New Generation of Meso and Antiprogestins (SPRMs) into the Osteoporosis Approach
Abstract
:1. Introduction
1.1. Molecular and Functional Characterization of RANK and RANKL
1.2. The Role of RANK and RANKL in Bone Homeostasis
1.3. Effect of RANK/RANKL Inhibition in Osteoclastogenesis
2. Prevention of Osteoporosis—Women’s Health
2.1. Hormone-Dependent Diseases with Bone Density Loss
2.2. The Popular Drugs Effective to Osteoporosis Prevention
2.2.1. Denosumab (Prolia, Xgeva)
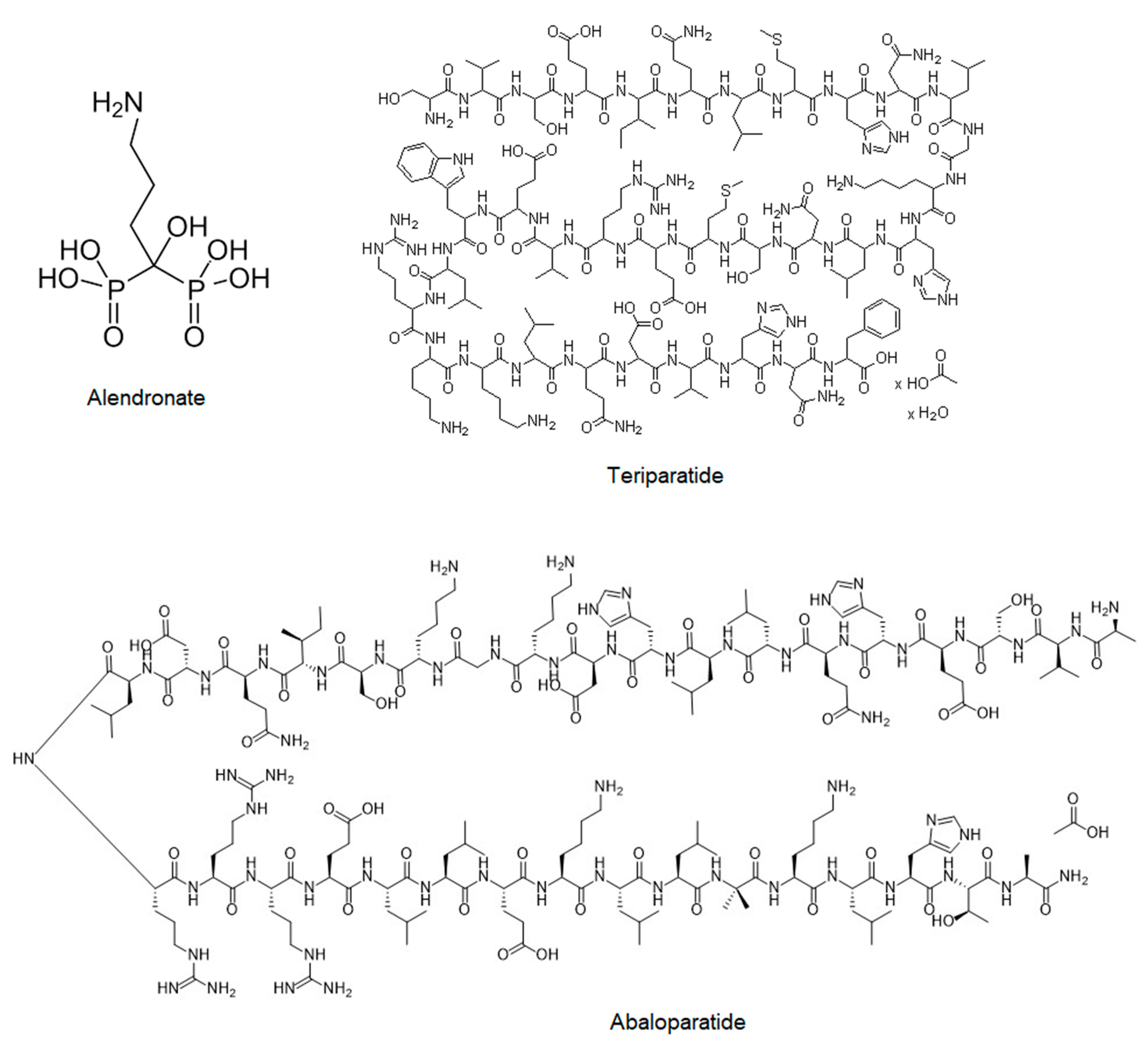
2.2.2. Bisphosphonates (Fosamax, Bonviva, Aclasta, and Zometa)
2.2.3. Teriparatide (Forteo)
2.2.4. Abaloparatide (Tymlos)
3. New Trends in RANK/RANKL Signaling Inhibition—The Role of Meso and Antiprogestins—New SPRMs
4. Bone Homeostasis Evaluation—In Vivo and In Vitro Tests for Studying Osteoclastogenesis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Idriss, H.T.; Naismith, J.H. TNFα and the TNF receptor superfamily: Structure-function relationship(s). Microsc. Res. Tech. 2000, 50, 184–195. [Google Scholar] [CrossRef]
- Beth Erlichman, O.M. Zack Howard, CD27 signals through PKC in human B cell lymphomas. Cytokine 1999, 11, 476–484. [Google Scholar] [CrossRef]
- Theill, L.E.; Boyle, W.J.; Penninger, J.M. RANK-L and RANK: T Cells, Bone Loss, and Mammalian Evolution. Annu. Rev. Immunol. 2002, 20, 795–823. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ai, C.; Ma, G.; Deng, Y.; Zheng, Q.; Gen, Y.; Li, W.; Li, Y.; Zu, L.; Zhou, Q. Nm23-H1 inhibits lung cancer bone-specific metastasis by upregulating miR-660-5p targeted SMARCA5. Thorac. Cancer 2020, 11, 640–650. [Google Scholar] [CrossRef] [Green Version]
- Whang, P.; Schwarz, E.; Gamradt, S.; Dougall, W.; Lieberman, J. The effects of RANK blockade and osteoclast depletion in a model of pure osteoblastic prostate cancer metastasis in bone. J. Orthop. Res. 2005, 23, 1475–1483. [Google Scholar] [CrossRef] [PubMed]
- Sordillo, E.M.; Pearse, R.N. RANK-Fc: A therapeutic antagonist for RANK-L in myeloma. Cancer 2003, 97, 802–812. [Google Scholar] [CrossRef]
- Ono, T.; Hayashi, M.; Sasaki, F.; Nakashima, T. RANKL biology: Bone metabolism, the immune system, and beyond. Inflamm. Regen. 2020, 40, 1–16. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sigl, V.; Penninger, J.M. RANKL/RANK—From bone physiology to breast cancer. Cytokine Growth Factor Rev. 2014, 25, 205–214. [Google Scholar] [CrossRef] [PubMed]
- Boyce, B.F.; Yao, Z.; Xing, L. Osteoclasts Have Multiple Roles in Bone in Addition to Bone Resorption. Crit. Rev. Eukaryot. Gene Expr. 2009, 19, 171–180. [Google Scholar] [CrossRef] [PubMed]
- Son, H.S.; Lee, J.; Lee, H.I.; Kim, N.; Jo, Y.-J.; Lee, G.-R.; Hong, S.-E.; Kwon, M.; Kim, N.Y.; Kim, H.J.; et al. Benzydamine inhibits osteoclast differentiation and bone resorption via down-regulation of interleukin-1 expression. Acta Pharm. Sin. B 2020, 10, 462–474. [Google Scholar] [CrossRef] [PubMed]
- Donnaloja, F.; Jacchetti, E.; Soncini, M.; Raimondi, M.T. Natural and Synthetic Polymers for Bone Scaffolds Optimization. Polymers 2020, 12, 905. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kylmaoja, E.; Nakamura, M.; Tuukkanen, J. Osteoclasts and Remodeling Based Bone Formation. Curr. Stem Cell Res. Ther. 2016, 11, 626–633. [Google Scholar] [CrossRef]
- Lo, C.H.; Baratchart, E.; Basanta, D.; Lynch, C.C. Computational modeling reveals a key role for polarized myeloid cells in controlling osteoclast activity during bone injury repair. Sci. Rep. 2021, 11, 6055. [Google Scholar] [CrossRef] [PubMed]
- Etich, J.; Rehberg, M.; Eckes, B.; Sengle, G.; Semler, O.; Zaucke, F. Signaling pathways affected by mutations causing osteogenesis imperfecta. Cell. Signal. 2020, 76, 109789. [Google Scholar] [CrossRef]
- Lee, S.Y.; Park, K.H.; Yu, H.-G.; Kook, E.; Song, W.-H.; Lee, G.; Koh, J.-T.; Shin, H.-I.; Choi, J.-Y.; Huh, Y.H.; et al. Controlling hypoxia-inducible factor-2α is critical for maintaining bone homeostasis in mice. Bone Res. 2019, 7, 14. [Google Scholar] [CrossRef] [PubMed]
- Boyce, B.F.; Xing, L. Functions of RANKL/RANK/OPG in bone modeling and remodeling. Arch. Biochem. Biophys. 2008, 473, 139–146. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jorde, R.; Stunes, A.K.; Kubiak, J.; Grimnes, G.; Thorsby, P.M.; Syversen, U. Smoking and other determinants of bone turnover. PLoS ONE 2019, 14, e0225539. [Google Scholar] [CrossRef] [PubMed]
- Hofbauer, L.C. Estrogen Stimulates Gene Expression and Protein Production of Osteoprotegerin in Human Osteoblastic Cells. Endocrinology 1999, 140, 4367–4370. [Google Scholar] [CrossRef] [PubMed]
- Lochlin, R.M.; Khosla, S.; Turner, R.T.; Riggs, B.L. Mediators of the bisphasic responses of bone to intermittent and continuously administered parathyroid hormone. J. Cell Biochem. 2003, 89, 180–190. [Google Scholar] [CrossRef] [PubMed]
- Gunsser, J.; Hermann, R.; Roth, A.; Lupp, A. Comprehensive assessment of tissue and serum parameters of bone metabolism in a series of orthopaedic patients. PLoS ONE 2019, 14, e0227133. [Google Scholar] [CrossRef]
- Ricarte, F.R.; Le Henaff, C.; Kolupaeva, V.G.; Gardella, T.J.; Partridge, N.C. Parathyroid hormone(1–34) and its analogs differentially modulate osteoblastic Rankl expression via PKA/SIK2/SIK3 and PP1/PP2A–CRTC3 signaling. J. Biol. Chem. 2018, 293, 20200–20213. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hadji, P.; Colli, E.; Regidor, P.-A. Bone health in estrogen-free contraception. Osteoporos. Int. 2019, 30, 2391–2400. [Google Scholar] [CrossRef] [PubMed]
- Zuo, H.-L.; Xin, H.; Yan, X.-N.; Huang, J.; Zhang, Y.-P.; Du, H. 17β-Estradiol improves osteoblastic cell function through the Sirt1/NF-κB/MMP-8 pathway. Climacteric 2020, 23, 404–409. [Google Scholar] [CrossRef] [PubMed]
- Manokawinchoke, J.; Ritprajak, P.; Osathanon, T.; Pavasant, P. Estradiol induces osteoprotegerin expression by human dental pulp cells. Odontology 2014, 104, 10–18. [Google Scholar] [CrossRef] [PubMed]
- Hofbauer, L.C.; Kühne, C.A.; Viereck, V. The OPG/RANKL/RANK system in metabolic bone diseases. J. Musculoskelet. Neuronal Interact. 2004, 4, 268–275. [Google Scholar] [PubMed]
- Wang, L.; Shi, X.; Zhao, R.; Halloran, B.P.; Clark, D.J.; Jacobs, C.R.; Kingery, W.S. Calcitonin-gene-related peptide stimulates stromal cell osteogenic differentiation and inhibits RANKL induced NF-κB activation, osteoclastogenesis and bone resorption. Bone 2010, 46, 1369–1379. [Google Scholar] [CrossRef] [Green Version]
- Huebner, A.K.; Schinke, T.; Priemel, M.; Schilling, S.; Schilling, A.F.; Emeson, R.B.; Rueger, J.M.; Amling, M. Calcitonin Deficiency in Mice Progressively Results in High Bone Turnover. J. Bone Miner. Res. 2006, 21, 1924–1934. [Google Scholar] [CrossRef] [PubMed]
- Zhong, Y.; Harvey, J. Armbrecht, Sylvia Christakos, Calcitonin, a Regulator of the 25-Hydroxyvitamin D3 1α-Hydroxylase Gene. J. Biol. Chem. 2009, 284, 11059–11069. [Google Scholar] [CrossRef] [Green Version]
- Akopian, A.; Demulder, A.; Ouriaghli, F.; Corazza, F.; Fondu, P.; Bergmann, P. Effects of CGRP on human osteoclast-like cell formation: A possible connection with the bone loss in neurological disorders? Peptides 2000, 21, 559–564. [Google Scholar] [CrossRef]
- Millet, I.; Phillips, R.J.; Sherwin, R.S.; Ghosh, S.; Voll, R.E.; Flavell, R.A.; Vignery, A.; Rincón, M. Inhibition of NF-κB Activity and Enhancement of Apoptosis by the Neuropeptide Calcitonin Gene-related Peptide. J. Biol. Chem. 2000, 275, 15114–15121. [Google Scholar] [CrossRef] [Green Version]
- Miles, D.T.; Voskuil, R.T.; Dale, W.; Mayerson, J.L.; Scharschmidt, T.J. Integration of denosumab therapy in the management of giant cell tumors of bone. J. Orthop. 2020, 22, 38–47. [Google Scholar] [CrossRef] [PubMed]
- Miller, R.E.; Branstetter, D.; Armstrong, A.P.; Kennedy, B.; Jones, J.; Cowan, L.; Bussiere, J.L.; Dougall, W.C. Receptor Activator of NF-κB Ligand Inhibition Suppresses Bone Resorption and Hypercalcemia but Does Not Affect Host Immune Responses to Influenza Infection. J. Immunol. 2007, 179, 266–274. [Google Scholar] [CrossRef] [Green Version]
- Akiyama, T.; Choong, P.; Dass, C.R. RANK-Fc inhibits malignancy via inhibiting ERK activation and evoking caspase-3-mediated anoikis in human osteosarcoma cells. Clin. Exp. Metastasis 2010, 27, 207–215. [Google Scholar] [CrossRef] [PubMed]
- Lamoureux, F.; Richard, P.; Wittrant, Y.; Battaglia, S.; Pilet, P.; Trichet, V.; Blanchard, F.; Gouin, F.; Pitard, B.; Heymann, D.; et al. Therapeutic Relevance of Osteoprotegerin Gene Therapy in Osteosarcoma: Blockade of the Vicious Cycle between Tumor Cell Proliferation and Bone Resorption. Cancer Res. 2007, 67, 7308–7318. [Google Scholar] [CrossRef] [Green Version]
- Castellano, D.; Sepulveda, J.M.; García-Escobar, I.; Rodriguez-Antolín, A.; Sundlöv, A.; Cortes-Funes, H. The Role of RANK-Ligand Inhibition in Cancer: The Story of Denosumab. Oncologist 2011, 16, 136–145. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McClung, M.R.; Lewiecki, E.M.; Cohen, S.B.; Bolognese, M.A.; Woodson, G.C.; Moffett, A.H.; Peacock, M.; Miller, P.D.; Lederman, S.N.; Chesnut, C.H.; et al. Denosumab in Postmenopausal Women with Low Bone Mineral Density. N. Engl. J. Med. 2006, 354, 821–831. [Google Scholar] [CrossRef] [PubMed]
- McClung, M. Role of RANKL inhibition in osteoporosis. Arthritis Res. Ther. 2007, 9 (Suppl. 1), S3. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- An, H.; Zhao, J.; Wang, J.; Li, C.; Jiang, Z.; Yao, J.; Zhang, X.; Wu, J. Comparison of efficacy and safety of Chinese patent medicine in the treatment of postmenopausal osteoporosis. Medicine 2020, 99, e22734. [Google Scholar] [CrossRef] [PubMed]
- Bone, H.G.; Wagman, R.B.; Brandi, M.L.; Brown, J.P.; Chapurlat, R.; Cummings, S.R.; Czerwiński, E.; Fahrleitner-Pammer, A.; Kendler, D.L.; Lippuner, K.; et al. 10 years of denosumab treatment in postmenopausal women with osteoporosis: Results from the phase 3 randomised FREEDOM trial and open-label extension. Lancet Diabetes Endocrinol. 2017, 5, 513–523. [Google Scholar] [CrossRef]
- Emerton, K.; Hu, B.; Woo, A.; Sinofsky, A.; Hernandez, C.; Majeska, R.; Jepsen, K.; Schaffler, M. Osteocyte apoptosis and control of bone resorption following ovariectomy in mice. Bone 2010, 46, 577–583. [Google Scholar] [CrossRef] [Green Version]
- Ganova, P.; Zhivkova, R.; Kolarov, A.; Ivanovska, N. Influence of estradiol treatment on bone marrow cell differentiation in collagenase-induced arthritis. Inflamm. Res. 2020, 69, 533–543. [Google Scholar] [CrossRef]
- Guder, C.; Gravius, S.; Burger, C.; Wirtz, D.C.; Schildberg, F.A. Osteoimmunology: A Current Update of the Interplay Between Bone and the Immune System. Front. Immunol. 2020, 11, 58. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hayashi, M.; Nakashima, T.; Yoshimura, N.; Okamoto, K.; Tanaka, S.; Takayanagi, H. Autoregulation of Osteocyte Sema3A Orchestrates Estrogen Action and Counteracts Bone Aging. Cell Metab. 2019, 29, 627–637. [Google Scholar] [CrossRef] [Green Version]
- Nagy, V.; Penninger, J. The RANKL-RANK Story. Gerontology 2015, 61, 534–542. [Google Scholar] [CrossRef] [PubMed]
- Engdahl, C.; Bondt, A.; Harre, U.; Raufer, J.; Pfeifle, R.; Camponeschi, A.; Wuhrer, M.; Seeling, M.; Mårtensson, I.-L.; Nimmerjahn, F.; et al. Estrogen induces St6gal1 expression and increases IgG sialylation in mice and patients with rheumatoid arthritis: A potential explanation for the increased risk of rheumatoid arthritis in postmenopausal women. Arthritis Res. 2018, 20, 84. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Charatcharoenwitthaya, N.; Khosla, S.; Atkinson, E.J.; McCready, L.K.; Riggs, B.L. Effect of Blockade of TNF-α and Interleukin-1 Action on Bone Resorption in Early Postmenopausal Women. J. Bone Miner. Res. 2007, 22, 724–729. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Qian, W.-P.; Dark, K.; Toraldo, G.; Lin, A.S.P.; Guldberg, R.E.; Flavell, R.A.; Weitzmann, M.N.; Pacifici, R. Estrogen prevents bone loss through transforming growth factor signaling in T cells. Proc. Natl. Acad. Sci. USA 2004, 101, 16618–16623. [Google Scholar] [CrossRef] [Green Version]
- Wakkach, A.; Mansour, A.; Dacquin, R.; Coste, E.; Jurdic, P.; Carle, G.F.; Blin-Wakkach, C. Bone marrow microenvironment controls the in vivo differentiation of murine dendritic cells into osteoclasts. Blood 2008, 112, 5074–5083. [Google Scholar] [CrossRef] [PubMed]
- Madel, M.-B.; Ibáñez, L.; Wakkach, A.; De Vries, T.J.; Teti, A.; Apparailly, F.; Blin-Wakkach, C. Immune Function and Diversity of Osteoclasts in Normal and Pathological Conditions. Front. Immunol. 2019, 10, 1408. [Google Scholar] [CrossRef] [PubMed]
- Kiesel, L.; Kohl, A. Role of the RANK/RANKL pathway in breast cancer. Maturitas 2016, 86, 10–16. [Google Scholar] [CrossRef]
- Simatou, A.; Sarantis, P.; Koustas, E.; Papavassiliou, A.G.; Karamouzis, M.V. The Role of the RANKL/RANK Axis in the Prevention and Treatment of Breast Cancer with Immune Checkpoint Inhibitors and Anti-RANKL. Int. J. Mol. Sci. 2020, 21, 7570. [Google Scholar] [CrossRef]
- Muhammad, A.; Mada, S.B.; Malami, I.; Forcados, G.E.; Erukainure, O.; Sani, H.; Abubakar, I.B. Postmenopausal osteoporosis and breast cancer: The biochemical links and beneficial effects of functional foods. Biomed. Pharmacother. 2018, 107, 571–582. [Google Scholar] [CrossRef] [PubMed]
- Tan, W.; Zhang, W.; Strasner, A.; Grivennikov, S.; Cheng, J.Q.; Hoffman, R.M.; Karin, M. Tumour-infiltrating regulatory T cells stimulate mammary cancer metastasis through RANKL–RANK signalling. Nature 2011, 470, 548–553. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Grimm, S.L.; Hartig, S.M.; Edwards, D.P. Progesterone Receptor Signaling Mechanisms. J. Mol. Biol. 2016, 428, 3831–3849. [Google Scholar] [CrossRef] [PubMed]
- Tchakarska, G.; Sola, B. The double dealing of cyclin D1. Cell Cycle 2020, 19, 163–178. [Google Scholar] [CrossRef] [Green Version]
- Hardy, D.B.; Janowski, B.A.; Corey, D.R.; Mendelson, C. Progesterone Receptor Plays a Major Antiinflammatory Role in Human Myometrial Cells by Antagonism of Nuclear Factor-κB Activation of Cyclooxygenase 2 Expression. Mol. Endocrinol. 2006, 20, 2724–2733. [Google Scholar] [CrossRef]
- Feng, J.; Liu, S.; Ma, S.; Zhao, J.; Zhang, W.; Qi, W.; Cao, P.; Wang, Z.; Lei, W. Protective effects of resveratrol on postmenopausal osteoporosis: Regulation of SIRT1-NF-κB signaling pathway. Acta Biochim. et Biophys. Sin. 2014, 46, 1024–1033. [Google Scholar] [CrossRef] [Green Version]
- Hu, H.; Wang, J.; Gupta, A.; Shidfar, A.; Branstetter, D.; Lee, O.; Ivancic, D.; Sullivan, M.; Chatterton, R.T.; Dougall, W.C.; et al. RANKL expression in normal and malignant breast tissue responds to progesterone and is up-regulated during the luteal phase. Breast Cancer Res. Treat. 2014, 146, 515–523. [Google Scholar] [CrossRef]
- Olver, I.N. Prevention of breast cancer. Med. J. Aust. 2016, 205, 475–479. [Google Scholar] [CrossRef]
- Roepman, P.; Horlings, H.M.; Krijgsman, O.; Kok, M.; Bueno-De-Mesquita, J.M.; Bender, R.; Linn, S.C.; Glas, A.; Van De Vijver, M.J. Microarray-Based Determination of Estrogen Receptor, Progesterone Receptor, and HER2 Receptor Status in Breast Cancer. Clin. Cancer Res. 2009, 15, 7003–7011. [Google Scholar] [CrossRef] [Green Version]
- Kuran, D.; Pogorzelska, A.; Wiktorska, K. Breast Cancer Prevention-Is there a Future for Sulforaphane and Its Analogs? Nutrients 2020, 12, 1559. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, S.; Azad, K.A. Study of Receptor Status in Carcinoma Breast Patient. Chattagram Maa-O-Shishu Hosp. Med Coll. J. 2018, 16, 48–50. [Google Scholar] [CrossRef]
- Brodowicz, T.; Hemetsberger, M.; Windhager, R. Denosumab for the treatment of giant cell tumor of the bone. Futur. Oncol. 2015, 11, 1881–1894. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gibiansky, L.; Sutjandra, L.; Doshi, S.; Zheng, J.; Sohn, W.; Jang, G.R.; Chow, A.T.; Pérez-Ruixo, J.J.; Peterson, M.C.; Pérez-Ruixo, J.J. Population Pharmacokinetic Analysis of Denosumab in Patients with Bone Metastases from Solid Tumours. Clin. Pharmacokinet. 2012, 51, 247–260. [Google Scholar] [CrossRef] [PubMed]
- Sutjandra, L.; Rodriguez, R.D.; Doshi, S.; Ma, M.; Jang, G.R.; Chow, A.T.; Pérez-Ruixo, J.J.; Peterson, M.C.; Pérez-Ruixo, J.J. Population Pharmacokinetic Meta-Analysis of Denosumab in Healthy Subjects and Postmenopausal Women with Osteopenia or Osteoporosis. Clin. Pharmacokinet. 2011, 50, 793–807. [Google Scholar] [CrossRef]
- Smith, M.R.; Egerdie, B.; Toriz, N.H.; Feldman, R.; Tammela, T.L.; Saad, F.; Heracek, J.; Szwedowski, M.; Ke, C.; Kupic, A.; et al. Denosumab in Men Receiving Androgen-Deprivation Therapy for Prostate Cancer. N. Engl. J. Med. 2009, 361, 745–755. [Google Scholar] [CrossRef] [Green Version]
- Fizazi, K.; Lipton, A.; Mariette, X.; Body, J.-J.; Rahim, Y.; Gralow, J.R.; Gao, G.; Wu, L.; Sohn, W.; Jun, S. Randomized Phase II Trial of Denosumab in Patients with Bone Metastases From Prostate Cancer, Breast Cancer, or Other Neoplasms After Intravenous Bisphosphonates. J. Clin. Oncol. 2009, 27, 1564–1571. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Atkins, G.J.; Haynes, D.R.; Graves, S.E.; Evdokiou, A.; Hay, S.; Bouralexis, S.; Findlay, D.M. Expression of Osteoclast Differentiation Signals by Stromal Elements of Giant Cell Tumors. J. Bone Miner. Res. 2010, 15, 640–649. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Aleza, C.; Nguyen, B.; Yoldi, G.; Ciscar, M.; Barranco, A.; Hernández-Jiménez, E.; Maetens, M.; Salgado, R.; Zafeiroglou, M.; Pellegrini, P.; et al. Inhibition of RANK signaling in breast cancer induces an anti-tumor immune response orchestrated by CD8+ T cells. Nat. Commun. 2020, 11, 6335. [Google Scholar] [CrossRef] [PubMed]
- Ming, J.; Cronin, S.J.F.; Penninger, J.M. Targeting the RANKL/RANK/OPG Axis for Cancer Therapy. Front. Oncol. 2020, 10, 1283. [Google Scholar] [CrossRef]
- Widschwendter, M.; Rosenthal, A.N.; Philpott, S.; Rizzuto, I.; Fraser, L.; Hayward, J.; Intermaggio, M.P.; Edlund, C.K.; Ramus, S.J.; Gayther, S.A.; et al. The sex hormone system in carriers of BRCA1/2 mutations: A case-control study. Lancet Oncol. 2013, 14, 1226–1232. [Google Scholar] [CrossRef]
- Bains, J.S.; Carter, E.M.; Citron, K.P.; Boskey, A.L.; Shapiro, J.R.; Steiner, R.D.; Smith, P.A.; Bober, M.B.; Hart, T.; Cuthbertson, D.; et al. A Multicenter Observational Cohort Study to Evaluate the Effects of Bisphosphonate Exposure on Bone Mineral Density and Other Health Outcomes in Osteogenesis Imperfecta. JBMR Plus 2019, 3, e10118. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Baron, R.; Ferrari, S.; Russell, R.G.G. Denosumab and bisphosphonates: Different mechanisms of action and effects. Bone 2011, 48, 677–692. [Google Scholar] [CrossRef] [PubMed]
- Ashrafi, M.; Ghalichi, F.; Mirzakouchaki, B.; Doblare, M. On the effect of antiresorptive drugs on the bone remodeling of the mandible after dental implantation: A mathematical model. Sci. Rep. 2021, 11, 2792. [Google Scholar] [CrossRef]
- Roelofs, A.J.; Coxon, F.P.; Ebetino, F.H.; Lundy, M.W.; Henneman, Z.J.; Nancollas, G.H.; Sun, S.; Blazewska, K.; Bala, J.L.F.; Kashemirov, B.A.; et al. Fluorescent risedronate analogues reveal bisphosphonate uptake by bone marrow monocytes and localization around osteocytes in vivo. J. Bone Miner. Res. 2010, 25, 606–616. [Google Scholar] [CrossRef] [PubMed]
- Russell, R.G.G.; Watts, N.B.; Ebetino, F.H.; Rogers, M. Mechanisms of action of bisphosphonates: Similarities and differences and their potential influence on clinical efficacy. Osteoporos. Int. 2008, 19, 733–759. [Google Scholar] [CrossRef] [PubMed]
- Fisher, J.E.; Rogers, M.; Halasy, J.M.; Luckman, S.P.; Hughes, D.E.; Masarachia, P.J.; Wesolowski, G.; Russell, R.G.G.; Rodan, G.A.; Reszka, A.A. Alendronate mechanism of action: Geranylgeraniol, an intermediate in the mevalonate pathway, prevents inhibition of osteoclast formation, bone resorption, and kinase activation in vitro. Proc. Natl. Acad. Sci. USA 1999, 96, 133–138. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dwan, K.; Phillipi, C.A.; Steiner, R.D. Bisphosphonate therapy for osteogenesis imperfecta. Cochrane Database Syst. Rev. 2014, 23, CD005088. [Google Scholar] [CrossRef] [Green Version]
- Reid, I.R.I.; Brown, J.; Burckhardt, P.; Horowitz, Z.Z.; Richardson, P.P.; Trechsel, U.; Widmer, A.A.; Devogelaer, J.-P.; Kaufman, J.-M.; Jaeger, P.P.; et al. Intravenous Zoledronic Acid in Postmenopausal Women with Low Bone Mineral Density. N. Engl. J. Med. 2002, 346, 653–661. [Google Scholar] [CrossRef] [PubMed]
- Kanis, J.A.; Burlet, N.; Cooper, C.; Delmas, P.D.; Reginster, J.-Y.; Borgstrom, F.; Rizzoli, R.; (Esceo). European guidance for the diagnosis and management of osteoporosis in postmenopausal women. Osteoporos. Int. 2008, 19, 1103–1104. [Google Scholar] [CrossRef] [Green Version]
- Perazella, M.A.; Markowitz, G.S. Bisphosphonate nephrotoxicity. Kidney Int. 2008, 74, 1385–1393. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mohsenzade, P.; Amirhakimi, A.; Honar, N.; Saki, F.; Omrani, G.H.R.; Dabbaghmanesh, M. Bone density, fractures and the associated factors in iranian children and adolescent with Osteogenesis Imperfecta. BMC Pediatr. 2021, 21, 37. [Google Scholar] [CrossRef]
- Lindahl, K.; Kindmark, A.; Rubin, C.-J.; Malmgren, B.; Grigelioniene, G.; Söderhäll, S.; Ljunggren, Ö.; Åström, E. Decreased fracture rate, pharmacogenetics and BMD response in 79 Swedish children with osteogenesis imperfecta types I, III and IV treated with Pamidronate. Bone 2016, 87, 11–18. [Google Scholar] [CrossRef] [PubMed]
- Almazrooa, S.A.; Woo, S.B. Bisphosphonate and nonbisphosphonate-associated osteonecrosis of the jaw: A review. J. Am. Dent. Assoc. 2009, 140, 864–875. [Google Scholar] [CrossRef]
- Censi, S.; Iacobone, M.; Simmini, S.; Manso, J.; Franceschet, G.; Plebani, M.; Frigo, A.C.; Zaninotto, M.; Torresan, F.; De Silvestro, G.; et al. PTH: Redefining Reference Ranges in a Healthy Population—The Role of Interfering Factors and the Type of Laboratory Assay. Int. J. Endocrinol. 2020, 2020, 1053719. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gorvin, C.M. Calcium-sensing receptor signaling—How human disease informs biology. Curr. Opin. Endocr. Metab. Res. 2021, 16, 10–18. [Google Scholar] [CrossRef]
- Jiang, Y.; Zhao, J.J.; Mitlak, B.H.; Wang, O.; Genant, H.K.; Eriksen, E.F. Recombinant Human Parathyroid Hormone (1-34) [Teriparatide] Improves Both Cortical and Cancellous Bone Structure. J. Bone Miner. Res. 2003, 18, 1932–1941. [Google Scholar] [CrossRef]
- Neer, R.M.; Arnaud, C.D.; Zanchetta, J.R.; Prince, R.; Gaich, G.A.; Reginster, J.-Y.; Hodsman, A.B.; Eriksen, E.F.; Ish-Shalom, S.; Genant, H.K.; et al. Effect of Parathyroid Hormone (1-34) on Fractures and Bone Mineral Density in Postmenopausal Women with Osteoporosis. N. Engl. J. Med. 2001, 344, 1434–1441. [Google Scholar] [CrossRef] [PubMed]
- Hodsman, A.B.; Bauer, D.C.; Dempster, D.W.; Dian, L.; Hanley, D.A.; Harris, S.T.; Kendler, D.; McClung, M.; Miller, P.D.; Olszynski, W.P.; et al. Parathyroid Hormone and Teriparatide for the Treatment of Osteoporosis: A Review of the Evidence and Suggested Guidelines for Its Use. Endocr. Rev. 2005, 26, 688–703. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Paschalis, E.P.; Dempster, D.W.; Gamsjaeger, S.; Rokidi, S.; Hassler, N.; Brozek, W.; Chan-Diehl, F.W.; Klaushofer, K.; Taylor, K.A. Mineral and organic matrix composition at bone forming surfaces in postmenopausal women with osteoporosis treated with either teriparatide or zoledronic acid. Bone 2021, 145, 115848. [Google Scholar] [CrossRef] [PubMed]
- Sato, M.; Zeng, G.Q.; Turner, C.H. Biosynthetic Human Parathyroid Hormone (1–34) Effects on Bone Quality in Aged Ovariectomized Rats. Endocrinology 1997, 138, 4330–4337. [Google Scholar] [CrossRef] [PubMed]
- Jerome, C.; Burr, D.; Van Bibber, T.; Hock, J.; Brommage, R. Treatment with human parathyroid hormone (1-34) for 18 months increases cancellous bone volume and improves trabecular architecture in ovariectomized cynomolgus monkeys (Macaca fascicularis). Bone 2001, 28, 150–159. [Google Scholar] [CrossRef]
- Dempster, D.W.; Roschger, P.; Misof, B.M.; Zhou, H.; Paschalis, E.P.; Alam, J.; Ruff, V.A.; Klaushofer, K.; Taylor, K.A. Differential Effects of Teriparatide and Zoledronic Acid on Bone Mineralization Density Distribution at 6 and 24 Months in the SHOTZ Study. J. Bone Miner. Res. 2016, 31, 1527–1535. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, D.; Zhou, Q.; Labroska, V.; Qin, S.; Darbalaei, S.; Wu, Y.; Yuliantie, E.; Xie, L.; Tao, H.; Cheng, J.; et al. G protein-coupled receptors: Structure- and function-based drug discovery. Signal Transduct. Target. Ther. 2021, 6, 7. [Google Scholar] [CrossRef] [PubMed]
- Leder, B.Z.; O’Dea, L.S.; Zanchetta, J.R.; Kumar, P.; Banks, K.; McKay, K.; Lyttle, C.R.; Hattersley, G. Effects of Abaloparatide, a Human Parathyroid Hormone-Related Peptide Analog, on Bone Mineral Density in Postmenopausal Women with Osteoporosis. J. Clin. Endocrinol. Metab. 2015, 100, 697–706. [Google Scholar] [CrossRef]
- Makino, A.; Hasegawa, T.; Takagi, H.; Takahashi, Y.; Hase, N.; Amizuka, N. Frequent administration of abaloparatide shows greater gains in bone anabolic window and bone mineral density in mice: A comparison with teriparatide. Bone 2021, 142, 115651. [Google Scholar] [CrossRef]
- Arlt, H.; Mullarkey, T.; Hu, D.; Baron, R.; Ominsky, M.S.; Mitlak, B.; Lanske, B.; Besschetnova, T. Effects of abaloparatide and teriparatide on bone resorption and bone formation in female mice. Bone Rep. 2020, 13, 100291. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, S.; Matsuda, M.; Hou, Z.; Bailey, J.P.; Kitazawa, R.; Herbst, M.P.; Horseman, N.D. Receptor Activator of NF-κB Ligand Induction via Jak2 and Stat5a in Mammary Epithelial Cells. J. Biol. Chem. 2003, 278, 46171–46178. [Google Scholar] [CrossRef] [Green Version]
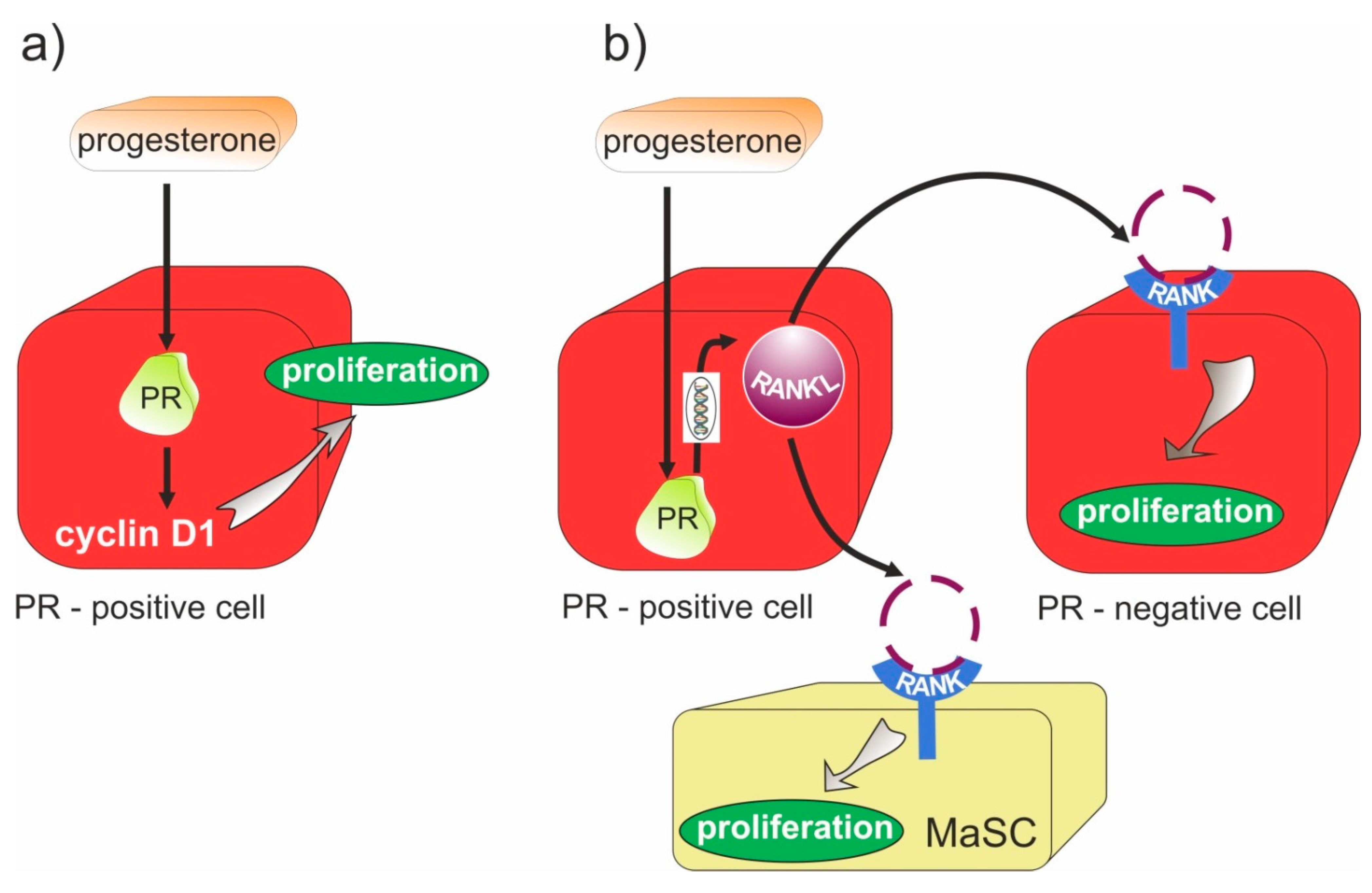
- Mukherjee, A.; Soyal, S.M.; Li, J.; Ying, Y.; He, B.; Demayo, F.J.; Lydon, J.P. Targeting RANKL to a specific subset of murine mammary epithelial cells induces ordered branching morphogenesis and alveologenesis in the absence of progesterone receptor expression. FASEB J. 2010, 24, 4408–4419. [Google Scholar] [CrossRef] [Green Version]
- Tsubaki, M.; Komai, M.; Fujimoto, S.-I.; Itoh, T.; Imano, M.; Sakamoto, K.; Shimaoka, H.; Takeda, T.; Ogawa, N.; Mashimo, K.; et al. Activation of NF-κB by the RANKL/RANK system up-regulates snail and twist expressions and induces epithelial-to-mesenchymal transition in mammary tumor cell lines. J. Exp. Clin. Cancer Res. 2013, 32, 62. [Google Scholar] [CrossRef] [Green Version]
- Widschwendter, M.; Burnell, M.; Fraser, L.; Rosenthal, A.N.; Philpott, S.; Reisel, D.; Dubeau, L.; Cline, M.; Pan, Y.; Yi, P.-C.; et al. Osteoprotegerin (OPG), The Endogenous Inhibitor of Receptor Activator of NF-κB Ligand (RANKL), is Dysregulated in BRCA Mutation Carriers. EBioMedicine 2015, 2, 1331–1339. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gonzalez-Suarez, E.; Jacob, A.P.; Jones, J.; Miller, R.; Roudier-Meyer, M.P.; Erwert, R.; Pinkas, J.; Branstetter, D.G.; Dougall, W.C. RANK ligand mediates progestin-induced mammary epithelial proliferation and carcinogenesis. Nat. Cell Biol. 2010, 468, 103–107. [Google Scholar] [CrossRef] [PubMed]
- Schramek, D.; Leibbrandt, A.; Sigl, V.; Kenner, L.; Pospisilik, J.A.; Lee, H.J.; Hanada, R.; Joshi, P.A.; Aliprantis, A.; Glimcher, L.; et al. Osteoclast differentiation factor RANKL controls development of progestin-driven mammary cancer. Nature 2010, 468, 98–102. [Google Scholar] [CrossRef] [Green Version]
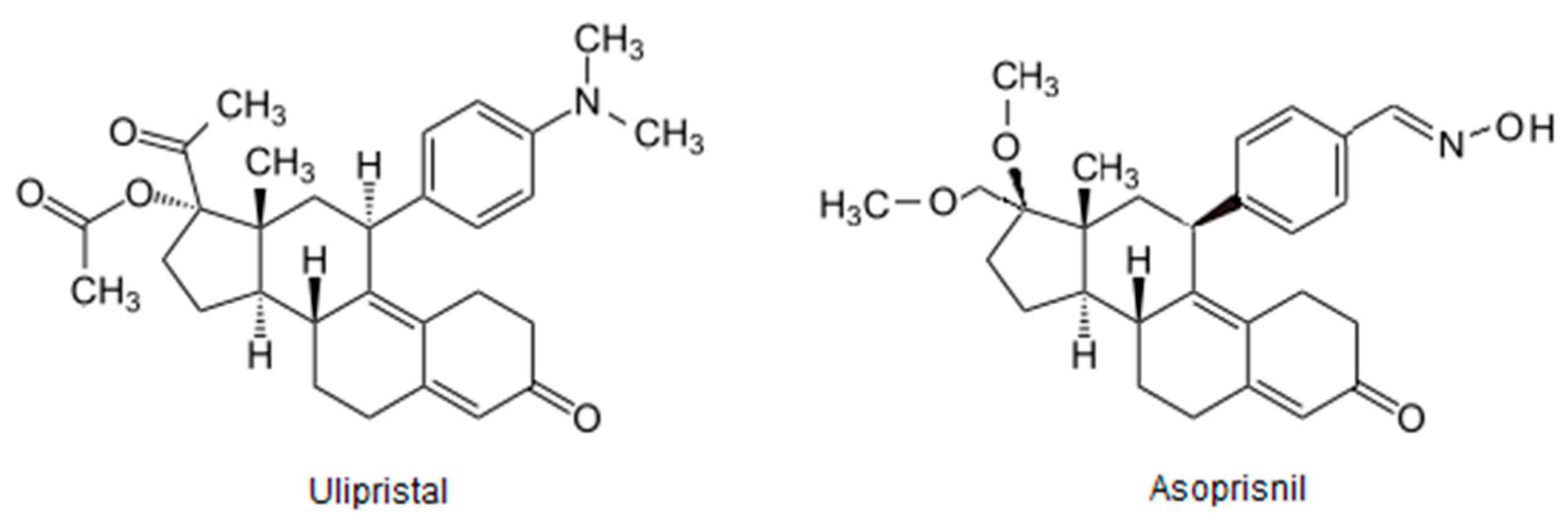
- Chwalisz, K.; Perez, M.C.; DeManno, D.; Winkel, C.; Schubert, G.; Elger, W. Selective Progesterone Receptor Modulator Development and Use in the Treatment of Leiomyomata and Endometriosis. Endocr. Rev. 2005, 26, 423–438. [Google Scholar] [CrossRef]
- Bestel, E.; Donnez, J. The potential of selective progesterone receptor modulators for the treatment of uterine fibroids. Expert Rev. Endocrinol. Metab. 2013, 9, 79–92. [Google Scholar] [CrossRef] [PubMed]
- Błaszczak-Świątkiewicz, K. New Selective Progesterone Receptor Modulators and Their Impact on the RANK/RANKL Complex Activity. Molecules 2020, 25, 1321. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kolb, A.D.; Dai, J.; Keller, E.T.; Bussard, K.M. ‘Educated’ Osteoblasts Reduce Osteoclastogenesis in a Bone-Tumor Mimetic Microenvironment. Cancers 2021, 13, 263. [Google Scholar] [CrossRef]
- Eosta, B.; Ebenedetti, G.; Emiossec, P. Classical and Paradoxical Effects of TNF-α on Bone Homeostasis. Front. Immunol. 2014, 5, 48. [Google Scholar] [CrossRef]
- Donnez, J.; Tomaszewski, J.; Vázquez, F.; Bouchard, P.; Lemieszczuk, B.; Baró, F.; Nouri, K.; Selvaggi, L.; Sodowski, K.; Bestel, E.; et al. Ulipristal Acetate versus Leuprolide Acetate for Uterine Fibroids. N. Engl. J. Med. 2012, 366, 421–432. [Google Scholar] [CrossRef] [Green Version]
- Sun, X.; Wei, B.; Peng, Z.; Fu, Q.; Wang, C.; Zhen, J.; Sun, J. Protective effects of Dipsacus asper polysaccharide on osteoporosis in vivo by regulating RANKL/RANK/OPG/VEGF and PI3K/Akt/eNOS pathway. Int. J. Biol. Macromol. 2019, 129, 579–587. [Google Scholar] [CrossRef] [PubMed]
- Cai, C.; Liu, C.; Zhao, L.; Liu, H.; Li, W.; Guan, H.; Zhao, L.; Xiao, J. Effects of Taxifolin on Osteoclastogenesis in vitro and in vivo. Front. Pharmacol. 2018, 9, 1286. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sun, X.; Xie, Z.; Hu, B.; Zhang, B.; Ma, Y.; Pan, X.; Huang, H.; Wang, J.; Zhao, X.; Jie, Z.; et al. The Nrf2 activator RTA-408 attenuates osteoclastogenesis by inhibiting STING dependent NF-κb signaling. Redox Biol. 2020, 28, 101309. [Google Scholar] [CrossRef] [PubMed]
- Zhi, X.; Wang, L.; Chen, H.; Fang, C.; Cui, J.; Hu, Y.; Cao, L.; Weng, W.; Zhou, Q.; Qin, L.; et al. l -tetrahydropalmatine suppresses osteoclastogenesis in vivo and in vitro via blocking RANK-TRAF6 interactions and inhibiting NF-κB and MAPK pathways. J. Cell. Mol. Med. 2020, 24, 785–798. [Google Scholar] [CrossRef] [Green Version]
- Menarim, B.C.; Gillis, K.H.; Oliver, A.; Mason, C.; Ngo, Y.; Werre, S.R.; Barrett, S.H.; Luo, X.; Byron, C.R.; Dahlgren, L.A. Autologous bone marrow mononuclear cells modulate joint homeostasis in an equine in vivo model of synovitis. FASEB J. 2019, 33, 14337–14353. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Culemann, S.; Grüneboom, A.; Ávila, J.; Ángel, N.; Weidner, D.; Lämmle, K.F.; Rothe, T.; Quintana, J.A.; Kirchner, P.; Krljanac, B.; et al. Locally renewing resident synovial macrophages provide a protective barrier for the joint. Nat. Cell Biol. 2019, 572, 670–675. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, A.; Fearon, U.; Veale, D.J.; Godson, C. Macrophages in Synovial Inflammation. Front. Immunol. 2011, 2, 52. [Google Scholar] [CrossRef] [Green Version]
- Chen, W.; Sun, Y.; Gu, X.; Hao, Y.; Liu, X.; Lin, J.; Chen, J.; Chen, S. Conditioned medium of mesenchymal stem cells delays osteoarthritis progression in a rat model by protecting subchondral bone, maintaining matrix homeostasis, and enhancing autophagy. J. Tissue Eng. Regen. Med. 2019, 13, 1618–1628. [Google Scholar] [CrossRef]
- Marini, H.; Minutoli, L.; Polito, F.; Bitto, A.; Altavilla, D.; Atteritano, M.; Gaudio, A.; Mazzaferro, S.; Frisina, A.; Frisina, N.; et al. OPG and sRANKL serum concentrations in osteopenic, postmenopausal women after 2-year genistein administration. Bone Miner Res. 2008, 23, 715–720. [Google Scholar] [CrossRef]
- Arcoraci, V.; Atteritano, M.; Squadrito, F.; D’Anna, R.; Marini, H.; Santoro, D.; Minutoli, L.; Messina, S.; Altavilla, D.; Bitto, A. Antiosteoporotic Activity of Genistein Aglycone in Postmenopausal Women: Evidence from a Post-Hoc Analysis of a Multicenter Randomized Controlled Trial. Nutrients 2017, 9, 179. [Google Scholar] [CrossRef] [Green Version]
- Zhang, J.; Wang, F.; Qu, G.; Wang, B. Murrayanine suppresses the proliferation and metastasis of human breast cancer cells via induction of apoptosis and inhibition of RANK/RANKL signaling pathway. Appl. Biol. Chem. 2021, 64, 32. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Woźniczka, M.; Błaszczak-Świątkiewicz, K. New Generation of Meso and Antiprogestins (SPRMs) into the Osteoporosis Approach. Molecules 2021, 26, 6491. https://doi.org/10.3390/molecules26216491
Woźniczka M, Błaszczak-Świątkiewicz K. New Generation of Meso and Antiprogestins (SPRMs) into the Osteoporosis Approach. Molecules. 2021; 26(21):6491. https://doi.org/10.3390/molecules26216491
Chicago/Turabian StyleWoźniczka, Magdalena, and Katarzyna Błaszczak-Świątkiewicz. 2021. "New Generation of Meso and Antiprogestins (SPRMs) into the Osteoporosis Approach" Molecules 26, no. 21: 6491. https://doi.org/10.3390/molecules26216491
APA StyleWoźniczka, M., & Błaszczak-Świątkiewicz, K. (2021). New Generation of Meso and Antiprogestins (SPRMs) into the Osteoporosis Approach. Molecules, 26(21), 6491. https://doi.org/10.3390/molecules26216491





