Anti-Inflammatory Effect of Charantadiol A, Isolated from Wild Bitter Melon Leaf, on Heat-Inactivated Porphyromonas gingivalis-Stimulated THP-1 Monocytes and a Periodontitis Mouse Model
Abstract
:1. Introduction
2. Results and Discussion
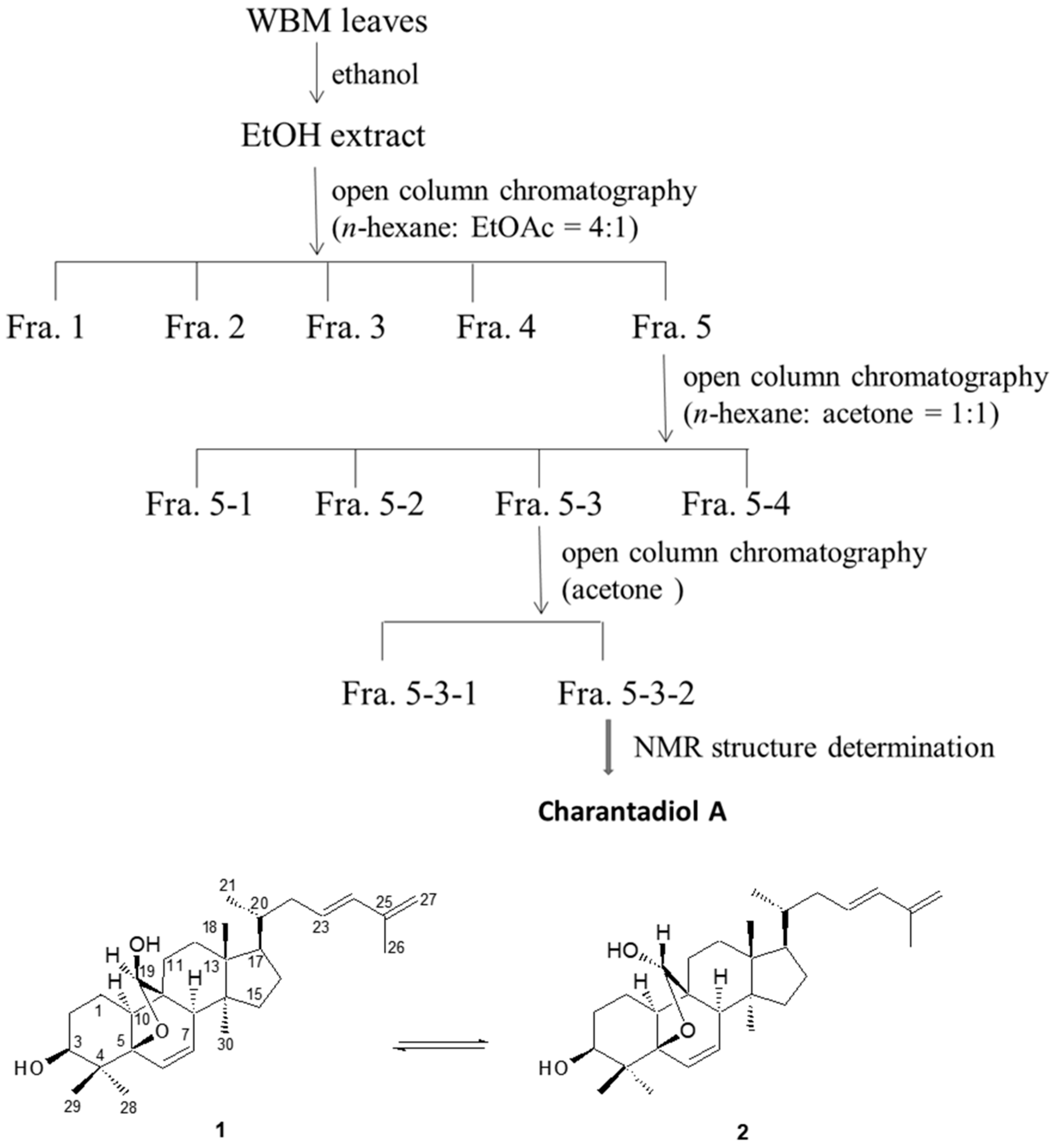
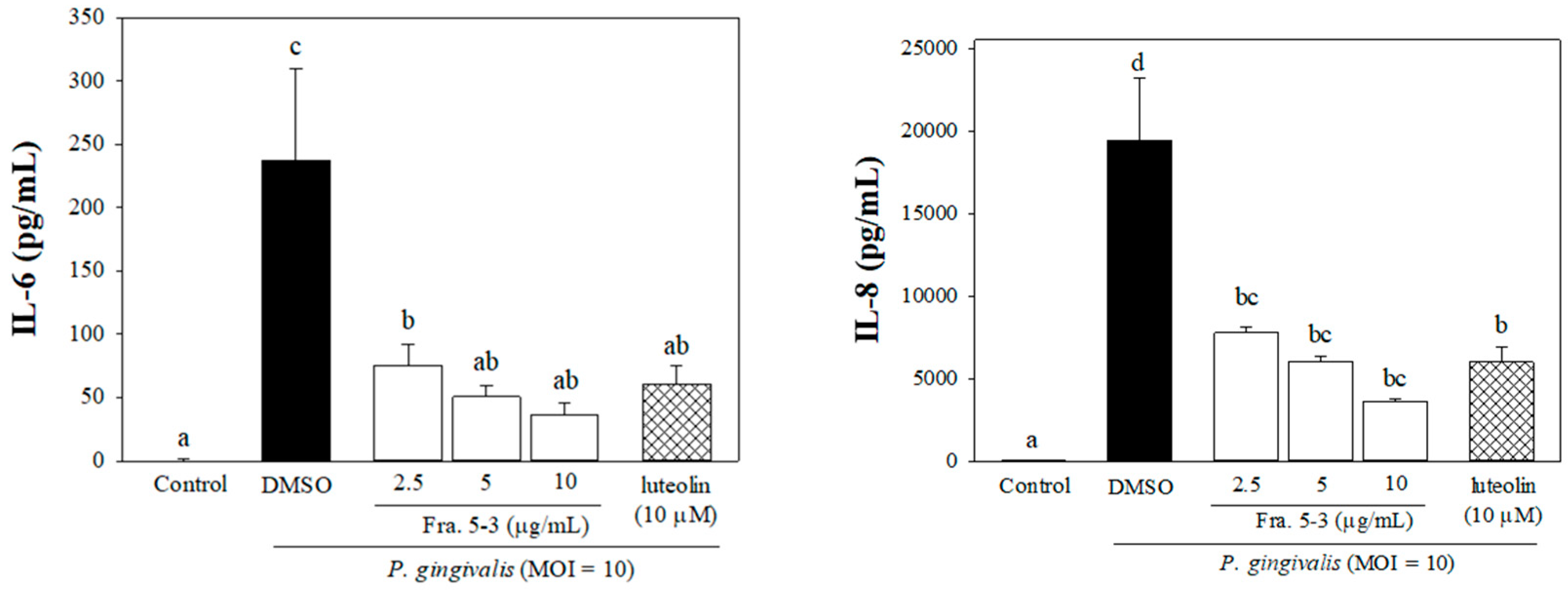
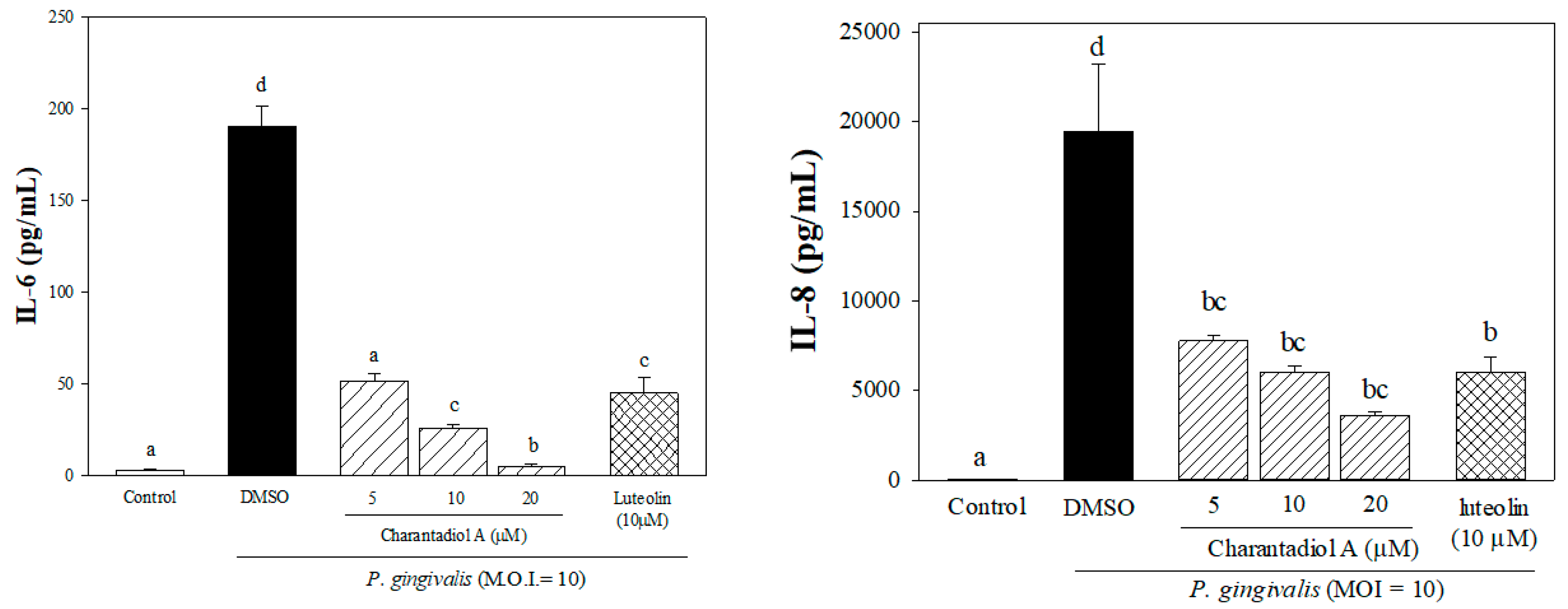
2.1. Effects of Sub-Fractions from Leaf Extract of Wild Bitter Melon and Charantadiol A on P. gingivalis-Induced Cytokines in THP-Cells
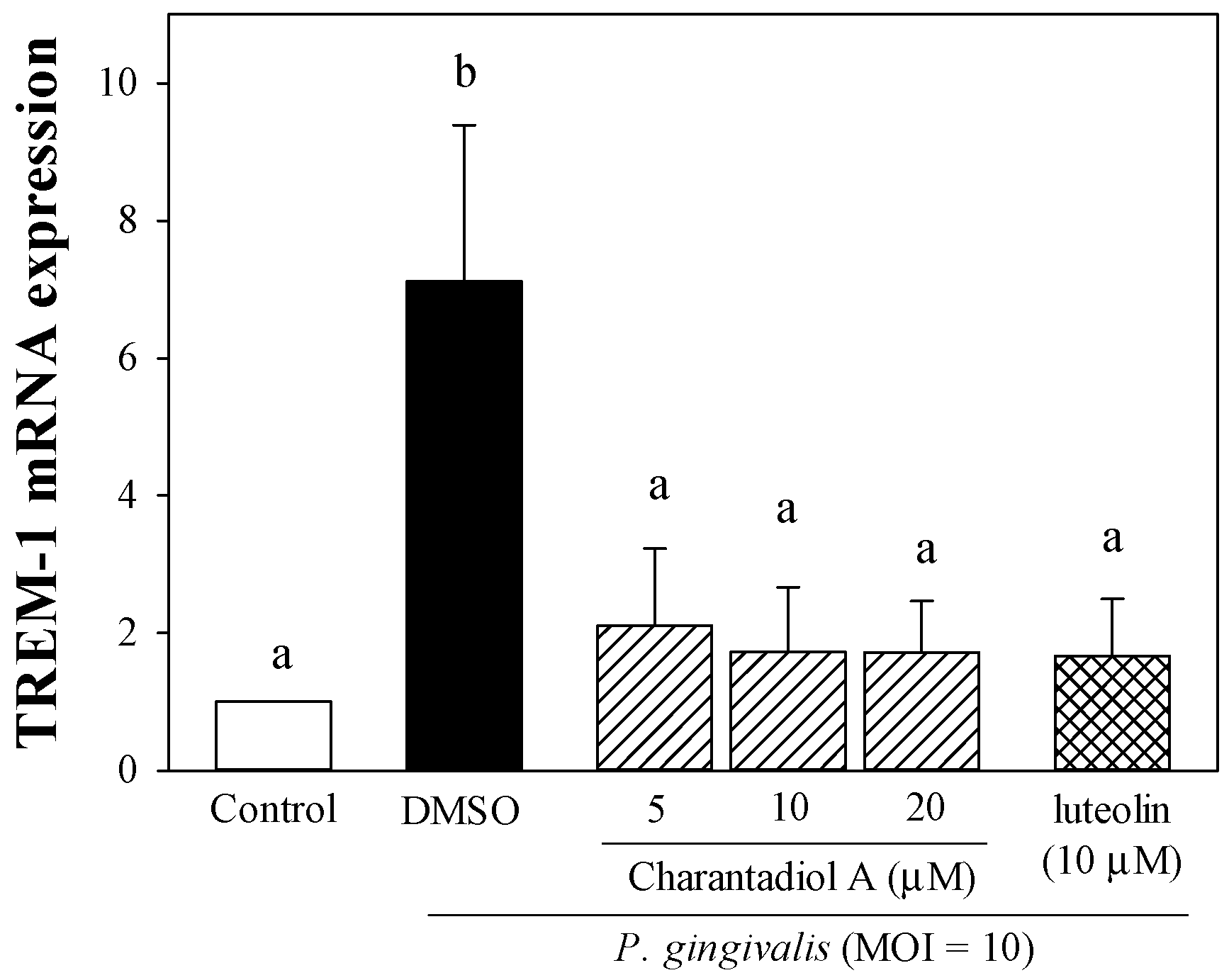
2.2. Effect of Charantadiol A on TREM-1 mRNA Expression Level in P. gingivalis-Stimulated THP-1 Cells
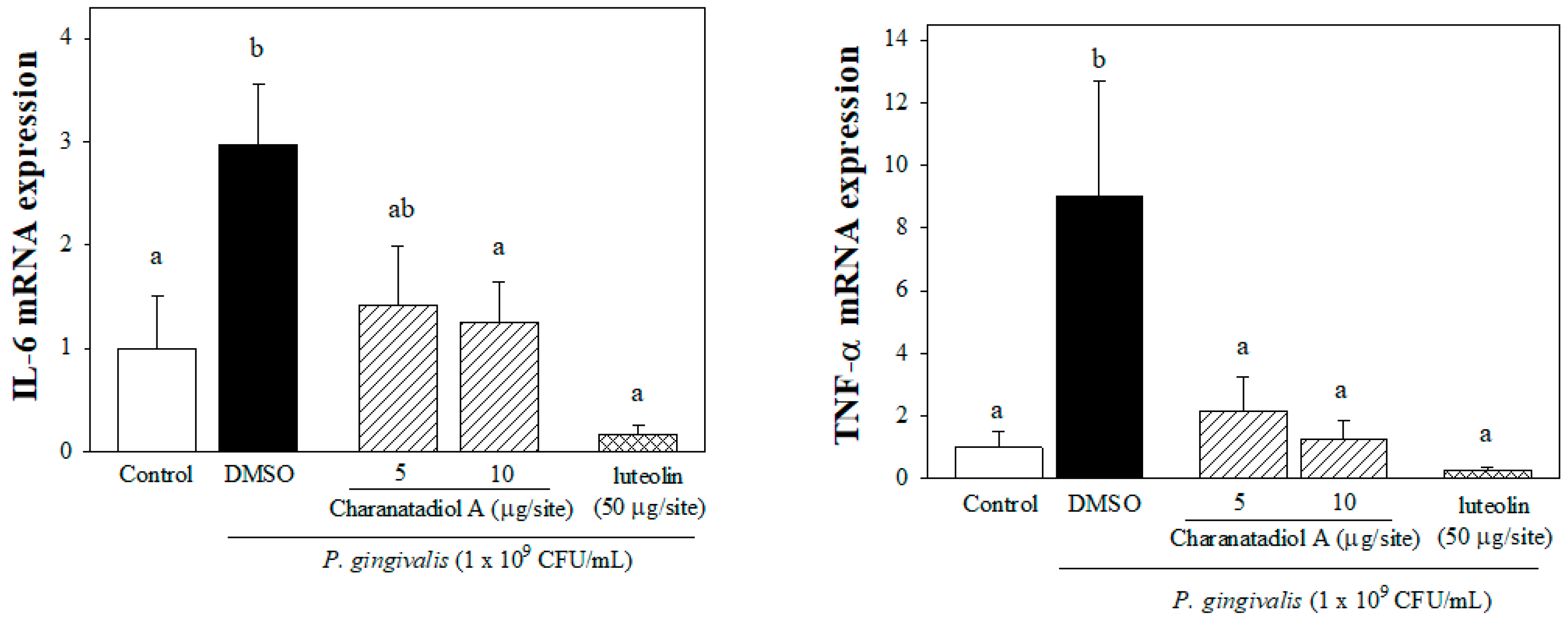
2.3. Effect of Charantadiol A on IL-6 and TNFα mRNA expression in P. gingivalis-Stimulated Gingival Tissue of Mice
3. Materials and Methods
3.1. Plant Materials
3.2. Isolation and Determination of Charantadiol A
3.3. Cell Culture
3.4. Preparation of Heat-Inactivated P. gingivalis
3.5. Stimulation of THP-1 Cells with P. gingivalis and Cytokine Measurements
3.6. RNA Extraction of THP-1 Cells and Quantitative Real-Time Polymerase Chain Reaction (PCR)
3.7. Effect of Charantadiol A on P. gingivalis-Induced Cytokine Expression In Vivo
3.8. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Nakayama, M.; Ohara, N. Molecular mechanisms of Porphyromonas gingivalis-host cell interaction on periodontal diseases. Jpn. Dent. Sci. Rev. 2017, 53, 134–140. [Google Scholar] [CrossRef]
- How, K.Y.; Song, K.P.; Chan, K.G. Porphyromonas gingivalis: An overview of periodontopathic pathogen below the gum line. Front. Microbiol. 2016, 7, 53. [Google Scholar] [CrossRef]
- Ramadan, D.E.; Hariyani, N.; Indrawati, R.; Ridwan, R.D.; Diyatri, I. Cytokines and chemokines in periodontitis. Eur. J. Dent. 2020, 14, 483–495. [Google Scholar] [PubMed]
- Cekici, A.; Kantarci, A.; Hasturk, H.; Van Dyke, T.E. Inflammatory and immune pathways in the pathogenesis of periodontal disease. Periodontology 2000 2014, 64, 57–80. [Google Scholar] [CrossRef] [Green Version]
- Huang, Y.K.; Tseng, K.F.; Tsai, P.H.; Wang, J.S.; Lee, C.Y.; Shen, M.Y. IL-8 as a potential therapeutic target for periodontitis and its inhibition by caffeic acid phenethyl ester in vitro. Int. J. Mol. Sci. 2021, 22, 3641. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Choi, S.I.; Kim, G.H.; Imm, J.Y. Anti-inflammatory effect of Ecklonia cava extract on Porphyromonas gingivalis lipopolysaccharide-stimulated macrophages and a periodontitis rat model. Nutrients 2019, 11, 1143. [Google Scholar] [CrossRef] [Green Version]
- Tsou, S.H.; Hu, S.W.; Yang, J.J.; Yan, M.; Lin, Y.Y. Potential oral health care agent from coffee against virulence factor of periodontitis. Nutrients 2019, 11, 2235. [Google Scholar] [CrossRef] [Green Version]
- Seong, J.; Lee, J.; Lim, Y.K.; Yoon, W.J.; Jung, S.; Kook, J.K.; Lee, T.H. Osmunda japonica extract suppresses pro-inflammatory cytokines by downregulating NF-κB activation in periodontal ligament fibroblasts infected with oral pathogenic bacteria. Int. J. Mol. Sci. 2020, 21, 2453. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jia, S.; Shen, M.; Zhang, F.; Xie, J. Recent advances in Momordica charantia: Functional components and biological activities. Int. J. Mol. Sci. 2017, 18, 2555. [Google Scholar] [CrossRef] [Green Version]
- Chang, M.L.; Lin, Y.T.; Kung, H.N.; Hou, Y.C.; Liu, J.J.; Pan, M.H.; Chen, H.L.; Yu, C.H.; Tsai, P.J. Triterpenoid-enriched extract of wild bitter melon leaf alleviates hepatic fibrosis by inhibiting inflammatory responses in carbon tetrachloride-treated mice. Food Funct. 2021, 12, 7805–7815. [Google Scholar] [CrossRef]
- Hsu, C.; Tsai, T.H.; Li, Y.Y.; Wu, W.H.; Huang, C.J.; Tsai, P.J. Wild bitter melon (Momordica charantia Linn. var. abbreviata Ser.) extract and its bioactive components suppress Propionibacterium acnes-induced inflammation. Food Chem. 2012, 135, 976–984. [Google Scholar] [CrossRef]
- Huang, W.C.; Tsai, T.H.; Huang, C.J.; Li, Y.Y.; Chyuan, J.H.; Chuang, L.T.; Tsai, P.J. Inhibitory effects of wild bitter melon leaf extract on Propionibacterium acnes-induced skin inflammation in mouse and cytokine production in vitro. Food Funct. 2015, 6, 2550–2560. [Google Scholar] [CrossRef]
- Tsai, T.H.; Huang, W.C.; Ying, H.T.; Kuo, Y.H.; Shen, C.C.; Lin, Y.K.; Tsai, P.J. Wild bitter melon leaf extract inhibits Porphyromonas gingivalis-induced inflammation: Identification of active compounds through bioassay-guided isolation. Molecules 2016, 21, 454. [Google Scholar] [CrossRef] [Green Version]
- Yee, M.; Kim, S.; Sethi, P.; Düzgüneş, N.; Konopka, K. Porphyromonas gingivalis stimulates IL-6 and IL-8 secretion in GMSM-K, HSC-3 and H413 oral epithelial cells. Anaerobe 2014, 28, 62–67. [Google Scholar] [CrossRef]
- Uehara, A.; Takada, H. Functional TLRs and NODs in human gingival fibroblasts. J. Dent. Res. 2007, 86, 249–254. [Google Scholar] [CrossRef]
- Application of Charantadiol A in Preparation of Drugs Treating Diabetes. Available online: https://patents.google.com/patent/CN102895235B/en (accessed on 8 December 2020).
- Huang, H.T.; Zhang, L.J.; Huang, H.C.; Hwang, S.Y.; Wu, C.L.; Lin, Y.C.; Liaw, C.C.; Cheng, Y.Y.; Morris-Natschke, S.L.; Huang, C.Y.; et al. Cucurbitane-type triterpenoids from the vines of Momordica charantia and their anti-inflammatory activities. J. Nat. Product. 2020, 83, 1400–1408. [Google Scholar] [CrossRef]
- Bostanci, N.; Thurnheer, T.; Aduse-Opoku, J.; Curtis, M.A.; Zinkernagel, A.S.; Belibasakis, G.N. Porphyromonas gingivalis regulates TREM-1 in human polymorphonuclear neutrophils via its gingipains. PLoS ONE 2013, 8, e75784. [Google Scholar]
- Rudick, C.P.; Miyamoto, T.; Lang, M.S.; Agrawal, D.K. Triggering receptor expressed on myeloid cells in the pathogenesis of periodontitis: Potential novel treatment strategies. Expert. Rev. Clin. Immunol. 2017, 13, 1189–1197. [Google Scholar] [CrossRef] [PubMed]
- Bouchon, A.; Dietrich, J.; Colonna, M. Cutting edge: Inflammatory responses can be triggered by TREM-1, a novel receptor expressed on neutrophils and monocytes. J. Immunol. 2000, 164, 4991–4995. [Google Scholar] [CrossRef] [PubMed]
- Bostanci, N.; Thurnheer, T.; Belibasakis, G.N. Involvement of the TREM-1/DAP12 pathway in the innate immune responses to Porphyromonas gingivalis. Mol. Immunol. 2011, 49, 387–394. [Google Scholar] [CrossRef] [Green Version]
- Varanat, M.; Haase, E.M.; Kay, J.G.; Scannapieco, F.A. Activation of the TREM-1 pathway in human monocytes by periodontal pathogens and oral commensal bacteria. Mol. Oral Microbiol. 2017, 32, 275–287. [Google Scholar] [CrossRef] [PubMed]
- Willi, M.; Belibasakis, G.N.; Bostanci, N. Expression and regulation of triggering receptor expressed on myeloid cells 1 in periodontal diseases. Clin. Exp. Immunol. 2014, 178, 190–200. [Google Scholar] [CrossRef] [Green Version]
- Bostanci, N.; Belibasakis, G.N. Doxycycline inhibits TREM-1 induction by Porphyromonas gingivalis. FEMS Immunol. Med. Microbiol. 2012, 66, 37–44. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nikolopoulos, G.K.; Dimou, N.L.; Hamodrakas, S.J.; Bagos, P.G. Cytokine gene polymorphisms in periodontal disease: A meta-analysis of 53 studies including 4178 cases and 4590 controls. J. Clin. Periodontol. 2008, 35, 754–767. [Google Scholar] [CrossRef]
- Pan, W.; Wang, Q.; Chen, Q. The cytokine network involved in the host immune response to periodontitis. Int. J. Oral Sci. 2019, 11, 30. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tawfig, N. Proinflammatory cytokines and periodontal disease. J. Dent. Probl. Solut. 2016, 3, 012–017. [Google Scholar] [CrossRef] [Green Version]
- Takahashi, K.; Takashiba, S.; Nagai, A.; Takigawa, M.; Myoukai, F.; Kurihara, H.; Murayama, Y. Assessment of interleukin-6 in the pathogenesis of periodontal disease. J. Periodontol. 1994, 65, 147–153. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Booij-Vrieling, H.E.; Bronkhorst, E.M.; Shao, J.; Kouwer, P.H.J.; Jansen, J.; Walboomers, X.F.; Yang, F. Antimicrobial and anti-inflammatory thermo-reversible hydrogel for periodontal delivery. Acta Biomater. 2020, 116, 259–267. [Google Scholar] [CrossRef] [PubMed]
- Polak, D.; Martin, C.; Sanz-Sánchez, I.; Beyth, N.; Shapira, L. Are anti-inflammatory agents effective in treating gingivitis as solo or adjunct therapies? A systematic review. J. Clin. Periodontol. 2015, 42 (Suppl. 16), S139–S151. [Google Scholar] [CrossRef]
- Groeger, S.; Jarzina, F.; Domann, E.; Meyle, J. Porphyromonas gingivalis activates NFκB and MAPK pathways in human oral epithelial cells. BMC Immunol. 2017, 18, 1. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Y.; Cui, J.M.; Zhao, Y.Q. Novel compounds in Momordica charantia. Chin. Tradit. Herb. Drugs 2009, 40, 509–512. [Google Scholar]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tsai, T.-H.; Chang, C.-I.; Hung, Y.-L.; Huang, W.-C.; Chang, H.; Kuo, Y.-H.; Chyuan, J.-H.; Chuang, L.-T.; Tsai, P.-J. Anti-Inflammatory Effect of Charantadiol A, Isolated from Wild Bitter Melon Leaf, on Heat-Inactivated Porphyromonas gingivalis-Stimulated THP-1 Monocytes and a Periodontitis Mouse Model. Molecules 2021, 26, 5651. https://doi.org/10.3390/molecules26185651
Tsai T-H, Chang C-I, Hung Y-L, Huang W-C, Chang H, Kuo Y-H, Chyuan J-H, Chuang L-T, Tsai P-J. Anti-Inflammatory Effect of Charantadiol A, Isolated from Wild Bitter Melon Leaf, on Heat-Inactivated Porphyromonas gingivalis-Stimulated THP-1 Monocytes and a Periodontitis Mouse Model. Molecules. 2021; 26(18):5651. https://doi.org/10.3390/molecules26185651
Chicago/Turabian StyleTsai, Tzung-Hsun, Chi-I Chang, Ya-Ling Hung, Wen-Cheng Huang, Hsiang Chang, Yueh-Hsiung Kuo, Jong-Ho Chyuan, Lu-Te Chuang, and Po-Jung Tsai. 2021. "Anti-Inflammatory Effect of Charantadiol A, Isolated from Wild Bitter Melon Leaf, on Heat-Inactivated Porphyromonas gingivalis-Stimulated THP-1 Monocytes and a Periodontitis Mouse Model" Molecules 26, no. 18: 5651. https://doi.org/10.3390/molecules26185651
APA StyleTsai, T.-H., Chang, C.-I., Hung, Y.-L., Huang, W.-C., Chang, H., Kuo, Y.-H., Chyuan, J.-H., Chuang, L.-T., & Tsai, P.-J. (2021). Anti-Inflammatory Effect of Charantadiol A, Isolated from Wild Bitter Melon Leaf, on Heat-Inactivated Porphyromonas gingivalis-Stimulated THP-1 Monocytes and a Periodontitis Mouse Model. Molecules, 26(18), 5651. https://doi.org/10.3390/molecules26185651






