicolous Aspergillus versicolor strain. Mar Drugs 2012, 10, 131–139. [Google Scholar] [CrossRef]
- Zheng, W.; Liu, T.; Xiang, X.; Gu, Q. Sterol composition in field-grown and cultured mycelia of Inonotus obliquus. Yaoxue Xuebao 2007, 42, 750–756. [Google Scholar]
- Zuo, W.; Luo, D.Q. Research on the chemical components of the fruit bodies of Boletus calopus. Anhui Nongye Kexue 2010, 38, 2356–2357. [Google Scholar]
- Yaoita, Y.; Matsuki, K.; Iijima, T.; Nakano, S.; Kakuda, R.; Machida, K.; Kikuchi, M. New sterols and triterpenoids from four edible mushrooms. Chem. Pharm. Bull. 2001, 49, 589–594. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.; Li, X.M.; Li, C.S.; Wang, B.G. Nigerasterols A and B, antiproliferative sterols from the mangrovederived endophytic fungus Aspergillus niger MA-132. Helv. Chim. Acta 2013, 96, 1055–1061. [Google Scholar] [CrossRef]
- Yaoita, Y.; Endo, K.; Tani, Y.; Machida, K.; Amemiya, K. Sterol constituents from seven mushrooms. Chem. Pharm. Bull. 1999, 47, 847–851. [Google Scholar] [CrossRef]
- Liu, D.Z.; Luo, M.H. Two new chamigrane metabolites from fermentation broth of Steccherinum ochraceum. Fitoterapia 2010, 81, 1205–1207. [Google Scholar] [CrossRef]
- Liu, D.Z.; Dong, Z.J.; Wang, F.; Liu, J.K. Two novel norsesquiterpene peroxides from basidiomycete Steccherinum ochraceum. Tetrahedron Lett. 2010, 51, 3152–3153. [Google Scholar] [CrossRef]
- Li, L.B.; Ren, J.; Lai, R.; Cheng, Z.M.; Zhu, H.J. Natural cyclic peroxide echinobithiophene A with antimicrobial activity from Echinops ritro L. Chem. J. Chinese Univ. 2011, 32, 891–896. [Google Scholar]
- Rahaman, M.S.; Siraj, M.A.; Sultana, S.; Seidel, V.; Islam, M.A. Molecular phylogenetics and biological potential of fungal endophytes from plants of the sundarbans Mangrove. Front. Microbiol. 2020, 11, 570855. [Google Scholar] [CrossRef]
- Li, H.; Huang, H.; Shao, C.; Huang, H.; Jiang, J. Cytotoxic norsesquiterpene peroxides from the endophytic fungus Talaromyces flavus isolated from the mangrove plant Sonneratia apetala. J. Nat. Prod. 2011, 74, 1230–1235. [Google Scholar] [CrossRef]
- She, Z.; Li, H.; Li, M.; Zhu, X.; Lin, Y. Norsesquiterpenoid Peroxide with Antitumor Activity and Preparation and Application Thereof. Chinese Patent CN 2011-10031487, 27 December 2011. Faming Zhuanli Shenqing. [Google Scholar]
- Li, Y.; Niu, S.; Sun, B.; Liu, S.; Liu, X. Cytosporolides A-C, antimicrobial meroterpenoids with a unique peroxylactone skeleton from Cytospora sp. Org. Lett. 2010, 12, 3144–3147. [Google Scholar] [CrossRef]
- Linington, R.; Navarro, G.; Pudhom, K.; McKerrow, J. Novel Semisynthetic Small Molecules for the Treatment Parasitic Disease. Patent WO 2012-US48743, 7 February 2013. PCT Int. Appl.. [Google Scholar]
- Chokpaiboon, S.; Sommit, D.; Teerawatananond, T.; Muangsin, N.; Bunyapaiboonsri, T.; Pudhom, K. Cytotoxic nor-chamigrane and chamigrane endoperoxides from a Basidiomycetous fungus. J. Nat. Prod. 2010, 73, 1005–1007. [Google Scholar] [CrossRef] [PubMed]
- Spence, J.T.J.; George, J.H. Structural reassignment of cytosporolides A–C via biomimetic synthetic studies and reinterpretation of NMR data. Org. Lett. 2011, 13, 5318–5321. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Niu, S.; Lu, X.; Chen, X.; Zhang, H. Unique metabolites of Pestalotiopsis fici suggest a biosynthetic hypothesis involving a Diels-Alder reaction and then mechanistic diversification. Chem. Commun. 2010, 46, 460–462. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.-S.; Chen, Y.-T.; Wan, X.-Y.; Edmund, F.; Puff, H.; Breitmaier, E. Die Struktur des Hypocrellins und seines Photooxidationsproduktes Peroxyhypocrellin. Liebigs Ann. Chem. 1981, 10, 1880–1885. [Google Scholar]
- Dembitsky, V.M. Bioactive fungal endoperoxides. Med Mycol. 2015, 1, 1–10. [Google Scholar] [CrossRef]
- Liu, D.Z.; Liu, J.K. Peroxy natural products. Nat. Prod. Bioprospect. 2013, 3, 161–206. [Google Scholar] [CrossRef]
- Ma, N.; Zhang, Z.; Liao, F.; Jiang, T.; Tu, Y. The birth of artemisinin. Pharmacol. Therapeut. 2020, 216, 107658. [Google Scholar] [CrossRef]
- Liu, K.; Zuo, H.; Li, G.; Yu, H.; Hu, Y. Global research on artemisinin and its derivatives: Perspectives from patents. Pharmacol. Res. 2020, 159, 105048. [Google Scholar] [CrossRef]
- Czechowski, T.; Weathers, P.J.; Brodelius, P.E.; Brown, G.D.; Graham, I.A. Editorial: Artemisinin–from traditional Chinese medicine to artemisinin combination therapies; Four decades of research on the biochemistry, physiology, and breeding of Artemisia annua. Front. Plant Sci. 2020, 11, 594565. [Google Scholar] [CrossRef]
- Luo, S.; Zhang, Q. A new peroxide-sesquetepene: Nardosaldehyde. Nat. Prod. Res. Develop. 1997, 9, 7–9. [Google Scholar]
- Jayachandran, K.; Sekar, I.; Parthiban, K.T.; Amirtham, D.; Suresh, K.K. Analysis of different grades of agarwood (Aquilaria malaccensis Lamk.) oil through GC-MS. Indian J. Nat. Prod. Res. 2014, 5, 44–47. [Google Scholar]
- Caniato, R.; Puricelli, L. Review: Natural antimalarial agents (1995–2001). Crit. Rev. Plant Sci. 2003, 22, 79–105. [Google Scholar] [CrossRef]
- Takaya, Y.; Kurumada, K.I.; Takeuji, Y.; Kim, H.H.; Shibata, Y.; Ikemoto, N.; Wataya, Y.; Oshima, Y. Novel antimalarial guaiane-type sesquiterpenoids from Nardostachys chinensis roots. Tetrahedron Lett. 1998, 39, 1361–1364. [Google Scholar] [CrossRef]
- Chatterjee, A.; Dutta, U.; Bandyopadhyay, D.; Nayak, A.; Basak, B.; Banerji, A.; Banerji, J. An overview of the genus Nardostachys. Nat. Prod. Commun. 2007, 2, 1163–1173. [Google Scholar] [CrossRef]
- Takaya, Y.; Takeuji, Y.; Akasaka, M.; Nakagawasai, O.; Tadano, T.; Kisara, K.; Kim, H.S.; Wataya, Y.; Niwa, M.; Oshima, Y. Novel guaiane endoperoxides, nardoguaianone A–D, from Nardostachys chinensis roots and their antinociceptive and antimalarial activities. Tetrahedron 2000, 56, 7673–7678. [Google Scholar] [CrossRef]
- Poornima, B.; Siva, B.; Shankaraiah, G.; Venkanna, A.; Nayak, V.L.; Ramakrishna, S.; Rao, C.V.; Babu, K.B. Novel sesquiterpenes from Schisandra grandiflora: Isolation, cytotoxic activity and synthesis of their triazole derivatives using “click” reaction. Eur. J. Med. Chem. 2015, 92, 449–458. [Google Scholar] [CrossRef]
- Dong, J.Y.; Ma, X.Y.; Cai, X.Q.; Yan, P.C.; Yue, L.; Lin, C. Sesquiterpenoids from Curcuma wenyujin with anti-influenza viral activities. Phytochemistry 2013, 85, 122–128. [Google Scholar] [CrossRef] [PubMed]
- Zaugg, J.M. Discovery of New Scaffolds for GABA (A) Receptor Modulators from Natural Origin. Ph.D. Thesis, Universität Basel, Basel, Switzerland, 2011. [Google Scholar]
- Sy, L.K.; Brown, G.D. Labdane diterpenoids from Alpinia chinensis. J. Nat. Prod. 1997, 60, 904–908. [Google Scholar] [CrossRef]
- Zhao, X.; Zheng, G.W.; Niu, X.M.; Li, W.Q.; Wang, F.S.; Li, S.H. Terpenes from Eupatorium adenophorum and their allelopathic effects on Arabidopsis seeds germination. J. Agric. Food Chem. 2009, 57, 478–482. [Google Scholar] [CrossRef]
- Loyola, L.A.; Morales, G.; Rodriguez, B.; Jiménez-Barbero, J.; De La Torre, M.C.; Perales, A.; Torres, M.R. Mulinic and isomulinic acids. Rearranged diterpenes with a new carbon skeleton from Mulinum crassifolium. Tetrahedron 1990, 46, 5413–5420. [Google Scholar] [CrossRef]
- Loyola, L.A.; Morales, G.; De La Torre, M.C.; Pedreros, S.; Rodríguez, B. 17-Acetoxymulinic acid, a rearranged diterpenoid from Mulinum crassifolium. Phytochemistry 1990, 29, 3950–3951. [Google Scholar] [CrossRef]
- Rojas-Alvareza, F.; Campos-Brionesa, C.; Lima, C.; Pérez, E.G.; Sepúlveda, B. Further mulinane diterpenoids from Azorella compacta Carlos Arechea. J. Pharm. Pharmacol. 2013, 65, 1231–1238. [Google Scholar]
- de Jesús Dzul-Beh, A.; Uc-Cachón, A.H.; Bórquez, J.; Loyola, L.A.; Peña-Rodríguez, L.M.; Molina-Salinas, G.M. Mulinane- and azorellane-type diterpenoids: A systematic review of their biosynthesis. Chem. Pharm. Biomol. 2020, 10, 1333. [Google Scholar]
- Itokawa, H.; Tachi, Y.; Kamano, Y.; Iitaka, Y. Structure of gilvanol, a new triterpene isolated from Quercus gilva Bume. Chem. Phar. Bull. 1978, 26, 331–333. [Google Scholar] [CrossRef]
- Itokawa, H.; Morita, H.; Katou, I.; Takeya, K.; Cavalheiro, A.J.; de Oliveira, R.C.B.; Ishige, M.; Motidome, M. Cytotoxic diterpenes from the rhizomes of Hedychium coronarium. Planta Med. 1988, 54, 311–315. [Google Scholar] [CrossRef]
- Kamchonwongpaisan, S.; Nilanonta, C.; Tarnchompoo, B.; Thebtaranonth, C.; Thebtaranonth, Y.; Yuthavong, Y.; Kongsaeree, P.; Clardy, J. An antimalarial peroxide from Amomum krervanh Pierre. Tetrahedron Lett. 1995, 36, 1821–1824. [Google Scholar] [CrossRef]
- Qinghaosu Antimalaria Coordinating Research Group. Antimalaria studies on qinghaosu. Chin. Med. J. 1979, 92, 811–816. [Google Scholar]
- Balint, G.A. Artemisinin and its derivatives: An important new class of antimalarial agents. Pharmacol. Therapeut. 2001, 90, 261–265. [Google Scholar] [CrossRef]
- McIntosh, H.; Olliaro, P. Cochrane infectious diseases group. Artemisinin derivatives for treating severe malaria. Cochrane Database Syst. Rev. 1998, 3, CD000527. [Google Scholar]
- Crespo-Ortiz, M.P.; Wei, M.Q. Antitumor activity of artemisinin and its derivatives: From a well-known antimalarial agent to a potential anticancer drug. J. Biomed. Biotech. 2012, 18, 247597. [Google Scholar] [CrossRef]
- Kiani, B.H.; Kayani, W.K.; Khayam, A.U.; Dilshad, E.; Ismail, H.; Mirza, B. Artemisinin and its derivatives: A promising cancer therapy. Mol. Biol. Rep. 2020, 47, 6321–6336. [Google Scholar] [CrossRef] [PubMed]
- Carolino, K.; Winzeler, E.A. The antimalarial resistome–finding new drug targets and their modes of action. Current Opin. Microbiol. 2020, 57, 49–55. [Google Scholar] [CrossRef] [PubMed]
- Gao, F.; Sun, Z.; Kong, F.; Xiao, J. Artemisinin-derived hybrids and their anticancer activity. Eur. J. Med. Chem. 2020, 188, 112044. [Google Scholar] [CrossRef] [PubMed]
- Schlesinger, P.H.; Krogstad, D.J.; Herwaldt, B.L. Antimalarial agents: Mechanisms of action. Antimicrob. Agents Chemther. 1988, 12, 793–798. [Google Scholar] [CrossRef] [PubMed]
- Slezakova, S.; Ruda-Kucerova, J. Anticancer activity of artemisinin and its derivatives. Anticancer Res. 2017, 37, 5995–6003. [Google Scholar] [PubMed]
- Wong, Y.K.; Xu, C.; Kalesh, K.A.; He, Y.; Lin, Q.; Wong, W.S.F.; Shen, H.M.; Wang, J. Artemisinin as an anticancer drug: Recent advances in target profiling and mechanisms of action. Med. Res. Rev. 2017, 37, 1–26. [Google Scholar] [CrossRef]
- Bhattacharjee, M.K. Antifungals, antimalarials, and antivirals. Chem. Antibiot. Relat. Drugs 2016, 12, 175–195. [Google Scholar]
- Nagashima, F.; Suzuki, M.; Takaoka, S.; Asakawa, Y. New sesqui- and diterpenoids from the Japanese liverwort Jungermannia infusca (Mitt.) Sterh. Chem. Pharm. Bull. 1998, 46, 1184–1191. [Google Scholar] [CrossRef][Green Version]
- Nagashima, F.; Suzuki, M.; Takaoka, S.; Asakawa, Y. New acorane-and cuparane-type sesqui-and new labdane-and seco-labdanne-type diterpenoids from the Japanese liverwort Jungermannia infusca (Mitt.) Steph. Tetrahedron 1999, 55, 9117–9121. [Google Scholar] [CrossRef]
- Chokpaiboon, S.; Sommit, D.; Bunyapaiboonsri, T.; Matsubara, K.; Pudhom, K. Antiangiogenic effect of chamigrane endoperoxides from a Thai mangrove-derived fungus. J. Nat. Prod. 2011, 74, 2290–2299. [Google Scholar] [CrossRef]
- Chen, H.J.; Wu, Y. Expeditious entry to the chamigrane endoperoxide family of natural products. Org. Lett. 2015, 17, 592–595. [Google Scholar] [CrossRef] [PubMed]
- Ngo, K.S.; Brown, G.D. Allohimachalane, seco-allohimachalane and himachalane sesquiterpenes from Illicium tsangii. Tetrahedron 1999, 55, 759–766. [Google Scholar] [CrossRef]
- Ngo, K.S.; Brown, G.D. Santalane and isocampherenane sesquiterpenoids from Illicium tsangii. Phytochemistry 1999, 50, 1213–1219. [Google Scholar] [CrossRef]
- Ngo, K.S.; Wong, W.T.; Brown, G.D. Muurolane sesquiterpenes from Illicium tsangii. J. Nat. Prod. 1999, 62, 549–556. [Google Scholar] [CrossRef]
- Ma, W.H.; Tan, C.M.; He, J.C.; Duan, P.S.; Qin, L.P. A novel eudesmene sesquiterpenoid from Schisandra sphenanthera stems. Chem. Nat. Comp. 2011, 47, 713–717. [Google Scholar] [CrossRef]
- Adio, A.M.; König, W.A. Sesquiterpene constituents from the essential oil of the liverwort Plagiochila asplenioides. Phytochemistry 2005, 66, 599–609. [Google Scholar] [CrossRef]
- Nagashima, F.; Matsumura, N.; Ashigaki, Y.; Asakawa, Y. Chemical constituents of the liverworts Bryopteris filicina, Plagiochila asplenioides and Porella canariensis. J. Hattori Bot. Lab. 2003, 94, 197–204. [Google Scholar]
- Kundu, A.; Saha, S.; Ahluwalia, V.; Walia, S. Plant growth inhibitory terpenes from Eupatorium adenophorum leaves. J. Appl. Bot. Food Qual. 2013, 86, 33–36. [Google Scholar]
- Wang, C.F.; Zhao, Y.; Liu, Y.Z.; Zhang, Z.Z. Occurrence and biological activities of eremophilane-type sesquiterpenes. Chem. Res. Chinese Univ. 2009, 25, 480–484. [Google Scholar]
- He, L.; Hou, J.; Gan, M.; Shi, J.; Chantrapromma, F.S.; Williams, I.D.; Sung, H.H.Y. Cadinane sesquiterpenes from the leaves of Eupatorium adenophorum. J. Nat. Prod. 2008, 71, 1485–1488. [Google Scholar] [CrossRef]
- Moreira, I.C.; Roque, N.F.; Contini, K.; Lago, J.H.G. Sesquiterpenes and hydrocarbons from Xylopia emarginata (Annonaceae) fruits. Rev. Bras. Farmacogn. 2007, 17, 55–62. [Google Scholar] [CrossRef]
- Müller, S.; Murillo, R.; Castro, V.; Brecht, V.; Merfort, I. Sesquiterpene lactones from Montanoa hibiscifolia that inhibit the transcription factor NF-κB. J. Nat. Prod. 2004, 67, 622–630. [Google Scholar] [CrossRef] [PubMed]
- Mahmoud, A.A. Xanthanolides and xanthane epoxide derivatives from Xanthium strumarium. Planta Med. 1998, 64, 724–727. [Google Scholar] [CrossRef] [PubMed]
- Marco, J.A.; Sanz, J.F.; Falco, E.; Jakupovic, J.; Lex, J. New oxygenated eudesmanolides from Artemisia herba-alba. Tetrahedron 1990, 46, 7941–7950. [Google Scholar] [CrossRef]
- Aguilar-Guadarrama, A.B.; Rios, M.Y. Three new sesquiterpenes from Croton arboreous. J. Nat. Prod. 2004, 67, 914–917. [Google Scholar] [CrossRef] [PubMed]
- Rustaiyan, A.; Faridchehr, A.; Bakhtiyar, M. Sesquiterpene lactones of Iranian Compositae family (Astraceae); their chemical constituents and anti-plasmodial properties of tehranolide (A. Review). Orient J. Chem. 2017, 33. [Google Scholar] [CrossRef]
- Taniguchi, Y.; Taniguchi, H.; Matsukura, Y.; Kawachi, Y.; Shindo, K. Structural elucidation of humulone autoxidation products and analysis of their occurrence in stored hops. J. Nat. Prod. 2014, 77, 1252–1261. [Google Scholar] [CrossRef]
- Yin, S.; Wang, X.N.; Fan, C.Q.; Liao, S.G.; Yue, J.M. The first limonoid peroxide in the Meliaceae family: Walsuronoid A from Walsura robusta. Org. Lett. 2007, 9, 2353–2356. [Google Scholar] [CrossRef]
- Chen, G.F.; Li, Z.L.; Tang, C.M.; He, X.; Chen, K.; Pan, D.J.; Hu, C.Q.; McPhail, D.R.; McPhail, A.T.; Lee, K.H. Structure and stereochemistry of pseudolarolide-I, a novel cytotoxic peroxytriterpene dilactone from Pseudolarix kaempferi. Heterocycles 1990, 31, 1903–1906. [Google Scholar]
- Zhou, T.; Zhang, H.; Zhu, N.; Chiu, P. New triterpene peroxides from Pseudolarix kaempferi. Tetrahedron 2004, 60, 4931–4936. [Google Scholar] [CrossRef]
- Ali, Z.; Khan, I.A.; Fronczek, F.R. Revision of the structure of podocarpaside E, from Actaea podocarpa. Acta Crystallograph. 2007, 63, o2101–o2103. [Google Scholar]
- Ding, Y.; Liang, C.; Kim, J.H.; Lee, Y.M.; Hyun, J.H.; Kang, H.K.; Kim, J.A.; Min, B.S.; Kim, Y.H. Triterpene compounds isolated from Acer mandshuricum and their anti-inflammatory activity. Bioorg. Med. Chem. Lett. 2010, 20, 1528–1531. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.P.; Li, N.; Gao, J.; Fu, K.L.; Qin, Y.; Li, G.Y.; Wang, J.H. A new peroxy-multiflorane triterpene ester from the processed seeds of Trichosanthes kirilowii. Helv. Chim. Acta 2011, 94, 1881–1889. [Google Scholar] [CrossRef]
- Chiamg, Y.M.; Kuo, Y.H. New peroxy triterpenes from the aerial roots of Ficus microcarpa. J. Nat. Prod. 2001, 64, 436–439. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.J.; Fei, D.Q.; Chen, S.G.; Gao, K. Antimicrobial triterpenoids from Vladimiria muliensis. J. Nat. Prod. 2008, 71, 547–550. [Google Scholar] [CrossRef]
- Saha, B.; Naskar, D.B.; Misra, D.R.; Pradhan, B.P.; Khastgir, H.N. Baccatin, a novel nor-triterpene peroxide isolated from Sapium baccatum roxb. Tetrahedron Lett. 1977, 26, 3095–3098. [Google Scholar] [CrossRef]
- Wu, Q.X.; Liu, X.; Shi, Y.P. Chemical components from Gentiana aristata. Chem. Biodiver. 2007, 4, 175–179. [Google Scholar] [CrossRef]
- Makino, B.; Kawai, M.; Iwata, Y.; Yamamura, H.; Butsugan, Y.; Ogawa, K.; Hayashi, M. Physalins possessing an endoperoxy structure from Physalis alkekengi var. francheti. Structural revision of physalin K. Bull. Chem. Soc. Japan 1995, 68, 219–223. [Google Scholar] [CrossRef]
- Cirigliano, A.M.; Veleiro, A.S.; Oberti, J.C.; Burton, G. Spiranoid withanolides from Jaborosa odonelliana. J. Nat. Prod. 2002, 65, 1049–1054. [Google Scholar] [CrossRef]
- Casero, C.N.; Oberti, J.C.; Orozco, C.I.; Cárdenas, A.; Brito, I.; Barboza, G.E.; Nicotra, V.E. Withanolides from three species of the genus Deprea (Solanaceae). Chemotaxonomical considerations. Phytochemistry 2015, 110, 83–90. [Google Scholar] [CrossRef]
- Rocha, M.R.; de Souza, J.J.; Barcellos, L.T.; Sant’Anna, C.M.; Braz-Filho, R.; Vieira, I.J. A novel 3,9-(1,2,3-trioxocine)-type steroid of Rauia nodosa (Rutaceae). Molecules 2014, 19, 14637–14648. [Google Scholar] [CrossRef] [PubMed]
- He, F.; Pu, J.X.; Huang, S.X.; Wang, Y.Y.; Xiao, W.L.; Li, L.M.; Liu, J.P.; Zhang, H.B.; Li, Y.; Sun, H.D. Schinalactone A, a new cytotoxic triterpenoid from Schisandra sphenanthera. Org. Lett. 2010, 12, 1208–1211. [Google Scholar] [CrossRef] [PubMed]
- Van Der Vijver, L.M. Distribution of plumbag in in the Plumbaginaceae. Phytochemistry 1974, 11, 3247–3248. [Google Scholar] [CrossRef]
- Uchiyama, T.; Hara, S.; Makino, M.; Fujimoto, Y. Seco-Adianane-type triterpenoids from Dorstenia brasiliensis (Moraceae). Phytochemistry 2002, 60, 761–764. [Google Scholar] [CrossRef]
- Wautie, A. Prophylaxis and treatment of the chief parasitic diseases of the gastrointestinal tract of the horse. Parasitica 1946, 2, 44–67. [Google Scholar]
- Herz, W.; Watanabe, K.; Kulanthaivel, P.; Blount, J.F. Cycloartanes from Lindheimera texana. Phytochemistry 1985, 24, 2645–2654. [Google Scholar] [CrossRef]
- Dev, S. Handbook of Terpenoids: Triterpenoids, 1st ed.; CRC Press: Boca Raton, FL, USA, 1989; Volume I. [Google Scholar]
- Ageta, H.; Shiojima, K.; Kamaya, R.; Masuda, K. Fern constituent: Naturally occurring adian-5-ene ozonide in the leaves of Adiantum monochlamys and Oleandra wallichii. Tetrahedron Lett. 1978, 19, 899–900. [Google Scholar] [CrossRef]
- Rücker, G.; Manns, D.; Schenkel, E.P.; Hartmann, R.; Heinzmann, B.M. A triterpene ozonide from Senecio Selloi. Arch. Pharm. Med. Chem. 2003, 336, 205–207. [Google Scholar] [CrossRef]
- Kazakova, O.B.; Kazakov, D.V.; Yamansarov, E.Y.; Medvedeva, N.I.; Tolstikov, G.A.; Suponitsky, K.Y.; Arkhipov, D.E. Synthesis of triterpenoid-based 1,2,4-trioxolanes and 1,2,4-dioxazolidines by ozonolysis of allobetulin derivatives. Tetrahedron Lett. 2011, 52, 976–979. [Google Scholar] [CrossRef]
- Omathúna, D.P.; Doskotch, R.W. Amoenolide K and amoenolide K 19-acetate, two grindelane peroxides from Amphiachyris amoena. Isolation, structure determination, and preparation of amoenolide K from amoenolide A by photochemical oxygenation. J. Nat. Prod. 1995, 58, 1407. [Google Scholar] [CrossRef]
- Habtemariam, S.; Gray, A.I.; Lavaud, C.; Massiot, G.; Skelton, B.W.; Waterman, P.G.; White, A.H. ent-12-Oxolabda-8,13(16)-dien-15-oic acid and ent-8β,12α-epidioxy-12β-hydroxylabda-9(11),13-dien-15-oic acidγ-lactone: Two new diterpenes from the aerial parts of Premna oligotricha. J. Chem. Soc. Perkin Trans. 1991, 1, 893–899. [Google Scholar] [CrossRef]
- Barrero, A.F.; del Moral, J.F.Q.; Aitigri, M. Oxygenated diterpenes and other constituents from Moroccan Juniperus phoenicea and Juniperus thurifera var. africana. Phytochemistry 2004, 65, 2507–2515. [Google Scholar] [CrossRef] [PubMed]
- Escudero, J.; Perez, L.; Rabanal, R.M.; Valverde, S. Diterpenoids from Salvia oxyodon and Salvia lavandulifolia. Phyfochemistry 1983, 22, 585–588. [Google Scholar] [CrossRef]
- Barrero, A.F.; Sanchez, J.F.; Alvarez-Mansaneda, E.J.; Dorado, M.M.; Haidour, A. Endoperoxide diterpenoids and other constituents from Abies marocana. Phytochemistry 1991, 30, 593–597. [Google Scholar] [CrossRef]
- Delgado, G.; Sanchez, E.; Hernandez, J.; Chavez, M.I.; Alvarez, L.; Martinez, E. Abietanoid acid from Lepechinia caulescens. Phytochemistry 1992, 31, 3159–3161. [Google Scholar] [CrossRef]
- San Feliciano, A.; del Corral, J.M.M.; Gordaliza, M.; Castro, M.A. Two diterpenoids from leaves of Juniperus sabina. Phytochemistry 1991, 30, 695–697. [Google Scholar] [CrossRef]
- Maslovskaya, L.A.; Savchenko, A.I.; Pierce, C.J.; Gordon, V.A.; Reddell, P.W.; Parsons, P.G.; Williams, C.M. Unprecedented 1,14-seco-crotofolanes from Croton insularis: Oxidative cleavage of crotofolin C by a putative homo-baeyer-villiger rearrangement. Chem. Eur. J. 2014, 20, 226–14230. [Google Scholar] [CrossRef]
- Maslovskaya, L.A.; Savchenko, A.I.; Gordon, V.A.; Reddell, P.W.; Pierce, C.J.; Parsons, P.G.; Williams, C.M. Isolation and confirmation of the proposed cleistanthol biogentic link from Croton insularis. Org. Lett. 2011, 13, 1032–1035. [Google Scholar] [CrossRef]
- Qu, J.B.; Zhu, R.L.; Zhang, Y.L.; Guo, H.F.; Wang, X.N.; Xie, C.F.; Yu, W.T.; Ji, M.; Lou, H.X. ent-Kaurane diterpenoids from the liverwort Jungermannia atrobrunnea. J. Nat. Prod. 2008, 71, 1418–1422. [Google Scholar] [CrossRef]
- Guo, F.; Xi, M.; Li, Y. Triptotin A and B, two novel diterpenoids from Tripterygium wilfordii. Tetrahedron Lett. 1999, 40, 947–950. [Google Scholar] [CrossRef]
- Kong, L.Y.; Min, Z.D.; Shi, J.X. Chemical constituents from roots of Jatropha curcas. J. China Pharm. Univ. 1996, 38, 161–166. [Google Scholar]
- Adelekan, A.M.; Prozesky, E.A.; Hussein, A.A.; Urena, L.D.; van Rooyen, P.H.; Liles, D.C.; Meyer, J.J.M.; Rodriguez, B. Bioactive diterpenes and other constituents of Croton steenkampianus. J. Nat. Prod. 2008, 71, 1919–1922. [Google Scholar] [CrossRef] [PubMed]
- Bridi, H.; de Carvalho Meirelles, G.; von Poser, G.L. Structural diversity and biological activities of phloroglucinol derivatives from Hypericum species. Phytochemistry 2018, 155, 203–232. [Google Scholar] [CrossRef] [PubMed]
- Liao, Y.; Liu, X.; Yang, J.; Lao, Y.Z.; Yang, X.W.; Li, X.N.; Zhang, J.J.; Ding, Z.J.; Xu, H.X.; Xu, G. Hypersubones A and B, new polycyclic acylphloroglucinols with intriguing adamantane type cores from Hypericum subsessile. Org. Lett. 2015, 17, 1172–1175. [Google Scholar] [CrossRef] [PubMed]
- Zhu, H.C.; Chen, C.M.; Zhang, J.W.; Guo, Y.; Tan, D.D.; Wei, G.Z. Hyperisampsins N and O, two new benzoylated phloroglucinol derivatives from Hypericum sampsonii. Chin. Chem. Lett. 2017, 28, 986–990. [Google Scholar] [CrossRef]
- Xiao, Z.Y.; Zeng, Y.H.; Mu, Q.; Ka, W.; Shiu, P.; Gibbons, S. Prenylated benzophenone peroxide derivatives from Hypericum sampsonii. Chem. Biodiver. 2010, 7, 953–958. [Google Scholar] [CrossRef]
- Henry, G.E.; Jacobs, H.; Carrington, C.M.S.; McLean, S.; Reynolds, W.F. Prenylated benzophenone derivatives from Caribbean Clusia species (Guttiferae). Plukenetiones B-G and xerophenone A. Tetrahedron 1999, 55, 1581–1596. [Google Scholar] [CrossRef]
- Ishida, Y.; Shirota, O.; Sekita, S. Polyprenylated benzoylphloroglucinoltype derivatives including novel cage compounds from Hypericum erectum. Chem. Pharm. Bull. 2010, 58, 336–343. [Google Scholar] [CrossRef]
- Ting, C.W.; Hwang, T.L.; Chen, I.S.; Cheng, M.J.; Sung, P.J.; Yen, M.H.; Chen, J.J. Garcimultiflorone G, a novel benzoylphloroglucinol derivative from Garcinia multiflora with inhibitory activity on neutrophil pro-inflammatory. Responses. Chem. Biodivers. 2014, 11, 819–824. [Google Scholar] [CrossRef]
- Novais, C.; Kato, L.; Terra, F.; Vaz, M.B.G.; de Oliveira, C.M.A. Goianone, a new natural polyprenylated benzophenone with a homo-adamantyl ketone core spiro-fused to a cyclic peroxide. Planta Med. 2016, 82S, S1–S381. [Google Scholar] [CrossRef]
- Itokawa, H.; Xu, J.; Takeya, K.; Watanabe, K. Studies on chemical constituents of antitumor fraction from Periploca sepium. II.: Structures of new pregnane glycosides, periplocosides A, B and C. Chem. Pharm. Bull. 1988, 36, 982–987. [Google Scholar] [CrossRef] [PubMed]
- Itokawa, H.; XU, J.; Takeya, K. Studies on chemical constituents of antitumor fraction from Periploca sepium. IV.: Structures of new pregnane glycosides, periplocosides D, E, L, and M. Chem. Pharm. Bull. 1988, 36, 2084–2089. [Google Scholar] [CrossRef] [PubMed]
- Itokawa, H.; Xu, J.; Takeya, K. Studies on chemical constituents of antitumor fraction from Periploca sepium. V. Structures of new pregnane glycosides, periplocosides J, K, F and O. Chem. Pharm. Bull. 1988, 36, 4441–4446. [Google Scholar] [CrossRef] [PubMed]
- Vázquez, J.; López, M.; Gibert, E.; Herrero, E.; Luque, F.J. Merging ligand-based and structure-based methods in drug discovery: An overview of combined virtual screening approaches. Molecules 2020, 25, 4723. [Google Scholar] [CrossRef] [PubMed]
- Filimonov, D.A.; Poroikov, V.V. Probabilistic approach in activity prediction. In Chemoinformatics Approaches to Virtual Screening; Varnek, A., Tropsha, A., Eds.; RSC Publishing: Cambridge, UK, 2008; pp. 182–216. [Google Scholar]
- Stumpfe, D.; Hu, H.; Bajorath, J. Evolving concept of activity cliffs. ACS Omega 2019, 4, 14360–14368. [Google Scholar] [CrossRef] [PubMed]
- Wermuth, C.G.; Aldous, D.; Raboisson, R.; Rognan, D. (Eds.) The Practice of Medicinal Chemistry, 4th ed.; Academic Press: Amsterdam, The Netherlands, 2015; p. 902. [Google Scholar]
- Mervin, L.H.; Afzal, A.M.; Drakakis, G.; Lewis, R.; Engkvist, O.; Bender, A. Target prediction utilizing negative bioactivity data covering large chemical space. J. Cheminform. 2015, 7, 51. [Google Scholar] [CrossRef] [PubMed]
- Burov, Y.V.; Poroikov, V.V.; Korolchenko, L.V. National system for registration and biological testing of chemical compounds: Facilities for new drugs search. Bull. Natl. Center Biol. Act. Comp. 1990, 1, 4–25. [Google Scholar]
- Filimonov, D.A.; Lagunin, A.A.; Gloriozova, T.A.; Rudik, A.V.; Druzhilovskii, D.S.; Pogodin, P.V.; Poroikov, V.V. Prediction of the biological activity spectra of organic compounds using the PASS online web resource. Chem. Heterocycl. Comp. 2014, 50, 444–457. [Google Scholar] [CrossRef]
- Filimonov, D.A.; Druzhilovskiy, D.S.; Lagunin, A.A.; Gloriozova, T.A.; Rudik, A.V.; Dmitriev, A.V.; Pogodin, P.V.; Poroikov, V.V. Computer-aided prediction of biological activity spectra for chemical compounds: Opportunities and limitations. Biomed. Chem. Res. Meth. 2018, 1, e00004. [Google Scholar] [CrossRef]
- Available online: http://www.way2drug.com/passonline/ (accessed on 20 October 2020).
- Druzhilovskiy, D.S.; Rudik, A.V.; Filimonov, D.A.; Gloriozova, T.A.; Lagunin, A.A.; Dmitriev, A.V.; Pogodin, P.V.; Dubovskaya, V.I.; Ivanov, S.M.; Tarasova, O.A.; et al. Computational platform Way2Drug: From the prediction of biological activity to drug repurposing. Russ. Chem. Bull. 2017, 66, 1832–1841. [Google Scholar] [CrossRef]
- Dembitsky, V.M.; Gloriozova, T.A.; Poroikov, V.V. Pharmacological and predicted activities of natural azo compounds. Nat. Prod. Bioprospec. 2017, 7, 151–169. [Google Scholar] [CrossRef] [PubMed]
- Horishny, V.; Kartsev, V.; Matiychuk, V.; Geronikaki, A.; Anthi, P.; Pogodin, P.; Poroikov, V.; Ivanov, M.; Kostic, M.; Soković, M.D.; et al. 3-Amino-5-(indol-3-yl) methylene-4-oxo-2-thioxothiazolidine derivatives as antimicrobial agents: Synthesis, computational and biological evaluation. Pharmaceuticals 2020, 13, 229. [Google Scholar] [CrossRef] [PubMed]
- Amiranashvili, L.; Nadaraia, N.; Merlani, M.; Kamoutsis, C.; Petrou, A.; Geronikaki, A.; Pogodin, P.; Druzhilovskiy, D.; Poroikov, V.; Ciric, A.; et al. Antimicrobial activity of nitrogencontaining 5-alpha-androstane derivatives: In silico and experimental studies. Antibiotics 2020, 9, 224. [Google Scholar] [CrossRef] [PubMed]
- Dembitsky, V.M.; Gloriozova, T.A.; Poroikov, V.V. Biological activities of organometalloid (As, At, B, Ge, Si, Se, Te) steroids. J. Appl. Pharm. Sci. 2017, 7, 184–202. [Google Scholar]
- Singh, S.; Bharti, N.; Mohapatra, P.P. Chemistry and biology of synthetic and naturally occurring antiamoebic agents. Chem. Rev. 2009, 109, 1900–1947. [Google Scholar] [CrossRef]
- Teixeira, C.; Vale, N.; Pérez, B.; Gomes, A.; Gomes, J.R.B.; Gomes, P. “Recycling” classical drugs for malaria. Chem. Rev. 2014, 114, 11164–11220. [Google Scholar] [CrossRef]
- Salas, P.F.; Herrmann, C.; Orvig, C. Metalloantimalarials. Chem. Rev. 2013, 5, 3450–3492. [Google Scholar] [CrossRef]
- Vizer, S.A.; Sycheva, E.S.; Quntar, A.A.A.; Kurmankulov, N.B.; Yerzhanov, K.B.; Dembitsky, V.M. Propargylic sulfides: Synthesis, properties, and application. Chem. Rev. 2015, 115, 1475–1502. [Google Scholar] [CrossRef]
- Barnett, D.S.; Guy, R.K. Antimalarials in development in 2014. Chem. Rev. 2014, 114, 11221–11241. [Google Scholar] [CrossRef]
- Dembitsky, V.M.; Quntar, A.A.A.; Srebnik, M. Natural and synthetic small boron-containing molecules as potential inhibitors of bacterial and fungal quorum sensing. Chem. Rev. 2011, 111, 209–237. [Google Scholar] [CrossRef]
- Smoum, R.; Rubinstein, A.; Dembitsky, V.M.; Srebnik, M. Boron containing compounds as protease inhibitors. Chem. Rev. 2012, 112, 4156–4220. [Google Scholar] [CrossRef] [PubMed]
- Dembitsky, V.M. Anticancer activity of natural and synthetic acetylenic lipids. Lipids 2006, 41, 883–924. [Google Scholar] [CrossRef] [PubMed]
- Siddiq, A.; Dembitsky, V. Acetylenic anticancer agents. Anti-Cancer Agents Med. Chem. 2008, 8, 132–170. [Google Scholar] [CrossRef] [PubMed]
- Dembitsky, V.M.; Levitsky, D.O. Acetylenic terrestrial anticancer agents. Nat. Prod. Commun. 2006, 1, 405–429. [Google Scholar] [CrossRef]
- Kuklev, D.V.; Dembitsky, V.M. Epoxy acetylenic lipids: Their analogues and derivatives. Prog. Lipid Res. 2014, 56, 67–91. [Google Scholar] [CrossRef]
- Levitsky, D.O.; Dembitsky, V.M. Anti-breast cancer agents derived from plants. Nat. Prod. Bioprosp. 2015, 5, 1–16. [Google Scholar] [CrossRef]
- Dembitsky, V.M.; Levitsky, D.O.; Gloriozova, T.A.; Poroikov, V.V. Acetylenic aquatic anticancer agents and related compounds. Nat. Prod. Commun. 2006, 1, 773–811. [Google Scholar] [CrossRef]
- Abu-Lafi, S.; Dembicki, J.W.; Goldshlag, P.; Hanuš, L.O.; Dembitsky, V.M. The use of the Cryogenic GC/MS and on-column injection for study of organosulfur compounds of the Allium sativum. J. Food Composit. Anal. 2004, 17, 235–245. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).