Orientin Improves Substrate Utilization and the Expression of Major Genes Involved in Insulin Signaling and Energy Regulation in Cultured Insulin-Resistant Liver Cells
Abstract
1. Introduction
2. Results
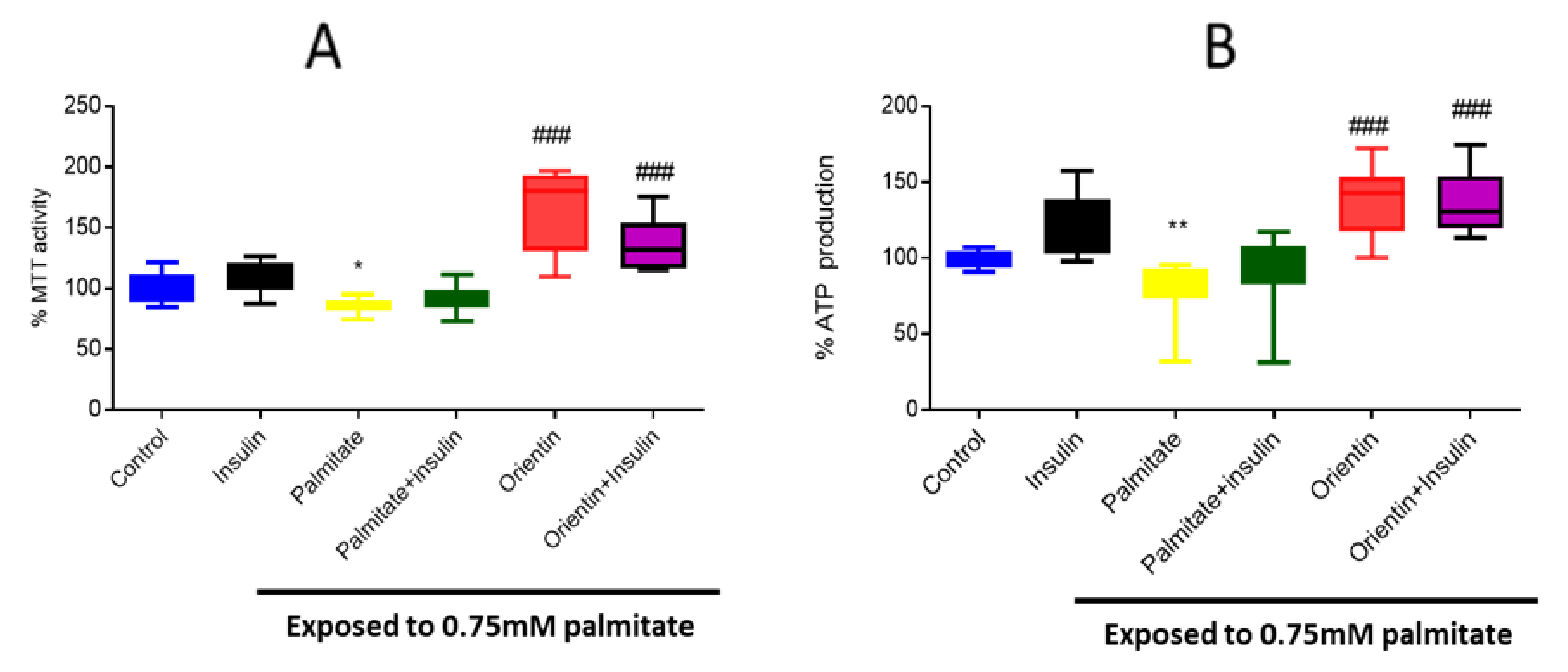
2.1. Effect of Orientin on Metabolic Activity in Palmitate-Exposed Liver Cells
2.2. Effect of Orientin on Substrate Metabolism in Palmitate-Exposed Liver Cells
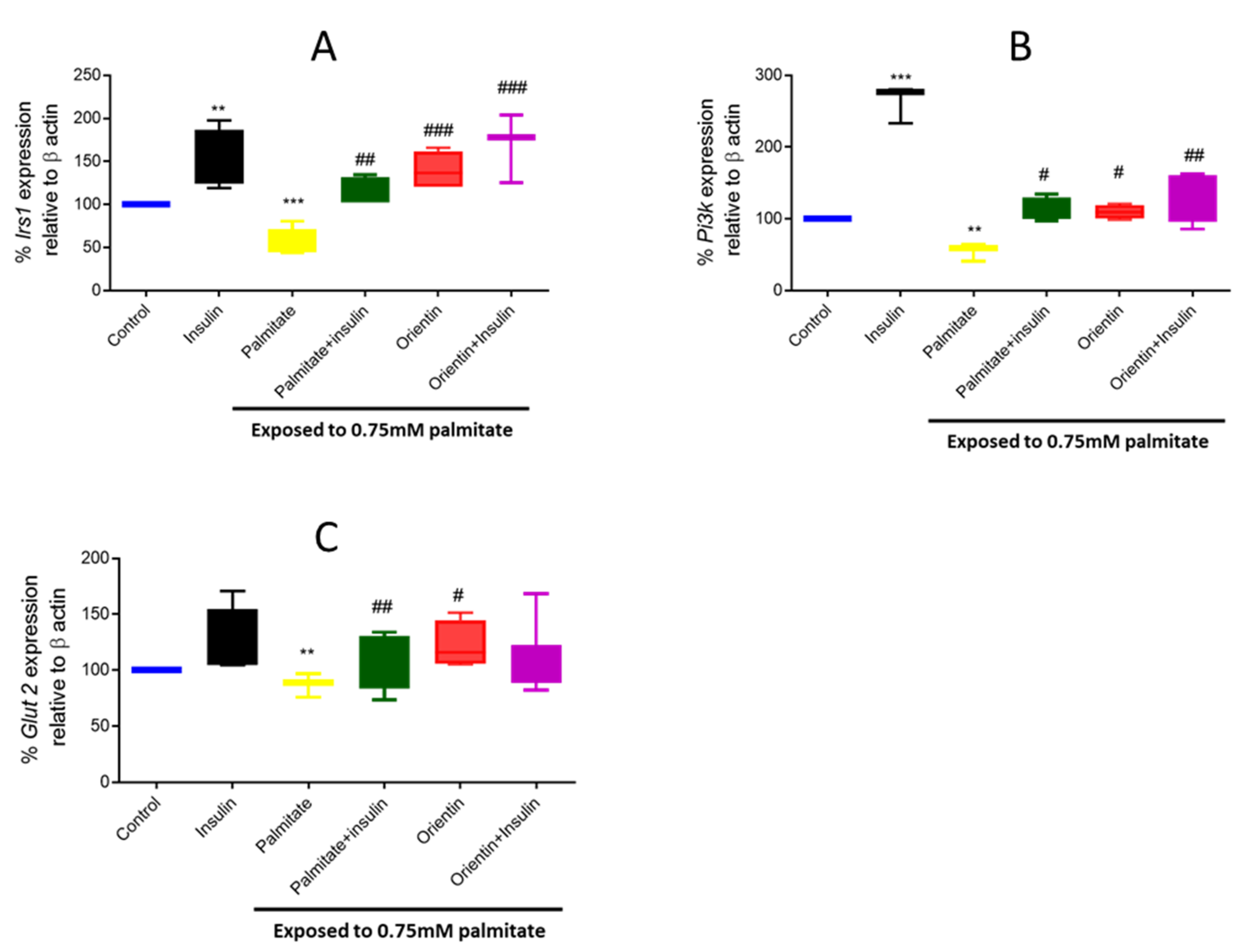
2.3. Effect of Orientin in Modulating the Expression of Different Genes Involved in Insulin Signaling in Palmitate-Exposed Liver Cells
2.4. Orientin Modulate Energy Metabolism in Palmitate-Induced Insulin-Resistant C3A Liver Cells
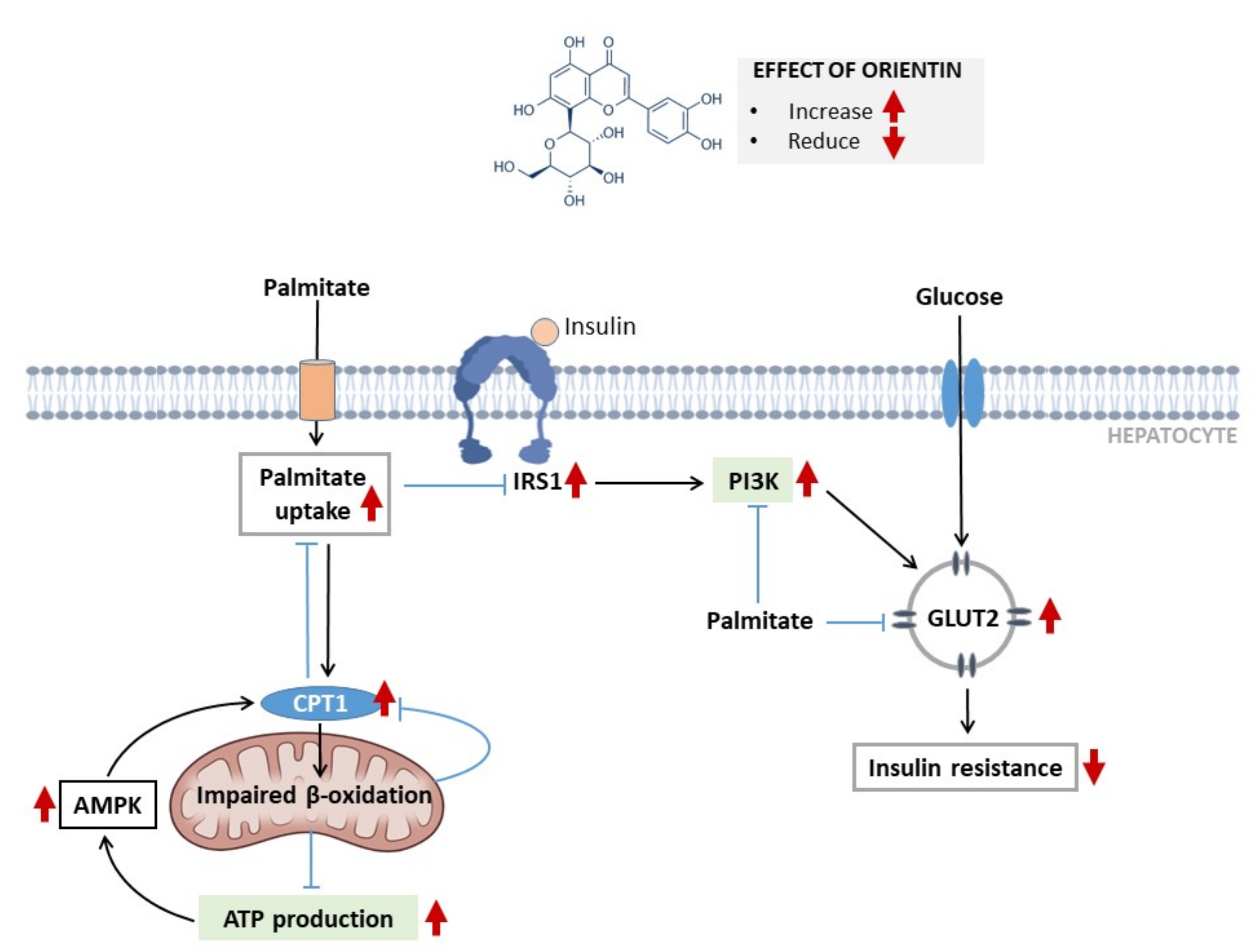
3. Discussion and Conclusions
4. Materials and Methods
4.1. Reagents and Kits Used
4.2. Cell Culture, Establishing the Experimental Model of Insulin Resistance, and Orientin Treatment
4.3. Determining Metabolic Activity
4.4. Assessing Substrate Metabolism: Glucose and Palmitate Uptake
4.5. mRNA Expression Analysis Using RT-PCR
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
Abbreviations
References
- World Health Organization (WHO). The Top Ten Leading Causes of Death. Available online: https://www.who.int/news-room/fact-sheets/detail/the-top-10-causes-of-death#:~:text=The%20top%20global%20causes%20of,birth%20asphyxia%20and%20birth%20trauma%2C (accessed on 3 August 2021).
- International Diabetes Federation (IDF). IDF Diabetes Atlas 2019. Available online: https://www.diabetesatlas.org/en/ (accessed on 1 August 2021).
- World Health Organization (WHO). World Health Statistics 2012. Available online: https://www.who.int/gho/publications/world_health_statistics/EN_WHS2012_Full.pdf (accessed on 29 July 2021).
- Trikkalinou, A.; Papazafiropoulou, A.K.; Melidonis, A. Type 2 diabetes and quality of life. World J. Diabetes 2017, 8, 120–129. [Google Scholar] [CrossRef]
- Jeong, S.U.; Kang, D.G.; Lee, D.H.; Lee, K.W.; Lim, D.M.; Kim, B.J.; Park, K.Y.; Chin, H.J.; Koh, G. Clinical characteristics of type 2 diabetes patients according to family history of diabetes. Korean Diabetes J. 2010, 34, 222–228. [Google Scholar] [CrossRef][Green Version]
- Eckel, R.H.; Grundy, S.M.; Zimmet, P.Z. The metabolic syndrome. Lancet 2005, 365, 1415–1428. [Google Scholar] [CrossRef]
- Mazibuko-Mbeje, S.E.; Dludla, P.V.; Roux, C.; Johnson, R.; Ghoor, S.; Joubert, E.; Louw, J.; Opoku, A.R.; Muller, C.J.F. Aspalathin-Enriched Green Rooibos Extract Reduces Hepatic Insulin Resistance by Modulating PI3K/AKT and AMPK Pathways. Int. J. Mol. Sci. 2019, 20, 633. [Google Scholar] [CrossRef]
- Dludla, P.V.; Silvestri, S.; Orlando, P.; Mazibuko-Mbeje, S.E.; Johnson, R.; Marcheggiani, F.; Cirilli, I.; Muller, C.J.F.; Louw, J.; Chellan, N.; et al. Palmitate-induced toxicity is associated with impaired mitochondrial respiration and accelerated oxidative stress in cultured cardiomyocytes: The critical role of coenzyme Q (9/10). Toxicol. Vitr. 2020, 68, 104948. [Google Scholar] [CrossRef]
- Muller, C.J.F.; Malherbe, C.J.; Chellan, N.; Yagasaki, K.; Miura, Y.; Joubert, E. Potential of rooibos, its major C-glucosyl flavonoids, and Z-2-(β-D-glucopyranosyloxy)-3-phenylpropenoic acid in prevention of metabolic syndrome. Crit. Rev. Food Sci. Nutr. 2018, 58, 227–246. [Google Scholar] [CrossRef]
- Stander, M.A.; Van Wyk, B.E.; Taylor, M.J.; Long, H.S. Analysis of Phenolic Compounds in Rooibos Tea (Aspalathus linearis) with a Comparison of Flavonoid-Based Compounds in Natural Populations of Plants from Different Regions. J. Agric. Food Chem. 2017, 65, 10270–10281. [Google Scholar] [CrossRef] [PubMed]
- Johnson, R.; de Beer, D.; Dludla, P.V.; Ferreira, D.; Muller, C.J.; Joubert, E. Aspalathin from Rooibos (Aspalathus linearis): A Bioactive C-glucosyl Dihydrochalcone with Potential to Target the Metabolic Syndrome. Planta Med. 2018, 84, 568–583. [Google Scholar] [CrossRef]
- Mazibuko-Mbeje, S.E.; Ziqubu, K.; Dludla, P.V.; Tiano, L.; Silvestri, S.; Orlando, P.; Nyawo, T.A.; Louw, J.; Kappo, A.P.; Muller, C.J.F. Isoorientin ameliorates lipid accumulation by regulating fat browning in palmitate-exposed 3T3-L1 adipocytes. Metabol. Open 2020, 6, 100037. [Google Scholar] [CrossRef] [PubMed]
- Alonso-Castro, A.J.; Zapata-Bustos, R.; Gómez-Espinoza, G.; Salazar-Olivo, L.A. Isoorientin reverts TNF-α-induced insulin resistance in adipocytes activating the insulin signaling pathway. Endocrinology 2012, 153, 5222–5230. [Google Scholar] [CrossRef] [PubMed]
- Ziqubu, K.; Dludla, P.V.; Joubert, E.; Muller, C.J.; Louw, J.; Tiano, L.; Nkambule, B.B.; Kappo, A.P.; Mazibuko-Mbeje, S. E. Isoorientin: A dietary flavone with the potential to ameliorate diverse metabolic complications. Pharmacol. Res. 2020, 158, 104867. [Google Scholar] [CrossRef] [PubMed]
- Ku, S.K.; Kwak, S.; Bae, J.S. Orientin inhibits high glucose-induced vascular inflammation in vitro and in vivo. Inflammation 2014, 37, 2164–2173. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Lan, N.; Ren, J.; Wu, Y.; Wang, S.T.; Huang, X.F.; Yu, Y. Orientin improves depression-like behavior and BDNF in chronic stressed mice. Mol. Nutr. Food Res. 2015, 59, 1130–1142. [Google Scholar] [CrossRef]
- Lam, K.Y.; Ling, A.P.K.; Koh, R.Y.; Wong, Y.P.; Say, Y. H. A Review on Medicinal Properties of Orientin. Adv. Pharmacol. Sci. 2016, 2016, 4104595. [Google Scholar] [CrossRef] [PubMed]
- Santoleri, D.; Titchenell, P.M. Resolving the Paradox of Hepatic Insulin Resistance. Cell Mol. Gastroenterol. Hepatol. 2019, 7, 447–456. [Google Scholar] [CrossRef]
- Meshkani, R.; Adeli, K. Hepatic insulin resistance, metabolic syndrome and cardiovascular disease. Clin. Biochem. 2009, 42, 1331–1346. [Google Scholar] [CrossRef]
- Stanford, K.I.; Goodyear, L.J. Exercise and type 2 diabetes: Molecular mechanisms regulating glucose uptake in skeletal muscle. Adv. Physiol. Educ. 2014, 38, 308–314. [Google Scholar] [CrossRef] [PubMed]
- Ziqubu, K.; Muller, C.J.; Dludla, P.V.; Mthembu, S.X.; Obonye, N.; Louw, J.; Kappo, A.P.; Silvestri, S.; Orlando, P.; Tiano, L.; et al. Impact of Isoorientin on Metabolic Activity and Lipid Accumulation in Differentiated Adipocytes. Molecules 2020, 25, 1773. [Google Scholar] [CrossRef] [PubMed]
- Rena, G.; Hardie, D.G.; Pearson, E.R. The mechanisms of action of metformin. Diabetologia 2017, 60, 1577–1585. [Google Scholar] [CrossRef] [PubMed]
- Home, P.; Blonde, L.; Kalra, S.; Ji, L.; Guyot, P.; Brulle-Wohlhueter, C.; Murray, E.; Shah, R.; Sayre, T.; Shaunik, A. Insulin glargine/lixisenatide fixed-ratio combination (iGlarLixi) compared with premix or addition of meal-time insulin to basal insulin in people with type 2 diabetes: A systematic review and Bayesian network meta-analysis. Diabetes Obes. Metab. 2020, 22, 2179–2188. [Google Scholar] [CrossRef] [PubMed]
- Nasri, H.; Baradaran, A.; Shirzad, H.; Rafieian-Kopaei, M. New concepts in nutraceuticals as alternative for pharmaceuticals. Int. J. Prev. Med. 2014, 5, 1487–1499. [Google Scholar] [PubMed]
- Nyambuya, T.M.; Nkambule, B.B.; Mazibuko-Mbeje, S.E.; Mxinwa, V.; Mokgalaboni, K.; Orlando, P.; Silvestri, S.; Louw, J.; Tiano, L.; Dludla, P. V. A Meta-Analysis of the Impact of Resveratrol Supplementation on Markers of Renal Function and Blood Pressure in Type 2 Diabetic Patients on Hypoglycemic Therapy. Molecules 2020, 25, 5645. [Google Scholar] [CrossRef]
- Derosa, G.; Limas, C.P.; Macías, P.C.; Estrella, A.; Maffioli, P. Dietary and nutraceutical approach to type 2 diabetes. Arch. Med. Sci. 2014, 10, 336–344. [Google Scholar] [CrossRef] [PubMed]
- Mirmiran, P.; Bahadoran, Z.; Azizi, F. Functional foods-based diet as a novel dietary approach for management of type 2 diabetes and its complications: A review. World J. Diabetes 2014, 5, 267–281. [Google Scholar] [CrossRef] [PubMed]
- Hewlings, S.J.; Kalman, D.S. Curcumin: A Review of Its Effects on Human Health. Foods 2017, 6, 92. [Google Scholar] [CrossRef] [PubMed]
- Mthembu, S.X.H.; Dludla, P.V.; Ziqubu, K.; Nyambuya, T.M.; Kappo, A.P.; Madoroba, E.; Nyawo, T.A.; Nkambule, B.B.; Silvestri, S.; Muller, C.J.F.; et al. The Potential Role of Polyphenols in Modulating Mitochondrial Bioenergetics within the Skeletal Muscle: A Systematic Review of Preclinical Models. Molecules 2021, 26, 2791. [Google Scholar] [CrossRef] [PubMed]
- Dludla, P.V.; Jack, B.; Viraragavan, A.; Pheiffer, C.; Johnson, R.; Louw, J.; Muller, C.J. A dose-dependent effect of dimethyl sulfoxide on lipid content, cell viability and oxidative stress in 3T3-L1 adipocytes. Toxicol. Rep. 2018, 5, 1014–1020. [Google Scholar] [CrossRef] [PubMed]
- Se, M. In Vitro and In Vivo Effect of Aspalathus Linearis and Its Major Polyphenols on Carbohydrate and Lipid Metabolism in Insulin Resistant Models; Department of Biochemistry, University of Zululand: Richards Bay, South Africa, 2014. [Google Scholar]
- Mosmann, T. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar] [CrossRef]
- Mazibuko-Mbeje, S.E.; Mthembu, S.X.H.; Dludla, P.V.; Madoroba, E.; Chellan, N.; Kappo, A.P.; Muller, C.J. Antimycin A-induced mitochondrial dysfunction is consistent with impaired insulin signaling in cultured skeletal muscle cells. Toxicol. In Vitro 2021, 76, 105224. [Google Scholar] [CrossRef]


| Gene Probes | Assay ID |
|---|---|
| Beta-2 Microglobulin (B2m) | Hs99999907-m1 |
| Phosphatidylinositol 3 Kinase (Pi3k) | Hs00933163-R1 |
| Insulin receptor substrate 1 (Irs1) | Hs00178563-R1 |
| Glucose transporter 2 (Glut2) | Hs00165775-R1 |
| Carnitine Palmitoyl transferase 1 (Cpt1) | Hs00912671-m1 |
| 5’adenosine monophosphate activated protein kinase (Ampk) | Hs00178903-m1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mazibuko-Mbeje, S.E.; Mthembu, S.X.H.; Tshiitamune, A.; Muvhulawa, N.; Mthiyane, F.T.; Ziqubu, K.; Muller, C.J.F.; Dludla, P.V. Orientin Improves Substrate Utilization and the Expression of Major Genes Involved in Insulin Signaling and Energy Regulation in Cultured Insulin-Resistant Liver Cells. Molecules 2021, 26, 6154. https://doi.org/10.3390/molecules26206154
Mazibuko-Mbeje SE, Mthembu SXH, Tshiitamune A, Muvhulawa N, Mthiyane FT, Ziqubu K, Muller CJF, Dludla PV. Orientin Improves Substrate Utilization and the Expression of Major Genes Involved in Insulin Signaling and Energy Regulation in Cultured Insulin-Resistant Liver Cells. Molecules. 2021; 26(20):6154. https://doi.org/10.3390/molecules26206154
Chicago/Turabian StyleMazibuko-Mbeje, Sithandiwe E., Sinenhlanhla X. H. Mthembu, Andani Tshiitamune, Ndivhuwo Muvhulawa, Fikile T. Mthiyane, Khanyisani Ziqubu, Christo J. F. Muller, and Phiwayinkosi V. Dludla. 2021. "Orientin Improves Substrate Utilization and the Expression of Major Genes Involved in Insulin Signaling and Energy Regulation in Cultured Insulin-Resistant Liver Cells" Molecules 26, no. 20: 6154. https://doi.org/10.3390/molecules26206154
APA StyleMazibuko-Mbeje, S. E., Mthembu, S. X. H., Tshiitamune, A., Muvhulawa, N., Mthiyane, F. T., Ziqubu, K., Muller, C. J. F., & Dludla, P. V. (2021). Orientin Improves Substrate Utilization and the Expression of Major Genes Involved in Insulin Signaling and Energy Regulation in Cultured Insulin-Resistant Liver Cells. Molecules, 26(20), 6154. https://doi.org/10.3390/molecules26206154









