Effect of Microwave and Conventional Modes of Heating on Sintering Behavior, Microstructural Evolution and Mechanical Properties of Al-Cu-Mn Alloys
Abstract
1. Introduction
2. Materials and Methods
3. Results & Discussion
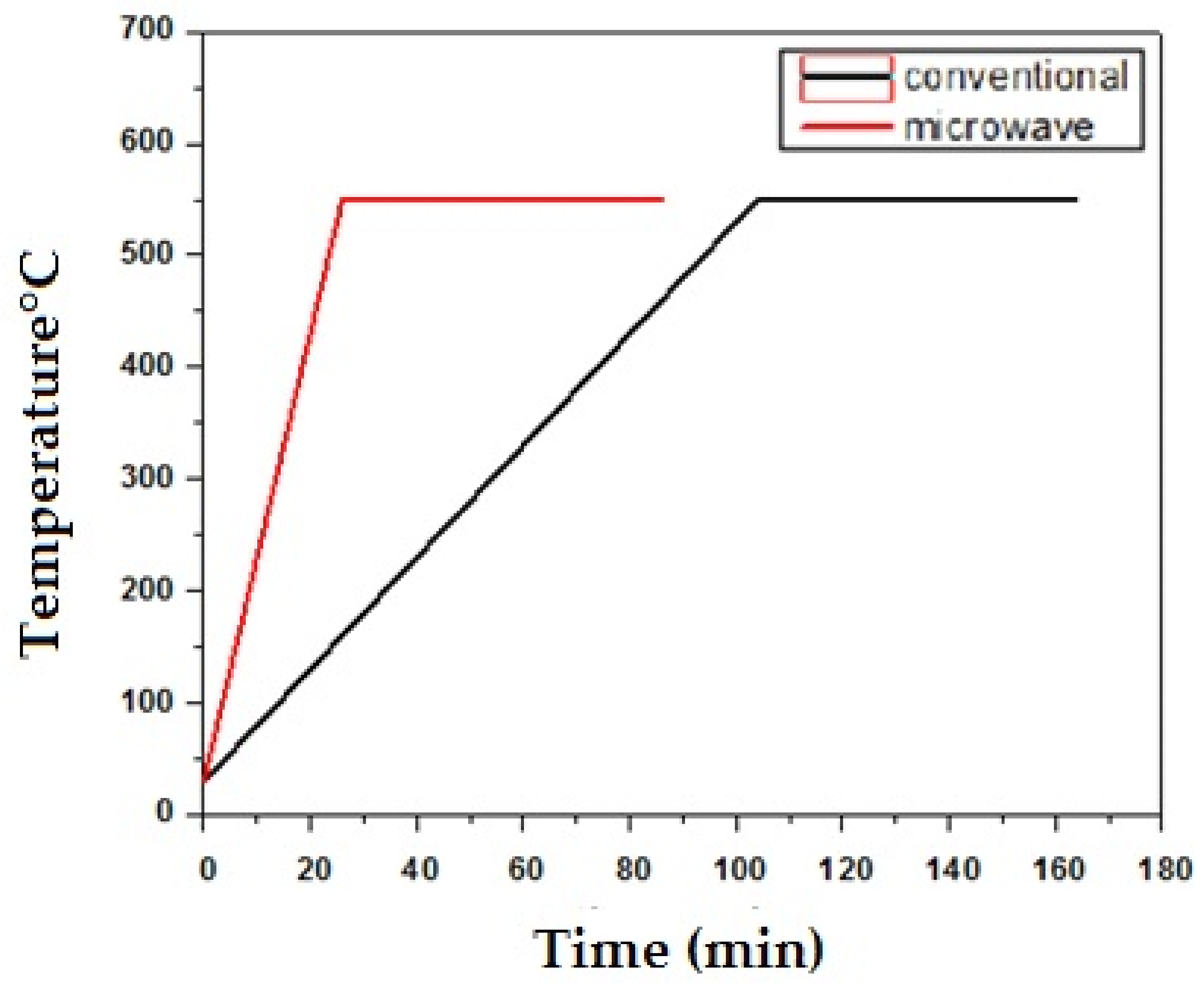
3.1. Densification Response
3.2. Micro–Hardness
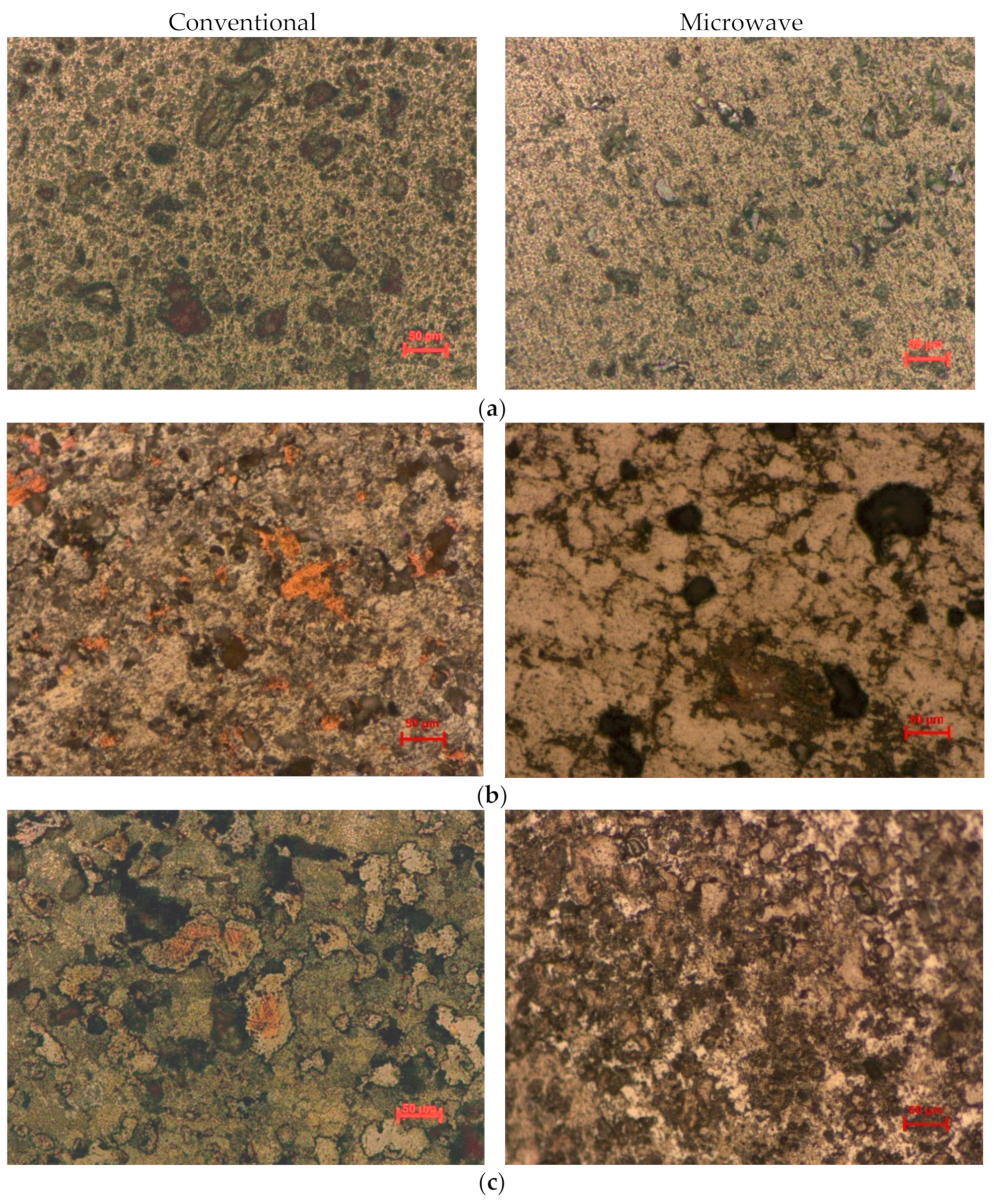
3.3. Microstructural Results
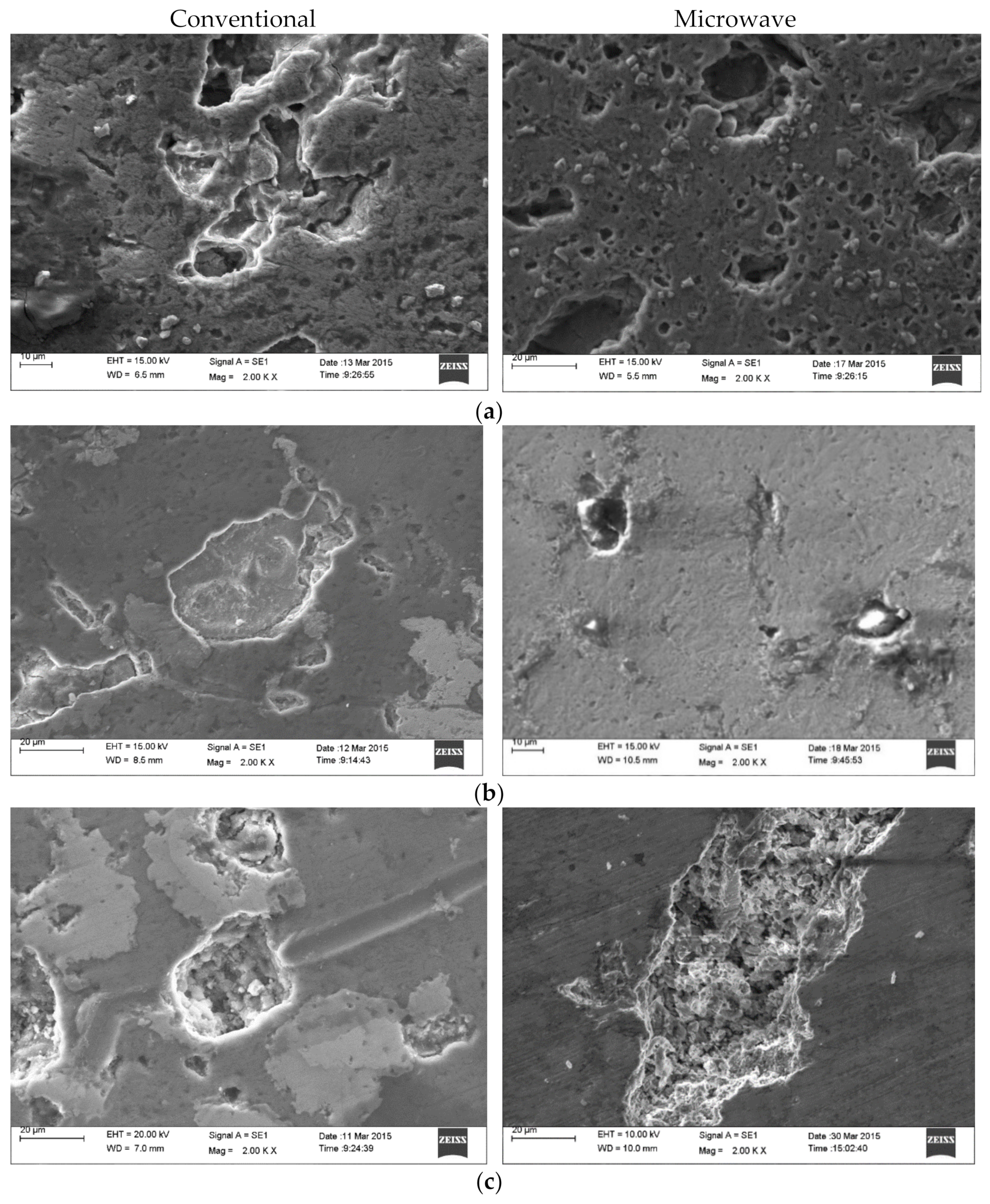
3.4. Scanning Electron Microscopy
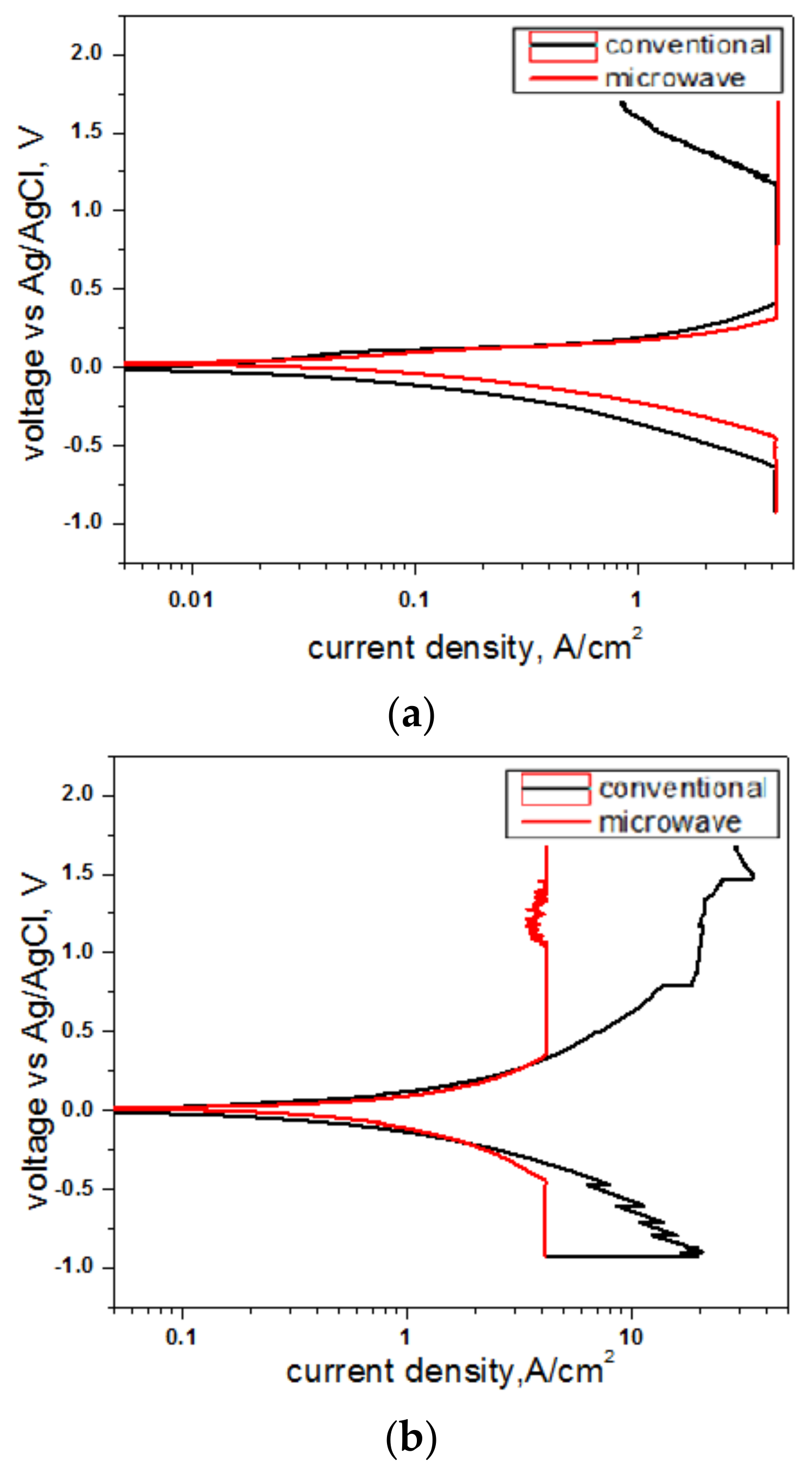
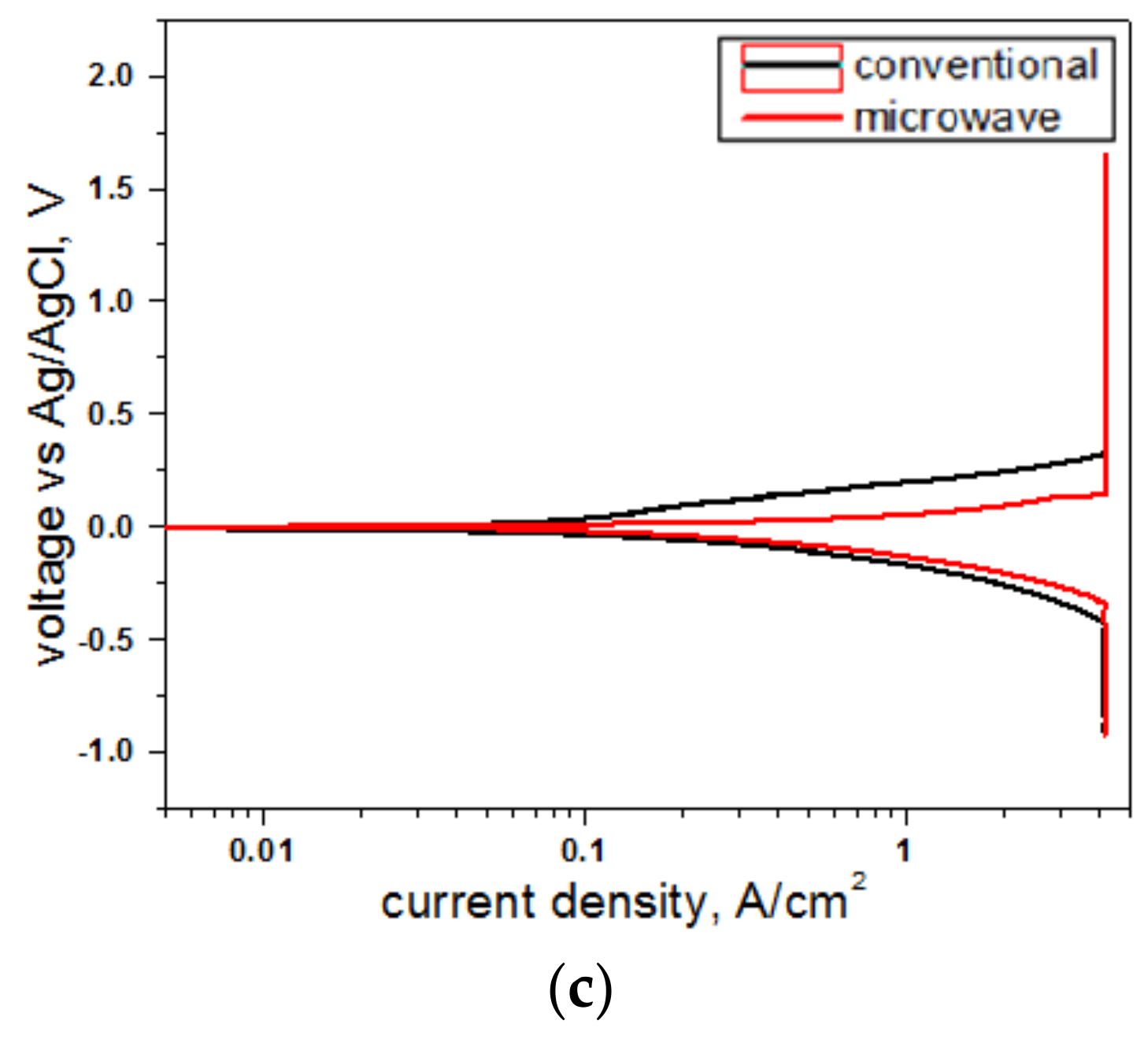
3.5. Electrochemical Study
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Cobden, R.; Banbury, A. Aluminum: Physical Properties, Characteristics, and Alloys. Talat Lect. 1994, 1501, 144–155. [Google Scholar]
- Zheng, R.R.; Wu, Y.; Liao, S.L.; Wang, W.Y.; Wang, W.B.; Wang, A.H. Microstructure and mechanical properties of Al/(Ti,W)C composites prepared by microwave sintering. J. Alloys Compd. 2014, 590, 168–175. [Google Scholar] [CrossRef]
- Lall, C.; Heath, W. P/M aluminium structural parts -manufacturing and metallurgical fundamentals. Int. J. Powder Met. 2000, 36, 45–50. [Google Scholar]
- Kumar, M.G.A.; Seetharamua, S.; Nayak, J.; Satapathy, L.N. A Studyon Thermal Behavior of Aluminum Cenosphere Powder Metallurgy Composites Sinteredin Microwave. Procedia Mater. Sci. 2014, 5, 1066–1074. [Google Scholar] [CrossRef]
- Surappa, M.K. Aluminummatrixcomposites: Challengesandopportunities. Sadhana 2003, 28, 319–334. [Google Scholar] [CrossRef]
- Sever, S.; Liviu, R. Introduction to Powder Metallurgy. In Proceedings of the “ConstantinBrâncusi” University, 8th International Conference, Targu Jiu, Romania, 24 May 2002. [Google Scholar]
- Groover, M.P. Fundamentals of Modern Manufacturing; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2002. [Google Scholar]
- Mascarenhas, J.; Marcelo, T.; Inverno, A.; Castanho, J.; Vieira, T. Microwave Sintering of Sputter Coated 316l Powders. In Proceedings of the Euro PM2008 International Powder Metallurgy Congress & Exhibition, Mannheim, Germany, 29 September–1 October 2008; pp. 21–26. [Google Scholar]
- Oghbaei, M.; Mirzaee, O. Microwave versus conventional sintering: A review of fundamentals, advantages, and applications. J. Alloys Compd. 2010, 494, 175–189. [Google Scholar] [CrossRef]
- Mahmoud, M.M.; Link, G.; Thumm, M. The role of the native oxide shellon the microwaves intering of copper metal powder compacts. J. Alloys Compd. 2015, 627, 231–237. [Google Scholar] [CrossRef]
- Muthuchamy, A.; Annamalai, A.R.; Acharyya, S.G.; Nagaraju, N.; Agrawal, D.K. Microstructural and Electrochemical behaviour of Aluminium Alloy Composites Produced Using Different Sintering Techniques. Mat. Res. 2018, 21. [Google Scholar] [CrossRef]
- Chen, B.; Wang, R.; Peng, C.; Feng, Y.; Wang, N. Influence of Al_Mn master alloys on microstructures an delect rochemical properties of Mg_Al_Pb_Mn alloys. Trans. Nonferrous Met. Soc. China 2014, 24, 423–430. [Google Scholar] [CrossRef]
- Khvan, A.V.; Cheverikin, V.V.; Dinsdale, A.T.; Watson, A.; Levchenko, V.V.; Zolotorevskiy, V.S. Formation of metastable phases during solidification of Al–3.2wt%Mn. J. Alloys Compd. 2015, 622, 223–228. [Google Scholar] [CrossRef]
- Udomphol, T. Aluminum and Its Alloys; Suranaree University of Technology: Nakhon Ratchasima, Thailand, 2007. [Google Scholar]
- Wolla, D.W.; Davidson, M.J.; Khanra, A.K. Studies on the formability of powder metallurgical aluminum–copper composite. Mater. Des. 2014, 59, 151–159. [Google Scholar] [CrossRef]
- Wang, Y.; Rainforth, W.M.; Jones, H.; Lieblich, M. Dry wear behavior and its relationto microstructure of novel 6092 Aluminum alloy–Ni3 Al powder metallurgy composite. Wear 2001, 251, 1421–1432. [Google Scholar] [CrossRef]
- Voort, V.; George, F. Metallography, Principles and Practice; ASM International: Almere, The Netherlands, 1999; p. 197. ISBN 978-0-87170-672-0. [Google Scholar]
- Liu, G.; Zhang, G.J.; Wang, R.H.; Hu, W.; Sun, J.; Chen, K.H. Heat treatment-modulated coupling effect of multi-scale second-phase particles on the ductile fracture of aged aluminum alloys. Acta Mater. 2007, 55, 273–284. [Google Scholar] [CrossRef]
- Padmavathi, C.; Upadhyaya, A.; Agrawal, D. Effect of Microwave and conventional heating on sintering behavior and properties of Al-Mg-Si–Cu alloy. Mater. Chem. Phys. 2011, 130, 449–457. [Google Scholar] [CrossRef]
- Ghazali, M.J.; Rainforth, W.M.; Jones, H. Dry sliding wear behavior of some wrought, rapidly solidified powder metallurgy Aluminum alloys. Wear 2005, 259, 490–500. [Google Scholar] [CrossRef]
- Kloc, L.; Cerri, E.; Spigarelli, S.; Evangelista, E.; Terence, G.; Langdon, B. Significance of continuous precipitation during creep of a powder metallurgy aluminum alloy. Mater. Sci. Eng. A 1996, 216, 161–168. [Google Scholar] [CrossRef]
- Ellner, M. Crystal chemical parameters of the liquid-quenched MnAl0.8. J. Alloys Compd. 2006, 422, 192–193. [Google Scholar] [CrossRef]
- George, F. Chromium-Free Conversion Coating of Aluminum-Copper Alloys. Ph.D. Thesis, The University of Manchester, Manchester, UK, 2010. [Google Scholar]
- Namahoot, J. Effect of Deformation on Corrosion of Al-Mn Alloys. Ph.D. Thesis, The University of Birmingham, Birmingham, UK, 2004. [Google Scholar]
| Sample No. | Aluminum Wt% * | Copper Wt% * | Manganese Wt% * |
|---|---|---|---|
| 1 | 77.5 | 0 | 22.5 |
| 2 | 57.28 | 18.6 | 24.12 |
| 3 | 65 | 20 | 15 |
| Cumulative Powder Size | μm |
|---|---|
| D10 | 12.6 |
| D50 | 31.0 |
| D90 | 44.2 |
| Composition | Sintering Mode | Radial Shrinkage | Axial Shrinkage | % wt Loss | Green Density (% Theoretical) | Sinter Density (% Theoretical) | Densification Parameter |
|---|---|---|---|---|---|---|---|
| 77.5Al-22.5Mn | Conventional | 0.001 | 0.533 | 0.072 | 78.8 | 80.77 | 0.029 |
| Microwave | 0.059 | 2.967 | 0.297 | 80.19 | 81.54 | 0.125 | |
| 57.28Al-18.6Cu-24.12Mn | Conventional | 0.039 | 0.125 | 0.34 | 71.12 | 71.51 | 0.013 |
| Microwave | 0.078 | 0.152 | 3.721 | 70.4 | 73.30 | 0.096 | |
| 65Al-20Cu-15Mn | Conventional | 0.023 | 0.035 | 0.097 | 71.94 | 72.06 | 0.004 |
| Microwave | 0.551 | 4.189 | 0.194 | 71.4 | 75.40 | 0.138 |
| Composition | Conventional | Microwave |
|---|---|---|
| 77.5Al-22.5Mn | 52.67 ± 5 Hv | 86.65 ± 5 Hv |
| 57.28Al-18.6Cu-24.12Mn | 94.46 ± 10 Hv | 745.8 ± 10 Hv |
| 65Al-20Cu-15Mn | 114.03 ± 5 Hv | 580.37 ± 10 Hv |
| Sample | Al-22.5%Mn | Al-18.6%Cu-24.12%Mn | Al-20%Cu-15%Mn | |||
|---|---|---|---|---|---|---|
| Conventional | Microwave | Conventional | Microwave | Conventional | Microwave | |
| Al wt % | 97.65 | 98.16 | 81.98 | 54.70 | 39.52 | 60.57 |
| Cu wt % | 0 | 0 | 1.79 | 22.73 | 13.99 | 20.78 |
| Mn wt % | 2.35 | 1.84 | 16.22 | 22.56 | 46.48 | 18.63 |
| Composition | Sintering Mode | Icorr | Ecorr | Corrosion Rate |
|---|---|---|---|---|
| (mA/cm2) | (V) | (mpy) | ||
| 77.5Al-22.5Mn | Conventional | 1.135 | 0.003 | 1.407 |
| Microwave | 1.103 | −0.034 | 1.355 | |
| 57.28Al-18.6Cu-24.12Mn | Conventional | 2.171 | 0.005 | 2.398 |
| Microwave | 1.672 | −0.14 | 1.803 | |
| 65Al-20Cu-15Mn | Conventional | 1.29 | 0.019 | 1.276 |
| Microwave | 1.164 | −0.002 | 1.101 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Muthuchamy, A.; Srikanth, M.; Agrawal, D.K.; Annamalai, A.R. Effect of Microwave and Conventional Modes of Heating on Sintering Behavior, Microstructural Evolution and Mechanical Properties of Al-Cu-Mn Alloys. Molecules 2021, 26, 3675. https://doi.org/10.3390/molecules26123675
Muthuchamy A, Srikanth M, Agrawal DK, Annamalai AR. Effect of Microwave and Conventional Modes of Heating on Sintering Behavior, Microstructural Evolution and Mechanical Properties of Al-Cu-Mn Alloys. Molecules. 2021; 26(12):3675. https://doi.org/10.3390/molecules26123675
Chicago/Turabian StyleMuthuchamy, A., Muthe Srikanth, Dinesh K. Agrawal, and A. Raja Annamalai. 2021. "Effect of Microwave and Conventional Modes of Heating on Sintering Behavior, Microstructural Evolution and Mechanical Properties of Al-Cu-Mn Alloys" Molecules 26, no. 12: 3675. https://doi.org/10.3390/molecules26123675
APA StyleMuthuchamy, A., Srikanth, M., Agrawal, D. K., & Annamalai, A. R. (2021). Effect of Microwave and Conventional Modes of Heating on Sintering Behavior, Microstructural Evolution and Mechanical Properties of Al-Cu-Mn Alloys. Molecules, 26(12), 3675. https://doi.org/10.3390/molecules26123675








