Antioxidative and Anti-Inflammatory Phytochemicals and Related Stable Paramagnetic Species in Different Parts of Dragon Fruit
Abstract
1. Introduction
2. Results and Discussion
2.1. TPC, TFC, and TAC Content
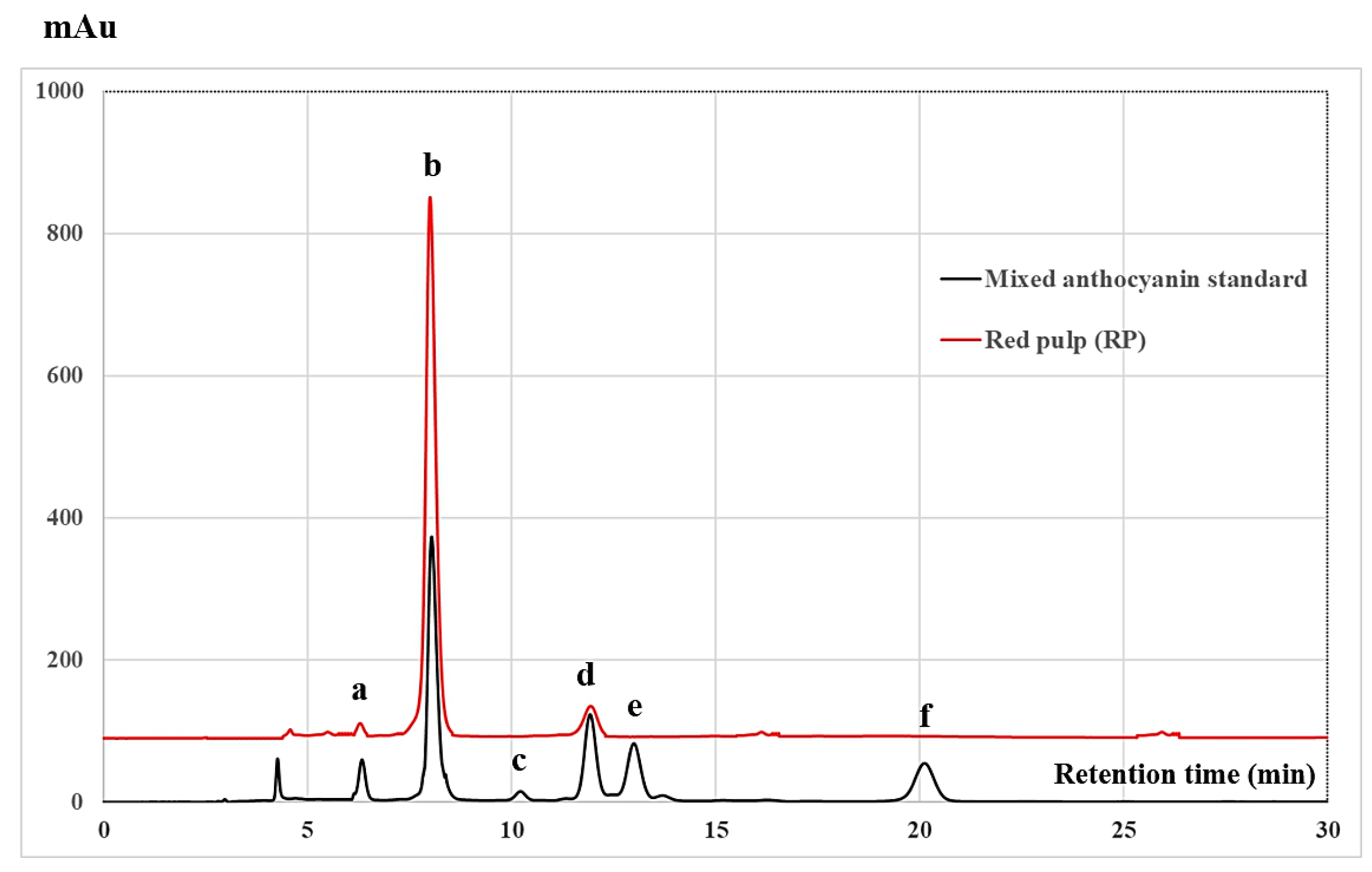
2.2. Chromatographic Analysis of Antioxidative Phytochemicals
2.3. Determination of the Inhibitory Effects on Intracellular ROS and RNS Production
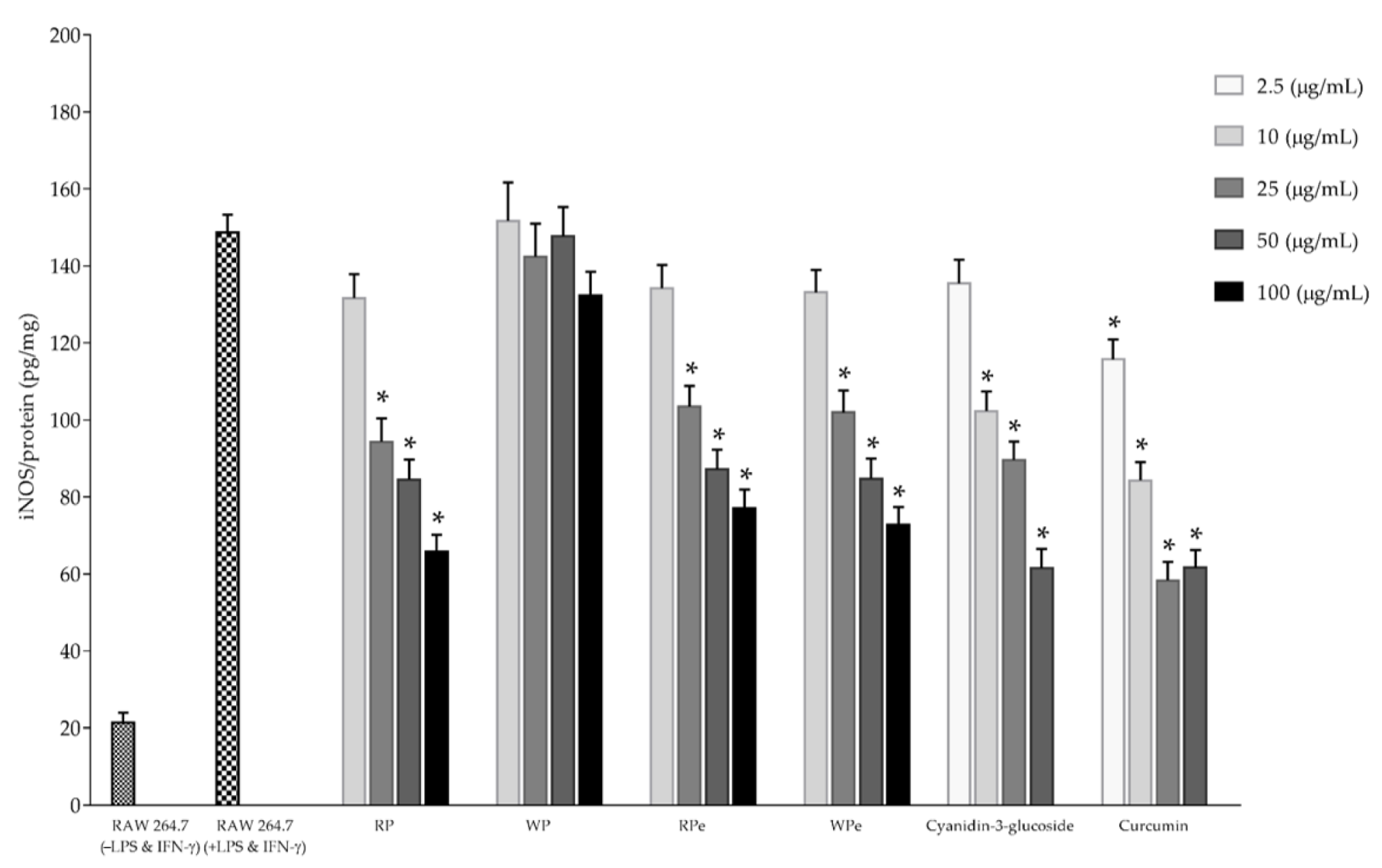
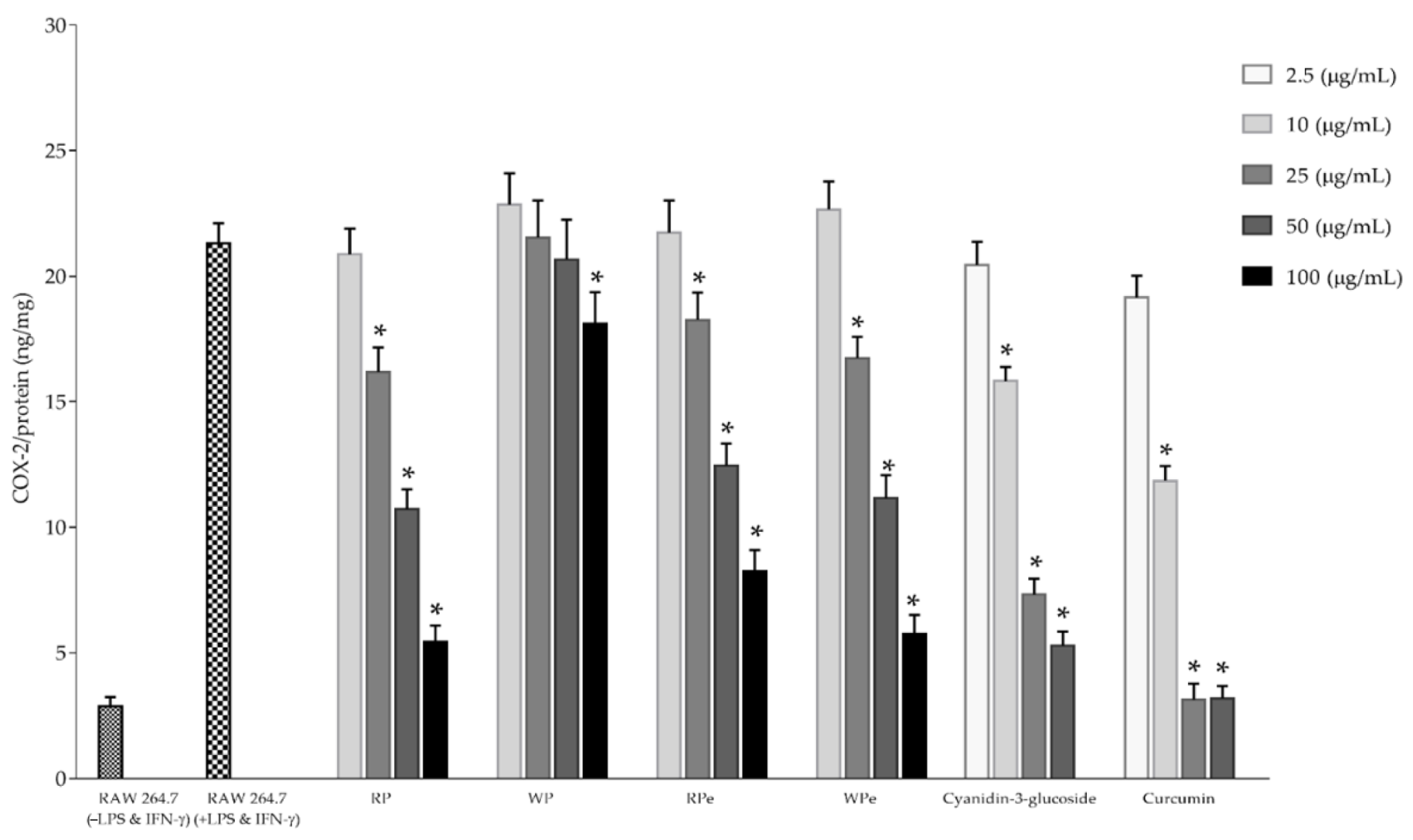
2.4. Determination of Anti-Inflammatory Activities
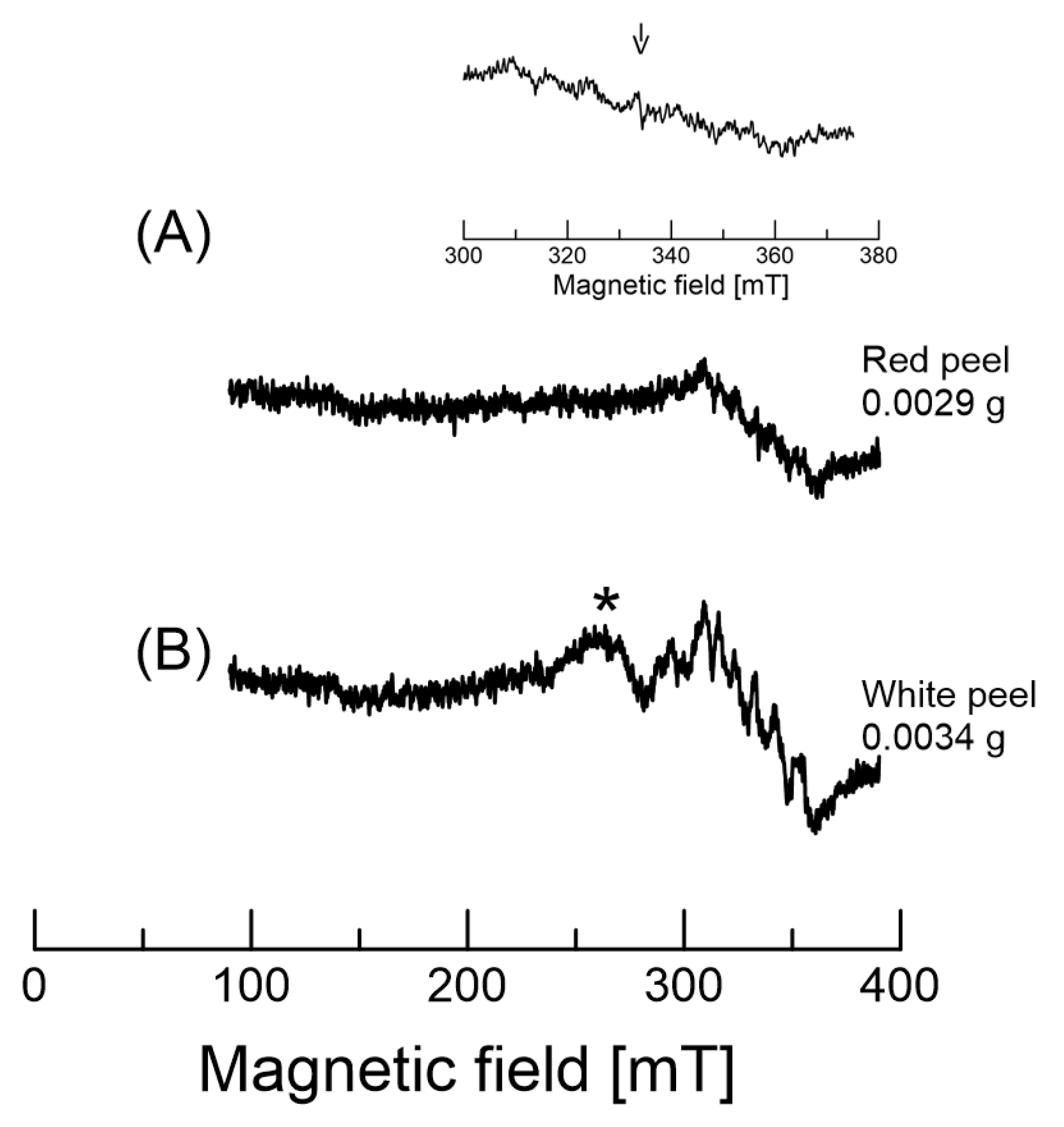
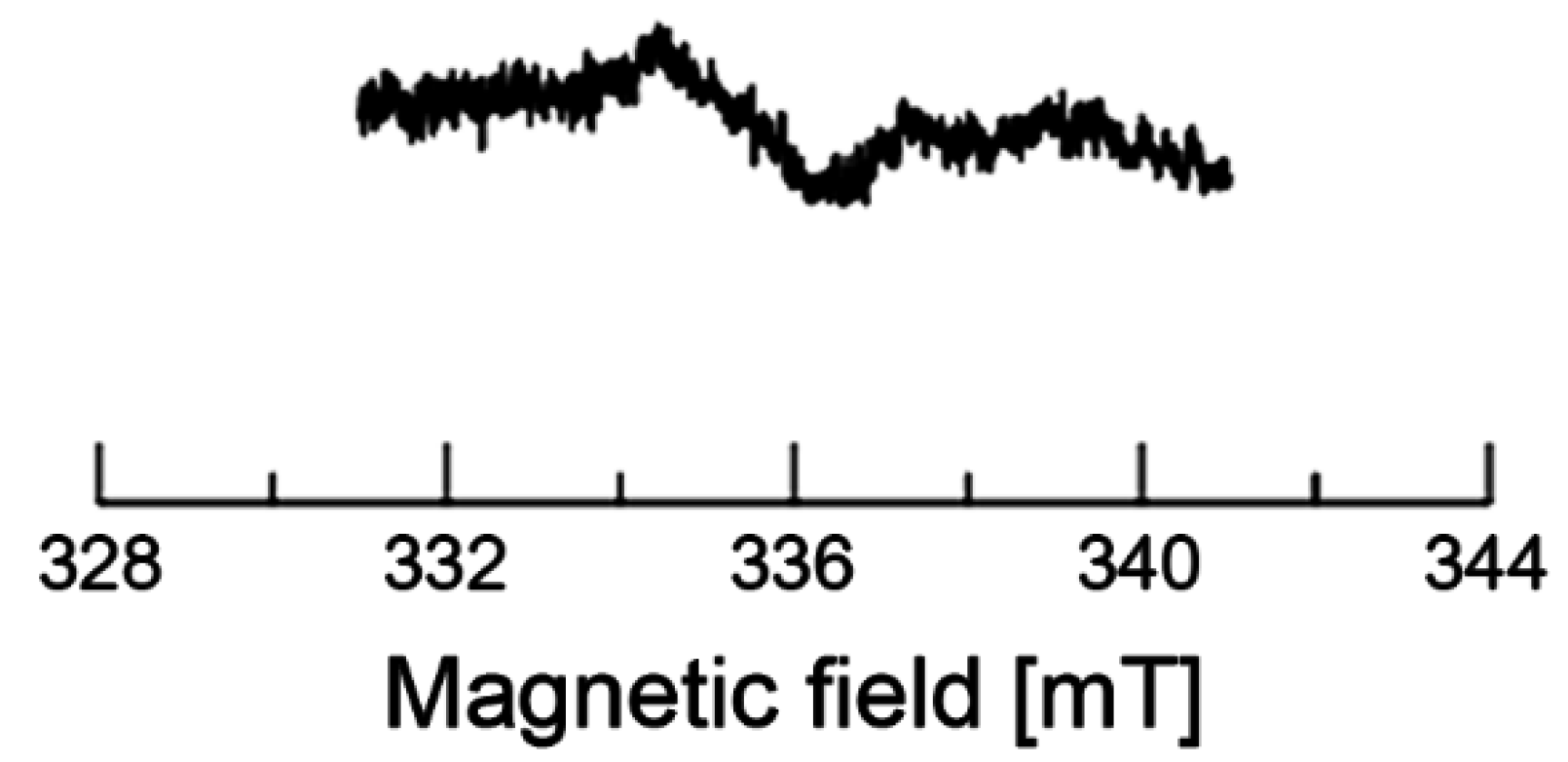
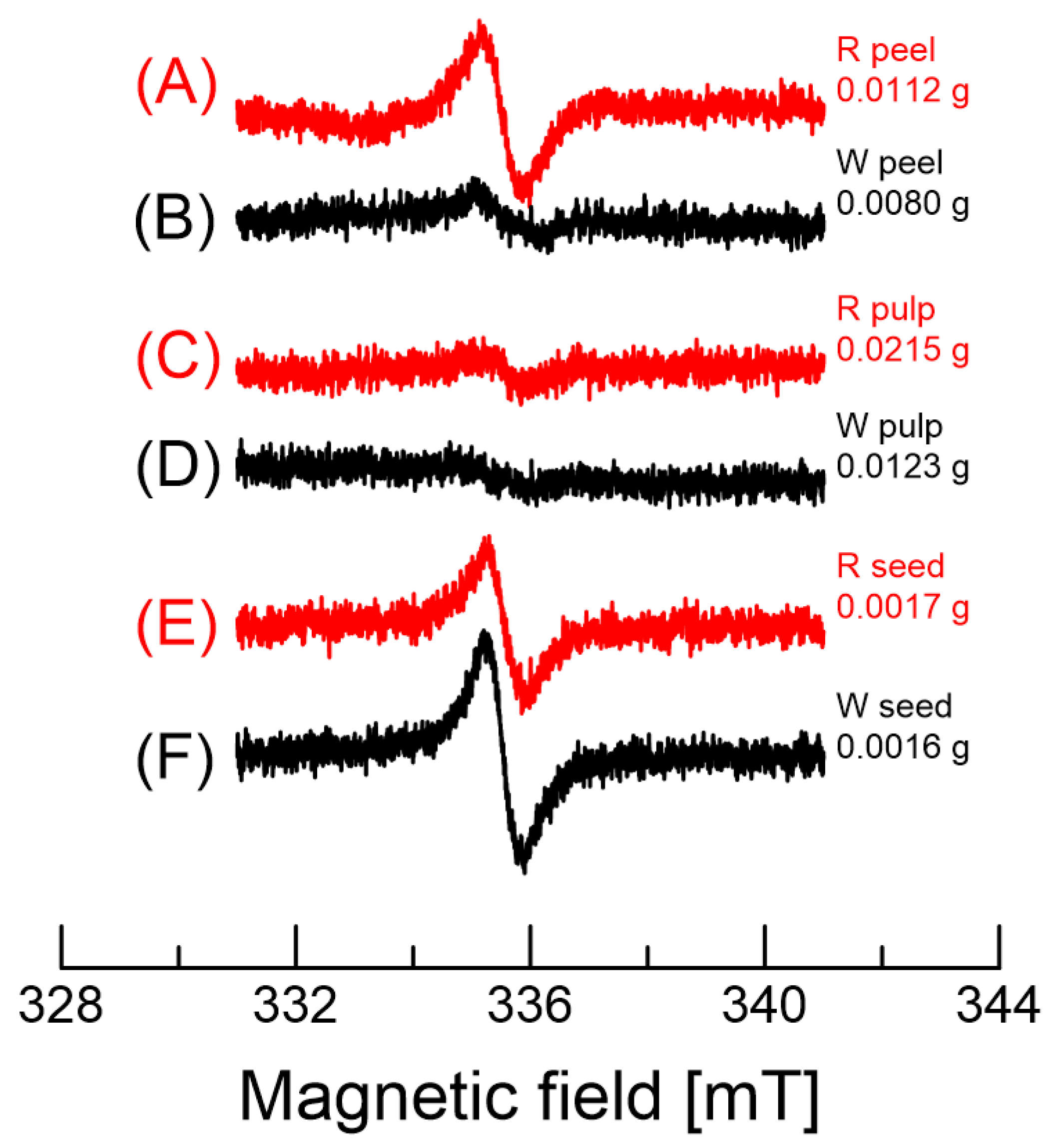
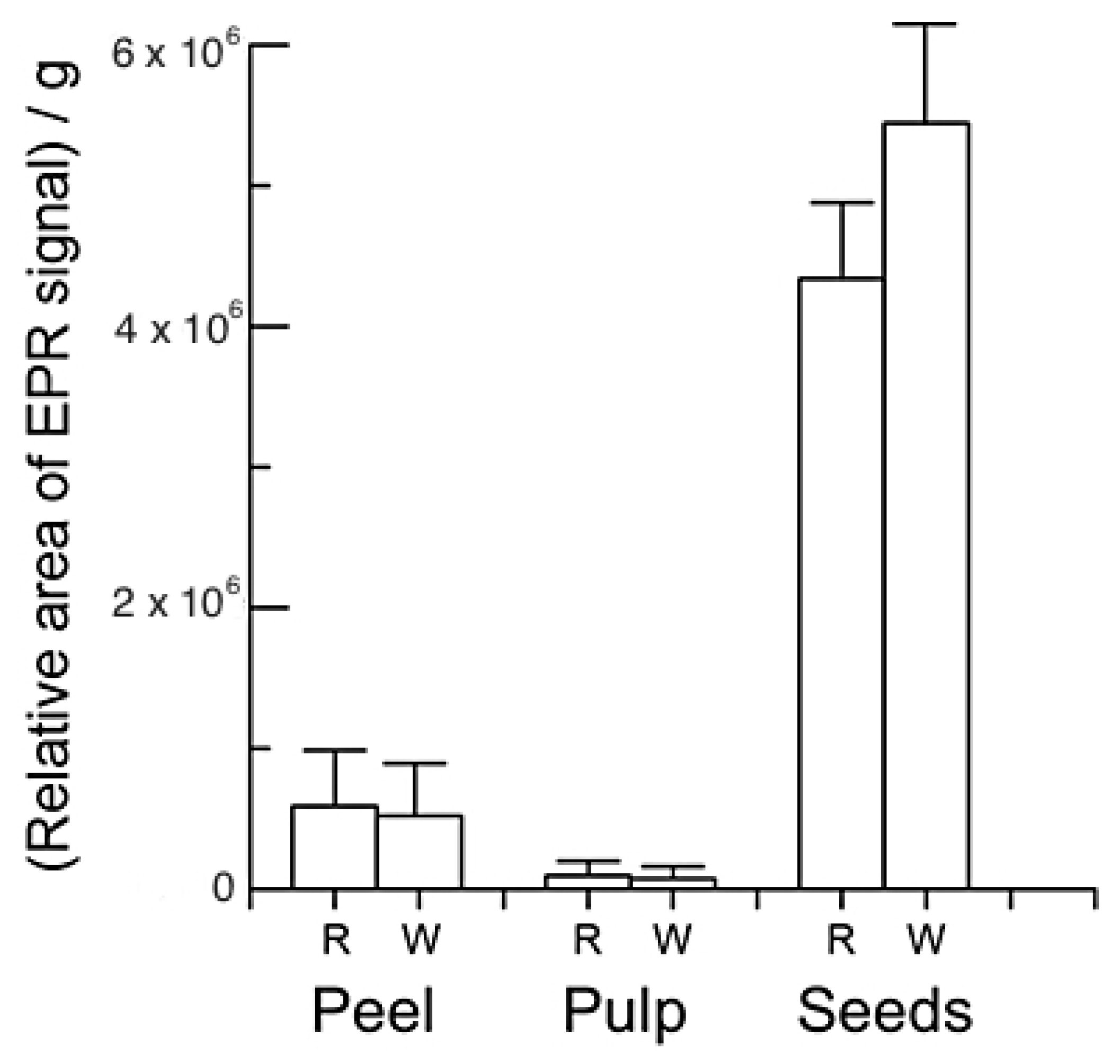
2.5. EPR of Dragon Fruits
3. Materials and Methods
3.1. Samples Preparation
3.2. Total Phenolic Measurement
3.3. Total Flavonoid Measurement
3.4. Total Anthocyanin Measurement
3.5. Chromatographic Analysis of Anthocyanins
3.6. Chromatographic Analysis of Catechin and Related Compounds
3.7. Determination on Inhibition Effect on Intracellular Reactive Oxygen Species (ROS) Production
3.8. Determination of the Inhibitory Effect on Intracellular Reactive Nitrogen Species (RNS) Production and Anti-Inflammatory Activities
3.9. EPR Measurements
3.10. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Choo, W.-S.; Yong, W. Antioxidant properties of two species of Hylocereus fruits. Adv. Appl. Sci. Res. 2011, 2, 418–425. [Google Scholar]
- De Mello, F.R.; Bernardo, C.; Dias, C.O.; Gonzaga, L.; Amante, E.R.; Fett, R.; Candido, L.M.B. Antioxidant properties, quantification and stability of betalains from pitaya (Hylocereus undatus) peel. Ciência Rural 2015, 45, 323–328. [Google Scholar] [CrossRef]
- Al-Mekhlafi, N.A.; Mediani, A.; Ismail, N.H.; Abas, F.; Dymerski, T.; Lubinska-Szczygeł, M.; Vearasilp, S.; Gorinstein, S. Metabolomic and antioxidant properties of different varieties and origins of dragon fruit. Microchem. J. 2021, 160, 105687. [Google Scholar] [CrossRef]
- Luo, H.; Cai, Y.; Peng, Z.; Liu, T.; Yang, S. Chemical composition and in vitroevaluation of the cytotoxic and antioxidant activities of supercritical carbon dioxide extracts of pitaya (dragon fruit) peel. Chem. Cent. J. 2014, 8, 1. [Google Scholar] [CrossRef] [PubMed]
- Arivalagan, M.; Karunakaran, G.; Roy, T.K.; Dinsha, M.; Sindhu, B.C.; Shilpashree, V.M.; Satisha, G.C.; Shivashankara, K.S. Biochemical and nutritional characterization of dragon fruit (Hylocereus species). Food Chem. 2021, 353, 129426. [Google Scholar] [CrossRef]
- Paśko, P.; Galanty, A.; Zagrodzki, P.; Luksirikul, P.; Barasch, D.; Nemirovski, A.; Gorinstein, S. Dragon fruits as a reservoir of natural polyphenolics with chemopreventive properties. Molecules 2021, 26, 2158. [Google Scholar] [CrossRef]
- Barber, T.M.; Kabisch, S.; Pfeiffer, A.F.H.; Weickert, M.O. The health benefits of dietary fibre. Nutrients 2020, 12, 3209. [Google Scholar] [CrossRef]
- Zhang, F.F.; Barr, S.I.; McNulty, H.; Li, D.; Blumberg, J.B. Health effects of vitamin and mineral supplements. BMJ (Clin. Res. Ed.) 2020, 369, m2511. [Google Scholar] [CrossRef]
- Wybraniec, S.; Mizrahi, Y. Fruit flesh betacyanin pigments in Hylocereus cacti. J. Agric. Food Chem. 2002, 50, 6086–6089. [Google Scholar] [CrossRef]
- Ding, P.; Chew, M.; Aziz, S.; Lai, O.; Ong Abdullah, J. Red-fleshed pitaya (Hylocereus polyrhizus) fruit colour and betacyanin content depend on maturity. Int. Food Res. J. 2009, 16, 233–242. [Google Scholar]
- Yee, P.; Wah, C. Application of red pitaya powder as a natural food colourant in fruit pastille. J. Gizi Klin. Indones. 2017, 13, 111–120. [Google Scholar] [CrossRef]
- Adnan, L.; Osman, A.; Abdul Hamid, A. Antioxidant activity of different extracts of red pitaya (Hylocereus polyrhizus) seed. Int. J. Food Prop. 2011, 14, 1171–1181. [Google Scholar] [CrossRef]
- Vargas, M.; Cortez, J.; Sauri, E.; Lizama, A.; Mendez, C. Extraction and stability of anthocyanins present in the skin of the dragon fruit (Hylocereus undatus). Food Sci. Nutr. 2013, 4, 1221–1228. [Google Scholar]
- Nakagawa, K.; Hara, H. Investigation of radical locations in various sesame seeds by CW EPR and 9-GHz EPR imaging. Free Radic. Res. 2015, 49, 1–6. [Google Scholar] [CrossRef]
- Nakagawa, K.; Hara, H. X-band electron paramagnetic resonance investigation of stable organic radicals present under cold stratification in ‘Fuji’ apple seeds. J. Oleo Sci. 2017, 66, 1375–1379. [Google Scholar] [CrossRef]
- Wojdyło, A.; Oszmiański, J.; Czemerys, R. Antioxidant activity and phenolic compounds in 32 selected herbs. Food Chem. 2007, 105, 940–949. [Google Scholar] [CrossRef]
- Paixão, N.; Perestrelo, R.; Marques, J.C.; Câmara, J.S. Relationship between antioxidant capacity and total phenolic content of red, rosé and white wines. Food Chem. 2007, 105, 204–214. [Google Scholar] [CrossRef]
- Nguyen, N.M.P.; Le, T.T.; Vissenaekens, H.; Gonzales, G.B.; Van Camp, J.; Smagghe, G.; Raes, K. In vitro antioxidant activity and phenolic profiles of tropical fruit by-products. Int. J. Food Sci. Technol. 2019, 54, 1169–1178. [Google Scholar] [CrossRef]
- Nurliyana, R.; Zahir, I.; Suleiman, K.; Aisyah, M.R.; Kamarudin, K.R. Antioxidant study of pulps and peels of dragon fruits: A comparative study. Int. Food Res. J. 2010, 17, 367–375. [Google Scholar]
- Hernandez-Ramos, L.; García-Mateos, R.; Castillo-González, A.M.; Ybarra-Moncada, M.C.; Nieto-Angel, R. Fruits of the pitahaya Hylocereus undatus and H. ocamponis: Nutritional components and antioxidants. J. Appl. Bot. Food Qual. 2020, 93, 197–203. [Google Scholar]
- Prabowo, I.; Utomo, E.P.; Nurfaizy, A.; Widodo, A.; Widjajanto, E.; Rahadju, P. Characteristics and antioxidant activities of anthocyanin fraction in red dragon fruit peels (Hylocereus polyrhizus) extract. Drug Invent. Today 2019, 12, 670–678. [Google Scholar]
- Fan, R.; Sun, Q.; Zeng, J.; Zhang, X. Contribution of anthocyanin pathways to fruit flesh coloration in pitayas. BMC Plant Biol. 2020, 20, 361. [Google Scholar] [CrossRef]
- Mahli, Y.; Husmaini, H.; Warnita, W.; Mirzah, M.; Kobayashi, M.; Mahata, M.E. The processing effects of anthocyanins extracted from dragon fruit (Hylocereus polyrhizus) peel on total amount of anthocyanins and SEM image in poultry nutrition. J. World Poult. Res. 2020, 10, 513–519. [Google Scholar] [CrossRef]
- Giangrieco, I.; Proietti, S.; Moscatello, S.; Tuppo, L.; Battistelli, A.; La Cara, F.; Tamburrini, M.; Famiani, F.; Ciardiello, M.A. Influence of geographical location of orchards on green kiwifruit bioactive components. J. Agric. Food Chem. 2016, 64, 9172–9179. [Google Scholar] [CrossRef] [PubMed]
- Paśko, P.; Tyszka-Czochara, M.; Trojan, S.; Bobis-Wozowicz, S.; Zagrodzki, P.; Namieśnik, J.; Haruenkit, R.; Poovarodom, S.; Pinsirodom, P.; Gorinstein, S. Glycolytic genes expression, proapoptotic potential in relation to the total content of bioactive compounds in durian fruits. Food Res. Int. 2019, 125, 108563. [Google Scholar] [CrossRef]
- Aryal, S.; Baniya, M.K.; Danekhu, K.; Kunwar, P.; Gurung, R.; Koirala, N. Total phenolic content, flavonoid content and antioxidant potential of wild vegetables from western Nepal. Plants 2019, 8, 96. [Google Scholar] [CrossRef] [PubMed]
- Kiokias, S.; Proestos, C.; Oreopoulou, V. Phenolic Acids of Plant Origin-A Review on Their Antioxidant Activity In Vitro (O/W Emulsion Systems) Along with Their in Vivo Health Biochemical Properties. Foods 2020, 9, 534. [Google Scholar] [CrossRef]
- Wang, W.; Goodman, M.T. Antioxidant property of dietary phenolic agents in a human LDL-oxidation ex vivo model: Interaction of protein binding activity. Nutr. Res. 1999, 19, 191–202. [Google Scholar] [CrossRef]
- Kuskoski, E.M.; Asuero, A.G.; García-Parilla, M.C.; Troncoso, A.M.; Fett, R. Actividad antioxidante de pigmentos antociánicos. Food Sci. Technol. 2004, 24, 691–693. [Google Scholar] [CrossRef]
- Ali, H.M.; Almagribi, W.; Al-Rashidi, M.N. Antiradical and reductant activities of anthocyanidins and anthocyanins, structure-activity relationship and synthesis. Food Chem. 2016, 194, 1275–1282. [Google Scholar] [CrossRef]
- Jung, H.; Kwak, H.-K.; Hwang, K.T. Antioxidant and antiinflammatory activities of cyanidin-3-glucoside and cyanidin-3-rutinoside in hydrogen peroxide and lipopolysaccharide-treated RAW264.7 cells. Food Sci. Biotechnol. 2014, 23, 2053–2062. [Google Scholar] [CrossRef]
- Limtrakul, P.; Yodkeeree, S.; Pitchakarn, P.; Punfa, W. Suppression of inflammatory responses by black rice extract in RAW 264.7 macrophage cells via downregulation of NF-kB and AP-1 signaling pathways. Asian Pac. J. Cancer Prev. 2015, 16, 4277–4283. [Google Scholar] [CrossRef] [PubMed]
- Ferrari, D.; Speciale, A.; Cristani, M.; Fratantonio, D.; Molonia, M.S.; Ranaldi, G.; Saija, A.; Cimino, F. Cyanidin-3-O-glucoside inhibits NF-¦ÊB signalling in intestinal epithelial cells exposed to TNF-¦Á and exert protective effects via Nrf2 pathway activation. Toxicol. Lett. 2016, 264, 51–58. [Google Scholar] [CrossRef]
- Weil, J.A.; Bolton, J.R.; Wert, J.E. Relaxation Times, Linewidths and Spin Kinetic Phenomena. In Electron Paramagnetic Resonance; John Wiley & Sons Inc.: Hoboken, NJ, USA, 1994; pp. 301–356. [Google Scholar]
- Kang, H.-J.; Ko, M.-J.; Chung, M.-S. Anthocyanin Structure and pH Dependent Extraction Characteristics from Blueberries (Vaccinium corymbosum) and Chokeberries (Aronia melanocarpa) in Subcritical Water State. Foods 2021, 10, 527. [Google Scholar] [CrossRef]
- AOAC. Official Methods of Analysis; Association of Official Analytical Chemists: Washington, DC, USA, 1990; Volume 15. [Google Scholar]
- Phromnoi, K.; Suttajit, M.; Saenjum, C. Polyphenols and rosmarinic acid contents, antioxidant and anti-inflammatory activities of different solvent fractions from Nga-Mon (Perilla frutescens) leaf. J. Pharm. Nutr. Sci. 2019, 9, 239–246. [Google Scholar]
- Pengkumsri, N.; Chaiyasut, C.; Saenjum, C.; Sirilun, S.; Peerajan, S.; Suwannalert, P.; Sirisattha, S.; Sivamaruthi, B.S. Physicochemical and antioxidative properties of black, brown and red rice varieties of northern Thailand. Food Sci. Technol. 2015, 35, 331–338. [Google Scholar] [CrossRef]
- Saenjum, C.; Pattananandecha, T.; Nakagawa, K. Detection of antioxidant phytochemicals isolated from Camellia japonica seeds using HPLC and EPR imaging. Antioxidants 2020, 9, 493. [Google Scholar] [CrossRef]
- Banjerdpongchai, R.; Wudtiwai, B.; Khaw-On, P.; Rachakhom, W.; Duangnil, N.; Kongtawelert, P. Hesperidin from Citrus seed induces human hepatocellular carcinoma HepG2 cell apoptosis via both mitochondrial and death receptor pathways. Tumour Biol. 2016, 37, 227–237. [Google Scholar] [CrossRef]
- Sirithunyalug, B.; Saenjum, C.; Charumanee, S.; Sivamaruthi, B.S.; Chaiyasut, C.; Sirithunyalug, J.; Tipduangta, P. Development of colorectal-targeted dietary supplement tablets containing natural purple rice bran oil as a colorectal chemopreventive. Nutrients 2018, 10, 444. [Google Scholar] [CrossRef]
- Nakagawa, K.; Maeda, H.; Yamaya, Y.; Tonosaki, Y. Maillard reaction intermediates and related phytochemicals in black garlic determined by EPR and HPLC analyses. Molecules 2020, 25, 4578. [Google Scholar] [CrossRef]


| Sample Extracts | Total Phenolic Content (TPC) | Total Flavonoid Content (TFC) | Total Anthocyanin Content (TAC) |
|---|---|---|---|
| mg GAE/g Dried Weight | mg QE/g Dried Weight | mg CCE/g Dried Weight | |
| Fresh red pulp (RP) | 277.6 ± 14.2 c | 177.4 ± 12.5 c | 159.7 ± 8.9 a |
| Fresh white pulp (WP) | 48.4 ± 6.8 e | 39.5 ± 7.3 e | ND |
| Red peel (RPe) | 294.8 ± 12.9 c | 193.8 ± 11.7 c | 135.4 ± 9.3 b |
| White peel (WPe) | 207.3 ± 8.5 d | 142.9 ± 12.6 d | 106.8 ± 7.4 c |
| Red pulp seed (RP-S) | 375.1 ± 12.6 a | 264.4 ± 10.8 a | ND |
| White pulp seed (WP-S) | 338.7 ± 13.3 b | 227.6 ± 11.6 b | ND |
| Sample Extracts | IC50 (µg/mL) | |
|---|---|---|
| RNS | ROS | |
| Fresh red pulp (RP) | 13.72 ± 1.08 b | 18.73 ± 1.17 d |
| Fresh white pulp (WP) | >100 | >100 |
| Red peel (RPe) | 17.32 ± 1.26 c | 24.37 ± 1.41 e |
| White peel (WPe) | 22.49 ± 1.15 d | 27.85 ± 1.83 e |
| Red pulp seed (RP-S) | 29.78 ± 1.42 e | 65.42 ± 2.53 f |
| White pulp seed (WP-S) | 32.49 ± 1.36 e | 73.08 ± 2.17 e |
| Cyanidin 3-glucoside | 6.25 ± 0.58 a | 8.48 ± 0.63 b |
| Curcumin | 5.84 ± 0.73 a | ND |
| N-Acetylcysteine | ND | 5.18 ± 0.42 a |
| L-Ascorbic acid | ND | 12.54 ± 0.79 c |
| Red | White | |||||
|---|---|---|---|---|---|---|
| Peel | Pulp | Seed | Peel | Pulp | Seed | |
| ΔHpp (mT) | 0.66 ± 0.06 | 0.64 ± 0.05 | 0.61 ± 0.06 | 1.45 ± 0.13 | 0.59 ± 0.05 | 0.61 ± 0.06 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Saenjum, C.; Pattananandecha, T.; Nakagawa, K. Antioxidative and Anti-Inflammatory Phytochemicals and Related Stable Paramagnetic Species in Different Parts of Dragon Fruit. Molecules 2021, 26, 3565. https://doi.org/10.3390/molecules26123565
Saenjum C, Pattananandecha T, Nakagawa K. Antioxidative and Anti-Inflammatory Phytochemicals and Related Stable Paramagnetic Species in Different Parts of Dragon Fruit. Molecules. 2021; 26(12):3565. https://doi.org/10.3390/molecules26123565
Chicago/Turabian StyleSaenjum, Chalermpong, Thanawat Pattananandecha, and Kouichi Nakagawa. 2021. "Antioxidative and Anti-Inflammatory Phytochemicals and Related Stable Paramagnetic Species in Different Parts of Dragon Fruit" Molecules 26, no. 12: 3565. https://doi.org/10.3390/molecules26123565
APA StyleSaenjum, C., Pattananandecha, T., & Nakagawa, K. (2021). Antioxidative and Anti-Inflammatory Phytochemicals and Related Stable Paramagnetic Species in Different Parts of Dragon Fruit. Molecules, 26(12), 3565. https://doi.org/10.3390/molecules26123565







