Anisotropic Photoluminescence of Poly(3-hexyl thiophene) and Their Composites with Single-Walled Carbon Nanotubes Highly Separated in Metallic and Semiconducting Tubes
Abstract
1. Introduction
2. Results and Discussion
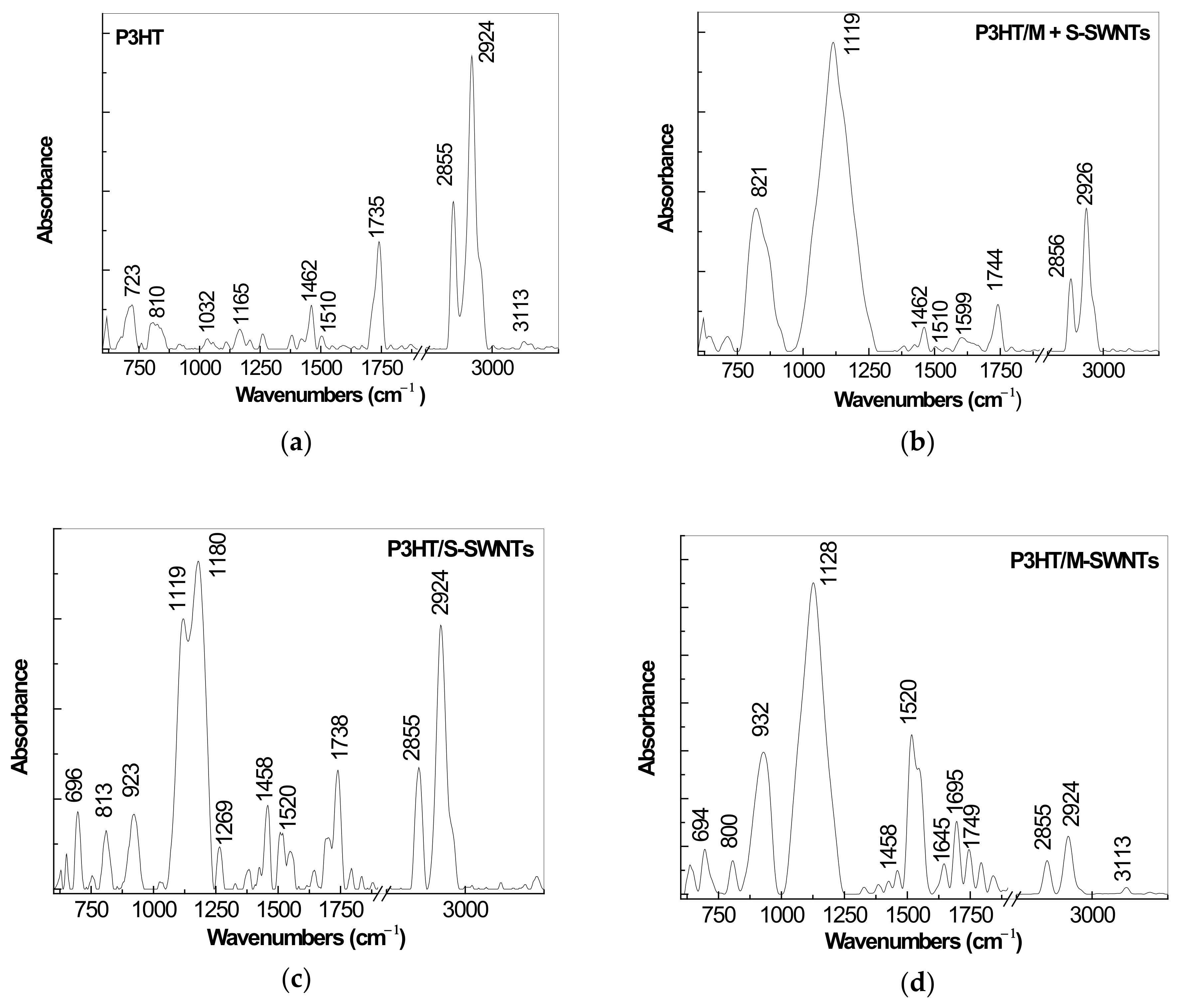
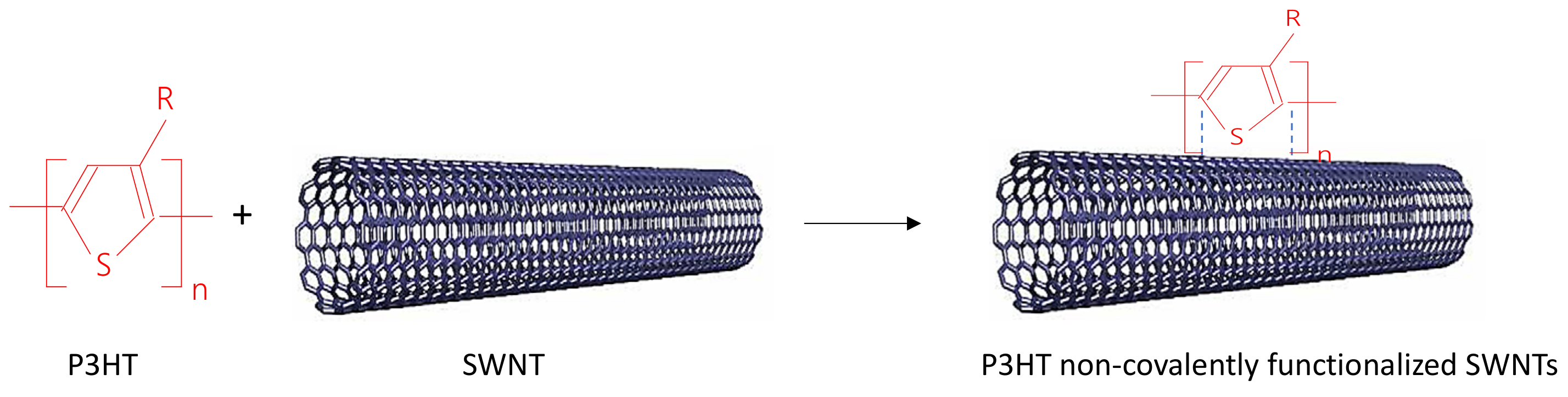
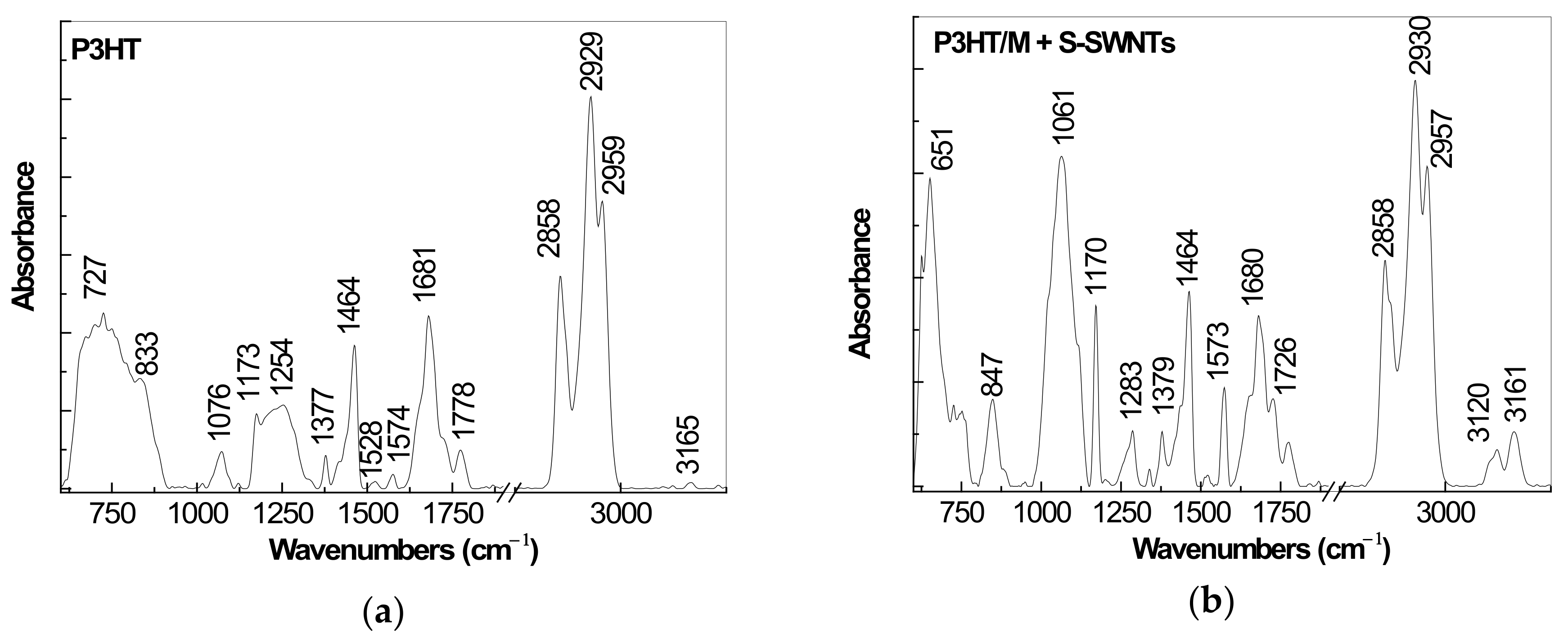
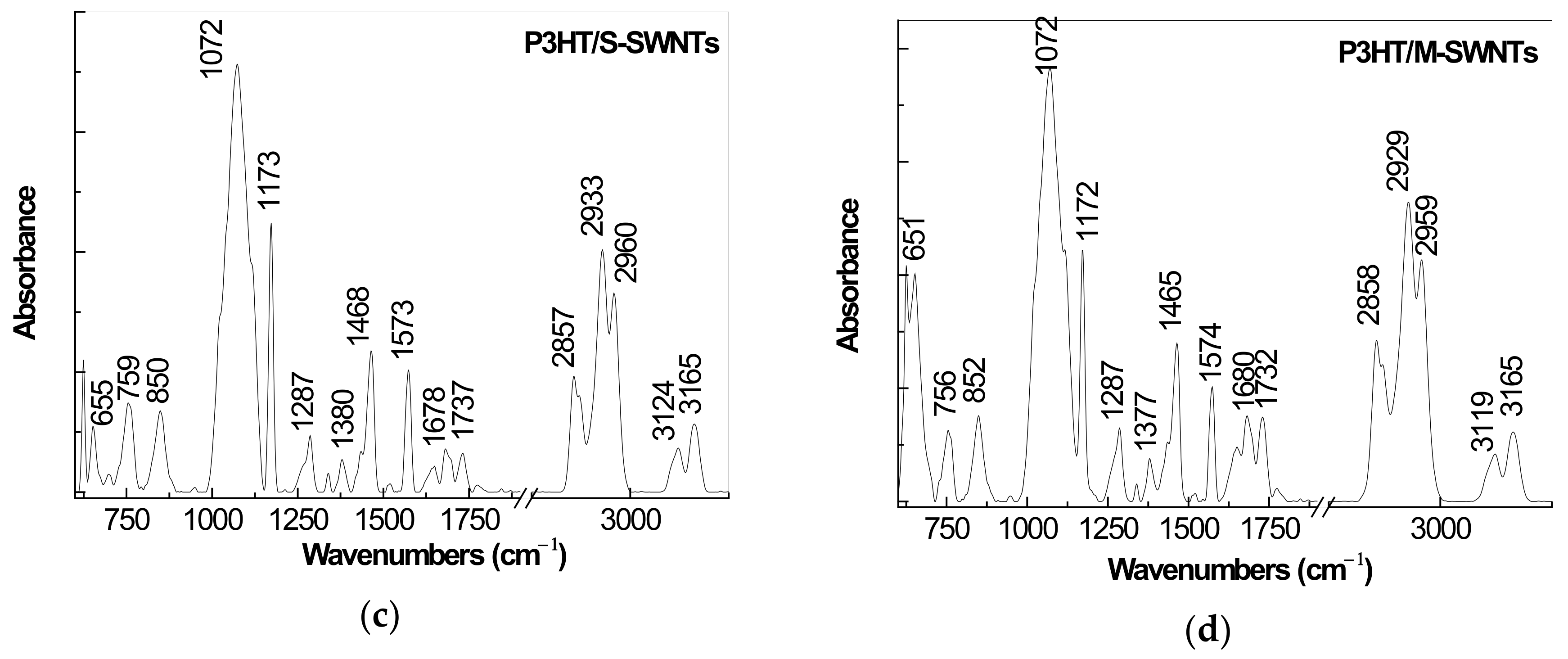
2.1. IR Spectra of P3HT and Its Composites with CNTs Prepared by Chemical and Electrochemical Methods
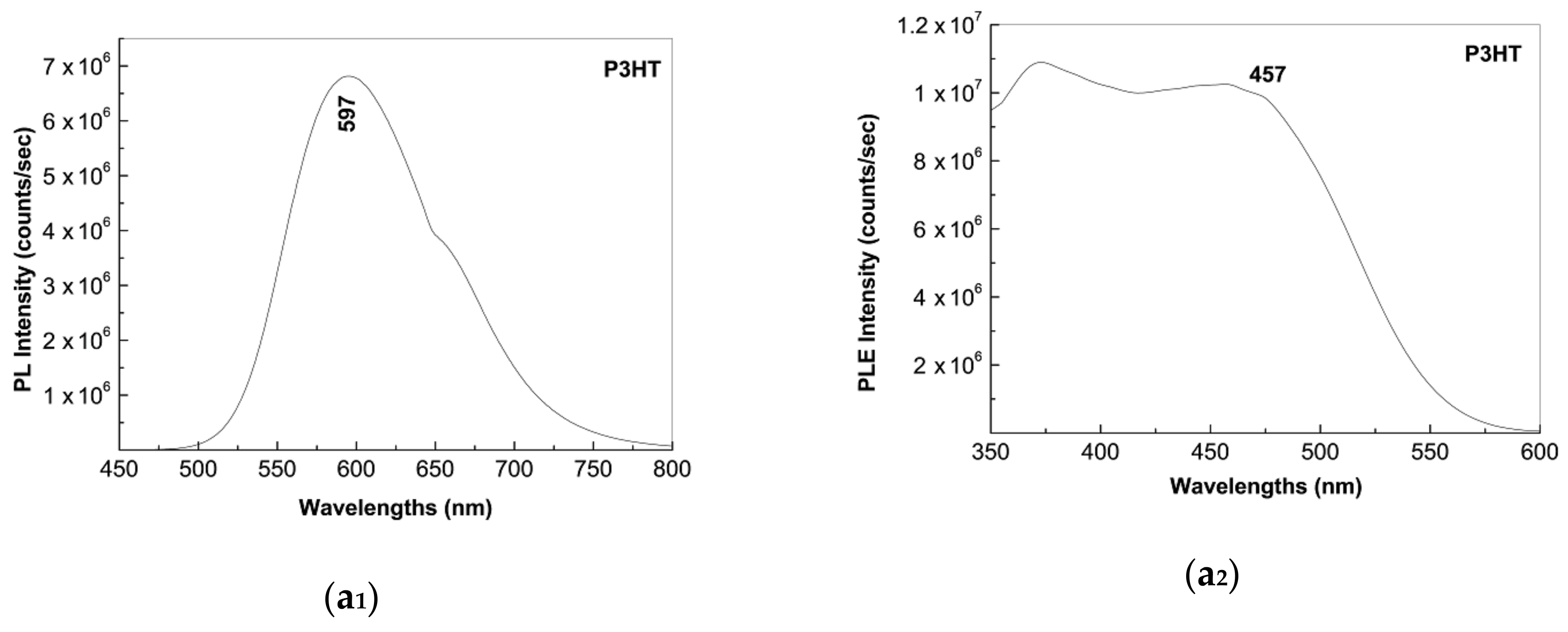
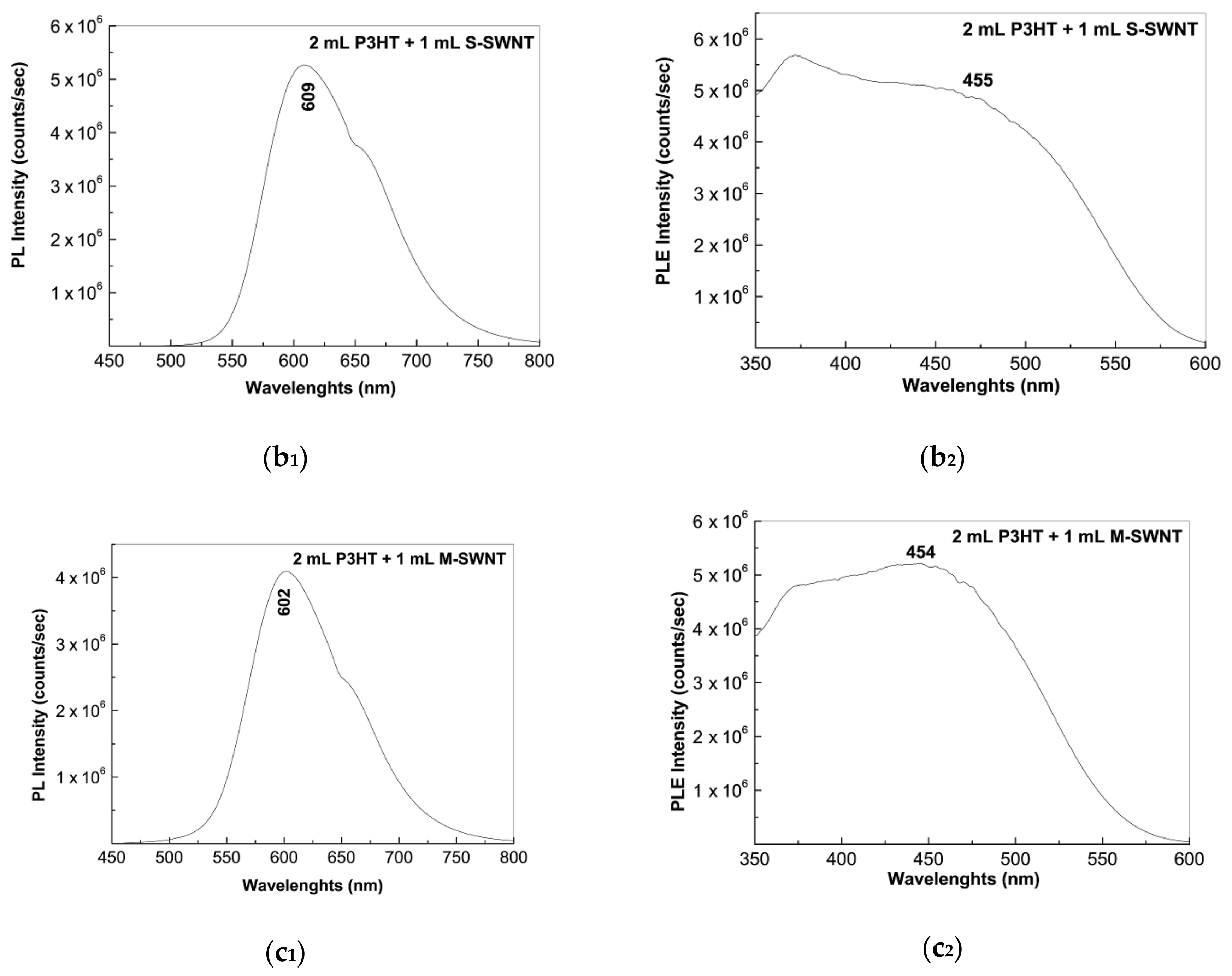
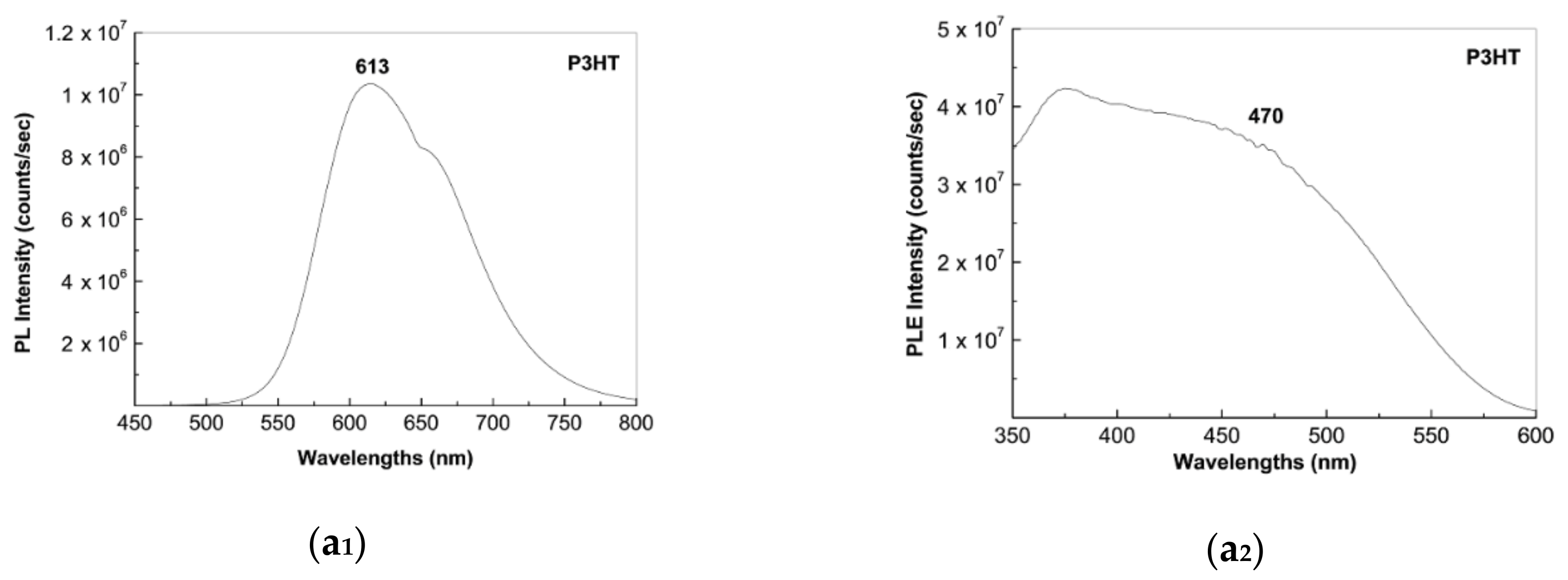
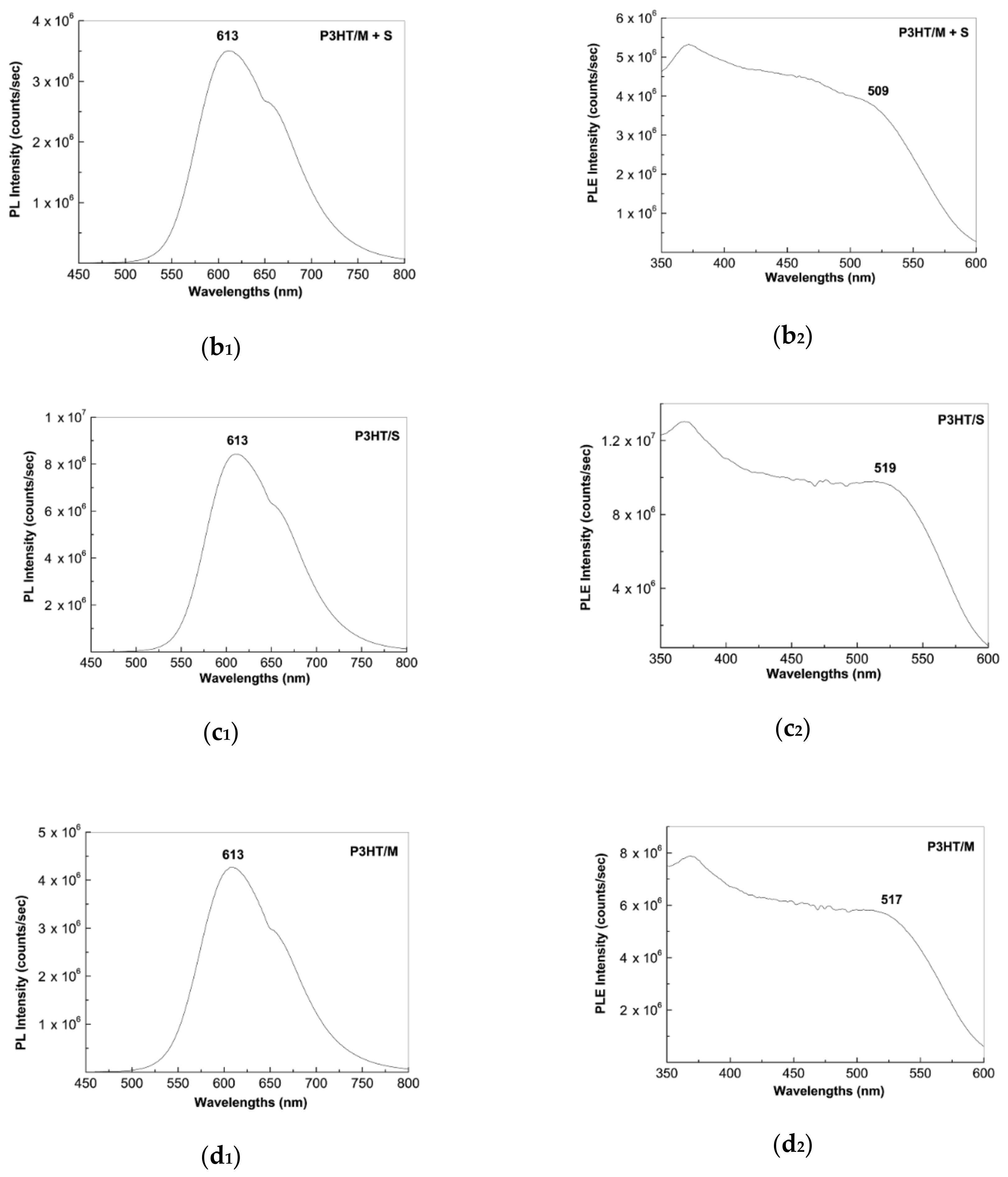
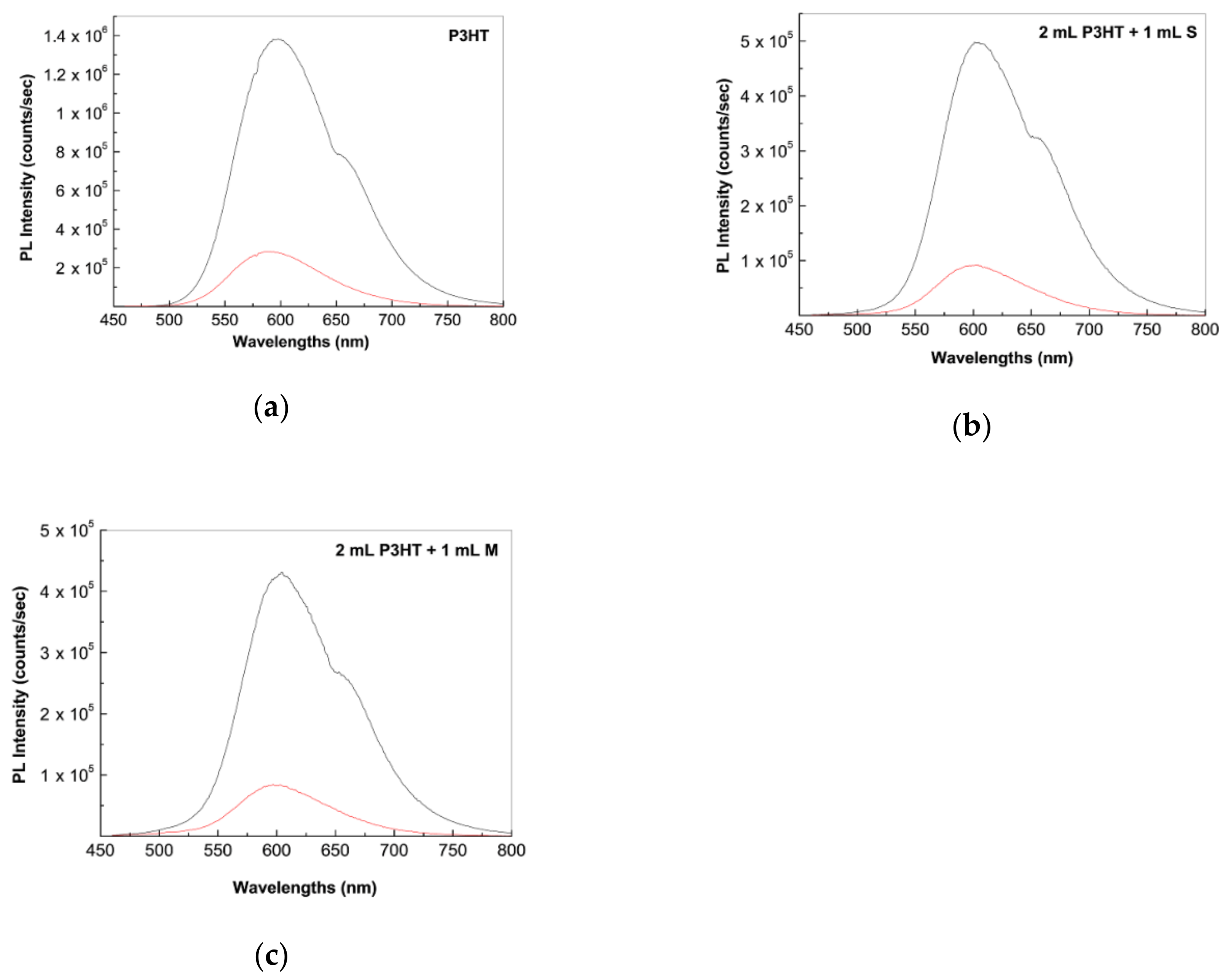
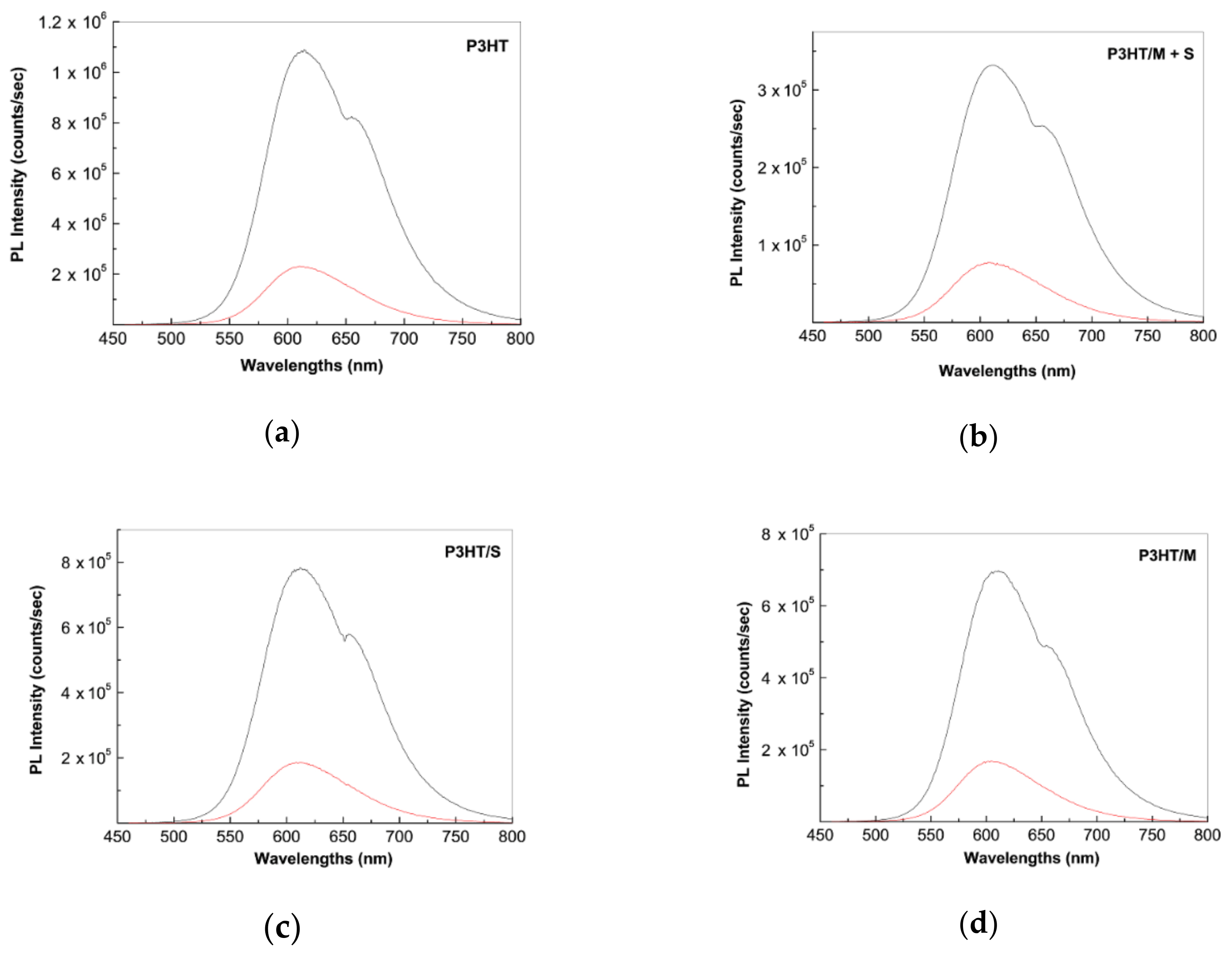
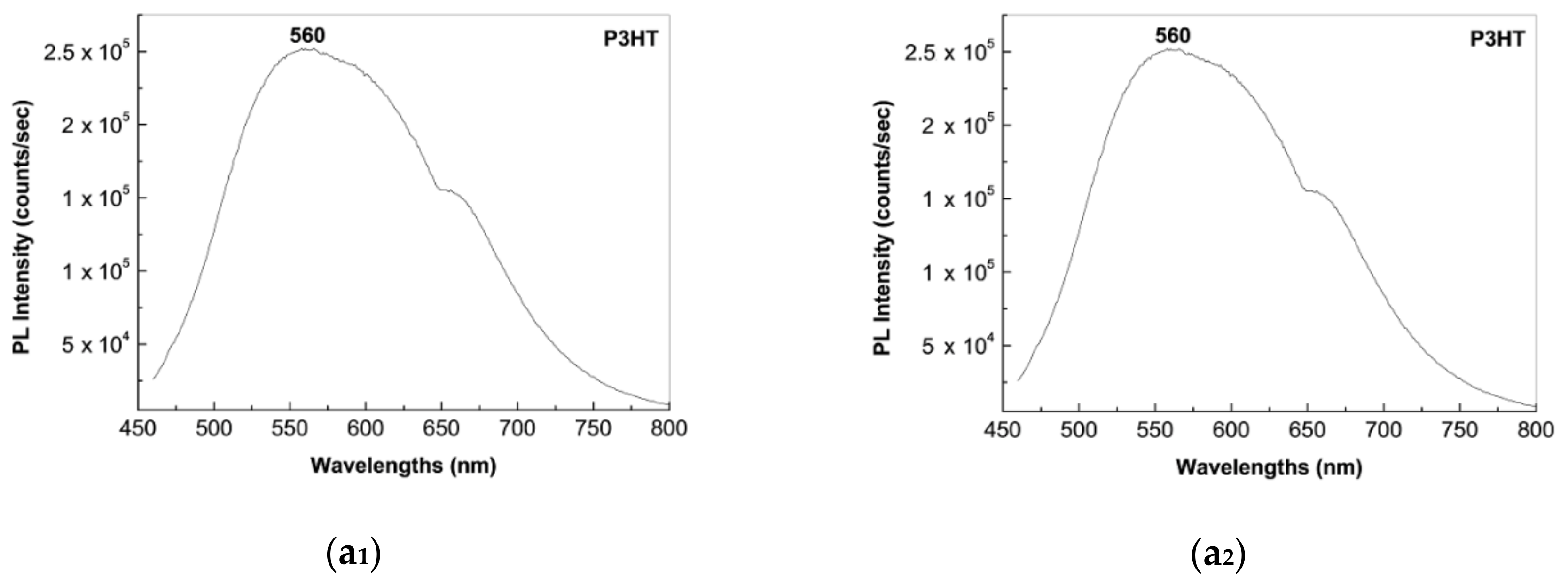
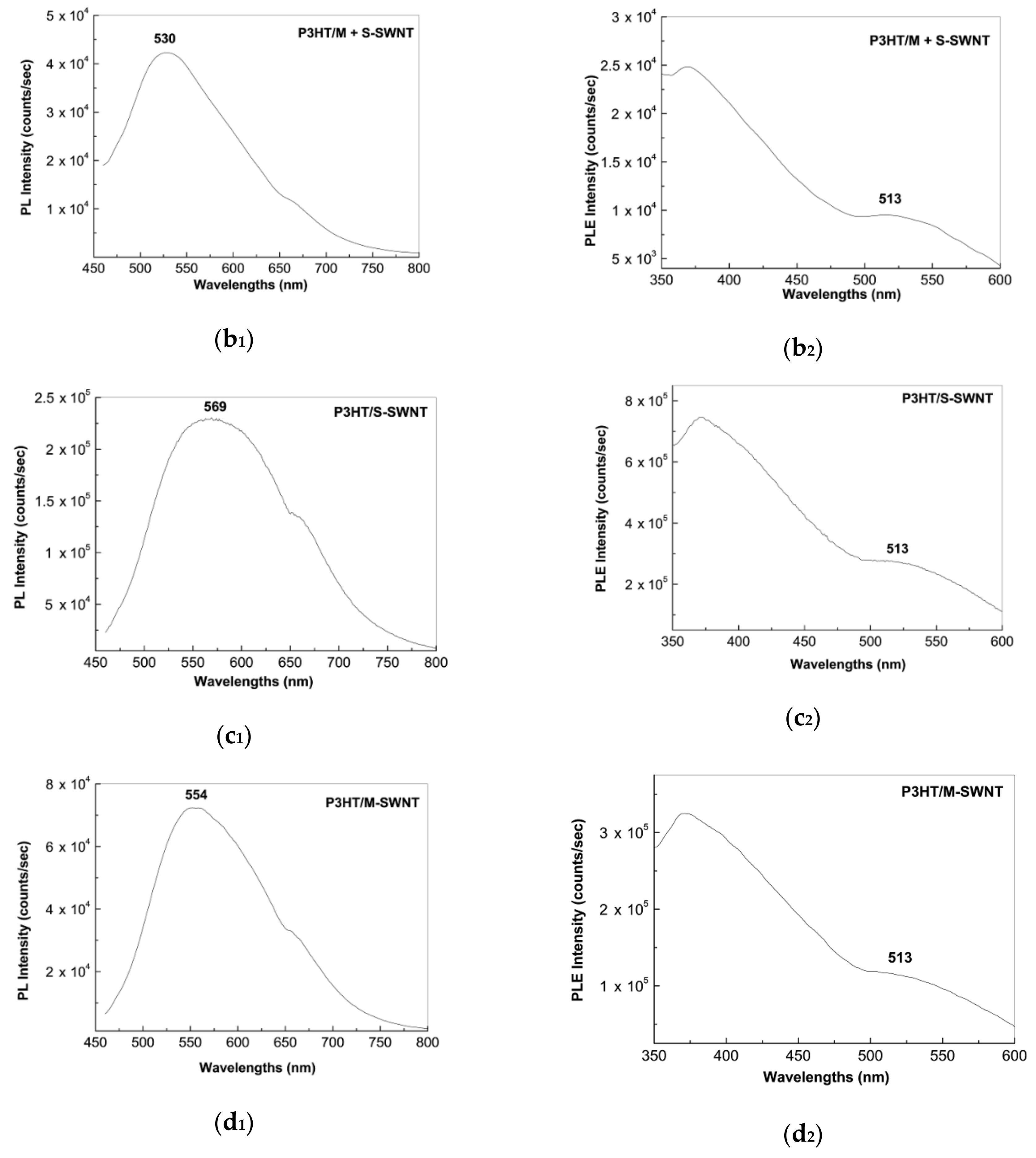
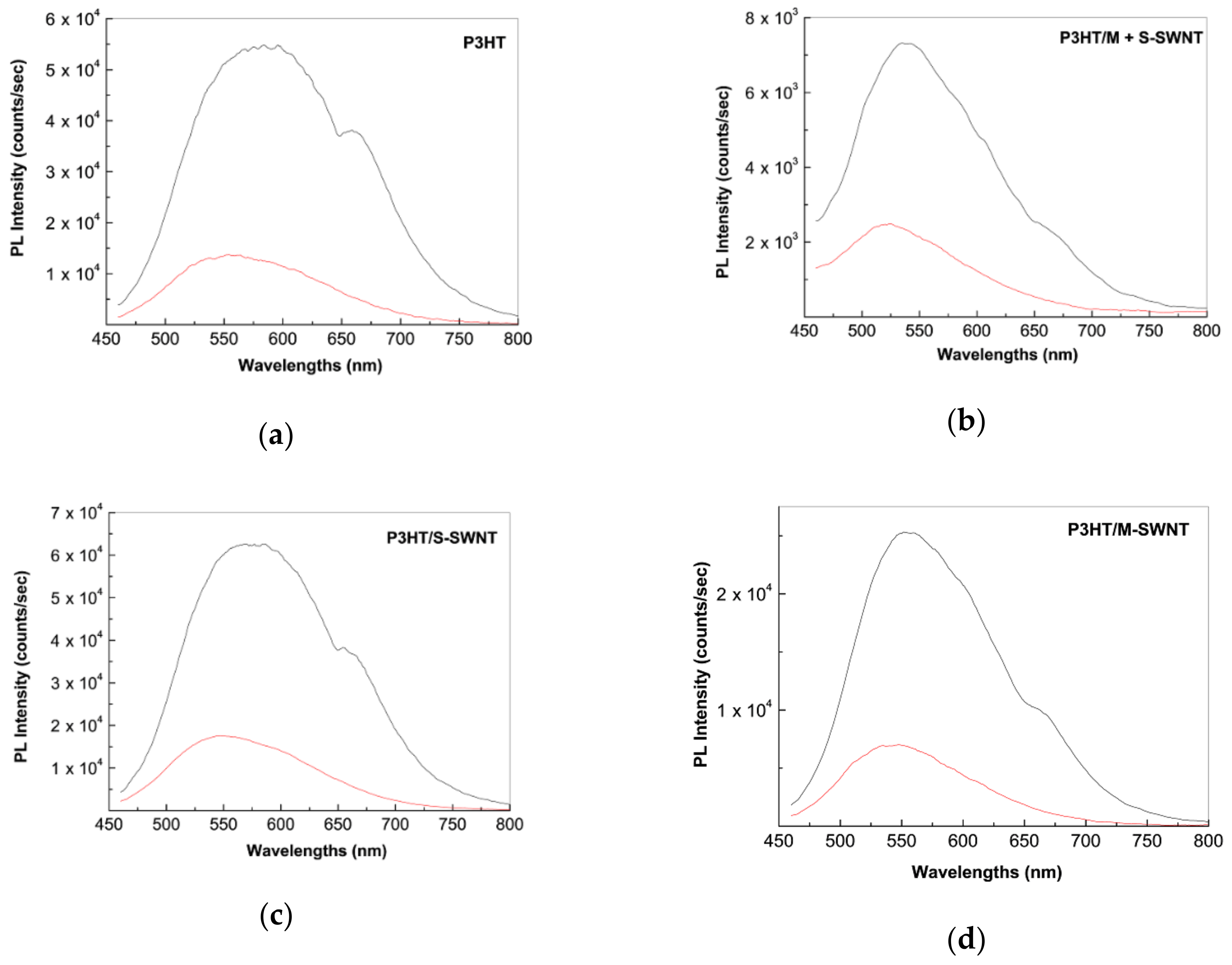
2.2. Photoluminescence of the P3HT/CNTs Composites Prepared by Chemical and Electrochemical Methods
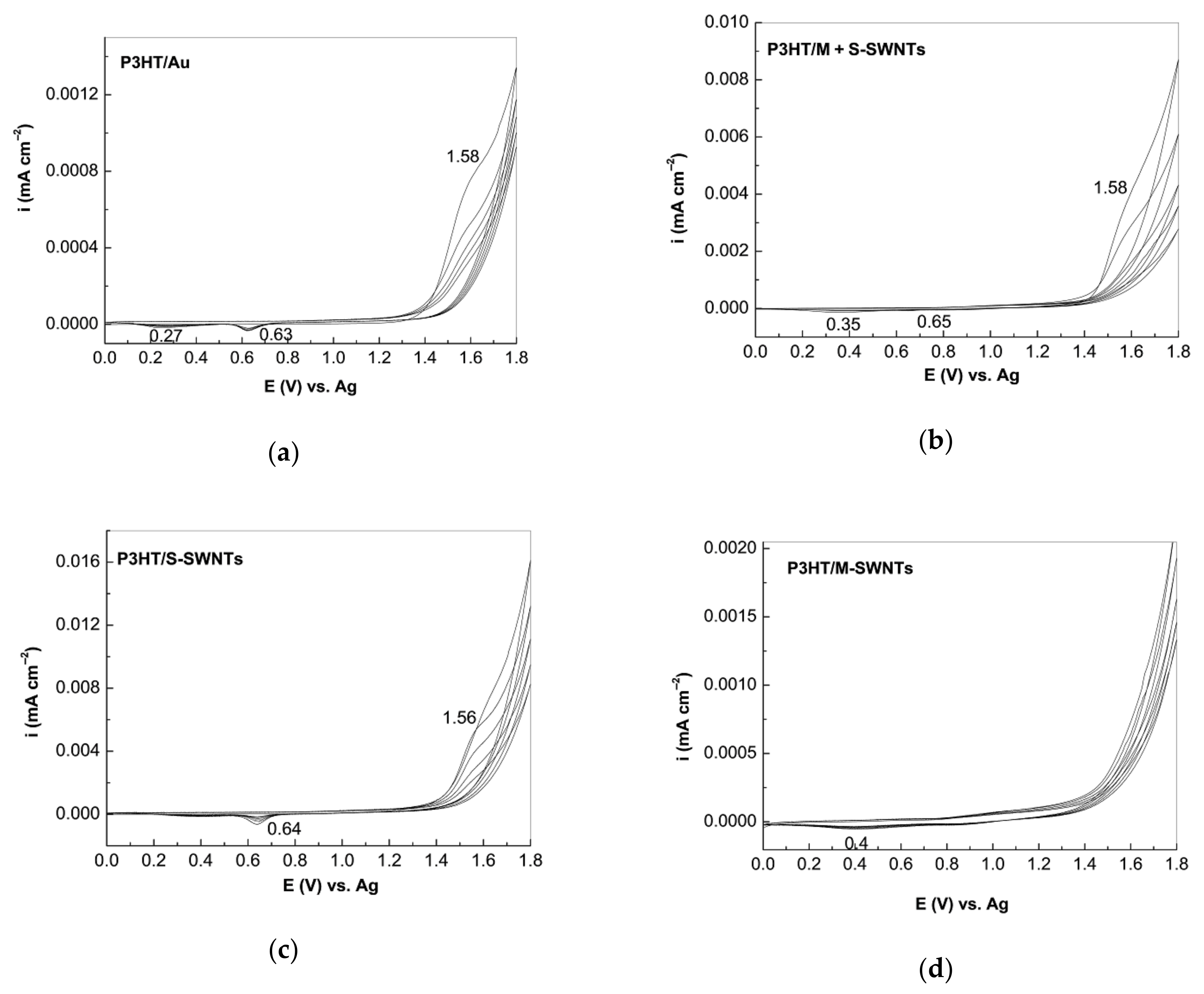
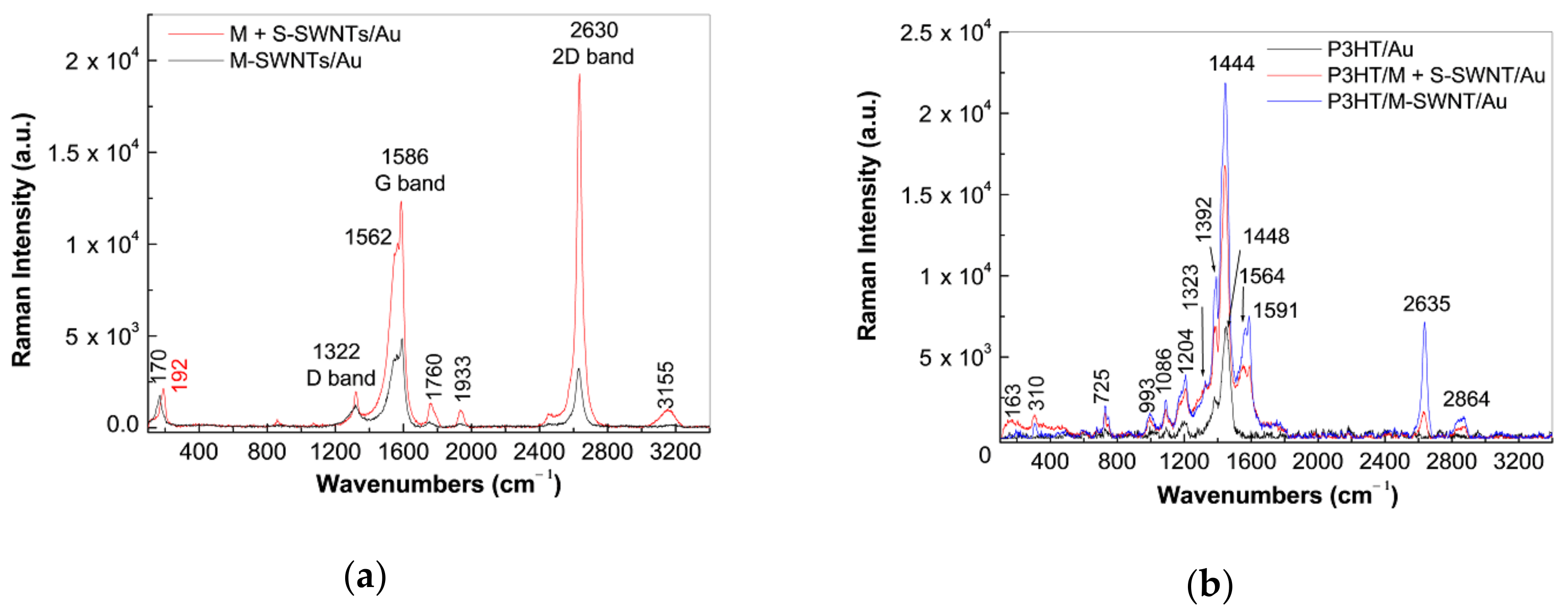
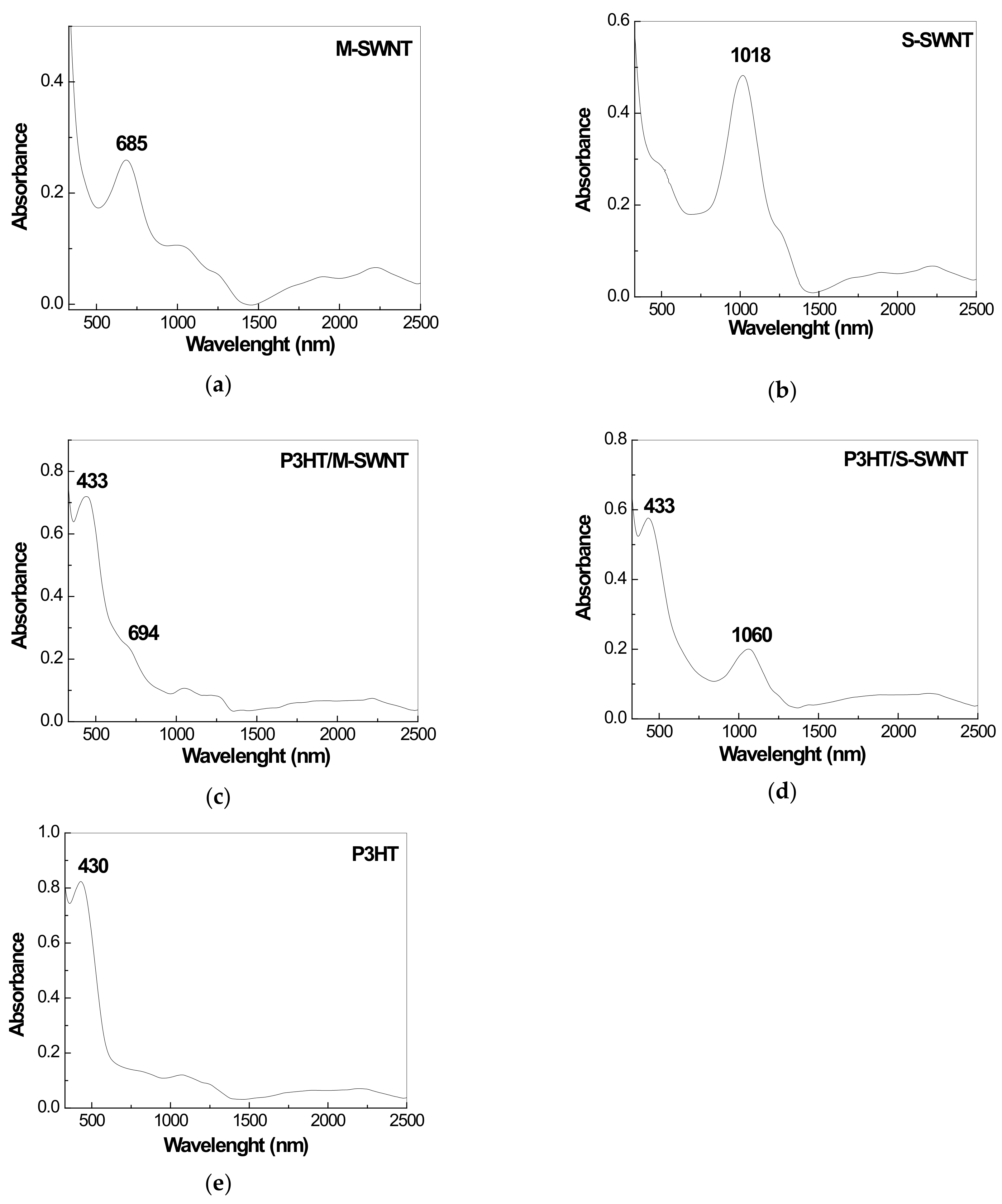
2.3. The SERS and UV-VIS-NIR Spectra of the P3HT/CNTs Composites
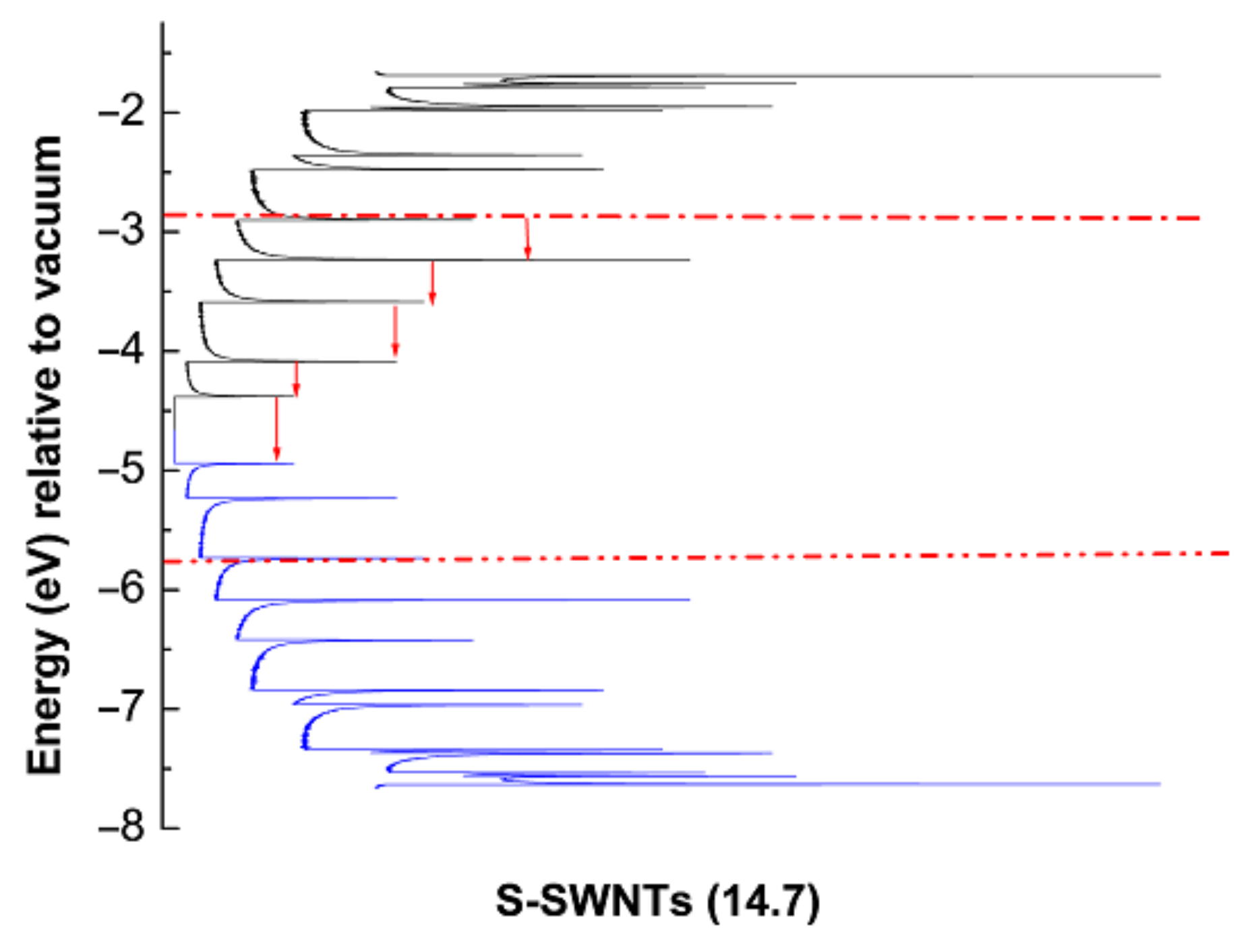
2.4. The Mechanism of P3HT PL Quenching Process
3. Materials and Methods
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Zhang, Y.; Zhang, X.; Shi, Y.; Sun, C.; Zhou, N.; Wen, H. The synthesis and functional study of multicolor nitrogen-doped carbon dots for live cell nuclear imaging. Molecules 2020, 25, 306. [Google Scholar] [CrossRef] [PubMed]
- Muhulet, A.; Miculescu, F.; Voicu, S.I.; Schutt, F.; Thakur, V.K.; Mishra, Y.K. Fundamentals and scopes of doped carbon nanotubes towards energy and biosensing applications. Mater. Today Energy 2018, 9, 154–186. [Google Scholar] [CrossRef]
- Jariwala, D.; Sangwan, V.K.; Lauhon, L.J.; Marks, T.J.; Hersam, M.C. Carbon nanomaterials for electronics, optoelectronics, photovoltaics and sensing. Chem. Soc. Rev. 2013, 42, 2824–2860. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Xie, Z.; Yeow, J.T.W. Review-state-of-art organic solar cells based on carbon nanotubes and graphene. ECS J. Solid State Sci. Technol. 2020, 9, 105004. [Google Scholar] [CrossRef]
- Bhatia, R.; Kumar, L. Functionalized carbon nanotube doping of P3HT: PCBM photovoltaic devices for enhancing short circuit current and efficiency. J. Saudi Chem. Soc. 2017, 21, 366–376. [Google Scholar] [CrossRef]
- Agbolaghi, S.; Charoughchi, S.; Aghapour, S.; Abbasi, F.; Bahadori, A.; Sarvari, R. Bulk heterojunction photovoltaics with iproved efficiencies using stem-leaf, shish-kebab and double-fibrillar nano-hybrids based on modified carbon nanotubes and poly(3-hexylthiophene). Sol. Energy 2018, 170, 138–150. [Google Scholar] [CrossRef]
- Nganou, C.; Lackner, G.; Teschome, B.; Deen, M.J.; Adir, N.; Pouhe, D.; Lupascu, D.C.; Mkandawire, M. Energy transfer kinetics in photosynthesis as an inspiration for improving organic solar cells. ACS Appl. Mater. Interfaces 2017, 9, 19030–19039. [Google Scholar] [CrossRef]
- Lanzi, M.; Di-Nicola, F.P.; Errani, F.; Paganin, L.; Mucci, A. Soventless deposition of oligo- and polythiophenes for bulk heterojunction solar cells. Synth. Met. 2014, 195, 61–68. [Google Scholar] [CrossRef]
- Saha, S.; Singh, J.P.; Saha, U.; Goswami, T.H.; Rao, K.U.B. Structure-property relationship of SELF-sustained homogeneous ternary nanocomposites: Key issues to evaluate properties of rrP3HT wrapped MWNT dispersed in TPU. Comp. Sci. Technol. 2011, 71, 397–405. [Google Scholar] [CrossRef]
- Lefrant, S.; Baibarac, M.; Baltog, I. Raman and FTIR spectroscopy as valuable tools for the characterization of polymer and carbon nanotube based composites. J. Mater. Chem. 2009, 19, 5690–5704. [Google Scholar] [CrossRef]
- Baibarac, M.; Baltog, I.; Lefrant, S. Recent progress in synthesis, vibrational characterization and applications trend of conjugated polymers/carbon nanotubes composites. Curr. Org. Chem. 2011, 15, 1160–1196. [Google Scholar] [CrossRef][Green Version]
- Hamzah, N.; Yasin, M.F.M.; Yusop, M.Z.M.; Saat, A.; Subha, N.A.M. Rapid production of carbon nanotubes: A review on advancement in growth control and morphology manipulations of flame synthesis. J. Mater. Chem. A 2017, 5, 25144–25170. [Google Scholar] [CrossRef]
- Adamska, M.; Narkiewicz, U. Purifiation of carbon nanotubes—A review of methodology. J. Nanosci. Nanotechnol. 2018, 10, 1329–1342. [Google Scholar]
- Maeda, Y.; Kimura, S.; Kanda, M.; Hirashima, Y.; Hasegawa, T.; Wakahara, T.; Lian, Y.F.; Nakahodo, T.; Tsuchiya, T.; Akasaka, T.; et al. Large-scale separation of metallic and semiconducting single-walled carbon nanotubes. J. Am. Chem. Soc. 2005, 127, 10287–10290. [Google Scholar] [CrossRef]
- Baibarac, M.; Nila, A.; Baltog, I.; Lefrant, S.; Mevellec, J.Y.; Quillard, S.; Humbert, B. Influence of single-walled carbon nanotubes enriched in semiconducting and metallic tubes on the electropolymerization of tetrabromo ortho-xylene: Insights on the synthesis mechanism of poly(ortho-phenylenevinylene). Eur. Polym. Sci. 2017, 88, 109–128. [Google Scholar] [CrossRef]
- Muguruma, H.; Hoshino, T.; Nowaki, K. Electronically type-sorted carbon nanotube-based electrochemical biosensors with glucose oxidase and dehydrogenase. ACS Appl. Mater. Interfaces 2015, 7, 584–592. [Google Scholar] [CrossRef]
- Wang, C.; Zhang, J.; Ryu, K.; Badmaev, A.; Gomez De Arco, L.; Zhou, C. Wafer-scale fabrication of separated carbon nanotube thin-film transistors for display applications. Nano Lett. 2009, 9, 4285–4291. [Google Scholar] [CrossRef]
- Debera, G.D.M.R.; Jayawardena, K.D.G.I.; Prabhath, M.R.R.; Yahya, I.; Tan, Y.Y.; Nismy, N.A.; Shiozawa, H.; Sauer, M.; Ruiz-Soria, G.; Ayala, P.; et al. Hybrid carbon nanotube networks as efficient hole extraction layers for organic photovoltaics. ACS Nano 2013, 7, 556–565. [Google Scholar] [CrossRef]
- Baibarac, M.; Matea, A.; Daescu, M.; Mercioniu, I.; Quillard, S.; Mevellec, J.Y.; Lefrant, S. Polyaniline photoluminescence quenching induced by single-walled carbon nanotubes enriched in metallic and semiconducting tubes. Sci. Rep. 2018, 8, 9518. [Google Scholar] [CrossRef]
- Khlaifia, D.; Desert, A.; Barek, M.; Garreau, A.; Mevellec, J.Y.; Massuyeau, F.; Faulques, E.; Alimi, K.; Duvail, J.L. Self-ordering promoted by the nanoconfinement of poly(3-hexylthiophene) and its nanocomposite with single-walled carbon nanotubes. Nanotechnology 2019, 30, 055603. [Google Scholar] [CrossRef]
- Ansari, M.A.; Mohiuddin, S.; Kandemirli, F.; Malik, M.I. Synthesis and characterization of poly(3-hexylthiophene): Improvement of regioregularity and energy band gap. RSC Adv. 2018, 8, 8319–8328. [Google Scholar] [CrossRef]
- Furukawa, Y.; Akimoto, M.; Harada, I. Vibrational key bands and electrical conductivity of polythiophene. Synth. Met. 1987, 18, 151–156. [Google Scholar] [CrossRef]
- Chazaro-Ruiz, L.F.; Kellenberger, A.; Dunsch, L. In situ ESR/UV-VIS-NIR and ATR-FTIR spectroelectrochemical study on the p-doping of copolymers of 3-methylthiophene and 3-hexylthiophene. J. Phys. Chem. B 2009, 113, 2310–2316. [Google Scholar] [CrossRef]
- Kim, U.J.; Liu, X.M.; Furtado, C.A.; Chen, G.; Saito, R.; Jiang, J.; Dresselhaus, M.S.; Eklund, P.C. Infrared-active vibrational modes of single-walled carbon nanotubes. Phys. Rev. Lett. 2005, 95, 157402. [Google Scholar] [CrossRef]
- Ikeda, A.; Nobusawa, K.; Hamano, T.; Kikuchi, J. Single-walled carbon nanotubes template the one-dimensional ordering of a polythiophene derivative. Org. Lett. 2006, 8, 5489–5492. [Google Scholar] [CrossRef]
- Wagner, M.; Kvarnstrom, C.; Ivaska, A. Room temperature ionic liquids in electrosynthesis and spectroelectrochemical characterization of poly(para-phenylene). Electrochim. Acta 2010, 55, 2527–2535. [Google Scholar] [CrossRef]
- Gurban, A.M.; Rotariu, L.; Baibarac, M.; Baltog, I.; Bala, C. Sensistive detection of endocrine disrupters using ionic liquid-single walled carbon nanotubes modified screen-printed based biosensors. Talanta 2011, 85, 2007–2013. [Google Scholar] [CrossRef]
- Sea, M.J.; Mehlenbacher, R.D.; Zanni, M.T.; Arnold, M.S. Experimental measurement of the binding configuration and coverage of chirality–sorting polyfluorenes on carbon nanotubes. J. Phys. Chem. Lett. 2014, 5, 3742–3749. [Google Scholar] [CrossRef]
- Baibarac, M.; Radu, A.; Cristea, M.; Cercel, R.; Smaranda, I. UV light effects on cationic photopolymerization of the SU8 photoresist and its composites with carbon nanotubes: New evidence shown by photoluminescence studies. J. Phys. Chem. C 2020, 124, 7467–7476. [Google Scholar] [CrossRef]
- Jorio, A.; Saito, R.; Hafner, J.H.; Lieber, C.M.; Junter, M.; McClure, T.; Dresselhaus, G.; Dresselhaus, M.S. Structural (n, m) determination of isolated single-wall carbon nanotubes by resonant Raman scattering. Phy. Rev. Lett. 2001, 86, 1118–1121. [Google Scholar] [CrossRef]
- Trznadel, M.; Zagorska, M.; Lapkowski, M.; Louarn, G.; Lefrant, S.; Pron, A. UV-VIS-NIR and Raman spectroelectrochemistry of regioregular poly(3-octylthiophene): Comparison with its non-regioregular analoque. J. Chem. Soc. Faraday Trans. 1996, 92, 1387–1393. [Google Scholar] [CrossRef]
- Baibarac, M.; Baltog, I.; Lefrant, S.; Mevellec, J.Y.; Chauvet, O. Polyaniline and carbon nanotubes based composites containing whole units and fragments of nanotubes. Chem. Mater. 2003, 15, 4149–4156. [Google Scholar] [CrossRef]
- Ding, L.; Zhou, W.; McNicholas, T.P.; Wang, J.; Chu, H.; Li, Y.; Liu, J. Direct observation of the strong interaction between carbon nanotubes and quartz substrate. Nano Res. 2009, 2, 903–910. [Google Scholar] [CrossRef]
- Mingyang, J.; McKensie, L.M.; Modarelli, D.A.; Parquette, J.R. Threading carbon nanotubes through a self-assembled nanotubes. Chem. Sci. 2019, 10, 7868–7877. [Google Scholar]
- Homma, Y.; Chiashi, S.; Yamamoto, T.; Kono, K.; Matsumoto, D.; Shitaba, J.; Sato, S. Photoluminescence measurements and molecular dynamics simulations of water adsorption on the hydrophobic surface of a carbon nanotube in water vapour. Phys. Rev. Lett. 2013, 110, 157402. [Google Scholar] [CrossRef] [PubMed]
- Kataura, H.; Kamazawa, Y.; Maiwa, Y.; Umezu, I.; Suzuki, S.; Ohtsuka, Y.; Achiba, S. Optical properties of single-wall carbon nanotubes. Synth. Met. 1999, 103, 2555–2558. [Google Scholar] [CrossRef]
- Li, X.Y.; Pang, Y.H.; Shi, G.Y.; Zhu, J.R.; Wang, F.; Jin, L.T. Synthesis and electrochromic properties of poly(3-hexyxl-thiophene) in a room temperature ionic liquic and its application to an electrochromic device. Chin. J. Chem. 2008, 26, 677–680. [Google Scholar] [CrossRef]
- Vedhanarayanan, B.; Nair, V.S.; Nair, V.C.; Ajayaghosh, A. Formation of coaxial nanocables with ampliefied supramolecular chirality through an interaction between carbon nanotubes and a chiral π-gelator. Angew. Chem. Int. Ed. 2016, 55, 1–6. [Google Scholar] [CrossRef]
- Shan, B.; Cho, K. First principle study of work functions of single wall carbon nanotubes. Phys. Rev. Lett. 2005, 94, 23602. [Google Scholar] [CrossRef]
- Karachevtsec, V.A.; Ploktnichenko, A.M.; Glamazda, A.Y.; Leontiev, V.S.; Levitsky, I.A. Excitonic energy transfer in polymer wrapped carbon nanotubes in gradually grown nanoassemblies. Phys. Chem. Chem. Phys. 2014, 16, 10914–10922. [Google Scholar] [CrossRef]
- Cardenas, J.F.; Glerup, M. The influence of surfactants on the distribution of the radial breathing modes of single walled carbon nanotubes. Nanotechnology 2006, 17, 5212–5215. [Google Scholar] [CrossRef]




Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Baibarac, M.; Arzumanyan, G.; Daescu, M.; Udrescu, A.; Mamatkulov, K. Anisotropic Photoluminescence of Poly(3-hexyl thiophene) and Their Composites with Single-Walled Carbon Nanotubes Highly Separated in Metallic and Semiconducting Tubes. Molecules 2021, 26, 294. https://doi.org/10.3390/molecules26020294
Baibarac M, Arzumanyan G, Daescu M, Udrescu A, Mamatkulov K. Anisotropic Photoluminescence of Poly(3-hexyl thiophene) and Their Composites with Single-Walled Carbon Nanotubes Highly Separated in Metallic and Semiconducting Tubes. Molecules. 2021; 26(2):294. https://doi.org/10.3390/molecules26020294
Chicago/Turabian StyleBaibarac, Mihaela, Grigory Arzumanyan, Monica Daescu, Adelina Udrescu, and Kahramon Mamatkulov. 2021. "Anisotropic Photoluminescence of Poly(3-hexyl thiophene) and Their Composites with Single-Walled Carbon Nanotubes Highly Separated in Metallic and Semiconducting Tubes" Molecules 26, no. 2: 294. https://doi.org/10.3390/molecules26020294
APA StyleBaibarac, M., Arzumanyan, G., Daescu, M., Udrescu, A., & Mamatkulov, K. (2021). Anisotropic Photoluminescence of Poly(3-hexyl thiophene) and Their Composites with Single-Walled Carbon Nanotubes Highly Separated in Metallic and Semiconducting Tubes. Molecules, 26(2), 294. https://doi.org/10.3390/molecules26020294






