Corn Silage Supplemented with Pomegranate (Punica granatum) and Avocado (Persea americana) Pulp and Seed Wastes for Improvement of Meat Characteristics in Poultry Production
Abstract
1. Introduction
2. Results
2.1. Quantitative Criteria
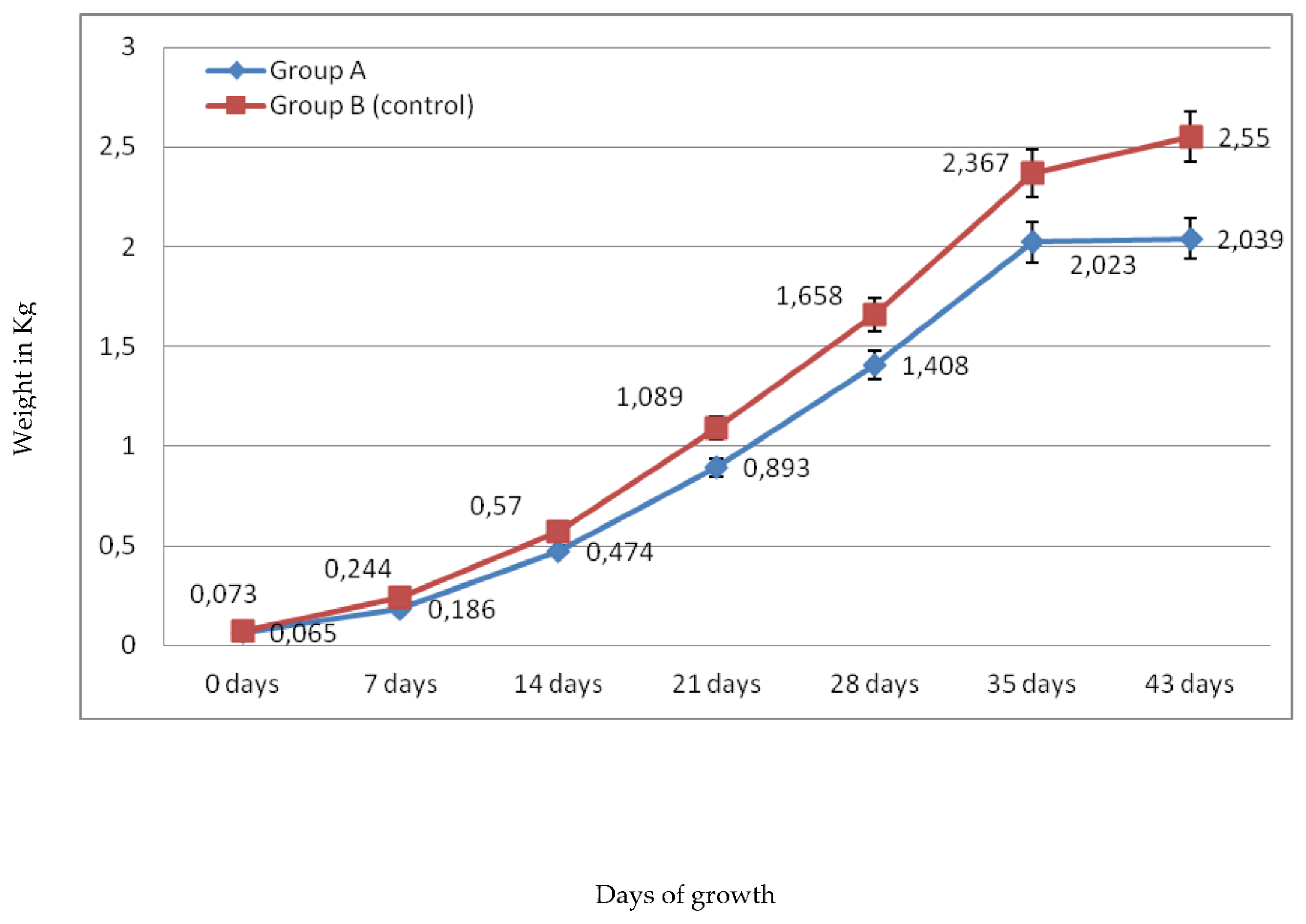
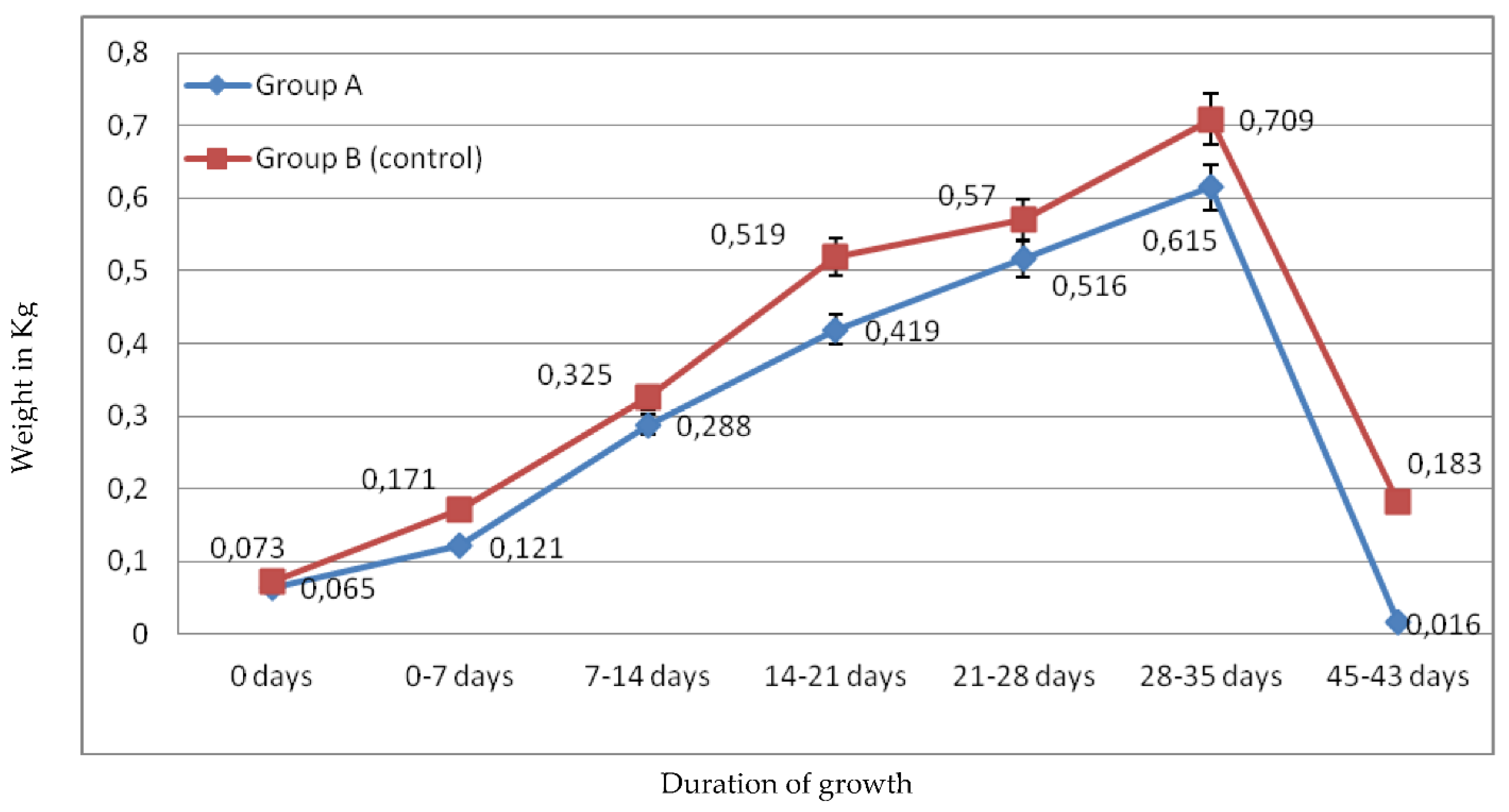
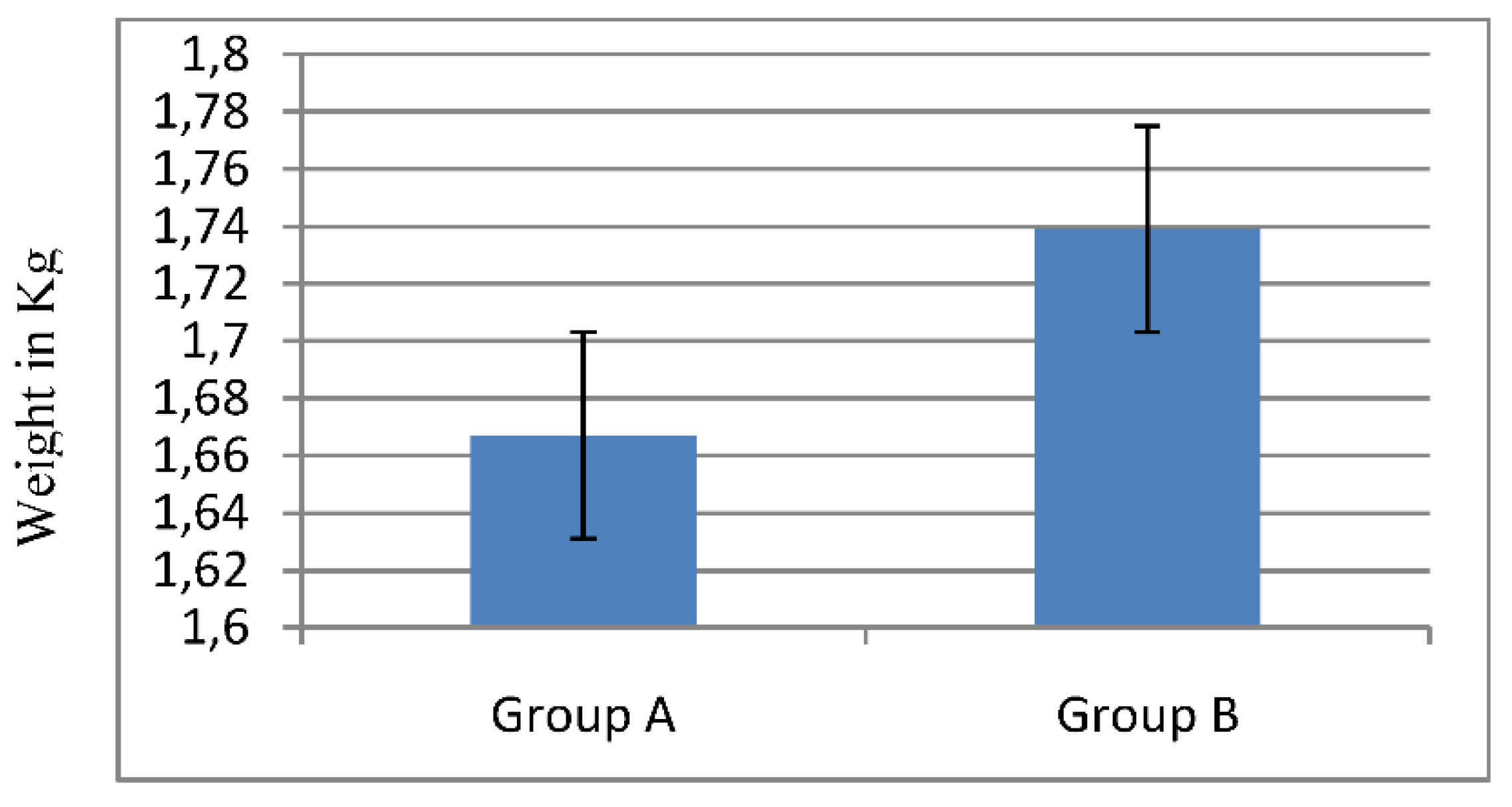
2.1.1. Broiler’s Growth Rate
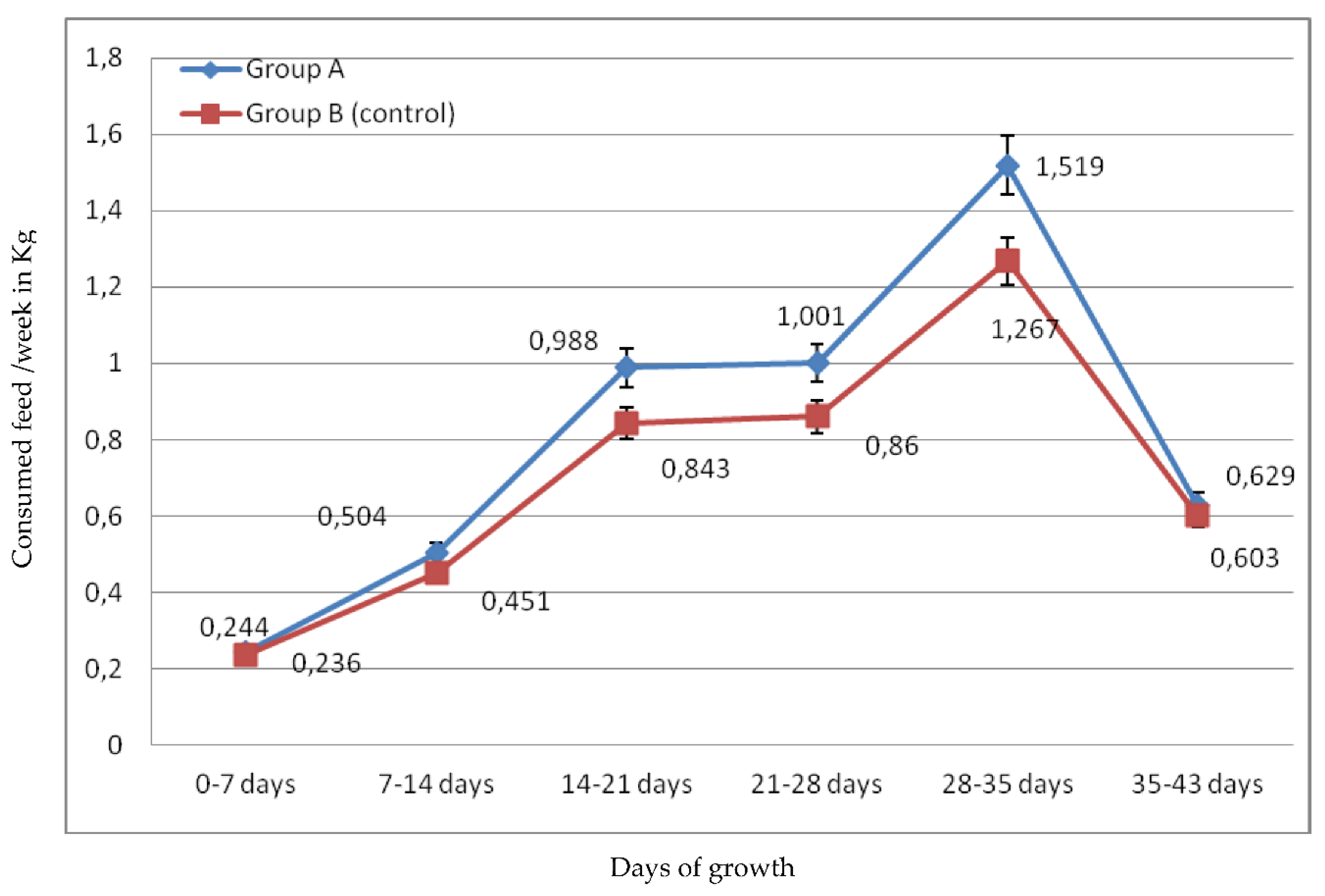
2.1.2. Feed Cost
2.2. Qualitive Criteria
2.2.1. Fat, Protein Moisture and Collagen Measurements
2.2.2. Thiobarbituric Acid–Reactive Substances (TBARS) Assay
2.2.3. Lipid and Fatty Acid Profile
3. Discussion
4. Materials and Methods
4.1. Plant Materials
4.2. Preparation of the Supplemented Silage
4.3. Broilers
4.4. Tissue Collection
4.5. TBARS Assay
4.6. Lipid Extraction and Fatty Acid Analysis
4.7. Statistical Analysis
5. Conclusions
Author Contributions
Funding

Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Patent Pending Notice
Sample Availability
References
- Bampidis, V.A.; Robinson, P.H. Citrus by-products as ruminant feeds: A review. Anim. Feed. Sci. Technol. 2006, 128, 175–217. [Google Scholar] [CrossRef]
- Makri, S.; Kafantaris, I.; Stagos, D.; Chamokeridou, T.; Petrotos, K.; Gerasopoulos, K.; Mpesios, A.; Goutzourelas, N.; Kokkas, S.; Goulas, P.; et al. Novel feed including bioactive compounds from winery wastes improved broilers’ redox status in blood and tissues of vital organs. Food Chem. Toxicol. 2017, 102, 24–31. [Google Scholar] [CrossRef]
- Pinotti, L.; Krogdahl, A.; Givens, I.; Knight, C.; Baldi, A.; Baeten, V.; Raamsdonk, L.; van Woodgate, S.; Perez Marin, D.; Luten, J. The role of animal nutrition in designing optimal foods of animal origin as reviewed by the COST Action Feed for Health (FA0802). Biotechnol. Agron. Société Environ. 2014, 18, 471–479. [Google Scholar]
- van Ryssen, J.B.J.; Skenjana, A.; van Niekerk, W.A. Can avocado meal replace maize meal in broiler diets? Appl. Anim. Husb. Rural. Dev. 2013, 6, 22–27. [Google Scholar]
- Ismail, A.; Marjan, Z.M.; Foong, C.W. Total antioxidant activity and phenolic content in selected vegetables. Food Chem. 2004, 87, 581–586. [Google Scholar] [CrossRef]
- Kumar, A.; Roy, B.; Lakhani, G.P.; Jain, A. Evaluation of dried bread waste as feedstuff for growing crossbred pigs. Vet. World 2014, 7, 698–701. [Google Scholar] [CrossRef]
- Garg, A.K.; Pathak, N.N.; Anjaneyulu, A.S.R.; Lakshmanan, V. Utilization of cane molasses as a source of energy in the diet of young pigs. Agric. Wastes 1986, 17, 225–228. [Google Scholar] [CrossRef]
- Kjos, N.P.; Øverland, M.; Arnkværn, B.E.; Sørheim, O. Food waste products in diets for growing-finishing pigs: Effect on growth performance, carcass characteristics and meat quality. Acta Agric. Scand. Sect. Anim. Sci. 2000, 50, 193–204. [Google Scholar] [CrossRef]
- Myer, R.O.; Brendemuh, J.H.; Johnson, D.D. Evaluation of dehydrated restaurant food waste products as feedstuffs for finishing pigs. J. Anim. Sci. 1999, 77, 685–692. [Google Scholar] [CrossRef]
- De Oliveira, A.C.; Valentim, I.B.; Silva, C.A.; Henríquez, B.E.J.; de Barros, M.P.; Mano, C.M. Total phenolic content and free radical scavenging activities of methanolic extract powders of tropical fruit residues. Food Chem. 2009, 115, 469–475. [Google Scholar] [CrossRef]
- Ganhão, R.; Estévez, M.; Kylli, P.; Heinonen, M.; Morcuende, D. Characterization of selected wild Mediterranean fruits and comparative efficacy as inhibitors of oxidative reactions in emulsified raw pork burger patties. J. Agric. Food Chem. 2010, 58, 8854–8861. [Google Scholar] [CrossRef] [PubMed]
- Wojdylo, A.; Oszmianski, J.; Czemerys, R. Antioxidant activity and phenolic compounds in 32 selected herbs. Food Chem. 2007, 105, 940–949. [Google Scholar] [CrossRef]
- Yoo, K.M.; Lee, C.H.; Lee, H.; Moon, B.; Lee, C.Y. Relative antioxidant and cytoprotective activities of common herbs. Food Chem. 2008, 106, 929–936. [Google Scholar] [CrossRef]
- Moure, A.; Cruz, J.M.; Franco, D.; Doínguez, J.M.; Sineiro, J.; Dominguez, H. Natural antioxidant from residual sources. Food Chem. 2001, 72, 145–171. [Google Scholar] [CrossRef]
- Gaztambide, C. Alimentación de Animales en los Trópicos; Editorial Diana: Mexico City, Mexico, 1975; p. 275. [Google Scholar]
- Montoya, N.; Pino, I.D.; Correa, H.J. Evaluación de la suplementación con papa (Solanum tuberosum) durante la lactancia en vacas Holstein. (Evaluation of the supplementation with potato (Solanum tuberosum) during the lactation of Holstein cows). Rev. Colomb. Cienc. Pecu. 2004, 17, 241–249. [Google Scholar]
- Ulloa, J.B.; Van Weerd, J.H.; Huisman, E.A.; Verreth, J.A. Tropical agricultural residues and their potential uses in fish feeds: The Costa Rican situation. Waste Manag. 2004, 24, 87–97. [Google Scholar] [CrossRef]
- Ding, H.; Chin, Y.W.; Kinghorn, A.D.; D’Ambrosio, S.M. Chemopreventive characteristics of avocado fruit. Semin. Cancer Biol. 2007, 17, 386–394. [Google Scholar] [CrossRef]
- Zafar, T.; Sidhu, J.S. Avocado: Production, Quality, and Major Processed Products. In Handbook of Vegetables and Vegetable Processing; Sinha, N., Ed.; Wiley-Blackwell: Oxford, UK, 2011; Volume 1871, pp. 525–543. [Google Scholar]
- Ranade, S.S.; Thiagarajan, P. A review on Persea americana Mill. (Avocado)-its fruit and oil. Int. J. Pharm. Tech. Res. 2015, 8, 72–77. [Google Scholar]
- Wang, W.; Bostic, T.R.; Gu, L. Antioxidant capacities, procyanidins and pigments in avocado of different strains and cultivars. Food Chem. 2010, 122, 1193–1198. [Google Scholar] [CrossRef]
- Rodríguez-Carpena, J.G.; Morcuende, D.; Andrade, M.-J.; Kylli, P.; Estévez, M. Avocado (Persea americana Mill.) phenolics, in vitro antioxidant and antimicrobial activities, and inhibition of lipid and protein oxidation in porcine patties. J. Agric. Food Chem. 2011, 59, 5625–5635. [Google Scholar] [CrossRef]
- Rodríguez-Carpena, J.G.; Morcuende, D.; Estévez, M. Avocado by-products as inhibitors of color deterioration and lipid and protein oxidation in raw porcine patties subjected to chilled storage. Meat Sci. 2011, 89, 166–173. [Google Scholar] [CrossRef]
- Grageola, F.; Sangines, L.; Díaz, C.; Gómez, A.; Cervantes, M.; Lemus, C.; Ly, J. The effect of breed and dietary level of avocado fat on the N and energy balance in young pigs. J. Anim. Feed. Sci. 2010, 19, 37–49. [Google Scholar] [CrossRef][Green Version]
- Oswell, N.J.; Thippareddi, H.; Pegg, R.B. Practical use of natural antioxidants in meat products in the U.S.: A review. Meat Sci. 2018, 145, 469–479. [Google Scholar] [CrossRef]
- Hygreeva, D.; Pandey, M.C.; Radhakrishna, K. Potential applications of plant based derivatives as fat replacers, antioxidants and antimicrobials in fresh and processed meat products. Meat Sci. 2014, 98, 47–57. [Google Scholar] [CrossRef]
- Hess-Pierce, B.; Kader, A.A. Responses of “Wonderful” pomegranates to controlled atmospheres. Acta Hortic. 2003, 60, 751–757. [Google Scholar] [CrossRef]
- Wu, P.; Gu, Y.; Zhao, R.; Liu, Y.; Wang, Y.; Lv, G.; Li, Z.; Bao, Y. Residual pomegranate affecting the nonspecific immunity of juvenile Darkbarbel catfish. Fish Shellfish. Immunol. 2019, 95, 190–194. [Google Scholar] [CrossRef] [PubMed]
- Wadhwa, M.; Bakshi, M.P.S. Utilization of Fruit and Vegetable Wastes as Livestock Feed and as Substrates for Generation of Other value-added Products; Makkar, H.P.S., Ed.; FAO: Rome, Italy, 2013. [Google Scholar]
- Emami, A.; Ganjkhanlou, M.; Fathi Nasri, M.H.; Zali, A.; Rashidi, L.; Sharifi, M. Antioxidant status of dairy goats fed diets containing pomegranate seed oil or linseed oil. Small Rumin. Res. 2017, 153, 175–179. [Google Scholar] [CrossRef]
- Marcos, C.N.; Carro, M.D.; Fernández-Yepes, J.E.; Arbesu, L.; Molina-Alcaide, E. Utilization of avocado and mango fruit wastes in multi-nutrient blocks for goats feeding: In vitro evaluation. Animals 2020, 10, 2279. [Google Scholar] [CrossRef]
- de Evan, T.; Carro, M.D.; Fernández Yepes, J.E.; Haro, A.; Arbesú, L.; Romero-Huelva, M.; Molina-Alcaide, E. Effects of feeding multinutrient blocks including avocado pulp and peels to dairy goats on feed intake and milk yield and composition. Animals 2020, 10, 194. [Google Scholar] [CrossRef] [PubMed]
- Natalello, A.; Priolo, A.; Valenti, B.; Codini, M.; Mattioli, S.; Pauselli, M.; Puccio, M.; Lanza, M.; Stergiadis, S.; Luciano, G. Dietary pomegranate by-product improves oxidative stability of lamb meat. Meat Sci. 2020, 162, 108037. [Google Scholar] [CrossRef]
- Khorsandi, S.; Riasi, A.; Khorvash, M.; Hashemzadeh, F. Nutrients digestibility, metabolic parameters and milk production in postpartum Holstein cows fed pomegranate (Punica granatum L.) by-products silage under heat stress condition. Anim. Feed. Sci. Technol. 2019, 255, 114213. [Google Scholar] [CrossRef]
- Skenjana, A.; Van Ryssen, J.B.J.; Van Niekerk, W.A. In vitro digestibility and in situ degradability of avocado meal and macadamia waste products in sheep. South Afr. J. Anim. Sci. 2006, 36, 78–81. [Google Scholar]
- Abdel Aziz, A.R.; AbouLail, M.R.; Aziz, M.; Omar, M.A.; Sultan, K. In vitro and in vivo anthelmintic activity of pumpkin seeds and pomegranate peels extracts against Ascaridia galli. Beni Suef Univ. J. Basic Appl. Sci. 2018, 7, 231–234. [Google Scholar] [CrossRef]
- Basu, S.; Eriksson, M. Vitamin E in relation to lipid peroxidation in experimental septic shock. Prostaglandins Leukot. Essent. Fat. Acids 2000, 62, 195–199. [Google Scholar] [CrossRef]
- Iqbal, M.; Pumford, N.R.; Tang, Z.X.; Lassiter, K.; Wing, T.; Cooper, M. Low feed efficient broilers within a single genetic line exhibit higher oxidative stress and protein expression in breast muscle with lower mitochondrial complex activity. Poult. Sci. 2004, 83, 474–484. [Google Scholar] [CrossRef]
- Lauritzen, B.; Lykkesfeldt, J.; Friis, C. Evaluation of a single dose versus a di-vided dose regimen of amoxicillinin treatment of Actinobacillus pleuropneumoniae in fectionin pigs. Res. Vet. Sci. 2005, 79, 61–67. [Google Scholar] [CrossRef]
- Lykkesfeldt, J.; Svendsen, O. Oxidants and antioxidants in disease: Oxidative stress in farm animals. Vet. J. 2007, 173, 502–511. [Google Scholar] [CrossRef]
- Goto, S.; Radák, Z. Hormetic effects of reactive oxygen species by exercise: A view from animal studies for successful aging in human. Dose Response 2009, 8, 68–72. [Google Scholar] [CrossRef]
- Esteban, M.B.; García, A.J.; Ramos, P.; Márquez, M.C. Evaluation of fruit-vegetable and fish wastes as alternative feedstuffs in pig diets. Waste Manag. 2007, 27, 193–200. [Google Scholar] [CrossRef]
- García, A.J.; Esteban, M.B.; Márquez, M.C.; Ramos, P. Biodegradable municipal solid waste: Characterization and potential use as animal feedstuffs. Waste Manag. 2005, 25, 780–787. [Google Scholar] [CrossRef]
- Katongole, C.B.; Bareeba, F.B.; Sabiiti, E.N.; Ledin, I. Nutritional characterization of some tropical urban market crop wastes. Anim. Feed. Sci. Technol. 2008, 142, 275–291. [Google Scholar] [CrossRef]
- Smaoui, S.; Hlim, H.B.; Mtiba, A.C.; Fourati, M.; Sellem, I.; Elhad, K.; Ennouri, K.; Mellouli, L. Pomegranate peel as phenolic compounds source: Advanced analytical strategies and practical use in meat products. Meat Sci. 2019, 158, 107914. [Google Scholar] [CrossRef]
- Mahfuz, S.; Piao, X.S. Application of Moringa (Moringa oleifera) as natural feed supplement in poultry diets. Animals 2019, 9, 431. [Google Scholar] [CrossRef]
- Meagher, E.A.; FitzGerald, G.A. Indices of lipid peroxidation in vivo: Strengths and limitations. Free. Radic. Biol. Med. J. 2000, 28, 1745–1750. [Google Scholar] [CrossRef]
- World Health Organization. Fats and Fatty Acids in Human Nutrition; FAO Food and Nutrition Paper 91; World Health Organization: Geneva, Switzerland, 2008; ISBN 9789251067338. [Google Scholar]
- Sarmah, A.K.; Meyer, M.T.; Boxall, B.A. A global perspective on the use, sales, exposure pathways, occurrence, fate and effects of veterinary antibiotics (VAs) in the environment. Chemosphere 2006, 65, 725–759. [Google Scholar] [CrossRef]
- Rebah, B.; Prevost, F.; Yezza, D.; Tyagi, A.R.D. Agro-industrial waste materials and wastewater sludge for rhizobial inoculant production: A review. Bioresour. Technol. 2007, 98, 3535–3546. [Google Scholar]
- Sud, D.; Mahajan, G.; Kaur, M.P. Agricultural waste material as potential adsorbent for sequestering heavy metal ions from aqueous solutions e a review. Bioresour. Technol. 2008, 99, 6017–6027. [Google Scholar] [CrossRef]
- Ayala-Zavala, J.F.; Vega-Vega, V.; Rosas-Domínguez, C.; Palafox-Carlos, H.; Villa-Rodriguez, J.A.; Siddiqui, M.W.; Dávila-Aviña, J.E.; González-Aguilar, G.A. Agro-industrial potential of exotic fruit byproducts as a source of food additives. Food Res. Int. 2011, 44, 1866–1874. [Google Scholar] [CrossRef]
- Vinha, A.F.; Barreira, S.V.; Castro, A.; Costa, A.; Oliveira, M.B.P. Influence of the storage conditions on the physicochemical properties, antioxidant activity and microbial flora of different tomato (Lycopersicon esculentum L.) cultivars. J. Agric. Sci. 2013, 5, 118. [Google Scholar] [CrossRef]
- Mensah, J.K.; Golomeke, D. Antioxidant and antimicrobial activities of the extracts of the Calyx of Hibiscus sabdariffa Linn. Curr. Sci. Perspect. 2015, 1, 69–76. [Google Scholar]
- Guil-Guerrero, J.L.; Ramos, L.; Moreno, C.; Zúñiga-Paredes, J.C.; Carlosama-Yepez, M.; Ruales, P. Antimicrobial activity of plant-food by-products: A review focusing on the tropics. Livest. Sci. 2016, 189, 32–49. [Google Scholar] [CrossRef]
- Reddy, P.R.K.; Elghandour, M.M.M.Y.; Salem, A.Z.M.; Yasaswini, D.; Reddy, P.P.R.; Nagarjuna Reddy, A.; Hyder, I. Plant secondary metabolites as feed additives in calves for antimicrobial stewardship. Anim. Feed. Sci. Technol. 2020, 264, 114469. [Google Scholar] [CrossRef]
- Abuelo, A.; Hernandez, J.; Benedito, J.L.; Castillo, C. The importance of the oxidative status of dairy cattle in the periparturient period: Revisiting antioxidant supplementation. J. Anim. Physiol. Anim. Nutr. 2015, 95, 1003–1016. [Google Scholar] [CrossRef] [PubMed]
- Deaton, C.M.; Marlin, D.J.; Smith, N.C.; Harris, P.A.; Schroter, R.C.; Kelly, F.J. Antioxidant supplementation in horses affected by recurrent airway obstruction. J. Nutr. 2004, 134, S2065–S2067. [Google Scholar] [CrossRef] [PubMed]
- Estévez, M.; Cava, R. Effectiveness of rosemary essential oil as an inhibitor of lipid and protein oxidation: Contradictory effects in different types of frankfurters. Meat Sci. 2006, 72, 348–355. [Google Scholar] [CrossRef] [PubMed]
- Estévez, M.; Heinonen, M. Effect of phenolic compounds on the formation of α-amoniadipic and γ-glutamic semialdehydes from myofibrillar proteins oxidized by cooper, iron, and myoglobin. J. Agric. Food Chem. 2010, 58, 4448–4455. [Google Scholar] [CrossRef]
- Lund, M.N.; Hviid, M.S.; Skibsted, L.H. The combined effect of antioxidants and modified atmosphere packaging on protein and lipid oxidation in beef patties during chill storage. Meat Sci. 2007, 76, 226–233. [Google Scholar] [CrossRef]
- Pokorný, J.; Yanishlieva, N.; Gordon, M. Antioxidants in Food; CRC Press: Cambridge, UK, 2001. [Google Scholar]
- Soong, Y.Y.; Barlow, P.J. Antioxidant activity and phenolic content of selected fruit seeds. Food Chem. 2004, 88, 411–417. [Google Scholar] [CrossRef]
- Wu, X.; Gu, L.; Holden, J.; Haytowitz, D.B.; Gebhardt, S.E.; Beecher, G. Development of a database for total antioxidant capacity in foods: A preliminary study. J. Food Compos. Anal. 2004, 17, 407–422. [Google Scholar] [CrossRef]
- Angulo, J.; Mahecha, L.; Yepes, S.A.; Yepes, A.M.; Bustamante, G.; Jaramillo, H.; Valencia, E.; Villamil, T.; Gallo, J. Nutritional evaluation of fruit and vegetable waste as feedstuff for diets of lactating Holstein cows. J. Environ. Manag. 2012, 95, 210–214. [Google Scholar] [CrossRef]
- Rodríguez-Carpena, J.G.; Morcuende, D.; Estévez, M. Avocado, sunflower and olive oils as replacers of pork back-fat in burger patties: Effect on lipid composition, oxidative stability and quality traits. Meat Sci. 2012, 90, 106–115. [Google Scholar] [CrossRef]
- Utrera, M.; Rodríguez-Carpena, J.G.; Morcuende, D.; Estévez, M. Formation of lysine-derived oxidation products and loss of tryptophan during processing of porcine patties with added avocado byproducts. J. Agric. Food Chem. 2012, 60, 3917–3926. [Google Scholar] [CrossRef]
- Naht Dinh, T.T. Lipid and cholesterol composition of the longissimus muscle from Angus, Brahman and Romosinuano. Master’s Thesis, Texas Tech University, Lubbock, TX, USA, 2006. [Google Scholar]
- Simopoulos, A.P. Essential fatty acids in health and chronic disease. Am. J. Nutr. 1999, 70, 560S–569S. [Google Scholar] [CrossRef] [PubMed]
- Hernández-López, S.H.; Rodríguez-Carpena, J.G.; Lemus-Flores, C.; Grageola-Nuñez, F.; Estévez, M. Avocado waste for finishing pigs: Impact on muscle composition and oxidative stability during chilled storage. Meat Sci. 2016, 116, 186–192. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.P.; Chen, X.; Zhang, H.; Zhou, Y.M. Effects of dietary concentrations of methionine on growth performance and oxidative status of broiler chickens with different hatching weight. Breed. Poult. Sci. 2013, 54, 531–537. [Google Scholar] [CrossRef]
- Oskoueian, E.; Abdullah, N.; Idrus, Z.; Ebrahimi, M.; Goh, Y.M.; Shakeri, M. Palm kernel cake extract exerts hepatoprotective activity in heat-induced oxidative stress in chicken hepatocytes. BMC Complement. Altern. Med. 2014, 14, 368. [Google Scholar] [CrossRef] [PubMed]
- Cardinali, A.; Pati, S.; Minervini, F.; D’Antuono, I.; Linsalata, V.; Lattanzio, V. Verbascoside, isoverbascoside, and their derivatives recovered from olive mill wastewater as possible food antioxidants. J. Agric. Food Chem. 2012, 60, 1822–1829. [Google Scholar] [CrossRef] [PubMed]
- Rubio-Senent, F.; de Roos, B.; Duthie, G.; Fernandez-Bolanos, J.; Rodriguez-Gutierrez, G. Inhibitory and synergistic effects of natural olive phenols on human platelet aggregation and lipid peroxidation of microsomes from vitamin E-deficient rats. Eur. J. Nutr. 2015, 54, 1287–1295. [Google Scholar] [CrossRef] [PubMed]
- Arshad, M.S.; Anjum, F.M.; Khan, M.I.; Shahid, M.; Akhtar, S.; Sohaib, M. Wheat germ oil enrichment in broiler feed with α-lipoic acid to enhance the antioxidant potential and lipid stability of meat. Lipids Health Dis. 2013, 4, 164. [Google Scholar] [CrossRef]
- Nam, K.C.; Ahn, D.U. Use of antioxidants to reduce lipid oxidation and off-odor volatiles of irradiated pork homogenates and patties. Meat Sci. 2003, 63, 1–8. [Google Scholar] [CrossRef]
- Naveena, B.M.; Sen, A.R.; Vaithiyanathan, S.; Babji, Y.; Kondaiah, N. Comparative efficacy of pomegranate juice, pomegranate rind power extract and BHT as antioxidants in cooked chicken patties. Meat Sci. 2008, 80, 1304–1308. [Google Scholar] [CrossRef]
- Gerasopoulos, K.; Stagos, D.; Kokkas, S.; Petrotos, K.; Kantas, D.; Goulas, P.; Kouretas, D. Feed supplemented with byproducts from olive oil mill wastewater processing increases antioxidant capacity in broiler chickens. Food Chem. Toxicol. 2015, 82, 42–49. [Google Scholar] [CrossRef]
- Mujahid, A.; Akiba, Y.; Toyomizu, M. Olive oil-supplemented diet alleviates acute heat stress-induced mitochondrial ROS production in chicken skeletal muscle. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2009, 297, 690–698. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, R.A.; Narciso, D.D.; Bisinott, R.S.; Perdromo, M.C.; Ballou, M.A.; Dreher, M.; Santos, J.E.P. Effects of feeding polyphenols from pomegranate extract on health, growth, nutrient digestion, and immunocompetence of calves. J. Dairy Sci. 2010, 93, 4280–4291. [Google Scholar] [CrossRef] [PubMed]
- Jami, E.; Shabtay, A.; Nikbachat, M.; Yosef, E.; Miron, J.; Mizrahi, I. Effects of adding a concentrated pomegranate-residue extract to the ration of lactating cows on in vivo digestibility and profile of rumen bacterial population. J. Dairy Sci. 2012, 95, 5996–6005. [Google Scholar] [CrossRef] [PubMed]
- Shabtay, A.; Nikbachat, M.; Zenou, A.; Yosef, E.; Arkin, O.; Sneer, O.; Shwimmer, A.; Yaari, A.; Budmand, E.; Agmond, G.; et al. Effects of adding a concentrated pomegranate extract to the ration of lactating cows on performance and udder health parameters. Anim. Feed. Sci. Technol. 2012, 175, 24–32. [Google Scholar] [CrossRef]
- Safari, M.; Ghasemi, E.; Alikhani, M.; Ansari-Mahyari, S. Supplementation effects of pomegranate by-products on oxidative status, metabolic profile, and performance in transition dairy cows. J. Dairy Sci. 2018, 101, 11297–11309. [Google Scholar] [CrossRef]
- Natalello, A.; Hervas, G.; Toral, P.G.; Luciano, G.; Valenti, B.; Mendoza, A.G.; Pauselli, M.; Priolo, A.; Frutos, P. Bioactive compounds from pomegranate by-products increase the in vitro ruminal accumulation of potentially health promoting fatty acids. Anim. Feed. Sci. Technol. 2020, 259, 114355. [Google Scholar] [CrossRef]
- Andrés, A.I.; Petrón, M.J.; Adámez, J.D.; López, M.; Timón, M.L. Food by-products as potential antioxidant and antimicrobial additives in chill stored raw lamb patties. Meat Sci. 2017, 129, 62–70. [Google Scholar] [CrossRef]
- Kotsampasi, B.; Christodoulou, V.; Zotos, A.; Liakopoulou-Kyriakides, M.; Goulas, P.; Petrotos, K.; Natas, P.; Bampidis, V.A. Effects of dietary pomegranate byproduct silage supplementation on performance, carcass characteristics and meat quality of growing lambs. Anim. Feed. Sci. Technol. 2014, 197, 92–102. [Google Scholar] [CrossRef]
- Leontopoulos, S.V.; Petrotos, K.B.; Kokkora, M.I.; Giavasis, I.; Papaioannou, C. In vivo evaluation of liquid polyphenols obtained from OMWW as natural bio-chemicals against several fungal pathogens on tomato plants. Desalination Water Treat. 2016, 57, 20646–20660. [Google Scholar]
- Leontopoulos, S.V.; Giavasis, I.; Petrotos, K.; Makridis, C. Effect of different formulations of polyphenolic compounds obtained from OMWW on the growth of several fungal plant and food borne pathogens. Studies in vitro and in vivo. Agric. Agric. Sci. Procedia 2015, 4, 327–337. [Google Scholar] [CrossRef]
- Leontopoulos, S.; Mitsagga, C.; Giavasis, I.; Papaioannou, C.; Vasilakoglou1, I.; Petrotos, K. Potential synergistic action of liquid olive fruit polyphenol extract with aqueous extracts of solid wastes of pomegranate or/and orange juice industry as organic phyto-protective agents against important plant pathogens–Part 1 (in vitro Studies). Univers. J. Agric. Res. 2020, 8, 202–222. [Google Scholar] [CrossRef]
- Leistner, L. Basic aspects of food preservation by hurdle technology. Int. J. Food Microbiol. 2000, 55, 181–186. [Google Scholar] [CrossRef]
- Skenderidis, P.; Leontopoulos, S.; Petrotos, K.; Giavasis, I. Optimization of vacuum Microwave-Assisted Extraction of pomegranate fruits peels by the evaluation of extracts’ phenolic content and antioxidant activity. Foods 2020, 9, 1655. [Google Scholar] [CrossRef]
- Skenderidis, P.; Leontopoulos, S.; Petrotos, K.; Giavasis, I. Polyphenolic compounds derived from avocado (Persea americana) solid waste with Vacuum Microwave-Assisted Extraction. Sustainability 2021, 13, 2166. [Google Scholar] [CrossRef]
- Lambakis, D.; Skenderidis, P.; Leontopoulos, S. Technologies and extraction methods of polyphenolic compounds derived from pomegranate (Punica granatum) peels. A mini review. Processes 2021, 9, 236. [Google Scholar] [CrossRef]
- AOAC. Official Methods of Analysis, 16th ed.; Association of Official Analytical Chemists: Washington, DC, USA, 1999. [Google Scholar]
- Richard, E. Muck Silage microbiology and its control through additives. Rev. Bras. Zootec. 2010, 39, 183–191. [Google Scholar]
- Keles, M.S.; Taysi, S.; Sen, N.; Aksoy, H.; Akcay, F. Effect of corticosteroid therapy on serum and CSF malondialdehyde and antioxidant proteins in multiple sclerosis. Can. J. Neurol. Sci. 2001, 28, 141–143. [Google Scholar] [CrossRef]
- Gerasopoulos, K.; Stagos, D.; Petrotos, K.; Kokkas, S.; Kantas, D.; Goulas, P.; Kouretas, D. Feed supplemented with polyphenolic by-product from olive mill waste water processing improves the redox status in blood and tissues of piglets. Food Chem. Toxicol. 2015, 86, 319–327. [Google Scholar] [CrossRef] [PubMed]
- Folch, J.; Lees, M.; Stanley, G.H.S. A simple method for the isolation and purification of total lipids from animal tissues. J. Biol. Chem. 1957, 226, 497–509. [Google Scholar] [CrossRef]

| Days | pH | Lactic Acid Bacteria Log cfu/g | Yeasts Log cfu/g |
|---|---|---|---|
| 1 | 4.56 ± 0.03 | 7.35 ± 0.04 | 6.08 ± 0.08 |
| 3 | 4.50 ± 0.01 | 7.62 ± 0.05 | 7.36 ± 0.06 |
| 5 | 4.48 ± 0.02 | 8.79 ± 0.10 | 7.95 ± 0.05 |
| 12 | 4.27 ± 0.01 | 8.93 ± 0.04 | 8.56 ± 0.07 |
| 19 | 4.16 ± 0.01 | 8.88 ± 0.07 | 8.52 ± 0.08 |
| 26 | 4.42 ± 0.01 | 9.09 ± 0.06 | 8.70 ± 0.1 |
| 32 | 4.53 ± 0.02 | 9.02 ± 0.07 | 8.58 ± 0.11 |
| 40 | 4.59 ± 0.01 | 8.84 ± 0.04 | 8.18 ± 0.08 |
| Broiler’s Tissue Muscle | TBARS mg/kg MDA | |
|---|---|---|
| Group A (Avocado + pomegranate) | Leg | 0.076 ± 0.2 * |
| Breast | 0.076 ± 0.2 | |
| Group B (Control) | Leg | 0.149 ± 0.3 |
| Breast | 0.080 ± 0.2 |
| Fatty Acid % | ||||
|---|---|---|---|---|
| Group A (Avocado + Pomegranate + Corn) | Group B (Control: Corn Sillage) | Group A (Avocado + Pomegranate + Corn) | Group B (Control: Corn Sillage) | |
| (Chicken Breast) | (Chicken Breast) | (Chicken Leg) | (Chicken Leg) | |
| Myristic (C14:0) | 0.49 ± 0.0 | 0.55 ± 0.0 | 0.50 ± 0.0 | 0.53 ± 0.0 |
| Myristoleic (C14:1 cis-9) | 0.14 ± 0.0 | 0.16 ± 0.0 | 0.15 ± 0.0 | 0.16 ± 0.0 |
| Pentadecanoic (C15:0) | 0.08 ± 0.0 | 0.09 ± 0.0 | 0.08 ± 0.0 | 0.09 ± 0.0 |
| Palmitic (C16:0) | 24.99 ± 0.1 | 24.45 ± 0.2 | 22.83 ± 0.2 | 23.61 ± 0.2 |
| Palmitoleic cis (C16:1 cis-9) | 5.97 ± 0.1 | 5.17 ± 0.1 | 6.05 ± 0.1 | 5.54 ± 0.1 |
| Stearic (C18:0) | 6.19 ± 0.0 | 6.35 ± 0.1 | 5.84 ± 0.1 | 6.63 ± 0.1 |
| Oleic (C18:1 cis ω9) | 44.32 ± 0.2 | 43.27 ± 0.2 | 45.07 ± 0.2 | 43.18 ± 0.2 |
| Linoleic (C18:2 cis ω6) | 16.27 ± 0.1 | 18.44 ± 0.2 | 17.65 ± 0.2 | 18.37 ± 0.1 |
| Linolenic trans (C18:3 ω3) | 1.08 ± 0.0 | 0.27 ± 0.0 | 1.18 ± 0.0 | 0.27 ± 0.0 |
| g-Linolenic cis (C18:3 cis ω6) | 0.23 ± 0.0 | 0.17 ± 0.0 | 0.25 ± 0.0 | 0.29 ± 0.0 |
| Eicosatrienic (C20:3 cis ω6) | 0.11 ± 0.0 | 0.13 ± 0.0 | 0.15 ± 0.0 | 0.14 ± 0.0 |
| Eicositrienoic (C23:0) | 0.11 ± 0.0 | <0.01 | 0.20 ± 0.0 | 0.23 ± 0.0 |
| SFA | 31.86 | 31.45 | 29.46 | 31.09 |
| MUFA | 50.44 | 48.57 | 51.27 | 48.88 |
| PUFA | 17.69 | 19.02 | 19.24 | 19.06 |
| (PUFA+MUFA)/SFA | 2.14 | 2.15 | 2.39 | 2.19 |
| PUFA/SFA | 0.56 | 0.60 | 0.65 | 0.61 |
| Total ω3 fatty acids | 1.19 | 0.41 | 1.33 | 0.41 |
| Total ω6 fatty acids | 16.5 | 18.6 | 17.91 | 18.65 |
| ω6/ω3 | 13.87 | 45.37 | 13.47 | 45.49 |
| Fat (%) | 1.99 ± 0.04 b | 1.79 ± 0.03 a | 5.19 ± 0.03 A | 5.09 ± 0.03 A |
| Protein (%) | 22.57 ± 0.3 a | 22.86 ± 0.4 a | 20.04 ± 0.03 B | 19.76 ± 0.03 A |
| Moisture (%) | 75.07 ± 1.13 a | 75.21 ± 0.98 a | 74.96 ± 0.03 A | 75.18 ± 0.03 A |
| Collagen (%) | 0.79 ± 0.08 a | 0.88 ± 0.03 a | 1.07 ± 0.03 A | 1.06 ± 0.03 A |
| Compounds | Group A | Group B |
|---|---|---|
| Total proteins * | 16.38 | 18.90 |
| Total fat * | 4.43 | 4.69 |
| Total fibre * | 2.61 | 2.34 |
| Total ash * | 4.23 | 4.41 |
| Carbohydrates * | 59.83 | 55.42 |
| Sugars * | 2.4 | 2.2 |
| Moisture * | 10.35 | 10.80 |
| Ca * | 0.25 | 0.9 |
| P * | 0.13 | 0.46 |
| Available P * | 0.32 | 0.41 |
| Na * | 0.14 | 0.16 |
| Methionine * | 0.40 | 0.43 |
| Lycine * | 0.93 | 1,08 |
| Energy per kg | 3.05 Mj/728.46 Kcal | 12.2 Mj/2.91 Kcal |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Leontopoulos, S.; Skenderidis, P.; Petrotos, K.; Giavasis, I. Corn Silage Supplemented with Pomegranate (Punica granatum) and Avocado (Persea americana) Pulp and Seed Wastes for Improvement of Meat Characteristics in Poultry Production. Molecules 2021, 26, 5901. https://doi.org/10.3390/molecules26195901
Leontopoulos S, Skenderidis P, Petrotos K, Giavasis I. Corn Silage Supplemented with Pomegranate (Punica granatum) and Avocado (Persea americana) Pulp and Seed Wastes for Improvement of Meat Characteristics in Poultry Production. Molecules. 2021; 26(19):5901. https://doi.org/10.3390/molecules26195901
Chicago/Turabian StyleLeontopoulos, Stefanos, Prodromos Skenderidis, Konstantinos Petrotos, and Ioannis Giavasis. 2021. "Corn Silage Supplemented with Pomegranate (Punica granatum) and Avocado (Persea americana) Pulp and Seed Wastes for Improvement of Meat Characteristics in Poultry Production" Molecules 26, no. 19: 5901. https://doi.org/10.3390/molecules26195901
APA StyleLeontopoulos, S., Skenderidis, P., Petrotos, K., & Giavasis, I. (2021). Corn Silage Supplemented with Pomegranate (Punica granatum) and Avocado (Persea americana) Pulp and Seed Wastes for Improvement of Meat Characteristics in Poultry Production. Molecules, 26(19), 5901. https://doi.org/10.3390/molecules26195901









