Analysis of Flavonoid Metabolites in Watercress (Nasturtium officinale R. Br.) and the Non-Heading Chinese Cabbage (Brassica rapa ssp. chinensis cv. Aijiaohuang) Using UHPLC-ESI-MS/MS
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Materials and Methods
2.2. Determination of Total Flavonoids Content
2.3. Extraction Process
2.4. Chromatographic and Mass Spectrometry of Analysis Conditions
2.5. Qualitative and Quantitative Principles of Metabolites
2.6. Statistical Analysis
3. Results
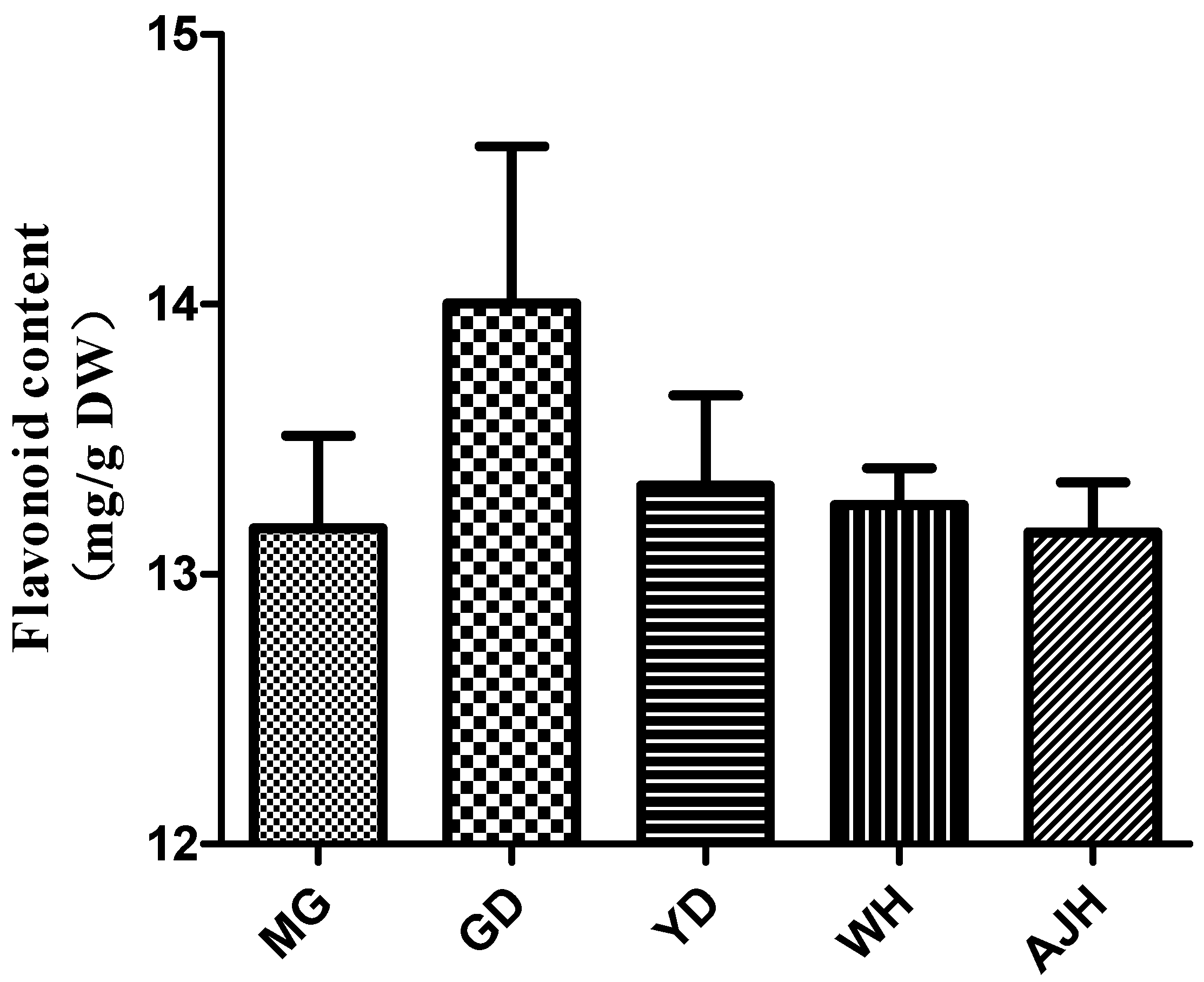
3.1. Determination of Total Flavonoid Content
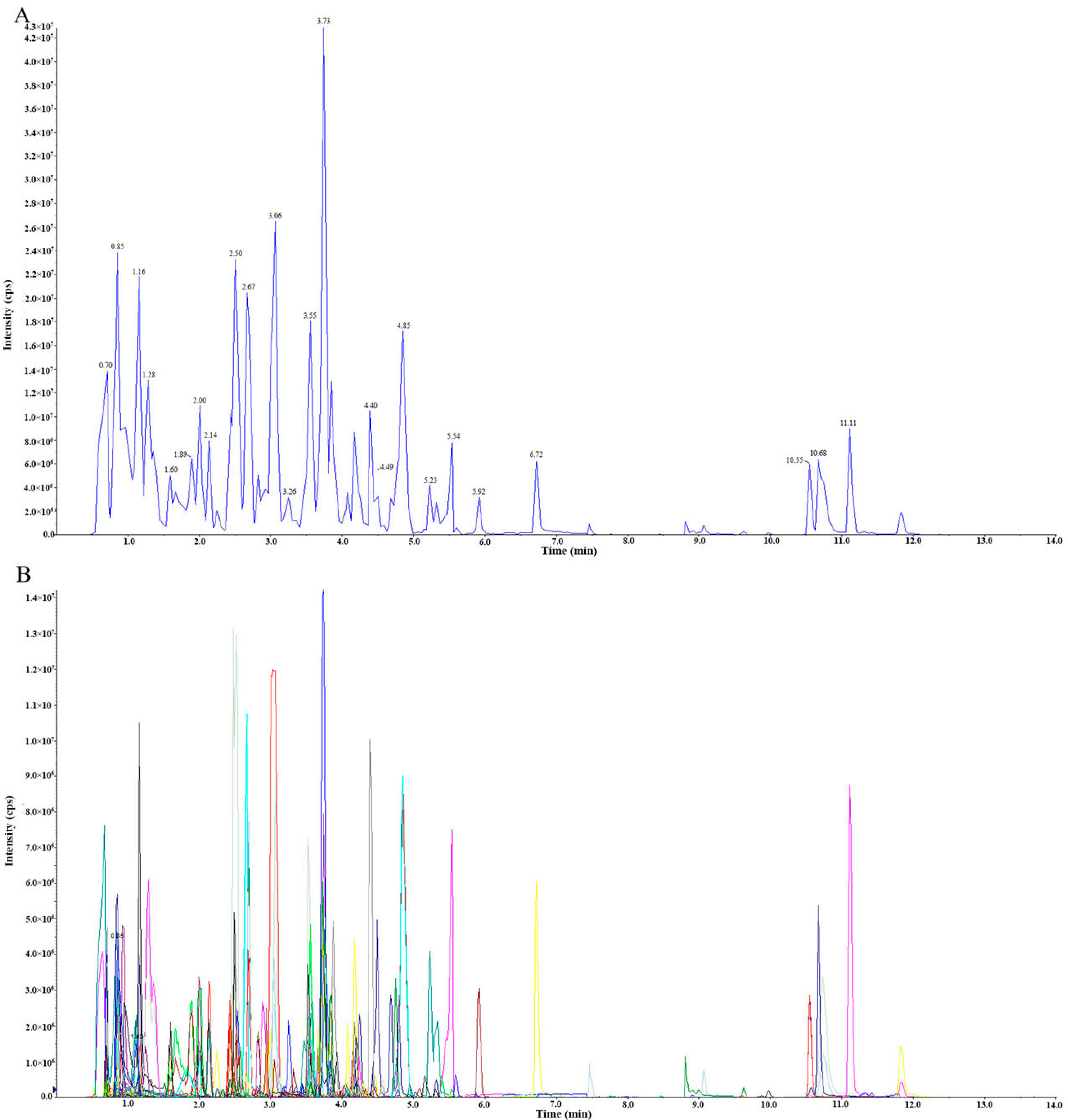
3.2. Qualitative and Quantitative Analyses of Metabolites and Quality Control (QC) Analysis of Samples
3.3. Principal Component Analysis (PCA) for the Different Varieties
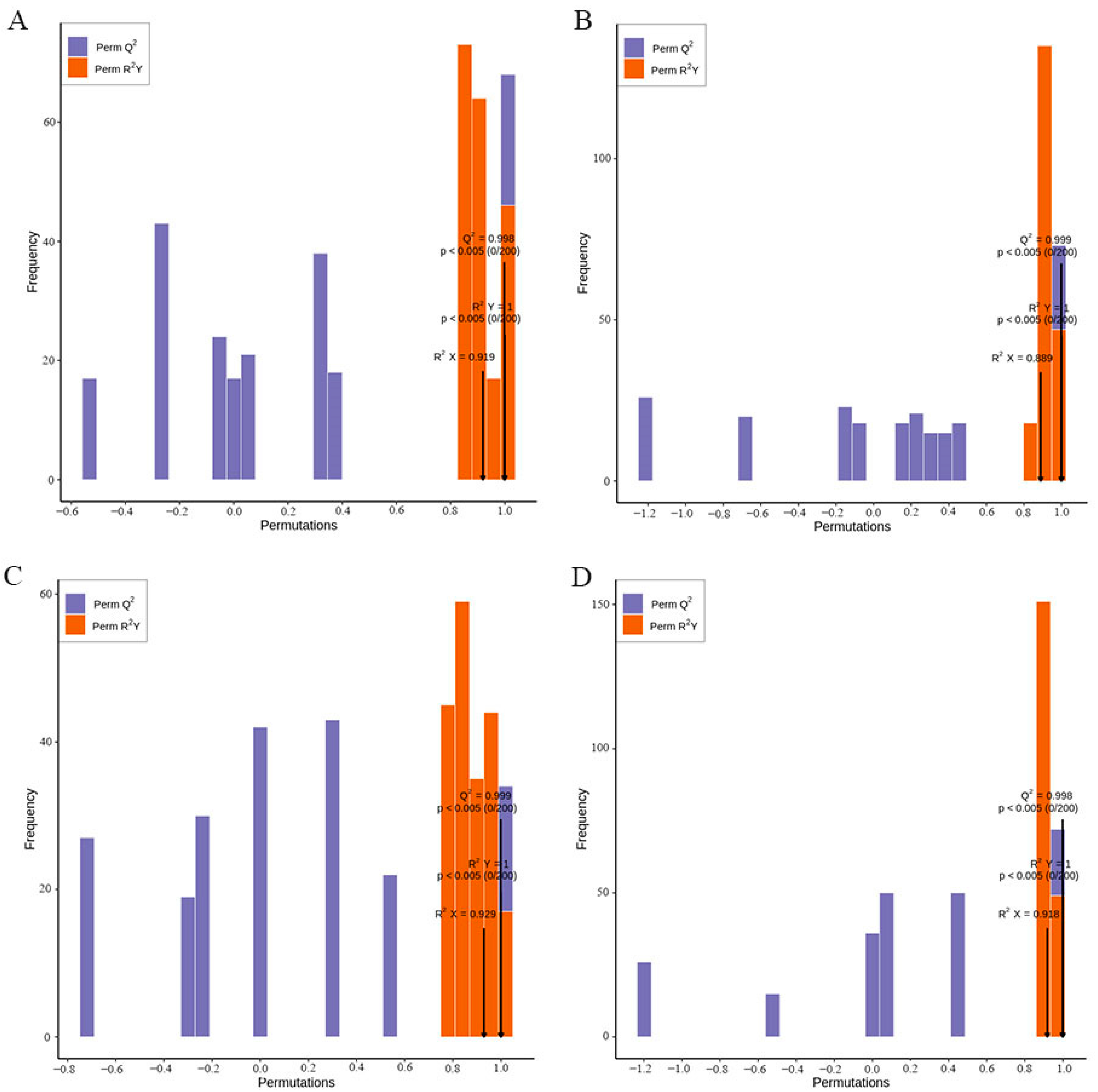
3.4. Orthogonal Projections to Latent Structure-Discriminant Analysis (OPLS-DA)
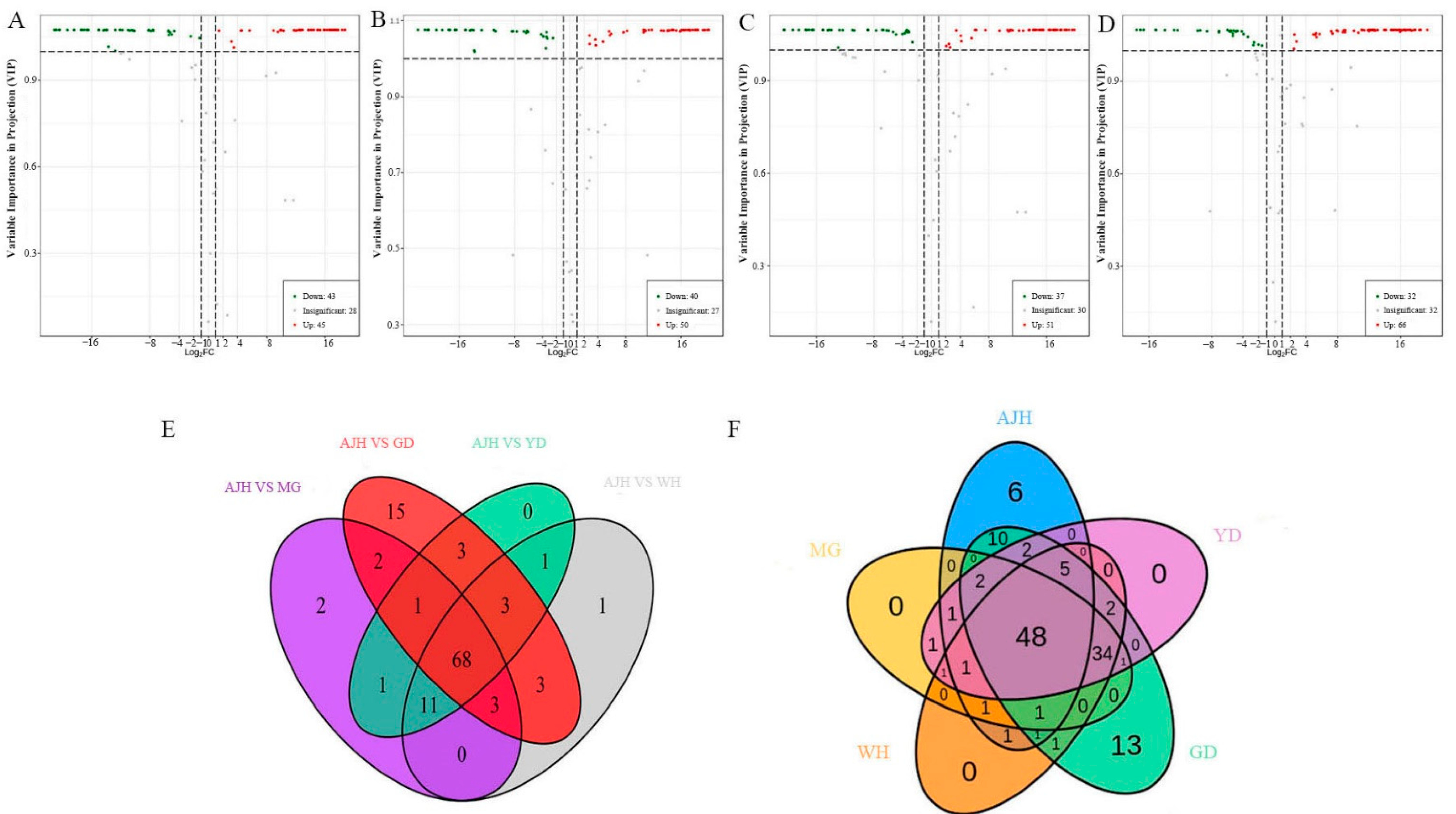
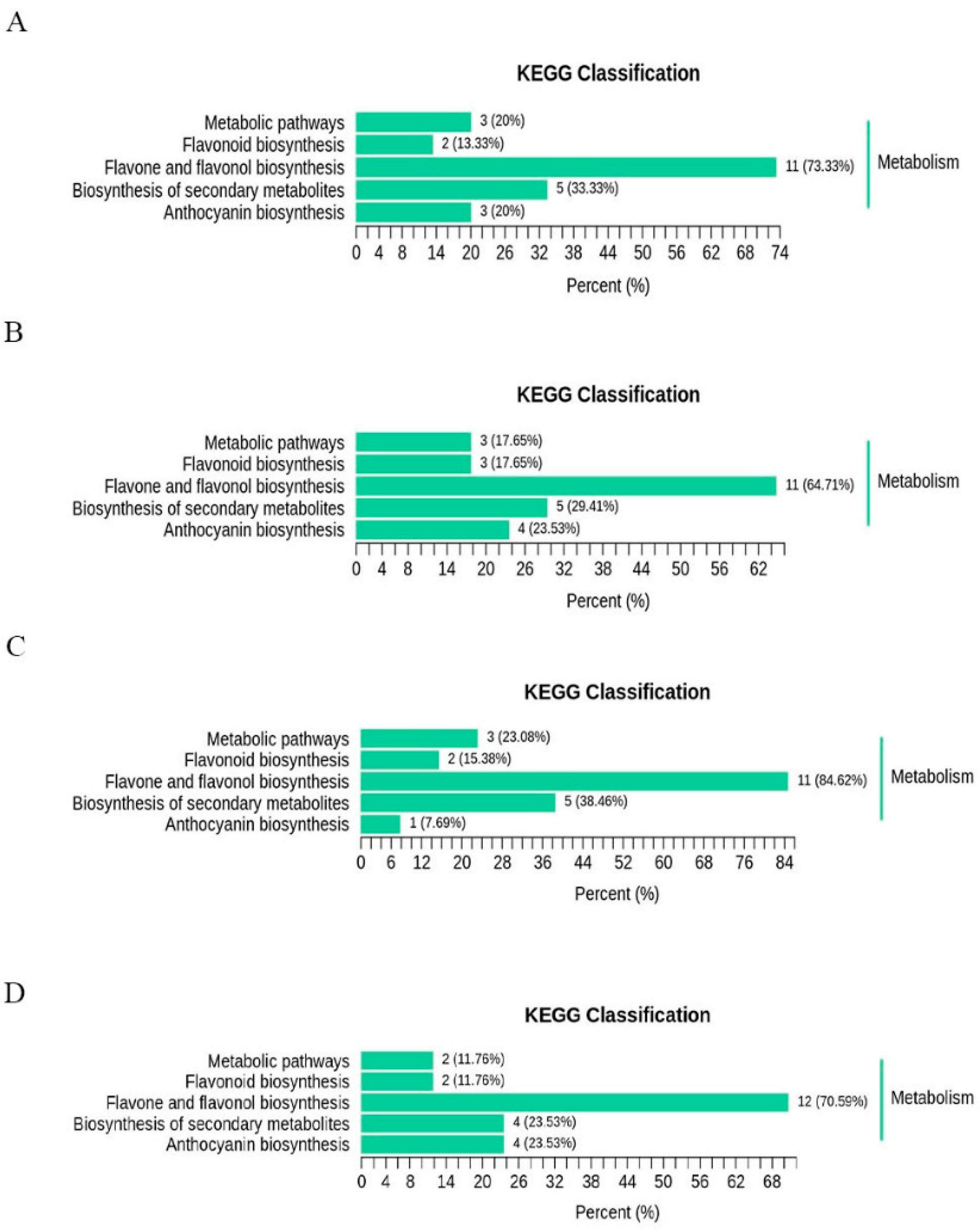
3.5. Screening and Kegg Analysis of Flavonoid Differential Metabolites
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Di Noia, J. Defining powerhouse fruits and vegetables: A nutrient density approach. Prev. Chronic Dis. 2014, 11, 3–7. [Google Scholar] [CrossRef] [PubMed]
- Goda, Y.; Hoshino, K.; Akiyama, H.; Ishikawa, T.; Abe, Y.; Nakamura, T.; Otsuka, H.; Takeda, Y.; Tanimura, A.; Toyoda, M. Constituents in watercress: Inhibitors of histamine release from RBL-2H3 cells induced by antigen stimulation. Biol. Pharm. Bull. 1999, 22, 1319–1326. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Sánchez, A.; Gil-Izquierdo, A.; Gil, M.I.; Ferreres, F. A comparative study of flavonoid compounds, vitamin C, and antioxidant properties of baby leaf cruciferae species. J. Agric. Food Chem. 2008, 56, 2330–2340. [Google Scholar] [CrossRef] [PubMed]
- Brennan, P.; Hsu, C.C.; Moullan, N.; Szeszenia-Dabrowska, N.; Lissowska, J.; Zaridze, D.; Rudnai, P.; Fabianova, E.; Mates, D.; Bencko, V.; et al. Effect of cruciferae vegetables on lung cancer in patients stratified by genetic status: A mendelian randomisation approach. Lancet 2005, 366, 1558–1560. [Google Scholar] [CrossRef]
- Cohen, J.H.; Kristal, A.R.; Stanford, J.L. Fruit and vegetable intakes and prostate cancer risk. J. Natl. Cancer Inst. 2000, 92, 61–68. [Google Scholar] [CrossRef]
- Gill, C.I.R.; Haldar, S.; Boyd, L.A.; Bennett, R.; Whiteford, J.; Butler, M.; Pearson, J.R.; Bradbury, I.; Rowland, I.R. Watercress supplementation in diet reduces lymphocyte DNA damage and alters blood antioxidant status in healthy adults. Am. J. Clin. Nutr. 2007, 85, 504–510. [Google Scholar] [CrossRef]
- Slattery, M.L.; Kampman, E.; Samowitz, W.; Caan, B.J.; Potter, J.D. Interplay between dietary inducers of GST and the GSTM-1 genotype in colon cancer. Int. J. Cancer 2000, 87, 728–733. [Google Scholar] [CrossRef]
- Voorrips, L.E.; Goldbohm, R.A.; Van Poppel, G.; Sturmans, F.; Hermus, R.J.J.; Van Den Brandt, P.A. Vegetable and fruit consumption and risks of colon and rectal cancer in a prospective cohort study: The Netherlands Cohort Study on Diet and Cancer. Am. J. Epidemiol. 2000, 152, 1081–1092. [Google Scholar] [CrossRef]
- Witte, J.S.; Longnecker, M.P.; Bird, C.L.; Lee, E.R.; Frankl, H.D.; Haile, R.W. Relation of vegetable, fruit, and grain consumption to colorectal adenomatous polyps. Am. J. Epidemiol. 1996, 144, 1015–1025. [Google Scholar] [CrossRef]
- Zhang, S.M.; Hunter, D.J.; Rosner, B.A.; Giovannucci, E.L.; Colditz, G.A.; Speizer, F.E.; Willett, W.C. Intakes of fruits, vegetables, and related nutrients and the risk of non-Hodgkin’s lymphoma among women. Cancer Epidemiol. Biomark. Prev. 2000, 9, 477–485. [Google Scholar]
- Cavell, B.E.; Syed Alwi, S.S.; Donlevy, A.; Packham, G. Anti-angiogenic effects of dietary isothiocyanates: Mechanisms of action and implications for human health. Biochem. Pharmacol. 2011, 81, 327–336. [Google Scholar] [CrossRef] [PubMed]
- Lai, K.C.; Hsu, S.C.; Kuo, C.L.; Ip, S.W.; Yang, J.S.; Hsu, Y.M.; Huang, H.Y.; Wu, S.H.; Chung, J.G. Phenethyl isothiocyanate inhibited tumor migration and invasion via suppressing multiple signal transduction pathways in human colon cancer HT29 cells. J. Agric. Food Chem. 2010, 58, 11148–11155. [Google Scholar] [CrossRef] [PubMed]
- Rose, P. 7-Methylsulfinylheptyl and 8-methylsulfinyloctyl isothiocyanates from watercress are potent inducers of phase II enzymes. Carcinogenesis 2000, 21, 1983–1988. [Google Scholar] [CrossRef] [PubMed]
- Rose, P.; Huang, Q.; Ong, C.N.; Whiteman, M. Broccoli and watercress suppress matrix metalloproteinase-9 activity and invasiveness of human MDA-MB-231 breast cancer cells. Toxicol. Appl. Pharmacol. 2005, 209, 105–113. [Google Scholar] [CrossRef]
- Syed Alwi, S.S.; Cavell, B.E.; Telang, U.; Morris, M.E.; Parry, B.M.; Packham, G. In vivo modulation of 4E binding protein 1 (4E-BP1) phosphorylation by watercress: A pilot study. Br. J. Nutr. 2010, 104, 1288–1296. [Google Scholar] [CrossRef]
- Steinkellner, H.; Rabot, S.; Freywald, C.; Nobis, E.; Scharf, G.; Chabicovsky, M.; Knasmüller, S.; Kassie, F. Effects of cruciferae vegetables and their constituents on drug metabolizing enzymes involved in the bioactivation of DNA-reactive dietary carcinogens. Mutat. Res. Fundam. Mol. Mech. Mutagenesis 2001, 480, 285–297. [Google Scholar] [CrossRef]
- Töpfer, N.; Kleessen, S.; Nikoloski, Z. Integration of metabolomics data into metabolic networks. Front. Plant Sci. 2015, 6, 49. [Google Scholar] [CrossRef]
- Gutermuth, J.; Bewersdorff, M.; Traidl-Hoffmann, C.; Ring, J.; Mueller, M.J.; Behrendt, H.; Jakob, T. Immunomodulatory effects of aqueous birch pollen extracts and phytoprostanes on primary immune responses in vivo. J. Allergy Clin. Immunol. 2007, 120, 293–299. [Google Scholar] [CrossRef]
- Zoerner, A.A.; Gutzki, F.M.; Batkai, S.; May, M.; Rakers, C.; Engeli, S.; Jordan, J.; Tsikas, D. Quantification of endocannabinoids in biological systems by chromatography and mass spectrometry: A comprehensive review from an analytical and biological perspective. Biochim. Et Biophys. Acta Mol. Cell Biol. Lipids 2011, 1811, 706–723. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Lu, Y.; Wang, R.; Liu, S.; Hu, X.; Wang, H. Transport and metabolic profiling studies of amentoflavone in Caco-2 cells by UHPLC-ESI-MS/MS and UHPLC-ESI-Q-TOF-MS/MS. J. Pharm Biomed. Anal. 2020, 189, 113441. [Google Scholar] [CrossRef]
- Zhou, C.; Luo, Y.; Lei, Z.; Wei, G. UHPLC-ESI-MS Analysis of Purified Flavonoids Fraction from Stem of Dendrobium denneaum Paxt. and Its Preliminary Study in Inducing Apoptosis of HepG2 Cells. Evid Based Complement. Alternat Med. 2018, 2018, 1–10. [Google Scholar] [CrossRef]
- Chen, W.; Gong, L.; Guo, Z.; Wang, W.; Zhang, H.; Liu, X.; Yu, S.; Xiong, L.; Luo, J. A novel integrated method for large-scale detection, identification, and quantification of widely targeted metabolites: Application in the study of rice metabolomics. Mol. Plant. 2013, 6, 1769–1780. [Google Scholar] [CrossRef]
- Wang, S.; Tu, H.; Wan, J.; Chen, W.; Liu, X.; Luo, J.; Xu, J.; Zhang, H. Spatio-temporal distribution and natural variation of metabolites in citrus fruits. Food Chem. 2016, 199, 8–17. [Google Scholar] [CrossRef] [PubMed]
- Boccard, J.; Rutledge, D.N. A consensus orthogonal partial least squares discriminant analysis (opls-da) strategy for multiblock omics data fusion. Anal. Chim. Acta 2013, 769, 30–39. [Google Scholar] [CrossRef]
- Yamamoto, S.; Sobue, T.; Kobayashi, M.; Sasaki, S.; Tsugane, S.; Tsugane, S.; Sasaki, S.; Sobue, T.; Ogata, J.; Baba, S.; et al. Soy, isoflavones, and breast cancer risk in Japan. J. Natl. Cancer Inst. 2003, 95, 1881–1882. [Google Scholar] [CrossRef][Green Version]
- Akhlaghi, M.; Ghasemi Nasab, M.; Riasatian, M.; Sadeghi, F. Soy isoflavones prevent bone resorption and loss, a systematic review and meta-analysis of randomized controlled trials. Crit. Rev. Food Sci. Nutr. 2020, 60, 2327–2341. [Google Scholar] [CrossRef]
- Fotsis, T.; Pepper, M.; Adlercreutz, H.; Fleischmann, G.; Hase, T.; Montesano, R.; Schweigerer, L. Genistein, a dietary-derived inhibitor of in vitro angiogenesis. Proc. Natl. Acad. Sci. USA 1993, 90, 2690–2694. [Google Scholar] [CrossRef]
- Shao, Z.M.; Shen, Z.Z.; Liu, C.H.; Sartippour, M.R.; Go, V.L.; Heber, D.; Nguyen, M. Curcumin exerts multiple suppressive effects on human breast carcinoma cells. Int. J. Cancer 2002, 98, 234–240. [Google Scholar] [CrossRef] [PubMed]
- Brown, C.R.; Culley, D.; Yang, C.P.; Durst, R.; Wrolstad, R. Variation of anthocyanin and carotenoid contents and associated antioxidant values in potato breeding lines. J. Am. Soc. Hortic. Sci. 2005, 130, 174–180. [Google Scholar] [CrossRef]
- Silva, S.; Costa, E.M.; Calhau, C.; Morais, R.M.; Pintado, M.E. Anthocyanin extraction from plant tissues: A review. Crit. Rev. Food Sci. Nutr. 2017, 57, 3072–3083. [Google Scholar] [CrossRef]
- Han, X.Z.; Ren, D.M.; Fan, P.H.; Shen, T.; Lou, H.X. Protective effects of naringenin-7-o-glucoside on doxorubicin-induced apoptosis in H9C2 cells. Eur. J. Pharmacol. 2008, 581, 47–53. [Google Scholar] [CrossRef]
- Wu, P.; Liu, A.L.; Li, L.J. Metabolomics and transcriptome analysis of the biosynthesis mechanism of flavonoids in the seeds of euryale ferox salisb at different developmental stages. Mol. Genet. Genom. 2021, 296, 953–970. [Google Scholar] [CrossRef]
- Qiu, W.W.; Su, W.Q.; Cai, Z.Y.; Dong, L.; Li, C.B.; Xin, M.; Fang, W.K.; Liu, Y.Q.; Wang, X.M.; Huang, Z.B.; et al. Combined Analysis of Transcriptome and Metabolome Reveals the Potential Mechanism of Coloration and Fruit Quality in Yellow and Purple Passiflora edulis Sims. J. Agric. Food Chem. 2020, 68, 12096–12106. [Google Scholar] [CrossRef]
- Nguyen, V.S.; Shi, L.; Luan, F.Q.; Wang, Q.A. Synthesis of kaempferide mannich base derivatives and their antiproliferative activity on three human cancer cell lines. Acta Biochim. Pol. 2015, 62, 547–552. [Google Scholar] [CrossRef] [PubMed]
- Kang, J.; Kim, E.; Kim, W.; Seong, K.M.; Youn, H.; Kim, J.W.; Kim, J.; Youn, B. Rhamnetin and cirsiliol induce radiosensitization and inhibition of epithelial-mesenchymal transition (emt) by mir-34a-mediated suppression of notch-1 expression in non-small cell lung cancer cell lines. J. Biol. Chem. 2013, 288, 27343–27357. [Google Scholar] [CrossRef] [PubMed]
- Pollini, L.; Rocchi, R.; Cossignani, L.; Mañes, J.; Compagnone, D.; Blasi, F. Phenol Profiling and Nutraceutical Potential of Lycium spp. Leaf Extracts Obtained with Ultrasound and Microwave Assisted Techniques. Antioxidants 2019, 8, 260. [Google Scholar] [CrossRef] [PubMed]

| UHPLC Conditions | Parameters | |
|---|---|---|
| The chromatographic columns | Agilent SB-C18 1.8 µm, 2.1 mm × 100 mm | |
| Mobile phase | Phase A: ultra-pure water (0.1% formic acid was added) phase B: acetonitrile (0.1% formic acid was added) | |
| Gradient program | 0 min | 95:5 (v:v) |
| 9.0 min | 5:95 (v:v) | |
| 10.0 min | 5:95 (v:v) | |
| 11.0 min | 95:5 (v:v) | |
| 14.0 min | 95:5 (v:v) | |
| Flow rate | 0.40 mL/min | |
| Temperature | 40 °C | |
| Injection volume | 5 μL | |
| Mass Spectrometry Condition | Parameters | |
|---|---|---|
| Ion source | Turbo spray | |
| Source temperature | 550 °C | |
| Ion spray voltage | 5500 V | |
| Ion source | Gas I | 55 psi |
| Gas II | 60 psi | |
| Curtain gas | 25 psi | |
| Collision gas | High | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ma, X.; Ding, Q.; Hou, X.; You, X. Analysis of Flavonoid Metabolites in Watercress (Nasturtium officinale R. Br.) and the Non-Heading Chinese Cabbage (Brassica rapa ssp. chinensis cv. Aijiaohuang) Using UHPLC-ESI-MS/MS. Molecules 2021, 26, 5825. https://doi.org/10.3390/molecules26195825
Ma X, Ding Q, Hou X, You X. Analysis of Flavonoid Metabolites in Watercress (Nasturtium officinale R. Br.) and the Non-Heading Chinese Cabbage (Brassica rapa ssp. chinensis cv. Aijiaohuang) Using UHPLC-ESI-MS/MS. Molecules. 2021; 26(19):5825. https://doi.org/10.3390/molecules26195825
Chicago/Turabian StyleMa, Xiaoqing, Qiang Ding, Xilin Hou, and Xiong You. 2021. "Analysis of Flavonoid Metabolites in Watercress (Nasturtium officinale R. Br.) and the Non-Heading Chinese Cabbage (Brassica rapa ssp. chinensis cv. Aijiaohuang) Using UHPLC-ESI-MS/MS" Molecules 26, no. 19: 5825. https://doi.org/10.3390/molecules26195825
APA StyleMa, X., Ding, Q., Hou, X., & You, X. (2021). Analysis of Flavonoid Metabolites in Watercress (Nasturtium officinale R. Br.) and the Non-Heading Chinese Cabbage (Brassica rapa ssp. chinensis cv. Aijiaohuang) Using UHPLC-ESI-MS/MS. Molecules, 26(19), 5825. https://doi.org/10.3390/molecules26195825







