Chemical Constitution and Antimicrobial Activity of Kombucha Fermented Beverage
Abstract
1. Introduction
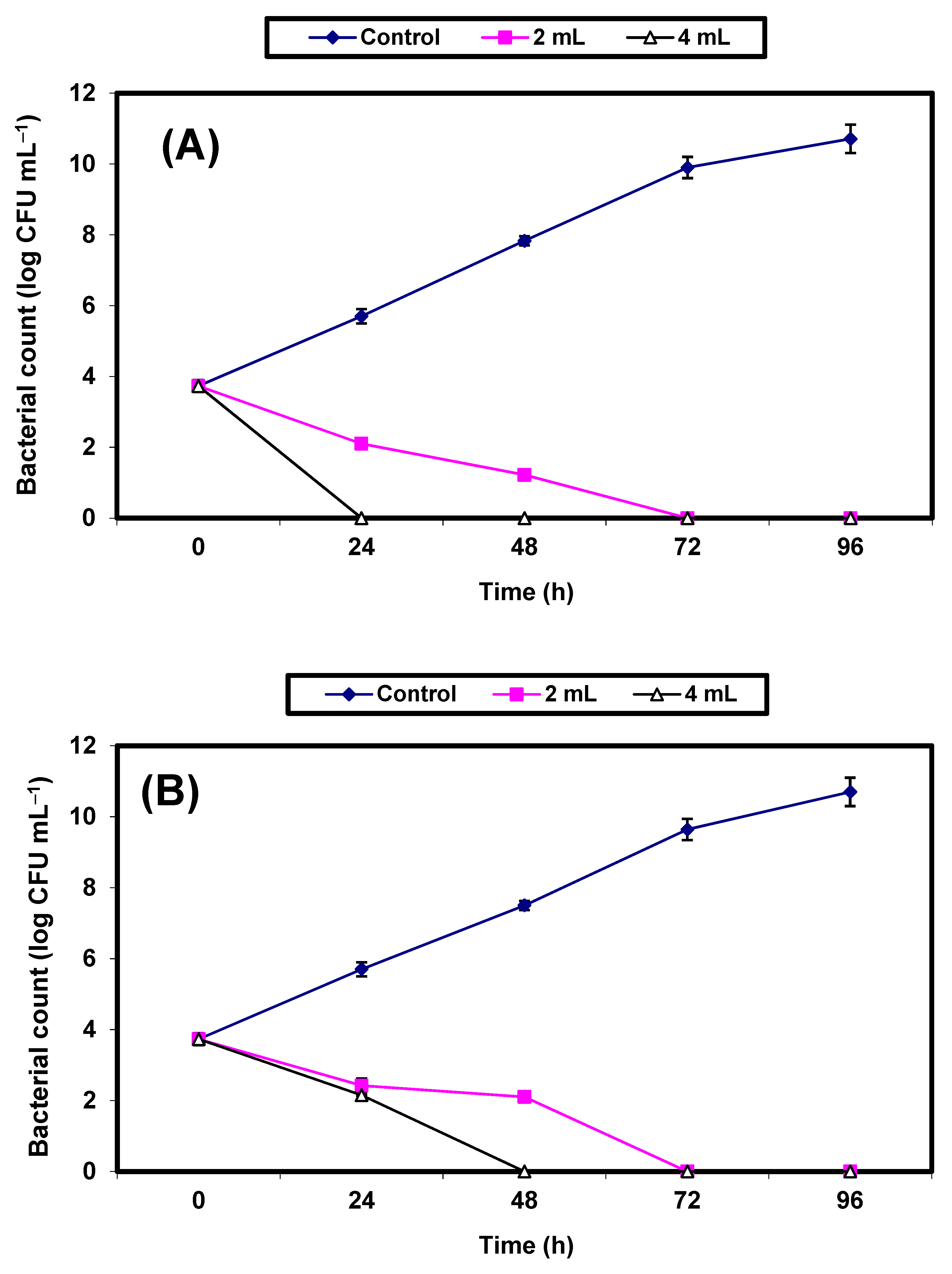
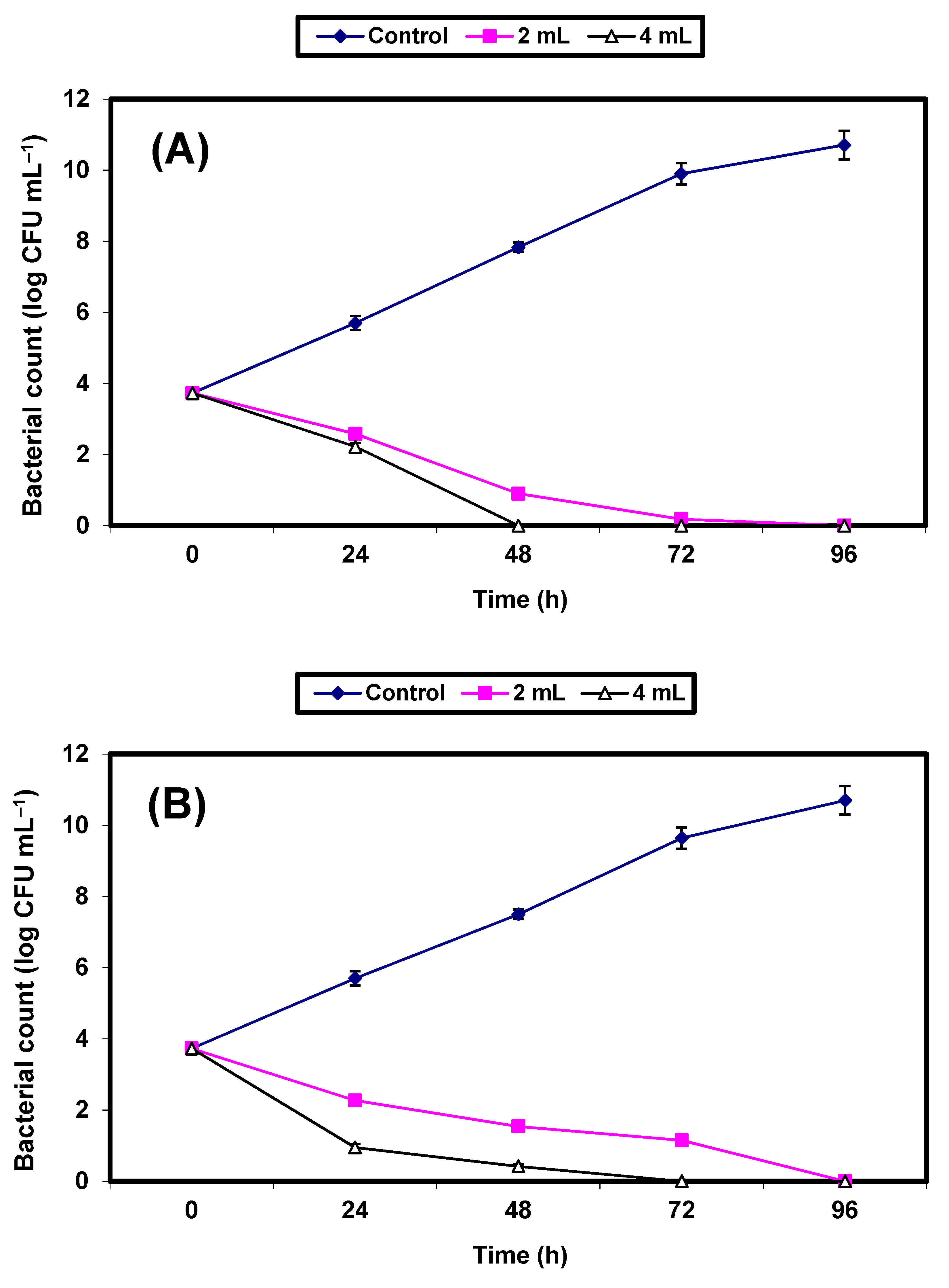
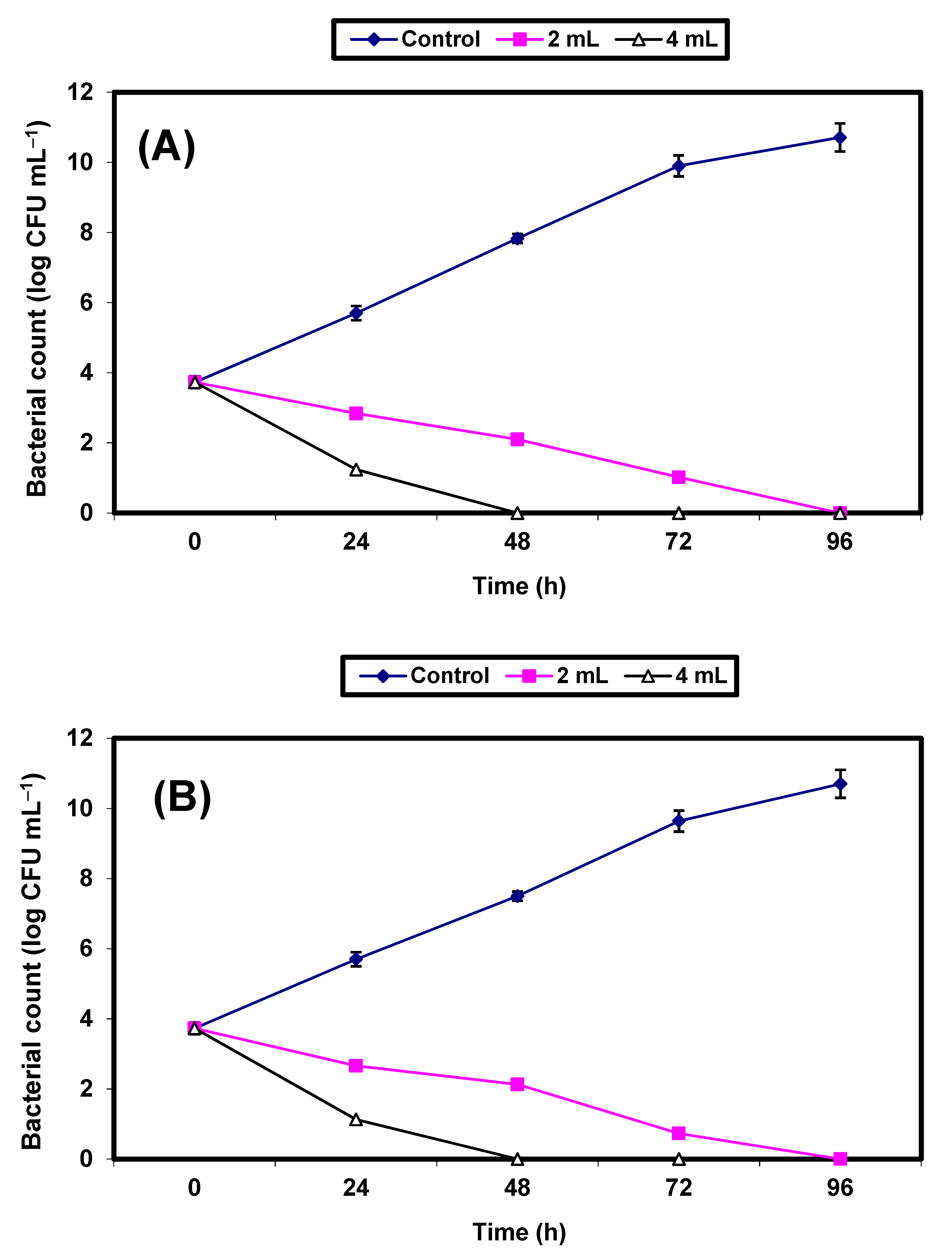
2. Results
3. Discussion
4. Materials and Methods
4.1. Kombucha Consortium
4.2. Preparation of Kombucha Fermented Beverage (KFB)
4.3. pH Determination
4.4. Instrumental Analysis of KFB
4.5. Isolation and Identification of Bacterial Species from the Kombucha
4.6. Microbial Test Strains
4.7. Antimicrobial Bioassays of Kombucha Beverage
4.8. Preparation of Fruit Juices Employed in the Antimicrobial Activity of KFB
4.9. Inhibition of Both E. coli and S. aureus in BHI Broth and Juices of Apple, Guava, Strawberry and Tomato
4.10. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Bermudez-Brito, M.; Plaza-Díaz, J.; Muñoz-Quezada, S.; Gómez-Llorente, C.; Gil, A. Probiotic mechanisms of action. Ann. Nutr. Metab. 2012, 61, 160–174. [Google Scholar] [CrossRef]
- Enan, G.; Abdel-Shafi, S.; Ouda, S.M.; El-Balat, I. Genetic linkage of the antibiotic resistance ability in the Escherichia coli UR4 strain isolated from urine. J. Med. Sci. 2013, 13, 261–268. [Google Scholar] [CrossRef][Green Version]
- Enan, G.; El-Essawy, A.A.; Uyttendaele, M.; Debevere, J. Antibacterial activity of Lactobacillus plantarum UG1 isolated from dry sausage: Characterization, production and bactericidal action of plantaricin UG1. Int. J. Food Microbiol. 1996, 30, 189–215. [Google Scholar] [CrossRef]
- Enan, G.; Abdel-Shafi, S.; Abdel-Haliem, M.F.; Negm, S. Characterization of probiotic lactic acid bacteria to be used as starter and protective cultures for dairy fermentations. Int. J. Probiotics Prebiotics 2013, 8, 157–163. [Google Scholar]
- Enan, G.; Abdel-Shafi, S.; Ouda, S.; Negm, S. Novel antibacterial activity of Lactococcus lactis subsp. Lactis Z11 isolated from zabady. Int. J. Biomed. Sci. 2013, 9, 174–180. [Google Scholar] [PubMed]
- Enan, G.; Osman, M.E.; Abdel-Haliem, M.E.F.; Abdel-Ghany, S.E. Advances in microbial and nucleic acids biotechnology. BioMed Res. Int. 2018, 2018, 3102374. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Shafi, S.; Al-Mohammadi, A.-R.; Negm, S.; Enan, G. Antibacterial activity of Lactobacillus delbreukii subspecies bulgaricus isolated from Zabady. Life Sci. J. 2014, 11, 264–270. [Google Scholar]
- Ouda, S.M.; Debevere, J.; Enan, G. Purification and biochemical characterization of plantiricin UG1: A bacteriocin produced by Lactobacillus plantarum UG1 isolated from dry sausage. Life Sci. J. 2014, 11, 271–279. [Google Scholar]
- Ivanišová, E.; Meňhartová, K.; Terentjeva, M.; Harangozo, Ľ.; Kántor, A.; Kántor, M. The evaluation of chemical, antioxidant, antimicrobial and sensory properties of kombucha tea beverage. J. Food Sci. Technol. 2020, 57, 1840–1846. [Google Scholar] [CrossRef]
- Al-Mohammadi, A.-R.; Ibrahim, R.A.; Moustafa, A.H.; Ismaiel, A.A.; Abou Zeid, A.; Enan, G. Chemical constitution and antimicrobial activity of kefir fermented beverage. Molecules 2021, 26, 2635. [Google Scholar] [CrossRef]
- Enan, G. Control of the regrowing bacteriocin resistant variants of Listeria monocytogenes LMG 10470 in vitro and in food by nisin- plantaricin UG1 mixture. Biotechnology 2006, 5, 143–147. [Google Scholar] [CrossRef]
- Ismaiel, A.A.; Ali, A.E.-S.; Enan, G. Incidence of Listeria in Egyptian meat and dairy samples. Food Sci. Biotechnol. 2014, 23, 179–185. [Google Scholar] [CrossRef]
- Osman, A.; El-Didamony, G.; Sitohy, M.; Khalifa, M.; Enan, G. Soybean glycinin basic subunit inhibits methicillin resistant vancomycin intermediate Staphylococcus aureus (MRSA-VISA) in vitro. Int. J. Appl. Res. Nat. Prod. 2016, 9, 17–26. [Google Scholar]
- McFarland, L.V. Meta-analysis of probiotics for the prevention of antibiotic associated diarrhea and the treatment of Clostridium difficile disease. Am. J. Gastroenterol. 2006, 101, 812–822. [Google Scholar] [CrossRef]
- Gaziano, R.; Sabbatini, S.; Roselletti, E.; Perito, S.; Monari, C. Saccharomyces cerevisiae-based probiotics as novel antimicrobial agents to prevent and treat vaginal infections. Front. Microbiol. 2020, 11, 718. [Google Scholar] [CrossRef]
- Jayabalan, R.; Malbaša, R.V.; Lončar, E.S.; Vitas, J.S.; Sathishkumar, M. A review on kombucha tea-microbiology, composition, fermentation, beneficial effects, toxicity, and “tea fungus”. Compr. Rev. Food Sci. Food Saf. 2014, 13, 538–550. [Google Scholar] [CrossRef]
- Villarreal-Soto, S.A.; Bouajila, J.; Souchard, J.; Taillandier, P. Understanding kombucha tea fermentation: A Review. J. Food Sci. 2018, 83, 580–588. [Google Scholar] [CrossRef] [PubMed]
- Kasper, Ed LAC. “The 3rd. Congress of Alternative Medicine and Acupuncture, Kombucha and AIDS Guadalajara Jalisco, México (11/98)”. Happy Herbalist Web Site. Available online: http://www.happyherbalist.com/kombucha.htm (accessed on 1 January 2009).
- Greenwalt, C.J.; Steinkraus, K.H.; Ledford, R.A. Kombucha, the fermented tea: Microbiology, composition, and claimed health effects. J. Food Prot. 2000, 63, 976–981. [Google Scholar] [CrossRef] [PubMed]
- Sreeramulu, G.; Zhu, Y.; Knol, W. Kombucha fermentation and its antimicrobial activity. J. Agric. Food Chem. 2000, 48, 2589–2594. [Google Scholar] [CrossRef] [PubMed]
- Hartmann, A.M.; Burleson, L.E.; Holmes, A.K.; Geist, C.R. Effects of chronic kombucha ingestion on open-field behaviors, longevity, appetitive behaviors, and organs in C57-BL/6 mice: A pilot study. Nutrition 2000, 16, 755–761. [Google Scholar] [CrossRef]
- Leal, J.M.; Suárez, L.V.; Jayabalan, R.; Oros, J.H.; Escalante-Aburto, A. A review on health benefits of kombucha nutritional compounds and metabolites. CyTA J. Food 2018, 16, 390–399. [Google Scholar] [CrossRef]
- Battikh, H.; Bakhrouf, A.; Ammar, E. Antimicrobial effect of Kombucha analogues. LWT Food Sci. Technol. 2012, 47, 71–77. [Google Scholar] [CrossRef]
- Watawana, M.I.; Jayawardena, N.; Gunawardhana, C.B.; Waisundara, V.Y. Health, wellness, and safety aspects of the consumption of kombucha. J. Chem. 2015, 11, 1–11. [Google Scholar] [CrossRef]
- Enan, G.; Al-Mohammadi, A.-R.; El-Didamony, C.; Abdel-Haliem, M.E.F.; Zakaria, A. Antimicrobial activity of Enterococcus faecium NM2 isolated from urine: Purification, Characterization and bacterial action of enterocin NM2. Asian J. Appl. Sci. 2014, 7, 621–634. [Google Scholar] [CrossRef]
- Enan, G.; Abdel-Haliem, M.E.F.; Tartour, E. Evaluation of the antimicrobial activity, starter capability and technological properties of some probiotic bacteria isolated from Egyptian pickles. Life Sci. J. 2014, 11, 976–985. [Google Scholar]
- Prado, F.C.; Parada, J.; Pandey, A.; Soccol, C.R. Trends in non-dairy probiotic beverages. Food Res. Int. 2008, 41, 111–123. [Google Scholar] [CrossRef]
- Ismaiel, A.A.; Bassyouni, R.H.; Kamel, Z.; Gabr, S.M. Detoxification of patulin by kombucha tea culture. CyTA J. Food 2016, 14, 271–279. [Google Scholar] [CrossRef]
- The, J.S. Toxicity of short-chain fatty acids and alcohols towards Cladosporium resinae. Appl. Microbiol. 1974, 28, 840–844. [Google Scholar] [CrossRef]
- Mithöfer, A.; Boland, W. Plant defense against herbivores: Chemical aspects. Annu. Rev. Plant Biol. 2012, 63, 431–450. [Google Scholar] [CrossRef]
- Zhao, L.; Zhang, H.; Hao, T.; Li, S. In vitro antibacterial activities and mechanism of sugar fatty acid esters against five food-related bacteria. Food Chem. 2015, 187, 370–377. [Google Scholar] [CrossRef]
- Espinoza, C.; Viniegra-González, G.; Loera, O.; Heredia, G.; Trigos, Á. Antibacterial activity against plant pathogens by cruded extracts and compounds from Idriella sp. Rev. Mex. Micol. 2008, 26, 9–15. [Google Scholar]
- Holtzman, J.L.; Crankshaw, D.L.; Peterson, F.J.; Polnaszek, C.F. The kinetics of the aerobic reduction on nitrofurantoin by NADPH-cytochrome P-450(c) reductase. Mol. Pharmacal. 1981, 20, 669–673. [Google Scholar]
- Beale, J.M.; Block, J.H. Wilson and Gisvold’s Textbook of Organic Medicinal and Pharmaceutical Chemistry, 12th ed.; A Wolters Kluwer Business; Lippincot & Wilkins: Philadelphia, PA, USA, 2011; ISBN 13-978-1609133986. [Google Scholar]
- Trombetta, D.; Bisignano, G.; Arena, S. Study on the mechanisms of the antibacterial action of some plant α-β- unsaturated aldehydes. Lett. Appl. Microbiol. 2002, 35, 285–290. [Google Scholar] [CrossRef]
- Wińska, K.; Grabarczyk, M.; Maczka, W.; Zarowska, B.; Maciejewska, G.; Anioł, M. Antimicrobial activity of new bicyclic lactones with three or four methyl groups obtained both synthetically and biosynthetically. J. Saudi Chem. Soc. 2016, 22, 363–371. [Google Scholar] [CrossRef]
- Mares, D. Antimicrobial activity of protoanemonin, a lactose from ranunculaceous plants. Mycopathologia 1987, 98, 133–140. [Google Scholar] [CrossRef]
- Atia, A.J.K. Synthesis and antibacterial activities of new metronidazole and imidazole derivatives. Molecules 2009, 14, 2431–2446. [Google Scholar] [CrossRef]
- Mayser, P.; Fromme, S.; Leitzmann, C.; Gründer, K. The yeast spectrum of the “tea fungus kombucha”. Mycoses 1995, 38, 289–295. [Google Scholar] [CrossRef]
- Sievers, M.; Lanini, C.; Weber, A.; Schuler-Schmid, U.; Teuber, M. Microbiology and fermentation balance in a kombucha beverage obtained from a tea fungus fermentation. Syst. Appl. Microbiol. 1995, 18, 590–594. [Google Scholar] [CrossRef]
- Marsh, A.J.; O’Sullivan, O.; Hill, C.; Ross, R.P.; Cotter, P.D. Sequence-based analysis of the bacterial and fungal compositions of multiple Kombucha (tea fungus) samples. Food Microbiol. 2014, 38, 171–178. [Google Scholar] [CrossRef] [PubMed]
- Greenwalt, C.J.; Ledford, R.A.; Steinkraus, K.H. Determination and characterization of the antimicrobial activity of the fermented tea kombucha. Lebensm. Wiss. Technol. 1998, 31, 291–296. [Google Scholar] [CrossRef]
- Sreeramulu, G.; Zhu, Y.; Knol, W. Characterization of antimicrobial activity in kombucha fermentation. Acta Biotechnol. 2001, 21, 49–56. [Google Scholar] [CrossRef]
- Steinkraus, K.H.; Shapiro, K.B.; Hotchkiss, J.H.; Mortlock, R.P. Investigations into the antibiotic activity of tea Fungus/Kombucha beverage. Acta Biotechnol. 1996, 16, 199–205. [Google Scholar] [CrossRef]
- Kaewkod, T.; Bovonsombut, S.; Tragoolpua, Y. Efficacy of Kombucha obtained from green, oolong, and black teas on inhibition of pathogenic bacteria, antioxidation, and toxicity on colorectal cancer cell line. Microorganisms 2019, 7, 700. [Google Scholar] [CrossRef] [PubMed]
- Lončar, E.; Djurić, M.; Malbaša, R.; Kolarov, L.J.; Klašnja, M. Influence of working conditions upon Kombucha conducted fermentation of black tea. Food Bioprod. Process. 2006, 84, 186–192. [Google Scholar] [CrossRef]
- Osman, A.; Abdel-Shafi, S.; Al-Mohammadi, A.-R.; Enan, G.; Sitohy, M. Catfish glycoprotein, a highly powerful safe preservative of minced beef stored at 4 °C for 15 days. Foods 2020, 9, 1115. [Google Scholar] [CrossRef] [PubMed]
- Osman, A.; Bin-Jumah, M.; Abd El-Hack, M.; Elaraby, G.; Swelum, A.A.; Taha, A.E.; Sitohy, M.; Allam, A.A.; Ashour, E.A. Dietary supplementation of soybean glycinin can alter the growth, carcases traits, blood biochemical indices, and meat quality of broilers. Poult. Sci. 2020, 99, 820–828. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Shafi, S.; Osman, A.; Enan, G.; El-Nemer, M.; Sitohy, M. Antibacterial activity of methylated egg white proteins against pathogenic G+ and G- bacteria matching antibiotics. SpringerPlus 2016, 5, 983. [Google Scholar] [CrossRef]
- Abdel-Shafi, S.; Al-Mohammadi, A.R.; Almanaa, T.N.; Moustafa, A.H.; Saad, T.M.M.; Ghonemy, A.; Anacarso, I.; Enan, G.; El-Gazzar, N. Identification and testing antidermatophyticoxaborole-6-derivative (OXBS) from Streptomyces atrovirens KM192347 isolated from soil. Antibiotics 2020, 9, 176. [Google Scholar] [CrossRef]
- Abdel-Shafi, S.; Osman, A.; Al-Mohammadi, A.-R.; Kamal, N.; Sitohy, M. Biochemical, biological characteristics and antibacterial activity of glycoprotein extracted from the epidermal mucus of African catfish (Clarias gariepinus). Int. J. Biol. Macromol. 2019, 138, 773–780. [Google Scholar] [CrossRef]
- El-Gazzar, N.S.; Enan, G. Advances in phage inspired nanoscience based therapy. In Nanobioscience; Saxena, S.K., Khurana, S.P., Eds.; Springer Nature Singapore Pte Ltd.: Singapore, 2020; pp. 237–257. ISBN 978-981-32-9898-9_10. [Google Scholar]
- Poolman, J.T.; Anderson, A.S. Escherichia coli and Staphylococcus aureus: Leading bacterial pathogens of healthcare associated infections and bacteremia in older-Age populations. Expert Rev. Vaccines 2018, 17, 607–618. [Google Scholar] [CrossRef]
- Enan, G.; Al-Mohammadi, A.-R.; Mahgoub, S.; Abdel-Shafi, S.; Askar, E.; Ghaly, M.F.; Taha, M.A.; El-Gazzar, N. Inhibition of Staphylococcus aureus LC554891 by Moringa oleifera seed extract either singly or in combination with antibiotics. Molecules 2020, 25, 4583. [Google Scholar] [CrossRef]
- Ayed, L.; Ben Abid, S.; Hamdi, M. Development of a beverage from red grape juice fermented with the kombucha consortium. Ann. Microbiol. 2017, 67, 111–121. [Google Scholar] [CrossRef]
- Zhao, Z.; Sui, Y.; Wu, H.; Zhou, C.; Hu, X.; Zhang, J. Flavour chemical dynamics during fermentation of kombucha tea. Emir. J. Food Agric. 2018, 30, 732–741. [Google Scholar]
- Polat, A.; Sat, I.G.; IIgaz, S. Comparison of black tea volatiles depending on the grades and different drying temperatures. J. Food Process. Preserv. 2018, 42, e13653. [Google Scholar] [CrossRef]
- Jayabalan, R.; Marimuthu, S.; Thangaraj, P.; Sathishkumar, M.; Binupriya, A.R.; Swaminathan, K.; Yun, S.E. Preservation of kombucha tea-effect of temperature on tea components and free radical scavenging properties. J. Agric. Food Chem. 2008, 56, 9064–9071. [Google Scholar] [CrossRef] [PubMed]
- El-Gazzar, N.; Almaary, K.; Ismail, A.; Polizzi, G. Influence of Funneliformis mosseae enhanced with titanium dioxide nanoparticles (TiO2NPs) on Phaseolus vulgaris L. under salinity stress. PLoS ONE 2020, 15, e0235355. [Google Scholar] [CrossRef] [PubMed]
- El-Sayed, A.; Enan, G.; Al-Mohammadi, A.-R.; Moustafa, H.A.; El-Gazzar, N. Detection, purification and elucidation of chemical structure and antiproliferative activity of taxol produced by Penicillium chrysogenum. Molecules 2020, 25, 4822. [Google Scholar] [CrossRef] [PubMed]
- El-Gazzar, N.; Ismail, A.M. The potential use of Titanium, Silver and Selenium nanoparticles in controlling leaf blight of tomato caused by Alternaria alternata. Biocatal. Agric. Biotechnol. 2020, 27, 101708. [Google Scholar] [CrossRef]
- El-Gazzar, N.; Almanaa, T.N.; Reda, R.M.; El Gaafary, M.; Rashwan, A.; Mahsoub, F. Assessment the using of silica nanoparticles (SiO2NPs) biosynthesized from rice husks by Trichoderma harzianum MF780864 as water lead adsorbent for immune status of Nile tilapia (Oreochromis niloticus). Saudi J. Biol. Sci. 2021, 28, 5119–5130. [Google Scholar] [CrossRef]
- De Man, J.C.; Rogosa, M.; Sharpe, M.E. A Medium for Larson. In Block S.S. Disinfection, Sterilization and Preservation, 4th ed.; Morton, E.L., Alcohols, H.E., Eds.; Lea & Febiger: Philadelphia, PA, USA, 1960. [Google Scholar]
- Osman, A.; El-Gazzar, N.; Almanaa, T.N.; El-Hadary, A.; Sitohy, M. Lipolytic postbiotic from Lactobacillus paracasei manages metabolic syndrome in albino wistar rats. Molecules 2021, 26, 472. [Google Scholar] [CrossRef] [PubMed]
- Holt, J.G. Facultatively anaerobic Gram-negative rods, subgroup 1: Family Enterobacteriaceae. In Bergey’s Manual of Determinative Bacteriology, 9th ed.; Holt, J.G., Ed.; Lippincott Williams and Wilkins: Baltimore, MA, USA, 1994; pp. 175–189. ISBN 9780683006032. [Google Scholar]
- Kurtzman, C.P.; Fell, J.W. The Yeasts a Taxonomic Study, 4th ed.; Elsevier: Amsterdam, The Netherlands, 1998; ISBN 978-0-123-84708-9. [Google Scholar]
- Performance Standards for Antimicrobial Disk Susceptibility Test: Approved Standard M2-A6, 6th ed.; National Committee for Clinical Laboratory Standards (NCCLS): Wayne, PA, USA, 2002.
- Clinical and Laboratory Standards Institute (CLSI). Performance Standards for Antimicrobial Susceptibility Testing: Eighteenth Informational Supplement; CLSI: Wayne, PA, USA, 2008. [Google Scholar]
- Durairaj, S.; Srinivasan, S.; Lakshmana-perumalsamy, P. In vitro antibacterial activity and stability of garlic extract at different pH and temperature. Electron. J. Biol. 2009, 5, 5–10. [Google Scholar]
- El-Bahr, S.; Elbakery, A.; El-Gazzar, N.; Amin, A.; Al-Sultan, S.; Alfattah, M.; Shousha, S.; Alhojaily, S.; Shathele, M.; Sabeq, I.; et al. Biosynthesized iron oxide nanoparticles from Petroselinum crispum leaf extract mitigate lead-acetate-induced anemia in male albino rats: Hematological, biochemical and histopathological features. Toxics 2021, 9, 123. [Google Scholar] [CrossRef] [PubMed]
- Victoria, C.N.; Harrison, J.; Cox, J.A.G. Dissecting the antimicrobial compostion of honey. Antibiotics 2019, 8, 251. [Google Scholar] [CrossRef]
- Armstrong, R.A.; Eperjesi, F.; Gilmartin, B. An introduction to analysis of variance (ANOVA) with special reference to data from clinical experiments in optometry. Opthalmic Physiol. Opt. 2002, 20, 235–241. [Google Scholar] [CrossRef]

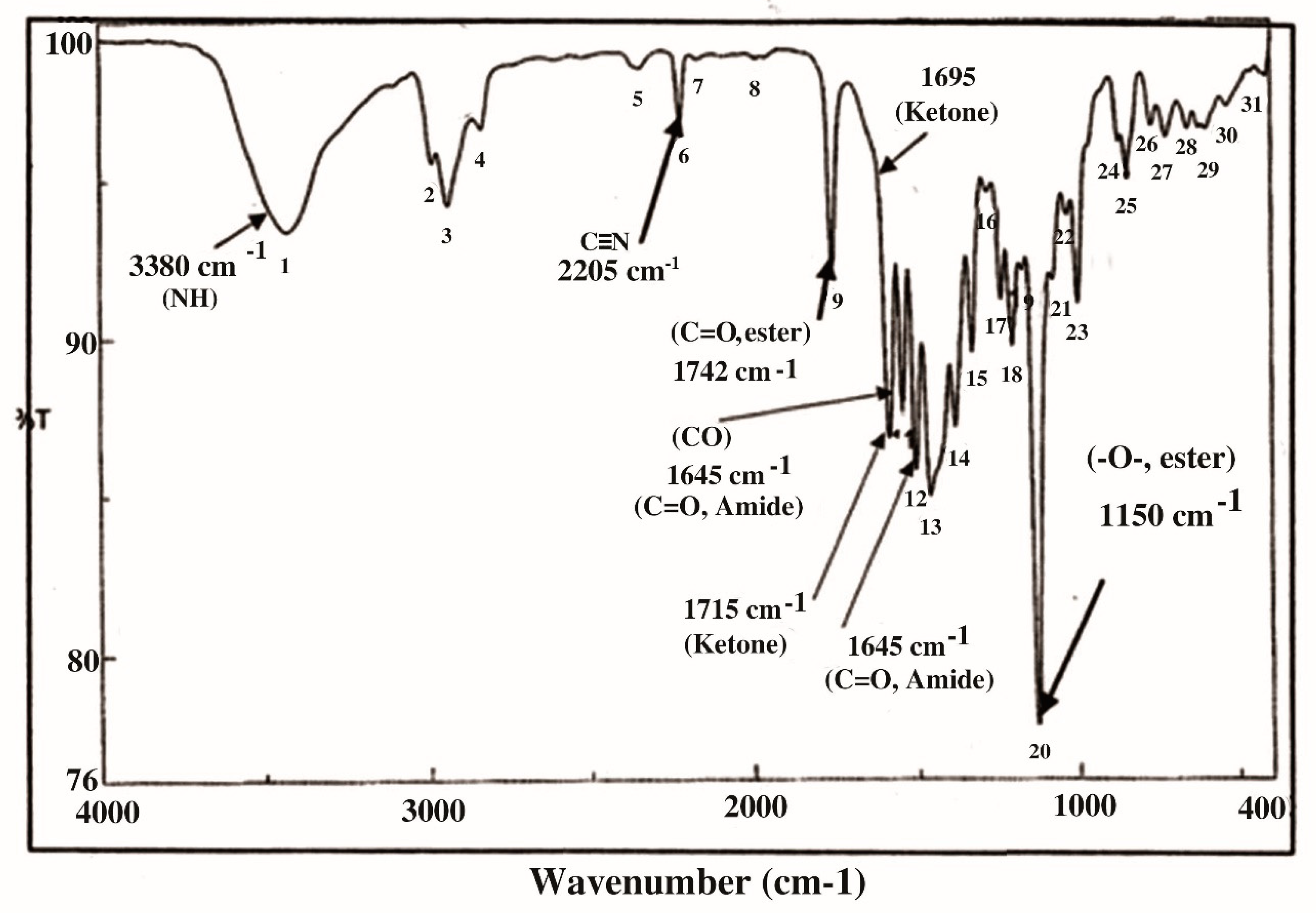


| Classification and Compound Name | Mol.wt & Mol. Formula | Parent Ion (M+) | Area | Base Peak (m/z) (100%) | |
|---|---|---|---|---|---|
| Group A: Heterocyclic Alcohols | |||||
| 1. | 2-Hydroxy methyl furan | C5H6O2 (98) | 99 | 1.35 | 98.0 |
| 2. | 2-(4-Hydroxyphenyl) ethanol | C8H10O2 (138.0) | 138.0 | 0.83 | 1.07 |
| Group B: Heterocyclic Acids | |||||
| 1. | 1,2,3,4-Tetrahydro-2,4-dioxo-5-carboxy methyl uracil | C6H6N2O4 (170.0) | 170.0 | 0.30 | 112.0 |
| 2. | Hexadecanoic acid CH3(CH2)14-COOH | C16H32O2 (256.0) | 256.0 | 0.66 | 73.0 |
| 3. | Ethyl-2-[2,2-dimethylcyclopropanecarboxylate (cyclopropane carboxylic acid) | C10H14Cl2O2 (236.0) | 236.0 | 0.42 | 163.0 |
| Group C: Lactone | |||||
| 1. | 3,5-Dihydroxy-6-methyl-2,3-diydroxy-4H-pyran-4-one | C6H8O4 (144.0) | 144.0 | 0.85 | 43.0 |
| 2. | 4-Hexyl-2,5-dihydro-2,5-dioxo-3-furan acetic acid | C12H16O5 (240.0) | 240.0 | 0.12 | 126.0 |
| 3. | 3-Methoxy-2,4,6-trimethylcyclohex-2-enone | C10H16O2 (168.0) | 168.0 | 0.12 | 126.0 |
| 4. | 3,7-Dimethyl-1-[2-(vinyloxy) ethyl-3,7-dihydro-1H-purine-2,6-dione | C11H14N4O (250.0) | 250.0 | 0.55 | 180.0 |
| 5. | S-[(2E)-1,3-diphenyl-2-butenyl] dimethylthiocarbamate | C19H21NOS (311.0) | 311.0 | 0.22 | 207.0 |
| Group D: Condensed Heterocyclic cpd | |||||
| 1. | 2,3-Dihydrobenzofuran | C8H8O (120.0) | 120.0 | 0.47 | 0.91 |
| 2. | 7,7-Dimethyl-1-Isobutyl hexahydro-2-benzofuran-3a (3H)-ol | C14H26O2 (226.0) | 226.0 | 0.51 | 159.0 |
| Group E: Antibiotics | |||||
| 1. | Cypermethrin | C22H19Cl2NO3 (415.0) | 415.0 | 0.30 | 163.0 |
| 2. | Cyhalothrin | C23H19Co F3NO3 (449.0) | 449.0 | 0.30 | 181.0 |
| Group F: Heterocyclic Esters | |||||
| 1. | 5-Acetoxymethyl-2-furaldehyde | C8H8O4 (168.0) | 168.0 | 0.38 | 126.0 |
| Group G: Heterocyclic Aldehydes | |||||
| 1. | 5-Hydroxymethyl furfural | C6H8O3 (126.0) | 126.0 | 13.45 | 97.0 |
| Group H: Unsat. Fatty Acids | |||||
| 1. | 2-Hexadecenoic acid | C16 H30O2 (254.0) | 254.0 | 0.40 | 43.0 |
| Group I: Alkaloids | |||||
| 1. | 1,3,7-Trimethyl-3,7-dihydro-1H-purine-2,6-dione (caffeine) | C8H10N4O2 (194.0) | 194.0 | 69.24 | 109.0 |
| Tested Organism | Inhibition Zone Diameters (mm) | ||||
|---|---|---|---|---|---|
| UKB | KFB | NKB | HKB | p-Value | |
| Salmonella typhimurium ATCC14028 | N.A. | 14.0 ± 0.2 | 1.3 ± 0.2 | 10 ± 0.1 | 00.000 |
| List. Monocytogenes ATCC4957 | N.A. | 15 ± 0.0 | 1.0 ± 0.0 | 7 ± 0.3 | 0.000 |
| B. cereus ATCC14579 | N.A. | 14.5 ± 0.18 | 1.4 ± 0.0 | 8.5 ± 0.0 | 0.000 |
| S. aureus ATCC6538 | N.A. | 19 ± 0.1 | 7 ± 0.0 | 12 ± 0.1 | 0.000 |
| E. coli ATCC 11229 | N.A. | 18 ± 0.25 | 5.3 ± 0.0 | 12 ± 0.0 | 0.000 |
| A. flavus ATCC16872 | N.A. | 12 ± 0.0 | 1.1 ± 0.1 | 5.0 ± 0.0 | 0.000 |
| A. niger ATCC20611 | N.A. | 9.5 ± 0.0 | 0.9 ± 0.2 | 4.6 ± 0.0 | 0.000 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Al-Mohammadi, A.-R.; Ismaiel, A.A.; Ibrahim, R.A.; Moustafa, A.H.; Abou Zeid, A.; Enan, G. Chemical Constitution and Antimicrobial Activity of Kombucha Fermented Beverage. Molecules 2021, 26, 5026. https://doi.org/10.3390/molecules26165026
Al-Mohammadi A-R, Ismaiel AA, Ibrahim RA, Moustafa AH, Abou Zeid A, Enan G. Chemical Constitution and Antimicrobial Activity of Kombucha Fermented Beverage. Molecules. 2021; 26(16):5026. https://doi.org/10.3390/molecules26165026
Chicago/Turabian StyleAl-Mohammadi, Abdul-Raouf, Ahmed A. Ismaiel, Rehab A. Ibrahim, Ahmed H. Moustafa, Azza Abou Zeid, and Gamal Enan. 2021. "Chemical Constitution and Antimicrobial Activity of Kombucha Fermented Beverage" Molecules 26, no. 16: 5026. https://doi.org/10.3390/molecules26165026
APA StyleAl-Mohammadi, A.-R., Ismaiel, A. A., Ibrahim, R. A., Moustafa, A. H., Abou Zeid, A., & Enan, G. (2021). Chemical Constitution and Antimicrobial Activity of Kombucha Fermented Beverage. Molecules, 26(16), 5026. https://doi.org/10.3390/molecules26165026







