Alpha-Lipoic Acid Plays a Role in Endometriosis: New Evidence on Inflammasome-Mediated Interleukin Production, Cellular Adhesion and Invasion
Abstract
1. Introduction
2. Results
2.1. Alpha Lipoic Acid Effects on Endometriotic Cells Viability
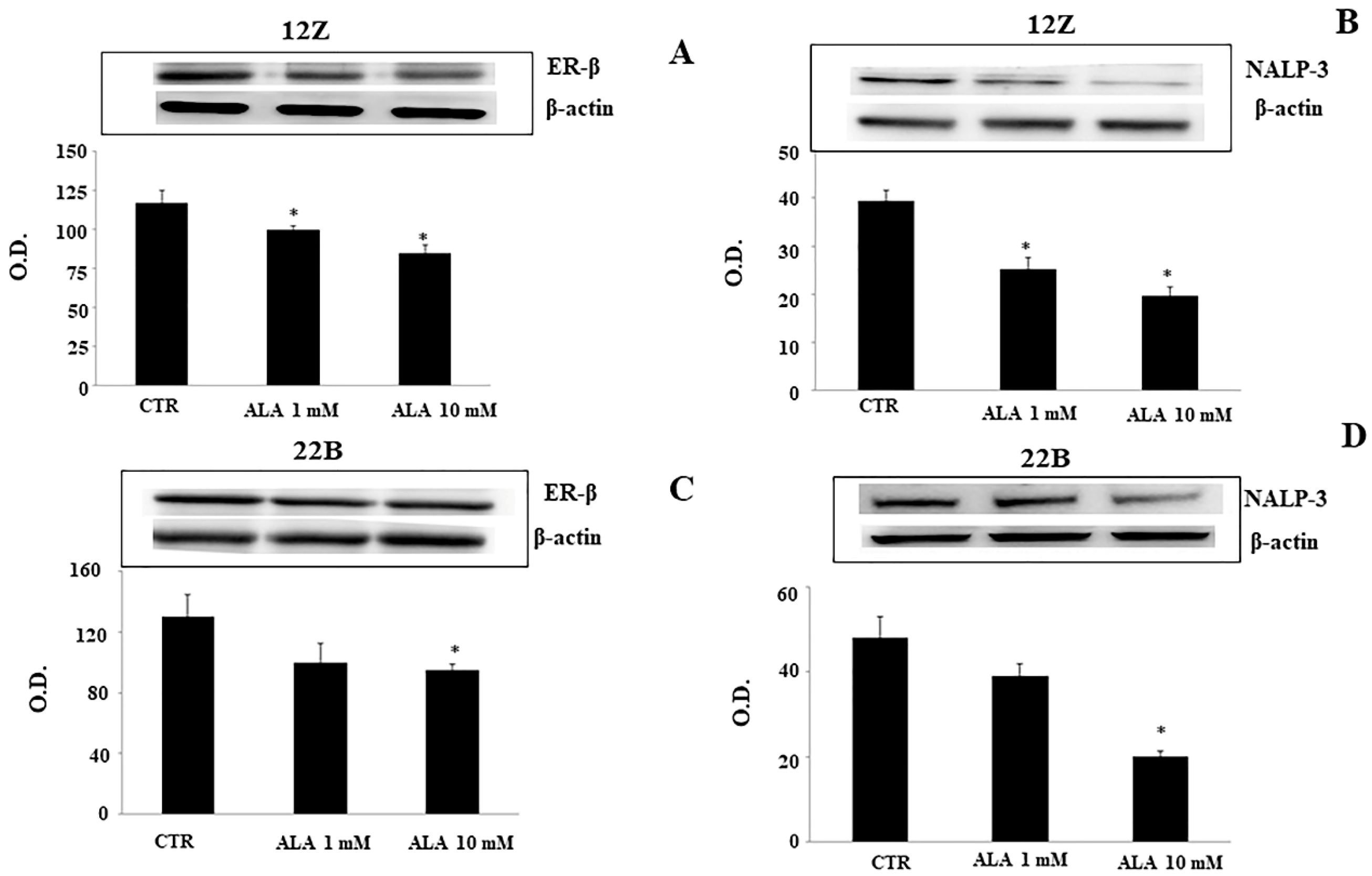
2.2. Alpha-Lipoic Acid Regulates ER-β and NALP-3 Proteins
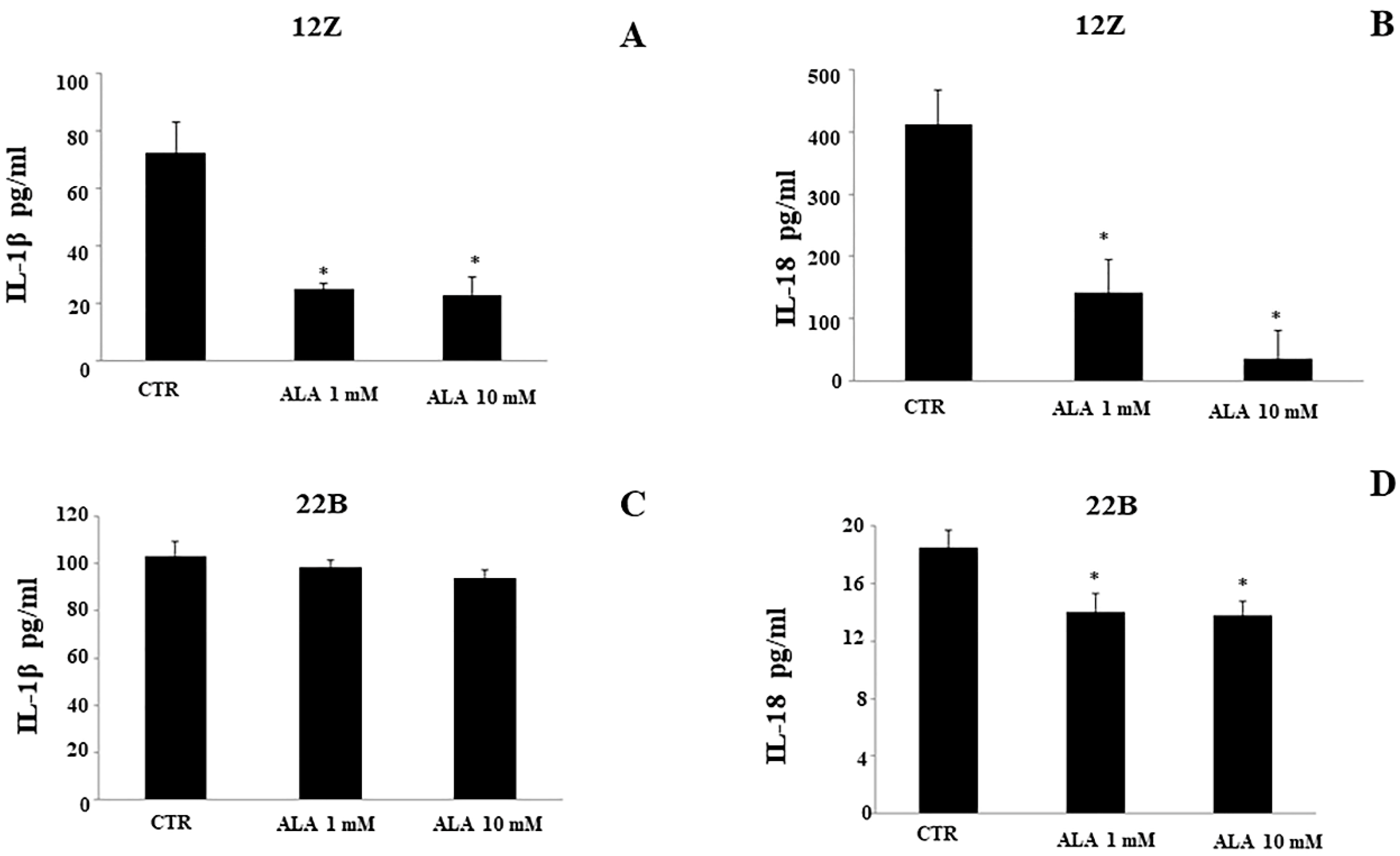
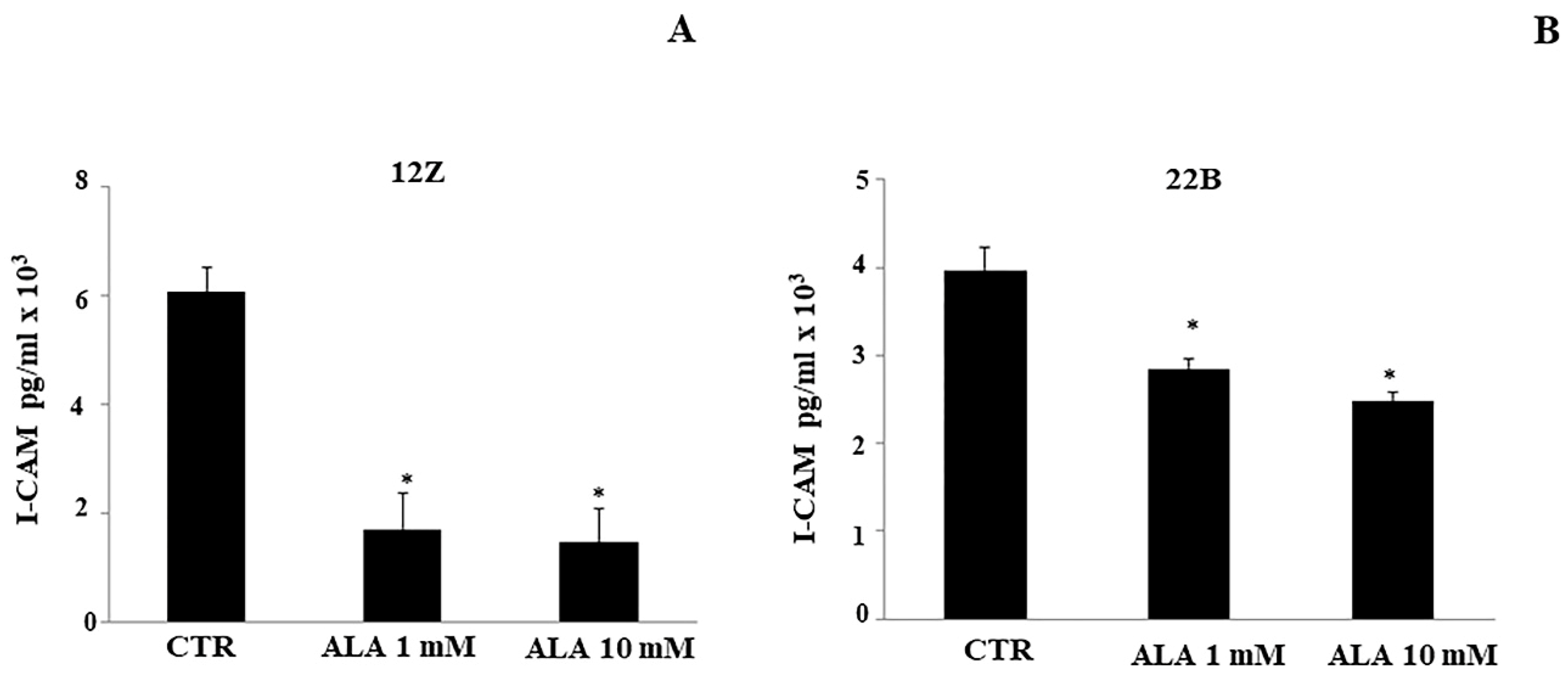
2.3. Cytokine Levels
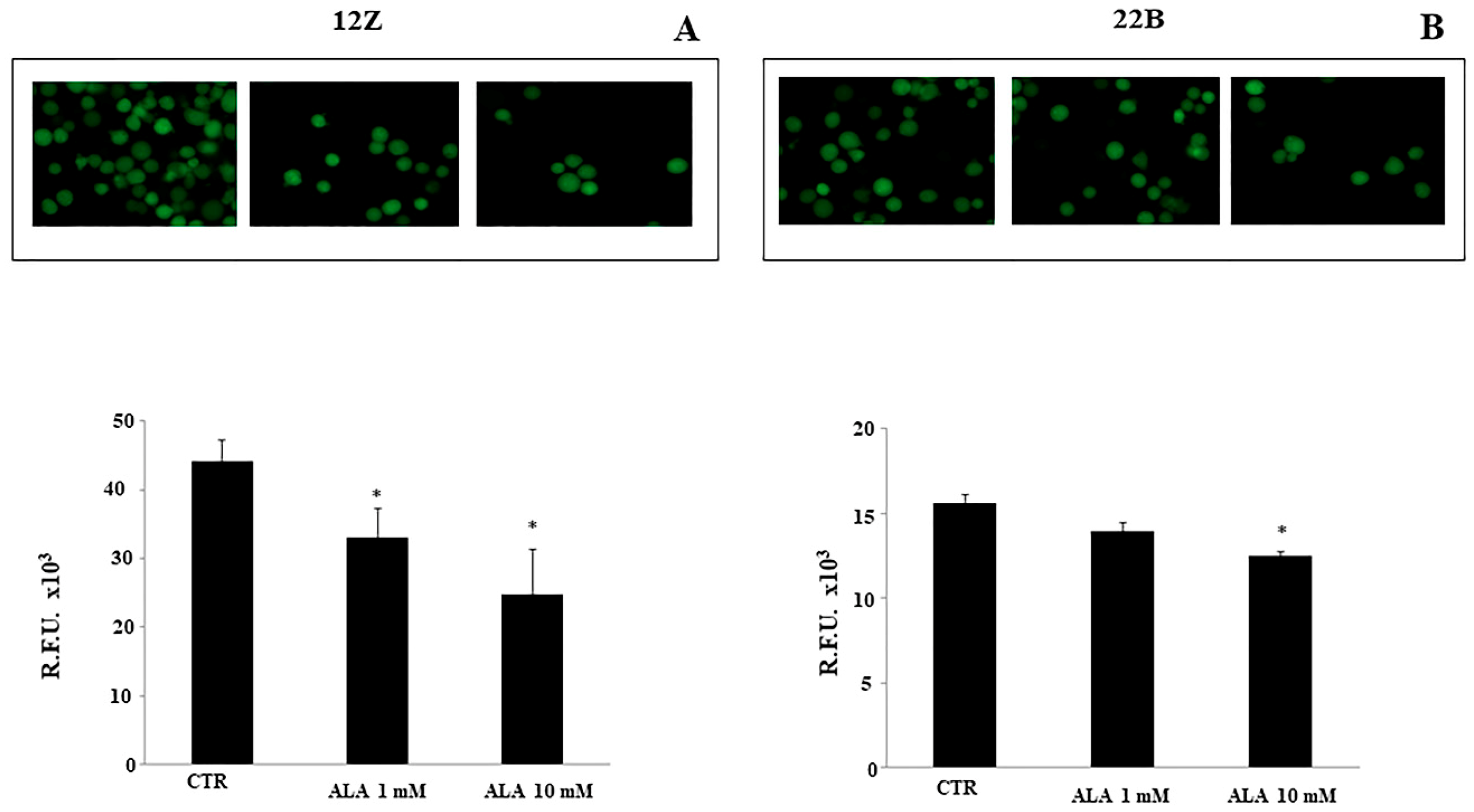
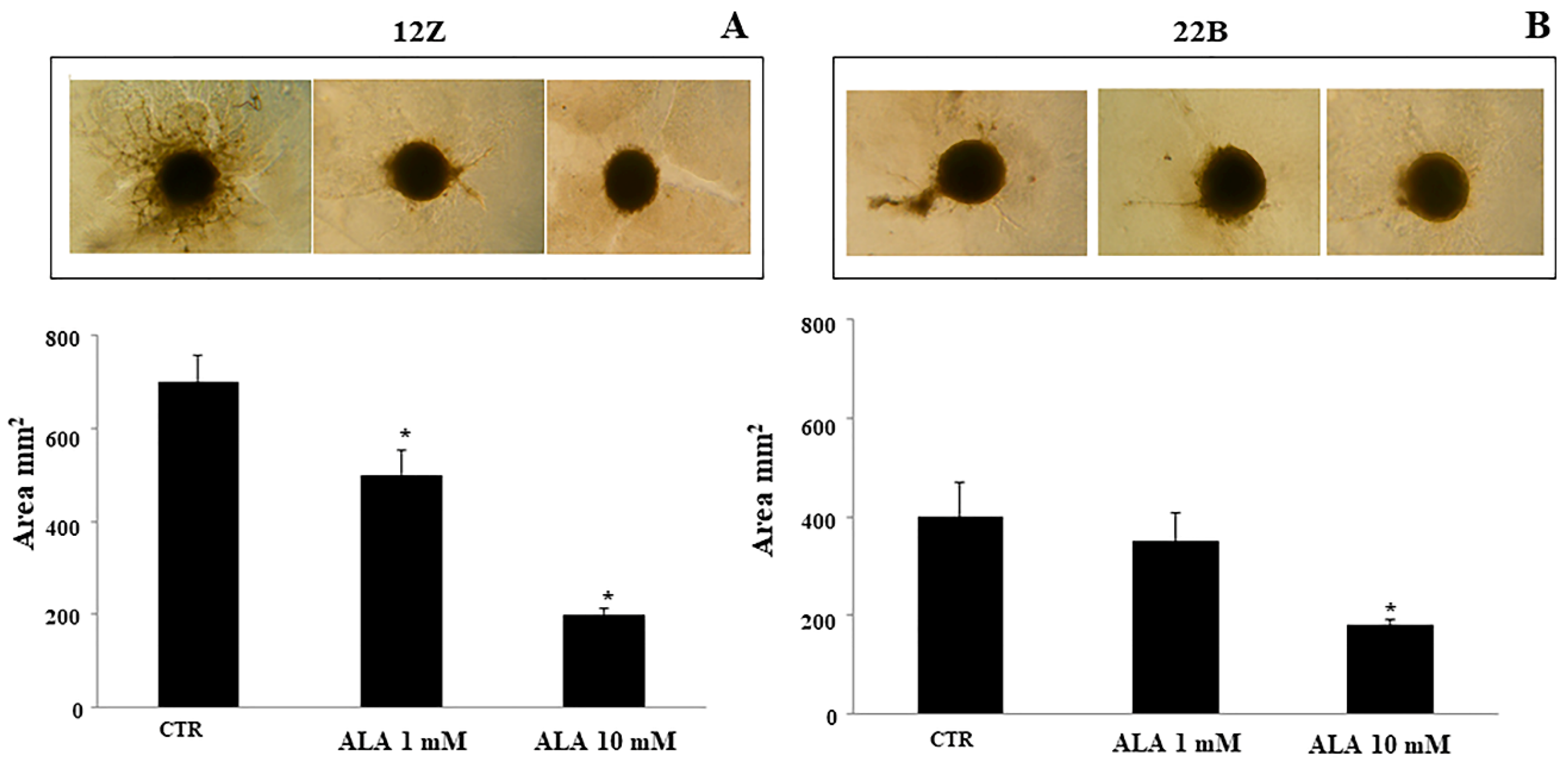
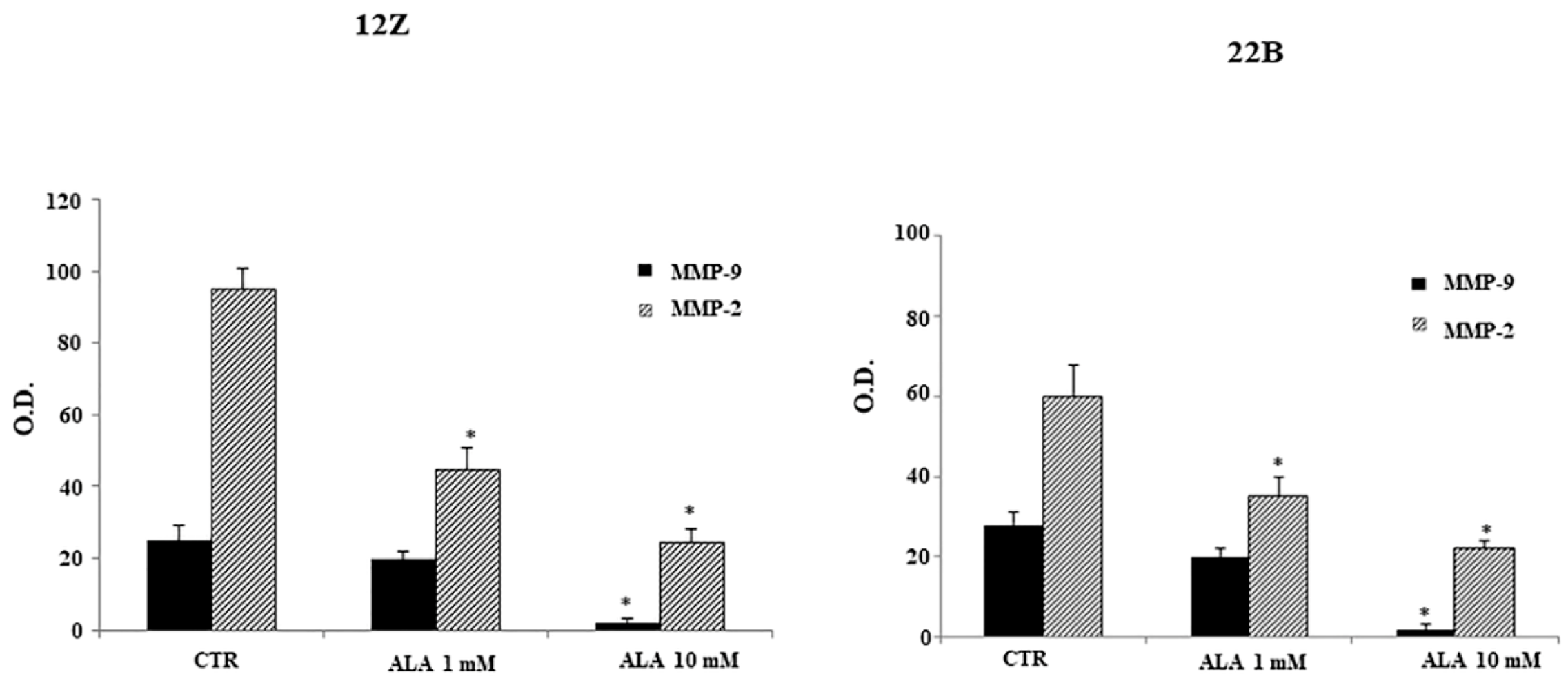
2.4. Alpha Lipoic Acid Inhibits Endometriotic Cell Adhesion and Invasion
3. Discussion
4. Materials and Methods
4.1. Cell Cultures
4.2. Pharmacological Treatments
4.3. Cell Viability Assay
4.4. SDS–PAGE and Immunoblotting
4.5. IL-18 and IL-1β Immunoassays
4.6. Cell Adhesion Assay
4.7. I-CAM Immunoassay
4.8. Cell Invasion Assay
4.9. Gelatin Zymography
4.10. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Vitagliano, A.; Noventa, M.; Quaranta, M.; Gizzo, S. Statins as Targeted “Magical Pills”for the Conservative Treatment of Endometriosis: May Potential Adverse Effects On Female Fertility Represent the “Dark side of the Same Coin”? A Systematic Review of Literature. Reprod. Sci. 2016, 23, 415–428. [Google Scholar] [CrossRef] [PubMed]
- Nezhat, C.; Falik, R.; McKinney, S.; King, L.P. Pathophysiology and management of urinary tract endometriosis. Nat. Rev. Urol. 2017, 14, 359–372. [Google Scholar] [CrossRef] [PubMed]
- Luisi, S.; Lazzeri, L.; Ciani, V.; Petraglia, F. Endometriosis in Italy: From cost estimates to new medical treatment. Gynecol. Endocrinol. 2009, 25, 734–740. [Google Scholar] [CrossRef]
- Nnoaham, K.E.; Hummelshoj, L.; Webster, P.; D’Hooghe, T.; De Cicco Nardone, F.; De Cicco Nardone, C.; Jenkinson, C.; Kennedy, S.H.; Zondervan, K.T. World Endometriosis Research Foundation Global Study of Women’s Health consortium. Impact of endometriosis on quality of life and work productivity: A multicentre study across ten countries. Fertil. Steril. 2011, 96, 366–373. [Google Scholar] [CrossRef] [PubMed]
- Aznaurova, Y.B.; Zhumataev, M.B.; Roberts, T.K.; Aliper, A.M.; Zhavoronkov, A.A. Molecular aspects of development and regulation of endometriosis. Reprod. Biol. Endocrinol. 2014, 13, 50–56. [Google Scholar] [CrossRef] [PubMed]
- Burney, R.O.; Giudice, L.C. Pathogenesis and pathophysiology of endometriosis. Fertil. Steril. 2012, 98, 511–519. [Google Scholar] [CrossRef]
- Sourial, S.; Tempest, N.; Hapangama, D.K. Theories on the pathogenesis of endometriosis. Int. J. Reprod. Med. 2014, 2014, 179515. [Google Scholar] [CrossRef]
- Liu, H.; Lang, J.H. Is abnormal eutopic endometrium the cause of endometriosis? The role of eutopic endometrium in pathogenesis of endoetriosis. Med. Sci. Monit. 2011, 17, RA92–RA99. [Google Scholar]
- Lee, D.H.; Kim, S.C.; Joo, J.K.; Kim, H.G.; Na, Y.J.; Kwak, J.Y.; Lee, K.S. Effects of 17β–estradiol on the release of monocyte chemotactic protein-1 and MAPK activity in monocytes stimulated with peritioneal fluid from endometriosis patients. J. Obstet. Gynaecol. Res. 2012, 38, 516–525. [Google Scholar] [CrossRef]
- Cakmak, H.; Basar, M.; Seval-Celik, Y.; Osteen, K.G.; Duleba, A.J.; Taylor, H.S.; Lockwood, C.J.; Arici, A. Statins inhibit monocyte chemotactic protein 1 expression in endometriosis. Reprod. Sci. 2012, 19, 572–579. [Google Scholar] [CrossRef]
- Iba, Y.; Harada, T.; Horie, S.; Deura, I.; Iwabe, T.; Terakawa, N. Lipopolysaccharide-promoted proliferation of endometriotic stromal cells via induction of tumor necrosis factor alpha and interleukin-8 expression. Fertil. Steril. 2004, 82, 1036–1042. [Google Scholar] [CrossRef] [PubMed]
- Delvoux, B.; Groothuis, P.; D’Hooghe, T.; Kyama, C.; Dunselman, G.; Romano, A. Increased production of 17 beta-estradiol in endometriosis lesions is the result of impaired metabolism. J. Clin. Endocrinol. Metab. 2009, 94, 876–883. [Google Scholar] [CrossRef] [PubMed]
- Burns, K.A.; Rodriguez, K.F.; Hewitt, S.C.; Janardhan, K.S.; Young, S.L.; Korach, K.S. Role of estrogen receptor signalling required for endometriosis-like lesion established in a mouse model. Endocrinology 2012, 153, 3960–3971. [Google Scholar] [CrossRef] [PubMed]
- Han, S.J.; Jung, S.Y.; Wu, S.P.; Hawkins, S.M.; Park, M.J.; Kyo, S.; Qin, J.; Lydon, J.P.; Tsai, S.Y.; Tsai, M.J.; et al. Estrogen receptor β modulates apoptosis complexes and the inflammasome to drive the pathogenesis of endometriosis. Cell 2015, 163, 960–974. [Google Scholar] [CrossRef]
- Dutartre, P. Inflammasomes and Natural Ingredients towards New Anti-Inflammatory Agents. Molecules 2016, 21, 1492. [Google Scholar] [CrossRef]
- Di Nicuolo, F.; Specchia, M.; Trentavizi, L.; Pontecorvi, A.; Scambia, G.; Di Simone, N. An Emerging Role of Endometrial Inflammasome in Reproduction: New Therapeutic Approaches. Protein Pept. Lett. 2018, 25, 455–462. [Google Scholar] [CrossRef]
- Vince, J.E.; Silke, J. The intersection of cell death and inflammasome activation. Cell. Mol. Life Sci. 2016, 73, 2349–2367. [Google Scholar] [CrossRef]
- D’Ippolito, S.; Tersigni, C.; Marana, R.; Di Nicuolo, F.; Gaglione, R.; Rossi, E.D.; Castellani, R.; Scambia, G.; Di Simone, N. Inflammosome in the human endometrium: Further step in the evaluation of the “maternal side”. Fertil. Steril. 2016, 105, 111–118. [Google Scholar] [CrossRef]
- Di Nicuolo, F.; D’ Ippolito, S.; Castellani, R.; Rossi, E.D.; Masciullo, V.; Specchia, M.; Mariani, M.; Pontecorvi, A.; Scambia, G.; Di Simone, N. Effect of alpha-lipoic acid and myoinositol on endometrial inflammasome from recurrent pregnancy loss women. Am. J. Reprod. Immunol. 2019, 82, e13153. [Google Scholar] [CrossRef]
- Maldonado-Rojas, W.; Olivero-Verbel, J.; Ortega-Zuñiga, C. Searching of Protein Targets for Alpha Lipoic Acid. J. Braz. Chem. Soc. 2011, 22, 2250–2259. [Google Scholar] [CrossRef]
- Salehi, B.; Berkay Yılmaz, Y.; Antika, G.; Tumer, T.B.; Mahomoodally, M.F.; Lobine, D.; Akram, M.; Riaz, M.; Capanoglu, E.; Shaporov, F.; et al. Insights on the Use of α-Lipoic Acid for Therapeutic Purposes. Biomolecules 2019, 9, 356. [Google Scholar] [CrossRef] [PubMed]
- Ghibu, S.; Richard, C.; Vergely, C.; Zeller, M.; Cottin, Y.; Rochette, L. Antioxidant properties of an endogenous thiol: Alpha-lipoic acid, useful in the prevention of cardiovascular diseases. J. Cardiovasc. Pharmacol. 2009, 54, 391–398. [Google Scholar] [CrossRef] [PubMed]
- Tibullo, D.; Li Volti, G.; Giallongo, C.; Grasso, S.; Tomassoni, D.; Anfuso, C.D.; Lupo, G.; Amenta, F.; Avola, R.; Bramanti, R. Biochemical and clinical relevance of alpha lipoic acid: Antioxidant and anti-inflammatory activity, molecular pathways and therapeutic potential. Inflamm. Res. 2017, 66, 947–959. [Google Scholar] [CrossRef] [PubMed]
- El Barky, A.R.; Hussein, S.A.; Mohamed, T.M. The potent antioxidant alpha lipoic acid. J. Plant Chem. Ecophysiol. 2017, 2, 1016. [Google Scholar]
- Wang, K.C.; Tsai, C.P.; Lee, C.; Chen, S.Y.; Lin, G.J.; Yen, M.H.; Sytwu, H.K.; Chen, S.J. α-Lipoic acid enhances endogenous peroxisome-proliferator-activated receptor-γ to ameliorate experimental autoimmune encephalomyelitis in mice. Clin. Sci. 2013, 125, 329–340. [Google Scholar] [CrossRef]
- Shay, K.P.; Moreau, R.F.; Smith, E.J.; Smith, A.R.; Hagen, T.M. Alpha-lipoic acid as a dietary supplement: Molecular mechanisms and therapeutic potential. Biochim. Biophys. Acta 2009, 1790, 1149–1160. [Google Scholar] [CrossRef]
- Jin, H.B.; Yang, Y.B.; Song, Y.L.; Zhang, Y.C.; Li, Y.R. Lipoic acid attenuates the expression of adhesion molecules by increasing endothelial mitric-oxide synthase activity. Mol. Biol. Rep. 2013, 40, 377–382. [Google Scholar] [CrossRef]
- Harris, H.A.; Bruner-Tran, K.L.; Zhang, X.; Osteen, K.G.; Lyttle, C.R. A selective estrogen receptor-beta agonist causes lesion regression in an experimentally induced model of endometriosis. Hum. Reprod. 2005, 20, 936–941. [Google Scholar] [CrossRef]
- Khan, R.N.; Hay, D.P. A clear and present danger: Inflammasomes DAMPing down disorders of pregnancy. Hum. Reprod. Update 2015, 21, 388–405. [Google Scholar] [CrossRef]
- Pontillo, A.; Girardelli, M.; Agostinis, C.; Masat, E.; Bulla, R.; Crovella, S. Bacterial LPS differently modulates inflammasome gene expression and IL-1β secretion in trophoblast cells, decidual stromal cells, and decidual endothelial cells. Reprod. Sci. 2013, 20, 563–566. [Google Scholar] [CrossRef]
- Dinarello, C.A. A clinical perspective of IL-1β as the gatekeeper of inflammation. Eur. J. Immunol. 2011, 41, 1203–1217. [Google Scholar] [CrossRef]
- Sims, J.E.; Smith, D.E. The IL-1 family: Regulators of immunity. Nat. Rev. Immunol. 2010, 10, 89–102. [Google Scholar] [CrossRef] [PubMed]
- Dinarello, C.A. Interleukin-18 and the pathogenesis of inflammatory diseases. Semin. Nephrol. 2007, 27, 98–114. [Google Scholar] [CrossRef] [PubMed]
- Dinarello, C.A.; Kaplanski, G. Interleukin-18 treatment options for inflammatory diseases. Expert Rev. Clin. Immunol. 2005, 1, 619–632. [Google Scholar] [CrossRef] [PubMed]
- Jin, C.; Flavell, R.A. Molecular mechanism of NLRP3 inflammasome activation. J. Clin. Immunol. 2010, 30, 628–631. [Google Scholar] [CrossRef] [PubMed]
- Netea, M.G.; Simon, A.; Van De Veerdonf, F.; Kullberg, B.J.; Van Der Meer, J.W.; Joosten, L.A. Il-1beta processing in host defence: Beyond the inflammasomes. PLoS Pathog. 2010, 6, e1000661. [Google Scholar] [CrossRef] [PubMed]
- Park, C.; Jeong, J.W.; Lee, D.S.; Yim, M.J.; Lee, J.M.; Han, M.H.; Kim, S.; Kim, H.S.; Kim, G.K.; Park, E.K.; et al. Sargassum serratifolium extract attenuates interleukin-1β-induced oxidative stress and inflammatory response in chondrocytes by suppressing the activation of NF-kB, p38 MAPK, and PI3K/Akt. Int. J. Mol. Sci. 2018, 19, 2308. [Google Scholar] [CrossRef] [PubMed]
- Sulistyowati, E.; Lee, M.Y.; Wu, L.C.; Hsu, J.H.; Dai, Z.K.; Wu, B.N.; Lin, M.C.; Yen, J.L. Exogenus heat shock cognate protein 70 suppresses LPS-Induced inflammation by down-regulating NF-kB through MAPK and MMP-2/-9 pathways in macrophages. Molecules 2018, 23, 2124. [Google Scholar] [CrossRef]
- Fratantonio, D.; Speciale, A.; Molonia, M.S.; Bashllari, R.; Palumbo, M.; Saija, A.; Cimino, F.; Monastra, G.; Virgili, F. Alpha-lipoic acid, but not di-hydrolipoic acid, activates Nrf2 response in primary human umbilical-vein endothelial cells and protects against TNF-α induced endothelium dysfunction. Arch. Biochem. Biophys. 2018, 655, 18–25. [Google Scholar] [CrossRef]
- Ying, Z.; Kampfrath, T.; Sun, Q.; Parthasarathy, S.; Rajagopalan, S. Evidence that α-lipoic acid inhibits NF-κB activation independent of its antioxidant function. Inflamm. Res. 2011, 60, 219–225. [Google Scholar] [CrossRef]
- Lee, J.; Banu, S.K.; Rodriguez, R.; Starzinski-Powitz, A.; Arosh, J.A. Selective blockade of prostaglandin E2 receptors EP2 and EP4 signalling inhibits proliferation of human endometriotic epithelial cells and stromal cells through distinct cell cycle arrest. Fertil. Steril. 2010, 93, 2498–2506. [Google Scholar] [CrossRef] [PubMed]
| 12Z | p Value | 22B | p Value | |
|---|---|---|---|---|
| ALA (mM) | ||||
| 0 | 100.0 ± 1.6 | 100.0 ± 1.0 | ||
| 1.0 | 93.4 ± 3.8 | NS | 96.8 ± 1.2 | NS |
| 10.0 | 98.0 ± 2.0 | NS | 90.9 ± 4.9 | NS |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Di Nicuolo, F.; Castellani, R.; De Cicco Nardone, A.; Barbaro, G.; Paciullo, C.; Pontecorvi, A.; Scambia, G.; Di Simone, N. Alpha-Lipoic Acid Plays a Role in Endometriosis: New Evidence on Inflammasome-Mediated Interleukin Production, Cellular Adhesion and Invasion. Molecules 2021, 26, 288. https://doi.org/10.3390/molecules26020288
Di Nicuolo F, Castellani R, De Cicco Nardone A, Barbaro G, Paciullo C, Pontecorvi A, Scambia G, Di Simone N. Alpha-Lipoic Acid Plays a Role in Endometriosis: New Evidence on Inflammasome-Mediated Interleukin Production, Cellular Adhesion and Invasion. Molecules. 2021; 26(2):288. https://doi.org/10.3390/molecules26020288
Chicago/Turabian StyleDi Nicuolo, Fiorella, Roberta Castellani, Alessandra De Cicco Nardone, Greta Barbaro, Carmela Paciullo, Alfredo Pontecorvi, Giovanni Scambia, and Nicoletta Di Simone. 2021. "Alpha-Lipoic Acid Plays a Role in Endometriosis: New Evidence on Inflammasome-Mediated Interleukin Production, Cellular Adhesion and Invasion" Molecules 26, no. 2: 288. https://doi.org/10.3390/molecules26020288
APA StyleDi Nicuolo, F., Castellani, R., De Cicco Nardone, A., Barbaro, G., Paciullo, C., Pontecorvi, A., Scambia, G., & Di Simone, N. (2021). Alpha-Lipoic Acid Plays a Role in Endometriosis: New Evidence on Inflammasome-Mediated Interleukin Production, Cellular Adhesion and Invasion. Molecules, 26(2), 288. https://doi.org/10.3390/molecules26020288






