The Chemical Compositions, and Antibacterial and Antioxidant Activities of Four Types of Citrus Essential Oils
Abstract
1. Introduction
2. Results and Discussion
2.1. Chemical Compositions of the Citrus EOs
2.2. Antibacterial Activity
2.3. Antioxidant Activity
3. Materials and Methods
3.1. Materials
3.2. Preparation of Citrus EO Samples
3.3. GC-MS Analyses
3.4. Antimicrobial Activity Assays
3.4.1. Bacterial Growth Conditions
3.4.2. Determination of the Inhibition Zone Diameter (IZD)
3.4.3. Determination of Minimum Inhibitory Concentration (MIC)
3.5. Free Radical-Scavenging Capacity
3.5.1. DPPH Radical-Scavenging Assay
3.5.2. ABTS Radical-Scavenging Assay
3.6. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Magare, M.E.; Sahu, N.; Kanade, G.S.; Chanotiya, C.S.; Thul, S.T. An Integrated Process of Value Addition to Citrus Waste and Performance of Fenton Process for Its Conversion to Biogas. Waste Biomass Valori. 2020, 11, 165–172. [Google Scholar] [CrossRef]
- Kamal, G.M.; Anwar, F.; Hussain, A.I.; Sarri, N.; Ashraf, M.Y. Yield and chemical composition of Citrus essential oils as affected by drying pretreatment of peels. Inter. Food Res. J. 2011, 18, 1275–1282. [Google Scholar]
- U.S. Department of Agriculture (USDA). Citrus: World Markets and Trade. Available online: http://apps.fas.usda.gov/psdonline/circulars/citrus.pdf (accessed on 4 March 2021).
- Zhang, X.; Du, Y.P.; Tong, P.J.; Wei, Y.L.; Wang, M. Geographical classification of Nanfeng mandarin by near infrared spectroscopy coupled with chemometrics methods. J. Innov. Opt. Health Sci. 2014, 7, 1450028. [Google Scholar] [CrossRef]
- Güney, M.; Oz, A.T.; Kafkas, E. Comparison of lipids, fatty acids and volatile compounds of various kumquat species using HS/GC/MS/FID techniques. J. Sci. Food Agric. 2014, 95, 1268–1273. [Google Scholar] [CrossRef] [PubMed]
- Hosseini, S.F.; Amraie, M.; Salehi, M.; Mohseni, M.; Aloui, H. Effect of chitosan-based coatings enriched with savory and/or tarragon essential oils on postharvest maintenance of kumquat (Fortunella sp.) fruit. Food Sci. Nutr. 2019, 7, 155–162. [Google Scholar] [CrossRef]
- Liu, X.F.; Liu, B.H.; Jiang, D.; Zhu, S.P.; Shen, W.X.; Yu, X.; Xue, Y.; Liu, M.Y.; Feng, J.Y.; Zhao, X.C. The accumulation and composition of essential oil in kumquat peel. Sci. Hortic. 2019, 252, 121–129. [Google Scholar] [CrossRef]
- Wang, Y.W.; Zeng, W.C.; Xu, P.Y.; Lan, Y.J.; Zhu, R.X.; Zhong, K.; Huang, Y.N.; Gao, H. Chemical Composition and Antimicrobial Activity of the Essential Oil of Kumquat (Fortunella crassifolia Swingle) Peel. Int. J. Mol. Sci. 2012, 13, 3382–3393. [Google Scholar] [CrossRef]
- Zhao, X.; Finlay, D.; Kneafsey, M. The effectiveness of contemporary geographical Indications (GIs) schemes in enhancing the quality of Chinese agrifoods—Experiences from the field. J. Rural. Stud. 2014, 36, 77–86. [Google Scholar] [CrossRef]
- Liu, G.D.; Chen, Y.H.; He, X.X.; Yao, F.X.; Guan, G.; Zhong, B.L.; Zhou, G.F. Seasonal changes of mineral nutrients in the fruit of navel orange plants grafted on trifoliate orange and citrange. Sci. Hortic. 2020, 264, 109156. [Google Scholar] [CrossRef]
- Li, X.; Kitajima, A.; Katsura, K.; Saito, H.; Koeda, S.; Takisawa, R.; Kawai, T.; Nakazaki, T.; Shimizu, T. Cell wall related gene expression during secondary physiological fruit drop in ponkan (Citrus reticulata Blanco) and hyuganatsu (C. tamurana hort. ex Tanaka). Acta Hortic. 2016, 1135, 47–52. [Google Scholar] [CrossRef]
- Ye, X.Q.; Chen, J.C.; Liu, D.H.; Jiang, P.; Shi, J.; Xue, S.; Wu, D.; Xu, J.G.; Kakuda, Y. Identification of bioactive composition and antioxidant activity in young mandarin fruits. Food Chem. 2011, 124, 1561–1566. [Google Scholar] [CrossRef]
- Sharma, K.; Mahato, N.; Cho, M.H.; Lee, Y.R. Converting citrus wastes into value added products: Economical and environment friendly approaches. Nutrition 2017, 34, 29–46. [Google Scholar] [CrossRef] [PubMed]
- Magalhães, M.L.; Ionta, M.; Ferreira, G.Á.; Campidelli, M.L.L.; Nelson, D.L.; Ferreira, V.R.F.; Rezende, D.A.C.S.; Cardoso, M.G. Biological activities of the essential oil from the Moro orange peel (Citrus sinensis (L.) Osbeck). Flavour Fragr. J. 2020, 35, 294–301. [Google Scholar] [CrossRef]
- Campoloa, O.; Puglisib, I.; Barbagallob, R.N.; Cherifbcd, A.; Ricuperob, M.; Biondib, A.; Palmeria, V.; Baglierib, A.; Zappalàb, L. Side effects of two citrus essential oil formulations on a generalist insect predator, plant and soil enzymatic activities. Chemosphere 2020, 257, 127252. [Google Scholar] [CrossRef] [PubMed]
- Bora, H.; Kamle, M.; Mahato, D.K.; Tiwari, P.; Kumar, P. Citrus Essential Oils (CEOs) and Their Applications in Food: An Overview. Plants 2020, 9, 357. [Google Scholar] [CrossRef]
- Frassinetti, S.; Caltavuturo, L.; Cini, M.; Della Croce, C.M.; Maserti, B.E. Antibacterial and antioxidant activity of essential oils from citrus spp. J. Essent. Oil Res. 2011, 23, 27–31. [Google Scholar] [CrossRef]
- Djamel, D. Chemical Profile, Antibacterial and Antioxidant Activity of Algerian Citrus Essential Oils and Their Application in Sardina pilchardus. Foods 2015, 4, 208–228. [Google Scholar]
- Yi, F.P.; Jin, R.Y.; Sun, J.; Ma, B.D.; Bao, X.L. Evaluation of mechanical-pressed essential oil from Nanfeng mandarin (Citrus reticulata Blanco cv. Kinokuni) as a food preservative based on antimicrobial and antioxidant activities. LWT-Food Sci. Technol. 2018, 95, 346–353. [Google Scholar] [CrossRef]
- Al-Saman, M.A.; Abdella, A.; Mazrou, K.E.; Tayel, A.A.; Irmak, S. Antimicrobial and antioxidant activities of different extracts of the peel of kumquat (Citrus japonica Thunb). J. Food Meas. Charact. 2019, 13, 3221–3229. [Google Scholar] [CrossRef]
- Raut, J.S.; Karuppayil, S.M. A status review on the medicinal properties of essential oils. Ind. Crop. Prod. 2014, 62, 250–264. [Google Scholar] [CrossRef]
- Shaaban, H.A.H.; El-Ghorab, A.H.; Takayuki, S. Bioactivity of essential oils and their volatile aroma components: Review. J. Essent. Oil Res. 2012, 24, 203–212. [Google Scholar] [CrossRef]
- Chi, P.T.L.; Chinh, V.K.; Hung, P.V.; Phi, N.T.L. Antimicrobial and antioxidant activities of essential oils extracted from leaves of Vinh orange, Dao lime and Thanh Tra pomelo in Vietnam. Int. J. Food Sci. Nutr. 2018, 3, 152–156. [Google Scholar]
- Kraśniewska, K.; Kosakowska, O.; Pobiega, K.; Gniewosz, M. The Influence of Two-Component Mixtures from Spanish Origanum Oil with Spanish Marjoram Oil or Coriander Oil on Antilisterial Activity and Sensory Quality of a Fresh Cut Vegetable Mixture. Foods 2020, 9, 1740. [Google Scholar] [CrossRef]
- Karadeniz, A.; Cinbilgel, I.; Gun, S.S.; Cetin, A. Antioxidant activity of some Turkish medicinal plants. Nat. Prod. Res. 2015, 29, 2308–2312. [Google Scholar] [CrossRef] [PubMed]
- Miguel, M.G. Antioxidant and Anti-Inflammatory Activities of Essential Oils: A Short Review. Molecules 2010, 15, 9252–9287. [Google Scholar] [CrossRef] [PubMed]
- Hsouna, A.B.; Halima, B.N.; Smaoui, S.; Hamdi, N. Citrus lemon essential oil: Chemical composition, antioxidant and antimicrobial activities with its preservative effect against Listeria monocytogenes inoculated in minced beef meat. Lipids Health Dis. 2017, 16, 146. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.H.; Yang, K.M.; Huang, T.C.; Wu, M.L. Traditional Small-Size Citrus from Taiwan: Essential Oils, Bioactive Compounds and Antioxidant Capacity. Medicines 2017, 4, 28. [Google Scholar] [CrossRef]
- The International Organization for Standardization. Essential Oil of Mandarin, Italian Type (Citrus Reticulata Blanco) ISO 3528; The International Organization for Standardization: Geneva, Switzerland, 2012. [Google Scholar]
- Choi, S.H. Characteristic Odor Components of Kumquat (Fortunella japonica Swingle) Peel Oil. J. Agric. Food Chem. 2005, 53, 1642–1647. [Google Scholar] [CrossRef]
- Ou, M.C.; Liu, Y.H.; Sun, Y.W.; Chan, C.F. The composition, antioxidant and antibacterial activities of cold-pressed and distilled essential oils of Citrus paradise and Citrus grandis (L.) Osbeck. Evid. Based Complement. Altern. Med. 2015, 2015, 804091. [Google Scholar] [CrossRef]
- Guo, Q.; Liu, K.; Deng, W.; Zhong, B.; Yang, W.; Chun, J. Chemical composition and antimicrobial activity of Gannan navel orange (Citrus sinensis Osbeck cv. Newhall) peel essential oils. Food Sci. Nutr. 2018, 6, 1431–1437. [Google Scholar] [CrossRef]
- Ambrosio, C.M.S.; de Alencar, S.M.; de Sousa, R.L.M.; Moreno, A.M.; Gloria, E.M.D. Antimicrobial activity of several essential oils on pathogenic and beneficial bacteria. Ind. Crop. Prod. 2017, 97, 128–136. [Google Scholar] [CrossRef]
- Sikkema, J.; de Bont, J.A.M.; Poolman, B. Mechanisms of membrane toxicity of hydrocarbons. Microbiol. Rev. 1995, 59, 201–222. [Google Scholar] [CrossRef]
- Aggarwal, K.K.; Khanuja, S.P.S.; Ahmad, A.; Kumar, T.R.S.; Gupta, V.K.; Kumar, S. Antimicrobial activity profiles of the two enantiomers of limonene and carvone isolated from the oils of Mentha spicata and Anethum sowa. Flavour Fragr. J. 2002, 17, 59–63. [Google Scholar] [CrossRef]
- Anton, P.A.; Kemp, J.A.; Butler, T.; Jacobs, M.R. Comparative efficacies of ceftriaxone, moxalactam, and ampicillin in experimental Salmonella typhimurium infection. Antimicrob. Agents Chemother. 1982, 22, 312–315. [Google Scholar] [CrossRef] [PubMed]
- Anomohanran, E.E.; Owhe-Ureghe, U.B.; Ehwarieme, D.A. Evaluating the Sensitivity of Pseudomonas aeruginosa, Escherichia coli and Salmonella typhi to various brands of Ampicillin and Amoxicillin available in Nigeria. Am. J. Agric. Biol. Sci. 2014, 9, 503–509. [Google Scholar] [CrossRef][Green Version]
- Gulcin, I. Antioxidants and antioxidant methods: An updated overview. Arch. Toxicol. 2020, 94, 651–715. [Google Scholar] [CrossRef] [PubMed]
- Hashim, N.A.; Ahmad, F.; Jani, N.A.; Susanti, D. In vitro Antioxidant, Antityrosinase, Antibacterial and Cytotoxicity Activities of the Leaf and Stem Essential Oil from Piper magnibaccum C. DC. J. Essent. Oil Bear. Plants 2017, 20, 223–232. [Google Scholar] [CrossRef]
- Farahmandfar, R.; Tirgarian, B.; Dehghan, B.; Nemati, A. Changes in chemical composition and biological activity of essential oil from Thomson navel orange (Citrus sinensis L. Osbeck) peel under freezing, convective, vacuum, and microwave drying methods. Food Sci. Nutr. 2020, 8, 124–138. [Google Scholar] [CrossRef] [PubMed]
- Dawidowicz, A.L.; Małgorzata, O. Does antioxidant properties of the main component of essential oil reflect its antioxidant properties? The comparison of antioxidant properties of essential oils and their main components. Nat. Prod. Res. 2014, 28, 1952–1963. [Google Scholar] [CrossRef]
- Klančnik, A.; Piskernik, S.; Jeršek, B.; Možina, S.S. Evaluation of diffusion and dilution methods to determine the antibacterial activity of plant extracts. J. Microbiol. Methods 2010, 81, 121–126. [Google Scholar] [CrossRef]
- Ksouda, G.; Sellimi, S.; Merlier, F.; Falcimaigne-cordin, A.; Thomasset, B.; Nasri, M.; Hajji, M. Composition, antibacterial and antioxidant activities of Pimpinella saxifraga essential oil and application to cheese preservation as coating additive. Food Chem. 2019, 288, 47–56. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.G.; Hu, Q.P.; Liu, Y. Antioxidant and DNA-Protective Activities of Chlorogenic Acid Isomers. J. Agric. Food Chem. 2012, 60, 11625–11630. [Google Scholar] [CrossRef] [PubMed]
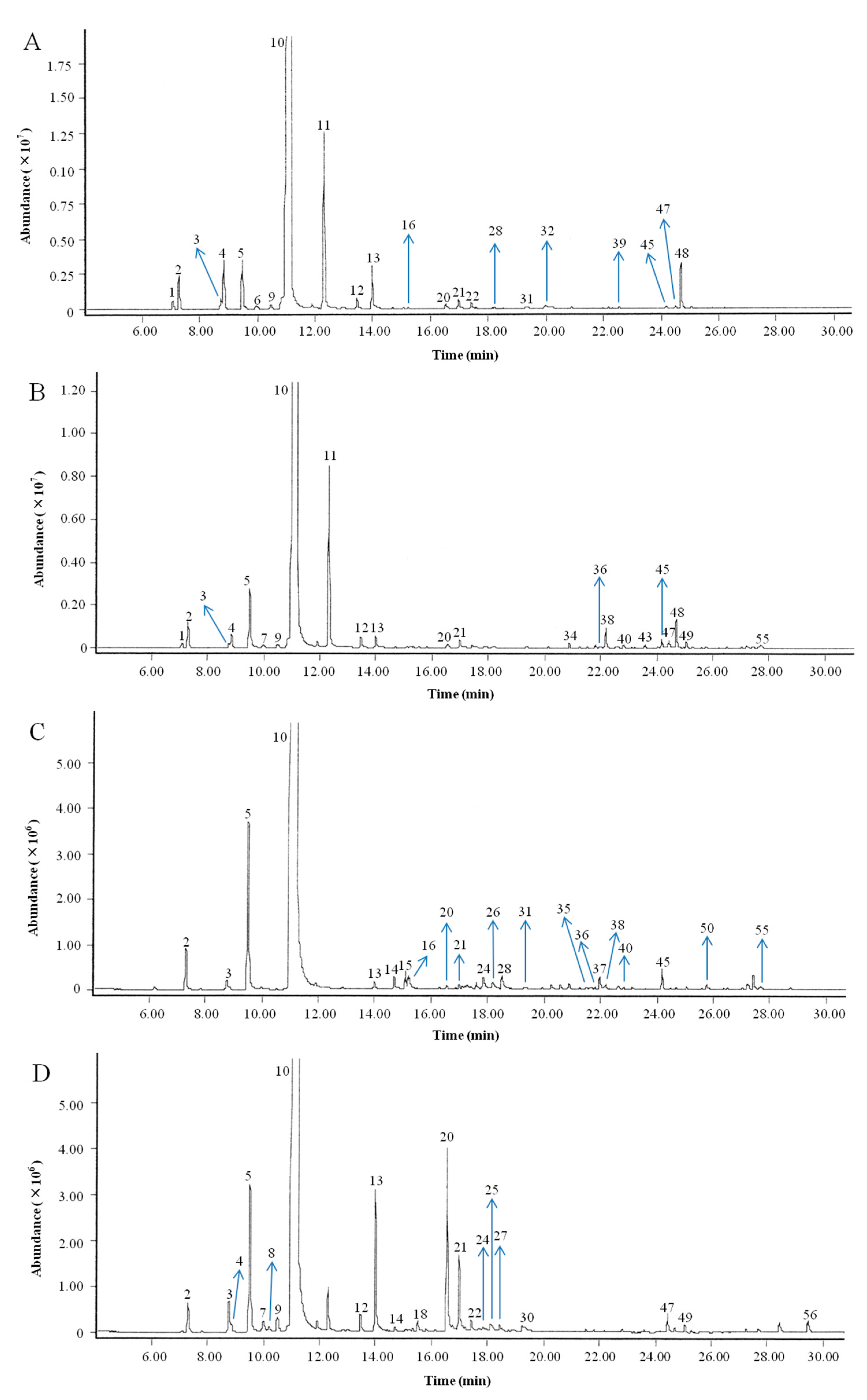
| No. | RI exp. | RI lit. | Compounds | Composition (%) | CharacteristicMass Ions (m/z) | |||
|---|---|---|---|---|---|---|---|---|
| NMEO | XMEO | YKEO | PDEO | |||||
| 1 | 923 | 927 | α-Thujene | 0.29 ± 0.00 a | 0.15 ± 0.01 b | – | – | 77, 91, 92, 93 |
| 2 | 929 | 929 | α-Pinene | 1.22 ± 0.03 a | 0.83 ± 0.05 b | 0.62 ± 0.07 c | 0.33 ± 0.01 d | 39, 77, 91, 93 |
| 3 | 970 | 963 | Sabinene | 0.35 ± 0.00 a | 0.08 ± 0.01 c | 0.15 ± 0.02 b | 0.33 ± 0.01 a | 77, 91, 93, 136 |
| 4 | 972 | 970 | β-Pinene | 2.00 ± 0.05 a | 0.46 ± 0.02 b | – | 0.09 ± 0.01 c | 41, 69, 91, 93 |
| 5 | 990 | 992 | β-Myrcene | 2.08 ± 0.01 b | 2.02 ± 0.12 b | 2.73 ± 0.22 a | 1.90 ± 0.04 b | 39, 41, 69, 93 |
| 6 | 1003 | 1002 | Octanal | 0.29 ± 0.03 a | – | – | – | 41, 43, 44, 56 |
| 7 | 1004 | 1006 | α-Phellandrene | – | 0.10 ± 0.00 b | – | 0.17 ± 0.03 a | 77, 91, 92, 93 |
| 8 | 1009 | 1007 | 3-Carene | – | – | – | 0.08 ± 0.00 a | 77, 79, 91, 93 |
| 9 | 1016 | 1020 | α-Terpinene | 0.21 ± 0.01 b | 0.14 ± 0.01 c | – | 0.26 ± 0.01 a | 91, 93, 121, 136 |
| 10 | 1030 | 1029 | Limonene | 79.13 ± 0.41 d | 86.03 ± 0.29 c | 91.54 ± 1.22 a | 88.25 ± 0.62 b | 67, 68, 79, 93 |
| 11 | 1060 | 1061 | γ-Terpinene | 8.19 ± 0.28 a | 5.80 ± 0.07 b | – | – | 77, 91, 93, 136 |
| 12 | 1088 | 1089 | Terpinolene | 0.49 ± 0.04 a | 0.33 ± 0.02 b | – | 0.18 ± 0.01 c | 93, 105, 121, 136 |
| 13 | 1101 | 1102 | Linalool | 1.81 ± 0.04 a | 0.43 ± 0.03 c | 0.12 ± 0.02 d | 1.50 ± 0.06 b | 41, 55, 69, 93 |
| 14 | 1122 | 1123 | trans-p-Mentha-2,8-dien-1-ol | – | – | 0.18 ± 0.00 a | 0.06 ± 0.02 b | 43, 79, 109, 134 |
| 15 | 1134 | 1136 | cis-Limonene oxide | 0.03 ± 0.00 b | 0.02 ± 0.00 b | 0.31 ± 0.01 a | 0.03 ± 0.00 b | 41, 43, 67, 93 |
| 16 | 1139 | 1139 | trans-Limonene oxide | 0.05 ± 0.00 b | 0.03 ± 0.01 c | 0.20 ± 0.01 a | 0.02 ± 0.01 c | 43, 67, 94, 108 |
| 17 | 1141 | 1145 | cis-β-Terpineol | – | – | – | 0.03 ± 0.01 a | 41, 43, 71, 93 |
| 18 | 1147 | 1151 | trans-β-Terpineol | – | – | – | 0.14 ± 0.02 a | 43, 71, 93, 136 |
| 19 | 1155 | 1155 | Citronellal | – | 0.01 ± 0.01 b | – | 0.03 ± 0.00 a | 41, 55, 69, 95 |
| 20 | 1177 | 1178 | Terpinen-4-ol | 0.22 ± 0.01 b | 0.14 ± 0.02 bc | 0.06 ± 0.00 c | 1.98 ± 0.07 a | 71, 77, 91, 93 |
| 21 | 1190 | 1191 | α-Terpineol | 0.45 ± 0.02 b | 0.25 ± 0.02 c | 0.07 ± 0.01 d | 1.01 ± 0.04 a | 59, 68, 79, 93 |
| 22 | 1203 | 1204 | Decanal | 0.57 ± 0.09 a | – | – | 0.25 ± 0.02 b | 41, 43, 55, 57 |
| 23 | 1220 | 1224 | trans-Carveol | – | – | 0.30 ± 0.04 a | 0.17 ± 0.03 b | 41, 55, 84, 109 |
| 24 | 1231 | 1232 | Citronellol | – | – | – | 0.17 ± 0.04 a | 41, 67, 69, 81 |
| 25 | 1233 | 1238 | cis-Carveol | – | – | 0.22 ± 0.04 a | – | 91, 105, 119, 134 |
| 26 | 1243 | 1242 | Neral | – | – | – | 0.12 ± 0.03 a | 27, 39, 41, 69 |
| 27 | 1246 | 1251 | Carvone | 0.05 ± 0.03 b | – | 0.34 ± 0.05 a | – | 39, 54, 82, 93 |
| 28 | 1253 | 1253 | Geraniol | – | – | – | 0.04 ± 0.01 a | 41, 68, 69, 93 |
| 29 | 1273 | 1272 | Geranial | – | – | – | 0.23 ± 0.13 a | 39, 41, 69, 84 |
| 30 | 1277 | 1276 | Perillyl aldehyde | 0.09 ± 0.01 a | 0.04 ± 0.01 b | 0.09 ± 0.01 a | – | 39, 67, 68, 79 |
| 31 | 1301 | 1295 | Thymol | 0.18 ± 0.04 a | – | – | – | 91, 115, 135, 150 |
| 32 | 1309 | 1310 | Undecanal | – | 0.03 ± 0.02 a | – | – | 41, 43, 55, 57 |
| 33 | 1340 | 1339 | δ-Elemene | 0.04 ± 0.00 b | 0.10 ± 0.00 a | – | – | 41, 93, 121, 136 |
| 34 | 1366 | 1371 | Neryl acetate | – | 0.03 ± 0.01 b | 0.07 ± 0.03 a | 0.02 ± 0.01 b | 41, 68, 69, 93 |
| 35 | 1379 | 1375 | Copaene | – | 0.07 ± 0.01 a | 0.06 ± 0.02 a | 0.02 ± 0.01 b | 93, 105, 119, 161 |
| 36 | 1386 | 1382 | Geranyl acetate | – | – | 0.25 ± 0.09 a | – | 41, 69, 79, 93 |
| 37 | 1395 | 1395 | β-Elemene | 0.04 ± 0.00 bc | 0.52 ± 0.02 a | 0.08 ± 0.02 b | 0.01 ± 0.01 c | 41, 68, 81, 93 |
| 38 | 1410 | 1411 | Dodecanal | 0.09 ± 0.02 a | – | – | 0.02 ± 0.01 b | 41, 43, 55, 57 |
| 39 | 1424 | 1428 | β-Caryophyllene | 0.02 ± 0.00 c | 0.09 ± 0.01 a | 0.05 ± 0.01 b | 0.03 ± 0.00 c | 41, 69, 79, 93 |
| 40 | 1437 | 1431 | γ-Elemene | – | – | 0.02 ± 0.01 a | – | 79, 93, 107, 121 |
| 41 | 1439 | 1438 | Perillyl acetate | – | – | 0.03 ± 0.01 a | – | 43, 68, 91, 119 |
| 42 | 1459 | 1455 | α-Humulene | – | 0.10 ± 0.02 a | 0.02 ± 0.01 bc | 0.03 ± 0.00 b | 41, 80, 93, 121 |
| 43 | 1480 | 1478 | β-Selinene | – | 0.03 ± 0.01 a | – | – | 41, 93, 105, 107 |
| 44 | 1486 | 1485 | Germacrene D | 0.09 ± 0.01 b | 0.22 ± 0.05 a | 0.27 ± 0.06 a | 0.02 ± 0.00 b | 105, 119, 161, 204 |
| 45 | 1492 | 1499 | α-Selinene | – | 0.03 ± 0.01 a | – | – | 161, 175, 189, 204 |
| 46 | 1502 | 1500 | Valencene | 0.09 ± 0.01 ab | 0.28 ± 0.07 a | – | 0.19 ± 0.01 bc | 147, 161, 189, 204 |
| 47 | 1511 | 1504 | α-Farnesene | 1.50 ± 0.17 a | 0.69 ± 0.18 b | 0.04 ± 0.01 c | 0.04 ± 0.01 c | 41, 55, 69, 93 |
| 48 | 1530 | 1527 | δ-Cadinene | 0.03 ± 0.01 b | 0.16 ± 0.04 a | 0.04 ± 0.02 b | 0.07 ± 0.00 b | 134, 161, 189, 204 |
| 49 | 1564 | 1560 | Germacrene B | – | 0.04 ± 0.01 ab | 0.08 ± 0.02 a | – | 93, 105, 107, 121 |
| 50 | 1568 | 1565 | trans-Nerolidol | – | – | 0.04 ± 0.01 a | – | 41, 43, 69, 93 |
| 51 | 1586 | 1583 | Caryophyllene oxide | 0.03 ± 0.00 a | – | – | – | 41, 79, 91, 105 |
| 52 | 1640 | 1643 | α-Eudesmol | – | – | – | 0.03 ± 0.01 a | 59, 149, 161, 189 |
| 53 | 1651 | 1651 | Cubenol | – | 0.04 ± 0.01 a | – | – | 119, 161, 189, 204 |
| 54 | 1664 | 1661 | β-Eudesmol | – | 0.12 ± 0.03 a | 0.12 ± 0.02 a | 0.04 ± 0.01 b | 59, 108, 149, 164 |
| 55 | 1762 | 1756 | α-Sinensal | – | – | – | 0.16 ± 0.04 a | 55, 79, 93, 134 |
| Total | 99.63 | 99.41 | 98.10 | 98.05 | ||||
| Monoterpene hydrocarbons | 93.96 | 95.94 | 95.04 | 91.59 | ||||
| Oxygenated monoterpenes | 2.88 | 0.95 | 2.24 | 5.55 | ||||
| Sesquiterpene hydrocarbons | 1.81 | 2.33 | 0.66 | 0.41 | ||||
| Oxygenated sesquiterpenes | 0.03 | 0.16 | 0.16 | 0.23 | ||||
| Others(straight-chain aldehydes) | 0.95 | 0.03 | 0.00 | 0.27 | ||||
| Bacterial Strain | Essential Oil | Ampicillin (100 µg/mL) | |||
|---|---|---|---|---|---|
| NMEO | XMEO | YKEO | PDEO | ||
| Bacillus subtilis (G+) | 10.57 ± 0.74 cd | 8.49 ± 0.41 d | 19.49 ± 1.41 b | 10.99 ± 1.02 c | 24.05 ± 0.78 a |
| Staphylococcus aureus (G+) | 9.54 ± 1.19 b | 6.90 ± 0.63 c | 8.02 ± 0.16 bc | 7.66 ± 0.41 c | 33.03 ± 0.24 a |
| Escherichia coli (G−) | 9.48 ± 1.22 bc | 7.44 ± 0.13 c | 21.58 ± 1.57 a | 10.77 ± 1.33 b | 7.95 ± 0.21 bc |
| Pseudomonas aeruginosa (G−) | 7.93 ± 0.49 a | 6.15 ± 0.05 b | 8.42 ± 0.66 a | 6.30 ± 0.13 b | 6.00 ± 0.00 b |
| Salmonella typhimurium (G−) | 6.35 ± 0.23 c | 6.00 ± 0.00 c | 25.39 ± 1.25 a | 6.00 ± 0.00 c | 13.20 ± 0.30 b |
| Bacterial Strain | Essential Oil (µL/mL) | Ampicillin (µg/mL) | |||
|---|---|---|---|---|---|
| NMEO | XMEO | YKEO | PDEO | ||
| Bacillus subtilis (G+) | 6.25 | 12.50 | 1.56 | 6.25 | 25.00 |
| Staphylococcus aureus (G+) | 25.00 | 50.00 | 12.50 | 50.00 | 0.10 |
| Escherichia coli (G−) | 25.00 | 12.50 | 1.56 | 6.25 | 3.12 |
| Pseudomonas aeruginosa (G−) | 25.00 | 50.00 | 100.00 | 100.00 | 800.00 |
| Salmonella typhimurium (G−) | 25.00 | 100.00 | 6.25 | 50.00 | 1.56 |
| Samples | DPPH IC50 (mg/mL) | ABTS IC50 (mg/mL) |
|---|---|---|
| NMEO | 15.20 ± 1.85 b | 0.80 ± 0.05 d |
| XMEO | 18.25 ± 0.74 b | 1.64 ± 0.18 c |
| YKEO | 30.01 ± 1.14 a | 6.62 ± 0.24 a |
| PDEO | 29.70 ± 1.02 a | 4.17 ± 0.09 b |
| BHT | 0.02 ± 0.01 c | 0.01 ± 0.00 e |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lin, X.; Cao, S.; Sun, J.; Lu, D.; Zhong, B.; Chun, J. The Chemical Compositions, and Antibacterial and Antioxidant Activities of Four Types of Citrus Essential Oils. Molecules 2021, 26, 3412. https://doi.org/10.3390/molecules26113412
Lin X, Cao S, Sun J, Lu D, Zhong B, Chun J. The Chemical Compositions, and Antibacterial and Antioxidant Activities of Four Types of Citrus Essential Oils. Molecules. 2021; 26(11):3412. https://doi.org/10.3390/molecules26113412
Chicago/Turabian StyleLin, Xiaocai, Shan Cao, Jingyu Sun, Dongliang Lu, Balian Zhong, and Jiong Chun. 2021. "The Chemical Compositions, and Antibacterial and Antioxidant Activities of Four Types of Citrus Essential Oils" Molecules 26, no. 11: 3412. https://doi.org/10.3390/molecules26113412
APA StyleLin, X., Cao, S., Sun, J., Lu, D., Zhong, B., & Chun, J. (2021). The Chemical Compositions, and Antibacterial and Antioxidant Activities of Four Types of Citrus Essential Oils. Molecules, 26(11), 3412. https://doi.org/10.3390/molecules26113412






