Immunomodulatory Effects of Green Tea Polyphenols †
Abstract
1. Introduction
2. Immune Potentiating Effects of Tea Polyphenols
2.1. Effects on Cellular Immunity
2.2. Effects on Humoral Immunity
3. Immunomodulation through Anti-Inflammatory Mechanism
4. Immunomodulation through Antioxidation Mechanism
5. Benefits on Human Microbiota and Corresponding Immunological Implications
6. Benefits towards Immune-Related Disease
6.1. Benefits in Autoimmune Diseases
6.2. Benefits in Cutaneousimmunity
6.3. Benefits towards Obesity-Related Immune Disease
7. Problems and Prospects
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| Ag | Antigen |
| ALT | Alanine aminotransaminase |
| APCs | Antigen-presenting cells |
| AST | Aspartate transaminase |
| BSA | Bovine serum albumin |
| CCL20 | CC chemokine ligand-20 |
| CHR | Contact hypersensitivity response |
| CPD | Cyclobutanepyrimidine dimers |
| COX | Cyclooxygenase |
| Con A | Concanavalin A |
| CYP450 | Cytochrome P450 |
| DCs | Dendritic cells |
| EAU | Experimental autoimmune uveoretinitis |
| EGCG | (–)-Epigallocatechin-3-gallate |
| EGCG3”Me | (–)-Epigallocatechin-3-O-(3-O-methyl) gallate |
| ECG | (–)-Epicatechin gallate |
| ERK | External signal-regulated kinases |
| FFA | Free fatty acid |
| GOT | Glutamic oxalacetic transaminase |
| GTE | Green tea extracts |
| GTPs | Green tea polyphenols |
| GSH | Glutathione |
| GSH-Pox | Glutathione peroxidase |
| HESI | Heated electrospray ionization |
| HDPC cells | Human dental pulp-derived cells |
| HDL-C | High-density lipoprotein cholesterol |
| HGFs | Human gingival fibroblasts |
| HILIC | Hydrophilic interaction liquid chromatography |
| HO-1 | Heme oxygenase-1 |
| HF/HS | High-fat/high-sucrose |
| Ig | Immunoglobulin |
| IgM | Immunoglobulin M |
| IFNγ | Interferon gamma |
| IL | Interleukin |
| IKKβ | IkB kinase |
| iNOS | Inducible nitric oxide synthetase |
| IRAKs | IL-1R associated kinases |
| LC-MS | Liquid chromatography mass spectrometry |
| LDL-C | Low-density lipoprotein cholesterol |
| LOX | Lipoxygenase |
| LPS | Lipopolysacharrides |
| LTs | Leukotrienes |
| LTT | T lymphocyte transformation |
| MAPK | Mitogen-activated protein kinase |
| MDA | Malondialdehyde |
| MHC | Major histocompatibility complex |
| MMP | Matrix metalloproteinase |
| MPTP | 1-Methyl-4-phenyl-1,2,3,6-tetrahydropyridine |
| MSU | Monosodium urate |
| MS | Multiple sclerosis |
| MyD88 | Myeloid differentiation primary response gene 88 |
| NF-κB | Nuclear factor-κB |
| NLRP3 | NOD-like receptor protein 3 |
| Nk | Natural killer cells |
| No | Nitric oxide |
| Nod | Non-obese diabetic |
| Pamps | Pathogen-associated molecular patterns |
| PARP | Poly(ADP-ribose) polymerase |
| PCNA | Proliferating cell nuclear antigen |
| PD | Parkinson’s disease |
| p-ERK | Phosphorylated extracellular signal-regulated kinase |
| PGE2 | Prostaglandin E2 |
| PFC | Plaque-forming cell |
| PGs | Prostaglandins |
| PI3K | Phosphatidylinositol-3-kinase |
| p-JNK | Phosphorylated c-Jun N-terminal |
| PPARγ | Peroxisome proliferator-activated receptor gamma |
| PRRs | Pattern-recognition receptors |
| RA | Rheumatoid arthritis |
| RCT | Retinal-choroidal thicknesses |
| RNF216 | Ringfinger protein 216 |
| ROS | Reactive oxygen species |
| SEB | Staphylococcus aureus enterotoxin B |
| SRBCs | Sheep red blood cells |
| SS | Sjogren’s syndrome |
| SOD | Serum superoxide dismutase |
| ssRNA | Single stranded RNA |
| TCA | Tricarboxylic acid |
| Th | T helper cell |
| TFG | Theaflavin digallate |
| TG | Triglyceride |
| TIR | Toll/interleukin 1 receptor |
| TLR | Toll-like receptor |
| Treg | Regulatory T cell |
| UV | Ultraviolet |
| UVR | Ultraviolet radiation |
| WSSV | White spot syndrome virus |
| WAT | White adipose tissue |
References
- Yoshida, S.H.; Gershwin, M.E. Immunity, infection, and nasal disease. Clin. Rev. Allerg. Immunol. 1998, 16, 3–24. [Google Scholar] [CrossRef] [PubMed]
- Patricia, O.W.; Heather, T.; Elma, B. Protective mechanisms of green tea polyphenols in skin. Oxid. Med. Cell. Longev. 2012, 2012, 560682. [Google Scholar]
- Mogensen, T.H. Pathogen recognition and inflammatory signaling in innate immune defenses. Clin. Microbiol. Rev. 2009, 22, 240–273. [Google Scholar] [CrossRef] [PubMed]
- Akira, S.; Uematsu, S.; Takeuchi, O. Pathogen recognition and innate immunity. Cell 2006, 124, 783–801. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Yu, J. Modulation of Toll-like receptor signaling in innate immunity by natural products. Int. Immunopharmacol. 2016, 37, 65–70. [Google Scholar] [CrossRef]
- He, S.; Chu, J.; Wu, L.C.; Mao, H.; Peng, Y.; Alvarez-Breckenridge, C.A.; Hughes, T.; Wei, M.; Zhang, J.; Yuan, S.; et al. MicroRNAs activate natural killer cells through Toll-like receptor signaling. Blood 2013, 121, 4663–4671. [Google Scholar] [CrossRef] [PubMed]
- Qiu, F.; Maniar, A.; Diaz, M.Q.; Chapoval, A.I.; Medvedev, A.E. Activation of cytokineproducing and antitumor activities of natural killer cells and macrophages by engagement of Toll-like and NOD-like receptors. Innate Immun. 2011, 17, 375–387. [Google Scholar] [CrossRef]
- Sallusto, F.; Lanzavecchia, A. The instructive role of dendritic cells on T-cell responses. Arthritis Res. 2002, 4 (Suppl. S3), S127–S132. [Google Scholar] [CrossRef]
- Kabelitz, D. Expression and function of Toll-like receptors in T lymphocytes. Curr. Opin. Immunol. 2007, 19, 39–45. [Google Scholar] [CrossRef]
- Gerondakis, S.; Grumont, R.J.; Banerjee, A. Regulating B-cell activation and survival in response to TLR signals. Immunol. Cell Biol. 2007, 85, 471–475. [Google Scholar] [CrossRef]
- Hennessy, E.J.; Parker, A.E.; O’Neill, L.A. Targeting Toll-like receptors: Emerging therapeutics? Nat. Rev. Drug Discov. 2010, 9, 293–307. [Google Scholar] [CrossRef]
- Tsai, C.C.; Lin, C.R.; Tsai, H.Y.; Chen, C.J.; Li, W.T.; Yu, H.M.; Ke, Y.Y.; Hsieh, W.Y.; Chang, C.C.Y.; Wu, C.Y.; et al. The immunologically active oligosaccharides isolated from wheat grass modulate monocytes via Tolllike receptor-2 signaling. J. Biol. Chem. 2013, 288, 17689–17697. [Google Scholar] [CrossRef]
- Vaure, C.; Liu, Y. A comparative review of toll-like receptor 4 expression and functionality in different animal species. Front. Immunol. 2014, 5, 316. [Google Scholar] [CrossRef]
- O’Neill, L.A.; Golenbock, D.; Bowie, A.G. The history of Toll-like receptors- redefining innate immunity. Nat. Rev. Immunol. 2013, 13, 453–460. [Google Scholar] [CrossRef] [PubMed]
- Shaw, A.C.; Panda, A.; Joshi, S.R.; Qian, F.; Allore, H.G.; Montgomery, R.R. Dysregulation of human Toll-like receptor function in aging. Ageing Res. Rev. 2011, 10, 346–353. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Zhou, Y.; Feng, G.; Su, S.B. The critical role of Toll-like receptor signaling pathways in the induction and progression of autoimmune diseases. Curr. Mol. Med. 2009, 9, 365–374. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.Y.; Hwang, D.H. The modulation of inflammatory gene expression by lipids: Mediation through Toll-like receptors. Mol. Cell 2006, 21, 174–185. [Google Scholar]
- Dias, D.A.; Urban, S.; Roessner, U. A historical overview of natural products in drug discovery. Metabolites 2012, 2, 303–336. [Google Scholar] [CrossRef] [PubMed]
- Haddad, P.S.; Azar, G.A.; Groom, S.; Boivin, M. Natural health products, modulation of immune function and prevention of chronic diseases. Evid. Based Complement. Alternat. Med. 2005, 2, 513–520. [Google Scholar]
- Kingston, D.G. Modern natural products drug discovery and its relevance to biodiversity conservation. J. Nat. Prod. 2011, 74, 496–511. [Google Scholar] [CrossRef]
- Pan, M.H.; Chiou, Y.S.; Wang, Y.J.; Ho, C.T.; Lin, J.K. Multistage carcinogenesis process as molecular targets in cancer chemoprevention by epicatechin-3-gallate. Food Funct. 2011, 2, 101. [Google Scholar] [CrossRef]
- Zhang, X.; Li, J.; Li, Y.; Liu, Z.; Lin, Y.; Huang, J. Anti-melanogenic effects of epigallocatechin-3-gallate (EGCG), epicatechin-3-gallate (ECG) and gallocatechin-3-gallate (GCG) via down-regulation of cAMP/CREB/MITF signaling pathway in B16F10 melanoma cells. Fitoterapia 2020, 145, 104634. [Google Scholar] [CrossRef]
- Way, T.D.; Lin, H.Y.; Kuo, D.H.; Tsai, S.J.; Shieh, J.C.; Wu, J.C.; Lee, M.R.; Lin, J.K. Pu-erh tea attenuates hyperlipogenesis and induces hepatoma cells growth arrest through activating AMP-activated protein kinase (AMPK) in human HepG2 cells. J. Agric. Food Chem. 2009, 57, 5257. [Google Scholar] [CrossRef]
- Wilasrusmee, C.; Kittur, S.; Siddiqui, J.; Bruch, D.; Wilasrusmee, S.; Kittur, D.S. In vitro immunomodulatory effects of ten commonly used herbs on murine lymphocytes. J. Altern. Complem. Med. 2002, 8, 467–475. [Google Scholar] [CrossRef] [PubMed]
- Braun, D.P.; Johnson, D.M.; Katsantonis, N.G.; Bhesaniya, K.; Staren, E.D. Apoptotic and immunomodulatory effects of green tea extracts (GTE) on chemoresistant human tumor cells. J. Clin. Oncol. 2009, 27 (Suppl. S15), e22101. [Google Scholar] [CrossRef]
- Youn, H.S.; Lee, J.Y.; Saitoh, S.I.; Miyake, K.; Kang, K.W.; Yong, J.C.; Hwang, D.H. Suppression of MyD88- and TRIF-dependent signaling pathways of Toll-like receptor by (−)-epigallocatechin-3-gallate, a polyphenol component of green tea. Biochem. Pharmacol. 2006, 72, 850–859. [Google Scholar] [CrossRef]
- Watson, J.L.; Vicario, M.; Wang, A.; Moreto, M.; McKay, D.M. Immune cell activation and subsequent epithelial dysfunction by Staphylococcus enterotoxin B is attenuated by the green tea polyphenol (−)-epigallocatechin gallate. Cell Immunol. 2005, 237, 7–16. [Google Scholar] [CrossRef] [PubMed]
- Katiyar, S.K.; Mukhtar, H. Green tea polyphenol (−)-epigallocatechin-3-gallate treatment to mouse skin prevents UVB-induced infiltration of leukocytes, depletion of antigen-presenting cells, and oxidative stress. J. Leukoc. Biol. 2001, 69, 719–726. [Google Scholar] [PubMed]
- Katiyar, S.K.; Vaid, M.; Van Steeg, H.; Meeran, S.M. Green tea polyphenols prevent UV-induced immunosuppression by rapid repair of DNA damage and enhancement of nucleotide excision repair genes. Cancer Prev. Res. 2010, 3, 179–189. [Google Scholar] [CrossRef]
- Ahn, S.C.; Kim, G.Y.; Kim, J.H.; Baik, S.W.; Han, M.K.; Lee, H.J.; Moon, D.O.; Lee, C.M.; Kang, J.H.; Kim, B.H. Epigallocatechin-3-gallate, constituent of green tea, suppresses the LPS-induced phenotypic and functional maturation of murine dendritic cells through inhibition of mitogen-activated protein kinases and NF-κB. Biochem. Biophys. Res. Commun. 2004, 313, 148–155. [Google Scholar] [CrossRef]
- Yoneyama, S.; Kawai, K.; Tsuno, N.H.; Okaji, Y.; Asakage, M.; Tsuchiya, T.; Yamada, J.; Sunami, E.; Osada, T.; Kitayama, J. Epigallocatechin gallate affects human dendritic cell differentiation and maturation. J. Allergy Clin. Immun. 2008, 121, 209–214. [Google Scholar] [CrossRef]
- Wang, Z.; Sun, B.; Zhu, F. Epigallocatechin-3-gallate protects Kuruma shrimp Marsupeneaus japonicus from white spot syndrome virus and Vibrio alginolyticus. Fish Shellfish Immun. 2018, 78, 1–9. [Google Scholar] [CrossRef]
- Varilek, G.W.; Yang, F.; Lee, E.Y.; de Villiers, W.J.; Zhong, J.; Oz, H.S.; Westberry, K.F.; McClain, C.J. Green tea polyphenol extract attenuates inflammation in interleukin-2-deficient mice, a model of autoimmunity. J. Nutr. 2001, 131, 2034–2039. [Google Scholar] [CrossRef] [PubMed]
- Yang, F.; Oz, H.S.; Barve, S.; de Villiers, W.J.; McClain, C.J.; Varilek, G.W. The green tea polyphenol (−)-epigallocatechin-3-gallate blocks nuclear factor-κB activation by inhibiting I κB kinase activity in the intestinal epithelial cell line IEC. Mol. Pharmacol. 2001, 60, 528–533. [Google Scholar] [PubMed]
- Tedeschi, E.; Suzuki, H.; Menegazzi, M. Antiinflammatory action of EGCG, the main component of green tea, through STAT-1 inhibition. Ann. N. Y. Acad. Sci. 2002, 973, 435–437. [Google Scholar] [CrossRef] [PubMed]
- Deng, Q.; Xu, J.; Yu, B.; He, J.; Zhang, K.; Ding, X.; Chen, D. Effect of dietary tea polyphenols on growth performance and cell mediated immune response of post-weaning piglets under oxidative stress. Arch. Anim. Nutr. 2010, 64, 12–21. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.Y.J.; Wan, J.M.F. Vitamin E supplementation improves cell-mediated immunity and oxidative stress of Asian men and women. J. Nutr. 2000, 130, 2932–2937. [Google Scholar] [CrossRef] [PubMed]
- Bayer, J.; Gomer, A.; Demir, Y.; Amano, H.; Kish, D.D.; Fairchild, R.; Heeger, P.S. Effects of green tea polyphenols on murine transplant-reactive T cell immunity. Clin. Immunol. 2004, 110, 100–108. [Google Scholar] [CrossRef]
- Gringhuis, S.I.; Papendrecht-van der Voort, E.A.M.; Leow, A.; Levarht, E.W.N.; Breedveld, F.C.; Verweij, C.L. Effect of redox balance alterations on cellular localization of LAT and downstream T-cell receptor signalling pathways. Mol. Cell Biol. 2002, 22, 400–411. [Google Scholar] [CrossRef]
- Wu, D.; Guo, Z.; Ren, Z.; Guo, W.; Meydani, S.N. Green tea EGCG suppresses T cell proliferation through impairment of IL-2/IL-2 receptor signaling. Free Radic. Biol. Med. 2009, 47, 636–643. [Google Scholar] [CrossRef]
- Pae, M.; Ren, Z.; Meydani, M.; Shang, F.; Meydani, S.N.; Wu, D. Epigallocatechin-3- gallate directly suppresses T cell proliferation through impaired IL-2 utilization and cell cycle progression. J. Nutr. 2010, 140, 1509–1515. [Google Scholar] [CrossRef]
- Zhou, T.; Zhu, M.; Liang, Z. (−)-Epigallocatechin-3-gallate modulates peripheral immunity in the MPTP-induced mouse model of Parkinson’s disease. Mol. Med. Rep. 2018, 17, 4883–4888. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.C.; Kao, Y.H.; Shih, S.F.; Tsai, M.C.; Lin, C.S.; Chen, L.W.; Chuang, Y.P.; Tsui, P.F.; Ho, L.J.; Lai, J.H.; et al. Epigallocatechin-3-gallate exhibits immunomodulatory effects in human primary T cells. Biochem. Biophys. Res. Commun. 2021, 550, 70–76. [Google Scholar] [CrossRef] [PubMed]
- Demeule, M.; Brossard, M.; Pagé, M.; Gingras, D.; Béliveau, R. Matrix metalloproteinase inhibition by green tea catechins. Biochim. Biophys. Acta 2000, 1478, 51. [Google Scholar] [CrossRef]
- Corbel, M.; Belleguic, C.; Boichot, E.; Lagente, V. Involvement of gelatinases (MMP-2 and MMP-9) in the development of airway inflammation and pulmonary fibrosis. Cell Biol. Toxicol. 2002, 18, 51. [Google Scholar] [CrossRef] [PubMed]
- Hofbauer, R.; Frass, M.; Gmeiner, B.; Handler, S.; Speiser, W.; Kapiotis, S. The green tea extract epigallocatechin gallate is able to reduce neutrophil transmigration through monolayers of endothelial cells. Wiener Klinische Wochenschrift 1999, 111, 278. [Google Scholar] [PubMed]
- Nguyen, T.; Payan, B.; Zambrano, A.; Du, Y.; Bondesson, M.; Mohan, C. Epigallocatechin-3-gallate suppresses neutrophil migration speed in a transgenic zebrafish model accompanied by reduced inflammatory mediators. J. Inflamm. Res. 2019, 12, 231–239. [Google Scholar] [CrossRef]
- Donà, M.; Dell’Aica, I.; Calabrese, F.; Benelli, R.; Morini, M.; Albini, A.; Garbisa, S. Neutrophil restraint by green tea: Inhibition of inflammation, associated angiogenesis, and pulmonary fibrosis. J. Immunol. 2003, 170, 4335–4341. [Google Scholar] [CrossRef]
- Yang, F.; de Villiers, W.J.S.; McClain, C.J.; Varilek, G.W. Green tea polyphenols block endotoxin-induced tumor necrosis factor production and lethality in a murine model. J. Nutr. 1998, 128, 2334–2340. [Google Scholar] [CrossRef]
- Lin, Y.L.; Lin, J.K. (−)-Epigallocatechin-3-gallate blocks the induction of nitric oxide synthase by down-regulating lipopolysaccharide-induced activity of transcription factor nuclear factor-kB. Mol. Pharmacol. 1997, 52, 465–472. [Google Scholar] [CrossRef]
- Zvetkova, E.; Wirleitner, B.; Tram, N.T.; Schennach, H.; Fuchs, D. Aqueous extracts of Crinum latifolium (L.) and Camellia sinensis show immunomodulatory properties in human peripheral blood mononuclear cells. Int. Immunopharmacol. 2001, 1, 2143–2150. [Google Scholar] [CrossRef]
- Khan, A.; Ali, N.H.; Santercole, V.; Paglietti, B.; Rubino, S.; Kazmi, S.U.; Farooqui, A. Camellia sinensis mediated enhancement of humoral immunity to particulate and non-particulate Antigens. Phytother. Res. 2016, 30, 41–48. [Google Scholar] [CrossRef]
- Haqqi, T.M.; Anthony, D.D.; Gupta, S.; Ahmad, N.; Lee, M.S.; Kumar, G.K.; Mukhtar, H. Prevention of collagen-induced arthritis in mice by a polyphenolic fraction from green tea. Proc. Natl. Acad. Sci. USA 1999, 96, 4524–4529. [Google Scholar] [CrossRef] [PubMed]
- Hu, Z.Q.; Toda, M.; Okubo, S.; Hara, Y.; Shimamura, T. Mitogenic activity of (−) epigallocatechin gallate on B-cells and investigation of its structure function relationship. Int. J. Immunopharmacol. 1992, 14, 1399–1407. [Google Scholar] [PubMed]
- Guo, H.; Lin, W.; Wang, L.; Zhang, D.; Wu, X.; Li, L.; Li, D.; Tang, R.; Yang, L.; Qiu, Y. The supplementation of dietary selenium yeast and green tea-derived polyphenols improves antioxidant capacity and immune response in juvenile Wuchang bream under ammonia stress. Aquac. Res. 2020, 51, 1–14. [Google Scholar] [CrossRef]
- Majdalawieh, A.; Ro, H.S. Regulation of IκBalpha function and NF-κB signaling: AEBP1 is a novel proinflammatory mediator in macrophages. Mediat. Inflamm. 2010, 2010, 823821. [Google Scholar] [CrossRef] [PubMed]
- Majdalawieh, A.F.; Fayyad, M.W. Immunomodulatory and anti-inflammatory action of Nigella sativa and thymoquinone: A comprehensive review. Int. Immunopharmacol. 2015, 28, 295–304. [Google Scholar] [CrossRef] [PubMed]
- Van Ryn, J.; Trummlitz, G.; Pairet, M. COX-2 selectivity and inflammatory processes. Curr. Med. Chem. 2000, 7, 1145–1161. [Google Scholar] [CrossRef]
- Magee, P.; Pearson, S.; Whittingham-Dowda, J.; Allen, J.J. PPARγ as a molecular target of EPA anti-inflammatory activity during TNF-α-impaired skeletal muscle cell differentiation. J. Nutr. Biochem. 2012, 23, 1440–1448. [Google Scholar] [CrossRef]
- Kim, H.R.; Rajaiah, R.; Wu, Q.L.; Satpute, S.R.; Tan, M.T.; Simon, J.E.; Berman, B.M.; Moudgil, K.D. Green tea protects rats against autoimmune arthritis by modulating disease-related immune events. J. Nutr. 2008, 138, 2111–2116. [Google Scholar] [CrossRef]
- Abboud, P.A.; Hake, P.W.; Burroughs, T.J.; Odoms, K.; O’Connor, M.; Mangeshkar, P.; Wong, H.R.; Zingarelli, B. Therapeutic effect of epigallocatechin-3-gallate in a mouse model of colitis. Eur. J. Pharmacol. 2008, 579, 411–417. [Google Scholar] [CrossRef] [PubMed]
- Tsai, P.Y.; Ka, S.M.; Chang, J.M.; Chen, H.C.; Sui, H.A.; Li, C.Y.; Hua, K.F.; Chang, W.L.; Huang, J.J.; Yang, S.S.; et al. Epigallocatechin-3-gallte prevents lupus nephritis development in mice via enhancing the Nrf2 antioxidant pathway and inhibiting NLRP3 inflammasome activation. Free Radic. Biol. Med. 2011, 51, 744–754. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Ren, Z.; Xu, Y.; Xiao, S.; Meydani, S.N.; Wu, D. Epigallocatechin-3-gallate ameliorates experimental autoimmune encephalomyelitis by altering balance among CD4+ T-cell subsets. Am. J. Pathol. 2012, 180, 221–234. [Google Scholar] [CrossRef]
- Hoshikawa, S.; Nakagawa, Y.; Ozaki, H.; Takahashi, Y.; Ito, S.; Yoshida, K.; Mori, K. Effects of green tea polyphenols on iodide-induced autoimmune thyroiditis in nonobese diabetic mice. Immunol. Invest. 2013, 42, 235–246. [Google Scholar] [CrossRef] [PubMed]
- Wheeler, D.S.; Catravas, J.D.; Odoms, K.; Denenberg, A.; Malhotra, V.; Wong, H.R. Epigallocatechin-3-gallate, a green tea-derived polyphenol, inhibits IL-1b- dependent proinflammatory signal transduction in cultured respiratory epithelial cells. J. Nutr. 2004, 134, 1039–1044. [Google Scholar] [CrossRef]
- Nakanishi, T.; Mukai, K.; Yumoto, H.; Hirao, K.; Hosokawa, Y.; Matsuo, T. Anti-inflammatory effect of catechin on cultured human dental pulp cells affected by bacteria-derived factors. Eur. J. Oral Sci. 2010, 118, 145–150. [Google Scholar] [CrossRef] [PubMed]
- Nakanishi, T.; Mukai, K.; Hosokawa, Y.; Takegawa, D.; Matsuo, T. Catechins inhibit vascular endothelial growth factor production and cyclooxygenase-2 expression in human dental pulp cells. Int. Endod. J. 2015, 48, 277–282. [Google Scholar] [CrossRef] [PubMed]
- el-Saadany, M.A.; Rawel, H.M.; Raila, J. Antioxidants modulate the IL-6 induced inhibition of negative acute-phase protein secretion in HepG2 cells. Cell Biochem. Funct. 2008, 26, 95–101. [Google Scholar] [CrossRef]
- Hosokawa, Y.; Hosokawa, I.; Ozaki, K.; Nakanishi, T.; Nakae, H.; Matsuo, T. Catechins inhibit CCL20 production in IL-17A-stimulated human jingival fibroblasts. Cell. Physiol. Biochem. 2009, 24, 391–396. [Google Scholar] [CrossRef]
- Chiou, Y.S.; Huang, Q.R.; Ho, C.T. Directly interact with keap1 and lps is involved in the anti-inflammatory mechanisms of (−)-epicatechin-3-gallate in LPS- induced macrophages and endotoxemia. Free Radic. Biol. Med. 2016, 94, 1–16. [Google Scholar] [CrossRef]
- Lee, H.E.; Yang, G.; Park, Y.B.; Kang, H.C.; Cho, Y.Y.; Lee, H.S.; Lee, J.Y. Epigallocatechin-3-gallate prevents acute gout by suppressing NLRP3 inflammasome activation and mitochondrial DNA Synthesis. Molecules 2019, 24, 2138. [Google Scholar] [CrossRef]
- Gao, Z.; Han, Y.; Hu, Y.; Wu, X.; Wang, Y.; Zhang, X.; Fu, J.; Zou, X.; Zhang, J.; Chen, X.; et al. Targeting HO-1 by epigallocatechin-3-gallate reduces contrast-induced renal injury via anti-oxidative stress and anti-inflammation pathways. PLoS ONE 2016, 11, e0149032. [Google Scholar] [CrossRef]
- Wang, D.X.; Gao, Q.; Wang, T.T.; Kan, Z.P.; Li, X.; Hu, L.Z.; Peng, C.Y.; Qian, F.; Wang, Y.; Granato, D. Green tea polyphenols and epigallocatechin-3-gallate protect against perfluorodecanoic acid induced liver damage and inflammation in mice by inhibiting NLRP3 inflammasome activation. Food Res. Int. 2020, 127, 108628. [Google Scholar] [CrossRef]
- Dobmeyer, T.S.; Findhammer, S.; Dobmeyer, J.M.; Klein, S.A.; Raffel, B.; Hoelzer, D.; Helm, E.B.; Kabelitz, D.; Rossol, R. Ex vivo induction of apoptosis in lymphocytes is mediated by oxidative stress: Role for lymphocyte loss in HIV infection. Free Radic. Biol. Med. 1997, 22, 775–785. [Google Scholar] [CrossRef]
- Sordillo, L.M.; Aitken, S.L. Impact of oxidative stress on the health and immune function of dairy cattle. Vet. Immunol. Immunopathol. 2009, 128, 104–109. [Google Scholar] [CrossRef] [PubMed]
- Sklavos, M.M.; Tse, H.M.; Piganelli, J.D. Redox modulation inhibits CD8 T cell effector function. Free Radic. Biol. Med. 2008, 45, 1477–1486. [Google Scholar] [CrossRef] [PubMed]
- Locatelli, F.; Canaud, B.; Eckardt, K.U.; Stenvinkel, P.; Wanner, C.; Zoccali, C. Oxidative stress in end-stage renal disease: An emerging threat to patient outcome. Nephrol. Dial. Transpl. 2003, 18, 1272–1280. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Safa, R.; Rusciano, D.; Osborne, N.N. Epigallocatechin gallate, an active ingredient from green tea, attenuates damaging influences to the retina caused by ischemia/reperfusion. Brain Res. 2007, 1159, 40–53. [Google Scholar] [CrossRef] [PubMed]
- Vidali, M.; Stewart, S.F.; Albano, E. Interplay between oxidative stress and immunity in the progression of alcohol-mediated liver injury. Trends Mol. Med. 2008, 14, 63–71. [Google Scholar] [CrossRef]
- Zhong, L.; Hu, Y.; Hu, Y.; Li, J.; Tian, Y.; Chen, J.; Ai, Q.; Xiao, T. Effects of dietary tea polyphenols on growth, immunity and lipid metabolism of juvenile black carp Mylopharyngodon piceus. Aquac. Res. 2019, 51, 1–8. [Google Scholar] [CrossRef]
- Burckhardt, I.C.; Gozal, D.; Dayyat, E.; Cheng, Y.; Li, R.C.; Row, B.W. Green tea catechin polyphenols attenuate behavioral and oxidative responses to intermittent hypoxia. Am. J. Resp. Crit. Care 2008, 177, 1135–1141. [Google Scholar] [CrossRef]
- Mohd, F.; Mohammad, O.; Sandesh, C.; Sheikh, M.H.; Aamir, A. Mobilization of nuclear copper by green tea polyphenol epicatechin-3-gallate and subsequent prooxidant breakage of cellular DNA: Implications for cancer chemotherapy. Int. J. Mol. Sci. 2017, 18, 34. [Google Scholar]
- Axling, U.; Olsson, C.; Xu, J.; Fernandez, C.; Larsson, S.; Ström, K.; Ahrnė, S.; Holm, C.; Molin, G.; Berger, K. Green tea powder and Lactobacillus plantarum affect gut microbiota, lipid metabolism and inflammation in high-fat fed C57BL/6J mice. Nutr. Metab. 2012, 9, 105. [Google Scholar] [CrossRef]
- Jin, J.S.; Touyama, M.; Hisada, T.; Benno, Y. Effects of green tea consumption on human fecal microbiota with special reference to Bifidobacterium species. Microbiol. Immunol. 2012, 11, 729–739. [Google Scholar] [CrossRef]
- Dae-Bang, S.; Jeong, H.W.; Cho, D.; Lee, B.J.; Lee, J.H.; Choi, J.Y.; Il-Hong, B.; Sung-Joon, L. Fermented green tea extract alleviates obesity and related complications and alters gut microbiota composition in diet-induced obese mice. J. Med. Food 2015, 18, 549–556. [Google Scholar]
- Liu, Z.; Chen, Z.; Guo, H.; He, D.; Zhao, H.; Wang, Z.; Zhang, W.; Liao, L.; Zhang, C.; Ni, L. The modulatory effect of infusions of green tea, oolong tea, and black tea on gut microbiota in high-fat-induced obese mice. Food Funct. 2016, 7, 4869–4879. [Google Scholar] [CrossRef] [PubMed]
- Guo, X.; Cheng, M.; Zhang, X.; Cao, J.; Wu, Z.; Weng, P. Green tea polyphenols reduce obesity in high-fat diet-induced mice by modulating intestinal microbiota composition. Int. J. Food Sci. Tech. 2017, 52, 1723–1730. [Google Scholar] [CrossRef]
- Zhou, J.; Tang, L.; Shen, C.L.; Wang, J.S. Green tea polyphenols boost gut-microbiota- dependent mitochondrial TCA and urea cycles in Sprague-Dawley rats. J. Nutr. Biochem. 2020, 81, 108395. [Google Scholar] [CrossRef] [PubMed]
- Molina, N.; Bolin, A.P.; Otton, R. Green tea polyphenols change the profile of inflammatory cytokine release from lymphocytes of obese and lean rats and protect against oxidative damage. Int. Immunopharmacol. 2015, 28, 985–996. [Google Scholar] [CrossRef] [PubMed]
- Wu, D.; Wang, J.; Pae, M.; Meydani, S.N. Green tea EGCG, T cells, and T cell- mediated autoimmune diseases. Mol. Aspects Med. 2012, 33, 107–118. [Google Scholar] [CrossRef] [PubMed]
- Hayashi, Y.; Arakaki, R.; Ishimaru, N. Apoptosis and estrogen deficiency in primary Sjögren’s syndrome. Curr. Opin. Rheumatol. 2004, 16, 522–526. [Google Scholar] [CrossRef]
- Gillespie, K.; Kodani, I.; Dickinson, D.P.; Ogbureke, K.; Camba, A.M.; Wu, M.; Looney, S.; Chu, T.C.; Qin, H.; Bisch, F.; et al. Effects of oral consumption of the green tea polyphenol EGCG in a murine model for human Sjogren’s syndrome, an autoimmune disease. Life Sci. 2008, 83, 581–588. [Google Scholar] [CrossRef]
- Van Woerkom, J.M.; Geertzema, J.G.; Nikkels, P.G.; Kruize, A.A.; Smeenk, R.J.; Vroom, T.M. Expression of Ro/SS-A and La/SS-B determined by immunohistochemistry in healthy, inflamed and autoimmune diseased human tissues: A generalized phenomenon. Clin. Exp. Rheumatol. 2004, 22, 285–292. [Google Scholar]
- Rosen, A.; Casciola-Rosen, L. Altered autoantigen structure in Sjögren’s syndrome: Implications for the pathogenesis of autoimmune tissue damage. Crit. Rev. Oral Biol. Med. 2004, 15, 156–164. [Google Scholar] [CrossRef]
- Hsu, S.; Dickinson, D.P.; Qin, H.; Borke, J.; Ogbureke, K.; Winger, J.N.; Walsh, D.S.; Bollag, W.B.; Stoppler, H.; Sharawy, M.; et al. Green tea polyphenols reduce autoimmune symptoms in a murine model for human Sjogren’s syndrome and protect human salivary acinar cells from TNF-alpha- induced cytotoxicity. Autoimmunity 2007, 40, 138–147. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Yip, Y.W.Y.; Ren, J.; Hui, W.K.; He, J.N.; Yu, Q.X.; Chu, K.O.; Ng, T.K.; Chan, S.O.; Pang, C.P.; et al. Green tea catechins alleviate autoimmune symptoms and visual impairment in a murine model for human chronic intraocular infammation by inhibiting Th17-associated pro-inflammatory gene expression. Sci. Rep. 2019, 9, 2301. [Google Scholar] [CrossRef]
- Meeran, S.M.; Mantena, S.K.; Katiyar, S.K. Prevention of ultraviolet radiation—induced immunosuppression by (−)-epigallocatechin-3-gallate in mice is mediated through interleukin 12-dependent DNA repair. Clin. Cancer Res. 2006, 12, 2272–2280. [Google Scholar] [CrossRef] [PubMed]
- Muthusamy, V.; Piva, T.J. The UV response of the skin: A review of the MAPK, NFκB and TNFα signal transduction pathways. Arch. Dermatol. Res. 2010, 302, 5–17. [Google Scholar] [CrossRef]
- Schwarz, A.; Ständer, S.; Berneburg, M.; Böhm, M.; Kulms, D.; van Steeg, H.; Grosse-Heitmeyer, K.; Krutmann, J.; Schwarz, T. Interleukin-12 suppresses ultraviolet radiation-induced apoptosis by inducing DNA repair. Nat. Cell Biol. 2002, 4, 26–31. [Google Scholar] [CrossRef] [PubMed]
- Kurukulasuriya, L.R.; Stas, S.; Lastra, G.; Manrique, C.; Sowers, J.R. Hypertension in obesity. Med. Clin. N. Am. 2011, 95, 903–917. [Google Scholar] [CrossRef]
- Richardson, V.R.; Smith, K.A.; Carter, A.M. Adipose tissue inflammation: Feeding the development of type 2 diabetes mellitus. Immunobiology 2013, 218, 1497–1504. [Google Scholar] [CrossRef] [PubMed]
- Albuquerque, K.F.F.S.; Marinovic, M.P.; Morandi, A.C.; Bolin, A.P.; Otton, R. Green tea polyphenol extract in vivo attenuates inflammatory features of neutrophils from obese rats. Eur. J. Nutr. 2016, 55, 1261–1274. [Google Scholar] [CrossRef] [PubMed]
- Fenton, J.I.; Nunez, N.P.; Yakar, S.; Perkins, S.N.; Hord, N.G.; Hursting, S.D. Diet-induced adiposity alters the serum profile of inflammation in C57BL/6 N mice as measured by antibody array. Diabetes Obes. Metab. 2009, 11, 343–354. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Akbar, S.M.F.; Miyake, T.; Abe, M.; Al-Mahtab, M.; Furukawa, S.; Bunzo, M.; Hiasa, Y.; Onji, M. Diminished immune response to vaccinations in obesity: Role of myeloid-derived suppressor and other myeloid cells. Obes. Res. Clin. Pract. 2015, 9, 35–44. [Google Scholar] [CrossRef] [PubMed]
- Grant, R.; Youm, Y.H.; Ravussin, A.; Dixit, V.D. Quantification of adipose tissue leukocytosis in obesity. Methods Mol. Biol. 2013, 1040, 195–209. [Google Scholar]
- Chatzigeorgiou, A.; Karalis, K.P.; Bornstein, S.R.; Chavakis, T. Lymphocytes in obesityrelated adipose tissue inflammation. Diabetologia 2012, 55, 2583–2592. [Google Scholar] [CrossRef]
- Huang, J.; Zhou, Y.; Wan, B.; Wang, Q.; Wan, X. Green tea polyphenols alter lipid metabolism in the livers of broiler chickens through increased phosphorylation of AMP-activated protein kinase. PLoS ONE 2017, 12, e187061. [Google Scholar] [CrossRef] [PubMed]
- Rothenberg, D.; Zhou, C.; Zhang, L. A review on the weight-loss effects of oxidized tea polyphenols. Molecules 2018, 23, 1176. [Google Scholar] [CrossRef]
- Daniel, G.R.; Schlachter, M.F.; Volker, E.; Weber, P. Effect of EGCG on lipid absorption and plasma lipid levels in rats. J. Nutr. Biochem. 2003, 14, 326–332. [Google Scholar]
- Kumazoe, M.; Nakamura, Y.; Yamashita, M.; Suzuki, T.; Takamatsu, K.; Huang, Y.; Bae, J.; Yamashita, S.; Murata, M.; Yamada, S.; et al. Green tea polyphenol epigallocatechin-3-gallate suppresses Toll-like receptor 4 expression via upregulation of E3 ubiquitin-protein ligase RNF. J. Biol. Chem. 2017, 292, 4077–4088. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.S.; Chen, G.; Wu, Q. Recent scientific studies of a traditional Chinese medicine, tea, on prevention of chronic diseases. J. Tradit. Complement. Med. 2014, 4, 17–23. [Google Scholar] [CrossRef] [PubMed]
- Tang, H.; Hao, S.; Chen, X.; Li, Y.; Yin, Z.; Zou, Y.; Song, X.; Lixia, L.; Ye, G.; Zhao, L.; et al. Epigallocatechin-3-gallate protects immunity and liver drug-metabolism function in mice loaded with restraint stress. Biomed. Pharmacother. 2020, 129, 110418. [Google Scholar] [CrossRef] [PubMed]
- Wong, C.P.; Nguyen, L.P.; Noh, S.K.; Bray, T.M.; Bruno, R.S.; Ho, E. Induction of regulatory T cells by green tea polyphenol EGCG. Immunol. Lett. 2011, 139, 7–13. [Google Scholar] [CrossRef] [PubMed]
- Kapetanovic, I.M.; Crowell, J.A.; Krishnaraj, R.; Zakharov, A.; Lindeblad, M.; Lyubimov, A. Exposure and toxicity of green tea polyphenols in fasted and non-fasted dogs. Toxicology 2009, 260, 28–36. [Google Scholar] [CrossRef]
- Isbrucker, R.A.; Bausch, J.; Edwards, J.A.; Wolz, E. Safety studies on epigallocatechin gallate (EGCG) preparations—Part 1: Genotoxicity. Food Chem. Toxicol. 2006, 44, 626–635. [Google Scholar] [CrossRef] [PubMed]
- Reygaert, W.C. Green tea catechins: Their use in treating and preventing infectious diseases. BioMed Res. Int. 2018, 2018, 9105261. [Google Scholar] [CrossRef]
- Harikrishnan, R.; Balasundaram, C.; Heo, M.S. Influence of diet enriched with green tea on innate humoral and cellular immune response of kelp grouper Epinephelusbruneus to Vibrio carchariae infection. Fish Shellfish Immun. 2011, 30, 972–979. [Google Scholar] [CrossRef] [PubMed]
- Camouse, M.M.; Domingo, D.S.; Swain, F.R.; Conrad, E.P.; Matsui, M.S.; Maes, D.; Declercq, L.; Cooper, K.D.; Stevens, S.R.; Baron, E.D. Topical application of green and white tea extracts provides protection from solar-simulated ultraviolet light in human skin. Exp. Dermatol. 2009, 18, 522–526. [Google Scholar] [CrossRef]
- Yang, H.Y.; Yang, S.C.; Chao, J.C.; Chen, J.R. Beneficial effects of catechin-rich green tea and inulin on the body composition of overweight adults. Brit. J. Nutr. 2012, 107, 749–754. [Google Scholar] [CrossRef]
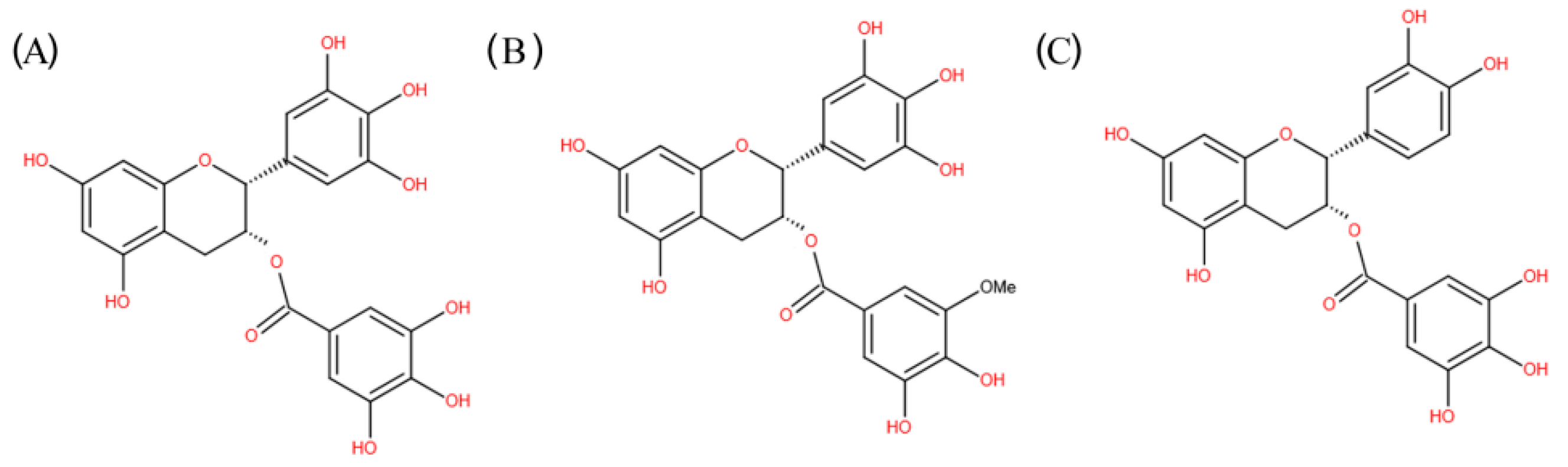
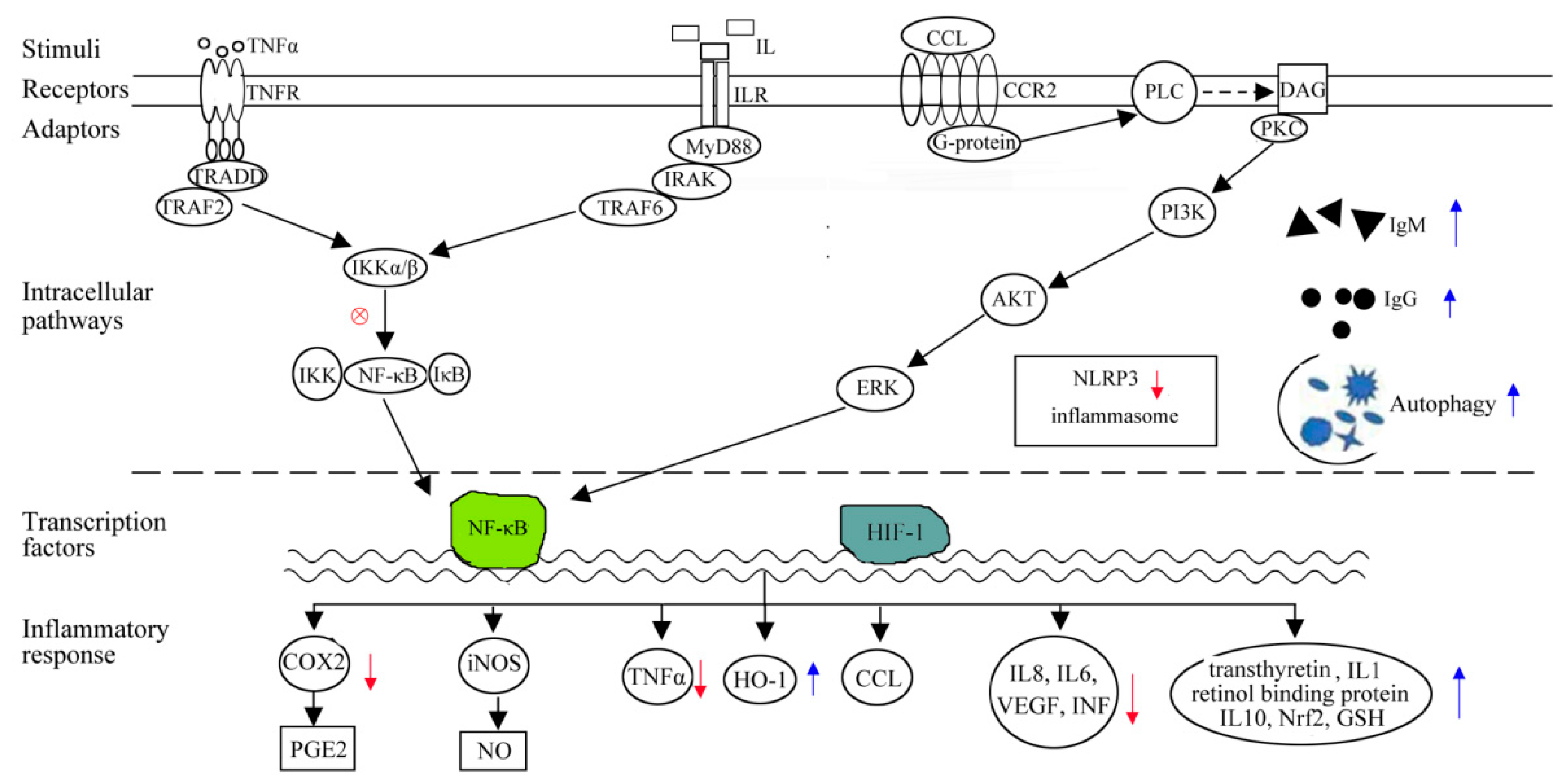
| TLR Subfamilies | Recognized Ligands |
|---|---|
| TLR1, TLR2, TLR6 | lipopeptides and glycolipids |
| TLR7, TLR8, TLR9 | nucleic acids (ssRNA and unmethylated CpG) |
| TLR3 | dsRNA associated with viral infection |
| TLR4 | fibronectin, lipopolysaccharides (LPS), and heat shock proteins |
| TLR5 | bacterial flagellin |
| TLR11, TLR12 | profilin and actin-binding protein |
| Component | Model | Effect | Reference |
|---|---|---|---|
| GTPs and their derivatives | Human cells | Stimulation of g multiple TLR signaling pathways | [24] |
| Murine lymphocytes | Inhibition of proliferation | ||
| EGCG | RAW 264.7 cells (a murine monocytic cell line, ATCC TIB-71) | Inhibition of IKKβ and NF-κB activation | [26] |
| 293T (human embryonic kidney cells) | |||
| EGCG | Human PBMC cells | Inhibition of the production of IFNγ | [27] |
| EGCG | C3H/HeN mice | Inhibition of the depletion of APCs and UV-induced immunosuppression | [28,29] |
| EGCG | Mouse bone marrow-derived DCs | Inhibition of DCs maturation | [29,30] |
| Human monocytes-derived DCs | |||
| EGCG | Kuruma shrimp Marsupeneaus japonicus challenged with WSSV | Stimulation of innate immune-related gene expressions (IMD, proPO, QM, myosin, Rho, Rab7, p53, TNF-alpha, MAPK, and NOS) | [32] |
| Gram-negative bacterium Vibrio alginolyticus | |||
| EGCG | shrimp innate immune system | Enhance of immune parameters, inhibition of apoptosis | [32] |
| GTPs | Interleukin-2-deficient mice, intestinal epithelial cell line IEC-6 | Inhibition of IFNγ-induced phosphorylation and function of STAT1; inhibition of iNOS and NF-κB upregulation; inhibition of transcription and secretion of TNFα and IFNγ | [33,34,35] |
| GTPs | MDAmb231, MDAmb468, MCF7, Hela, HepG2 | Inhibition of IFNγ-induced phosphorylation and function of STAT1 | [35] |
| GTPs | Piglets | Elevation of antiinflammatory cytokine IL-4; inhibition of the ratio of IFNγ/IL-4; stimulation of immune cell proliferation and T lymphocytes; elevation of CD4+ T cell percentage and the ratio of CD4+/CD8+; improvement of LTT | [36] |
| GTPs | Lymphocytes | Inhibition of IFNγ secretion; stimulation of T cells and APCs apoptosis; inhibition of T cell division and cycle progression in a dose-dependent manner | [38] |
| GTPs | T lymphocytes | influencing expression of T-cell receptor complex and antigen-specific T-cell responses | [39] |
| EGCG | Spleen cells isolated from C57BL mice | Inhibition ofsplenocyte proliferation, T cell division, and cell cycle progression | [40] |
| EGCG | murine DCs | Inhibition of MAPK and NF-kB | [30] |
| EGCG | MPTP-induced Parkinson’s disease mouse | Increase of the ratio of CD3+CD4+ to CD3+CD8+ T lymphocytes; modulating peripheral immune response | [42] |
| EGCG | Macrophages and neutrophils | Inhibition of metallo-elastase and serine-elastase | [43,44] |
| EGCG | Human umbilical vein endothelial cells | Inhibition of neutrophil transmigration | [45] |
| EGCG | Transgenic zebra fish | Inhibition of neutrophil response (accumulation, travel speed, and distance) expression of IL-1β and TNFα, as well as related signaling pathways | [46] |
| EGCG | Inflammatory angiogenesis model, pulmonary inflammation model | Apoptosis inhibition of activated neutrophils and chemokine-induced neutrophil chemotaxis; inhibition of neutrophil elastase, ROS activity, neutrophil-mediated angiogenesis, and fibrosis | [47] |
| GTPs | Macrophages | Inhibition of TNF-α induction and NF-kB activation | [48] |
| EGCG | peritoneal macrophages | Inhibition of LPS-stimulated NO production, iNOS expression, and NF-kB activities | [49] |
| EGCG | Human peripheral blood mononuclear cells | Inducement of neopterin production | [50] |
| Tea polyphenols | BALB/c mice | increase of antibody-secreted cells in spleen and IgM-mediated IgG-mediated immune response | [51] |
| GTPs | Mice | Decrease of total IgG and type II collagen-specific IgG levels in serum and arthritic joints, as well as the neutral endopeptidase activity | [52] |
| EGCG | Mouse | Enhancement of PFC response to sheep red blood cells, strong mitogenic activity towards splenic B-cells | [53] |
| GTPs | Wuchang bream juveniles | Elevation of content and mRNA levels of splenic IL-1β, TNFα and IgM | [54] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, S.; Li, Z.; Ma, Y.; Liu, Y.; Lin, C.-C.; Li, S.; Zhan, J.; Ho, C.-T. Immunomodulatory Effects of Green Tea Polyphenols. Molecules 2021, 26, 3755. https://doi.org/10.3390/molecules26123755
Wang S, Li Z, Ma Y, Liu Y, Lin C-C, Li S, Zhan J, Ho C-T. Immunomodulatory Effects of Green Tea Polyphenols. Molecules. 2021; 26(12):3755. https://doi.org/10.3390/molecules26123755
Chicago/Turabian StyleWang, Shuzhen, Zhiliang Li, Yuting Ma, Yan Liu, Chi-Chen Lin, Shiming Li, Jianfeng Zhan, and Chi-Tang Ho. 2021. "Immunomodulatory Effects of Green Tea Polyphenols" Molecules 26, no. 12: 3755. https://doi.org/10.3390/molecules26123755
APA StyleWang, S., Li, Z., Ma, Y., Liu, Y., Lin, C.-C., Li, S., Zhan, J., & Ho, C.-T. (2021). Immunomodulatory Effects of Green Tea Polyphenols. Molecules, 26(12), 3755. https://doi.org/10.3390/molecules26123755








