Analysis of Processing Effects on Glucosinolate Profiles in Red Cabbage by LC-MS/MS in Multiple Reaction Monitoring Mode
Abstract
:1. Introduction
2. Results and Discussion
2.1. Effect of Cooking Treatment on Proximate Composition in Red Cabbages
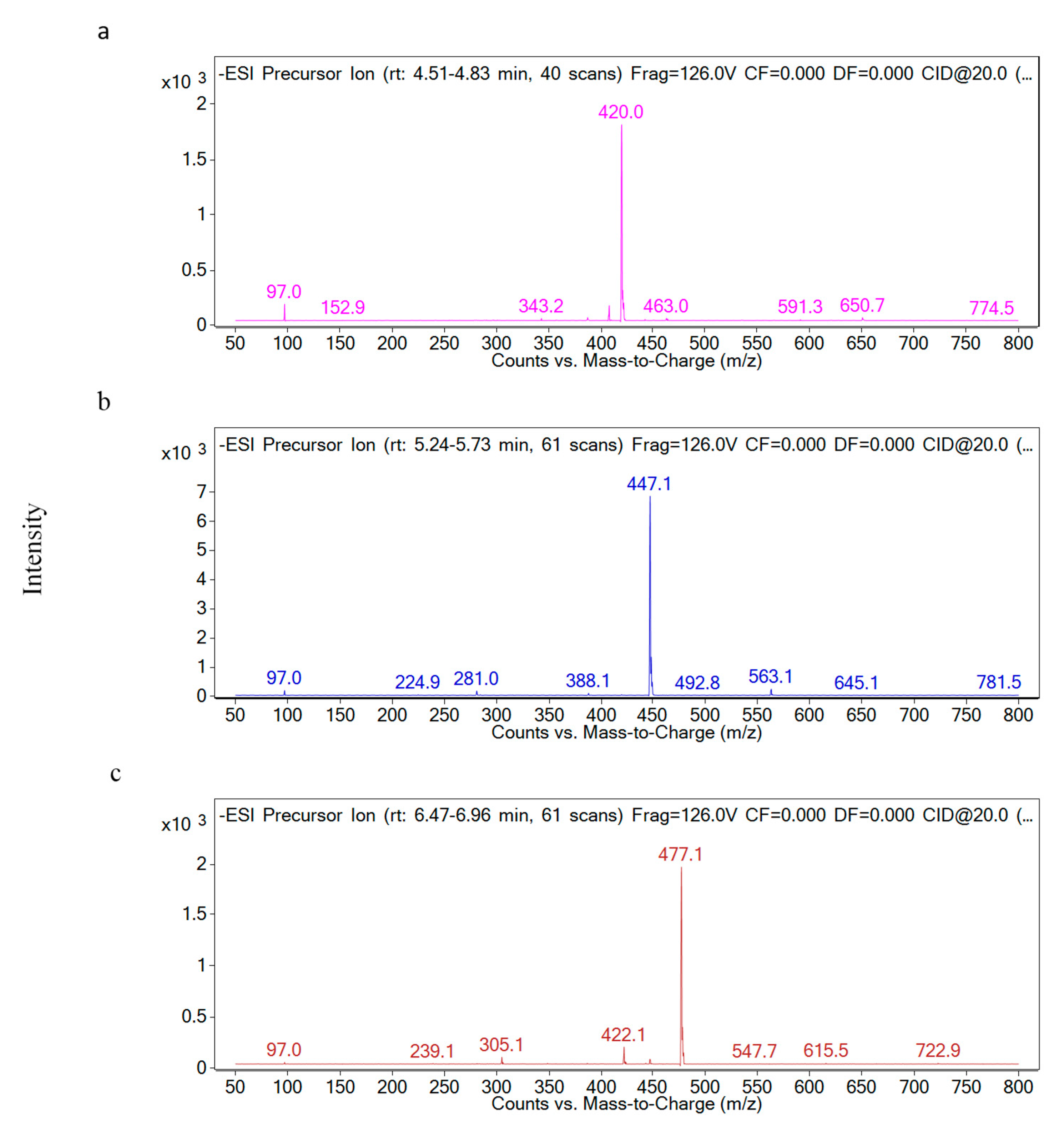
2.2. Identification of Individual Glucosinolate in Red Cabbage
2.3. Variation of Individual and Total Glucosinolate Content in Cooked Red Cabbage
2.3.1. Effect of Cooking on Indole Glucosinolates in Red Cabbage
2.3.2. Effect of Cooking on Aliphatic Glucosinolates in Red Cabbage
2.3.3. Effect of Boiling Time on Glucosinolates in Red Cabbage
3. Materials and Methods
3.1. Plant Materials
3.2. Cooking Treatments
3.3. Determination of Proximate Composition
3.4. Determination of Glucosinolates
3.4.1. Sample Extraction
3.4.2. Analysis of Intact Glucosinolates
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hanschen, F.S.; Lamy, E.; Schreiner, M.; Rohn, S. Reactivity and Stability of Glucosinolates and Their Breakdown Products in Foods. Angew. Chem. Int. Ed. 2014, 53, 11430–11450. [Google Scholar] [CrossRef]
- Lin, L.-Z.; Sun, J.; Chen, P.; Zhang, R.-W.; Fan, X.-E.; Li, L.-W.; Harnly, J.M. Profiling of Glucosinolates and Fla-vonoids in Rorippa indica (Linn.) Hiern. (Cruciferae) by UHPLC-PDA-ESI/HRMSn. J. Agric. Food Chem. 2014, 62, 6118–6129. [Google Scholar] [CrossRef]
- Oerlemans, K.; Barrett, D.M.; Suades, C.B.; Verkerk, R.; Dekker, M. Thermal degradation of glucosinolates in red cabbage. Food Chem. 2006, 95, 19–29. [Google Scholar] [CrossRef]
- Dekker, M.; Hennig, K.; Verkerk, R. Differences in Thermal Stability of Glucosinolates in Five Brassica Vegetables. Czech J. Food Sci. 2009, 27, S85–S88. [Google Scholar] [CrossRef] [Green Version]
- Hennig, K.; Verkerk, R.; Bonnema, G.; Dekker, M. Rapid Estimation of Glucosinolate Thermal Degradation Rate Constants in Leaves of Chinese Kale and Broccoli (Brassica oleracea) in Two Seasons. J. Agric. Food Chem. 2012, 60, 7859–7865. [Google Scholar] [CrossRef] [PubMed]
- Hanschen, F.S. Domestic boiling and salad preparation habits affect glucosinolate degradation in red cabbage (Brassica oleracea var. capitata f. rubra). Food Chem. 2020, 321, 126694. [Google Scholar] [CrossRef] [PubMed]
- Rungapamestry, V.; Duncan, A.; Fuller, Z.; Ratcliffe, B. Changes in Glucosinolate Concentrations, Myrosinase Activity, and Production of Metabolites of Glucosinolates in Cabbage (Brassica oleracea Var. capitata) Cooked for Different Durations. J. Agric. Food Chem. 2006, 54, 7628–7634. [Google Scholar] [CrossRef]
- Slominski, B.A.; Campbell, L.D. Gas-Chromatographic Determination Of Indole Glucosin-Olates—A Reexamination. J. Sci. Food Agric. 1987, 40, 131–143. [Google Scholar] [CrossRef]
- Heaney, R.K.; Fenwick, G.R. The Quantitative-Analysis of Indole Glucosinolates by Gas-Chromatography—The Importance of the Derivatization Conditions. J. Sci. Food Agric. 1982, 33, 68–70. [Google Scholar] [CrossRef]
- Śmiechowska, A.; Bartoszek, A.; Namieśnik, J. Determination of Glucosinolates and Their Decomposition Products—Indoles and Isothiocyanates in Cruciferous Vegetables. Crit. Rev. Anal. Chem. 2010, 40, 202–216. [Google Scholar] [CrossRef]
- Bianco, G.; Lelario, F.; Battista, F.G.; Bufo, S.A.; Cataldi, T.R. Identification of glucosinolates in capers by LC-ESI-hybrid linear ion trap with Fourier transform ion cyclotron resonance mass spectrometry (LC-ESI-LTQ-FTICR MS) and infrared multiphoton dissociation. J. Mass Spectrom. 2012, 47, 1160–1169. [Google Scholar] [CrossRef]
- Glauser, G.; Schweizer, F.; Turlings, T.C.J.; Reymond, P. Rapid Profiling of Intact Glucosinolates in Arabidopsis Leaves by UHPLC-QTOFMS Using a Charged Surface Hybrid Column. Phytochem. Anal. 2012, 23, 520–528. [Google Scholar] [CrossRef] [Green Version]
- Pasini, F.; Verardo, V.; Caboni, M.F.; D’Antuono, L.F. Determination of glucosinolates and phenolic compounds in rocket salad by HPLC-DAD–MS: Evaluation of Eruca sativa Mill. and Diplotaxis tenuifolia L. genetic re-sources. Food Chem. 2012, 133, 1025–1033. [Google Scholar] [CrossRef]
- Hennig, K.; Verkerk, R.; Bonnema, G.; Dekker, M. Pitfalls in the desulphation of glucosinolates in a high-throughput assay. Food Chem. 2012, 134, 2355–2361. [Google Scholar] [CrossRef] [PubMed]
- Tian, Q.; Rosselot, R.A.; Schwartz, S.J. Quantitative determination of intact glucosinolates in broccoli, broccoli sprouts, Brussels sprouts, and cauliflower by high-performance liquid chromatography–electrospray ionization–tandem mass spectrometry. Anal. Biochem. 2005, 343, 93–99. [Google Scholar] [CrossRef]
- Han, Y.; Yin, F.; John, G.F.; Clement, T.P. Understanding the relative performance of SCAN, SIM, PMRM and MRM methods for quantifying polycyclic aromatic hydrocarbons (PAHs) in crude oil samples. Rapid Commun. Mass Spectrom. 2020, 34, 8765. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.; Mu, T.; Xi, L.; Song, Z. Effects of Domestic Cooking Methods on Polyphenols and Antioxidant Activity of Sweet Potato Leaves. J. Agric. Food Chem. 2014, 62, 8982–8989. [Google Scholar] [CrossRef] [PubMed]
- Murniece, I.; Karklina, D.; Galoburda, R.; Santare, D.; Skrabule, I.; Costa, H. Nutritional composition of freshly harvested and stored Latvian potato (Solanum tuberosum L.) varieties depending on traditional cooking methods. J. Food Compos. Anal. 2011, 24, 699–710. [Google Scholar] [CrossRef]
- Gidamis, A.B.; Panga, J.T.; Sarwatt, S.V.; Chove, B.E.; Shayo, N.B. Nutrient and Antinutrient Contents in Raw and Cooked Young Leaves and Immature Pods OfMoringa oleifera, Lam. Ecol. Food Nutr. 2003, 42, 399–411. [Google Scholar] [CrossRef]
- Verkerk, R.; Dekker, M. Glucosinolates and Myrosinase Activity in Red Cabbage (Brassica oleracea L. Var. Capi-tata f. rubra DC.) after Various Microwave Treatments. J. Agric. Food Chem. 2004, 52, 7318–7323. [Google Scholar] [CrossRef]
- Volden, J.; Borge, G.I.A.; Bengtsson, G.; Hansen, M.; Thygesen, I.E.; Wicklund, T. Effect of thermal treatment on glucosinolates and antioxidant-related parameters in red cabbage (Brassica oleracea L. ssp. capitata f. rubra). Food Chem. 2008, 109, 595–605. [Google Scholar] [CrossRef]
- Kushad, M.M.; Brown, A.F.; Kurilich, A.C.; Juvik, J.A.; Klein, B.P.; Wallig, M.A.; Jeffery, E.H. Variation of Glucos-inolates in Vegetable Crops of Brassica oleracea. J. Agric. Food Chem. 1999, 47, 1541–1548. [Google Scholar] [CrossRef] [PubMed]
- Xu, F.; Zheng, Y.; Yang, Z.; Cao, S.; Shao, X.; Wang, H. Domestic cooking methods affect the nutritional quality of red cabbage. Food Chem. 2014, 161, 162–167. [Google Scholar] [CrossRef] [PubMed]
- Cieślik, E.; Leszczyńska, T.; Filipiak-Florkiewicz, A.; Sikora, E.; Pisulewski, P.M. Effects of some technological processes on glucosinolate contents in cruciferous vegetables. Food Chem. 2007, 105, 976–981. [Google Scholar] [CrossRef]
- Murillo, G.; Mehta, R.G. Cruciferous Vegetables and Cancer Prevention. Nutr. Cancer 2001, 41, 17–28. [Google Scholar] [CrossRef]
- Kim, J.K.; Chu, S.M.; Kim, S.J.; Lee, D.J.; Lee, S.Y.; Lim, S.H.; Ha, S.-H.; Kweon, S.J.; Cho, H.S. Variation of glucos-inolates in vegetable crops of Brassica rapa L. ssp. pekinensis. Food Chem. 2010, 119, 423–428. [Google Scholar] [CrossRef]
- Hanschen, F.S.; Rohn, S.; Mewis, I.; Schreiner, M.; Kroh, L.W. Influence of the chemical structure on the thermal degradation of the glucosinolates in broccoli sprouts. Food Chem. 2012, 130, 1–8. [Google Scholar] [CrossRef]
- Singh, K.; Connors, S.L.; Macklin, E.; Smith, K.D.; Fahey, J.W.; Talalay, P.; Zimmerman, A.W. Sulforaphane treatment of autism spectrum disorder (ASD). Proc. Natl. Acad. Sci. USA 2014, 111, 15550–15555. [Google Scholar] [CrossRef] [Green Version]
- Cartea, M.E.; Velasco, P.; Obregón, S.; Padilla, G.; de Haro, A. Seasonal variation in glucosinolate content in Brassica oleracea crops grown in northwestern Spain. Phytochemistry 2008, 69, 403–410. [Google Scholar] [CrossRef]
- Fenwick, G.R.; Heaney, R.K.; Mullin, W.J.; VanEtten, C.H. Glucosinolates and their breakdown products in food and food plants. CRC Crit. Rev. Food Sci. Nutr. 1983, 18, 123–201. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.-J.; Ishii, G. Glucosinolate profiles in the seeds, leaves and roots of rocket salad (Eruca sativa Mill.) and anti-oxidative activities of intact plant powder and purified 4-methoxyglucobrassicin. Soil Sci. Plant Nutr. 2006, 52, 394–400. [Google Scholar] [CrossRef]
- Rangkadilok, N.; Nicolas, M.E.; Bennett, R.N.; Premier, R.R.; Eagling, D.R.; Taylor, P.W.J. Determination of si-nigrin and glucoraphanin in Brassica species using a simple extraction method combined with ion-pair HPLC analysis. Sci. Hortic. 2002, 96, 27–41. [Google Scholar] [CrossRef]
- Ciska, E.; Kozłowska, H. The effect of cooking on the glucosinolates content in white cabbage. Eur. Food Res. Technol. 2001, 212, 582–587. [Google Scholar] [CrossRef]
- Van Doorn, H.E.; van der Kruk, G.C.; van Holst, G.-J.; Raaijmakers-Ruijs, N.C.M.E.; Postma, E.; Groeneweg, B.; Jongen, W.H.F. The glucosinolates sinigrin and progoitrin are important determinants for taste preference and bitterness of Brussels sprouts. J. Sci. Food Agric. 1998, 78, 30–38. [Google Scholar] [CrossRef]
- Rosa, E.A.S.; Heaney, R.K. The effect of cooking and processing on the glucosinolate content: Studies on four varieties of portuguese cabbage and hybrid white cabbage. J. Sci. Food Agric. 1993, 62, 259–265. [Google Scholar] [CrossRef]
- Hanschen, F.S.; Kühn, C.; Nickel, M.; Rohn, S.; Dekker, M. Leaching and degradation kinetics of glucosinolates during boiling of Brassica oleracea vegetables and the formation of their breakdown products. Food Chem. 2018, 263, 240–250. [Google Scholar] [CrossRef]
- Francisco, M.; Velasco, P.; Moreno-Fernández, D.Á.; Garcia-Viguera, C.; Cartea, M.E. Cooking methods of Brassica rapa affect the preservation of glucosinolates, phenolics and vitamin C. Food Res. Int. 2010, 43, 1455–1463. [Google Scholar] [CrossRef] [Green Version]
- Tabart, J.; Pincemail, J.; Kevers, C.; Defraigne, J.-O.; Dommes, J. Processing effects on antioxidant, glucosinolate, and sulforaphane contents in broccoli and red cabbage. Eur. Food Res. Technol. 2018, 244, 2085–2094. [Google Scholar] [CrossRef]
- Dekker, M.; Verkerk, R.; Jongen, W.M. Predictive modelling of health aspects in the food production chain: a case study on glucosinolates in cabbage. Trends Food Sci. Technol. 2000, 11, 174–181. [Google Scholar] [CrossRef]
- Jaiswal, A.K.; Gupta, S.; Abu-Ghannam, N. Kinetic evaluation of colour, texture, polyphenols and antioxidant capacity of Irish York cabbage after blanching treatment. Food Chem. 2012, 131, 63–72. [Google Scholar] [CrossRef] [Green Version]
- Kao, F.J.; Chiu, Y.S.; Tsou, M.J.; Chiang, W.D. Effects of Chinese domestic cooking methods on the carotenoid composition of vegetables in Taiwan. LWT 2012, 46, 485–492. [Google Scholar] [CrossRef]

| Poximate Composition | Thermal Processing Methods | |||||
|---|---|---|---|---|---|---|
| Raw | Boiled | Steamed | Microwaved | Fried | Stir-Fried | |
| Crude fat | 6.77 ± 0.50 a | 11.61 ± 0.16 c | 7.49 ± 0.82 a | 9.78 ± 0.19 b | 13.14 ± 0.32 d | 43.59 ± 0.25 e |
| Crude fiber | 4.52 ± 2.46 c | 6.17 ± 8.94 e | 4.18 ± 8.54 b | 5.01 ± 0.05 d | 12.67 ± 0.07 f | 3.28 ± 7.21 a |
| Crude protein | 19.12 | 18.31 | 18.56 | 18.7 | 12.58 | 8.3 |
| Ash | 7.68 ± 0.02 e | 7.30 ± 8.08 d | 7.36 ± 2.49 e | 6.86 ± 0.01 c | 5.01 ± 7.49 b | 4.07 ± 7.53 a |
| Poximate Composition | Boiling Time | ||||
|---|---|---|---|---|---|
| 1 min | 5 min | 10 min | 20 min | 30 min | |
| Crude fat | 12.96 ± 0.16 e | 9.56 ± 0.24 d | 8.08 ± 0.16 c | 7.02 ± 0.25 b | 6.20 ± 0.25 a |
| Crude fiber | 4.93 ± 5.17 b | 7.78 ± 8.12 c | 7.92 ± 2.49 e | 8.02 ± 3.39 d | 13.28 ± 0.01 f |
| Crude protein | 18.13 | 18.50 | 19.05 | 19.12 | 19.56 |
| Ash | 7.52 ± 8.10 f | 7.19 ± 0.01 d | 6.49 ± 9.48 c | 5.51 ± 0.03 a | 5.74 ± 9.14 b |
| Glucosinolates | Thermal Processing Methods | |||||
|---|---|---|---|---|---|---|
| Raw | Boiled | Steamed | Microwaved | Fried | Stir-Fried | |
| 4-Methoxyglucobrassicin | 1.2117 | 0.9107 | 0.8489 | 0.8112 | 0.4948 | 0.3602 |
| Neoglucobrassicin | 0.9424 | 0.7627 | 0.8827 | 0.7761 | 0.1642 | 0.0138 |
| Glucoalyssin | 0.8410 | 0.4827 | 0.5824 | 0.0697 | 0.0594 | 0.0558 |
| Glucobrassicin | 7.1302 | 4.2111 | 5.7305 | 6.1755 | 1.7267 | 1.5221 |
| Glucoraphanin | 1.3527 | 1.1332 | 1.2707 | 1.1766 | 0.2427 | 0.2172 |
| Glucoiberin | 0.6505 | 0.6155 | 0.5236 | 0.4819 | 0.0497 | 0.0269 |
| Glucoerucin | 1.3083 | 0.4457 | 0.1852 | 0.2768 | 0.0984 | 0. 0228 |
| Progoitrin | 1.6057 | 1.5446 | 1.3904 | 1.2824 | 0.1880 | 0.3221 |
| Gluconapin | 1.4480 | 1.0511 | 1.0714 | 1.3659 | 0.1116 | 0.0597 |
| Sinigrin | 0.1358 | 0.0879 | 0.0112 | 0.0083 | 0.0049 | 0.0046 |
| Indole glucosinolates | 9.2843 | 5.8845 | 7.4621 | 7.7628 | 2.3857 | 1.8961 |
| Aliphatic glucosinolates | 7.3420 | 5.3607 | 5.0349 | 4.6616 | 0.7547 | 0.6863 |
| Total | 16.6263 | 11.2452 | 12.4978 | 12.4244 | 3.1404 | 2.6112 |
| Glucosinolates | Boiling Time | ||||
|---|---|---|---|---|---|
| 1 min | 5 min | 10 min | 20 min | 30 min | |
| 4-Methoxyglucobrassicin | 1.0952 | 0.8098 | 0.6141 | 0.1830 | 0.1530 |
| Neoglucobrassicin | 0.8160 | 0.6798 | 0.3969 | 0.2424 | 0.1571 |
| Glucoalyssin | 0.4830 | 0.1410 | 0.0644 | 0.0636 | 0.0380 |
| Glucobrassicin | 5.0497 | 3.3872 | 2.2315 | 1.3152 | 1.1926 |
| Glucoraphanin | 1.2608 | 1.0481 | 0.9157 | 0.7667 | 0.6838 |
| Glucoiberin | 0.6202 | 0.6088 | 0.5259 | 0.4519 | 0.3657 |
| Glucoerucin | 0.8487 | 0.2978 | 0.1630 | 0.1420 | 0.0442 |
| Progoitrin | 1.6005 | 1.2534 | 1.1940 | 0.9083 | 0.8082 |
| Gluconapin | 1.2120 | 1.0296 | 0.9996 | 0.4541 | 0.3087 |
| Sinigrin | 0.1172 | 0.0800 | 0.0363 | 0.0182 | 0.0092 |
| Indole glucosinolates | 6.9609 | 4.8768 | 3.2425 | 1.7406 | 1.5027 |
| Aliphatic glucosinolates | 6.1424 | 4.4587 | 3.8989 | 2.8048 | 2.2578 |
| Total | 13.1032 | 9.3354 | 7.1414 | 4.5458 | 3.7604 |
| Temperature (Power) | Time | Material/Water (Oil) | Tool | |
|---|---|---|---|---|
| Boiling | 100 °C | 3 min | 300 g/2 L | Supor saucepan |
| Steaming | 100 °C | 3 min | None | FOTILE SCD20-01 steamer |
| Microwave heating | 1000 W | 3 min | None | FOTILE W25800-01AG microwave |
| Frying | 160 °C | 3 min | 300 g/500 mL | Royalstar YSF458 wok |
| Stir-frying | 190 °C | 3 s | 300 g/2 L | VESTA EF-81 frying pan |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, W.; Chen, J.; Yu, D.; Chen, S.; Ye, X.; Zhang, Z. Analysis of Processing Effects on Glucosinolate Profiles in Red Cabbage by LC-MS/MS in Multiple Reaction Monitoring Mode. Molecules 2021, 26, 5171. https://doi.org/10.3390/molecules26175171
Wu W, Chen J, Yu D, Chen S, Ye X, Zhang Z. Analysis of Processing Effects on Glucosinolate Profiles in Red Cabbage by LC-MS/MS in Multiple Reaction Monitoring Mode. Molecules. 2021; 26(17):5171. https://doi.org/10.3390/molecules26175171
Chicago/Turabian StyleWu, Weicheng, Jingqiu Chen, Dandan Yu, Shiguo Chen, Xingqian Ye, and Zhiguo Zhang. 2021. "Analysis of Processing Effects on Glucosinolate Profiles in Red Cabbage by LC-MS/MS in Multiple Reaction Monitoring Mode" Molecules 26, no. 17: 5171. https://doi.org/10.3390/molecules26175171
APA StyleWu, W., Chen, J., Yu, D., Chen, S., Ye, X., & Zhang, Z. (2021). Analysis of Processing Effects on Glucosinolate Profiles in Red Cabbage by LC-MS/MS in Multiple Reaction Monitoring Mode. Molecules, 26(17), 5171. https://doi.org/10.3390/molecules26175171







