Inclusion Compound of Efavirenz and γ-Cyclodextrin: Solid State Studies and Effect on Solubility
Abstract
1. Introduction
2. Results and Discussion
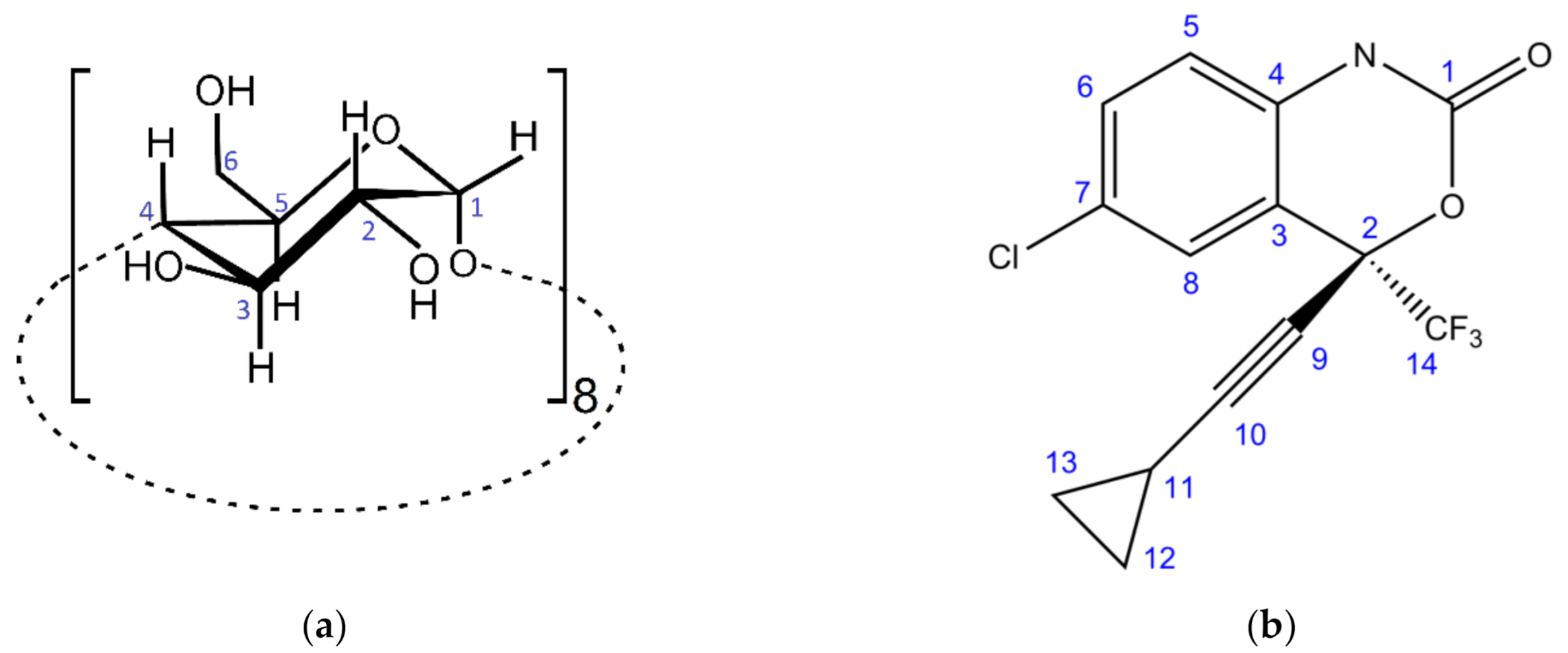
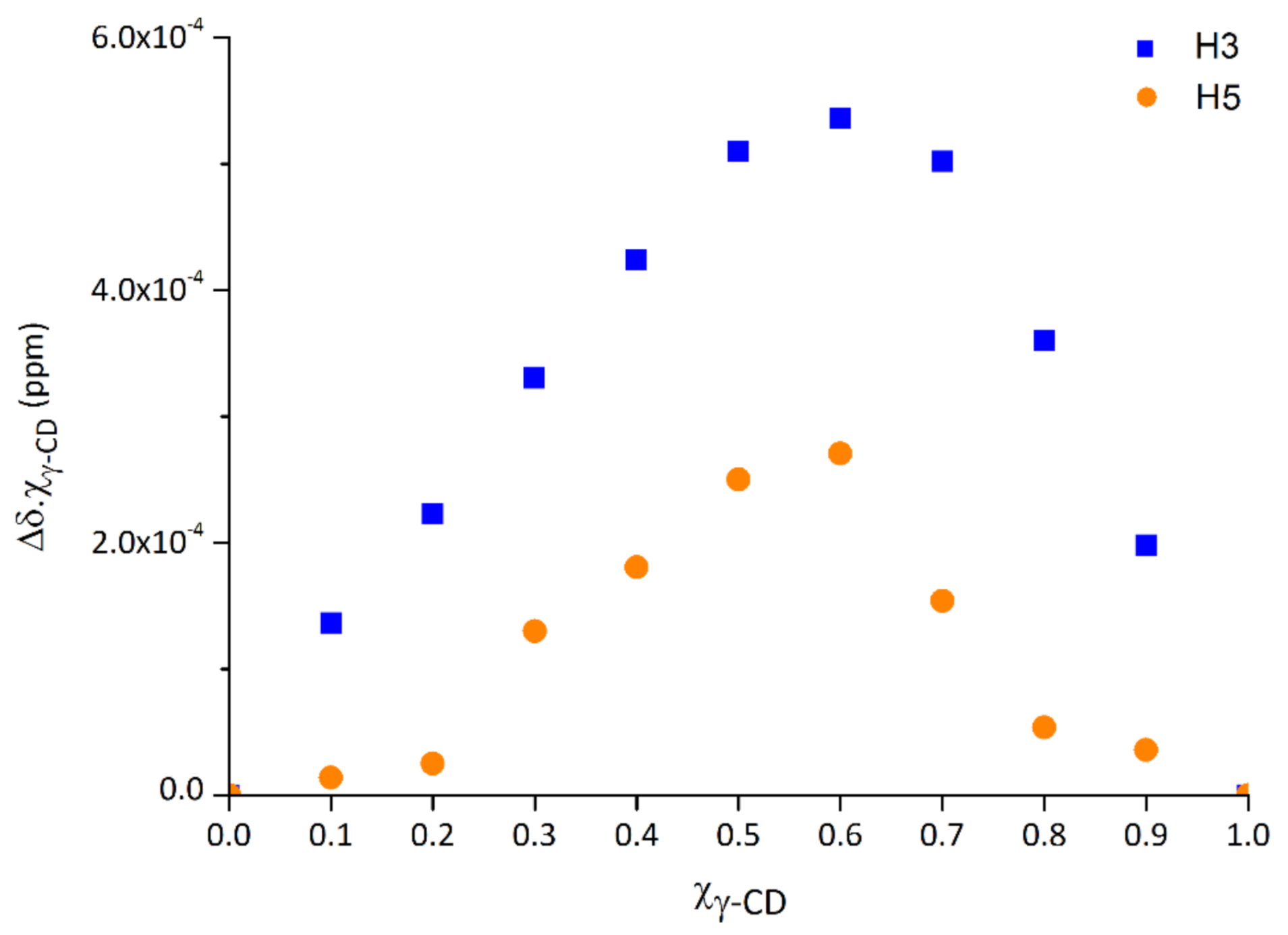
2.1. Stoichiometry in a Water–Methanol Solution
2.2. Solid-State Studies
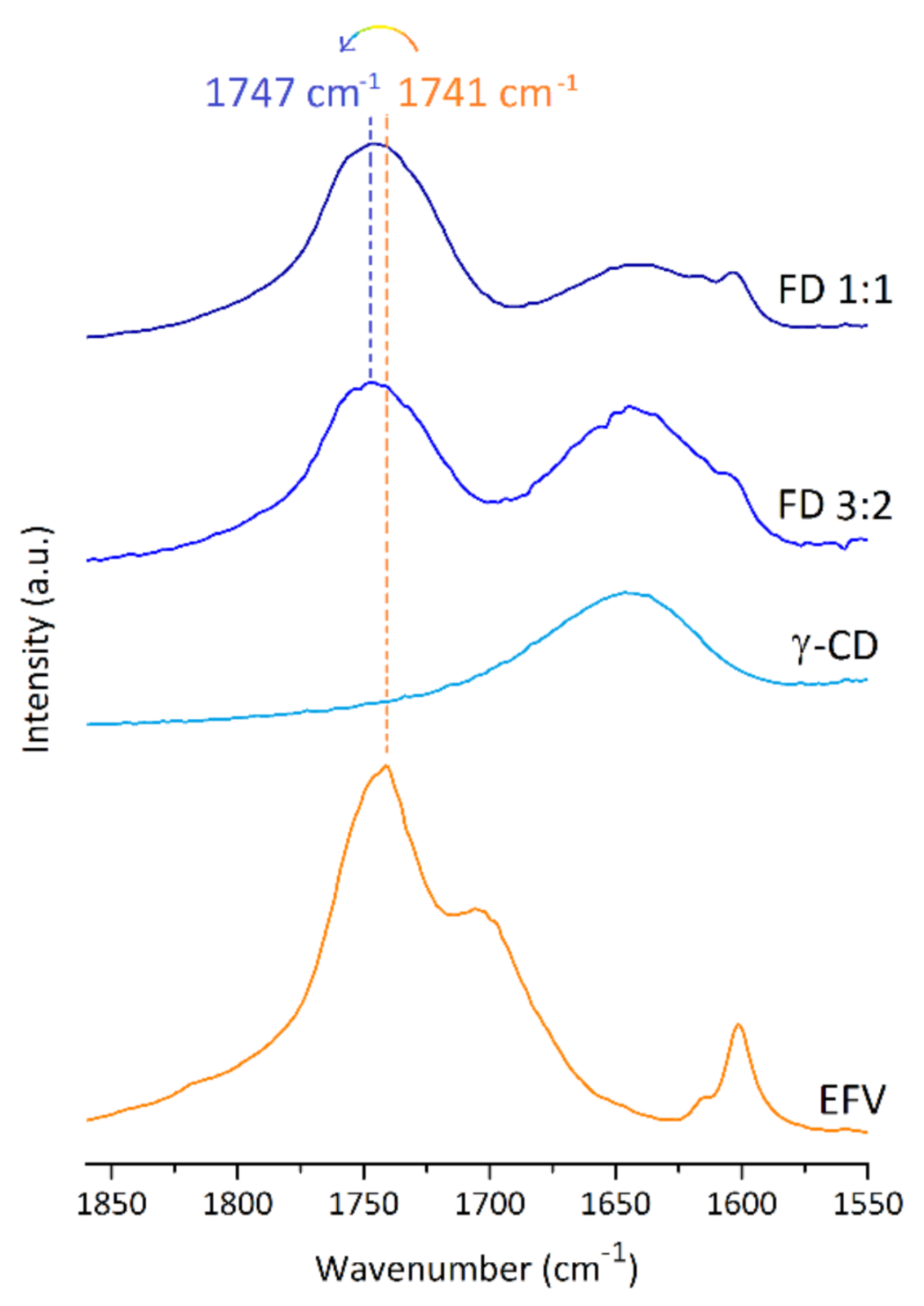
2.2.1. FT-IR Spectroscopy
2.2.2. Powder X-ray Diffraction
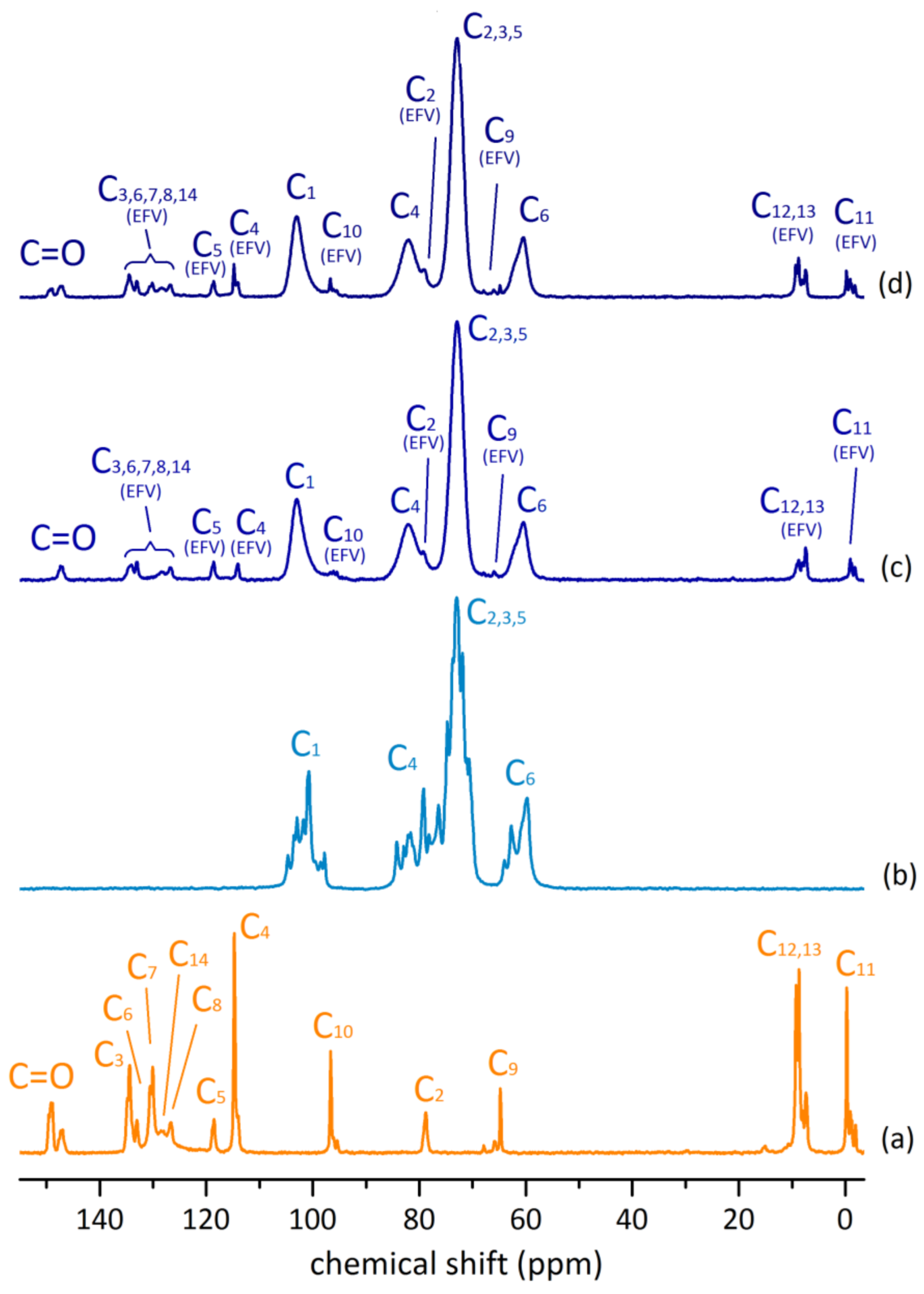
2.2.3. 13C{1H} CP-MAS NMR
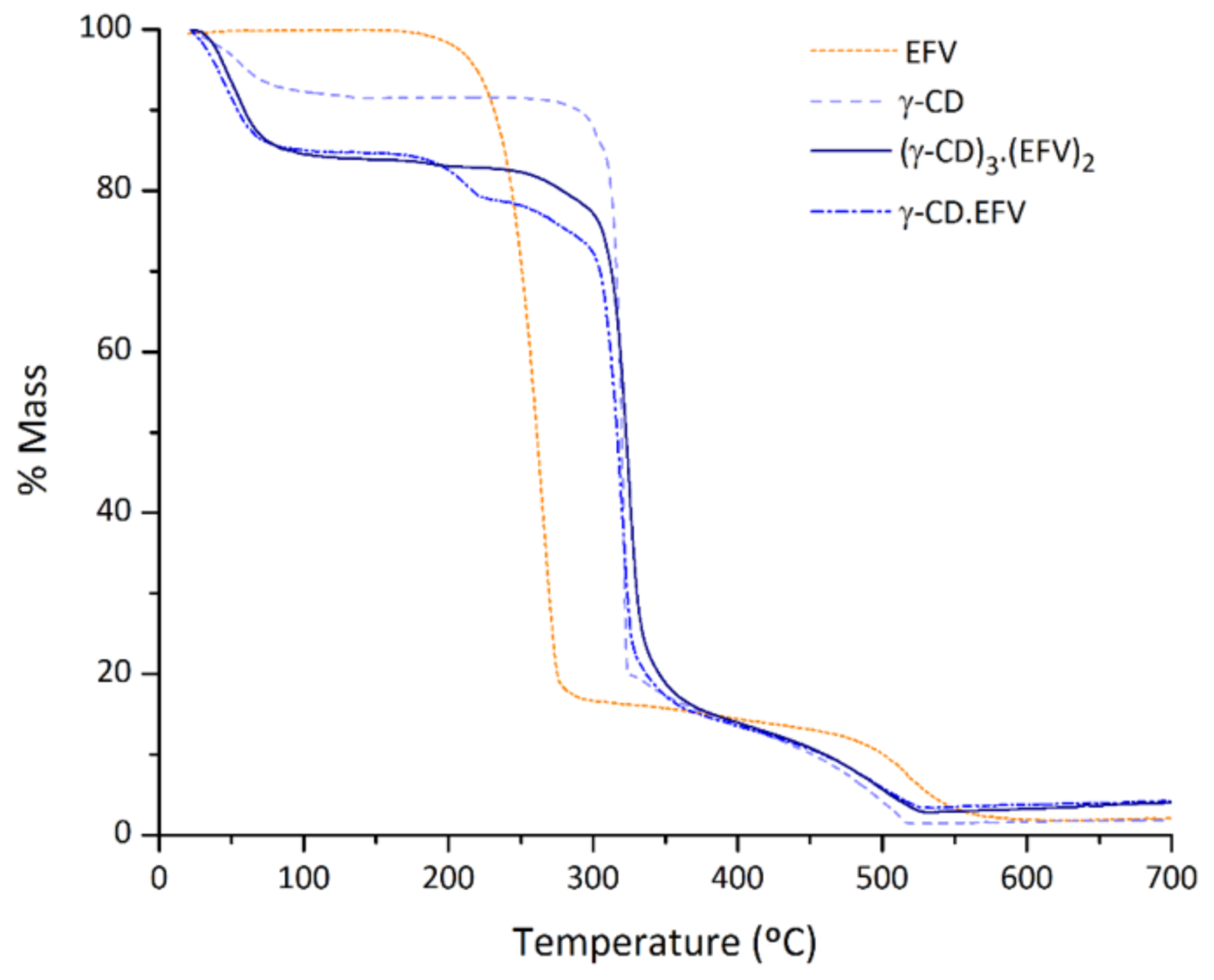
2.2.4. Thermogravimetric Analysis
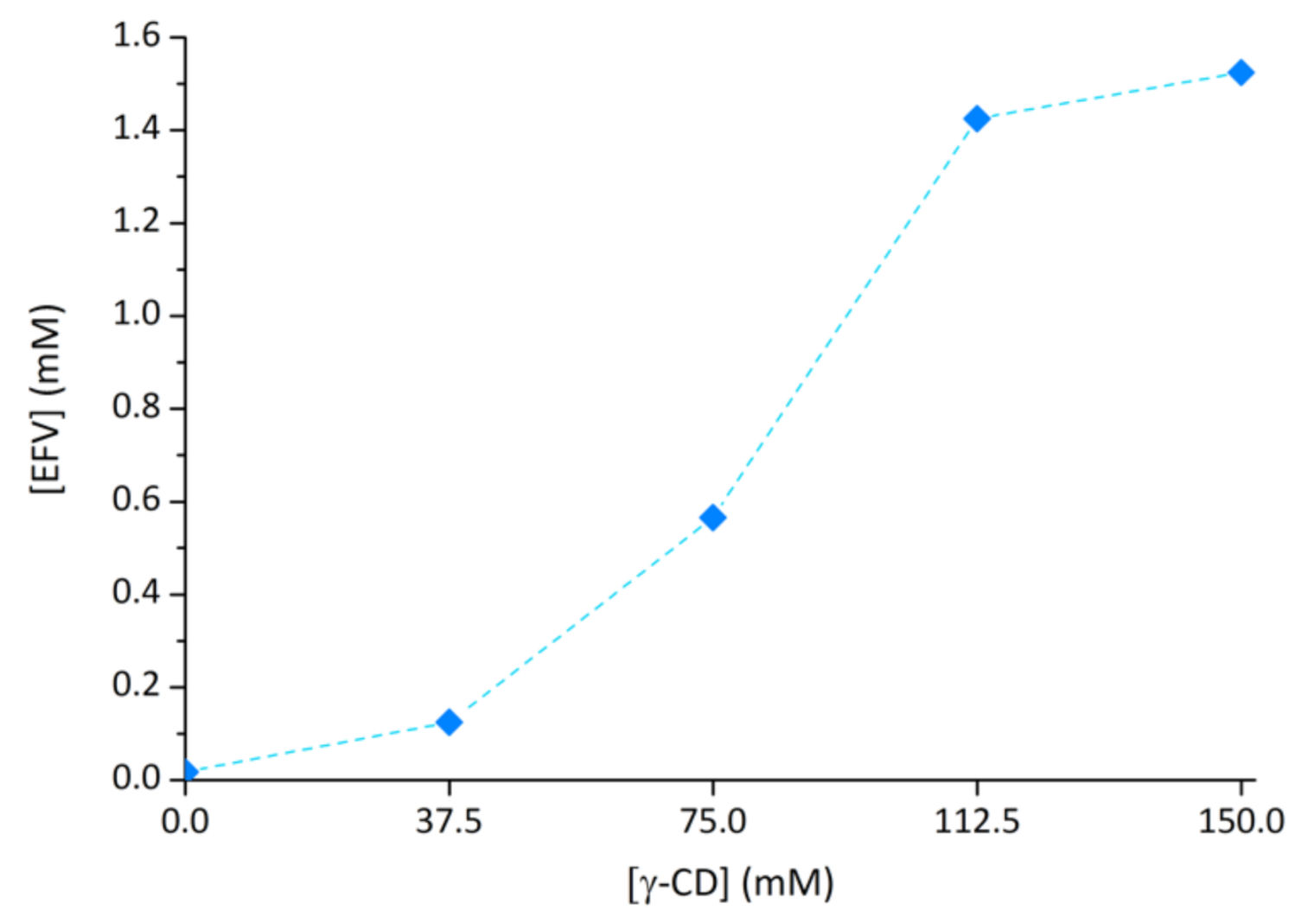
2.3. Effect of γ-CD on Efavirenz Solubility
3. Materials and Methods
3.1. Materials
3.2. Equipment
3.3. Continuous Variation Method
3.4. Preparation of the Inclusion Complexes as Solid Materials
3.4.1. γ-CD with EFV in the 3:2 Stoichiometry
3.4.2. γ-CD with EFV in the 1:1 Stoichiometry
3.5. Solubility Isotherms
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| API | Active pharmaceutical ingredient |
| CP-MAS | Cross-polarisation with magic angle spinning (a solid-state NMR method) |
| FT-IR | Fourier-transform infrared spectroscopy |
| γ-CD | Gamma-cyclodextrin |
| HPβCD | (2-hydroxy)propyl-beta-cyclodextrin |
| HPγCD | (2-hydroxy)propyl-gamma-cyclodextrin |
| EFV | Efavirenz |
| NMR | Nuclear magnetic resonance |
| ppm | Parts per million |
| PXRD | Powder X-ray diffraction |
| RAMEB | Randomly methylated beta-cyclodextrin |
| TGA | Thermogravimetric analysis |
| TMS | Tetramethylsilane |
References
- Best, B.M.; Goicoechea, M. Efavirenz—Still first line king? Expert Opin. Drug Metab. Toxicol. 2018, 4, 965–972. [Google Scholar] [CrossRef] [PubMed]
- Fandaruff, C.; Rauber, G.S.; Araya-Sibaja, A.M.; Pereira, R.N.; de Campos, C.E.M.; Rocha, H.V.A.; Monti, G.A.; Malaspina, T.; Silva, M.A.S.; Cuffini, S.L. Polymorphism of anti-HIV drug efavirenz: Investigations on thermodynamic and dissolution properties. Cryst. Growth Des. 2014, 14, 4968–4974. [Google Scholar] [CrossRef]
- Rajesh, Y.V.; Balasubramaniam, J.; Bindu, K.; Sridevi, R.; Swetha, M.; Rao, V.U. Impact of superdisintegrants on efavirenz release from tablet formulations. Acta Pharm. 2010, 60, 185–195. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Yang, J.; Grey, K.; Doney, J. An improved kinetics approach to describe the physical stability of amorphous solid dispersions. Int. J. Pharm. 2010, 384, 24–31. [Google Scholar] [CrossRef] [PubMed]
- Madhavi, B.B.; Kusum, B.; Charanya, C.K.; Madhu, M.N.; Harsa, V.S.; Banji, D. Dissolution enhancement of efavirenz by solid dispersion and PEGylation techniques. Int. J. Pharm. Investig. 2011, 1, 29–34. [Google Scholar] [CrossRef]
- Okafor, N.I.; Nkanga, C.I.; Walker, R.B.; Noundou, X.S.; Krause, R.W.M. Encapsulation and physicochemical evaluation of efavirenz in liposomes. J. Pharm. Investig. 2019, 50, 201–208. [Google Scholar] [CrossRef]
- Chiappetta, D.A.; Facorro, G.; de Celis, E.R.; Sosnik, A. Synergistic encapsulation of the anti-HIV agent efavirenz within mixed poloxamine/poloxamer polymeric micelles. Nanomed. Nanotech. Biol. Med. 2011, 7, 624–637. [Google Scholar] [CrossRef]
- Chiappetta, D.A.; Hocht, C.; Taira, C.; Sosnik, A. Efavirenz-loaded polymeric micelles for pediatric anti-HIV pharmacotherapy with significantly higher oral bioavailaibility. Nanomedicine 2010, 5, 11–23. [Google Scholar] [CrossRef]
- Varshosaz, J.; Taymouri, S.; Jahanian-Najafabadi, A.; Alizadeh, A. Efavirenz oral delivery via lipid nanocapsules: Formulation, optimisation, and ex-vivo gut permeation study. IET Nanobiotechnol. 2018, 12, 795–806. [Google Scholar] [CrossRef]
- Pokharkar, V.; Patil-Gadhe, A.; Palla, P. Efavirenz loaded nanostructured lipid carrier engineered for brain targeting through intranasal route: In-vivo pharmacokinetic and toxicity study. Biomed. Pharmacother. 2017, 94, 150–164. [Google Scholar] [CrossRef]
- Tshweu, L.; Katata, L.; Kalombo, L.; Chiappetta, D.A.; Hocht, C.; Sosnik, A.; Swai, H. Enhanced oral bioavailability of the antiretroviral efavirenz encapsulated in poly(epsilon-caprolactone) nanoparticles by a spray-drying method. Nanomedicine 2014, 9, 1821–1833. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.-H.; Lee, S.-E.; Pyo, Y.-C.; Tran, P.; Park, J.-S. Solubility enhancement and application of cyclodextrins in local drug delivery. J. Pharm. Investig. 2020, 50, 17–27. [Google Scholar] [CrossRef]
- Pereira, A.B.; Braga, S.S. Cyclodextrin Inclusion of Nutraceuticals, from the Bench to your Table. In Cyclodextrins: Synthesis, Chemical Applications and Role in Drug Delivery, 1st ed.; Ramirez, F.G., Ed.; NovaSience: Hauppage, NY, USA, 2015. [Google Scholar]
- Background Review for Cyclodextrins Used as Excipients; European Medicines Agency. 2014. Available online: http://www.ema.europa.eu/docs/en_GB/document_library/Report/2014/12/WC500177936.pdf (accessed on 9 December 2020).
- Veklury (Remdesivir) Prescribing Information. Available online: https://www.gilead.com/-/media/files/pdfs/medicines/covid-19/veklury/veklury_pi.pdf (accessed on 9 December 2020).
- Lyseng-Williamson, K.A. Midazolam oral solution (Ozalin®): A profile of its use for procedural sedation or premedication before anaesthesia in children. Drug Ther. Persp. 2019, 35, 255–262. [Google Scholar] [CrossRef]
- Phase 1 Study of Hydroxypropyl-Beta-Cyclodextrin(HP-beta-CyD)-Adjuvanted Influenza Split Vaccine. Available online: https://rctportal.niph.go.jp/en/detail?trial_id=UMIN000028530 (accessed on 2 August 2019).
- Braga, S.S. Cyclodextrins: Emerging medicines of the new millennium. Biomolecules 2019, 9, 801. [Google Scholar] [CrossRef]
- Sathigari, S.; Chadha, G.; Lee, Y.L.; Wright, N.; Parsons, D.L.; Rangari, V.K.; Fasina, R.; Babu, J. Physicochemical characterization of efavirenz cyclodextrin inclusion complexes. AAPS Pharm. Sci. Tech. 2009, 10, 81–87. [Google Scholar] [CrossRef]
- Braga, S.S.; Lysenko, K.; El-Saleh, F.; Paz, F.A.A. Cyclodextrin-efavirenz complexes investigated by solid state and solubility studies. Sciforum 2020. [Google Scholar] [CrossRef]
- Braga, S.S.; Aree, T.; Immamura, K.; Vertut, P.; Boal-Palheiros, I.; Sänger, W.; Teixeira-Dias, J.J.C. Structure of the β-cyclodextrin p-hydroxybenzaldehyde inclusion complex in aqueous solution and in the crystalline state. J. Incl. Phenom. Macrocycl. Chem. 2002, 43, 115–125. [Google Scholar] [CrossRef]
- Fernandes, J.A.; Paz, F.A.A.; Braga, S.S.; Ribeiro-Claro, P.; Rocha, J. Inclusion of potassium 4,40-biphenyldicarboxylate into b-cyclodextrin: The design and synthesis of an organic secondary building unit. New J. Chem. 2011, 35, 1280–1290. [Google Scholar] [CrossRef]
- Silva, R.N.; Costa, C.C.; Santos, M.J.G.; Alves, M.Q.; Braga, S.S.; Vieira, S.I.; Rocha, J.; Silva, A.M.S.; Guieu, S. Fluorescent light-up probe for the detection of protein aggregates. Chem. Asian J. 2019, 14, 859–863. [Google Scholar] [CrossRef]
- Kamitori, S.; Hirotsu, K.; Higuchi, T. Crystal and molecular structure of the γ-cyclodextrin–12-crown-4 1: 1 inclusion complex. J. Chem. Soc. Chem. Commun. 1986, 690–691. [Google Scholar] [CrossRef]
- Caira, M.R. On the isostructurality of cyclodextrin inclusion complexes and its practical utility. Rev. Roum. Chim. 2001, 46, 371–386. [Google Scholar]
- de Sousa, E.G.R.; de Carvalho, E.M.; Gil, R.A.S.S.; dos Santos, T.C.; Borré, L.B.; Santos-Filho, O.A.; Ellena, J. Solution and solid state nuclear magnetic resonance spectroscopic characterization of efavirenz. J. Pharm. Sci. 2016, 105, 2656–2664. [Google Scholar] [CrossRef] [PubMed]
- Heyes, S.J.; Clayden, N.J.; Dobson, C.M. 13C-CP/MAS NMR studies of the cyclomalto-oligosaccharide (cyclodextrin) hydrates. Carbohydr. Res. 1992, 233, 1–14. [Google Scholar] [CrossRef]
- Gidley, M.J.; Bociek, S.M. Carbon-13 CP/MAS NMR studies of amylose inclusion complexes, cyclodextrins, and the amorphous phase of starch granules: Relationships between glycosidic linkage conformation and solid-state carbon-13 chemical shifts. J. Am. Chem. Soc. 1988, 110, 3820–3829. [Google Scholar] [CrossRef]
- Pereira, A.B.; Silva, A.M.; Barroca, M.J.; Marques, M.P.M.; Braga, S.S. Physicochemical properties, antioxidant action and practical application in fresh cheese of the solid inclusion compound γ-cyclodextrin·quercetin, in comparison with β-cyclodextrin·quercetin. Arab. J. Chem. 2020, 13, 205–215. [Google Scholar] [CrossRef]
- Pais, J.M.; Barroca, M.J.; Marques, M.P.M.; Paz, F.A.A.; Braga, S.S. Solid-state studies and antioxidant properties of the γ-cyclodextrin·fisetin inclusion compound. Beilstein J. Org. Chem. 2017, 13, 2138–2145. [Google Scholar] [CrossRef] [PubMed]
- Catenacci, L.; Sorrenti, M.; Bonferoni, M.C.; Hunt, L.; Caira, M.R. Inclusion of the phytoalexin trans-resveratrol in native cyclodextrins: A thermal, spectroscopic, and X-ray structural study. Molecules 2020, 25, 998. [Google Scholar] [CrossRef]
- Kommavarapu, P.; Maruthapillai, A.; Palanisamy, K. Preparation and characterization of efavirenz nanosuspension with the application of enhanced solubility and dissolution rate. HIV AIDS Rev. 2016, 15, 170–176. [Google Scholar] [CrossRef]
- Cristofoletti, R.; Nair, A.; Abrahamsson, B.; Groot, D.W.; Kopp, S.; Langguth, P.; Polli, J.E.; Shah, V.P.; Dressman, J.B. Biowaiver monographs for immediate release solid oral dosage forms: Efavirenz. J. Pharm. Sci. 2013, 102, 318–329. [Google Scholar] [CrossRef]
- Job, P. Formation and stability of inorganic complexes in solution. Anal. Chim. Appl. 1928, 9, 113–203. [Google Scholar]
- Uekama, K.; Hirayama, F.; Irie, T. Cyclodextrin drug carrier systems. Chem. Rev. 1998, 98, 2045–2076. [Google Scholar] [CrossRef] [PubMed]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Braga, S.S.; El-Saleh, F.; Lysenko, K.; Paz, F.A.A. Inclusion Compound of Efavirenz and γ-Cyclodextrin: Solid State Studies and Effect on Solubility. Molecules 2021, 26, 519. https://doi.org/10.3390/molecules26030519
Braga SS, El-Saleh F, Lysenko K, Paz FAA. Inclusion Compound of Efavirenz and γ-Cyclodextrin: Solid State Studies and Effect on Solubility. Molecules. 2021; 26(3):519. https://doi.org/10.3390/molecules26030519
Chicago/Turabian StyleBraga, Susana Santos, Firas El-Saleh, Karyna Lysenko, and Filipe A. Almeida Paz. 2021. "Inclusion Compound of Efavirenz and γ-Cyclodextrin: Solid State Studies and Effect on Solubility" Molecules 26, no. 3: 519. https://doi.org/10.3390/molecules26030519
APA StyleBraga, S. S., El-Saleh, F., Lysenko, K., & Paz, F. A. A. (2021). Inclusion Compound of Efavirenz and γ-Cyclodextrin: Solid State Studies and Effect on Solubility. Molecules, 26(3), 519. https://doi.org/10.3390/molecules26030519







