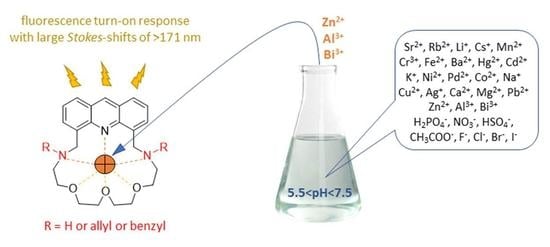
Acridino-Diaza-20-Crown-6 Ethers: New Macrocyclic Hosts for Optochemical Metal Ion Sensing
Abstract
:1. Introduction
2. Results and Discussion
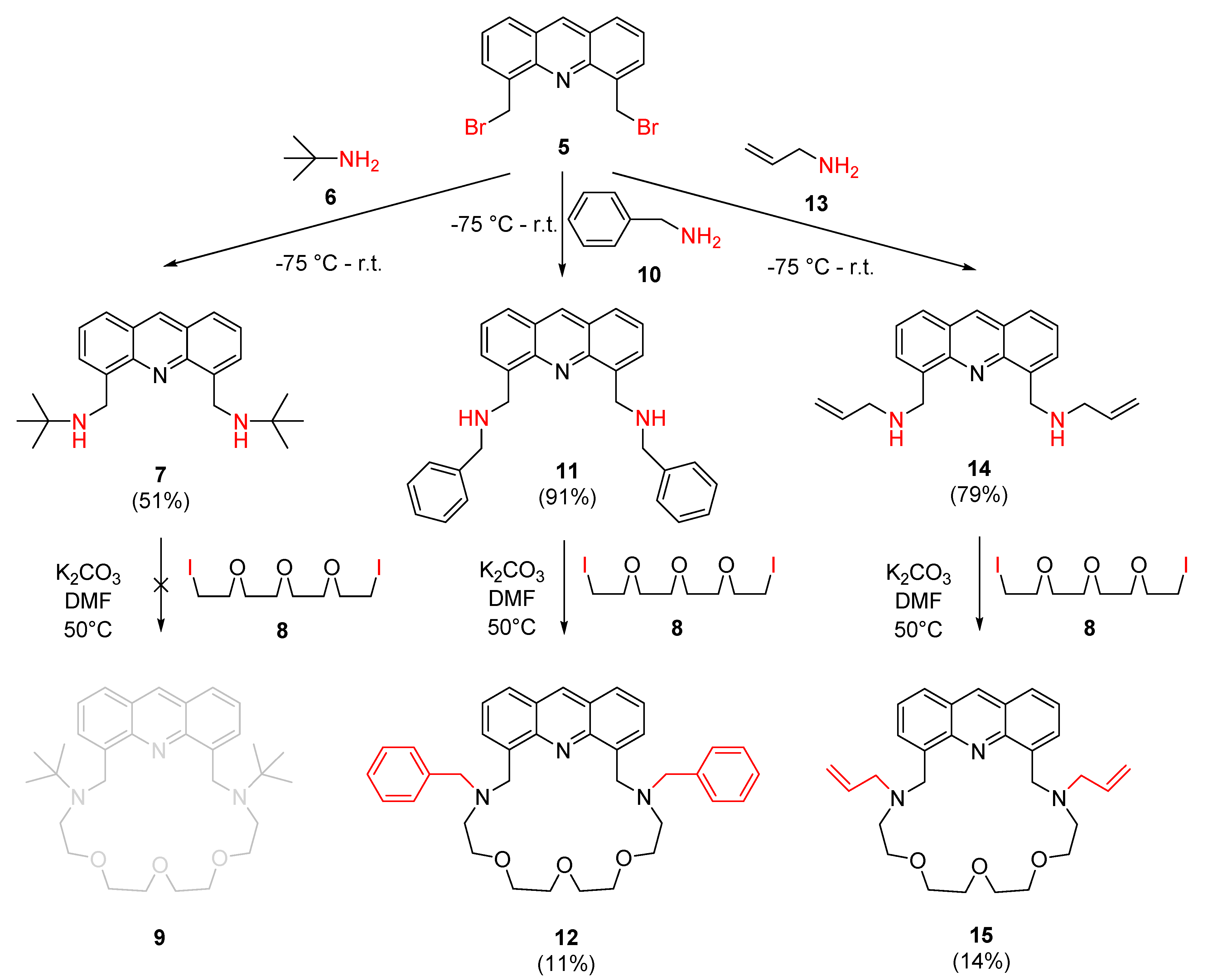
2.1. Synthesis
2.2. Spectral Properties
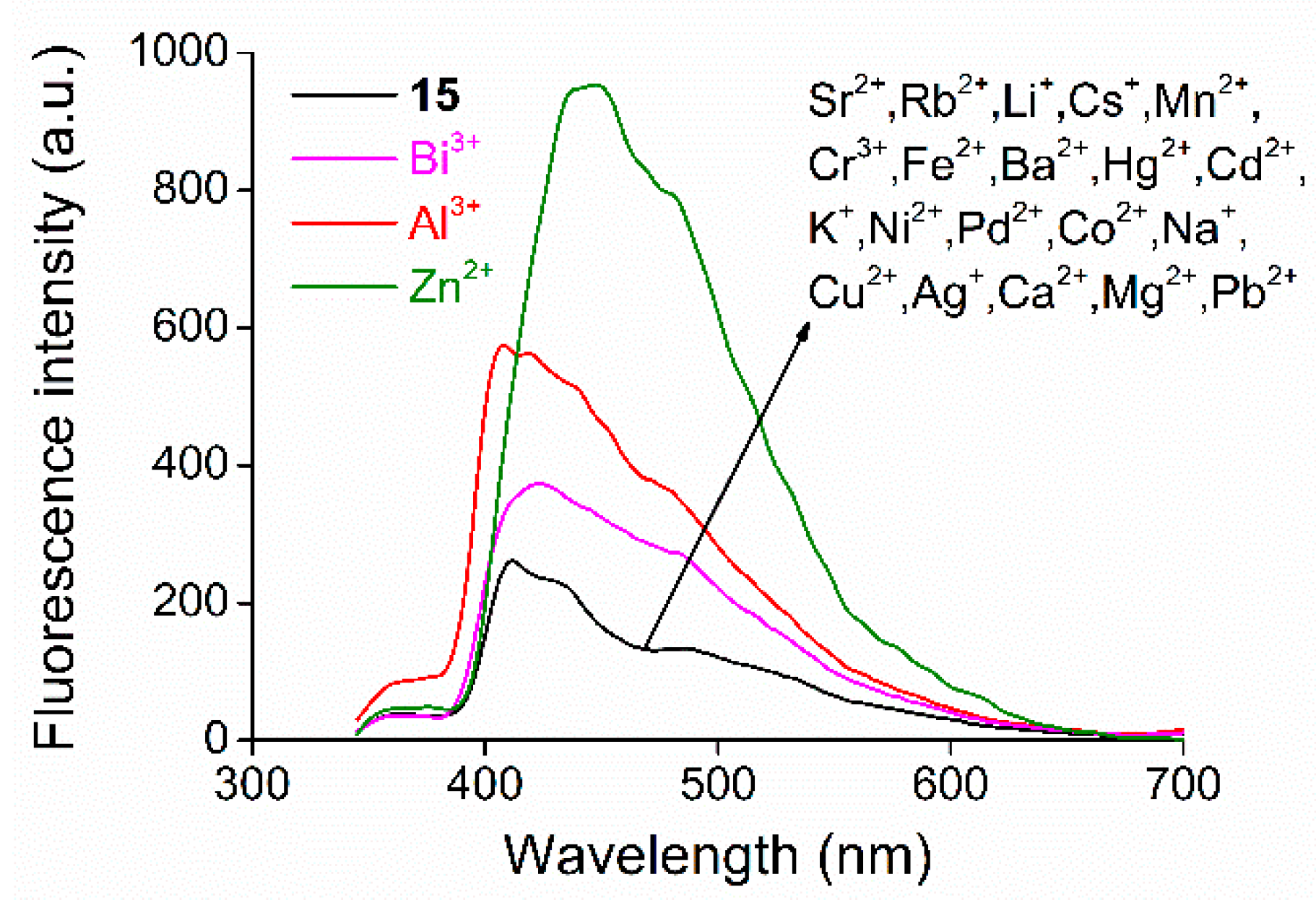
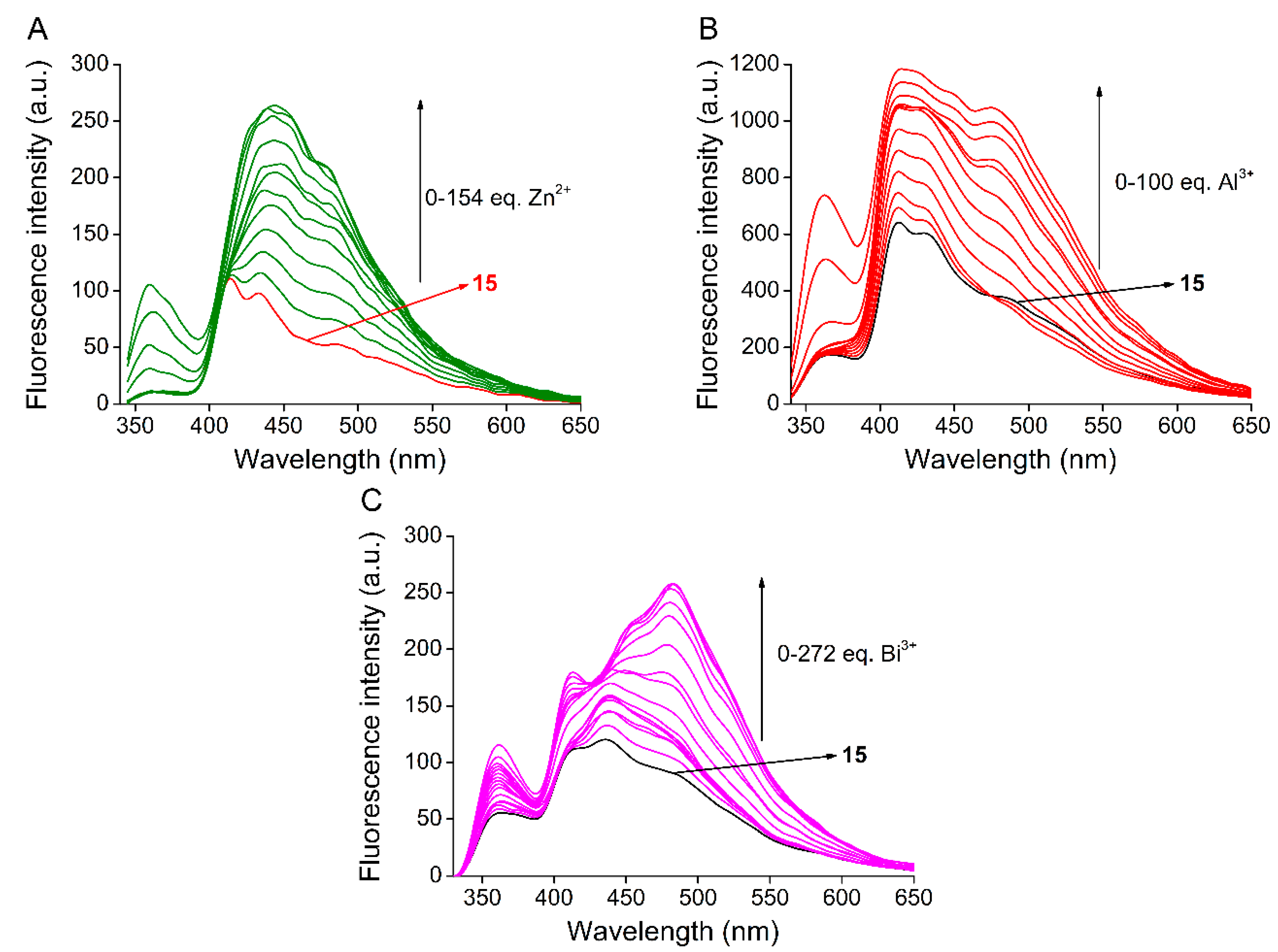
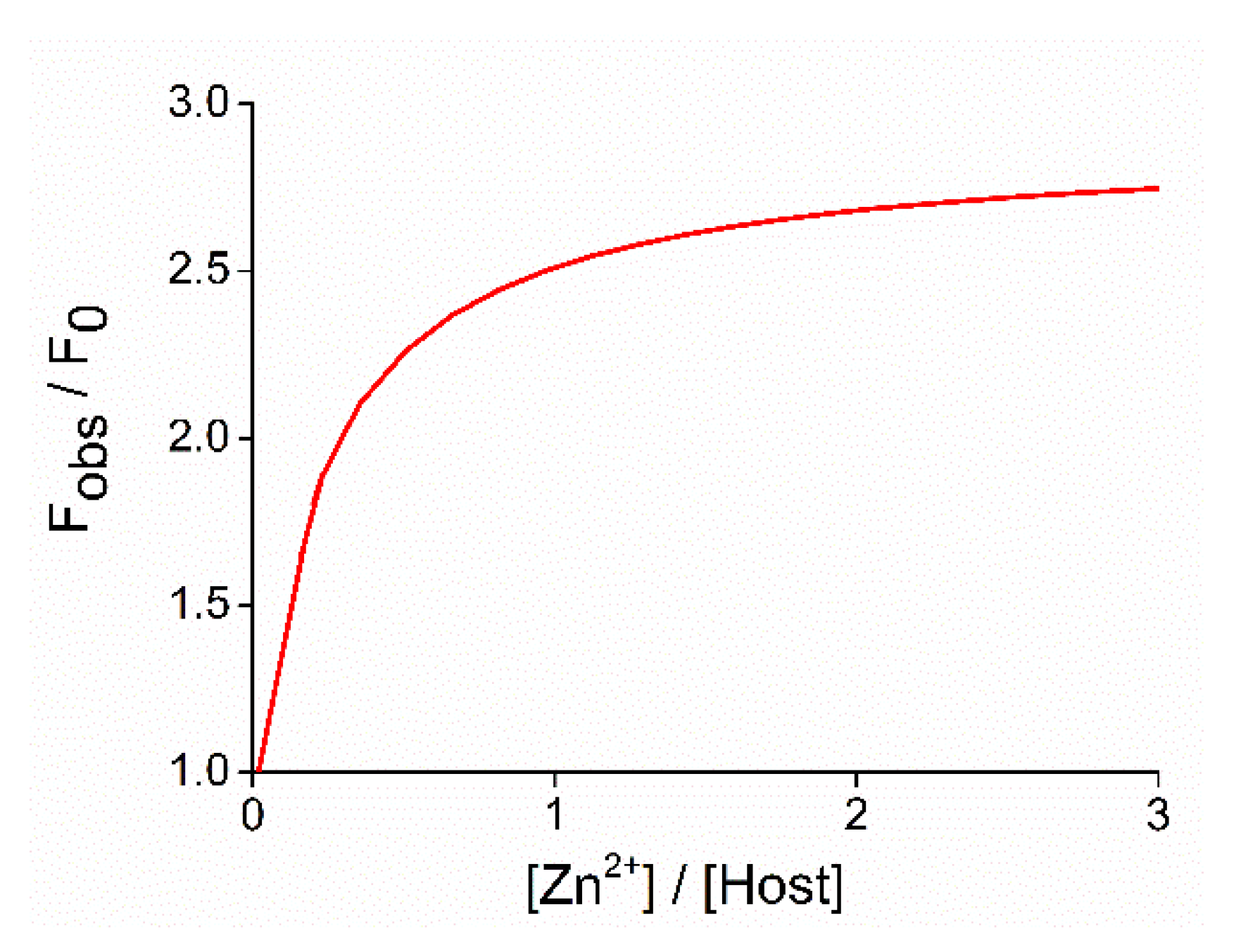
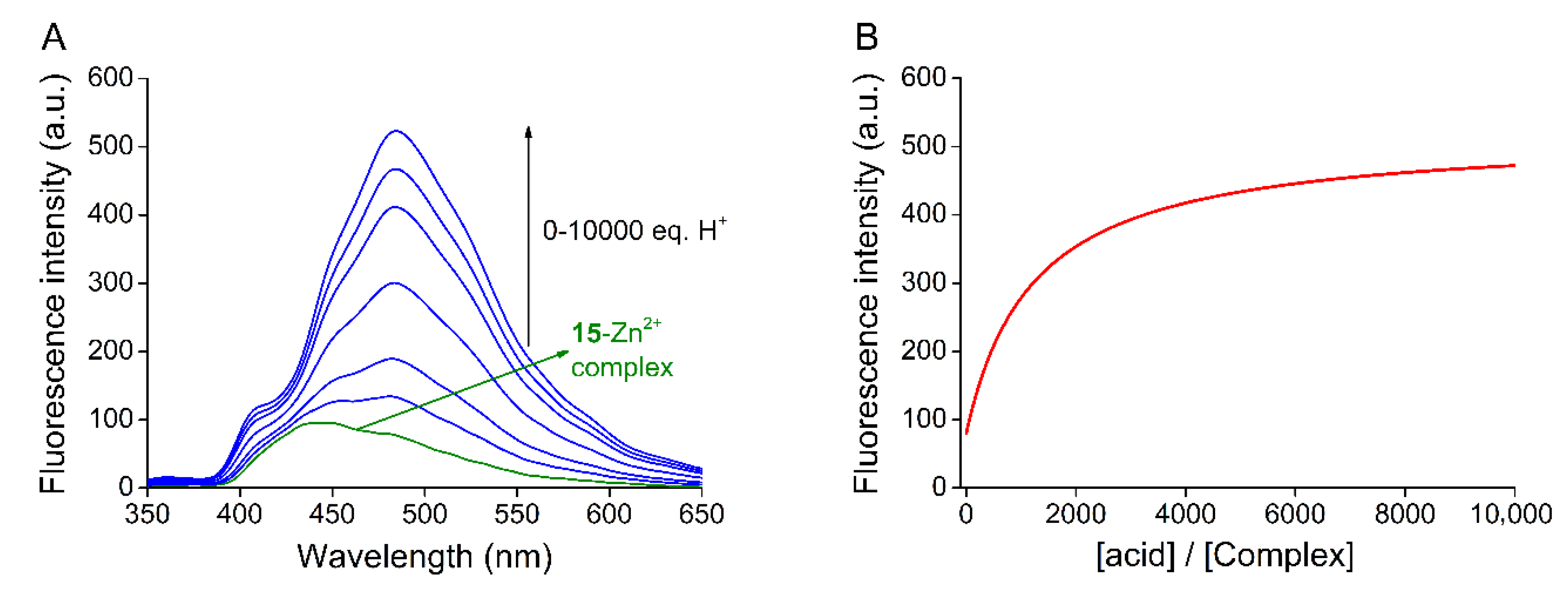
2.3. Studies on Metal Ion Complexation
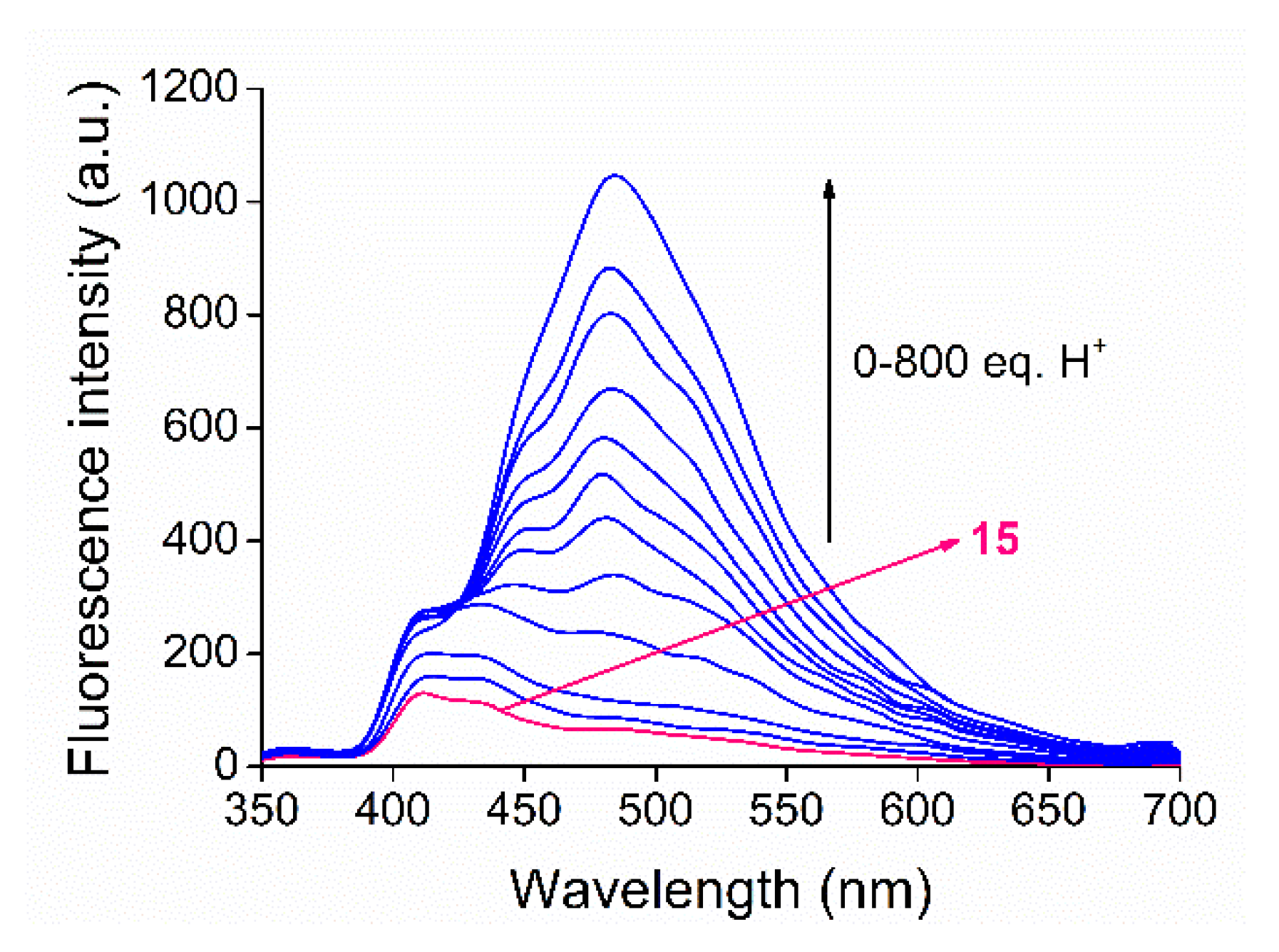
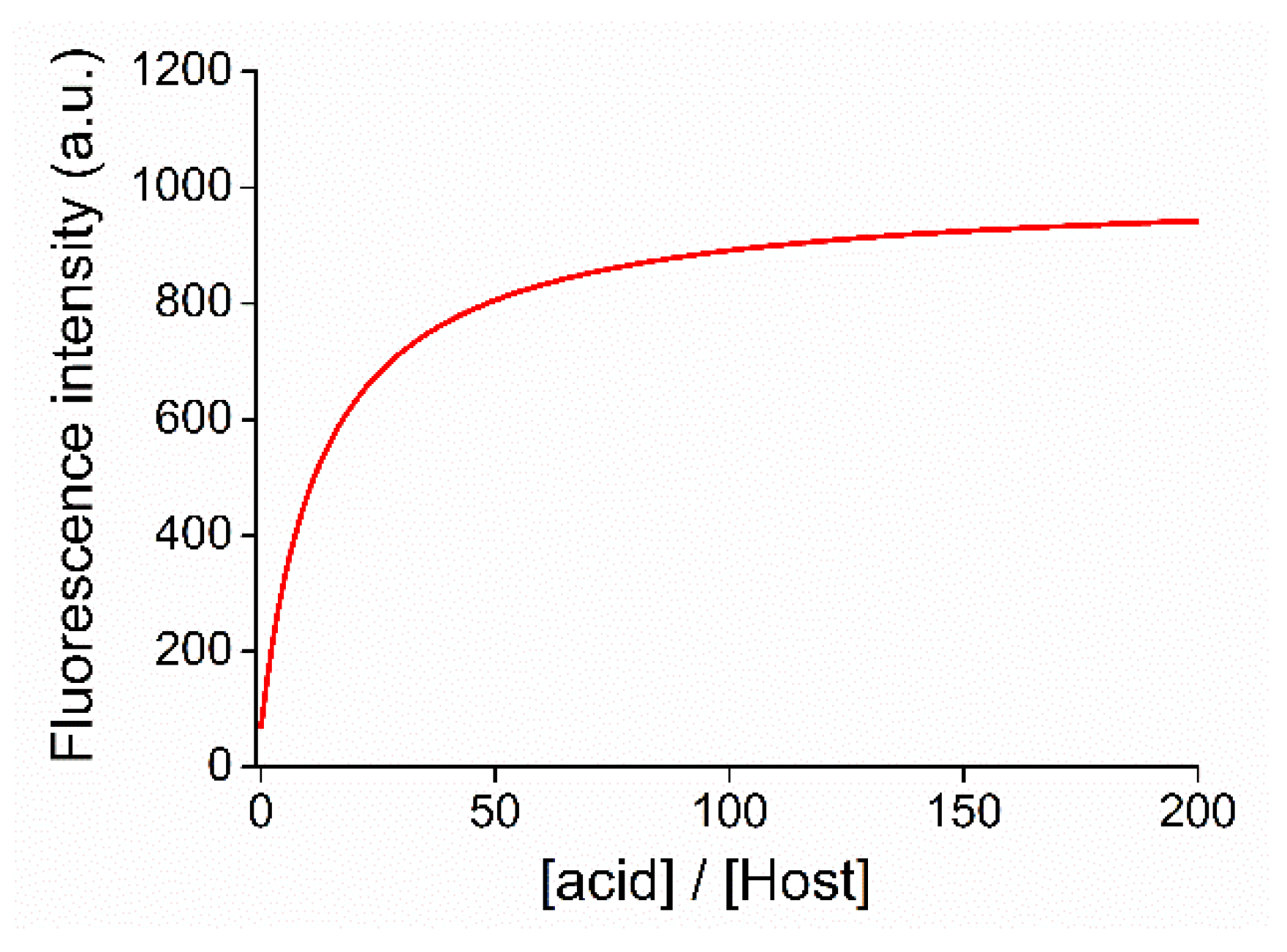
2.4. Acid–Base Properties
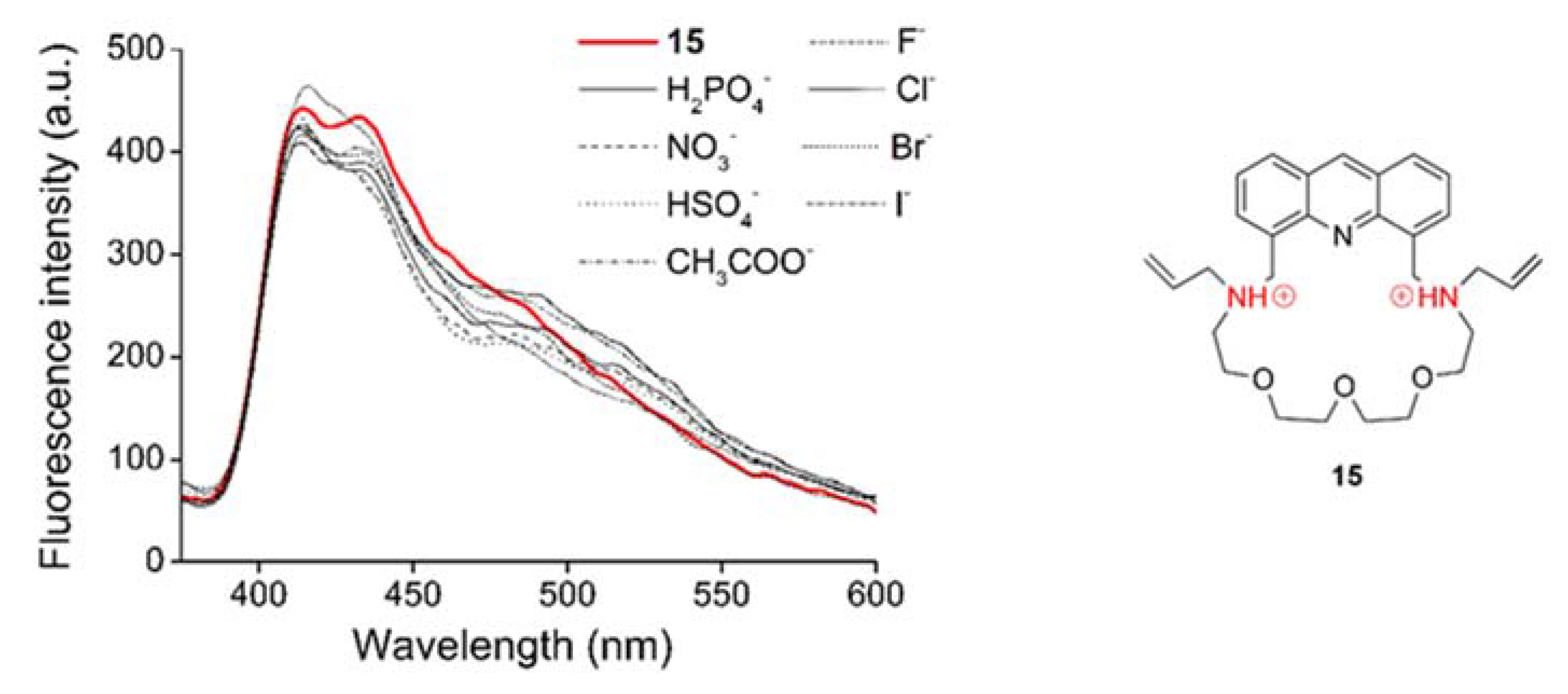
2.5. Coordination Ability toward Anions
3. Conclusions
4. Experimental Methods
4.1. Chemicals, Apparatus and Measurements
4.2. Evaluation of the Results
4.3. Synthesis of the New Compounds
4.3.1. Optimized Synthesis of 4,5-Bis(bromomethyl)acridine (5)
4.3.2. Preparation of N,N′-(Acridine-4,5-diylbis(methylene))bis(2-methylpropan-2-amine) (7)
4.3.3. Preparation of 1,1′-(Acridine-4,5-diyl)bis(N-benzylmethanamine) (11)
4.3.4. Preparation of N,N′-(Acridine-4,5-diylbis(methylene))bis(prop-2-en-1-amine) (14)
4.3.5. Preparation of 7,19-Dibenzyl-10,13,16-trioxa-7,19,27-triazatetracyclo [23.3.1.0⁵,2⁸.021,2⁶]nonacosa-1,3,5(28),21,23,25(29),26-heptaene (12)
4.3.6. Preparation of 7,19-Bis(prop-2-en-1-yl)-10,13,16-trioxa-7,19,27-triazatetracyclo[2 3.3.1.0⁵,2⁸.021,2⁶]nonacosa-1,3,5(28),21,23,25(29),26-heptaene (15)
4.3.7. Preparation of 10,13,16-Trioxa-7,19,27-triazatetracyclo[2 3.3.1.0⁵,2⁸.021,2⁶]nonacosa-1,3,5(28),21,23,25(29),26-heptaene (17) by the debenzylation of (12)
4.3.8. Preparation of 10,13,16-Trioxa-7,19,27-triazatetracyclo[2 3.3.1.0⁵,2⁸.021,2⁶]nonacosa-1,3,5(28),21,23,25(29),26-heptaene (17) starting from (15)
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Valeur, B.; Berberan-Santos, M.N. Molecular Fluorescence: Principles and Applications, 2nd ed.; Wiley VCH: Weinheim, Germany, 2012; pp. 20–374. ISBN 978-3-527-32837-6. [Google Scholar]
- Pál, D.; Baranyai, P.; Leveles, I.; Vértessy, B.G.; Huszthy, P.; Móczár, I. An acridone-based fluorescent chemosensor for cationic and anionic species, and its application for molecular logic operations. ChemistrySelect 2019, 4, 11936–11943. [Google Scholar] [CrossRef] [Green Version]
- dos Santos Carlos, F.; da Silva, L.A.; Zanlorenzi, C.; Nunes, F.S. A novel macrocycle acridine-based fluorescent chemosensor for selective detection of Cd2+ in Brazilian sugarcane spirit and tobacco cigarette smoke extract. Inorganica Chim. Acta 2020, 508, 119634–119641. [Google Scholar] [CrossRef]
- Visscher, A.; Bachmann, S.; Schnegelsberg, C.; Teuteberg, T.; Mata, R.A.; Stalke, D. Highly selective and sensitive fluorescence detection of Zn2+ and Cd2+ ions by using an acridine sensor. Dalton Trans. 2016, 45, 5689–5699. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, C.; Fu, J.; Yao, K.; Xue, K.; Xu, K.; Pang, X. Acridine-based fluorescence chemosensors for selective sensing of Fe3+ and Ni2+ ions. Spectrochim. Acta A 2018, 199, 403–411. [Google Scholar] [CrossRef]
- Zhou, J.; Yuan, Y.F.; Zhuo, J.B.; Lin, C.X. Synthesis and characterization of cyclophane: The highly selective recognition of Fe3+ in aqueous solution and H2PO4− in acetonitrile solution. Tetrahedron Lett. 2018, 59, 1059–1064. [Google Scholar] [CrossRef]
- Lee, H.N.; Kim, H.N.; Swamy, K.M.K.; Park, M.S.; Kim, J.; Lee, H.; Park, S.; Yoon, J. New acridine derivatives bearing immobilized azacrown or azathiacrown ligand as fluorescent chemosensors for Hg2+ and Cd2+. Tetrahedron Lett. 2008, 49, 1261–1265. [Google Scholar] [CrossRef]
- Wang, Y.; Hu, X.; Wang, L.; Shang, Z.; Chao, J.; Jin, W. A new acridine derivative as a highly selective ‘off–on’ fluorescence chemosensor for Cd2+ in aqueous media. Sens. Actuators B Chem. 2011, 156, 126–131. [Google Scholar] [CrossRef]
- Dai, Y.; Xu, K.; Li, Q.; Wang, C.; Liu, X.; Wang, P. Acridine-based complex as amino acid anion fluorescent sensor in aqueous solution. Spectrochim. Acta A 2016, 157, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Xu, K.; Song, P.; Dai, Y.; Yang, L.; Pang, X. Novel enantioselective fluorescent sensors for malate anion based on acridine. Dyes Pigm. 2014, 109, 169–174. [Google Scholar] [CrossRef]
- Yousuf, M.; Ahmed, N.; Shirinfar, B.; Miriyala, V.M.; Youn, I.S.; Kim, K.S. Precise tuning of cationic cyclophanes toward highly selective fluorogenic recognition of specific biophosphate anions. Org. Lett. 2014, 16, 2150–2153. [Google Scholar] [CrossRef]
- Bartoli, F.; Bencini, A.; Garau, A.; Giorgi, C.; Lippolis, V.; Lunghi, A.; Totti, F.; Valtancoli, B. Di- and triphosphate recognition and sensing with mono- and dinuclear fluorescent zinc(II) complexes: Clues for the design of selective chemosensors for anions in aqueous media. Chem. Eur. J. 2016, 22, 14890–14901. [Google Scholar] [CrossRef] [PubMed]
- Martí-Centelles, V.; Burguete, M.I.; Galindo, F.; Izquierdo, M.A.; Kumar, D.K.; White, A.J.; Luis, S.V.; Vilar, R. Fluorescent acridine-based receptors for H2PO4–. J. Org. Chem. 2012, 77, 490–500. [Google Scholar] [CrossRef]
- Chelli, R.; Pietraperzia, G.; Bencini, A.; Giorgi, C.; Lippolis, V.; Salvi, P.R.; Gellini, C. A fluorescent receptor for halide recognition: Clues for the design of anion chemosensors. Phys. Chem. Chem. Phys. 2015, 17, 10813–10822. [Google Scholar] [CrossRef]
- Xu, K.X.; Kong, H.J.; Li, P.; Yang, L.; Zhang, J.L.; Wang, C.J. Acridine-based enantioselective fluorescent sensors for the malate anion in water. New J. Chem. 2014, 38, 1004–1010. [Google Scholar] [CrossRef]
- Wang, C.; Wang, P.; Liu, X.; Fu, J.; Xue, K.; Xu, K. Novel enantioselective fluorescent sensors for tartrate anion based on acridinezswsxa. Luminescence 2017, 32, 1313–1318. [Google Scholar] [CrossRef] [PubMed]
- Martí-Centelles, V.; Izquierdo, M.A.; Burguete, M.I.; Galindo, F.; Luis, S.V. Recognition of free tryptophan in water by synthetic pseudopeptides: Fluorescence and thermodynamic studies. Chem. Eur. J. 2014, 20, 7465–7478. [Google Scholar] [CrossRef] [PubMed]
- Do-Thanh, C.L.; Rowland, M.M.; Best, M.D. Fluorescent bis-cyclen tweezer receptors for inositol (1,4,5)-trisphosphate. Tetrahedron 2011, 67, 3803–3808. [Google Scholar] [CrossRef]
- Read, S.A.; Obeid, S.; Ahlenstiel, C.; Ahlenstiel, G. The role of zinc in antiviral immunity. Adv. Nutr. 2019, 10, 696–710. [Google Scholar] [CrossRef] [Green Version]
- Yan, X.; Kim, J.J.; Jeong, H.S.; Moon, Y.K.; Cho, Y.K.; Ahn, S.; Jun, S.B.; Kim, H.; You, Y. Low-affinity zinc sensor showing fluorescence responses with minimal artifacts. Inorg. Chem. 2017, 56, 4332–4346. [Google Scholar] [CrossRef]
- Xia, S.; Shen, J.; Wang, J.; Wang, H.; Fang, M.; Zhou, H.; Tanasova, M. Ratiometric fluorescent and colorimetric BODIPY-based sensor for zinc ions in solution and living cells. Sens. Actuators B Chem. 2018, 258, 1279–1286. [Google Scholar] [CrossRef]
- Jang, H.J.; Kang, J.H.; Lee, M.; Lim, M.H.; Kim, C. Fluorescent sensor for sequentially monitoring zinc(II) and cyanide anion in near-perfect aqueous media. Ind. Eng. Chem. Res. 2018, 57, 54–62. [Google Scholar] [CrossRef]
- Liu, H.; Dong, Y.; Zhang, B.; Liu, F.; Tan, C.; Tan, Y.; Jiang, Y. An efficient quinoline-based fluorescence sensor for zinc(II) and its application in live-cell imaging. Sens. Actuators B Chem. 2016, 234, 616–624. [Google Scholar] [CrossRef]
- Liu, X.; Wang, P.; Fu, J.; Yao, K.; Xue, K.; Xu, K. Turn-on fluorescent sensor for zinc and cadmium ions based on quinolone and its sequential response to phosphate. J. Lumin. 2017, 186, 16–22. [Google Scholar] [CrossRef]
- Lin, N.; Zhang, Q.; Xia, X.; Liang, M.; Zhang, S.; Zheng, L.; Cao, Q.; Ding, Z. A highly zinc-selective ratiometric fluorescent probe based on AIE luminogen functionalized coordination polymer nanoparticles. RSC Adv. 2017, 7, 21446–21451. [Google Scholar] [CrossRef] [Green Version]
- Mandal, J.; Ghorai, P.; Pal, K.; Karmakar, P.; Saha, A. 2-Hydroxy-5-methylisophthalaldehyde based fluorescent-colorimetric chemosensor for dual detection of Zn2+ and Cu2+ with high sensitivity and application in live cell imaging. J. Lumin. 2019, 205, 14–22. [Google Scholar] [CrossRef]
- World Health Organization. Zinc in Drinking-Water. Background Document for Preparation of WHO Guidelines for Drinking-Water Quality; World Health Organization: Geneva, Switzerland, 2003; Available online: https://www.who.int/water_sanitation_health/dwq/chemicals/zinc.pdf (accessed on 10 May 2021).
- Nguyen, H.L.; Kumar, N.; Audibert, J.F.; Ghasemi, R.; Lefevre, J.P.; Ha-Thi, M.H.; Mongin, C.; Leray, I. Water-soluble aluminium fluorescent sensor based on aggregation-induced emission enhancement. New J. Chem. 2019, 43, 15302–15310. [Google Scholar] [CrossRef]
- Liu, Z.; Ren, K.; Chen, Y.; Li, Y.; Xie, J.; Ding, Y.; Li, L.; Li, K.; Zhu, W.; Yang, W.; et al. A supramolecular self-assembly host-guest system from cyclodextrin as an absolute water-soluble fluorescence sensor for aluminium ions: Synthesis, characterization and sensing activity. RSC Adv. 2017, 7, 38160–38165. [Google Scholar] [CrossRef] [Green Version]
- In, B.; Hwang, G.W.; Lee, K.H. Highly sensitive and selective detection of Al(III) ions in aqueous buffered solution with fluorescent peptide-based sensor. Bioorg. Med. Chem. Lett. 2016, 26, 4477–4482. [Google Scholar] [CrossRef] [PubMed]
- Balagurusamy, B.; Ilayaperumal, P.; Zorlu, Y.; Chellaiah, R. Selective turn-on aluminium ions detection of NBD(+) appended Schiff-base fluorophore. ChemistrySelect 2020, 5, 8086–8092. [Google Scholar] [CrossRef]
- Wang, J.H.; Gao, D.R.; Wang, X.Y.; Lu, Y.M.; Shen, W.X.; Lv, Y.Y. Near-infrared fluorescent probe for ratiometric Al3+ signaling and imaging through combined PET and ICT mechanisms. Sens. Actuators B Chem. 2019, 294, 14–23. [Google Scholar] [CrossRef]
- Balamurugan, G.; Velmathi, S.; Thirumalaivasan, N.; Wu, S.P. New phenazine based AIE probes for selective detection of aluminium(III) ions in presence of other trivalent metal ions in living cells. Analyst 2017, 142, 4721–4726. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Jin, L.; Kuang, Y.; Yuan, Z.; Wang, Q. Isonicotinoylhydrazide modified 3-acetylcoumarin scaffold as an efficient chemical reversible sensor to detect Al3+ selectively and its application in live cells imaging. Synth. Commun. 2019, 49, 2501–2511. [Google Scholar] [CrossRef]
- Saravanan, A.; Shyamsivappan, S.; Suresh, T.; Subashini, G.; Kadirvelu, K.; Bhuvanesh, N.; Nandhakumar, R.; Mohan, P.S. An efficient new dual fluorescent pyrene based chemosensor for the detection of bismuth(III) and aluminium(III) ions and its applications in bio-imaging. Talanta 2019, 198, 249–256. [Google Scholar] [CrossRef] [PubMed]
- Yue, X.L.; Wang, Z.Q.; Li, C.R.; Yang, Z.Y. Naphthalene-derived Al3+-selective fluorescent chemosensor based on PET and ESIPT in aqueous solution. Tetrahedron Lett. 2017, 58, 4532–4537. [Google Scholar] [CrossRef]
- Suresh, S.; Bhuvanesh, N.; Prabhu, J.; Thamilselvan, A.; Kannan, K.; Nandhakumar, R.K.V.R. Pyrene based chalcone as a reversible fluorescent chemosensor for Al3+ ion and its biological applications. J. Photochem. Photobiol. A 2018, 359, 172–182. [Google Scholar] [CrossRef]
- Shree, G.J.; Sivaraman, G.; Siva, A.; Chellappa, D. Anthracene- and pyrene-bearing imidazoles as turn-on fluorescent chemosensor for aluminum ion in living cells. Dyes Pigm. 2019, 163, 204–212. [Google Scholar] [CrossRef]
- Zhu, G.; Huang, Y.; Wang, C.; Lu, L.; Sun, T.; Wang, M.; Tang, Y.; Shan, D.; Wen, S.; Zhu, J. A novel coumarin-based fluorescence chemosensor for Al3+ and its application in cell imaging. Spectrochim. Acta A 2019, 210, 105–110. [Google Scholar] [CrossRef]
- Yu, C.; Jian, L.; Ji, Y.; Zhang, J. Al(III)-responsive “off-on” chemosensor based on rhodamine derivative and its application in cell imaging. RSC Adv. 2018, 8, 31106–31112. [Google Scholar] [CrossRef] [Green Version]
- Inan-Eroglu, E.; Ayaz, A. Is aluminum exposure a risk factor for neurological disorders? J. Res. Med. Sci. 2018, 23, 1–8. [Google Scholar] [CrossRef]
- World Health Organization. Aluminium in Drinking-Water. Background Document for Preparation of WHO Guidelines for Drinking-Water Quality; World Health Organization: Geneva, Switzerland, 2003; Available online: https://www.who.int/water_sanitation_health/publications/aluminium/en/ (accessed on 10 May 2021).
- Environmental Protection Agency. Drinking Water Parameters. Microbiological, Chemical and Indicator Parameters in the 2014 Drinking Water Regulations 2014—An Overview of Parameters and Their Importance; Environmental Protection Agency: Wexford, Ireland, 2014. Available online: https://www.epa.ie/pubs/advice/drinkingwater/2015_04_21_ParametersStandaloneDoc.pdf (accessed on 10 May 2021).
- Sadler, P.J.; Li, H.; Sun, H. Coordination chemistry of metals in medicine: Target sites for bismuth. Coord. Chem. Rev. 1999, 185, 689–709. [Google Scholar] [CrossRef]
- Tiwari, V.K.; Shukla, S.K.; Chauhan, V.S.; Kant, R.; Rani, S.; Tewari, I.C. Antimicrobial, antitumor and gastroprotective studies of some new water soluble organic derivatives of bismuth. Int. J. Pharm. Anal. 2010, 2, 9–14. [Google Scholar]
- Lei, P.; Zhang, P.; Yao, S.; Song, S.; Dong, L.; Xu, X.; Liu, X.; Du, K.; Feng, J.; Zhang, H. Optimization of Bi3+ in upconversion nanoparticles induced simultaneous enhancement of near-infrared optical and X-ray computed tomography imaging capability. ACS Appl. Mater. Interfaces 2016, 8, 27490–27497. [Google Scholar] [CrossRef] [PubMed]
- Moyano, S.; Wuilloud, R.G.; Olsina, R.A.; Gásquez, J.A.; Martinez, L.D. On-line preconcentration system for bismuth determination in urine by flow injection hydride generation inductively coupled plasma atomic emission spectrometry. Talanta 2001, 54, 211–219. [Google Scholar] [CrossRef]
- Okamoto, Y.; Fujiwara, T.; Notake, H. Determination of bismuth in nonferrous alloys by inductively coupled plasma mass spectrometry combined with use of solid-phase methylation and electrothermal vaporization technique. Appl. Spectrosc. 2001, 55, 514–516. [Google Scholar] [CrossRef]
- Yamini, Y.; Chaloosi, M.; Ebrahimzadeh, H. Solid phase extraction and graphite furnace atomic absorption spectrometric determination of ultra trace amounts of bismuth in water samples. Talanta 2002, 56, 797–803. [Google Scholar] [CrossRef]
- Tyszczuk-Rotko, K.; Sadok, I.; Barczak, M.; Jabłońska-Czapla, M. A new voltammetric sensor based on thiol-functionalized polysiloxane film modified by lead nanoparticles for detection of Bi(III) ions. Electrochim. Acta 2016, 208, 102–108. [Google Scholar] [CrossRef]
- Yang, M.S.; Jin, J.H.; An, X.; Cui, F.L. Determination of bismuth in pharmaceutical products by resonance light scattering using quaternary ammonium salts as molecular probe. J. Anal. Chem. 2014, 69, 942–947. [Google Scholar] [CrossRef]
- El-Sewify, I.M.; Shenashen, M.A.; Shahat, A.; Yamaguchi, H.; Selim, M.M.; Khalil, M.M.; El-Safty, S.A. Dual colorimetric and fluorometric monitoring of Bi3+ ions in water using supermicroporous Zr-MOFs chemosensors. J. Lumin. 2018, 198, 438–448. [Google Scholar] [CrossRef]
- Ramasamy, K.; Thambusamy, S. Dual emission and pH based naphthalimide derivative fluorescent sensor for the detection of Bi3+. Sens. Actuators B Chem. 2017, 247, 632–640. [Google Scholar] [CrossRef]
- Zhang, E.; Ju, P.; Li, Q.; Hou, X.; Yang, H.; Yang, X.; Zou, Y.; Zhang, Y. A novel rhodamine 6G-based fluorescent and colorimetric probe for Bi3+: Synthesis, selectivity, sensitivity and potential applications. Sens. Actuators B Chem. 2018, 260, 204–212. [Google Scholar] [CrossRef]
- Zhang, D.; Dong, Z.; Jiang, X.; Feng, M.; Li, W.; Gao, G. A proof-of-concept fluorescent strategy for highly selective detection of Cr(VI) based on inner filter effect using a hydrophilic ionic chemosensor. Anal. Methods 2013, 5, 1669–1675. [Google Scholar] [CrossRef]
- Berliner, M.A.; Belecki, K. Simple, rapid procedure for the synthesis of chloromethyl methyl ether and other chloro alkyl ethers1. J. Org. Chem. 2005, 70, 9618–9621. [Google Scholar] [CrossRef]
- Jousselme, B.; Blanchard, P.; Levillain, E.; Delaunay, J.; Allain, M.; Richomme, P.; Rondeau, D.; Gallego-Planas, N.; Roncali, J. Crown-annelated oligothiophenes as model compounds for molecular actuation. J. Am. Chem. Soc. 2003, 125, 1363–1370. [Google Scholar] [CrossRef] [PubMed]
- Garro-Helion, F.; Merzouk, A.; Guibe, F. Mild and selective palladium(0)-catalyzed deallylation of allylic amines. Allylamine and diallylamine as very convenient ammonia equivalents for the synthesis of primary amines. J. Org. Chem. 1993, 58, 6109–6113. [Google Scholar] [CrossRef]
- Lakowicz, J.R. Principles of Fluorescence Spectroscopy, 3rd ed.; Springer Science+Business Media: New York, NY, USA, 2006; pp. 9–60, ISBN-10: 0-387-31278-1, ISBN-13: 978-0387-31278-1. [Google Scholar]
- Diverdi, L.A.; Topp, M.R. Subnanosecond time-resolved fluorescence of acridine in solution. J. Phys. Chem. 1984, 88, 3447–3451. [Google Scholar] [CrossRef]
- Lide, D.R. CRC Handbook of Chemistry and Physics, 90th ed.; CRC Press/Taylor and Francis: Boca Raton, FL, USA, 2009; pp. 3–11. ISBN 978-1-4200-9084-0. [Google Scholar]
- Riddick, J.A.; Bunger, W.B.; Sakano, T.K. Organic Solvents: Physical Properties and Methods of Purification, 4th ed.; Wiley-Interscience: New York, NY, USA, 1986; pp. 1344–1400. ISBN 0471084670. [Google Scholar]
- Thordarson, P. Determining association constants from titration experiments in supramolecular chemistry. Chem. Soc. Rev. 2011, 40, 1305–1323. [Google Scholar] [CrossRef]
- Schalley, C.A. Analytical Methods in Supramolecular Chemistry, 2nd ed.; Wiley VCH: Weinheim, Germany, 2012; pp. 1–844. ISBN 978-3-527-32982-3. [Google Scholar]
- Long, G.L.; Winefordner, J.D. Limit of detection. A closer look at the IUPAC definition. Anal. Chem. 1983, 55, 712A–724A. [Google Scholar] [CrossRef]
- Baruah, M.; Qin, W.; Flors, C.; Hofkens, J.; Vallee, R.A.; Beljonne, D.; Van der Auweraer, M.; De Borggraeve, W.M.; Boens, N. Solvent and pH dependent fluorescent properties of a dimethylaminostyryl borondipyrromethene dye in solution. J. Phys. Chem. A 2006, 110, 5998–6009. [Google Scholar] [CrossRef] [PubMed]
- Kolthoff, I.M.; Chantooni, M.K. Dissociation constant, Ka, and stability constant, K(HA2−), of the 1:1 homoconjugate of sulfuric and nitric acids in acetonitrile at 298.1 K. Revised values. J. Chem. Eng. Data 1999, 44, 124–129. [Google Scholar] [CrossRef]
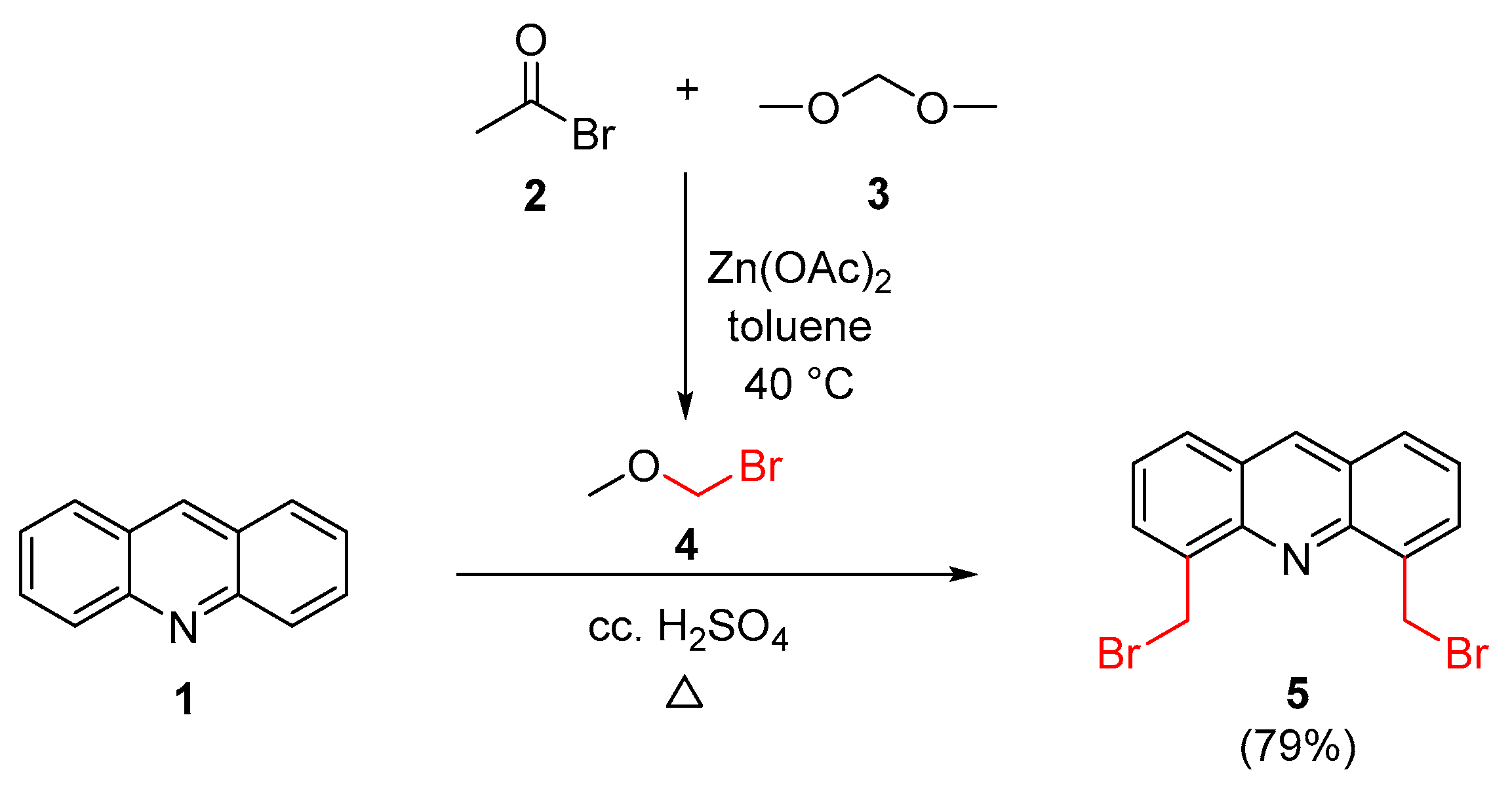




| Experiment No. | BMME (molar eq.) 1 | Temperature (°C) | Reaction Time (h) | Yield (%) |
|---|---|---|---|---|
| 1 | 4 | 50 | 18 | 48 [55] |
| 2 | 4 | 50 | 24 | 49 |
| 3 | 4 | 65 | 24 | 51 |
| 4 | 4 | 65 | 48 | 55 (chrom) 2/79 (cryst) 3 |
| 5 | 6 | 65 | 48 | 37 |
| 6 | 4 | 75 | 48 | 40 |
| Preferred Ion | Macrocycle | Peak Wavelength of the Complex 1 [nm] | LOD [M] | logK |
|---|---|---|---|---|
| Zn2+ | 12 | 444 | 7.0 × 10−8 | 5.5 ± 0.1 |
| 15 | 434 | 1.7 × 10−7 | 5.6 ± 0.2 | |
| 17 | 446 | 2.3 × 10−6 | 5.1 ± 0.3 | |
| Al3+ | 12 | 437 | 2.2 × 10−7 | 5.5 ± 0.3 |
| 15 | 412 | 5.9 × 10−8 | 5.6 ± 0.2 | |
| 17 | 419 | 1.9 × 10−6 | 4.9 ± 0.1 | |
| Bi3+ | 12 | 439 | 2.2 × 10−7 | 4.9 ± 0.3 |
| 15 | 483 | 4.6 × 10−6 | 5.0 ± 0.1 | |
| 17 | 438 | 2.0 × 10−7 | 4.8 ± 0.1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Golcs, Á.; Kovács, K.; Vezse, P.; Tóth, T.; Huszthy, P. Acridino-Diaza-20-Crown-6 Ethers: New Macrocyclic Hosts for Optochemical Metal Ion Sensing. Molecules 2021, 26, 4043. https://doi.org/10.3390/molecules26134043
Golcs Á, Kovács K, Vezse P, Tóth T, Huszthy P. Acridino-Diaza-20-Crown-6 Ethers: New Macrocyclic Hosts for Optochemical Metal Ion Sensing. Molecules. 2021; 26(13):4043. https://doi.org/10.3390/molecules26134043
Chicago/Turabian StyleGolcs, Ádám, Korinna Kovács, Panna Vezse, Tünde Tóth, and Péter Huszthy. 2021. "Acridino-Diaza-20-Crown-6 Ethers: New Macrocyclic Hosts for Optochemical Metal Ion Sensing" Molecules 26, no. 13: 4043. https://doi.org/10.3390/molecules26134043
APA StyleGolcs, Á., Kovács, K., Vezse, P., Tóth, T., & Huszthy, P. (2021). Acridino-Diaza-20-Crown-6 Ethers: New Macrocyclic Hosts for Optochemical Metal Ion Sensing. Molecules, 26(13), 4043. https://doi.org/10.3390/molecules26134043