Multidisciplinary Studies of Folk Medicine “Five Thieves’ Oil” (Olejek Pięciu Złodziei) Components
Abstract
1. Introduction
2. Results and Discussion
2.1. Volatile Compounds in Fresh and Stored Essential Oils
2.2. Low-Field NMR Studies
2.3. Spectroscopic Analysis
2.4. Microscopic and Color Studies
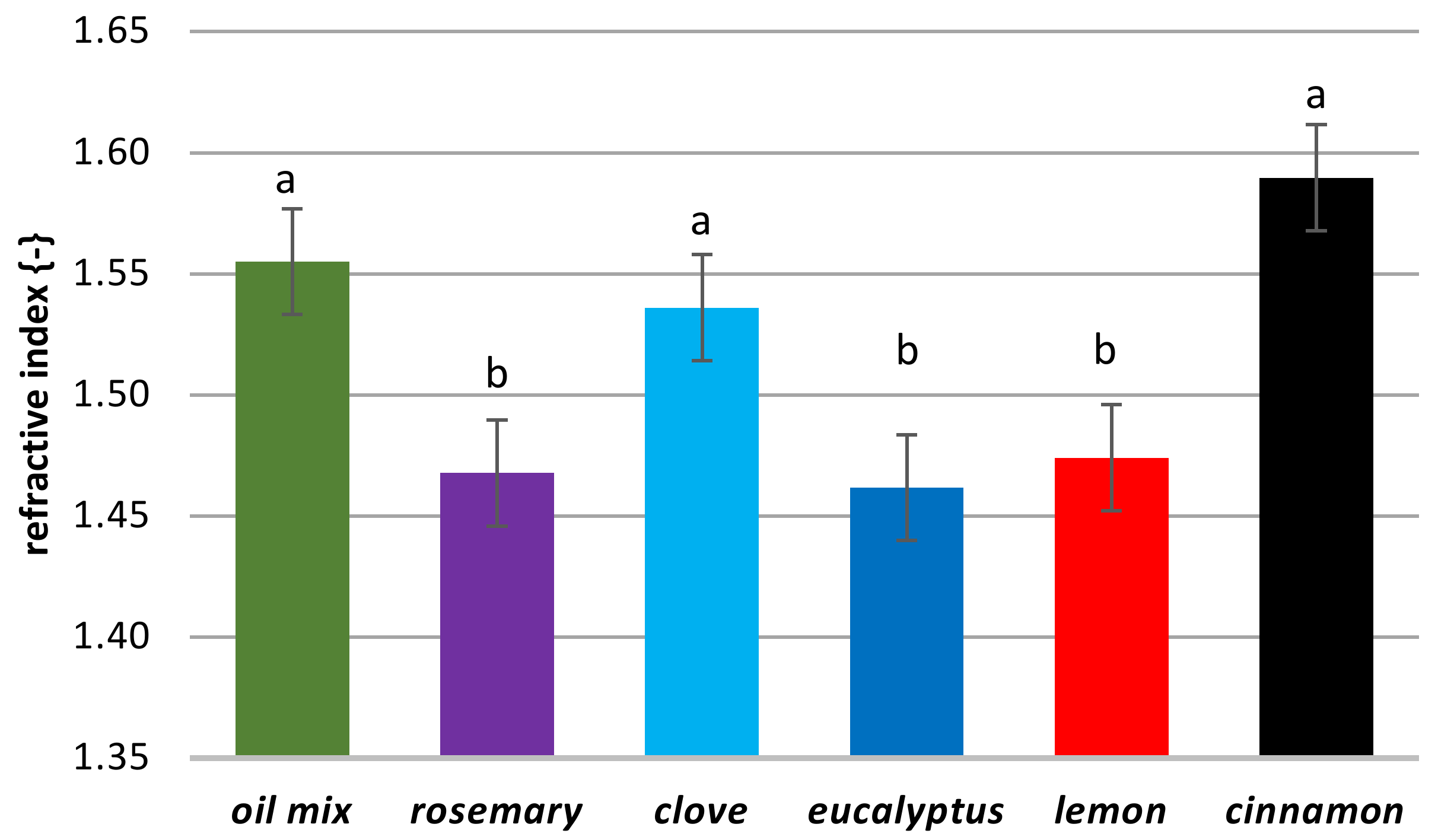
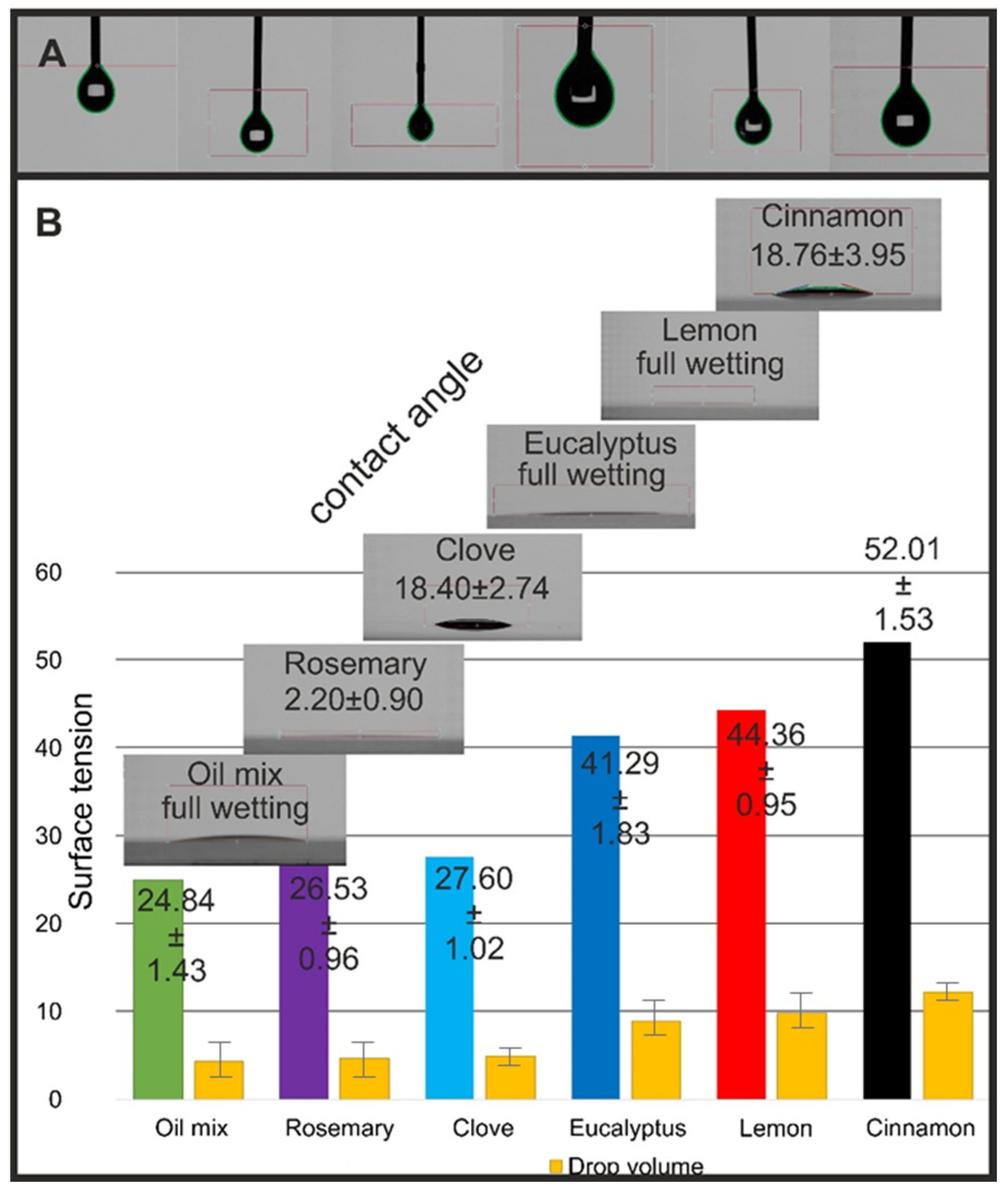
2.5. Surface Properties
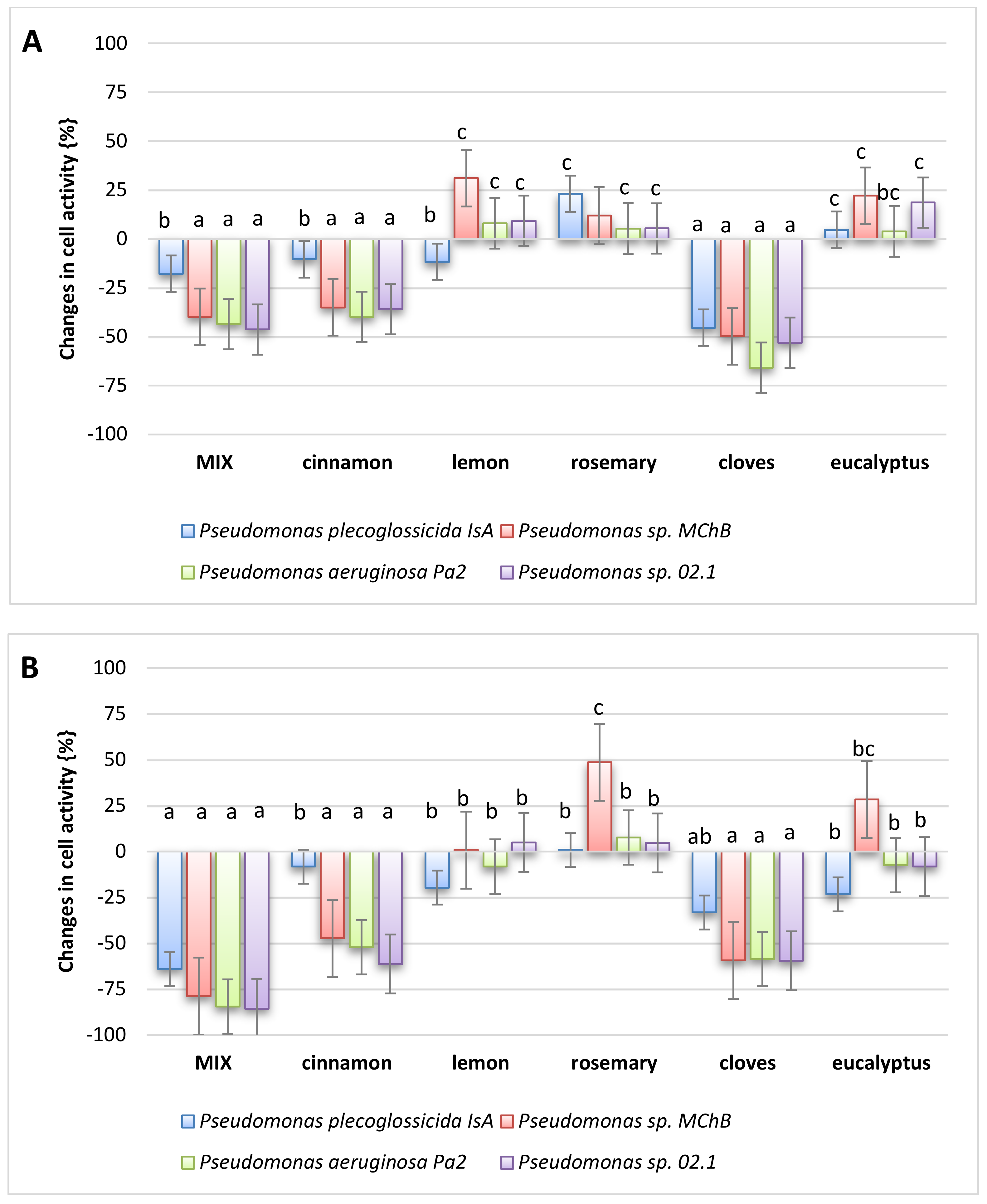
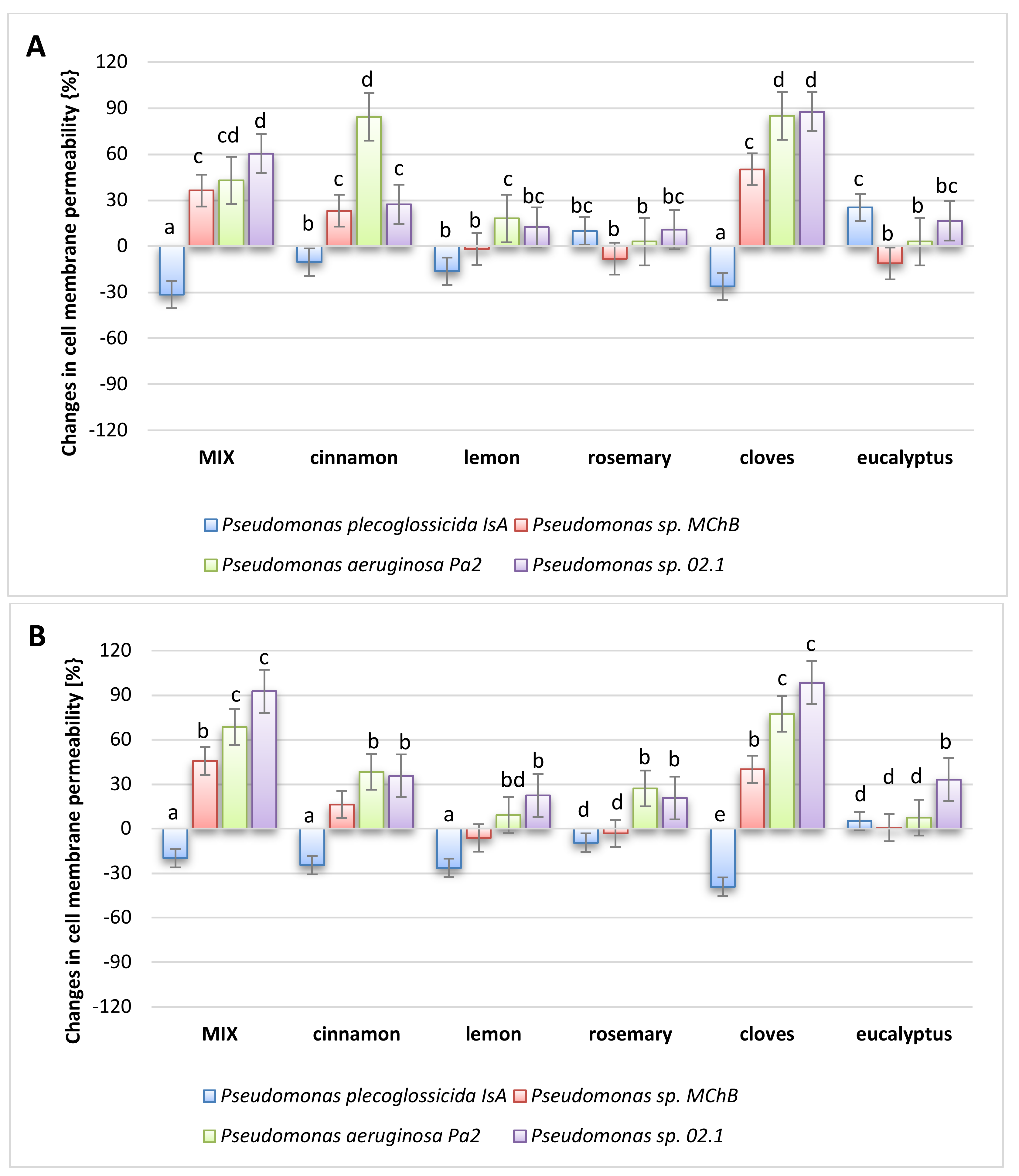
2.6. Antimicrobial Activity
3. Materials and Methods
3.1. Materials
3.2. Methods
3.2.1. GCMS
3.2.2. Essential Oil Evaporation Tests
3.2.3. Low-Field NMR
3.2.4. FTIR
3.2.5. UV–Vis
3.2.6. Fluorescence Behavior
3.2.7. Microscopic Investigations and Camera Imaging
3.2.8. L*a*b* Color Properties
3.2.9. Refractive Index
3.2.10. Contact Angle and Surface Tension Determination
3.2.11. Antibacterial Effect of Oils
3.3. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Macwan, S.R.; Dabhi, B.K.; Aparnathi, K.D.; Prajapati, J.B. Essential Oils of Herbs and Spices: Their Antimicrobial Activity and Application in Preservation of Food. Int. J. Curr. Microbiol. Appl. Sci. 2016, 5, 885–901. [Google Scholar] [CrossRef]
- Blackaby, A. The Hurst. Available online: http://www.hurstgreenhtc.org/wp-content/uploads/2020/12/The-Hurst-11lowres.pdf (accessed on 30 March 2021).
- Bakkali, F.; Averbeck, S.; Averbeck, D.; Idaomar, M. Biological effects of essential oils—A review. Food Chem. Toxicol. 2008, 46, 446–475. [Google Scholar] [CrossRef] [PubMed]
- Yen, P.P.-L.; Kitts, D.D.; Pratap Singh, A. Natural Acidification with Low-pH Fruits and Incorporation of Essential Oil Constituents for Organic Preservation of Unpasteurized Juices. J. Food Sci. 2018, 83, 2039–2046. [Google Scholar] [CrossRef] [PubMed]
- Kitts, D.D.; Singh, A.; Fathordoobady, F.; Doi, B.; Pratap Singh, A. Plant Extracts Inhibit the Formation of Hydroperoxides and Help Maintain Vitamin E Levels and Omega-3 Fatty Acids During High Temperature Processing and Storage of Hempseed and Soybean Oils. J. Food Sci. 2019, 84, 3147–3155. [Google Scholar] [CrossRef]
- Swamy, M.; Sinniah, U. A Comprehensive Review on the Phytochemical Constituents and Pharmacological Activities of Pogostemon cablin Benth.: An Aromatic Medicinal Plant of Industrial Importance. Molecules 2015, 20, 8521–8547. [Google Scholar] [CrossRef] [PubMed]
- Marongiu, B.; Porcedda, S.; Porta, G.D.; Reverchon, E. Extraction and isolation of Salvia desoleana and Mentha spicata subsp. insularis essential oils by supercritical CO2. Flavour Fragr. J. 2001, 16, 384–388. [Google Scholar] [CrossRef]
- Kumara Swamy, M.; Sudipta, K.M.; Lokesh, P.; Neeki, A.M.; Rashmi, W.; Bhaumik, H.S.; Darshil, H.S.; Vijay, R.; Kashyap, S.S.N. Phytochemical screening and in vitro antimicrobial activity of Bougainvillea spectabilis flower extracts. Int. J. Phytomedicine 2012, 4, 375–379. [Google Scholar]
- Akthar, M.S.; Degaga, B.; Azam, T. Antimicrobial activity of essential oils extracted from medicinal plants against the pathogenic microorganisms: A review. Issues Biol. Sci. Pharm. Res. 2014, 2, 1–7. [Google Scholar]
- Arumugam, G.; Swamy, M.; Sinniah, U. Plectranthus amboinicus (Lour.) Spreng: Botanical, Phytochemical, Pharmacological and Nutritional Significance. Molecules 2016, 21, 369. [Google Scholar] [CrossRef]
- Nazzaro, F.; Fratianni, F.; Coppola, R.; Feo, V. De Essential Oils and Antifungal Activity. Pharmaceuticals 2017, 10, 86. [Google Scholar] [CrossRef]
- Chorianopoulos, N.G.; Giaouris, E.D.; Skandamis, P.N.; Haroutounian, S.A.; Nychas, G.-J.E. Disinfectant test against monoculture and mixed-culture biofilms composed of technological, spoilage and pathogenic bacteria: Bactericidal effect of essential oil and hydrosol of Satureja thymbra and comparison with standard acid–base sanitizers. J. Appl. Microbiol. 2008, 104, 1586–1596. [Google Scholar] [CrossRef] [PubMed]
- Burt, S.A.; Reinders, R.D. Antibacterial activity of selected plant essential oils against Escherichia coli O157:H7. Lett. Appl. Microbiol. 2003, 36, 162–167. [Google Scholar] [CrossRef]
- De Martino, L.; De Feo, V.; Nazzaro, F. Chemical Composition and in Vitro Antimicrobial and Mutagenic Activities of Seven Lamiaceae Essential Oils. Molecules 2009, 14, 4213–4230. [Google Scholar] [CrossRef]
- Amri, I.; Mancini, E.; De Martino, L.; Marandino, A.; Lamia, H.; Mohsen, H.; Bassem, J.; Scognamiglio, M.; Reverchon, E.; De Feo, V. Chemical Composition and Biological Activities of the Essential Oils from Three Melaleuca Species Grown in Tunisia. Int. J. Mol. Sci. 2012, 13, 16580–16591. [Google Scholar] [CrossRef] [PubMed]
- Hammer, K.; Carson, C. Antibacterial and Antifungal Activities of Essential Oils Title. In Lipids and Essential Oils as Antimicrobial Agents; Thormar, H., Ed.; John Wiley & Sons: Hoboken, NJ, USA, 2011; pp. 255–306. ISBN 9780470741788. [Google Scholar]
- Raut, J.S.; Karuppayil, S.M. A status review on the medicinal properties of essential oils. Ind. Crops Prod. 2014, 62, 250–264. [Google Scholar] [CrossRef]
- Conner, D.E. Naturally Occurring Compounds. In Antimicrobials in Foods; Davidson, P., Branen, A.L., Eds.; Marcel Dekker, Inc.: New York, NY, USA, 1993; pp. 441–468. [Google Scholar]
- Kim, J.; Marshall, M.R.; Wei, C. Antibacterial activity of some essential oil components against five foodborne pathogens. J. Agric. Food Chem. 1995, 43, 2839–2845. [Google Scholar] [CrossRef]
- Mulyaningsih, S.; Sporer, F.; Zimmermann, S.; Reichling, J.; Wink, M. Synergistic properties of the terpenoids aromadendrene and 1,8-cineole from the essential oil of Eucalyptus globulus against antibiotic-susceptible and antibiotic-resistant pathogens. Phytomedicine 2010, 17, 1061–1066. [Google Scholar] [CrossRef] [PubMed]
- Ali, B.; Al-Wabel, N.A.; Shams, S.; Ahamad, A.; Khan, S.A.; Anwar, F. Essential oils used in aromatherapy: A systemic review. Asian Pac. J. Trop. Biomed. 2015, 5, 601–611. [Google Scholar] [CrossRef]
- Asif, M.; Saleem, M.; Saadullah, M.; Yaseen, H.S.; Al Zarzour, R. COVID-19 and therapy with essential oils having antiviral, anti-inflammatory, and immunomodulatory properties. Inflammopharmacology 2020, 28, 1153–1161. [Google Scholar] [CrossRef]
- Falkenberg Reads, T. Traditional and Complementary Medicines Policy. In MDS-3: Managing Access to Medicines and Health Technologies; Management Sciences for Health: Medford, MA, USA, 2012. [Google Scholar]
- Nowakowski, P.T. Wiedza studentów kierunków pomocowych na temat nadużywania leków. In Wokół Nadużywania Leków; Łuka, M., Ed.; Polskie Towarzystwo Socjologiczne: Warsaw, Poland, 2011; pp. 161–198. ISBN 9788393311132. [Google Scholar]
- Fonseca, M.J.; Santos, C.L.; Costa, P.; Lencastre, L.; Tavares, F. Increasing Awareness about Antibiotic Use and Resistance: A Hands-On Project for High School Students. PLoS ONE 2012, 7, e44699. [Google Scholar] [CrossRef]
- Aguilar-Hernández, M.G.; Sánchez-Bravo, P.; Hernández, F.; Carbonell-Barrachina, Á.A.; Pastor-Pérez, J.J.; Legua, P. Determination of the Volatile Profile of Lemon Peel Oils as Affected by Rootstock. Foods 2020, 9, 241. [Google Scholar] [CrossRef]
- Jirovetz, L.; Buchbauer, G.; Stoilova, I.; Stoyanova, A.; Krastanov, A.; Schmidt, E. Chemical Composition and Antioxidant Properties of Clove Leaf Essential Oil. J. Agric. Food Chem. 2006, 54, 6303–6307. [Google Scholar] [CrossRef] [PubMed]
- Marzoug, H.N.B.; Romdhane, M.; Lebrihi, A.; Mathieu, F.; Couderc, F.; Abderraba, M.; Khouja, M.L.; Bouajila, J. Eucalyptus oleosa Essential Oils: Chemical Composition and Antimicrobial and Antioxidant Activities of the Oils from Different Plant Parts (Stems, Leaves, Flowers and Fruits). Molecules 2011, 16, 1695–1709. [Google Scholar] [CrossRef]
- Tschiggerl, C. Investigation of the Volatile Fraction of Rosemary Infusion Extracts. Sci. Pharm. 2010, 78, 483–492. [Google Scholar] [CrossRef]
- Baritaux, O.; Richard, H.; Touche, J.; Derbesy, M. Effects of drying and storage of herbs and spices on the essential oil. Part I. Basil, ocimum basilicum L. Flavour Fragr. J. 1992, 7, 267–271. [Google Scholar] [CrossRef]
- Najafian, S. Storage conditions affect the essential oil composition of cultivated Balm Mint Herb (Lamiaceae) in Iran. Ind. Crops Prod. 2014, 52, 575–581. [Google Scholar] [CrossRef]
- Pratap Singh, A.; Fathordoobady, F.; Guo, Y.; Singh, A.; Kitts, D.D. Antioxidants help favorably regulate the kinetics of lipid peroxidation, polyunsaturated fatty acids degradation and acidic cannabinoids decarboxylation in hempseed oil. Sci. Rep. 2020, 10, 10567. [Google Scholar] [CrossRef]
- Pollard, D.; Fakayode, S.O.; Norman, V.; Stephenson, J.; Elzey, B. Purity Analysis of Adulterated Essential Oils by FT-IR Spectroscopy and Partial-Least-Squares Regression. Spectroscopy 2016, 31, 26–37. [Google Scholar]
- Gende, L.B.; Floris, I.; Fritz, R.; Eguaras, M.J. Antimicrobial activity of cinnamon (Cinnamomum zeylanicum) essential oil and its main components against paenibacillus larvae from argentine. Bull. Insectology 2008, 61, 1–4. [Google Scholar]
- Nagaraju, P.G.; Sengupta, P.; Chicgovinda, P.P.; Rao, P.J. Nanoencapsulation of clove oil and study of physicochemical properties, cytotoxic, hemolytic, and antioxidant activities. J. Food Process Eng. 2021, 44, e13645. [Google Scholar] [CrossRef]
- Jarzębski, M.; Smułek, W.; Siejak, P.; Rezler, R.; Pawlicz, J.; Trzeciak, T.; Jarzębska, M.; Majchrzak, O.; Kaczorek, E.; Kazemian, P.; et al. Aesculus hippocastanum L. as a Stabilizer in Hemp Seed Oil Nanoemulsions for Potential Biomedical and Food Applications. Int. J. Mol. Sci. 2021, 22, 887. [Google Scholar] [CrossRef] [PubMed]
- Tarhan, İ. A comparative study of ATR-FTIR, UV–visible and fluorescence spectroscopy combined with chemometrics for quantification of squalene in extra virgin olive oils. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2020, 241, 118714. [Google Scholar] [CrossRef]
- Kongbonga, Y.G.M.; Ghalila, H.; Onana, M.B.; Majdi, Y.; Lakhdar, Z.B.; Mezlini, H.; Sevestre-Ghalila, S. Characterization of Vegetable Oils by Fluorescence Spectroscopy. Food Nutr. Sci. 2011, 2, 692–699. [Google Scholar] [CrossRef]
- Wiktor, A.; Mandal, R.; Singh, A.; Pratap Singh, A. Pulsed Light treatment below a Critical Fluence (3.82 J/cm2) minimizes photo-degradation and browning of a model Phenolic (Gallic Acid) Solution. Foods 2019, 8, 380. [Google Scholar] [CrossRef]
- Siahbalaei, R.; Kavoosi, G.; Shakeri, R. In vitro antioxidant and antidiabetic activity of essential oils encapsulated in gelatin-pectin particles against sugar, lipid and protein oxidation and amylase and glucosidase activity. Food Sci. Nutr. 2020, 8, 6457–6466. [Google Scholar] [CrossRef] [PubMed]
- Kamrudi, N.; Akbari, S.; Haghighat Kish, M. Enhanced control release of thyme essential oils from electrospun nanofiber/polyamidoamine dendritic polymer for antibacterial platforms. Polym. Adv. Technol. 2020, 31, 1719–1731. [Google Scholar] [CrossRef]
- Sharifi-Rad, J.; Sureda, A.; Tenore, G.; Daglia, M.; Sharifi-Rad, M.; Valussi, M.; Tundis, R.; Sharifi-Rad, M.; Loizzo, M.; Ademiluyi, A.; et al. Biological Activities of Essential Oils: From Plant Chemoecology to Traditional Healing Systems. Molecules 2017, 22, 70. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Ren, L.; Li, M.; Qian, J.; Fan, J.; Du, B. Effects of clove essential oil and eugenol on quality and browning control of fresh-cut lettuce. Food Chem. 2017, 214, 432–439. [Google Scholar] [CrossRef]
- Escamilla-García, M.; Calderón-Domínguez, G.; Chanona-Pérez, J.J.; Mendoza-Madrigal, A.G.; Di Pierro, P.; García-Almendárez, B.E.; Amaro-Reyes, A.; Regalado-González, C. Physical, structural, barrier, and antifungal characterization of chitosan-zein edible films with added essential oils. Int. J. Mol. Sci. 2017, 18, 2370. [Google Scholar] [CrossRef]
- Kalemba, D.; Kunicka, A. Antibacterial and Antifungal Properties of Essential Oils. Curr. Med. Chem. 2003, 10, 813–829. [Google Scholar] [CrossRef]
- Haba, E.; Bouhdid, S.; Torrego-Solana, N.; Marqués, A.M.; Espuny, M.J.; García-Celma, M.J.; Manresa, A. Rhamnolipids as emulsifying agents for essential oil formulations: Antimicrobial effect against Candida albicans and methicillin-resistant Staphylococcus aureus. Int. J. Pharm. 2014, 476, 134–141. [Google Scholar] [CrossRef] [PubMed]
- Alexa, V.T.; Szuhanek, C.; Cozma, A.; Galuscan, A.; Borcan, F.; Obistioiu, D.; Dehelean, C.A.; Jumanca, D. Natural Preparations Based on Orange, Bergamot and Clove Essential Oils and Their Chemical Compounds as Antimicrobial Agents. Molecules 2020, 25, 5502. [Google Scholar] [CrossRef] [PubMed]
- Bahurmiz, O.M.; Ahmad, R.; Ismail, N.; Adzitey, F.; Sulaiman, S.-F. Antimicrobial Activity of Selected Essential Oils on Pseudomonas Species Associated with Spoilage of Fish with Emphasis on Cinnamon Essential Oil. J. Aquat. Food Prod. Technol. 2020, 29, 789–800. [Google Scholar] [CrossRef]
- Purkait, S.; Bhattacharya, A.; Bag, A.; Chattopadhyay, R.R. Synergistic antibacterial, antifungal and antioxidant efficacy of cinnamon and clove essential oils in combination. Arch. Microbiol. 2020, 202, 1439–1448. [Google Scholar] [CrossRef]
- Messaoudi Moussii, I.; Nayme, K.; Timinouni, M.; Jamaleddine, J.; Filali, H.; Hakkou, F. Synergistic antibacterial effects of Moroccan Artemisia herba alba, Lavandula angustifolia and Rosmarinus officinalis essential oils. Synergy 2020, 10, 100057. [Google Scholar] [CrossRef]
- Elcocks, E.R.; Spencer-Phillips, P.T.N.; Adukwu, E.C. Rapid bactericidal effect of cinnamon bark essential oil against Pseudomonas aeruginosa. J. Appl. Microbiol. 2020, 128, 1025–1037. [Google Scholar] [CrossRef]
- Kozics, K.; Bučková, M.; Puškárová, A.; Kalászová, V.; Cabicarová, T.; Pangallo, D. The Effect of Ten Essential Oils on Several Cutaneous Drug-Resistant Microorganisms and Their Cyto/Genotoxic and Antioxidant Properties. Molecules 2019, 24, 4570. [Google Scholar] [CrossRef]
- Yen, P.P.; Pratap-Singh, A. Vacuum microwave dehydration decreases volatile concentration and soluble protein content of pea (Pisum sativum L.) protein. J. Sci. Food Agric. 2021, 101, 167–178. [Google Scholar] [CrossRef] [PubMed]
- Brosio, E.; Gianferri, R. Basic NMR in foods characterization. In Basic NMR in Foods Characterization; Brosio, E., Ed.; Reseach Signpost: Karela, India, 2009; pp. 9–37. [Google Scholar]
- Weglarz, W.P.; Haranczyk, H. Two-dimensional analysis of the nuclear relaxation function in the time domain: The program CracSpin. J. Phys. D Appl. Phys. 2000, 33, 1909–1920. [Google Scholar] [CrossRef]
- Pacholak, A.; Burlaga, N.; Guzik, U.; Kaczorek, E. Investigation of the bacterial cell envelope nanomechanical properties after long-term exposure to nitrofurans. J. Hazard. Mater. 2021, 407, 124352. [Google Scholar] [CrossRef]
- Smułek, W.; Bielan, Z.; Pacholak, A.; Zdarta, A.; Zgoła-Grześkowiak, A.; Zielińska-Jurek, A.; Kaczorek, E. Nitrofurazone Removal from Water Enhanced by Coupling Photocatalysis and Biodegradation. Int. J. Mol. Sci. 2021, 22, 2186. [Google Scholar] [CrossRef] [PubMed]
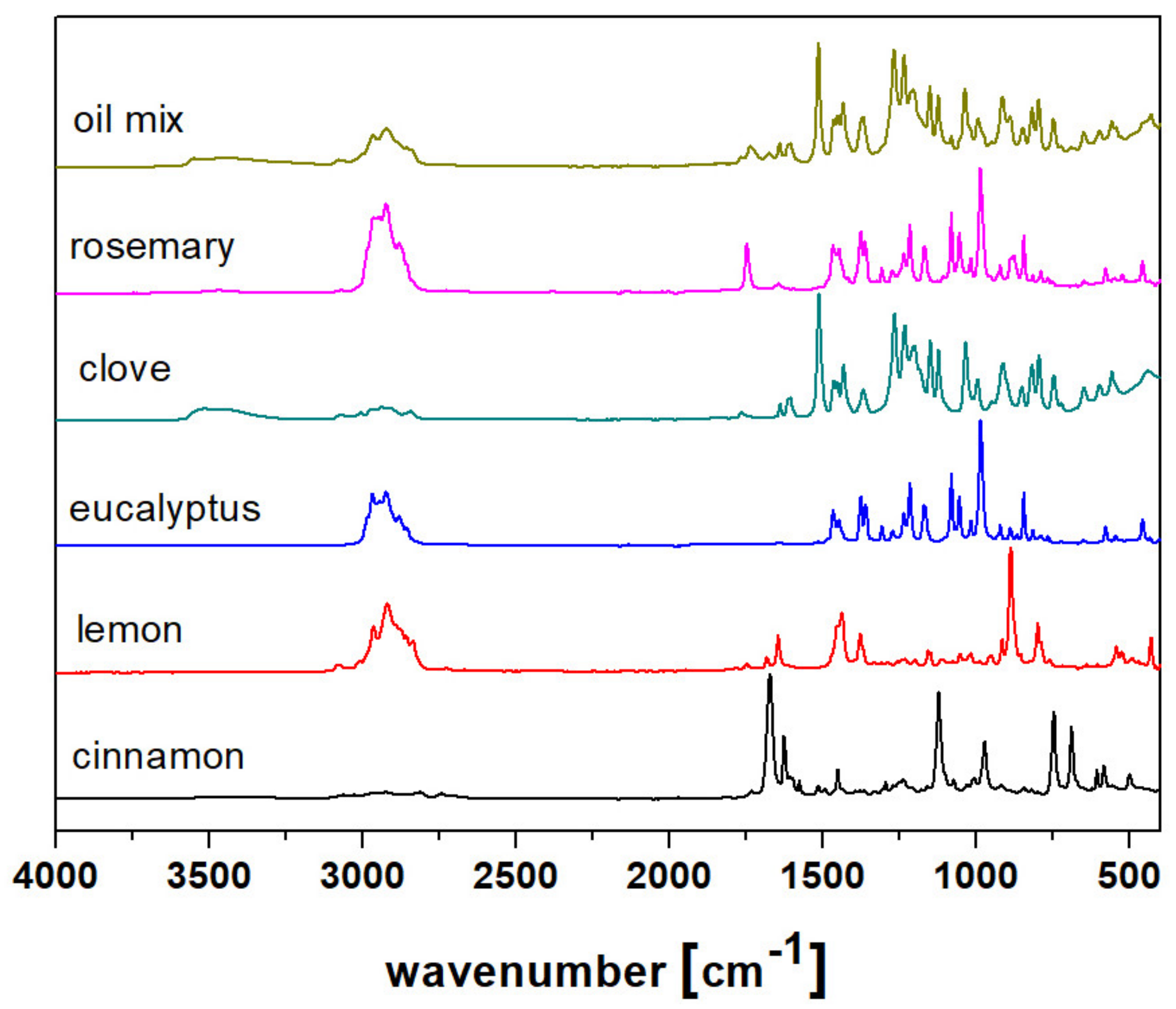
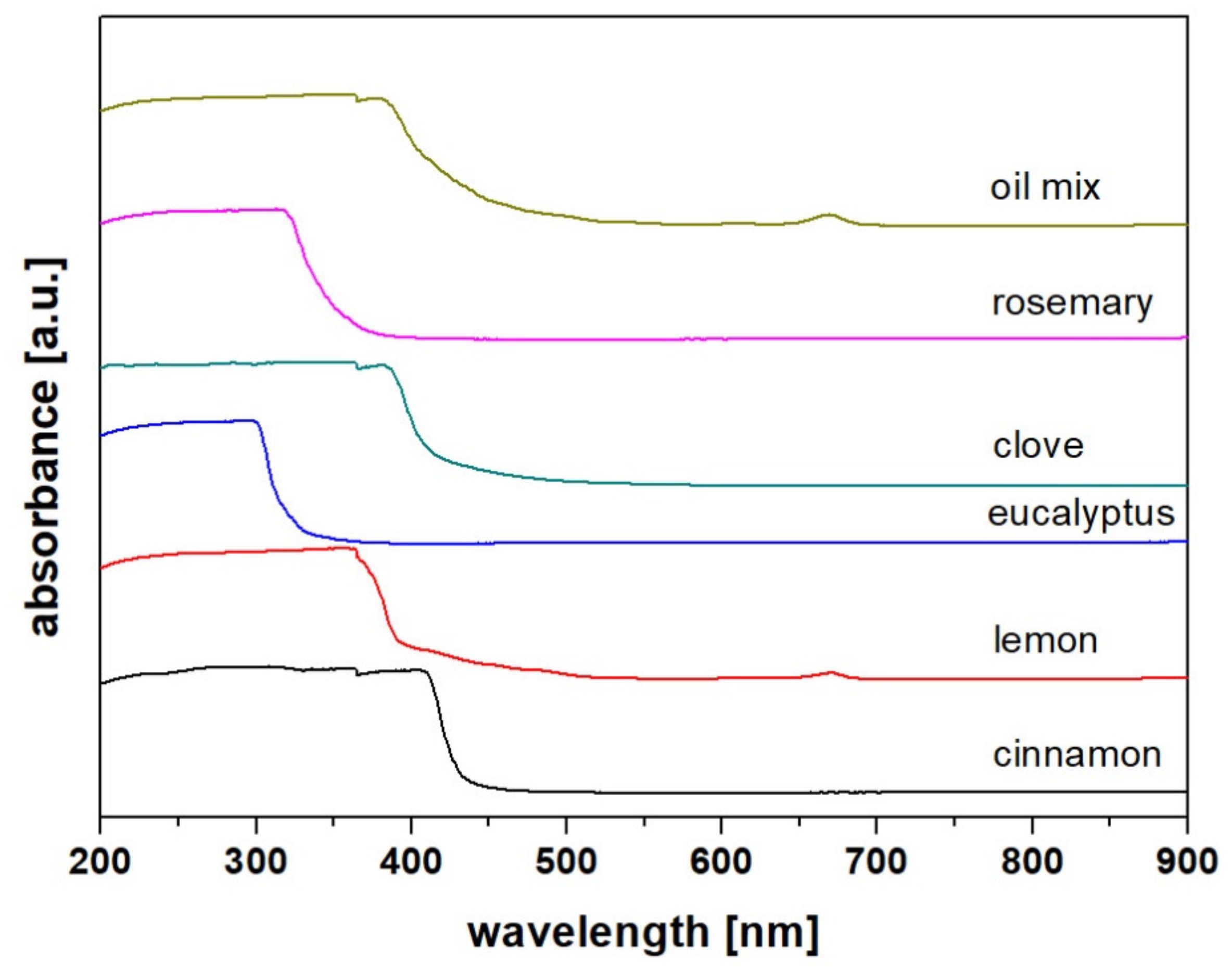
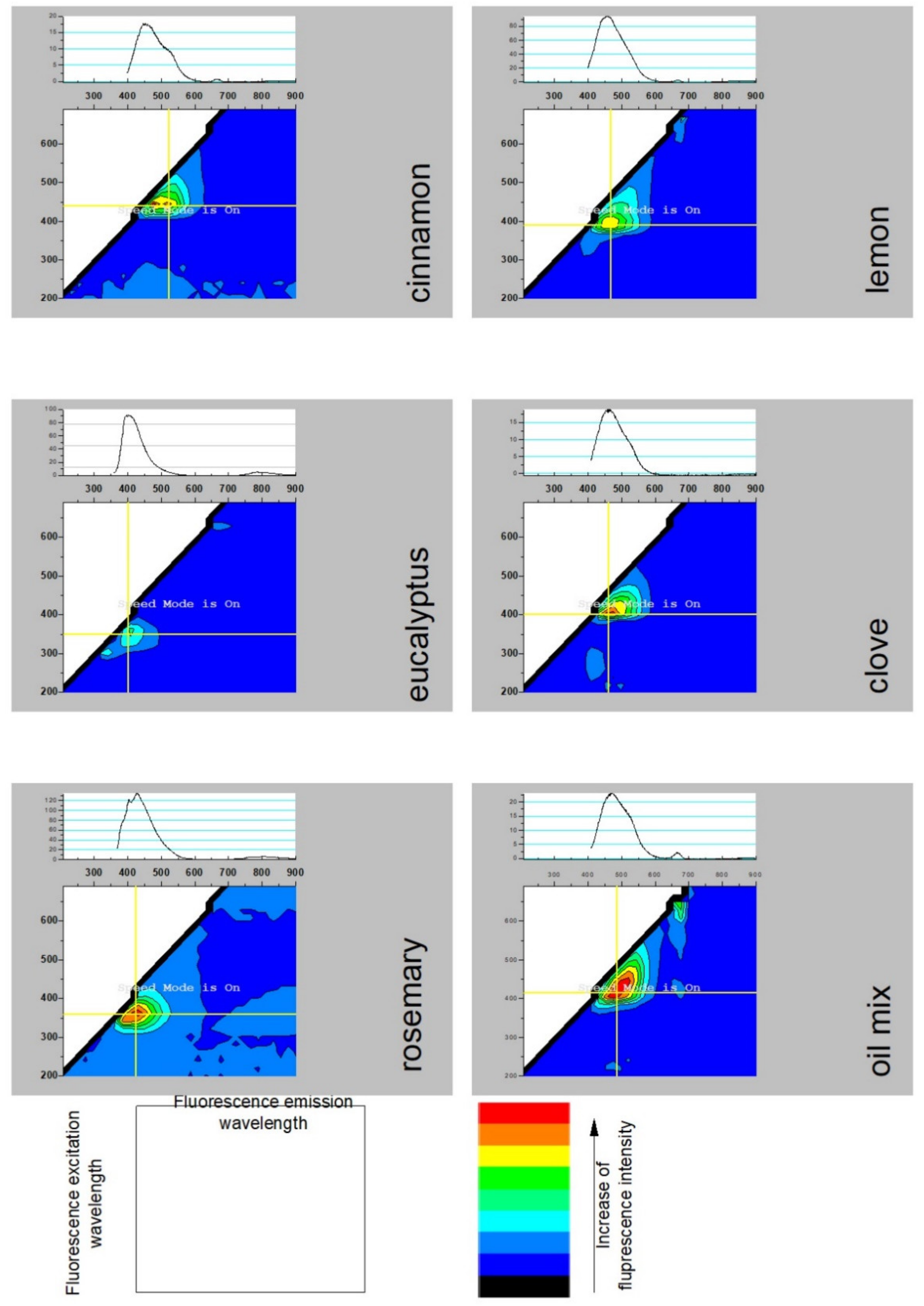

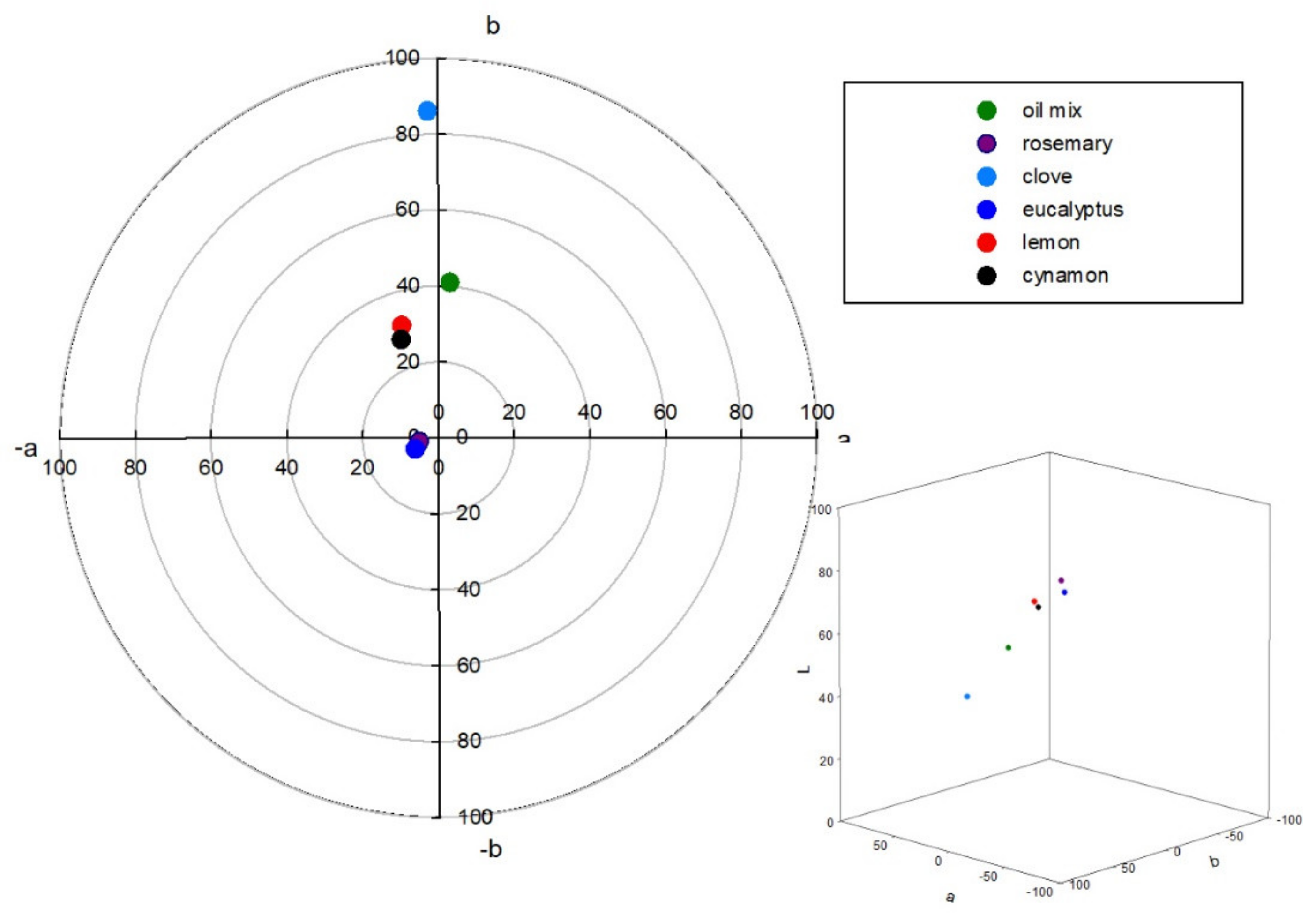
| Compound | Kovat’s Index | Fresh | After 1 Month | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Oil Mix | Rosemary | Lemon | Clove | Eucalyptus | Cinnamon | Oil Mix | Rosemary | Clove | Cinnamon | ||
| 1-Hexen-3-ol | 851 | 1.11 | |||||||||
| 2-Thujene | 873 | 0.45 | 4.04 | ||||||||
| α-Pinene | 939 | 16.46 | 30.06 | 11.66 | 20.65 | 9.34 | 0.13 | ||||
| Camphene | 953 | 1.41 | 8.66 | 0.5 | 1.2 | ||||||
| Benzaldehyde | 961 | 1.44 | 1 | ||||||||
| β-Pinene | 980 | 2.05 | 13.28 | 28.48 | 7.95 | 2.12 | 3.11 | 0.14 | |||
| 2-Carene | 1001 | 0.65 | 0.48 | ||||||||
| o-Cymene | 1022 | 17.94 | 0.5 | ||||||||
| m-Cymene | 1025 | 0.21 | 0.06 | 3.72 | |||||||
| Sylvestrene | 1027 | 28.69 | |||||||||
| Limonene | 1031 | 30.91 | 11.6 | 1.05 | 0.44 | ||||||
| Eucaliptol | 1033 | 23.66 | 60.98 | 10.79 | 23.7 | 0.18 | 3.67 | ||||
| γ-Terpine | 1062 | 3.43 | 2.12 | 11.01 | 8.46 | 0.8 | 3.07 | 1.74 | 0.05 | ||
| α-Terpinolene | 1088 | 1.36 | 2.15 | 0.89 | |||||||
| Isoterpinolene | 1090 | 7.64 | 2.07 | ||||||||
| Linalool | 1098 | 7.53 | 1.24 | 0.54 | 11.25 | 18.69 | 5.76 | 17.71 | |||
| 1-Ethyl-4-Isopropyl-Cyclohexyl 2-Hydroperfluorobutanoate | 1120 | 1.21 | |||||||||
| Camphor | 1143 | 0.94 | 10.06 | 1.98 | 26.99 | ||||||
| Borneol | 1165 | 2.02 | 11.23 | ||||||||
| α-Terpineol | 1189 | 0.35 | 0.36 | 0.59 | 1.08 | 1.08 | 2.54 | 5.92 | 2.13 | ||
| Linalyl Acetate | 1248 | 18.57 | |||||||||
| Cinnamaldehyde | 1266 | 1.95 | 30.98 | 53.24 | |||||||
| Bornyl Acetate | 1285 | 2.33 | 5.79 | ||||||||
| Carvacrol | 1298 | 0.69 | 1.34 | ||||||||
| β-Citral | 1316 | 2.09 | |||||||||
| α-Terpineol Acetate | 1340 | 3.05 | 1.76 | ||||||||
| α-Citral | 1341 | 0.33 | 2.47 | 0.56 | |||||||
| Eugenol | 1356 | 16.69 | 60.4 | 2 | 25.69 | 60.49 | 3.99 | ||||
| Copaene | 1376 | 0.42 | 0.72 | 1.07 | 2.19 | 0.1 | 1.94 | ||||
| β-Caryophyllene | 1404 | 4.28 | 3.85 | 0.22 | 27.18 | 6.66 | 9.95 | 8.54 | 28.82 | 12.47 | |
| α-Caryophyllene | 1418 | 0.59 | 0.59 | 6.68 | 0.92 | 1.71 | 1.21 | 7.39 | |||
| Isoeugenol | 1447 | 0.35 | 2.56 | 0.39 | 1.42 | ||||||
| Benzalmalonic Dialdehyde | 1454 | 1.42 | |||||||||
| Acetyleugenol | 1524 | 0.62 | 1.2 | 0.88 | 1.04 | ||||||
| Linalyl Anthranilate | 2157 | 7.26 | 0.48 | ||||||||
| Sample | R1 (s−1) | R2 (s−1) | pi |
|---|---|---|---|
| Oil mix | 1.585 ± 0.015 | 1.612 ± 0.012 | – |
| Rosemary | 1.215 ± 0.012 | 1.221 ± 0.031 | 0.26 ± 0.02 |
| Clove | 2.383 ± 0.008 | 2.403 ± 0.027 | 0.13 ± 0.02 |
| eucalyptus | 1.274 ± 0.009 | 1.241 ± 0.019 | 0.25 ± 0.03 |
| Lemon | 1.705 ± 0.013 | 1.724 ± 0.021 | 0.18 ± 0.03 |
| Cinnamon | 1.283 ± 0.004 | 1.332 ± 0.019 | 0.24 ± 0.02 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Siejak, P.; Smułek, W.; Fathordobady, F.; Grygier, A.; Baranowska, H.M.; Rudzińska, M.; Masewicz, Ł.; Jarzębska, M.; Nowakowski, P.T.; Makiej, A.; et al. Multidisciplinary Studies of Folk Medicine “Five Thieves’ Oil” (Olejek Pięciu Złodziei) Components. Molecules 2021, 26, 2931. https://doi.org/10.3390/molecules26102931
Siejak P, Smułek W, Fathordobady F, Grygier A, Baranowska HM, Rudzińska M, Masewicz Ł, Jarzębska M, Nowakowski PT, Makiej A, et al. Multidisciplinary Studies of Folk Medicine “Five Thieves’ Oil” (Olejek Pięciu Złodziei) Components. Molecules. 2021; 26(10):2931. https://doi.org/10.3390/molecules26102931
Chicago/Turabian StyleSiejak, Przemysław, Wojciech Smułek, Farahnaz Fathordobady, Anna Grygier, Hanna Maria Baranowska, Magdalena Rudzińska, Łukasz Masewicz, Małgorzata Jarzębska, Piotr T. Nowakowski, Aleksandra Makiej, and et al. 2021. "Multidisciplinary Studies of Folk Medicine “Five Thieves’ Oil” (Olejek Pięciu Złodziei) Components" Molecules 26, no. 10: 2931. https://doi.org/10.3390/molecules26102931
APA StyleSiejak, P., Smułek, W., Fathordobady, F., Grygier, A., Baranowska, H. M., Rudzińska, M., Masewicz, Ł., Jarzębska, M., Nowakowski, P. T., Makiej, A., Kazemian, P., Drobnik, P., Stachowiak, B., Jarzębski, M., & Pratap-Singh, A. (2021). Multidisciplinary Studies of Folk Medicine “Five Thieves’ Oil” (Olejek Pięciu Złodziei) Components. Molecules, 26(10), 2931. https://doi.org/10.3390/molecules26102931















