Recovery of Chlorogenic Acids from Agri-Food Wastes: Updates on Green Extraction Techniques
Abstract
:1. Introduction
2. Chlorogenic Acids in Agri-Food Wastes
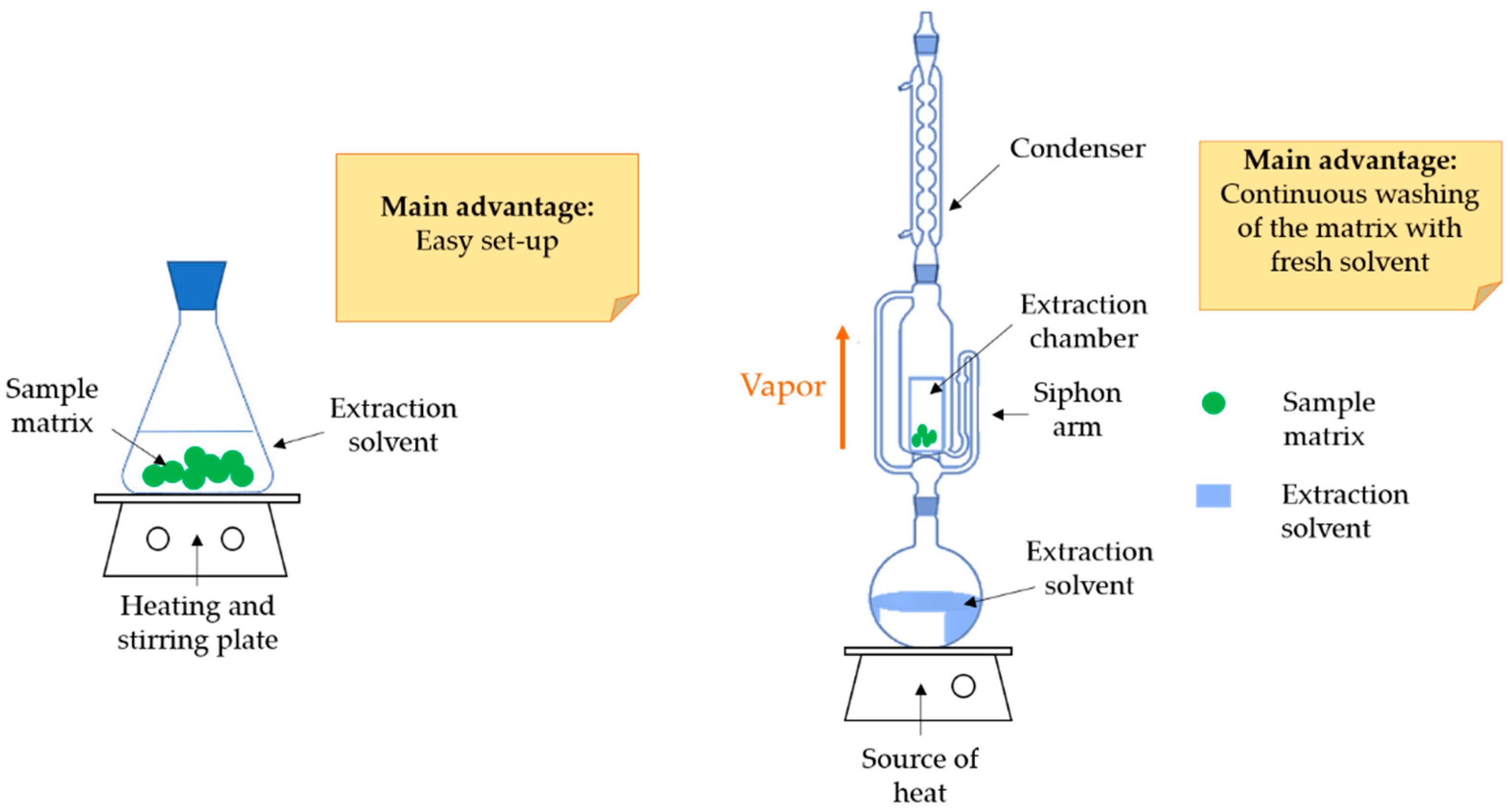
3. Conventional Extraction Methods
4. Innovative Green Extraction Techniques
4.1. Microwave Assisted Extraction (MAE)
4.2. Ultrasound-Assisted Extraction (UAE)
4.3. Supercritical Fluid Extraction (SFE)
4.4. Pressurized Liquid Extraction (PLE)
5. Comparison among the Reviewed Extraction Techniques: Is There a Better One to Recover CGAs?
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Available online: http://www.fao.org/food-loss-and-food-waste/flw-data (accessed on 26 June 2021).
- Available online: https://ec.europa.eu/environment/circular-economy/ (accessed on 26 June 2021).
- Panzella, L.; Moccia, F.; Nasti, R.; Marzorati, S.; Verrotta, L.; Napolitano, A. Bioactive phenolic compounds from agri-food wastes: An update on green and sustainable extraction methodologies. Front. Nutr. 2020, 7, 60. [Google Scholar] [CrossRef] [PubMed]
- Kumar, K.; Yadav, A.N.; Kumar, V.; Vyas, P.; Sigh Dhaliwal, H. Food waste: A potential bioresource for extraction of nutraceuticals and bioactive compounds. Bioresour. Bioprocess. 2017, 4, 18. [Google Scholar] [CrossRef] [Green Version]
- Sagar, N.A.; Pareek, S.; Sharma, S.; Yahia, E.M.; Lobo, M.G. Fruit and vegetable waste: Bioactive compounds, their extraction, and possible utilization. Compr. Rev. Food Sci. Food Saf. 2018, 17, 1–20. [Google Scholar] [CrossRef] [Green Version]
- Zuin, V.G.; Ramin, L.Z. Green and sustainable separation of natural products from agro-indutrial waste: Challenges, potentialities and perspectives on emerging approaches. Top Curr. Chem. 2018, 376, 229–282. [Google Scholar] [CrossRef] [Green Version]
- Upadhyay, R.; Rao, M.J. An outlook on chlorogenic acids—Occurrence, chemistry, technology, and biological activities. Crit. Rev. Food Sci. Nutr. 2013, 53, 968–984. [Google Scholar] [CrossRef]
- Liang, N.; Kitts, D.D. Role of chlorogenic acids in controlling oxidative and infiammatory stress conditions. Nutrients 2016, 8, 16. [Google Scholar] [CrossRef] [Green Version]
- Ding, Y.; Cao, Z.; Cao, L.; Ding, G.; Wang, Z.; Xiao, W. Antiviral activity of chlorogenic acid against influenza A (H1N1/H3N2) virus and its inhibition of neuraminidase. Sci. Rep. 2017, 7, 45723. [Google Scholar] [CrossRef] [Green Version]
- Lou, Z.; Wang, H.; Zhu, S.; Ma, C.; Wang, Z. Antibacterial activity and mechanism of action of chlorogenic acid. J. Food Sci. 2011, 76, 6. [Google Scholar] [CrossRef]
- Sun, Z.; Zhang, X.; Wu, H.; Wang, H.; Bian, H.; Zhu, Y.; Xu, W.; Liu, F.; Wang, D.; Fu, L. Antibacterial activity and action mode of chlorogenic acid against Salmonella Enteritidis, a foodborne pathogen in chilled fresh chicken. World J. Microb. Biot. 2020, 36, 24. [Google Scholar] [CrossRef]
- Chen, D.; Pan, D.; Tang, S.; Tan, Z.; Zhang, Y.; Fu, Y.; Lü, G.; Huang, Q. Administration of chlorogenic acid alleviates spinal cord inkury via TLR4/NF-kB and p38 signaling pathway anti-infiammatory activity. Mol. Med. 2018, 17, 1340–1349. [Google Scholar] [CrossRef] [Green Version]
- Li, L.; Su, C.; Chen, X.; Wang, Q.; Jiao, W.; Luo, H.; Tang, J.; Wang, W.; Li, S.; Guo, S. Chlorogenic acids in cardiovascular disease: A review of dietary consumption, pharmacology, and pharmacokinetics. J. Agric. Food Chem. 2020, 68, 6464–6484. [Google Scholar] [CrossRef]
- Heitman, E.; Ingram, D.K. Cognitive and neuroprotective effects of chlorogenic acid. Nutr. Neurosci. 2017, 20, 32–39. [Google Scholar] [CrossRef]
- Lu, H.; Tian, Z.; Cui, Y.; Liu, Z.; Ma, X. Chlorogenic acid: A comprehensive review of the dietary sources, processing effects, bioavailability, beneficial properties, mechanism of action, and future directions. Compr. Rev. Food Sci. Food Saf. 2020, 19, 3130–3158. [Google Scholar] [CrossRef] [PubMed]
- Naaved, M.; Hejezi, V.; Abbas, M.; Kamboh, A.A.; Khan, G.J.; Shumzaid, M.; Ahmad, F.; Babazadeh, D.; FangFang, X.; Modarresi-Ghazani, F.; et al. Chlorogenic acid (CGA): A pharmacological review and a call for further research. Biomed. Pharmacother. 2018, 97, 67–84. [Google Scholar] [CrossRef]
- Wianowska, D.; Gil, M. Recent advances in extraction and analyses procedures of natural chlorogenic acids. Phyrochem. Rev. 2019, 18, 273–302. [Google Scholar] [CrossRef] [Green Version]
- Clifford, M.N. Chlorogenic acids and other cinnamates-nature, occurrence and dietary burden. J. Sci. Food Agric. 1999, 79, 362–372. [Google Scholar] [CrossRef]
- Janissen, B.; Huynh, T. Chemical composition and value-adding applications of coffee industry byproducts: A review. Resour. Conserv. Recycl. 2018, 128, 110–117. [Google Scholar] [CrossRef]
- Angeloni, S.; Nzekoue, F.K.; Navarini, L.; Sagratini, G.; Torregiani, E.; Vittori, S.; Caprioli, G. An analytical method for the simultaneous quantification of 30 bioactive compounds in spent coffee ground by HPLC-MS/MS. J. Mass Spectrom. 2020, 55, e4519. [Google Scholar] [CrossRef]
- Fanali, C.; Della Posta, S.; Dugo, L.; Gentili, A.; Mondello, L.; De Gara, L. Choline-chloride and betaine-based deep eutectic solvents for greenextraction of nutraceutical compounds from spent coffee ground. J. Pharm. Biomed. Anal. 2020, 189, 113421. [Google Scholar] [CrossRef]
- Yoo, D.E.; Jeong, K.M.; Han, S.Y.; Kim, E.M.; Jin, Y.; Lee, J. Deep eutectic solvent-based valorization of spent coffee grounds. Food Chem. 2018, 255, 357–364. [Google Scholar] [CrossRef]
- Silva, M.d.O.; Honfoga, J.N.B.; Medeiros, L.L.; Madruga, M.S.; Bezerra, T.K.A. Obtaining bioactive compounds from the coffee husk (coffea arabica l.) using different extraction methods. Molecules 2021, 26, 46. [Google Scholar] [CrossRef]
- Guglielmetti, A.; D’Ignoti, V.; Ghirardello, D.; Belviso, S.; Zeppa, G. Optimisation of ultrasound and microwave-assisted extraction of caffeoyquinic acids and caffeine from coffee silverskin using response surface methodology. Ital. J. Food Sci. 2017, 29, 409–423. [Google Scholar]
- Joly, N.; Souidi, K.; Depraetere, D.; Wilsand, D.; Martin, P. Potato by-products as a source of natural chlorogenic acids and phenolic compounds: Extraction, characterization and antioxidant capacity. Molecules 2021, 26, 177. [Google Scholar] [CrossRef] [PubMed]
- Riciputi, Y.; Diaz-de-Cerio, E.; Akyolc, H.; Capanoglu, E.; Cerretani, L.; Caboni, M.F.; Verardo, V. Establishment of ultrasound-assisted extraction of phenolic compounds from industrial potato by-products using response surface methodology. Food Chem. 2018, 269, 258–263. [Google Scholar] [CrossRef] [PubMed]
- Jiménez-Moreno, N.; Cimminelli, M.J.; Volpe, F.; Ansó, R.; Esparza, I.; Mármol, I.; Rodríguez-Yoldi, M.J.; Ancín-Azpilicueta, C. Phenolic composition of artichoke waste and its antioxidant capacity on differentiated caco-2 cells. Nutrients 2019, 11, 1723. [Google Scholar] [CrossRef] [Green Version]
- Punzi, R.; Paradiso, A.; Fasciano, C.; Trani, A.; Faccia, M.; de Pinto, M.C.; Gambacorta, G. Phenols and antioxidant activity in vitro and in vivo of aqueous extracts obtained by ultrasound-assisted extraction from artichoke by-products. Nat. Prod. Commun. 2014, 9, 1315–1318. [Google Scholar] [CrossRef] [Green Version]
- Pagano, I.; Piccinelli, A.L.; Celano, R.; Campone, L.; Gazzerro, P.; Russo, M.; Rastrelli, L. Pressurized hot water extraction of bioactive compounds from artichoke by-products. Electrophoresis 2018, 39, 1899–1907. [Google Scholar] [CrossRef] [PubMed]
- Maietta, M.; Colombo, R.; Lavecchia, R.; Sorrenti, M.; Zuorro, A.; Papetti, A. Arthicoke (Cynara cardunuculus L. var. scolymus) waste as a natural source of carbonyl trapping and antiglycative agents. Food Res. Int. 2017, 100, 780–790. [Google Scholar] [CrossRef]
- Routeay, W.; Orsat, V. MAE of phenolic compounds form blueberry leaves and comparison with other extraction methods. Ind. Crops Prod. 2014, 58, 36–45. [Google Scholar] [CrossRef]
- Ma, X.; Zhou, X.Y.; Qiang, Q.Q.; Zhang, Z.Q. Ultrasound-assisted extraction and preliminary purification of proanthocyanidins and chlorogenic acid from almond (Prunus dulcis) skin. J. Sep. Sci. 2014, 14, 1834–1841. [Google Scholar] [CrossRef]
- Jabbar, S.; Abid, M.; Wu, T.; Hashim, M.M.; Saeeduddin, M.; Hu, B.; Lei, S.; Zeng, X. Ultrasound-assisted extraction of bioactive compounds and antioxidants from carrot pomace: A response surface approach. J. Food Process. Preserv. 2015, 39, 878–1888. [Google Scholar] [CrossRef]
- Kalantari, S.; Roufegarinejad, L.; Sajad Pirsa, S.; Gharekhani, M. Green extraction of bioactive compounds of pomegranate peel using β-Cyclodextrin and ultrasound. Main Group Chem. 2020, 19, 61–80. [Google Scholar] [CrossRef]
- Vieira, V.; Prieto, M.A.; Barros, L.; Coutinho, J.A.P.; Ferreira, O.; Ferreira, I.C.F.R. Optimization and comparison of maceration and microwave extraction systems for the production of phenolic compounds from Juglans regia L. for the valorization of walnuts leaves. Ind. Crops Prod. 2017, 107, 341–352. [Google Scholar] [CrossRef] [Green Version]
- Castaldo, L.; Izzo, L.; De Pascale, S.; Narváez, A.; Rodriguez-Carrasco, Y.; Ritieni, A. Chemical composition in vitro bioaccessibility and antioxidant acrtivity of polyphenolic compounds from nutraceutical fennel waste extract. Molecules 2021, 26, 1968. [Google Scholar] [CrossRef] [PubMed]
- Tranfić Bakić, M.; Pedisić, S.; Zorić, Z.; Dragović-Uzelac, V.; Ninčević Grassino, A. Effect of microwave-assisted extraction on polyphenols recovery from tomato peel waste. Acta Chim. Solv. 2019, 66, 367–377. [Google Scholar] [CrossRef] [Green Version]
- Pellicanò, T.M.; Sicari, V.; Loizzo, M.R.; Leporini, M.; Falco, F.; Poiana, M. Optimizing the supercritical fluid extraction process of bioactive compounds from processed tomato skin by-products. Food Sci. Technol. 2020, 40, 692–697. [Google Scholar] [CrossRef] [Green Version]
- Ninčević Grassino, A.; Djacović, S.; Bosiljkov, T.; Halambek, J.; Zorić, Z.; Dragović-Uzelac, V.; Petrović, M.; Rimac Brnčić, S. Valorisation of tomato peel waste as a sustainable source of pectin, polyphenols and fatty acids recovery using sequential extraction. Waste Biomass Valori. 2020, 11, 4593–4611. [Google Scholar] [CrossRef]
- Baiano, A.; Bevilacqua, L.; Terracone, C.; Contò, F.; Del Nobile, M.A. Single and interactive effects of process variables on microwave-assisted and conventional extraction of antioxidants from vegetable solid waste. J. Food Eng. 2014, 120, 135–145. [Google Scholar] [CrossRef]
- Rebollo-Hernanz, M.; Cañas, S.; Taladrid, D.; Benítez, V.; Bartolomé, B.; Aguilera, Y.; Martín-Cabrejas, M.A. Revalorization of coffee husk: Modeling and optimizing green sustainable extraction of phenolic compounds. Foods 2021, 10, 653. [Google Scholar] [CrossRef]
- Ramón-Gonçalves, M.; Gómez-Mejía, E.; Rosales-Conrado, N.; León-González, M.E.; Madrid, Y. Exraction, identification and quantification of polyphenols from spent coffee grounds by chromatographic methods and chemometric analyses. Waste Manag. 2019, 96, 15–24. [Google Scholar] [CrossRef]
- Al-Dhabi, N.A.; Ponmurugan, K.; Maran Jeganathan, P. Development and validation of ultrasound-assisted solid-liquid extraction of phenolic compounds from waste spent coffee grounds. Ultrason Sonochem 2017, 34, 206–213. [Google Scholar] [CrossRef]
- Caballero-Galván, A.S.; Restrepo-Serna, D.L.; Ortiz-Sánchez, M.; Cardona-Alzate, C.A. Analysis of extraction kinetics of bioactive compounds from spent coffee grounds (Coffea arábica). Waste Biomass Valorization 2018, 9, 2381–2389. [Google Scholar] [CrossRef]
- Mena-García, A.; Rodríguez-Sánchez, S.; Ruiz-Matute, A.I.; Sanz, M.L. Exploitation of artichoke byproducts to obtain bioactive extracts enriched in inositols and caffeoylquinic acids by microwave assisted extraction. J. Chromatogr. A 2020, 1613, 460703. [Google Scholar] [CrossRef]
- Rabelo, R.S.; Machado, M.T.C.; Martínez, J.; Hubinger, M.D. Ultrasound assisted extraction and nanofiltration of phenolic compounds from artichoke solid wastes. J. Food Eng. 2016, 178, 170–180. [Google Scholar] [CrossRef]
- Wu, T.; Yan, J.; Liu, R.; Marcone, M.F.; Akber Aisa, H.; Tsao, R. Optimization of microwave-assisted extraction of phenolics from potato and its downstream waste using orthogonal array design. Food Chem. 2012, 133, 1292–1298. [Google Scholar] [CrossRef]
- Rodríguez Amado, I.; Franco, D.; Sánchez, M.; Zapata, C.; Vázquez, J.A. Optimisation of antioxidant extraction from Solanum tuberosum potato peel waste by surface response methodology. Food Chem. 2014, 165, 290–299. [Google Scholar] [CrossRef] [Green Version]
- Singh, P.P.; Saldaña, M.D.A. Subcritical water extraction of phenolic compounds from potato peel. Food Res. Int. 2011, 44, 2452–2458. [Google Scholar] [CrossRef]
- Safdar, M.N.; Kausar, T.; Nadeem, M. Comparison of ultrasound and maceration techniques for the extraction of polyphenols from the mango peel. J. Food Process. Preserv. 2017, 41, e13028. [Google Scholar] [CrossRef]
- Mellinas, A.C.; Jiménez, A.; Garrígos, M.C. Optimization of microwave-assisted extraction of cocoa bean shell waste and evaluation of its antioxidant, physicochemical and functional properties. LWT-Food Sci. Technol. 2020, 127, 109361. [Google Scholar] [CrossRef]
- Safdar, M.N.; Kausar, T.; Jabbar, S.; Mumtz, A.; Ahad, K.; Saddozi, A.A. Extraction and quantification of polyphenols from kinnow (Citrus recticulate L.) peel using ultrasound and maceration techniques. J. Food Drug Anal. 2017, 25, 488–500. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Náthia-Neves, G.; Alonso, E. Valorization of sunflower by-product using microwave-assisted extraction to obtain a rich protein flour: Recovery of chlorogenic acid, phenolic content and antioxidant capacity. Food Bioprod. Process. 2021, 125, 57–67. [Google Scholar] [CrossRef]
- Banožić, M.; Banjari, I.; Jakovljević, M.; Šubarić, D.; Tomas, S.; Babić, J.; Jokić, S. Optimization of ultrasound-assisted extraction of some bioactive compounds from tobacco waste. Molecules 2019, 24, 1611. [Google Scholar] [CrossRef] [Green Version]
- Sifola, M.I.; Carrino, L.; Cozzolino, E.; del Piano, L.; Graziani, G.; Ritieni, A. Potential of pre-harvest wastes of tobacco (Nicotiana tabacum L.) crops, grown for smoke products, as source of bioactive compounds (phenols and flavonoids). Sustainability 2021, 13, 2087. [Google Scholar] [CrossRef]
- Caballero, A.S.; Romero-García, J.M.; Castro, E.; Cardona, C.A. Supercritical fluid extraction for enhancing polyphenolic compounds production from olive waste extracts. Chem. Technol. Biotechnol. 2020, 95, 356–362. [Google Scholar] [CrossRef]
- da Silva, L.C.; Souza, M.C.; Sumere, B.R.; Silva, L.G.S.; da Cunha, D.T.; Barbero, G.F.; Bezerra, R.M.N.; Rostagno, M.A. Simultaneous extraction and separation of bioactive compounds from apple pomace using pressurized liquids coupled on-line with solid-phase extraction. Food Chem. 2020, 318, 126450. [Google Scholar] [CrossRef]
- Baiano, A. Recovery of biomolecules from food waste—A review. Molecules 2014, 19, 14821–14842. [Google Scholar] [CrossRef] [Green Version]
- Narita, Y.; Inouye, K. Degradation kinetics of chlorogenic acid at various pH values and effects of ascorbic acid and epigallocatechin gallate on its stability under alkaline conditions. J. Agric. Food Chem. 2013, 61, 966–972. [Google Scholar] [CrossRef]
- Dai, Y.; Spronsen, J.V.; Witkamp, G.-J.; Verpoorte, R.; Hae Coi, Y. Natural deep eutectic solvents as new potential media for green technology. Anal. Chim. Acta 2013, 766, 61–68. [Google Scholar] [CrossRef]
- Ruesgas-Ramón, M.; Figueroa-Espinoza, M.C.; Durand, E. Application of deep eutectic solvents (DES) for phenolic compounds extraction: Overview, challenges and opportunities. J. Agric. Food Chem. 2017, 65, 3591–3601. [Google Scholar] [CrossRef]
- Ruesgas-Ramón, M.; Suárez-Quiroz, M.L.; González-Ríos, M.L.; González-Ríos, O.; Baréa, B.; Cazals, G.; Figueroa-Espinoza, M.C.; Durand, E. Biomolecules extraction from coffee and cocoa by- and co-products using deep eutectic solvents. J. Sci. Food Agric. 2020, 100, 81–91. [Google Scholar] [CrossRef]
- Saalem, M.; Tanvir, M.; Akhtar, M.F.; Iqbal, M.; Saalem, A. antidiabetic potential of Mangifera indica L. cv. anwar ratol leaves: Medicinal application of food waste. Medicina 2019, 55, 353. [Google Scholar] [CrossRef] [Green Version]
- Andres, A.I.; Petron, M.J.; Lopez, A.M.; Timon, M.L. Optimization of extraction conditions to improve phenolic content and in vitro antioxidant activity in crafts brewers’ spent grain using response surface methodology (RSM). Foods 2020, 9, 1398. [Google Scholar] [CrossRef]
- Dawidowicz, A.; Tyoek, R. Thermal stability of 5-o-caffeoyilquinic acid in aqueous solutions at different heating conditions. J. Agric. Food Chem. 2010, 58, 12578–12584. [Google Scholar] [CrossRef]
- Radojković, M.; Zeković, Z.; Jokić, S.; Vidović, S.; Lepojević, Z.; Milošević, S. Optimization of solid-liquid extraction of antioxidants from black mulberry leaves by response surface methodology. Food Technol. Biotechnol. 2012, 50, 167–176. [Google Scholar]
- Paleologou, I.; Vasiliou, A.; Grigorakis, S.; Makris, D.P. Optimisation of a green ultrasound-assisted extraction process for potato peel (Solanum tuberosum) polyphenols using bio-solvents and response surface methodology. Biomass Conv. Bioref. 2016, 6, 289–299. [Google Scholar] [CrossRef]
- Florez, N.; Conde, E.; Dominguez, H. Microwave assisted water extraction of plant compounds. J. Chem. Technol. Biotechnol. 2015, 90, 590–607. [Google Scholar] [CrossRef]
- Vilas-Boas, A.A.; Oliveira, A.; Ribeiro, T.B.; Ribeiro, S.; Nunes, C.; Gómez-García, R.; Nunes, J.; Pintado, M. Impact of extraction process in non-compliant ‘Bravo de Esmolfe’ apples towards the development of natural antioxidant extracts. Appl. Sci. 2021, 11, 5916. [Google Scholar] [CrossRef]
- Kaderides, K.; Papaoikonomou, L.; Serafim, M.; Goula, A.M. Microwave-assisted extraction from pomegranate peels: Optimization, kinetics, and comparison with ultrasound extraction. Chem. Eng. Process. 2019, 137, 1–11. [Google Scholar] [CrossRef]
- Pettinato, M.; Casazza, A.A.; Ferrari, P.F.; Palombo, D.; Perego, P. Eco-sustainable recovery of antioxidants from spent coffee grounds by microwave-assisted extraction: Process optimization, kinetic modeling and biological validation. Food Bioprod. Process. 2019, 114, 31–42. [Google Scholar] [CrossRef]
- Galan, A.-M.; Calinescu, I.; Trifan, A.; Winkworth-Smith, C.; Calvo-Carrascal, M.; Dodds, C.; Binner, E. New insights into the role of selective and volumetric heating during microwave extraction: Investigation of polyphenolic compounds from sea buckthorn leaves using microwave-assisted extraction and conventional solvent extraction. Chem. Eng. Process. 2017, 116, 29–39. [Google Scholar] [CrossRef]
- Ranic, M.; Nikolic, M.; Pavlovic, M.; Buntic, A.; Siler-Marinkovic, S.; Dimitrijevic-Brankovic, S. Optimization of microwave-assisted extraction of natural antioxidants from spent espresso coffee grouds by response surface methodology. J. Clean. Prod. 2014, 80, 69–79. [Google Scholar] [CrossRef]
- Chen, Y.; Yu, Q.J.; Li, X.; Luo, Y.; Liu, H. Extraction and characterization of chlorogenic acid from tobacco residuals. Sep. Sci. Technol. 2007, 42, 3481–3492. [Google Scholar] [CrossRef] [Green Version]
- Li, Z.; Huang, D.; Tang, Z.; Deng, C.; Zhang, X. Fast determination of chlorogenic acid in tobacco residues using microwave-assisted extraction and capillary zone electrophoresis technique. Talanta 2010, 82, 1181–1185. [Google Scholar] [CrossRef]
- Nayak, B.; Dahmoune, F.; Moussi, K.; Remini, H.; Dairi, S.; Aoun, O.; Khodir, M. Comparison of microwave, ultrasound and accelerated-assisted solvent extraction for recovery of polyphenols from Citrus sinensis peel. Food Chem. 2015, 187, 507–516. [Google Scholar] [CrossRef]
- Pavlovic, N.; Jokić, S.; Jakovljević, M.; Blazić, M.; Molnar, M. Green extaction methods for active compounds from food waste—Cocoa bean shell. Foods 2020, 9, 140. [Google Scholar] [CrossRef] [Green Version]
- Liazid, A.; Palma, M.; Brigui, J.; Barroso, C.G. Investigation on phenolic compounds stability during microwave-assisted extraction. J. Chromatogr. A 2007, 1140, 29–34. [Google Scholar] [CrossRef]
- Zardo, I.; De Espindola Sobczyk, A.; Damasceno Ferreira Marczak, L.; Sarkis, J. Optimization of ultrasound assisted extraction of phenolic compounds from sunflower seed cake using response surface methodology. Waste Biomass. Valor. 2019, 10, 33–44. [Google Scholar] [CrossRef]
- Alara, O.R.; Abdurahman, N.H.; Ukaegbu, A.I.; Azhari, N.H. Vernonia cinereal leaves as the source of phenolic compounds, antioxidants, and anti-diabetic activity using microwave—Assisted extraction technique. Ind. Crops Prod. 2018, 122, 533–544. [Google Scholar] [CrossRef]
- Aamer, K.; Shahbaz, H.M.; Kwon, J.-H. Green extraction methods for polyphenols from plant matrices and their byproducts: A review. Compr. Rev. Food Sci. Food Saf. 2017, 16, 295–315. [Google Scholar] [CrossRef] [Green Version]
- Dobrinčić, A.; Repajić, M.; Garofulić, I.E.; Tuđen, L.; Dragović-Uzelac, V.; Levaj, B. Comparison of different extraction methods for the recovery of olive leaves polyphenols. Processes 2020, 8, 1008. [Google Scholar] [CrossRef]
- Alves Filho, E.G.; Sousa, V.M.; Rodrigues, S.; de Brito, E.S.; Fernandes, F.A.N. Green ultrasound-assisted extraction of chlorogenic acids from sweet potato peels and sonochemical hydrolysis of caffeoylquinic acids derivatives. Ultrason. Sonochem. 2020, 63, 104911. [Google Scholar] [CrossRef] [PubMed]
- Chemat, F.; Rombaut, N.; Sicaire, A.G.; Meullemiestre, A.; Fabiano-Tixier, A.S.; Abert-Vian, M. Ultrasound assisted extraction of food and natural products. Mechanisms, techniques, combinations, protocols and applications. A review. Ultrason. Sonochem. 2017, 34, 540–560. [Google Scholar] [CrossRef]
- Santos, H.M.; Lodeiro, C.; Capelo-Martínez, J.L. The power of ultrasound. In Ultrasound in Chemistry: Analytical Applications; Capelo-Martínez, J.L., Ed.; John and Wiley and Sons: Hoboken, NJ, USA, 2009; pp. 1–16. [Google Scholar] [CrossRef]
- Suslick, K.S.; Price, G.J. Applications of ultrasound to materials chemistry. Annu. Rev. Mater. Sci. 1999, 29, 295–326. [Google Scholar] [CrossRef] [Green Version]
- Saleh, I.A.; Vinatoru, M.; Mason, T.J.; Abdel-Azim, N.S.; Aboutabl, E.A.; Hammouda, F.M. A possible general mechanism for ultrasound-assisted extraction (UAE) suggested from the results of UAE of chlorogenic acid from Cynara scolymus L. (artichoke) leaves. Ultrason. Sonochem. 2016, 31, 330–336. [Google Scholar] [CrossRef]
- Reche, C.; Rossellò, C.; Umaña, M.M.; Eim, V.; Simal, S. Mathematicalmodelling of ultrasound-assisted extraction kinetics of bioactive compounds from artichoke by-products. Foods 2021, 10, 931. [Google Scholar] [CrossRef]
- Szente, L.; Szejti, J. Cyclodextrins as food ingredient. Trends Food Sci. Technol. 2004, 15, 137–142. [Google Scholar] [CrossRef]
- Gullòn, P.; Gullòn, B.; Romanì, A.; Rocchetti, G.; Lorenzo, J.M. Smart advanced solvents for bioactive compounds recovery from agri-food by-products: A review. Trends Food Sci. Technol. 2020, 101, 182–197. [Google Scholar] [CrossRef]
- Sapkale, G.N.; Patil, S.M.; Surwase, U.S.; Bhatbhage, P.K. Supercritical fluid extraction. Int. J. Chem. Sci. 2010, 8, 729–743. [Google Scholar]
- Al Jitan, S.; Alkhoori, S.A.; Yousef, L.F. Phenolic acids from plants: Extraction and application to human health. Stud. Nat. Prod. Chem. 2018, 58, 389–417. [Google Scholar] [CrossRef]
- Anticona, M.; Blesa, J.; Frigola, A.; Esteve, M.J. High biological value compounds extraction from citrus waste with non-conventional methods. Foods 2020, 9, 811. [Google Scholar] [CrossRef]
- da Silva, R.P.F.F.; Rocha-Santos, T.A.P.; Duarte, A.C. Supercritical fluid extraction of bioactive compounds. Trends Anal. Chem. 2016, 76, 40–51. [Google Scholar] [CrossRef] [Green Version]
- Andrade, K.S.; Gonçalvez, R.T.; Maraschin, M.; Ribeiro-do-Valle, R.M.; Martínez, J.; Ferreira, S.R. Supercritical fluid extraction from spent coffee grounds and coffee husks: Antioxidant activity and effect of operational variables on extract composition. Talanta 2012, 88, 544–552. [Google Scholar] [CrossRef]
- Raventós, M.; Duarte, S.; Alarcón, R. Application and Possibilities of Supercritical CO2 Extraction in Food Processing Industry: An Overview. Food Sci. Tech. Int. 2002, 8, 269–284. [Google Scholar] [CrossRef]
- Sharif, K.M.; Rahman, M.M.; Azmir, J.; Mohamed, A.; Jahurul, M.H.A.; Sahena, F.; Zaidul, I.S.M. Experimental design of supercritical fluid extraction—A review. J. Food Eng. 2014, 124, 105–116. [Google Scholar] [CrossRef]
- Uwineza, P.A.; Waśkiewicz, A. Recent advances in supercritical fluid extraction of natural bioactive compounds from natural plant material. Molecules 2020, 25, 3847. [Google Scholar] [CrossRef] [PubMed]
- García, P.; Fredes, C.; Cea, I.; Lozano-Sánchez, J.; Leyva-Jiménez, F.J.; Robert, P.; Vergara, C.; Jimenez, P. Recovery of bioactive compounds from pomegranate (punica granatum L.) peel using pressurized liquid extraction. Foods 2021, 10, 203. [Google Scholar] [CrossRef]
- Paini, M.; Casazza, A.A.; Aliakbarian, B.; Perego, P.; Binello, A.; Cravotto, G. Influence of ethanol/water ratio in ultrasound and high-pressure/ high-temperature phenolic compound extraction from agri-food waste. Int. J. Food Sci. Technol. 2016, 51, 349–358. [Google Scholar] [CrossRef]
- Mustafa, A.; Turner, C. Pressurized liquid extraction as a green approach in food and herbal plants extraction: A review. Anal. Chim. Acta 2011, 703, 8–18. [Google Scholar] [CrossRef]
- Tunchaiyaphum, S.; Eshtiaghi, M.N.; Yoswathana, N. Extraction of bioactive compounds from mango peels using green technology. Int. J. Chem. Eng. 2013, 4, 194–198. [Google Scholar] [CrossRef] [Green Version]
- Herrero, M.; Cifuentes, A.; Ibañez, E. Sub- and supercritical fluid extraction of functional ingredients from different natural sources: Plants, food-by-products, algae and microalgae A review. Food Chem. 2006, 98, 136–148. [Google Scholar] [CrossRef] [Green Version]
- Kheirkhah, H.; Baroutian, S.; Quek, S.Y. Evaluation of bioactive compounds extracted from Hayward kiwifruit pomace by subcritical water extraction. Food Bioprod. Process. 2019, 115, 143–153. [Google Scholar] [CrossRef]
- Plaza, M.; Turner, C. Pressurized hot water extraction of bioactives. Trends Anal. Chem. 2015, 71, 39–54. [Google Scholar] [CrossRef] [Green Version]
- Osorio-Tobon, J.F. Recent advances and comparisons of conventional and alternative extraction techniques of phenolic compounds. J. Food Sci. Technol. 2020, 57, 4299–4315. [Google Scholar] [CrossRef]
- Plaza, M.; Abrahamsson, V.; Turner, C. Extraction and neoformation of antioxidant compounds by pressurized hot water extraction from apple byproducts. J. Agric. Food Chem. 2013, 61, 5500–5510. [Google Scholar] [CrossRef]





| Source | Type of Waste | CGAs Content | Ref. |
|---|---|---|---|
| Cauliflower | external leaves, stems | 21–98 mg/kg fw | [40] |
| Celery | external leaves, stems | 13–19 mg/kg fw | [40] |
| Coffee | husk | 337 μg/g dw 1 | [23] |
| 90,567 μg/g dw | [41] | ||
| spent ground | 22.08 mg/g dw | [42] | |
| 1.36 mg/g dw | [43] | ||
| 1700–1757 mg/kg dw | [20] | ||
| 19.6 mg/g dw | [22] | ||
| 0.93 mg/g fw | [44] | ||
| silverskin | 1.06–2.68 g/kg dw | [24] | |
| Artichoke | bracts | 3.73 mg/g dw | [30] |
| 12.98 mg/g dw | [45] | ||
| 0.02–16.47 mg/g dw | [46] | ||
| leaves, stems | 3–16 mg/g dw | [29] | |
| leaves, outer bracts, stems | 74.2 mg/kg dw | [28] | |
| stems | 8.86 mg/g dw | [30] | |
| stalks | 1.56 mg/g dw | [45] | |
| receptacles | 32.10 mg/g dw | [45] | |
| leaves | 2.39 mg/g dw | [45] | |
| Tomato | peel | 6–62 mg/kg fw | [36] |
| 3.04–4.54 mg/g dw | [38] | ||
| 4.05 mg/kg dw | [37] | ||
| Potato | peel | 6.63 mg/g dw | [47] |
| 0.032–1.03 mg/g dw | [48] | ||
| 0.15 mg/ g dw | [49] | ||
| 1.3–4.1 mg/g dw | [26] | ||
| Walnut | leaves | 8.6–10.7 mg/g dw | [35] |
| Blueberry | leaves | 47.271–51.631mg/g dw | [31] |
| Mango | peel | 33.97 μg/g dw | [50] |
| Carrot | pomace | 17.79 mg/g dw | [33] |
| Cocoa | bean shell | 0.76 mg/g dw | [51] |
| Citrus | peel | 20.52 μg/g dw | [52] |
| Sunflower | by-products | 3.2–15 mg/g dw | [53] |
| Almond | skin | 15.99 mg/g dw | [32] |
| Tobacco | scrap, midrib, dust | 36.4–804.2 μg/g dw | [54] |
| residual stalks | 1198.0–1998.6 μg/g dw | [55] | |
| Olive | exhaust olive pomace | 0.31 mg/g dw | [56] |
| tree biomass | 0.24 mg/g dw | [56] | |
| leaves | 0.09 mg/g dw | [56] | |
| Apple | pomace | 0.718 mg/g dw | [57] |
| Pomegranate | peel | 3.07 mg/g dw | [34] |
| Source | Type of Waste | Extraction Method | Optimal Extraction Conditions | Ref. |
|---|---|---|---|---|
| Coffee | Husk | UAE | 50% EtOH, 35 °C, 1 h, 1:10 w/v, 40 kHz, 220V | [23] |
| CSE | 50% EtOH, 60 °C, 1 h, 1:10 w/v | [23] | ||
| Spent ground | CSE | 15–20% EtOH, 40–60 °C, 15–25 min, 0.30:25 w/v | [42] | |
| UAE | 100% EtOH, 40 °C, 34 min, 1:17 w/v, 20 KHz, 244 W | [43] | ||
| UAE | betaine:triethylene glycol 1:2, 30% water, 65 °C, 20 min, 1:15 w/v, 37 KHz, 200 W | [21] | ||
| Silverskin | MAE | 60% EtOH, 43.5 °C, 31.5 min, 1:35 w/v, 280 W | [24] | |
| CSE | 60% EtOH, 67.5 °C, 36.5 min, 1:35 w/v | [24] | ||
| Artichoke | Bracts, stems | CSE | 80% EtOH, 60 ± 0.1 °C, 60 min, 0.5:15 w/v | [30] |
| Leaves, external bracts, stalks, receptacles | MAE | 50% EtOH, 50–69 °C, 3 min, 0.3:10 w/v, 900 W | [45] | |
| Internal and external bracts | UAE | 75% EtOH, 25 °C, 10 min, 1:10 w/v, 20 kHz, 240 W | [46] | |
| Outer bracts, stems | UAE | 100% water, 60 min, 1:3 w/v, 25 kHz, 1200 W, 50 W/L | [28] | |
| Bracts, leaves | PHWE | 10% EtOH, 93 °C, static time 5 min, 2 cycles, 103 bar | [29] | |
| Tomato | Peel | MAE | 70% EtOH, 55 °C, 5 min, 1: 50 w/v, 0–500 W | [37] |
| Potato | Peel | MAE | 60% EtOH, 80 °C, 2 min, 1:40 w/v, 300 W, 120 rpm magnetic stirring | [47] |
| UAE | 83% glycerol, 80 °C, 90 min, 1:81 w/v, 37 kHz, 140 W, 35 W/L | [67] | ||
| SCW | 100% water, 160 °C, 60 min, 60 bar, 2 mL/min | [49] | ||
| Walnut | Leaves | MAE | 100% water, 61.1 °C, 3 min, 0.030:1 w/v, 850 W, 600 rpm magnetic stirring | [35] |
| CSE | 100% water, 29.9 °C, 150 min, 0.030:1 w/v, 600 rpm magnetic stirring | [35] | ||
| Blueberry | Leaves | MAE | 30% EtOH+ 0.03% citric acid (1.5 M), 4 min, 0.5:80 w/v, 141.1 W | [31] |
| Cocoa | Bean shell | MAE | 100% water, pH 12, 97 °C, 5 min, 0.04:1 w/v, 500 W, 400 rpm magnetic stirring | [51] |
| Sunflower | By-products | MAE | 70% EtOH, 200 W, 30 s, 1:10 w/v | [53] |
| Citrus | Peel | CSE | 80% EtOH, 40 °C, 20 h, 1:15 w/v | [52] |
| UAE | 80% EtOH, 45 °C, 60 min, 1:20 w/v, 35 kHz | [52] | ||
| Cauliflower | External leaves, stems | CSE | 100% water, 70 °C, 20 min, 1:1 w/w | [58] |
| MAE | 100% water, 4 min, 1:1 w/w, 750 W | [58] | ||
| Celery | External leaves, stems | CSE | 100% water, 70 °C, 20 min, 1:2 w/w | [58] |
| MAE | 100% water, 4 min, 1:2 w/w, 750 W | [58] | ||
| Tobacco | Midrib, dust, scrap | UAE | 40–55.43% EtOH, 30.14–53.59 °C, 15.19–30.31 min 1:10–11 w/v, 37 kHz, 50 W | [54] |
| Carrot | Pomace | UAE | 80% EtOH, 20 °C, 10 min, 1:50 w/v, 20 kHz, 70% amplitude level, 48 W/cm2 | [33] |
| Almond | Skin | UAE | 50% PEG200, 75 °C, 20 min, 1:20 w/v, 40 kHz, 120W | [32] |
| Apple | Pomace | PLE | 100% water, 60 °C, 60 min, 100 bar, 2 mL/min | [57] |
| Olive | Tree biomass | SFE | CO2+ 60% EtOH, 50°C, 60 min, 1:3 w/v, 300 bar | [56] |
| Desirable Advantages | CSE | MAE | UAE | SFE | PLE |
|---|---|---|---|---|---|
| Low cost | + | − | + | − | − |
| Easy to perform | + | + | + | - | − |
| No thermal effect | − | − | + | + | − |
| Time-saving | − | + | + | − | + |
| Low solvent consumption | − | + | + | + | + |
| Low energy consumption | − | + | + | - | − |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Frosi, I.; Montagna, I.; Colombo, R.; Milanese, C.; Papetti, A. Recovery of Chlorogenic Acids from Agri-Food Wastes: Updates on Green Extraction Techniques. Molecules 2021, 26, 4515. https://doi.org/10.3390/molecules26154515
Frosi I, Montagna I, Colombo R, Milanese C, Papetti A. Recovery of Chlorogenic Acids from Agri-Food Wastes: Updates on Green Extraction Techniques. Molecules. 2021; 26(15):4515. https://doi.org/10.3390/molecules26154515
Chicago/Turabian StyleFrosi, Ilaria, Irene Montagna, Raffaella Colombo, Chiara Milanese, and Adele Papetti. 2021. "Recovery of Chlorogenic Acids from Agri-Food Wastes: Updates on Green Extraction Techniques" Molecules 26, no. 15: 4515. https://doi.org/10.3390/molecules26154515
APA StyleFrosi, I., Montagna, I., Colombo, R., Milanese, C., & Papetti, A. (2021). Recovery of Chlorogenic Acids from Agri-Food Wastes: Updates on Green Extraction Techniques. Molecules, 26(15), 4515. https://doi.org/10.3390/molecules26154515









