Zeaxanthin-Rich Extract from Superfood Lycium barbarum Selectively Modulates the Cellular Adhesion and MAPK Signaling in Melanoma versus Normal Skin Cells In Vitro
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material
2.2. Isolation of Carotenoids from L. barbarum Varieties Erma and Biglifeberry
2.3. Cell Lines
2.4. Cytotoxicity Testing
2.5. Antiproliferative Capacity
2.6. Flow-Cytometry Measurements of Membrane and Intracellular Markers
2.7. Immunoenzymatic Testing (ELISA)
2.8. Data Analysis
3. Results
3.1. Characterization of the Compounds through HPLC-PDA Analysis
3.2. Evaluation of the Extracts Effect on Skin-Derived Cells Growth
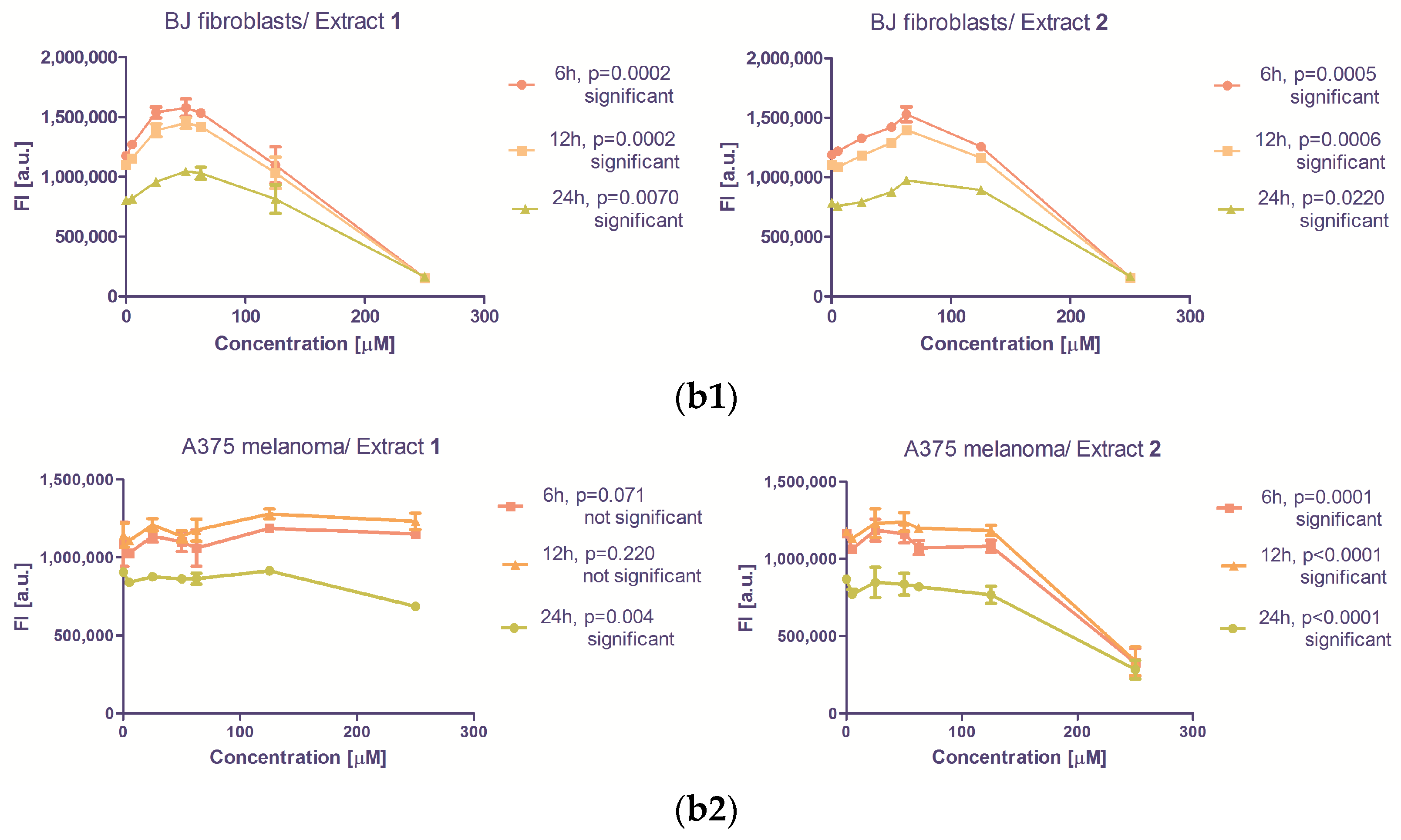
3.2.1. Cytotoxicity
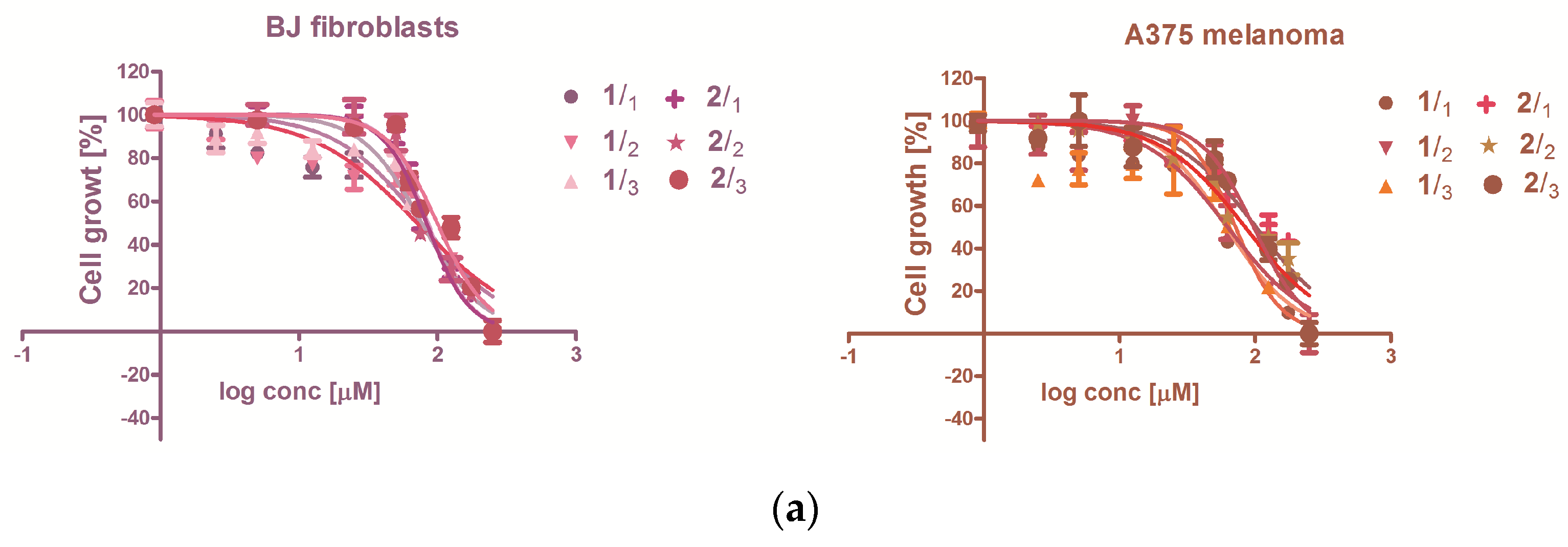
3.2.2. The Antiproliferative Capacity
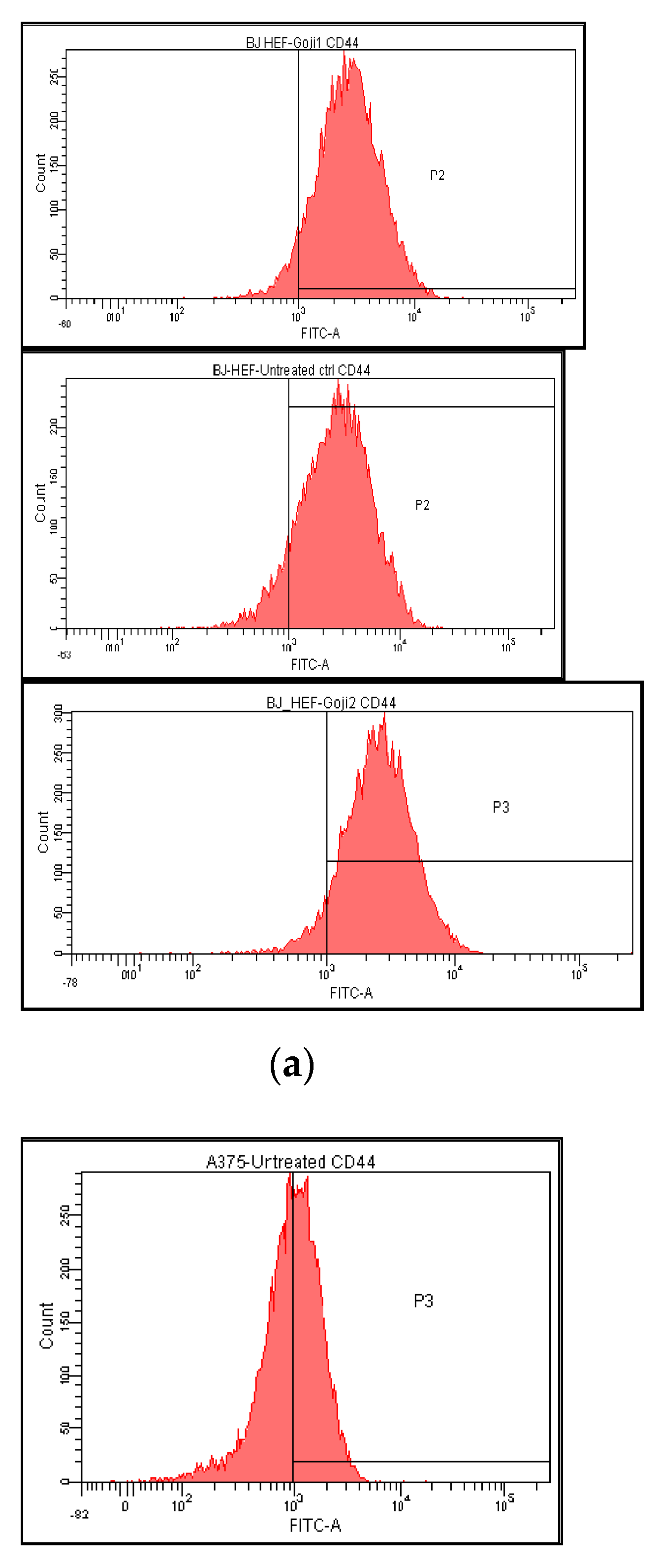
3.3. The Extracts 1 and 2 Effect on CD44 and CD105 Markers
3.4. The Evaluation of NF-kβ Transcription Factor
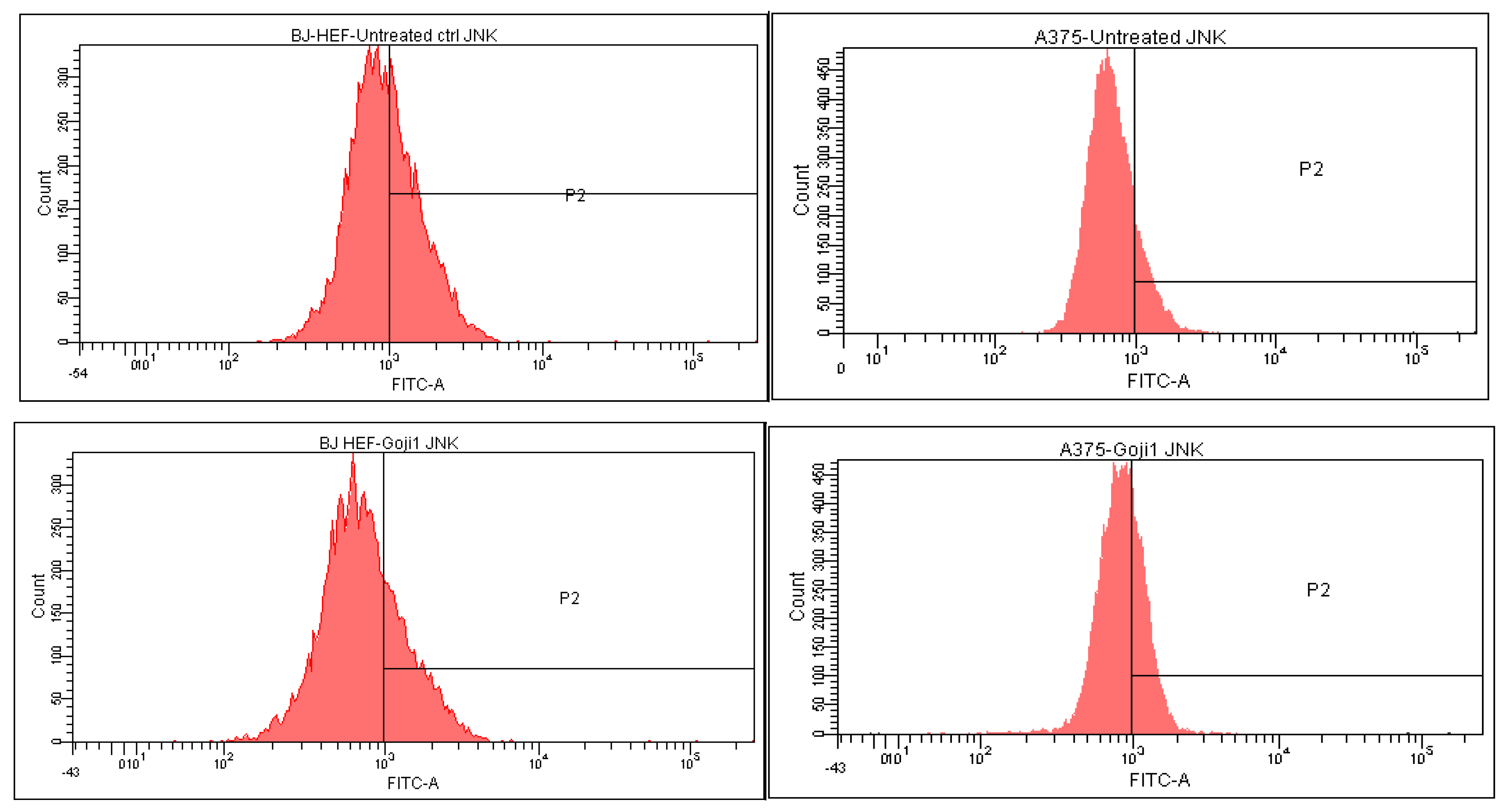
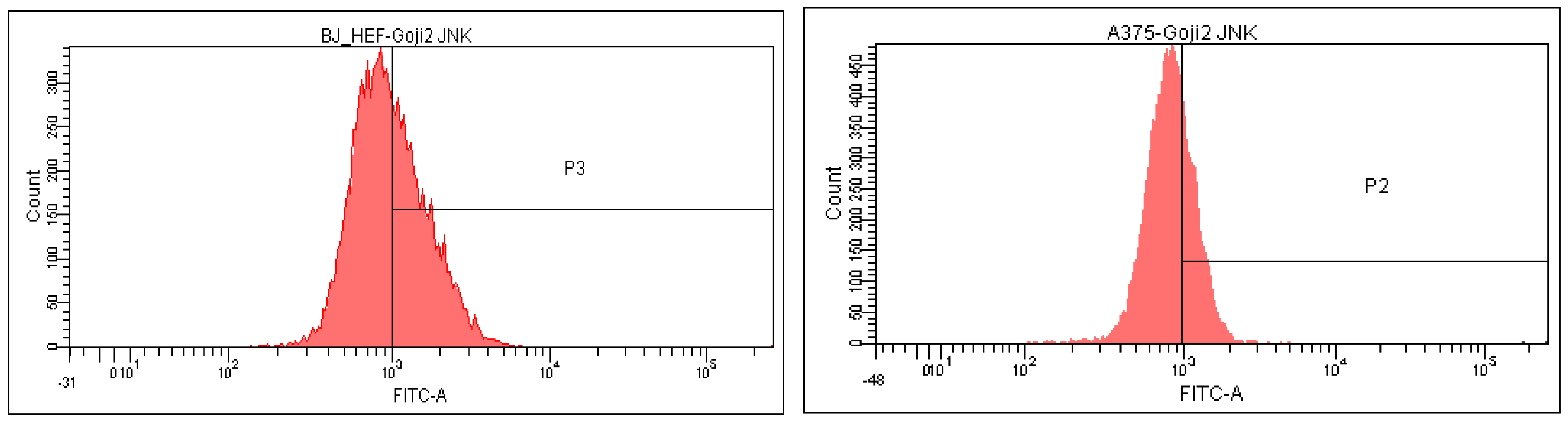
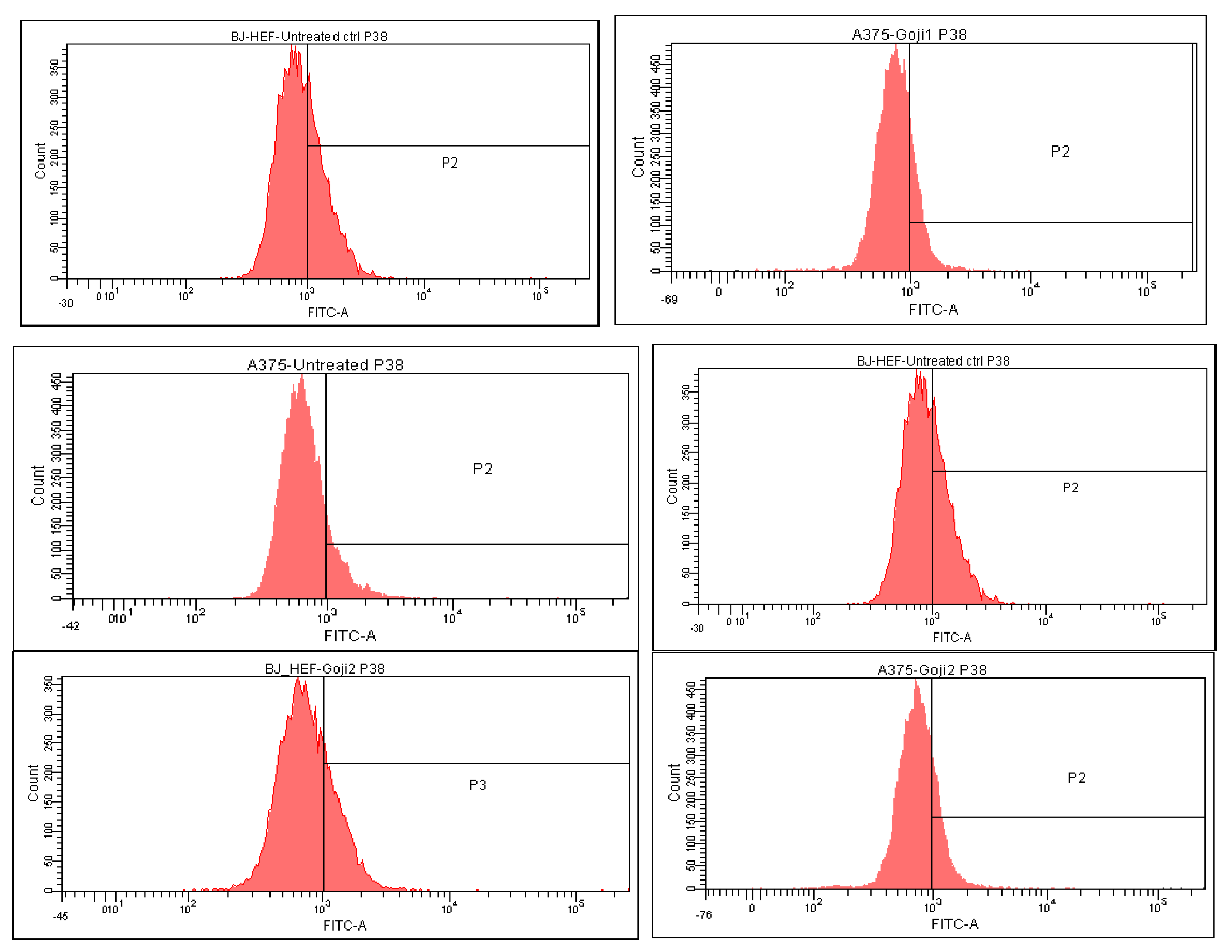
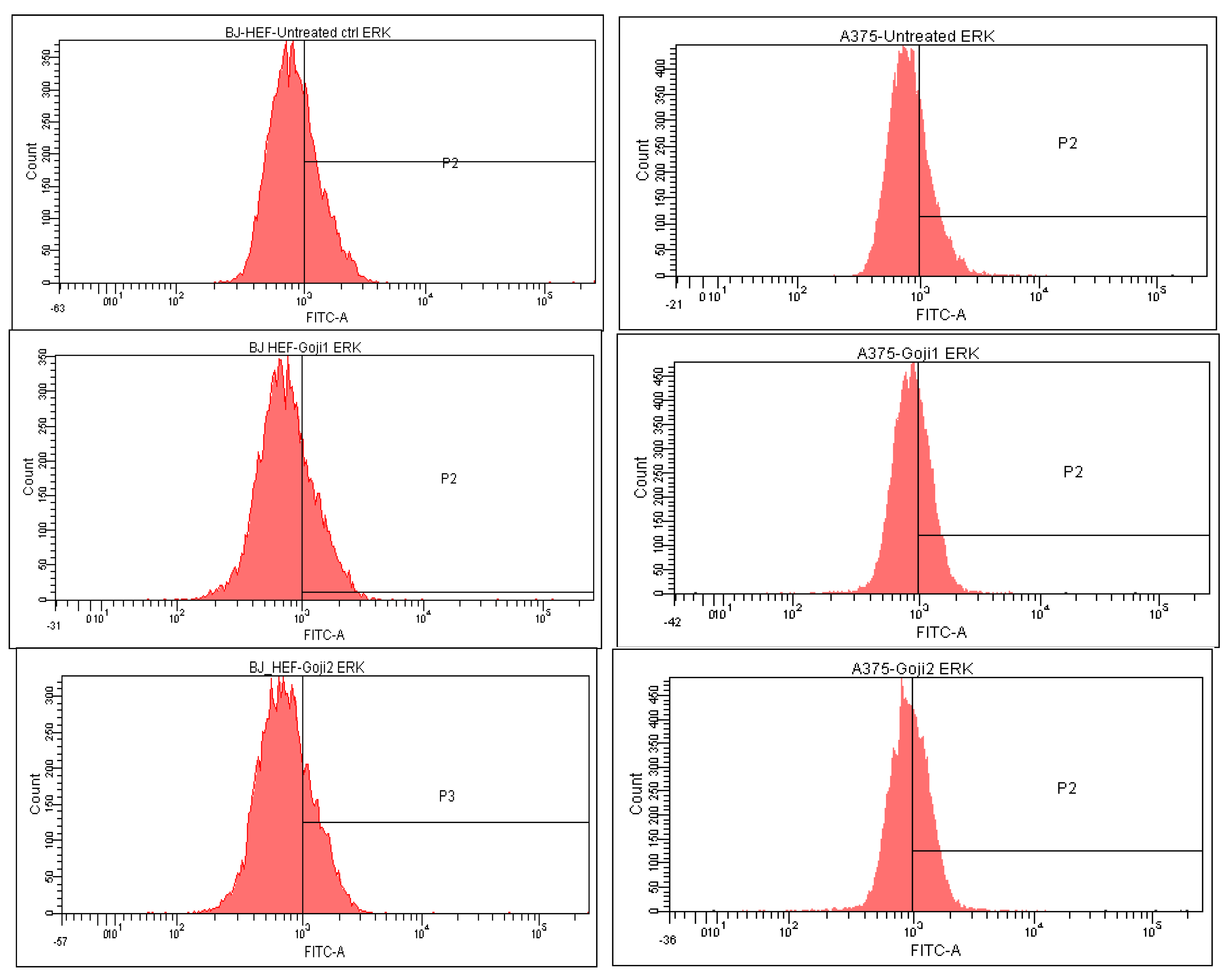
3.5. Modulation of MAPK Signaling
4. Discussion and Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Murillo, A.G.; Hu, S.; Fernandez, M.L. Zeaxanthin: Metabolism, Properties, and Antioxidant Protection of Eyes, Heart, Liver, and Skin. Antioxidants 2019, 8, 390. [Google Scholar] [CrossRef]
- Roberts, R.L.; Green, J.; Lewis, B. Lutein and zeaxanthin in eye and skin health. Clin. Dermatol. 2009, 27, 195–201. [Google Scholar] [CrossRef]
- Sies, H.; Stahl, W. Non-nutritive bioactive constituents of plants: Lycopene, lutein and zeaxanthin. Int. J. Vitam. Nutr. Res. 2003, 73, 95–100. [Google Scholar] [CrossRef]
- Li, R.; Turner, S.D.; Brautigan, D.L. Xanthophylls lutein and zeaxanthin modify gene expression and induce synthesis of hyaluronan in keratinocyte model of human skin. Biochem. Biophys. Rep. 2015, 4, 52–58. [Google Scholar] [CrossRef]
- Schwartz, S.; Frank, E.; Gierhart, D.; Simpson, P.; Frumento, R. Zeaxanthin-based dietary supplement and topical serum improve hydration and reduce wrinkle count in female subjects. J. Cosmet. Dermatol. 2016, 15, 13–20. [Google Scholar] [CrossRef] [PubMed]
- Wu, N.L.; Chiang, Y.C.; Huang, C.C.; Fang, J.Y.; Chen, D.F.; Hung, C.F. Zeaxanthin inhibits PDGF-BB-induced migration in human dermal fibroblasts. Exp. Dermatol. 2010, 19, e173–e181. [Google Scholar] [CrossRef]
- Bi, M.C.; Hose, N.; Xu, C.L.; Zhang, C.; Sassoon, J.; Song, E. Nonlethal levels of zeaxanthin inhibit cell Invasion, and secretion of MMP-2 via NF-κβ pathway in cultured human uveal melanoma cells. J. Ophtalmol. 2016, 2016, 8734309. [Google Scholar] [CrossRef]
- Kulczyński, B.; Gramza-Michałowska, A. Goji Berry (Lycium barbarum): Composition and Health Effects—A Review. Pol. J. Food Nutr. Sci. 2016, 66, 67–75. [Google Scholar] [CrossRef]
- Chang, R.C.; So, K.F. Use of anti-aging herbal medicine, Lycium barbarum, against aging-associated diseases. What do we know so far? Cell. Mol. Neurobiol. 2008, 28, 643–652. [Google Scholar] [CrossRef]
- Yang, X.; Bai, H.; Cai, W.; Li, J.; Zhou, Q.; Wang, Y.; Han, J.; Zhu, X.; Dong, M.; Hu, D. Lycium barbarum polysaccharides reduce intestinal ischemia/reperfusion injuries in rats. Chem. Biol. Interact. 2013, 204, 166–172. [Google Scholar] [CrossRef]
- Amagase, H.; Sun, B.; Borek, C. Lycium barbarum (goji) juice improves in vivo antioxidant biomarkers in serum of healthy adults. Nutr. Res. 2009, 29, 19–25. [Google Scholar] [CrossRef] [PubMed]
- Wawruszak, A.; Czerwonka, A.; Okła, K.; Rzeski, W. Anticancer effect of ethanol Lycium barbarum (Goji berry) extract on human breast cancer T47D cell line. Nat. Prod. Res. 2016, 30, 1993–1996. [Google Scholar] [CrossRef] [PubMed]
- Shen, L.; Du, G. Lycium barbarum polysaccharide stimulates proliferation of MCF-7 cells by the ERK pathway. Life Sci. 2012, 24, 353–357. [Google Scholar] [CrossRef] [PubMed]
- Ceccarini, M.R.; Vannini, S.; Cataldi, S.; Moretti, M.; Villarini, M.; Fioretti, B.; Albi, E.; Beccari, T.; Codini, M. In Vitro Protective Effects of Lycium barbarum Berries Cultivated in Umbria (Italy) on Human Hepatocellular Carcinoma Cells. Biomed. Res. Int. 2016, 2016, 7529521. [Google Scholar] [CrossRef] [PubMed]
- Nance, D.M.; Amagase, H.; Luczy-Bachman, G. Effects of Lycium barbarum on basal and LPS-induced cytokine production. Brain Behav. Immun. 2009, 23 (Suppl. S2), S51. [Google Scholar] [CrossRef]
- Potterat, O. Goji (Lycium barbarum and L. chinense): Phytochemistry, Pharmacology and Safety in the Perspective of Traditional Uses and Recent Popularity. Planta Med. 2010, 76, 7–19. [Google Scholar] [CrossRef] [PubMed]
- Hsu, H.J.; Huang, R.F.; Kao, T.H.; Inbaraj, B.S.; Chen, B.H. Preparation of carotenoid extracts and nanoemulsions from Lycium barbarum L. and their effects on growth of HT-29 colon cancer cells. Nanotechnology 2017, 28, 135103. [Google Scholar] [CrossRef] [PubMed]
- Amagase, H.; Farnsworth, N.R. A review of botanical characteristics, phytochemistry, clinical relevance in efficacy and safety of Lycium barbarum fruit (Goji). Food Res. Int. 2011, 44, 1702–1717. [Google Scholar] [CrossRef]
- Reeve, V.E.; Allanson, M.; Arun, S.J.; Domanski, D.; Painter, N. Mice drinking goji berry juice (Lycium barbarum) are protected from UV radiation-induced skin damage via antioxidant pathways. Photochem. Photobiol. Sci. 2010, 9, 601–607. [Google Scholar] [CrossRef]
- Zhao, H.; Alexeev, A.; Chang, E.; Greenburg, G.; Bojanowski, K. Lycium barbarum glycoconjugates: Effect on human skin and cultured dermal fibroblasts. Phytomedicine 2005, 12, 131–137. [Google Scholar] [CrossRef]
- Berra, E.; Diaz-Meco, M.T.; Moscat, J. The Activation of p38 and Apoptosis by the Inhibition of Erk Is Antagonized by the Phosphoinositide 3-Kinase/Akt Pathway. J. Biol. Chem. 1998, 273, 10792–10797. [Google Scholar] [CrossRef] [PubMed]
- Xiao, J.; Wang, J.; Xing, F.; Han, T.; Jiao, R.; Liong, E.C.; Fung, M.L.; So, K.F.; Tipoe, G.L. Zeaxanthin Dipalmitate Therapeutically Improves Hepatic Functions in an Alcoholic Fatty Liver Disease Model through Modulating MAPK Pathway. PLoS ONE 2014, 9, e95214. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Zhao, S.; Karnad, A.; Freeman, J.W. The biology and role of CD44 in cancer progression: Therapeutic implications. J Hematol. Oncol. 2018, 11, 64. [Google Scholar] [CrossRef] [PubMed]
- Magnoni, C.; Giudice, S.; Pellacani, G.; Bertazzoni, G.; Longo, C.; Veratti, E.; Morini, D.; Benassi, L.; Vaschieri, C.; Azzoni, P.; et al. Stem cell properties in cell cultures from different stage of melanoma progression. Appl. Immunohistochem. Mol. Morphol. 2014, 22, 171–181. [Google Scholar] [CrossRef]
- Alt, E.; Yan, Y.; Gehmert, S.; Song, Y.H.; Altman, A.; Gehmert, S.; Vykoukal, D.; Bai, X. Fibroblasts share mesenchymal phenotypes with stem cells, but lack their differentiation and colony-forming potential. Biol. Cell. 2011, 103, 197–208. [Google Scholar] [CrossRef]
- Tzellos, T.G.; Klagas, I.; Vahtsevanos, K.; Triaridis, S.; Printza, A.; Kyrgidis, A.; Karakiulakis, G.; Zouboulis, C.C.; Papakonstantinou, E. Extrinsic ageing in the human skin is associated with alterations in the expression of hyaluronic acid and its metabolizing enzymes. Exp. Dermatol. 2009, 18, 1028–1035. [Google Scholar] [CrossRef]
- Le Coz, V.; Zhu, C.; Devocelle, A.; Vazquez, A.; Boucheix, C.; Azzi, S.; Gallerne, C.; Eid, P.; Lecourt, S.; Giron-Michel, J. IGF-1 contributes to the expansion of melanoma-initiating cells through an epithelial-mesenchymal transition process. Oncotarget 2016, 13, 82511–82527. [Google Scholar] [CrossRef]
- Ahres, T.; Assmann, V.; Fieber, C.; Termeer, C.; Herrlich, P.; Hofmann, M.; Simon, J.C. CD44-is the principal mediator of hyaluronian acid induced melanoma cell proliferation. J. Investig. Dermatol. 2001, 116, 93–101. [Google Scholar] [CrossRef]
- Chen, F.; Bi, D.; Cheng, C.; Ma, S.; Liu, Y.; Cheng, K. Bone morphogenetic protein 7 enhances the osteogenic differentiation of human dermal-derived CD105+ fibroblast cells through the Smad and MAPK pathways. Int. J. Mol. Med. 2019, 43, 37–46. [Google Scholar] [CrossRef]
- Huang, H.C.; Huang, W.Y.; Tsai, T.C.; Hsieh, W.Y.; Ko, W.P.; Chang, K.J.; Chang, T.M. Supercritical fluid extract of Lycium chinense Miller root inhibition of melanin production and its potential mechanisms of action. BMC Complement. Altern. Med. 2014, 14, 208. [Google Scholar] [CrossRef]
- Cheng, J.; Zhou, Z.W.; Sheng, H.P.; He, L.J.; Fan, X.W.; He, Z.X.; Sun, T.; Zhang, X.; Zhao, R.J.; Gu, L.; et al. An evidence-based update on the pharmacological activities and possible molecular targets of Lycium barbarum polysaccharides. Drug Des. Devel. Ther. 2015, 9, 33–78. [Google Scholar] [CrossRef] [PubMed]
- Hempel, J.; Schädle, C.N.; Sprenger, J.; Heller, A.; Carle, R.; Schweiggert, R.M. Ultrastructural deposition forms and bioaccessibility of carotenoids and carotenoid esters from goji berries (Lycium barbarum L.). Food Chem. 2017, 218, 525–533. [Google Scholar] [CrossRef] [PubMed]
- Weller, P.; Breithaupt, D.E. Identification and Quantification of Zeaxanthin Esters in Plants Using Liquid Chromatography-Mass Spectrometry. J. Agric. Food Chem. 2003, 51, 7044–7049. [Google Scholar] [CrossRef] [PubMed]
- Britton, G.; Liaaen, S.; Pfander, H.P. Carotenoids: Isolation and Analysis; Birkhauser Verlag: Basel, Switzerland, 1995. [Google Scholar]
- Miklásová, N.; Fischer-Fodor, E.; Lönnecke, P.; Perde-Schrepler, M.; Virag, P.; Tatomir, C.; Cernea, V.I.; Hey-Hawkins, E.; Silaghi-Dumitrescu, L. Antiproliferative effect and genotoxicity of novel synthesized palladium complexes with organoarsenic ligands. J. Inorg. Biochem. 2009, 103, 1739–1747. [Google Scholar] [CrossRef]
- Perde-Schrepler, M.; Florea, A.; Brie, I.; Virag, P.; Fischer-Fodor, E.; Valcan, A.; Gurzau, E.; Lisencu, C.; Maniu, A. Size-Dependent Cytotoxicity and Genotoxicity of Silver Nanoparticles in Cochlear Cells In Vitro. J. Nanomater. 2019, 2019, 6090259. [Google Scholar] [CrossRef]
- Cenariu, D.; Fischer-Fodor, E.; Virag, P.; Tatomir, C.; Cenariu, M.; Pall, E.; Pintea, A.; Mocan, A.; Crisan, G. In vitro antitumour activity of tomato-extracted carotenoids on human colorectal carcinoma. Not. Bot. Horti. Agrobo. 2015, 43, 293–301. [Google Scholar] [CrossRef]
- Tudor, D.; Nenu, I.; Filip, G.A.; Olteanu, D.; Cenariu, M.; Tabaran, F.; Ion, R.M.; Gligor, L.; Baldea, I. Combined regimen of photodynamic therapy mediated by Gallium phthalocyanine chloride and Metformin enhances anti-melanoma efficacy. PLoS ONE. 2017, 12, e0173241. [Google Scholar] [CrossRef]
- Pintea, A.; Bunea, A.; Socaciu, C. Effect of esterification on thermal stability and antioxidant activity of zeaxanthin. In Proceedings of the 7th International Congress on Pigments in Food, Novara, Italy, 18–21 June 2013; pp. 174–177, ISBN 978-88-903360-3-4. [Google Scholar]
- Inbaraj, S.B.; Lu, H.; Kao, T.H.; Chen, B.H. Simultaneous determination of phenolic acids and flavonoids in Lycium barbarum Linnaeus by HPLC–DAD–ESI-MS. J. Pharm. Biomed. Anal. 2010, 51, 549–556. [Google Scholar] [CrossRef]
- Mocan, A.; Vlase, L.; Vodnar, D.C.; Bischin, C.; Hanganu, D.; Gheldiu, A.M.; Oprean, R.; Silaghi-Dumitrescu, R.; Crișan, G. Polyphenolic Content, Antioxidant and Antimicrobial Activities of Lycium barbarum L. and Lycium chinense Mill. Leaves. Molecules 2014, 19, 10056–10073. [Google Scholar] [CrossRef]
- Mocan, A.; Cairone, F.; Locatelli, M.; Cacciagrano, F.; Carradori, S.; Vodnar, D.C.; Crisan, G.; Simonetti, G.; Cesa, S. Polyphenols from Lycium barbarum (Goji) fruit European cultivars at different maturation steps: Extraction, HPLC-DAD analyses, and biological evaluation. Antioxidants 2019, 8, 562. [Google Scholar] [CrossRef]
- Fonsatti, E.; Del Vecchio, L.; Altomonte, M.; Sigalotti, L.; Nicotra, M.R.; Coral, S.; Natali, P.G.; Maio, M. Endoglin: An accessory component of the TGF-beta-binding receptor complex with diagnostic, prognostic, and bioimmunotherapeutic potential in human malignancies. J. Cell. Physiol. 2001, 188, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Ueda, Y.; Richmond, A. NF-κB activation in melanoma. Pigment. Cell. Res. 2006, 19, 112–124. [Google Scholar] [CrossRef] [PubMed]
- Breithaupt, D.E.; Weller, P.; Wolters, M.; Hahn, A. Comparison of plasma responses in human subjects after the ingestion of 3R, 3R9-zeaxanthin dipalmitate from wolfberry (Lycium barbarum) and non-esterified 3R, 3R9-zeaxanthin using chiral high-performance liquid chromatography. Br. J. Nutr. 2004, 91, 707–713. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.C.; Chang, S.C.; Inbaraj, B.S.; Chen, B.H. Isolation of carotenoids, flavonoids and polysaccharides from Lycium barbarum L. and evaluation of antioxidant activity. Food Chem. 2010, 120, 184–192. [Google Scholar] [CrossRef]
- Krinsky, N.I.; Johnson, E.J. Carotenoid actions and their relation to health and disease. Mol. Asp. Med. 2005, 26, 459–516. [Google Scholar] [CrossRef] [PubMed]
- Bunea, A.; Socaciu, C.; Pintea, A. Xanthophyll esters in fruits and vegetables. Not. Bot. Horti. Agrobo. 2014, 42, 310–324. [Google Scholar] [CrossRef]
- Cheng, C.Y.; Chung, W.Y.; Szeto, Y.T.; Benzie, I.F.F. Fasting plasma zeaxanthin response to Fructus barbarum L. (wolfberry; Kei Tze) in a food-based human supplementation trial. Br. J. Nutr. 2005, 93, 123–130. [Google Scholar] [CrossRef]
- Granado, F.; Olmedilla, B.; Gil-Martinez, E.; Blanco, I. Lutein ester in serum after lutein supplementation in human subjects. Br. J. Nutr. 1998, 80, 445–449. [Google Scholar] [CrossRef]
- Wingerath, T.; Sies, H.; Stahl, W. Xanthophyll esters in human skin. Arch Biochem. Biophys. 1998, 355, 271–274. [Google Scholar] [CrossRef]
- Chitchumroonchokchai, C.; Failla, M.L. Hydrolysis of Zeaxanthin Esters by Carboxyl Ester Lipase during Digestion Facilitates Micellarization and Uptake of the Xanthophyll by Caco-2 Human Intestinal Cells. J. Nutr. 2006, 136, 588–594. [Google Scholar] [CrossRef]
- Valluru, M.; Staton, C.A.; Reed, M.W.R.; Brown, N.J. Transforming Growth Factor-β and Endoglin Signaling Orchestrate Wound Healing. Front. Physiol. 2011, 2, 89. [Google Scholar] [CrossRef] [PubMed]
- Tesic, N.; Kamensek, U.; Sersa, G.; Kranjc, S.; Stimac, M.; Lampreht, U.; Preat, V.; Vandermeulen, G.; Butinar, M.; Turk, B.; et al. Endoglin (CD105) Silencing Mediated by shRNA Under the Control of Endothelin-1 Promoter for Targeted Gene Therapy of Melanoma. Mol. Ther. Nucleic Acids 2015, 4, 239. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Gómez, E.; Villa-Morales, M.; Santos, J.; Fernández-Piqueras, J.; Gamallo, C.; Dotor, J.; Bernabéu, C.; Quintanilla, M. A Role for endoglin as a suppressor of malignancy during mouse skin carcinogenesis. Cancer Res. 2007, 67, 10268–10277. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Ding, Q.Q.; Gao, S.S.; Yang, H.J.; Wang, M.; Shi, Y.; Cheng, B.F.; Bi, J.J.; Feng, Z.W. PRDM5 promotes the proliferation and invasion of murine melanoma cells through up-regulating JNK expression. Cancer Med. 2016, 5, 2558–2566. [Google Scholar] [CrossRef][Green Version]
- Yu, T.; Ji, J.; Guo, Y.L. MST1 activation by curcumin mediates JNK activation, Foxo3a nuclear translocation and apoptosis in melanoma cells. Biochem. Biophys. Res. Commun. 2013, 441, 53–58. [Google Scholar] [CrossRef]
- Braicu, C.; Buse, M.; Busuioc, C.; Drula, R.; Gulei, D.; Raduly, L.; Rusu, A.; Irimie, A.; Atanasov, A.G.; Slaby, O.; et al. A comprehensive review on MAPK: A promising therapeutic target in cancer. Cancers 2019, 11, 1618. [Google Scholar] [CrossRef]
- Sheng, Y.N.; Luo, Y.H.; Liu, S.B.; Xu, W.T.; Zhang, Y.; Zhang, T.; Xue, H.; Zuo, W.B.; Li, Y.N.; Wang, C.Y.; et al. Zeaxanthin induces apoptosis via ROS-regulated MAPK and AKT signaling pathway in human gastric cancer cells. Oncol. Targets Ther. 2020, 13, 10995. [Google Scholar] [CrossRef]
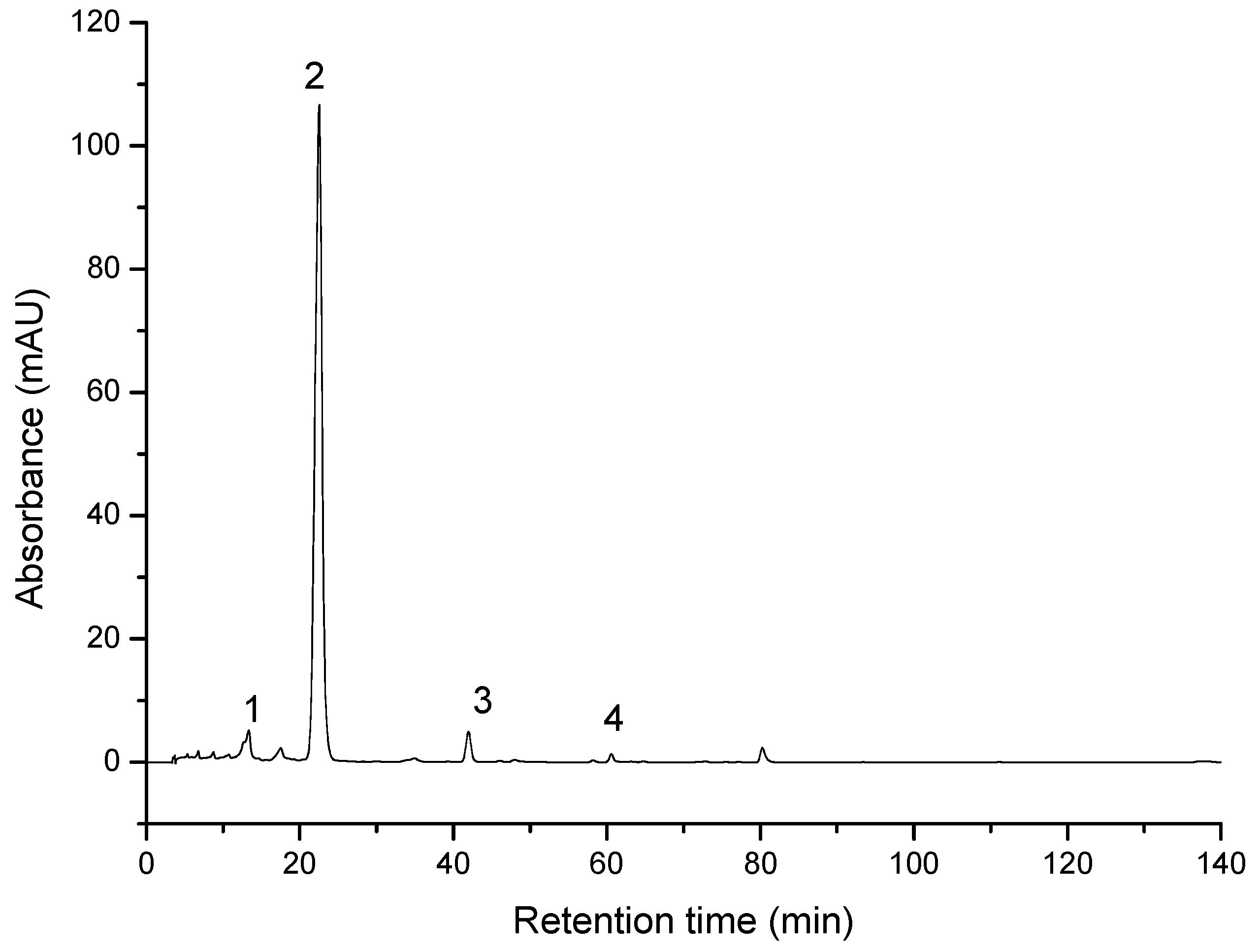
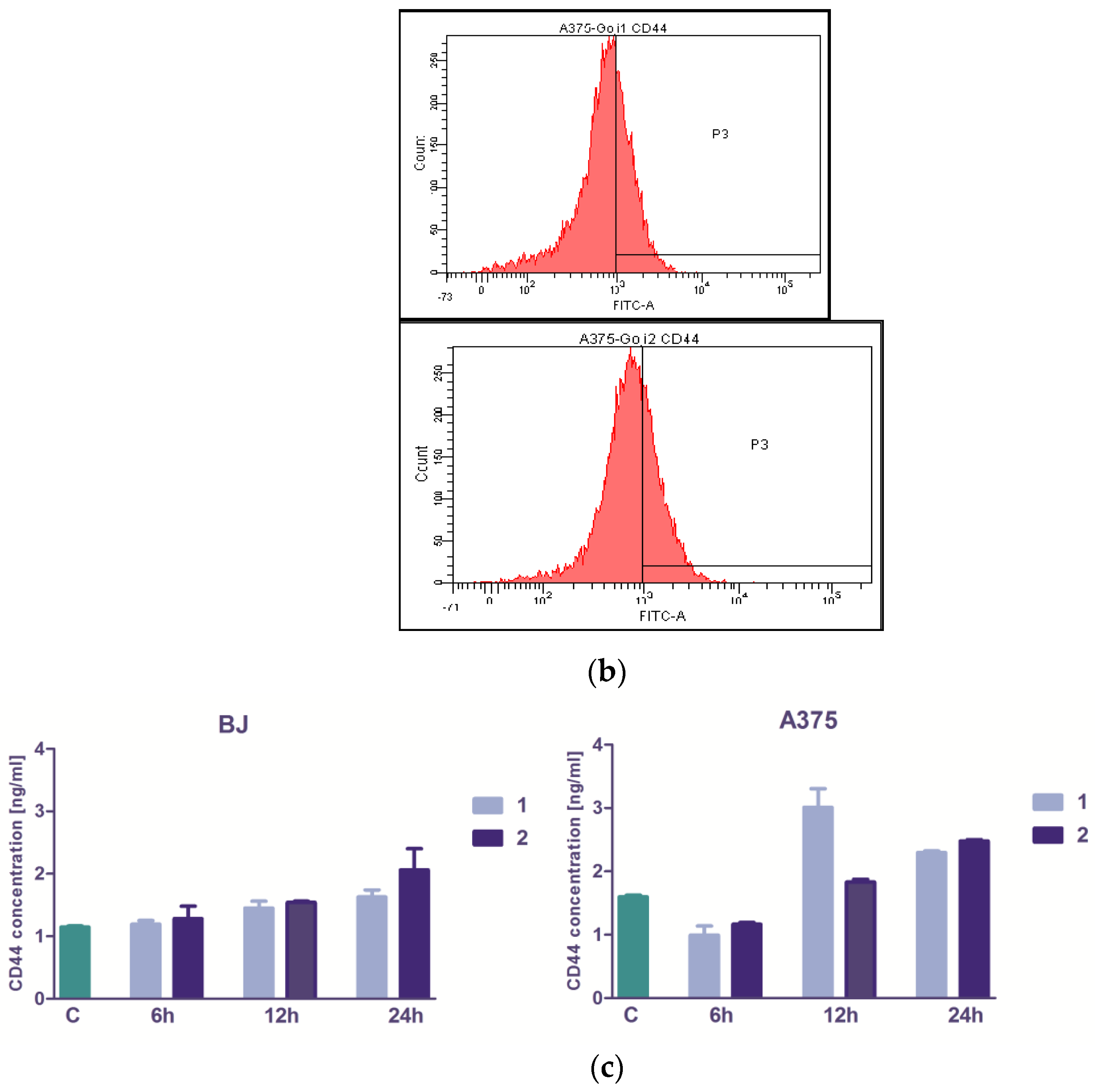
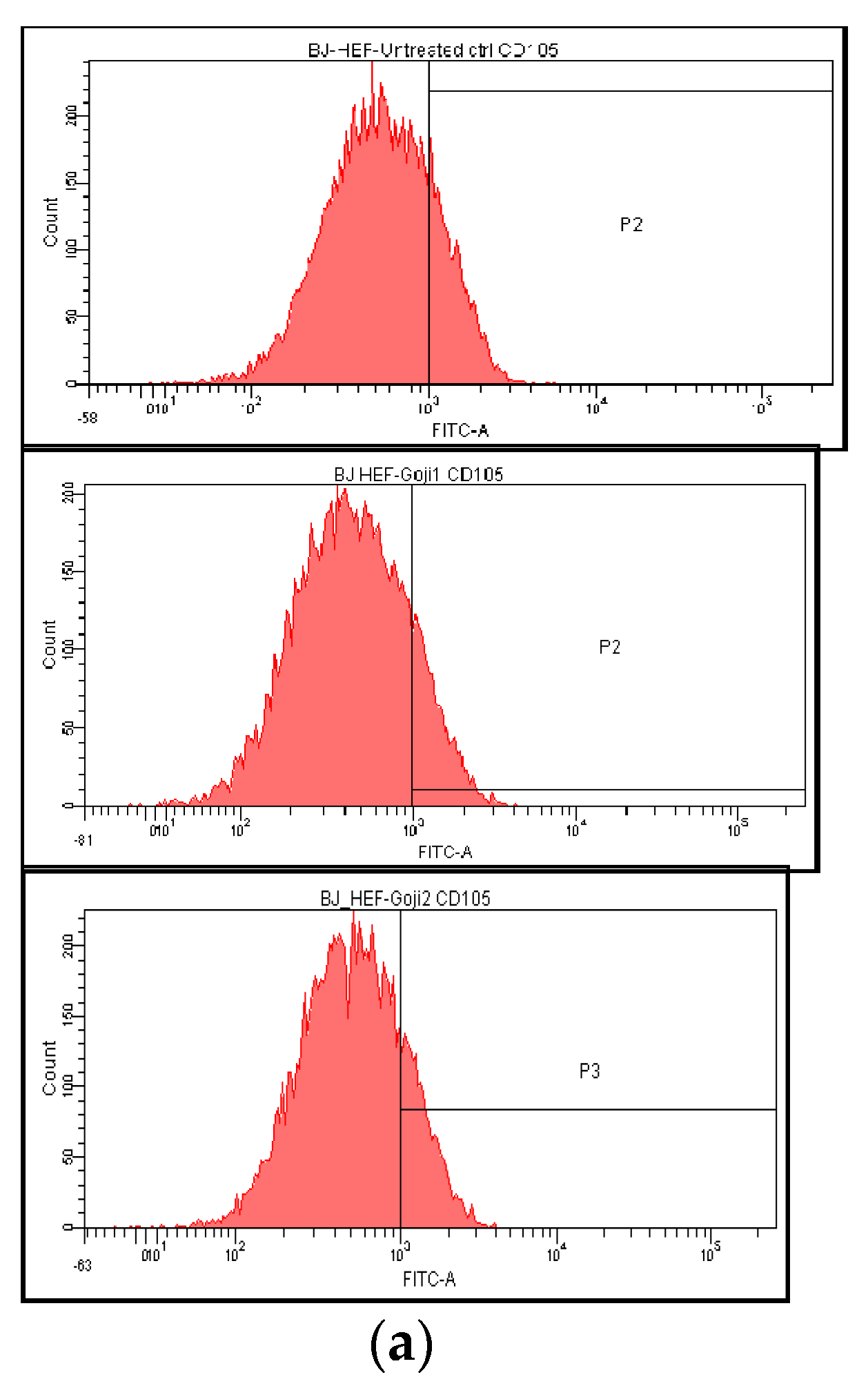
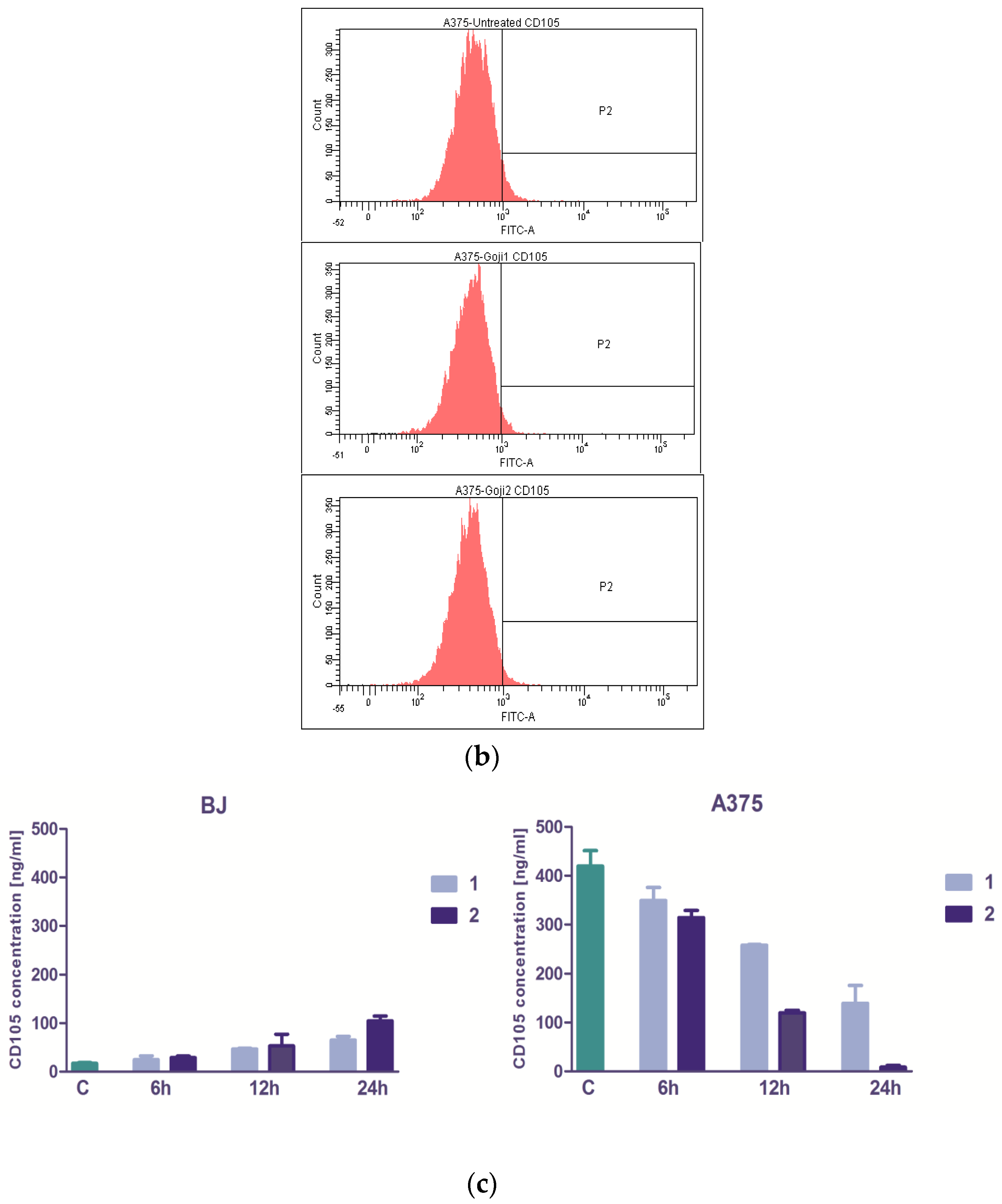
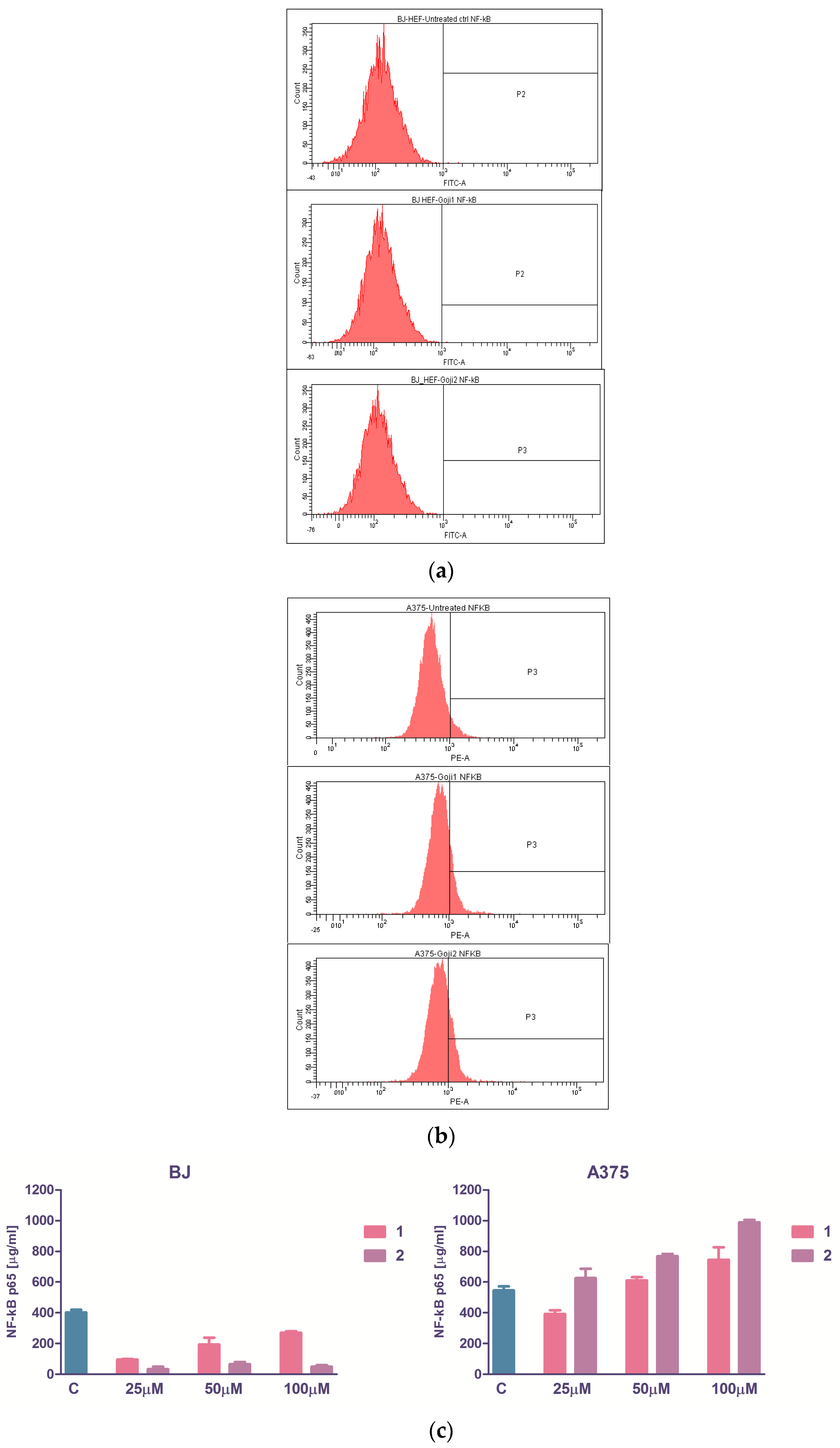
| ID | Compound | Retention Time | UV-Vis Maxima | Sample 1 mg/100 g FW | Sample 2 mg/100 g FW |
|---|---|---|---|---|---|
| 1 | Neoxanthin | 14.6 | 416, 439, 468 | 0.70 ± 0.14 | 0.75 ± 0.16 |
| 2 | Zeaxanthin | 22.1 | 425, 450, 476 | 23.44 ± 1.02 | 23.69 ± 1.13 |
| 3 | β-cryptoxanthin | 41.9 | 425, 451, 476 | 0.60 ± 0.15 | 0.49 ± 0.14 |
| 4 | β-carotene | 60.6 | 421, 452, 477 | 0.07 ± 0.02 | 0.07 ± 0.02 |
| IC50 Values (μM) | |||||
|---|---|---|---|---|---|
| Cell Line | BJ (CRL-2522) | A375 | |||
| 1 | 24 h | 75.15 ± 0.23 | 62.36 ± 0.08 | ||
| 2 | 24 h | 85.06 ± 11.34 | 92.59 ± 6.71 | ||
| Inhibition of Proliferation | |||||
| Cell line | BJ (CRL-2522) | A375 | |||
| Hillslope | p value | Hillslope | p value | ||
| 1 | 6 h | −4726 ± 905.0 | 0.0002 | 459.6 ± 228.7 | 0.0675 |
| 12 h | −4274 ± 822.5 | 0.0002 | 389.2 ± 301.3 | 0.2208 | |
| 24 h | −2754 ± 606.8 | 0.0007 | −654.8 ± 182.8 | 0.0038 | |
| 2 | 6 h | −4179 ± 877.0 | 0.0005 | −3166 ± 576.8 | 0.0001 |
| 12 h | −3706 ± 807.5 | 0.0006 | −3076 ± 482.9 | <0.0001 | |
| 24 h | −2253 ± 582.5 | 0.0022 | −2109 ± 345.0 | <0.0001 | |
| Cells and Treatments | CD44 | CD105 | NF-kβ | JNK | p38 | ERK |
|---|---|---|---|---|---|---|
| A375 | ||||||
| Untreated | 50.1 ± 0.40 | 4.0 ± 0.02 | 5.8 ± 0.05 | 14.3 ± 0.03 | 14.1 ± 0.01 | 29.2 ± 0.11 |
| Extract 1 | 33.4 ± 0.16 | 2.7 ± 0.01 | 14.6 ± 0.02 | 30.7 ± 0.05 | 19.4 ± 0.08 | 34.5 ± 0.14 |
| Extract 2 | 32.2 ± 0.18 | 2.0 ± 0.00 | 18.5 ± 0.12 | 29.3 ± 0.15 | 20.0 ± 0.04 | 42.4 ± 0.27 |
| BJ | ||||||
| Untreated | 89.5 ± 0.10 | 18.8 ± 0.03 | 0.02 ± 0.00 | 40.0 ± 0.14 | 35.4 ± 0.25 | 30.9 ± 0.13 |
| Extract 1 | 93.8 ± 0.02 | 13.7 ± 0.12 | 0.01 ± 0.00 | 27.4 ± 0.05 | 32.3 ± 0.04 | 25.9 ± 0.07 |
| Extract 2 | 93.5 ± 0.05 | 17.7 ± 0.07 | 0.05 ± 0.00 | 43.7 ± 0.11 | 25.4 ± 0.02 | 25.1 ± 0.06 |
| Mitogen Activated Protein Kinases (MAPK) | Cell Line | BJ | A375 | ||
|---|---|---|---|---|---|
| Treatment | 1 | 2 | 1 | 2 | |
| JNK | ↓ 31.68% | ↑ 9.12% | ↑ 114.40% | ↑ 104.61% | |
| ERK | ↓ 16.02% | ↓ 18.72% | ↑ 18.28% | ↑ 45.42% | |
| p38 | ↓ 9.00% | ↓ 28.41% | ↑ 36.75% | ↑ 41.63% | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cenariu, D.; Fischer-Fodor, E.; Țigu, A.B.; Bunea, A.; Virág, P.; Perde-Schrepler, M.; Toma, V.-A.; Mocan, A.; Berindan-Neagoe, I.; Pintea, A.; et al. Zeaxanthin-Rich Extract from Superfood Lycium barbarum Selectively Modulates the Cellular Adhesion and MAPK Signaling in Melanoma versus Normal Skin Cells In Vitro. Molecules 2021, 26, 333. https://doi.org/10.3390/molecules26020333
Cenariu D, Fischer-Fodor E, Țigu AB, Bunea A, Virág P, Perde-Schrepler M, Toma V-A, Mocan A, Berindan-Neagoe I, Pintea A, et al. Zeaxanthin-Rich Extract from Superfood Lycium barbarum Selectively Modulates the Cellular Adhesion and MAPK Signaling in Melanoma versus Normal Skin Cells In Vitro. Molecules. 2021; 26(2):333. https://doi.org/10.3390/molecules26020333
Chicago/Turabian StyleCenariu, Diana, Eva Fischer-Fodor, Adrian Bogdan Țigu, Andrea Bunea, Piroska Virág, Maria Perde-Schrepler, Vlad-Alexandru Toma, Andrei Mocan, Ioana Berindan-Neagoe, Adela Pintea, and et al. 2021. "Zeaxanthin-Rich Extract from Superfood Lycium barbarum Selectively Modulates the Cellular Adhesion and MAPK Signaling in Melanoma versus Normal Skin Cells In Vitro" Molecules 26, no. 2: 333. https://doi.org/10.3390/molecules26020333
APA StyleCenariu, D., Fischer-Fodor, E., Țigu, A. B., Bunea, A., Virág, P., Perde-Schrepler, M., Toma, V.-A., Mocan, A., Berindan-Neagoe, I., Pintea, A., Crișan, G., Cenariu, M., & Maniu, A. (2021). Zeaxanthin-Rich Extract from Superfood Lycium barbarum Selectively Modulates the Cellular Adhesion and MAPK Signaling in Melanoma versus Normal Skin Cells In Vitro. Molecules, 26(2), 333. https://doi.org/10.3390/molecules26020333















