Valorization of Artichoke Industrial By-Products Using Green Extraction Technologies: Formulation of Hydrogels in Combination with Paulownia Extracts
Abstract
1. Introduction
2. Results and Discussion
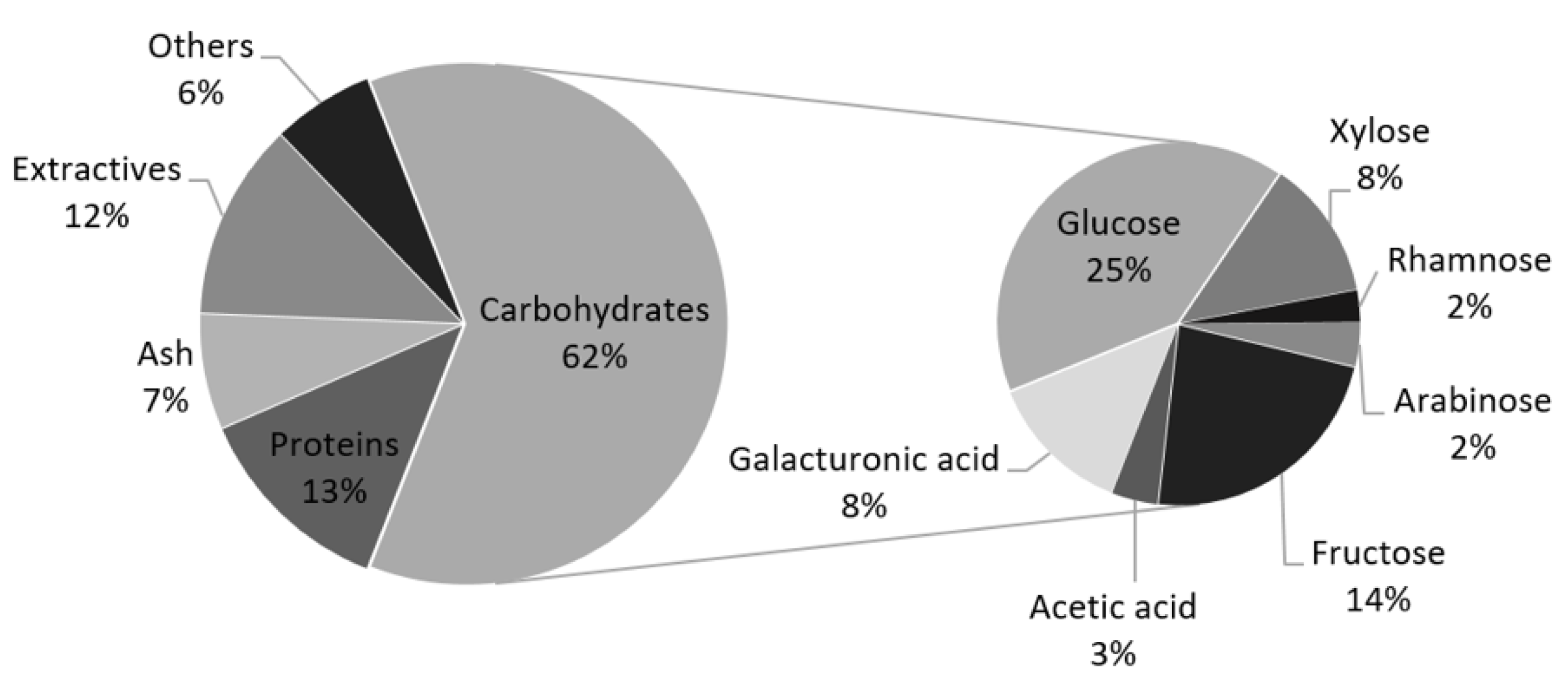
2.1. Artichoke Wastes Characterization
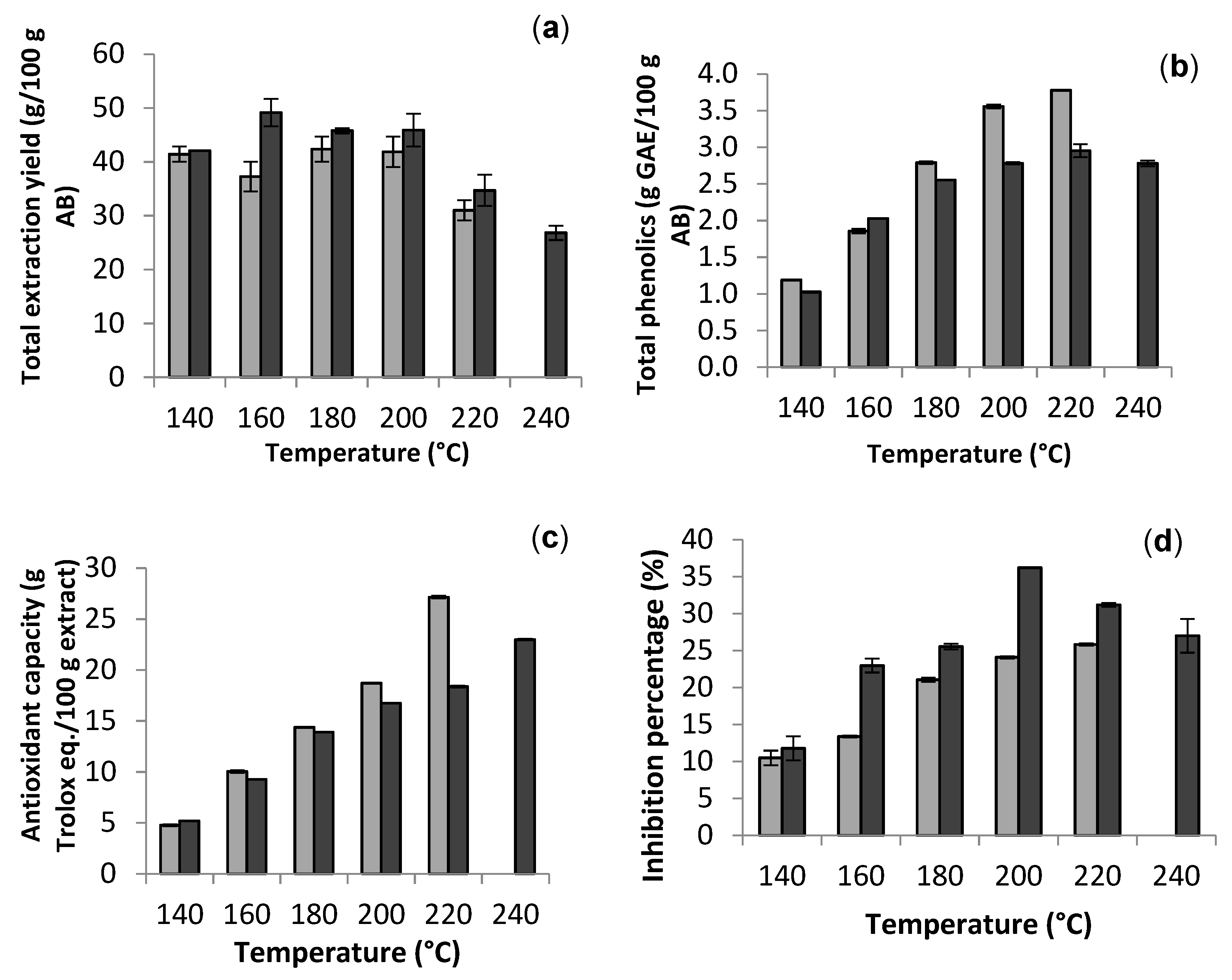
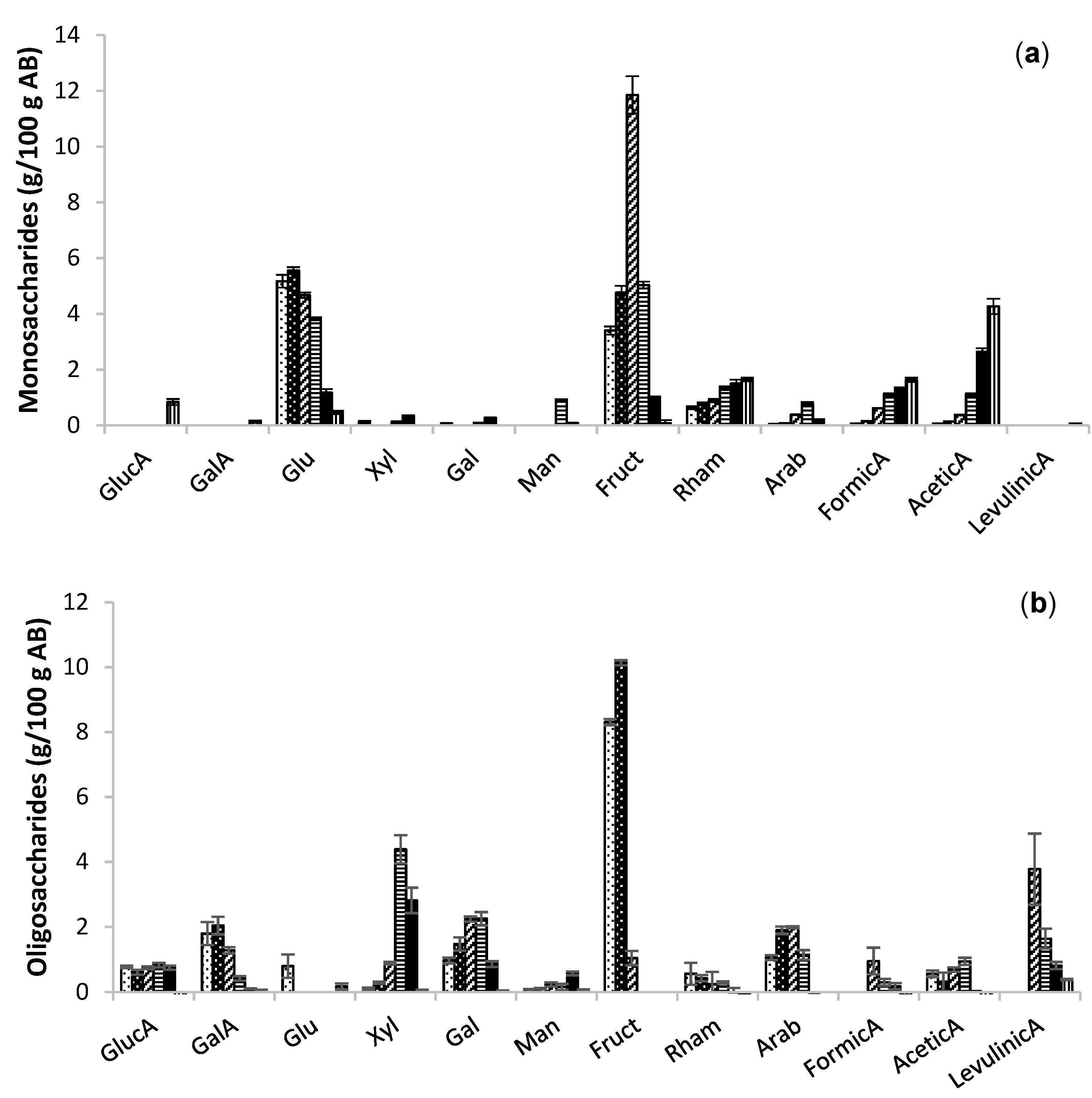
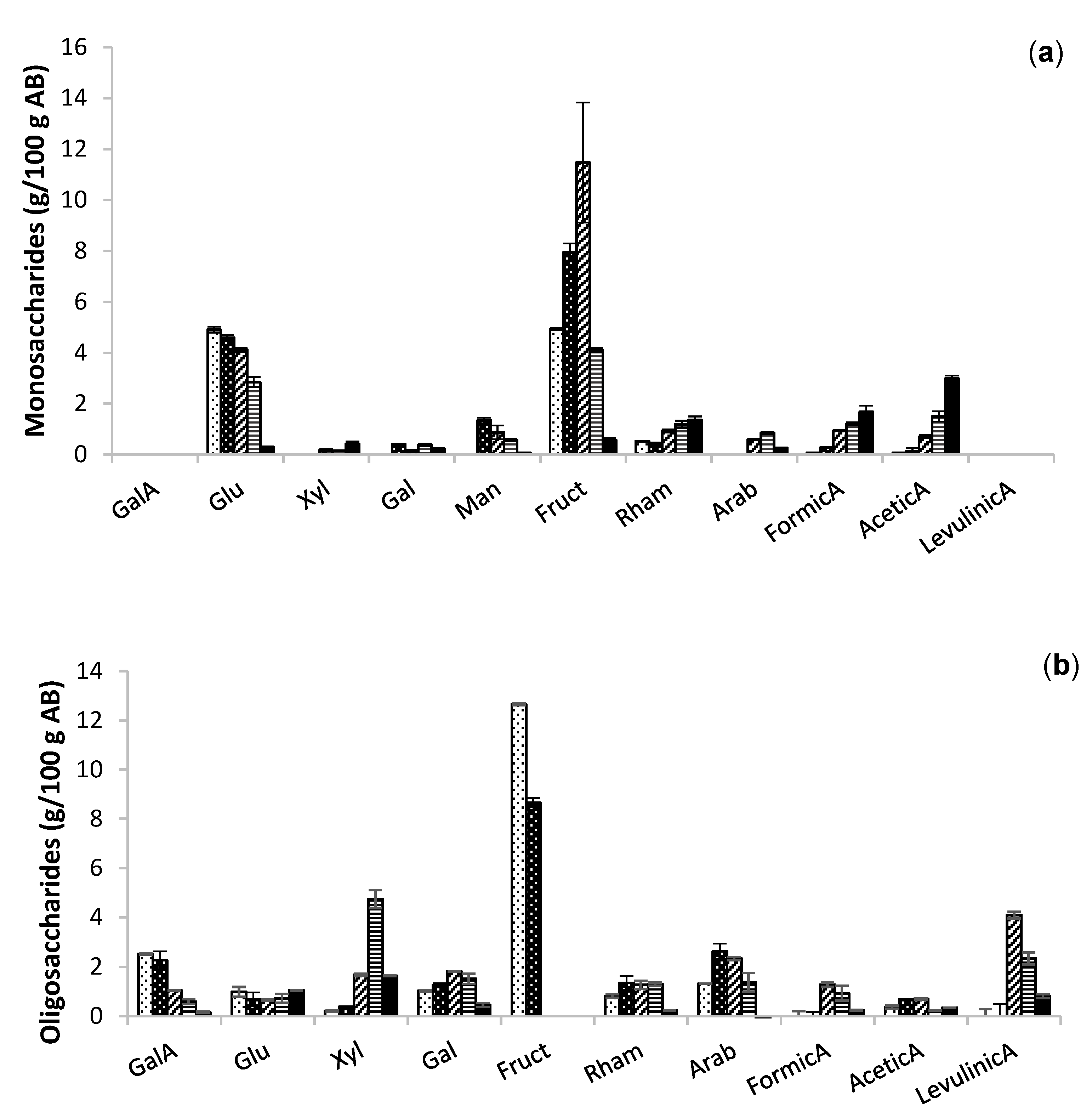
2.2. Extraction of Bioactive Fractions
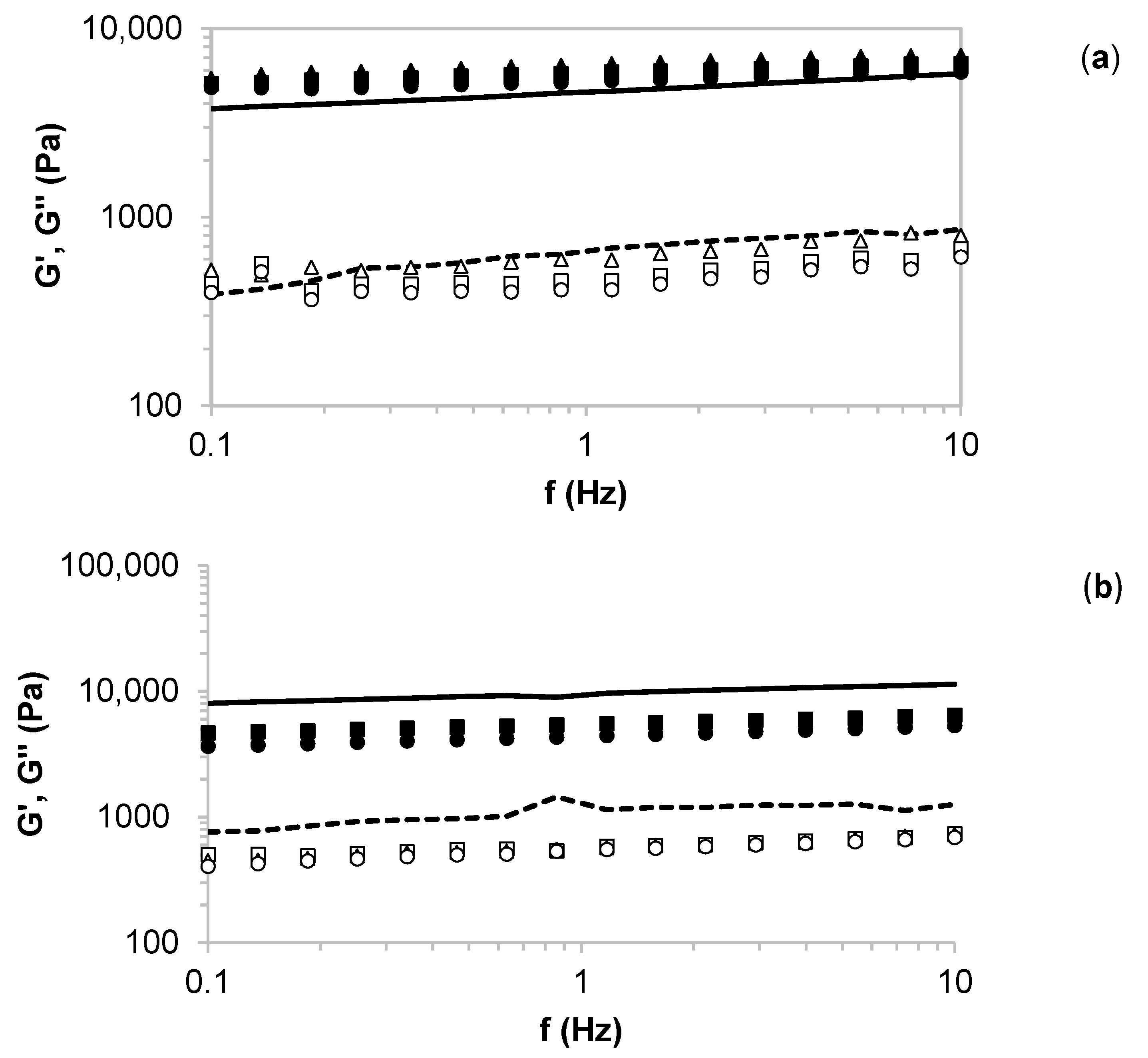
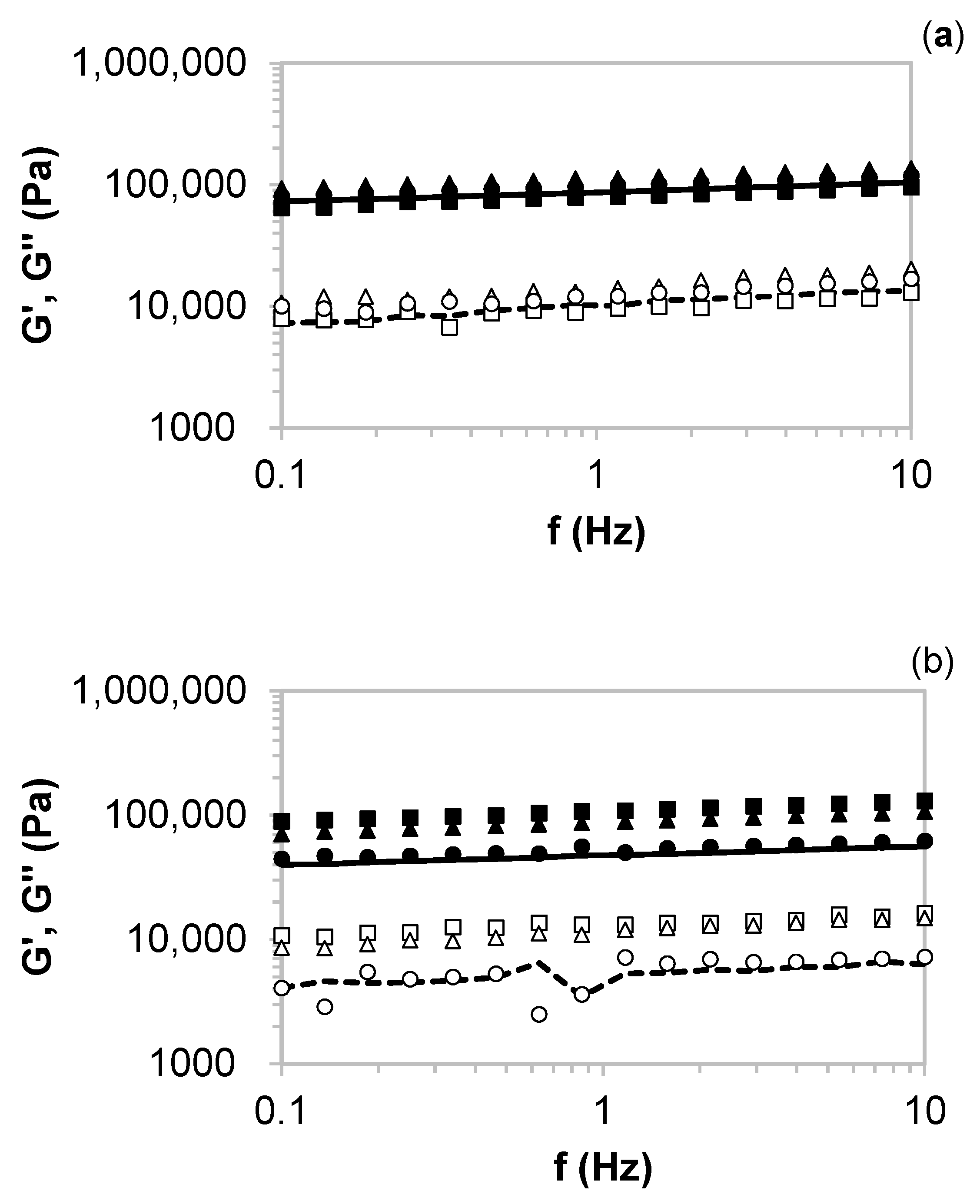
2.3. Rheology of the Formulated Hydrogels
3. Materials and Methods
3.1. Material
3.2. Extraction Method
3.3. Analytical Techniques
3.4. Hydrogels: Formulation and Rheology
3.5. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Galanakis, C.M. Recovery of high added-value components from food wastes: Conventional, emerging technologies and commercialized applications. Trends Food Sci. Technol. 2012, 26, 68–87. [Google Scholar] [CrossRef]
- Pandino, G.; Lombardo, S.; Mauromicale, G.; Williamson, G. Profile of polyphenols and phenolic acids in bracts and receptacles of globe artichoke (Cynara cardunculus var. scolymus) germplasm. J. Food Compos. Anal. 2011, 24, 148–153. [Google Scholar] [CrossRef]
- Sümer, E.; Senturk, G.E.; Demirel, Ö.U.; Yesilada, E. Comparative biochemical and histopathological evaluations proved that receptacle is the most effective part of Cynara scolymus against liver and kidney damages. J. Ethnopharmacol. 2020, 249, 112458. [Google Scholar] [CrossRef] [PubMed]
- Zuorro, A. Response surface methodology analysis of polyphenol recovery from artichoke waste. Am. J. Appl. Sci. 2014, 11, 1463–1471. [Google Scholar] [CrossRef][Green Version]
- Claus, T.; Maruyama, S.A.; Palombini, S.V.; Montanher, P.F.; Bonafé, E.G.; de Oliveira Santos Junior, O.; Matsushita, M.; Visentainer, J.V. Chemical characterization and use of artichoke parts for protection from oxidative stress in canola oil. LWT Food Sci. Technol. 2015, 61, 346–351. [Google Scholar] [CrossRef]
- Jiménez-moreno, N.; Cimminelli, M.J.; Volpe, F.; Ansó, R.; Esparza, I.; Mármol, I.; Rodríguez-yoldi, M.J.; Ancín-azpilicueta, C. Phenolic composition of artichoke waste and its antioxidant capacity on differentiated Caco-2 cells. Nutrients 2019, 11, 1723. [Google Scholar] [CrossRef] [PubMed]
- Mena-García, A.; Rodríguez-Sánchez, S.; Ruiz-Matute, A.I.; Sanz, M.L. Exploitation of artichoke byproducts to obtain bioactive extracts enriched in inositols and caffeoylquinic acids by Microwave Assisted Extraction. J. Chromatogr. A 2020, 1613, 460703. [Google Scholar] [CrossRef] [PubMed]
- Lavecchia, R.; Maffei, G.; Paccassoni, F.; Piga, L.; Zuorro, A. Artichoke waste as a source of phenolic antioxidants and bioenergy. Waste Biomass Valoriz. 2019, 10, 2975–2984. [Google Scholar] [CrossRef]
- Maietta, M.; Colombo, R.; Lavecchia, R.; Sorrenti, M.; Zuorro, A.; Papetti, A. Artichoke (Cynara cardunculus L. var. scolymus) waste as a natural source of carbonyl trapping and antiglycative agents. Food Res. Int. 2017, 100, 780–790. [Google Scholar] [CrossRef]
- Pagano, I.; Piccinelli, A.L.; Celano, R.; Campone, L.; Gazzerro, P.; Russo, M.; Rastrelli, L. Pressurized hot water extraction of bioactive compounds from artichoke by-products. Electrophoresis 2018, 39, 1899–1907. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Cano, D.; Pérez-Llamas, F.; Frutos, M.J.; Arnao, M.B.; Espinosa, C.; López-Jiménez, J.Á.; Castillo, J.; Zamora, S. Chemical and functional properties of the different by-products of artichoke (Cynara scolymus L.) from industrial canning processing. Food Chem. 2014, 160, 134–140. [Google Scholar] [CrossRef]
- Punzi, R.; Paradiso, A.; Fasciano, C.; Trani, A.; Faccia, M.; De Pinto, M.C.; Gambacorta, G. Phenols and antioxidant activity in vitro and in vivo of aqueous extracts obtained by ultrasound-assisted extraction from artichoke by-products. Nat. Prod. Commun. 2014, 9, 1315–1318. [Google Scholar] [CrossRef]
- Zeaiter, Z.; Regonesi, M.E.; Cavini, S.; Labra, M.; Sello, G.; Di Gennaro, P. BioMed Research International; Hindawi Limited: London, UK, 2019; pp. 1–8. [Google Scholar]
- Ruiz-Aceituno, L.; García-Sarrió, M.J.; Alonso-Rodriguez, B.; Ramos, L.; Sanz, M.L. Extraction of bioactive carbohydrates from artichoke (Cynara scolymus L.) external bracts using microwave assisted extraction and pressurized liquid extraction. Food Chem. 2016, 196, 1156–1162. [Google Scholar] [CrossRef] [PubMed]
- Benito-Román, Ó.; Blanco, B.; Sanz, M.T.; Beltrán, S. Subcritical water extraction of phenolic compounds from onion skin wastes (Allium cepa cv. Horcal): Effect of temperature and solvent properties. Antioxidants 2020, 9, 1233. [Google Scholar] [CrossRef]
- Ameer, K.; Shahbaz, H.M.; Kwon, J.H. Green Extraction Methods for Polyphenols from Plant Matrices and Their Byproducts: A Review. Compr. Rev. Food Sci. Food Saf. 2017, 16, 295–315. [Google Scholar] [CrossRef] [PubMed]
- Llompart, M.; Garcia-Jares, C.; Celeiro, M.; Dagnac, T. Extraction|Microwave-Assisted Extraction. In Encyclopedia of Analytical Science; Elsevier: Amsterdam, The Netherlands, 2019; pp. 67–77. ISBN 9780081019832. [Google Scholar]
- De Araújo, F.F.; de Paulo Farias, D.; Neri-Numa, I.A.; Pastore, G.M. Polyphenols and their applications: An approach in food chemistry and innovation potential. Food Chem. 2021, 338, 127535. [Google Scholar] [CrossRef] [PubMed]
- Kong, R.; Wang, J.; Cheng, M.; Lu, W.; Chen, M.; Zhang, R.; Wang, X. Development and characterization of corn starch/PVA active films incorporated with carvacrol nanoemulsions. Int. J. Biol. Macromol. 2020, 164, 1631–1639. [Google Scholar] [CrossRef]
- Feng, M.; Yu, L.; Zhu, P.; Zhou, X.; Liu, H.; Yang, Y.; Zhou, J.; Gao, C.; Bao, X.; Chen, P. Development and preparation of active starch films carrying tea polyphenol. Carbohydr. Polym. 2018, 196, 162–167. [Google Scholar] [CrossRef] [PubMed]
- Hawryl, A.; Hawryl, M.; Litwińczuk, W.; Bogucka-Kocka, A. Thin-layer chromatographic fingerprint of selected Paulownia species with chemometrics and antioxidant activity. J. Liq. Chromatogr. Relat. Technol. 2020, 43, 367–374. [Google Scholar] [CrossRef]
- Eldin Awad, O.M.; El-Sohaimy, S.A.; Ghareeb, D.A.; Aboulenein, A.M.; Saleh, S.R.; Abd El-Aziz, N.M. Phytochemical analysis and toxicity assessment of artichoke by-product extract. Pak. J. Biol. Sci. 2020, 23, 81–91. [Google Scholar] [CrossRef]
- Lutz, M.; Henríquez, C.; Escobar, M. Chemical composition and antioxidant properties of mature and baby artichokes (Cynara scolymus L.), raw and cooked. J. Food Compos. Anal. 2011, 24, 49–54. [Google Scholar] [CrossRef]
- Biel, W.; Witkowicz, R.; Piątkowska, E.; Podsiadło, C. Proximate composition, minerals and antioxidant activity of Artichoke leaf extracts. Biol. Trace Elem. Res. 2020, 194, 589–595. [Google Scholar] [CrossRef] [PubMed]
- Pesce, G.R.; Fernandes, M.C.; Mauromicale, G. Globe artichoke crop residues and their potential for bioethanol production by dilute acid hydrolysis. Biomass Bioenergy 2020, 134, 105471. [Google Scholar] [CrossRef]
- Chaves, J.O.; de Souza, M.C.; da Silva, L.C.; Lachos-Perez, D.; Torres-Mayanga, P.C.; da Fonseca Machado, A.P.; Forster-Carneiro, T.; Vázquez-Espinosa, M.; González-de-Peredo, A.V.; Barbero, G.F.; et al. Extraction of flavonoids from natural sources using modern techniques. Front. Chem. 2020, 8, 507887. [Google Scholar] [CrossRef]
- López-Hortas, L.; Conde, E.; Falqué, E.; Domínguez, H.; Torres, M.D. Preparation of hydrogels composed of bioactive compounds from aqueous phase of artichoke obtained by MHG technique. Food Bioprocess. Technol. 2019, 12, 1304–1315. [Google Scholar] [CrossRef]
- Nooeaid, P.; Chuysinuan, P.; Techasakul, S. Alginate/gelatine hydrogels: Characterisation and application of antioxidant release. Green Mater. 2017, 5, 153–164. [Google Scholar] [CrossRef]
- Lefatle, M.C.; John, M.J. Mechanical, rheological and viscoelastic properties of polysaccharide and protein based aerogels. In RSC Green Chemistry; The Royal Society of Chemistry: London, UK, 2018; Volume 2018, pp. 177–200. ISBN 9781782624127. [Google Scholar]
- Moreira, R.; Chenlo, F.; Torres, M.D.; Rama, B. Fine particle size chestnut flour doughs rheology: Influence of additives. J. Food Eng. 2014, 120, 94–99. [Google Scholar] [CrossRef]
- Alvarez, M.D.; Canet, W. Dynamic viscoelastic behavior of vegetable-based infant purees. J. Texture Stud. 2013, 44, 205–224. [Google Scholar] [CrossRef]
- Jiménez, A.; Fabra, M.J.; Talens, P.; Chiralt, A. Edible and biodegradable starch films: A Review. Food Bioprocess. Technol. 2012, 5, 2058–2076. [Google Scholar] [CrossRef]
- BeMiller, J.; Whistler, R. Starch, Chemistry and Technology, 3rd ed.; Elsevier: New York, NY, USA, 2009; ISBN 9780127462752. [Google Scholar]
- Charrondière, U.R.; Rittenschober, D.; Nowak, V.; Wijesinha-Bettoni, R.; Stadlmayr, B.; Haytowitz, D.; Persijn, D. FAO/INFOODS Guidelines for Converting Units, Denominators and Expressions, Version 1.0; The Food and Agriculture Organization: Rome, Italy, 2012; ISBN 9789251073780. [Google Scholar]
- Singleton, V.L.; Rossi, J.A. Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Am. J. Enol. Vitic. 1965, 16, 144–158. [Google Scholar]
- Von Gadow, A.; Joubert, E.; Hansmann, C.F. Comparison of the antioxidant activity of rooibos tea (Aspalathus linearis) with green, oolong and black tea. Food Chem. 1997, 60, 73–77. [Google Scholar] [CrossRef]
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic. Biol. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef]
- Torres, M.D.; Fradinho, P.; Rodríguez, P.; Falqué, E.; Santos, V.; Domínguez, H. Biorefinery concept for discarded potatoes: Recovery of starch and bioactive compounds. J. Food Eng. 2020, 275, 109886. [Google Scholar] [CrossRef]
- Rodríguez-Seoane, P.; del Pozo, C.; Puy, N.; Bartrolí, J.; Domínguez, H. Hydrothermal extraction of valuable components from leaves and petioles from Paulownia elongata × fortunei. Waste Biomass Valoriz. 2021, 12, 4525–4535. [Google Scholar] [CrossRef]
- Torres, M.D.; Chenlo, F.; Moreira, R. Rheological effect of gelatinisation using different temperature-time conditions on potato starch dispersions: Mechanical characterisation of the obtained gels. Food Bioprocess. Technol. 2018, 11, 132–140. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Órbenes, G.; Rodríguez-Seoane, P.; Torres, M.D.; Chamy, R.; Zúñiga, M.E.; Domínguez, H. Valorization of Artichoke Industrial By-Products Using Green Extraction Technologies: Formulation of Hydrogels in Combination with Paulownia Extracts. Molecules 2021, 26, 4386. https://doi.org/10.3390/molecules26144386
Órbenes G, Rodríguez-Seoane P, Torres MD, Chamy R, Zúñiga ME, Domínguez H. Valorization of Artichoke Industrial By-Products Using Green Extraction Technologies: Formulation of Hydrogels in Combination with Paulownia Extracts. Molecules. 2021; 26(14):4386. https://doi.org/10.3390/molecules26144386
Chicago/Turabian StyleÓrbenes, Gabriela, Paula Rodríguez-Seoane, María Dolores Torres, Rolando Chamy, María Elvira Zúñiga, and Herminia Domínguez. 2021. "Valorization of Artichoke Industrial By-Products Using Green Extraction Technologies: Formulation of Hydrogels in Combination with Paulownia Extracts" Molecules 26, no. 14: 4386. https://doi.org/10.3390/molecules26144386
APA StyleÓrbenes, G., Rodríguez-Seoane, P., Torres, M. D., Chamy, R., Zúñiga, M. E., & Domínguez, H. (2021). Valorization of Artichoke Industrial By-Products Using Green Extraction Technologies: Formulation of Hydrogels in Combination with Paulownia Extracts. Molecules, 26(14), 4386. https://doi.org/10.3390/molecules26144386







