A Metastable p-Type Semiconductor as a Defect-Tolerant Photoelectrode
Abstract
:1. Introduction
2. Results and Discussion
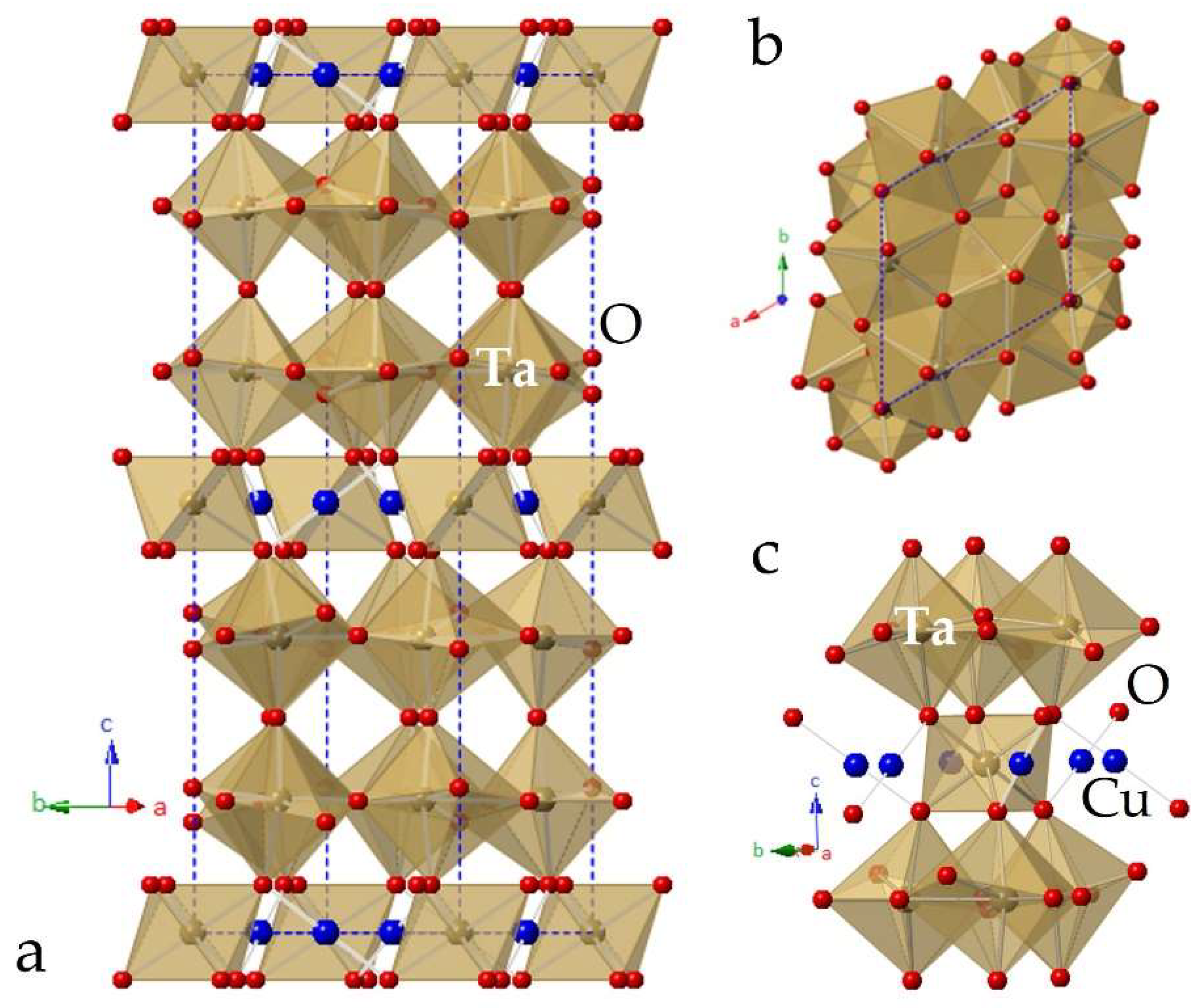
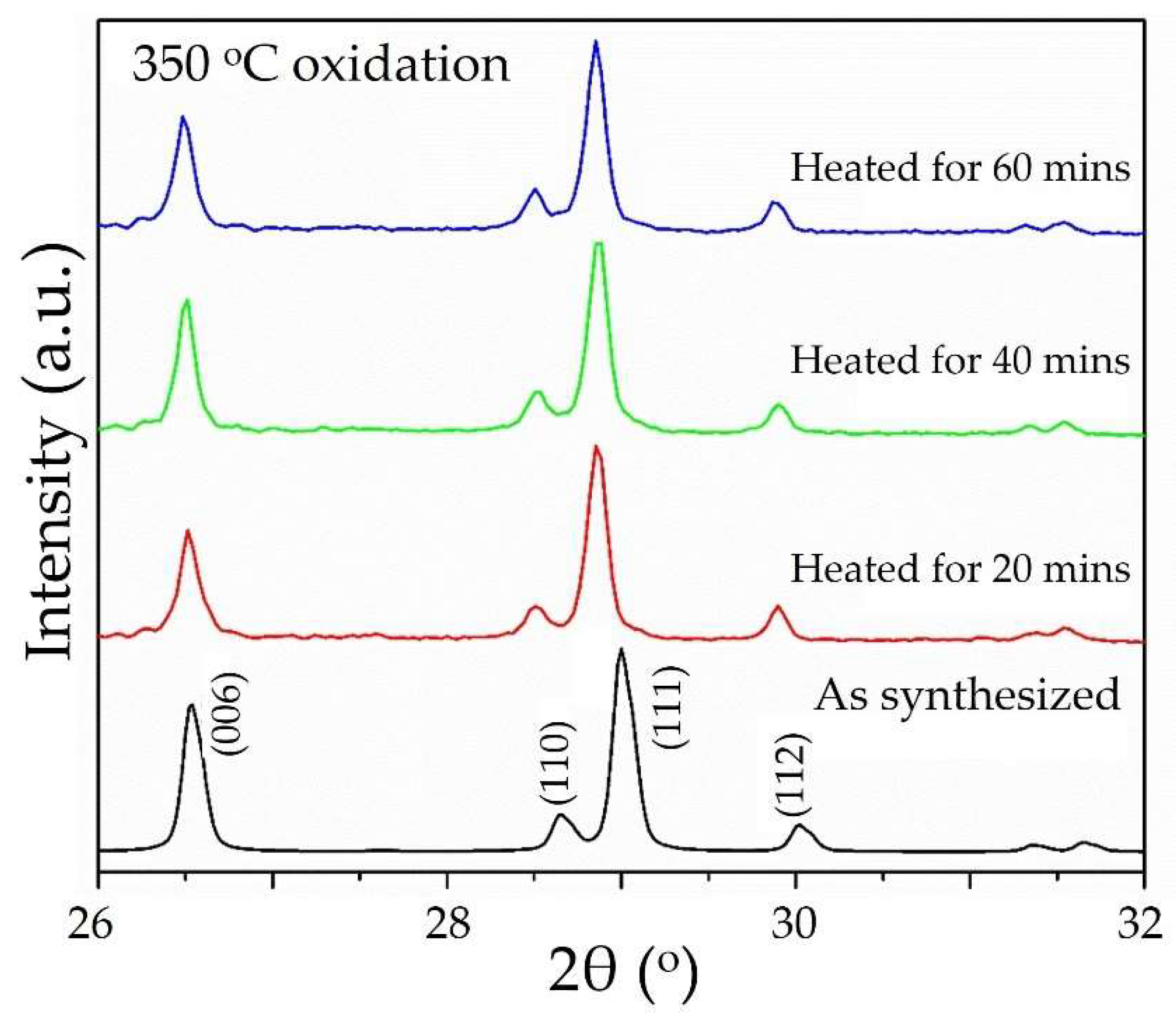
2.1. Bulk Phase Analysis and Thermal Stability
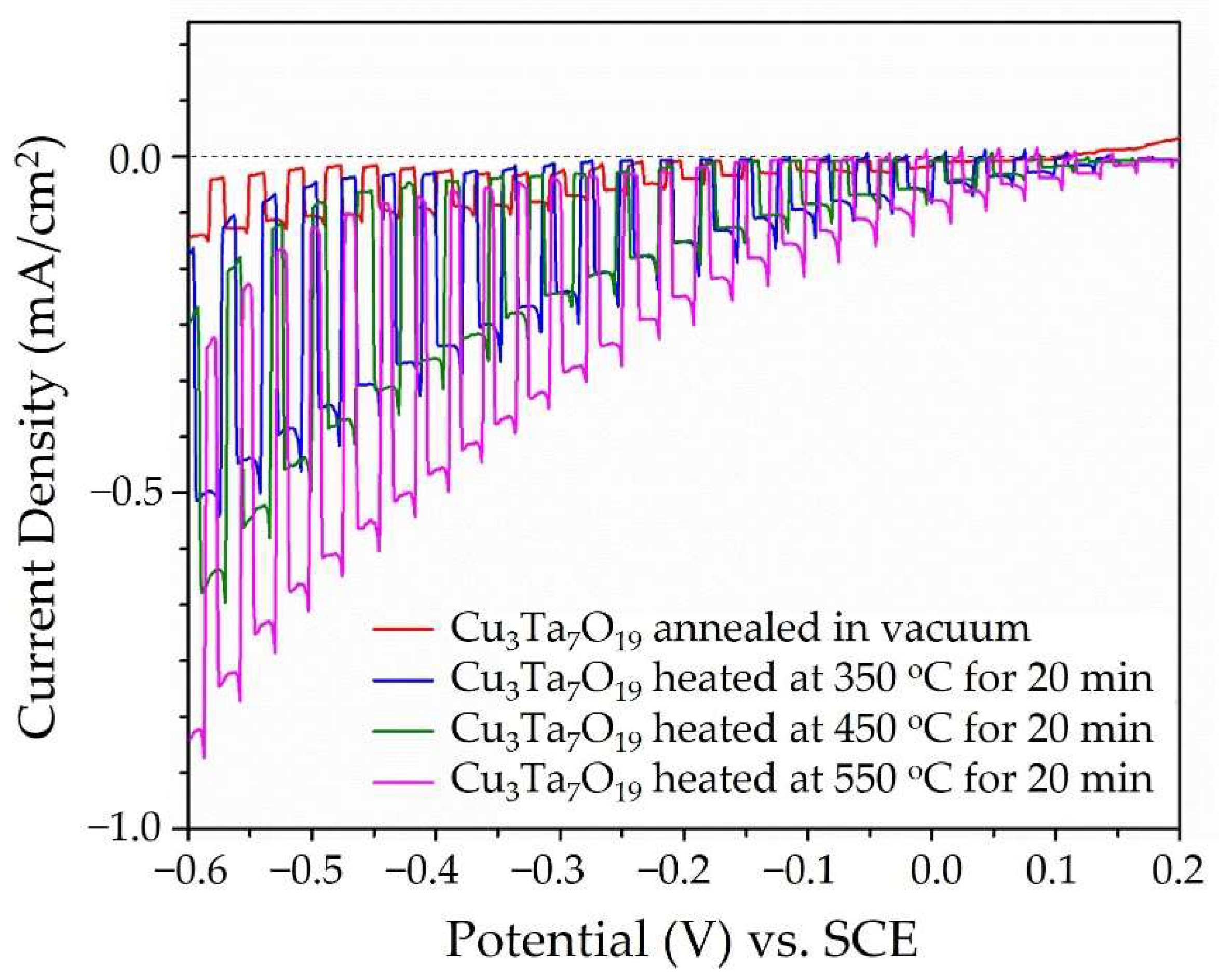
2.2. Optical and Photoelectrochemical Properties
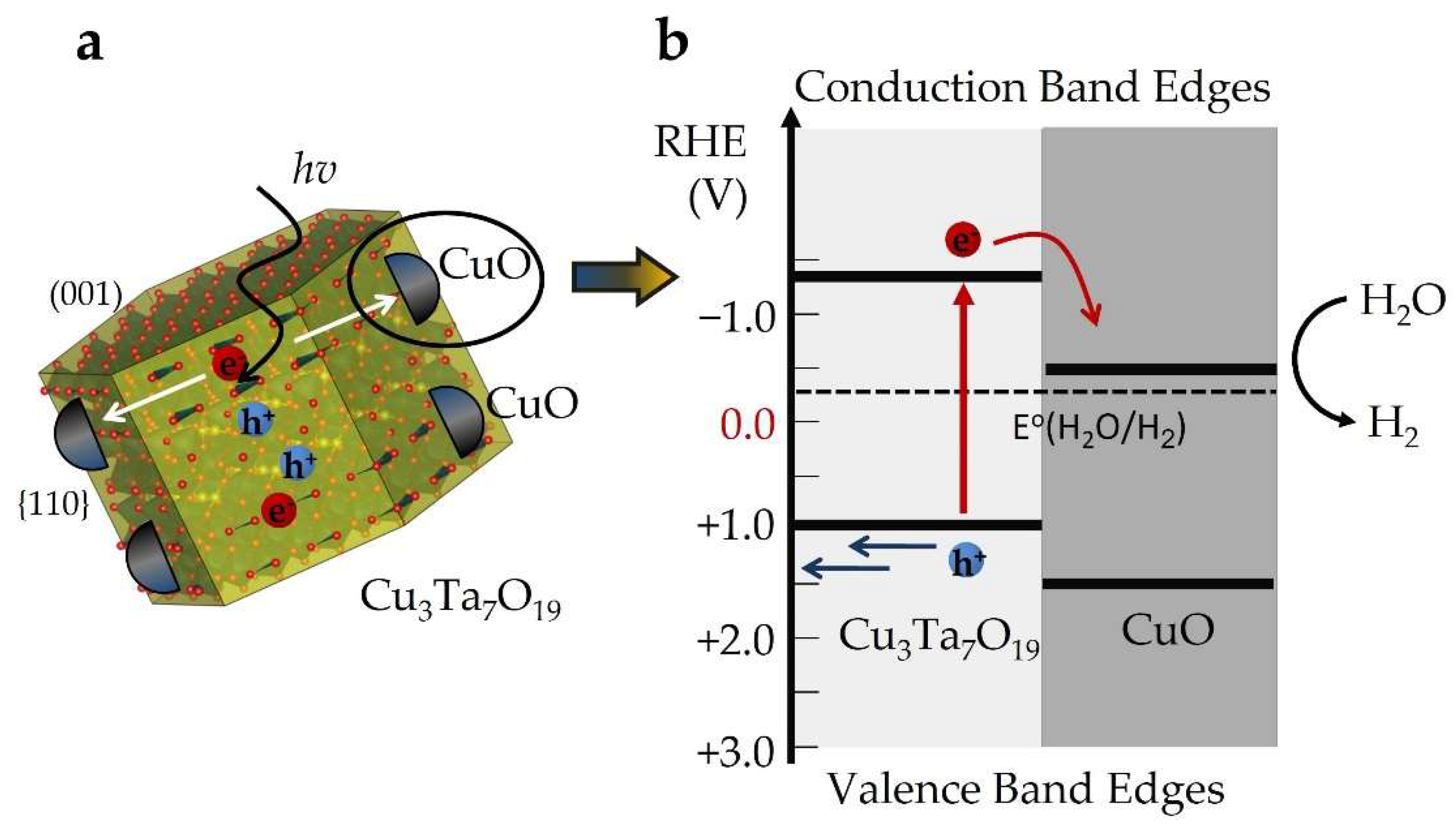
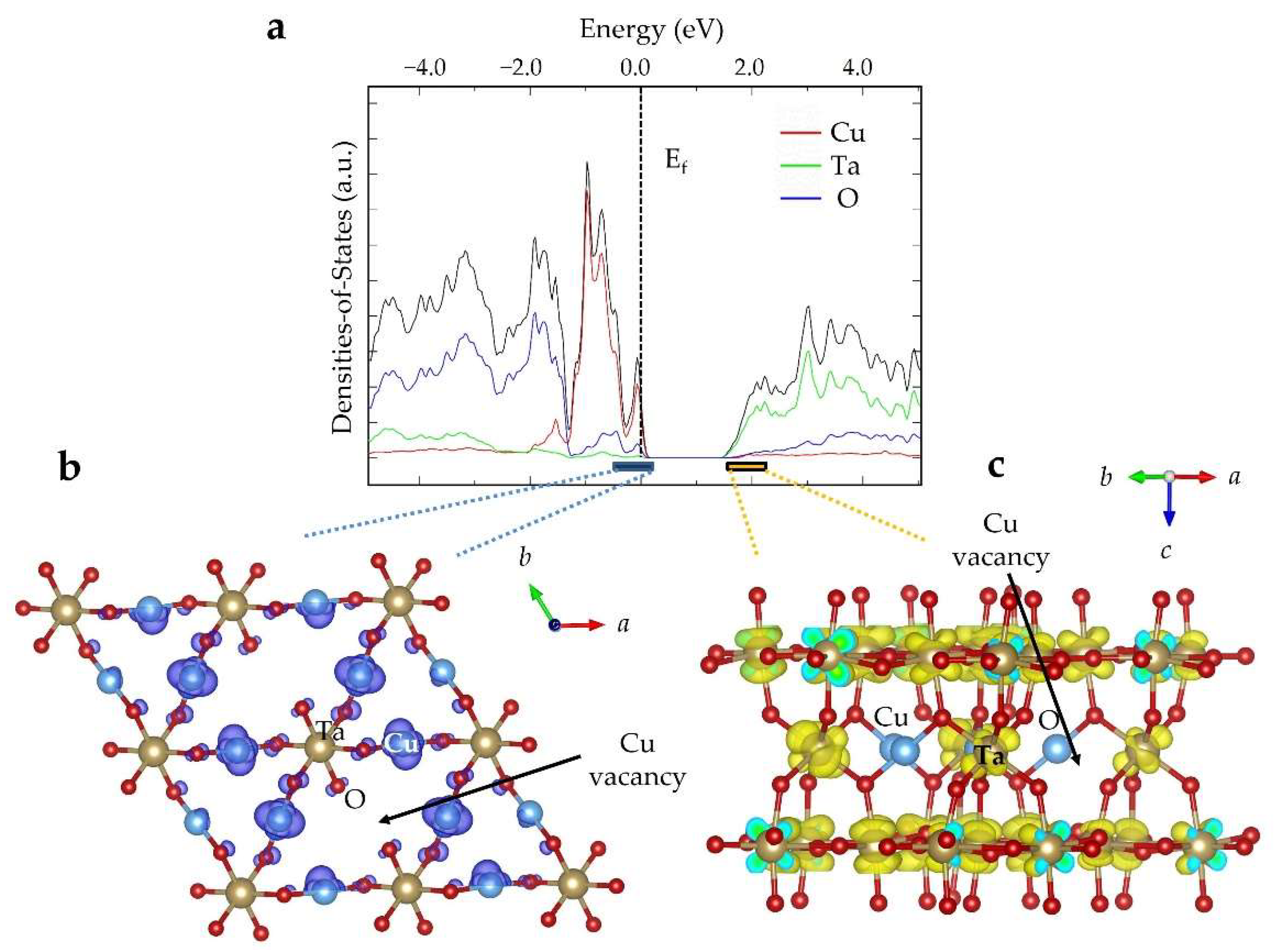
2.3. Electronic Structure and Defect Tolerance
3. Conclusions
4. Materials and Methods
4.1. Materials
4.2. Flux Synthesis
4.3. Characterization
4.4. Photoelectrochemical Measurements
4.5. Electronic Structure Calculations
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Sullivan, I.; Zoellner, B.; Maggard, P.A. Copper(I)-Based p-Type Oxides for Photoelectrochemical and Photovoltaic Solar Energy Conversion. Chem. Mater. 2016, 28, 5999–6016. [Google Scholar] [CrossRef]
- Rajeshwar, K.; Hossain, M.K.; Macaluso, R.T.; Janaky, C.; Varga, A.; Kulesza, P.J. Review-Copper Oxide-Based Ternary and Quaternary Oxides: Where Solid-State Chemistry Meets Photoelectrochemistry. J. Electrochem. Soc. 2018, 165, H3192–H3206. [Google Scholar] [CrossRef]
- Gu, J.; Yan, Y.; Krizan, J.W.; Gibson, Q.D.; Detweiler, Z.M.; Cava, R.J.; Bocarsly, A.B. P-type CuRhO2 as a Self-Healing Photoelectrode for Water Reduction under Visible Light. J. Am. Chem. Soc. 2014, 136, 830–833. [Google Scholar] [CrossRef]
- Galante, M.T.; Zivkovic, A.; Alvim, J.C.; Kleiner, C.C.C.; Sangali, M.; Taylor, S.F.R.; Greer, A.J.; Hardacre, C.; Rajeshwar, K.; Caram, R.; et al. Arc Synthesis, Crystal Structure, and Photoelectrochemistry of Copper(I) Tungstate. ACS Appl. Mater. Interfaces 2021, 13, 32865–32875. [Google Scholar] [CrossRef]
- Joshi, U.; Palasyuk, A.; Maggard, P.A. Photoelectrochemical Investigation and Electronic Structure of a p-Type CuNbO3 Photocathode. J. Phys. Chem. C 2011, 115, 13534–13539. [Google Scholar] [CrossRef]
- Sahoo, P.P.; Zoellner, B.; Maggard, P.A. Optical, Electronic, and Photoelectrochemical Properties of the p-type Cu3−xVO4 Semiconductor. J. Mater. Chem. 2015, 3, 4501–4509. [Google Scholar] [CrossRef]
- Paracchino, A.; Laporte, V.; Sivula, K.; Gratzel, M.; Thimsen, E. Highly Active Oxide Photocathode for Photoelectrochemical Water Reduction. Nat. Mater. 2011, 10, 456–461. [Google Scholar] [CrossRef]
- Morales-Guio, C.G.; Tilley, S.D.; Vrubel, H.; Gratzel, M.; Hu, X. Hydrogen Evolution from a Copper(I) Oxide Photocathode Coated with an Amorphous Molybdenum Sulphide Catalyst. Nat. Comm. 2014, 5, 4059. [Google Scholar] [CrossRef] [PubMed]
- Ida, S.; Yamada, K.; Matsunaga, T.; Hagiwara, H.; Matsumoto, Y.; Ishihara, T. Preparation of p-type CaFe2O4 Photocathodes for Producing Hydrogen from Water. J. Am. Chem. Soc. 2010, 132, 17343–17345. [Google Scholar] [CrossRef] [PubMed]
- Sekizawa, K.; Nonaka, T.; Arai, T.; Morikawa, T. Structural Improvement of CaFe2O4 by Metal Doping Toward Enhanced Photocathodic Current. ACS Appl. Mater. Interfac. 2014, 6, 10969–10973. [Google Scholar] [CrossRef] [PubMed]
- McLamb, N.; Sahoo, P.P.; Fuoco, L.; Maggard, P.A. Flux Growth of Single-Crystal Na2Ta4O11 Particles and their Photocatalytic Hydrogen Production. Cryst. Grow. Des. 2013, 13, 2322–2326. [Google Scholar] [CrossRef]
- Boltersdorf, J.; Maggard, P.A. Silver Exchange of Layered Metal Oxides and Their Photocatalytic Activities. ACS Catal. 2013, 3, 2547–2555. [Google Scholar] [CrossRef]
- Arney, D.; Maggard, P.A. Effect of Platelet-Shaped Surfaces and Silver-Cation Exchange on the Photocatalytic Hydrogen Production of RbLaNb2O7. ACS Catal. 2012, 2, 1711–1717. [Google Scholar] [CrossRef]
- Maggard, P.A. Capturing Metastable Oxide Semiconductors for Applications in Solar Energy Conversion. Acc. Chem. Res. 2021, 54, 3160–3171. [Google Scholar] [CrossRef]
- Choi, J.; King, N.; Maggard, P.A. Metastable Cu(I)-Niobate Semiconductor with a Low-Temperature Nanoparticle-Mediated Synthesis. ACS Nano. 2013, 7, 1699–1708. [Google Scholar] [CrossRef] [PubMed]
- King, N.; Sahoo, P.; Fuoco, L.; Stuart, S.; Dougherty, D.; Liu, Y.; Maggard, P.A. Cu-Deficiency in the p-Type Semiconductor Cu1-xNb3O8. Chem. Mater. 2014, 26, 2095–2104. [Google Scholar] [CrossRef]
- Sullivan, I.; Sahoo, P.P.; Fuoco, L.; Hewitt, A.S.; Stuart, S.; Dougherty, D.; Maggard, P.A. Cu-Deficiency in the p-Type Semiconductor Cu5−xTa11O30: Impact on its Crystalline Structure, Surfaces, and Photoelectrochemical Properties. Chem. Mater. 2014, 26, 6711–6721. [Google Scholar] [CrossRef]
- King, N.; Sommer, R.D.; Watkins-Curry, P.; Chan, J.Y.; Maggard, P.A. Synthesis, Structure, and Thermal Instability of the Cu2Ta4O11 Phase. Cryst. Growth Des. 2015, 15, 552–558. [Google Scholar] [CrossRef]
- Jain, A.; Ong, S.P.; Hautier, G.; Chen, W.; Richards, W.D.; Dacek, S.; Cholia, S.; Gunter, D.; Skinner, D.; Ceder, G.; et al. The Materials Project: A Materials Genome Approach to Accelerating Materials Innovation. APL Mater. 2013, 1, 011002. [Google Scholar] [CrossRef] [Green Version]
- Skorupska, K.; Maggard, P.A.; Eichberger, R.; Schwarzburg, K.; Shahbazi, P.; Zoellner, B.; Parksinson, B.A. Combinatorial Investigations of High Temperature CuNb Oxide Phases for Photoelectrochemical Water Splitting. ACS Comb. Sci. 2015, 17, 742–751. [Google Scholar] [CrossRef]
- Zhang, Z.; Lindley, S.A.; Dhall, R.; Bustillo, K.; Han, W.; Xie, E.; Cooper, J.K. Beneficial CuO Phase Segregation in the Ternary p-Type Oxide Photocathode CuBi2O4. ACS Appl. Energy Mater. 2019, 2, 4111–4117. [Google Scholar] [CrossRef]
- Brandt, R.E.; Stevanovic, V.; Ginley, D.S.; Buonassisi, T. Identifying Defect-Tolerant Semiconductors with High Minority-Carrier Lifetimes: Beyond Hybrid Lead Halide Perovskites. MRS Commun. 2015, 5, 265–275. [Google Scholar] [CrossRef] [Green Version]
- Palasyuk, O.; Palasyuk, A.; Maggard, P.A. Synthesis, Optical Properties and Electronic Structures of Copper(I) Tantalates: Cu5Ta11O30 and Cu3Ta7O19. J. Solid St. Chem. 2010, 183, 814–822. [Google Scholar] [CrossRef]
- King, N.; Boltersdorf, J.; Maggard, P.A.; Wong-Ng, W. Polymorphism and Structural Distortions of Mixed-Metal Oxide Photocatalysts Constructed with α-U3O8 Types of Layers. Crystals 2017, 7, 145. [Google Scholar] [CrossRef] [Green Version]
- Palasyuk, O.; Palasyuk, A.; Maggard, P.A. Site-Differentiated Solid Solution in (Na1-xCux)2Ta4O11 and its Electronic Structure and Optical Properties. Inorg. Chem. 2010, 49, 10571–10578. [Google Scholar] [CrossRef]
- Koffyberg, F.P.; Benko, F.A. A Photoelectrochemical Determination of the Conduction and Valence Band Edges of p-Type CuO. J. Appl. Phys. 1982, 53, 1173–1177. [Google Scholar] [CrossRef]
- Fuoco, L.; Joshi, U.A.; Maggard, P.A. Preparation and Photoelectrochemical Properties of p-Type Cu5Ta11O30 and Cu3Ta7O19 Semiconducting Polycrystalline Films. J. Phys. Chem. C 2012, 116, 10490–10497. [Google Scholar] [CrossRef]
- Jade 9 (Version 9.0); Materials Data Incorporated: Livermore, CA, USA, 2018.
- Dusek, M.; Petricek, V.; Wunschel, M.; Dinnebier, R.E.; van Smaalen, S. Refinement of Modulated Structures Against X-ray Powder Diffraction Data with JANA2000. J. Appl. Crystallogr. 2001, 34, 398–404. [Google Scholar] [CrossRef]
- Kresse, G.; Furthmuller, J. Efficiency of Ab-Initio Total Energy Calculations for Metals and Semiconductors Using a Plane-Wave Basis Set. Comput. Mater. Sci. 1996, 6, 15–50. [Google Scholar] [CrossRef]
- Payne, M.C.; Arias, T.A.; Joannopoulos, J.D. Iterative Minimization Techniques for Ab Initio Total-Energy Calculations: Molecular Dynamics and Conjugate Gradients. Rev. Mod. Phys. 1992, 64, 1045–1097. [Google Scholar] [CrossRef] [Green Version]
- Monkhorst, H.J.; Pack, J.D. Special Points for Brillouin-Zone Integrations. Phys. Rev. B 1976, 13, 5188–5192. [Google Scholar] [CrossRef]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sohag, Z.; O’Donnell, S.; Fuoco, L.; Maggard, P.A. A Metastable p-Type Semiconductor as a Defect-Tolerant Photoelectrode. Molecules 2021, 26, 6830. https://doi.org/10.3390/molecules26226830
Sohag Z, O’Donnell S, Fuoco L, Maggard PA. A Metastable p-Type Semiconductor as a Defect-Tolerant Photoelectrode. Molecules. 2021; 26(22):6830. https://doi.org/10.3390/molecules26226830
Chicago/Turabian StyleSohag, Zahirul, Shaun O’Donnell, Lindsay Fuoco, and Paul A. Maggard. 2021. "A Metastable p-Type Semiconductor as a Defect-Tolerant Photoelectrode" Molecules 26, no. 22: 6830. https://doi.org/10.3390/molecules26226830
APA StyleSohag, Z., O’Donnell, S., Fuoco, L., & Maggard, P. A. (2021). A Metastable p-Type Semiconductor as a Defect-Tolerant Photoelectrode. Molecules, 26(22), 6830. https://doi.org/10.3390/molecules26226830






