Dithieno[1,4]thiazines and Bis[1]benzothieno[1,4]thiazines—Organometallic Synthesis and Functionalization of Electron Density Enriched Congeners of Phenothiazine
Abstract
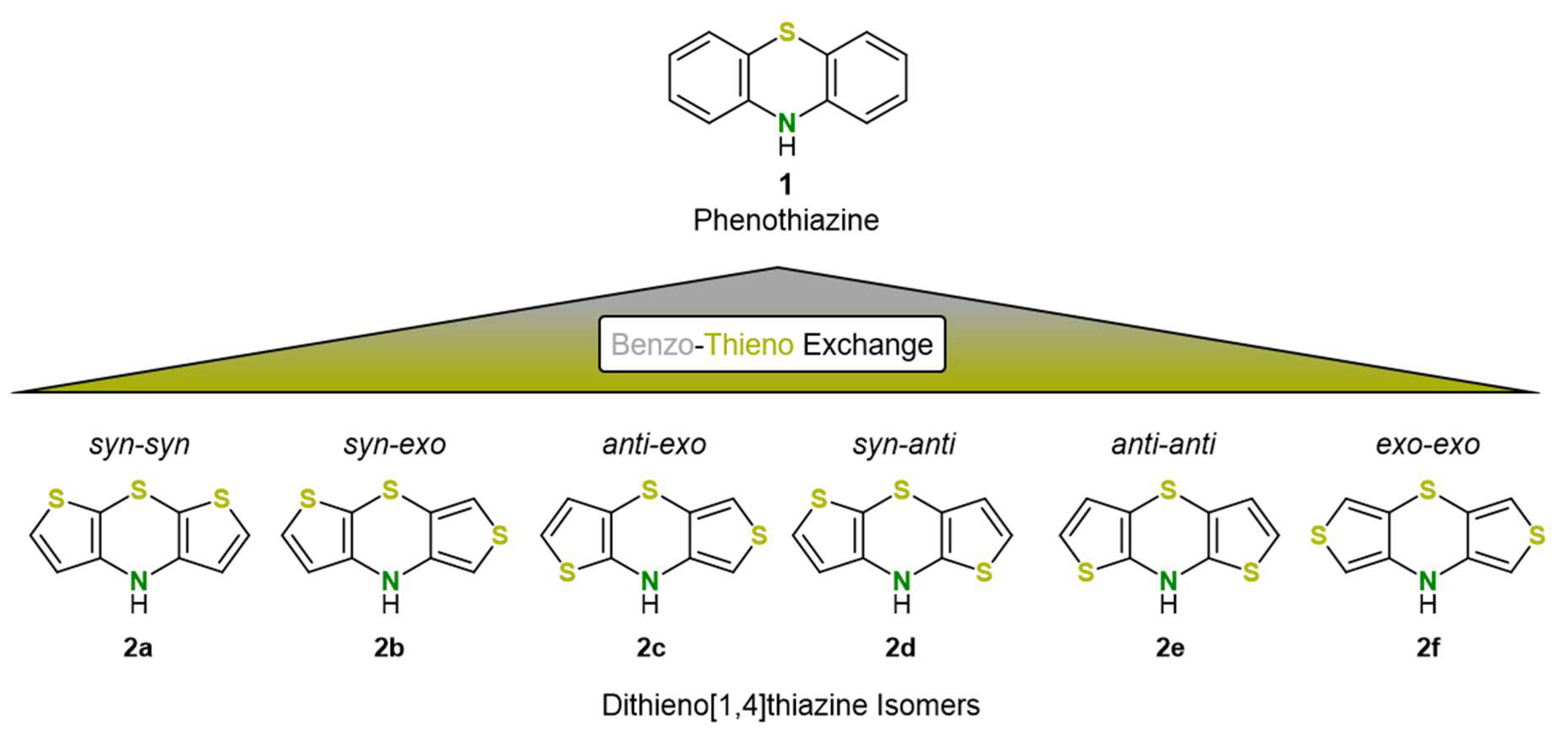
1. Introduction
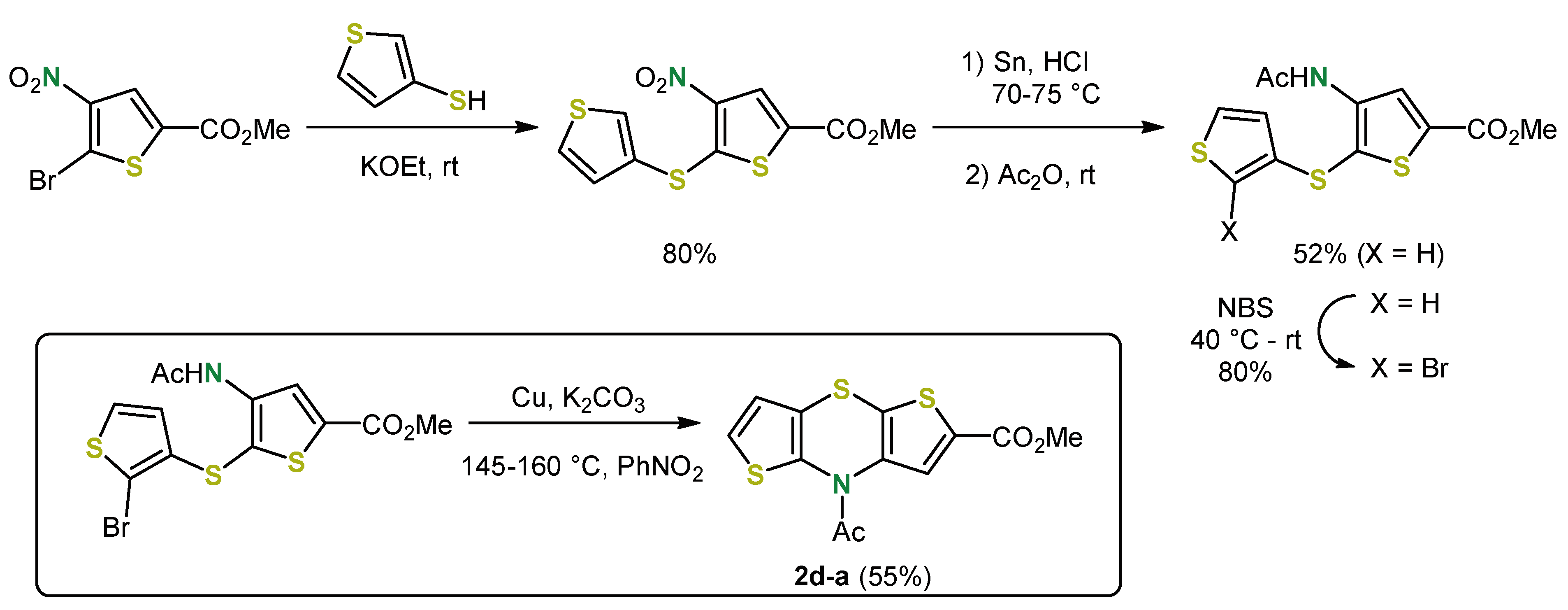
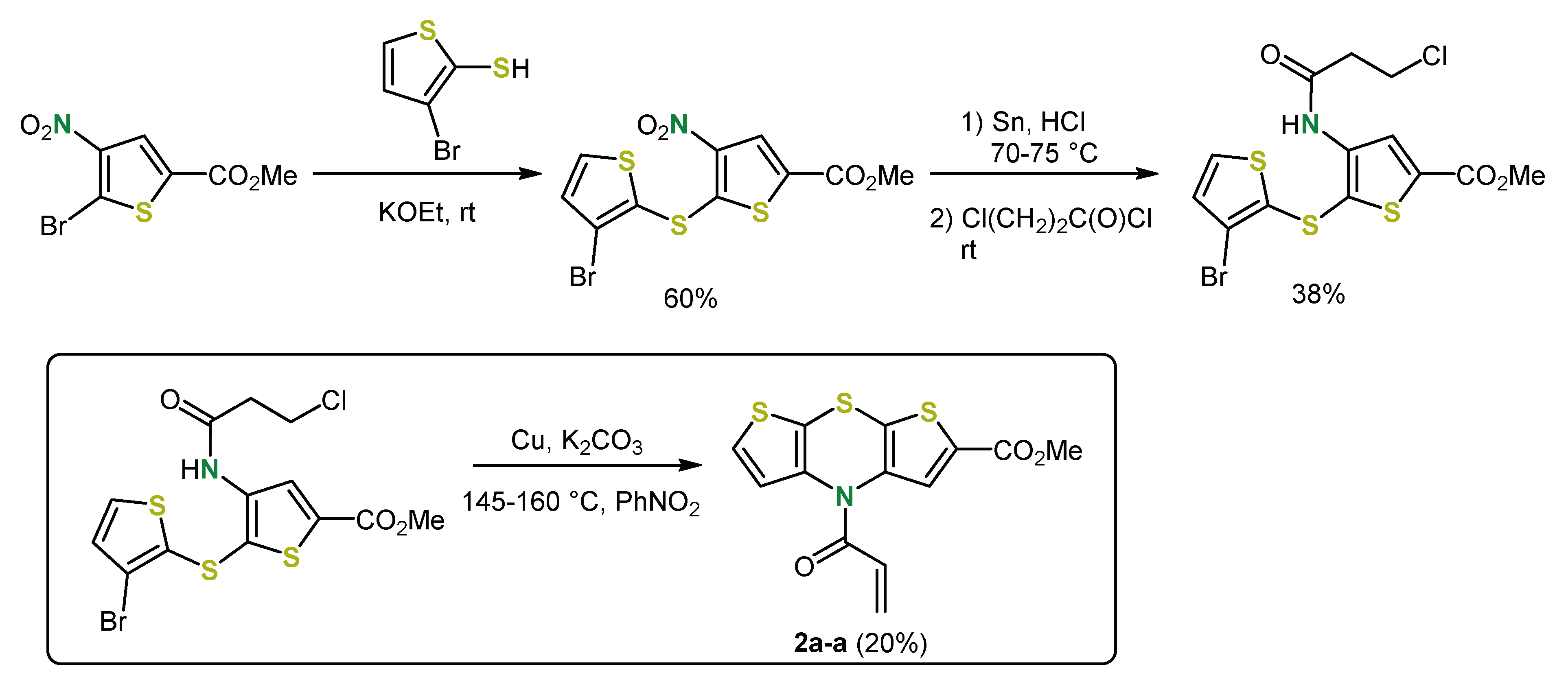
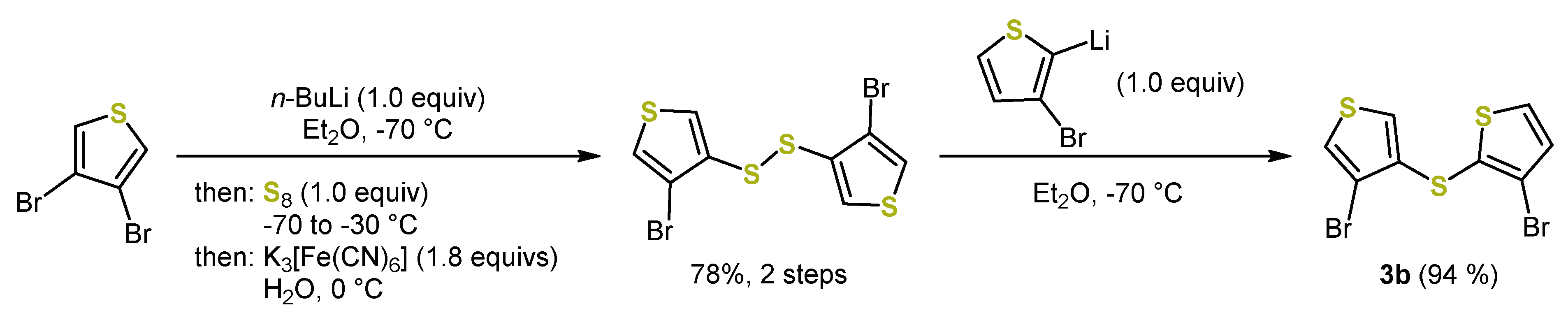
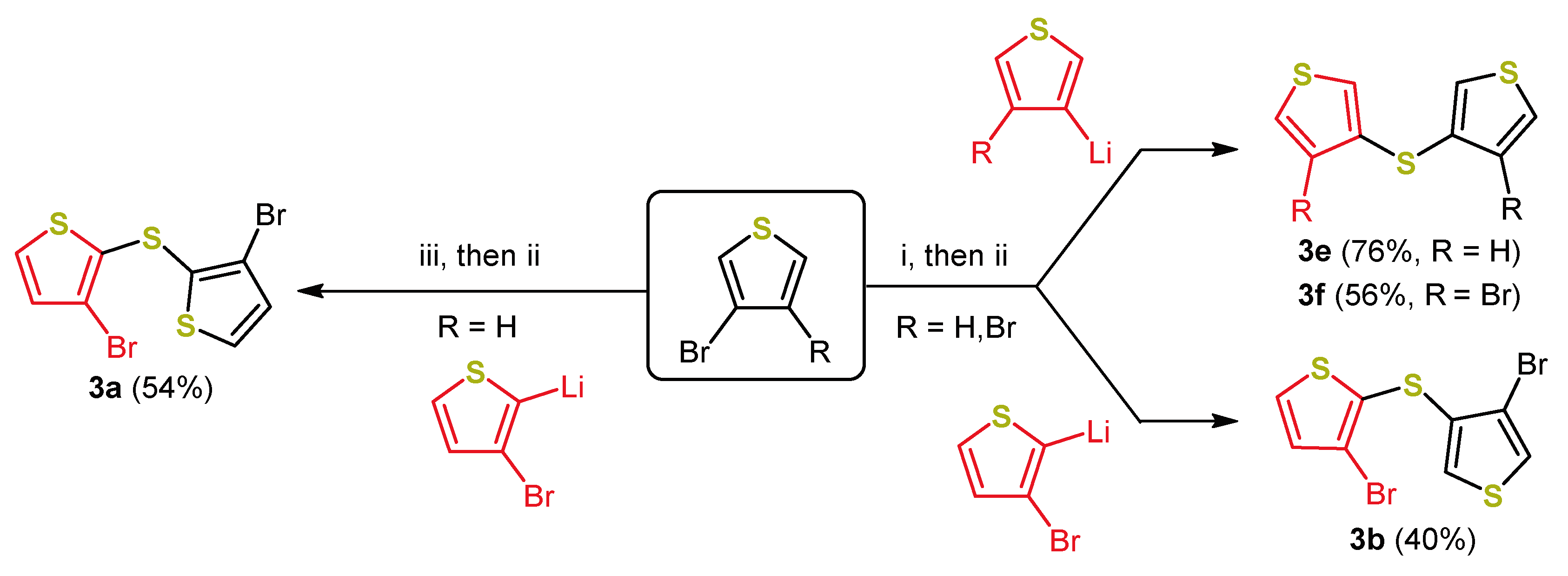
2. Early Syntheses of Dithieno[1,4]thiazines Based on Ullmann-Type Coupling
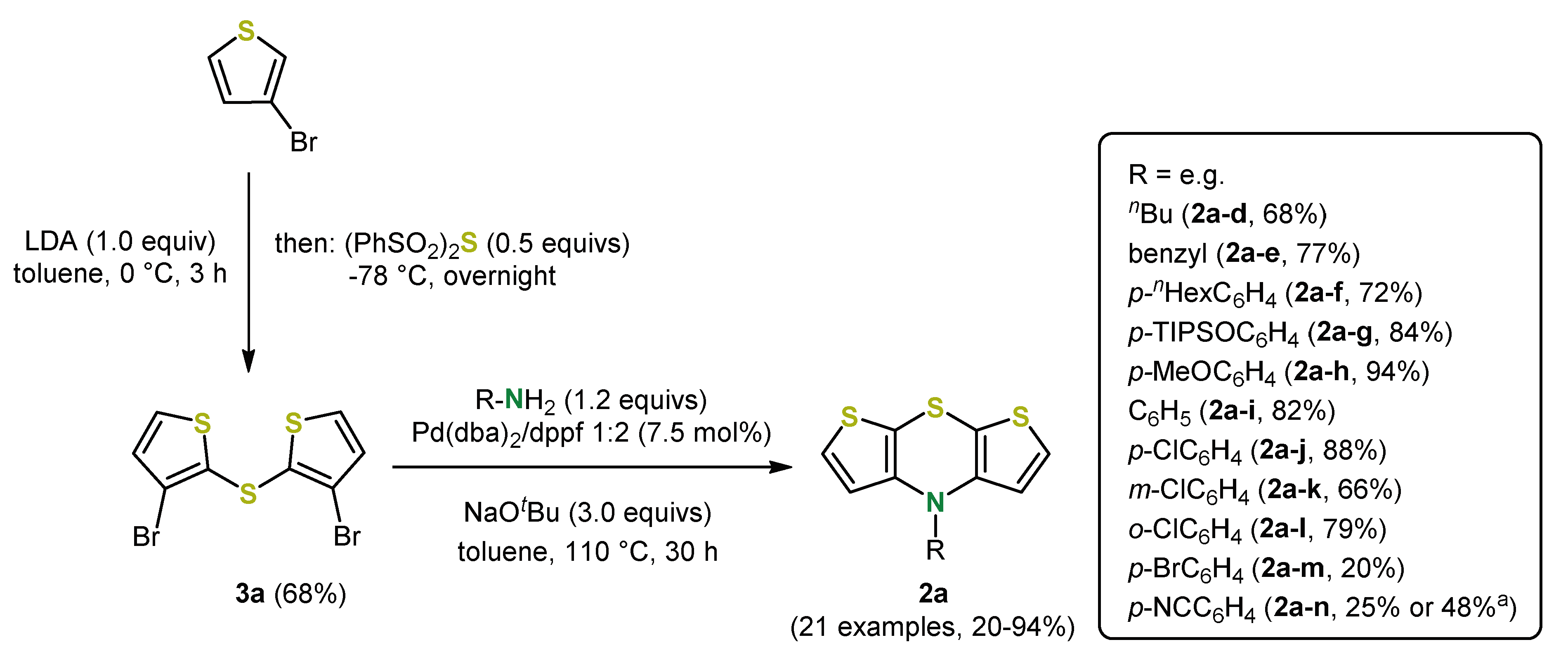
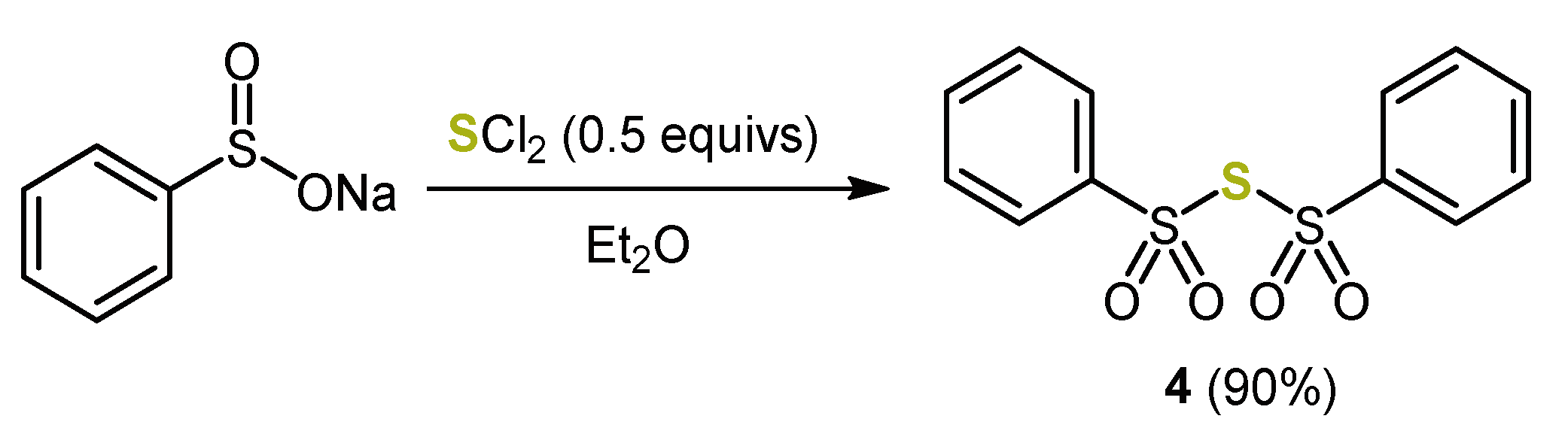
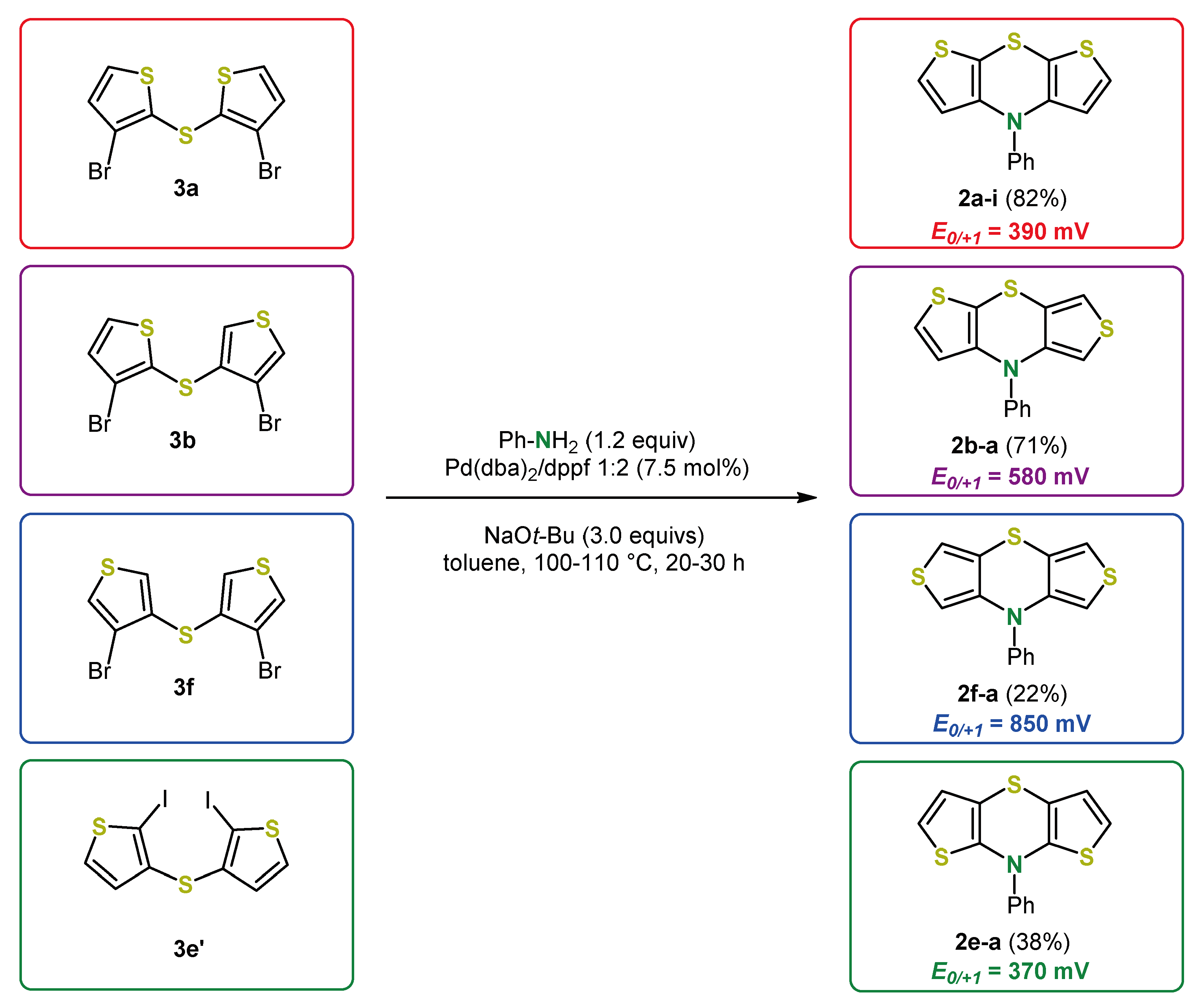
3. Syntheses of Dithieno[1,4]thiazines by Cyclizing Buchwald–Hartwig Coupling
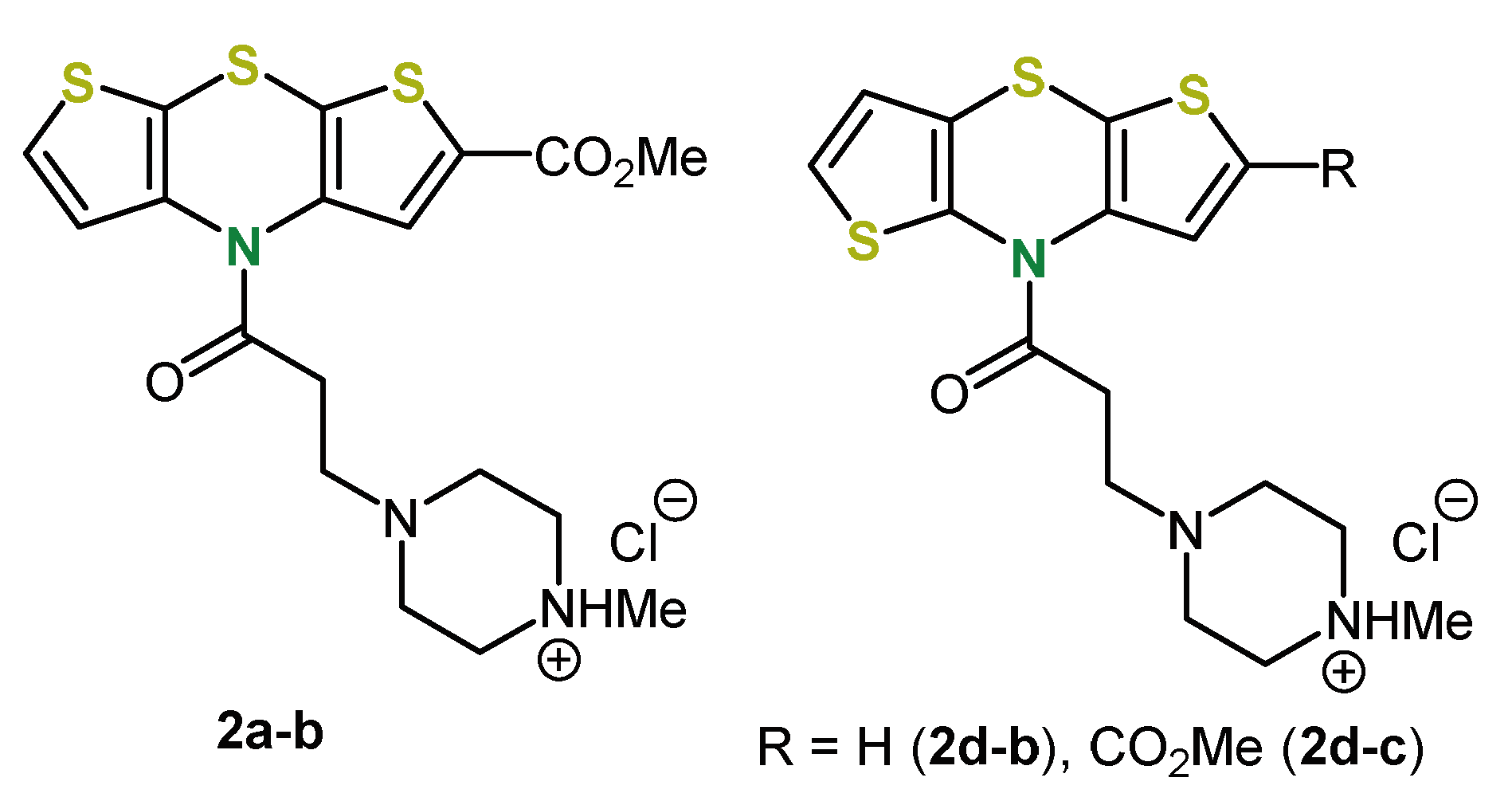
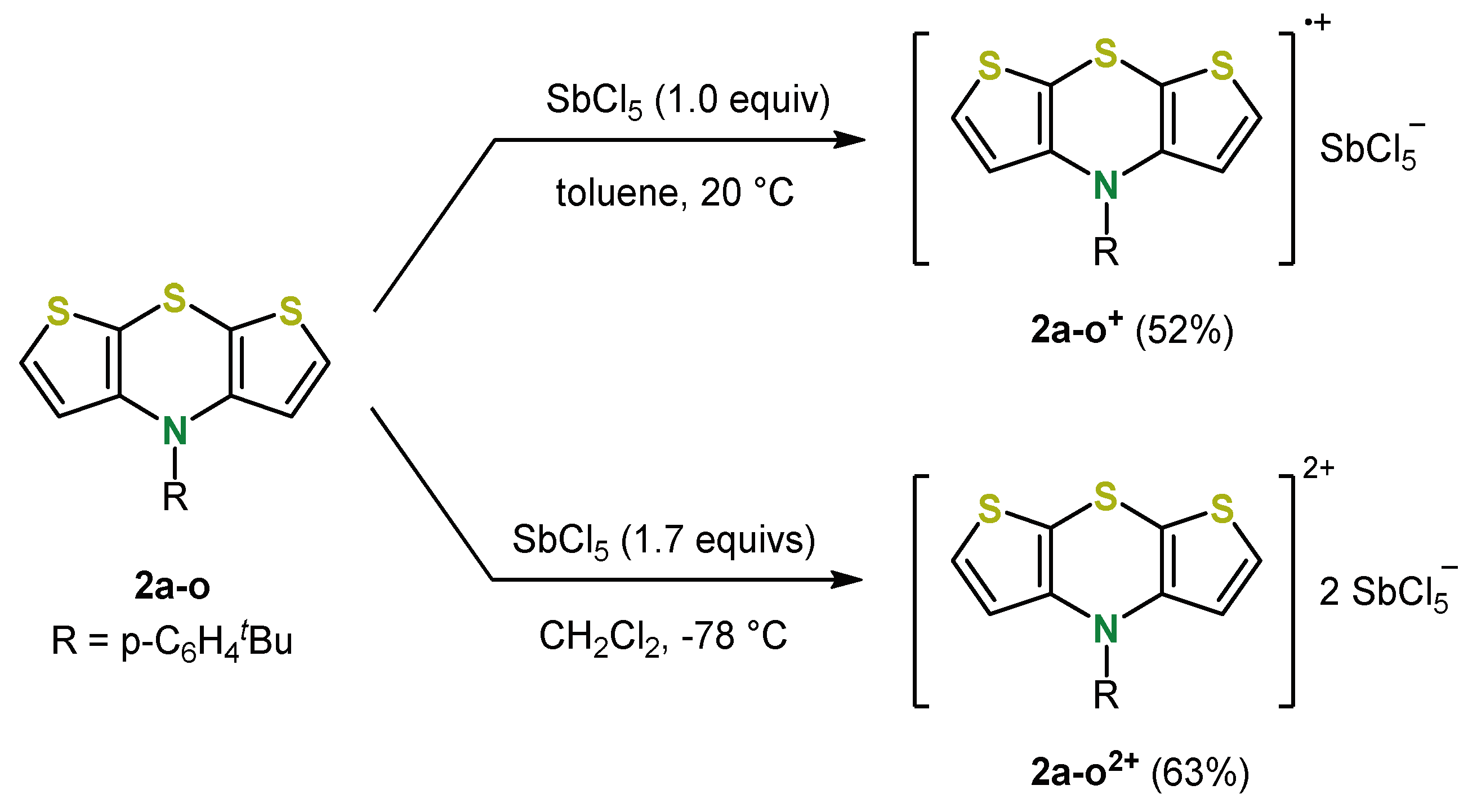
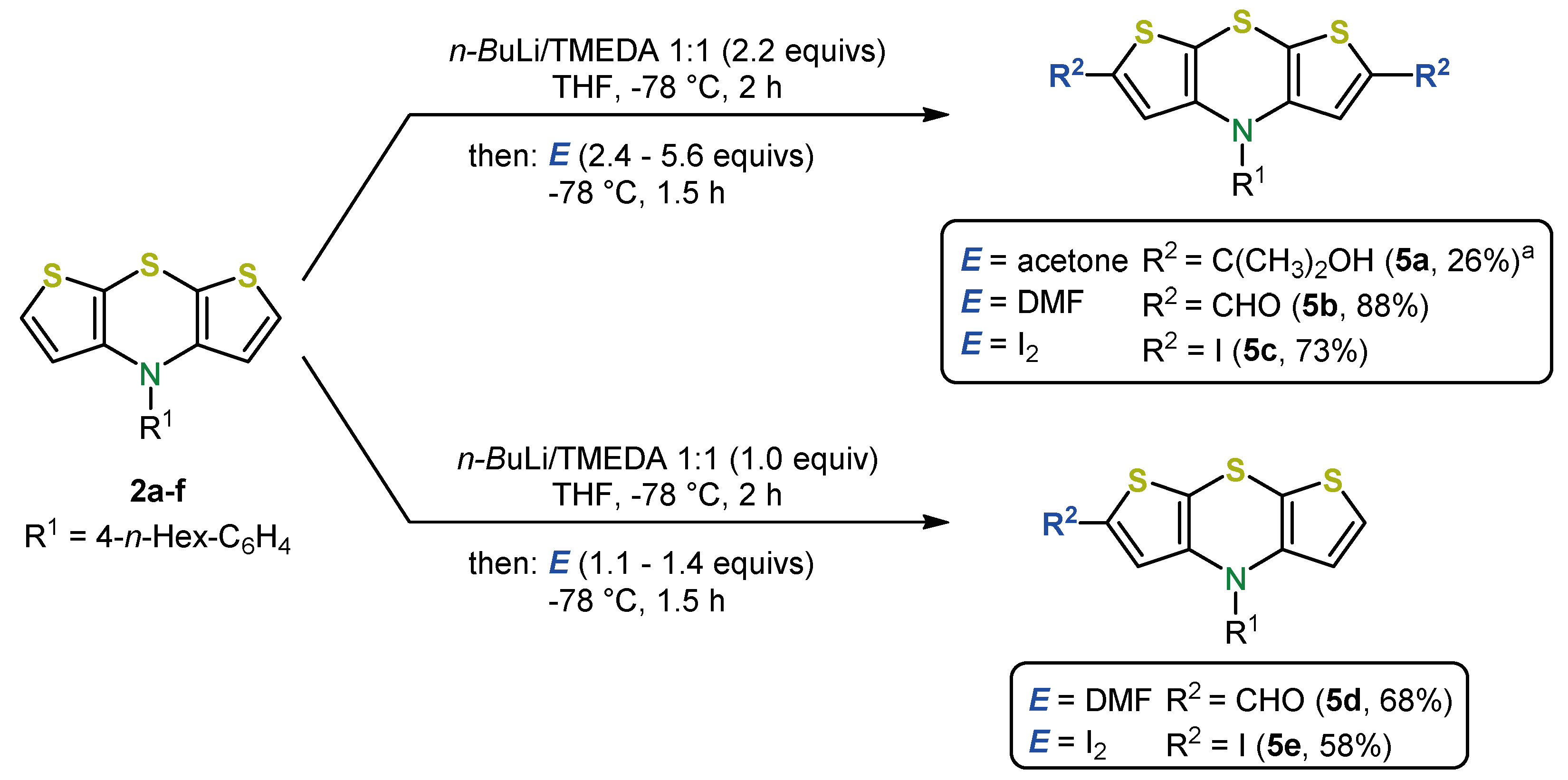
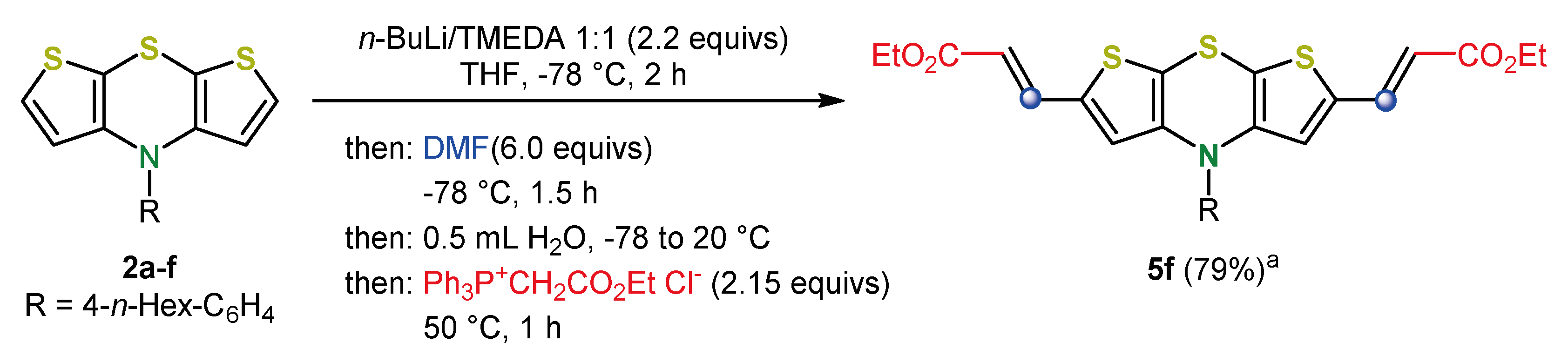
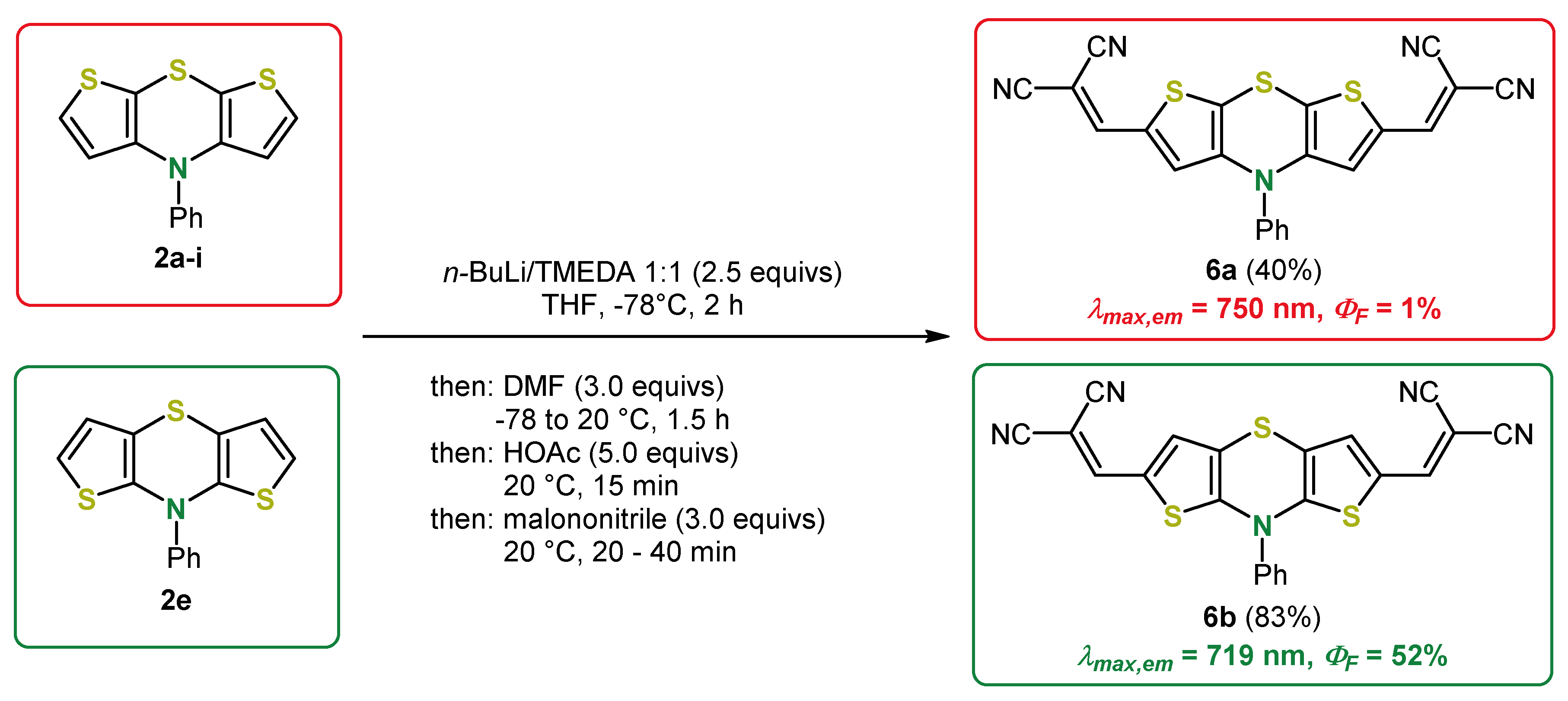
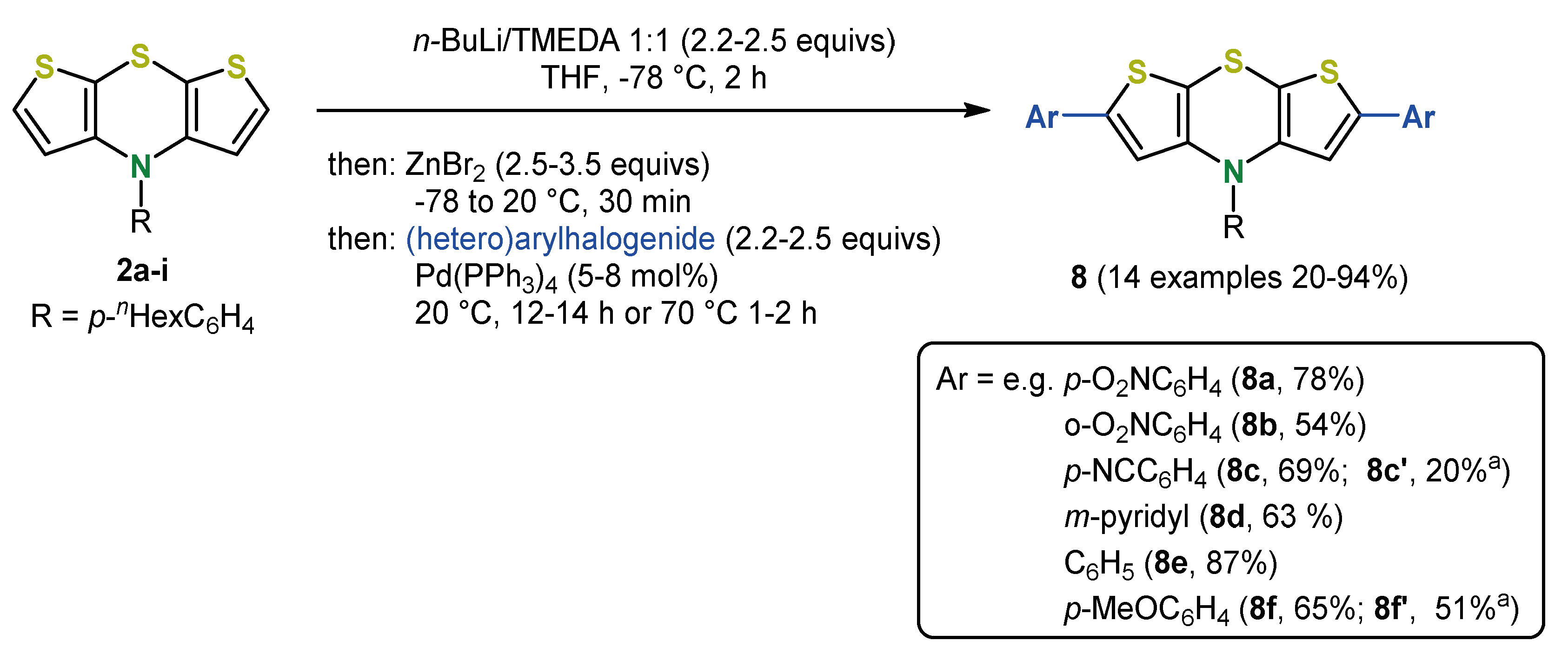
4. 2,6-Functionalization of Dithieno[1,4]thiazines: Expanding the π-Systems
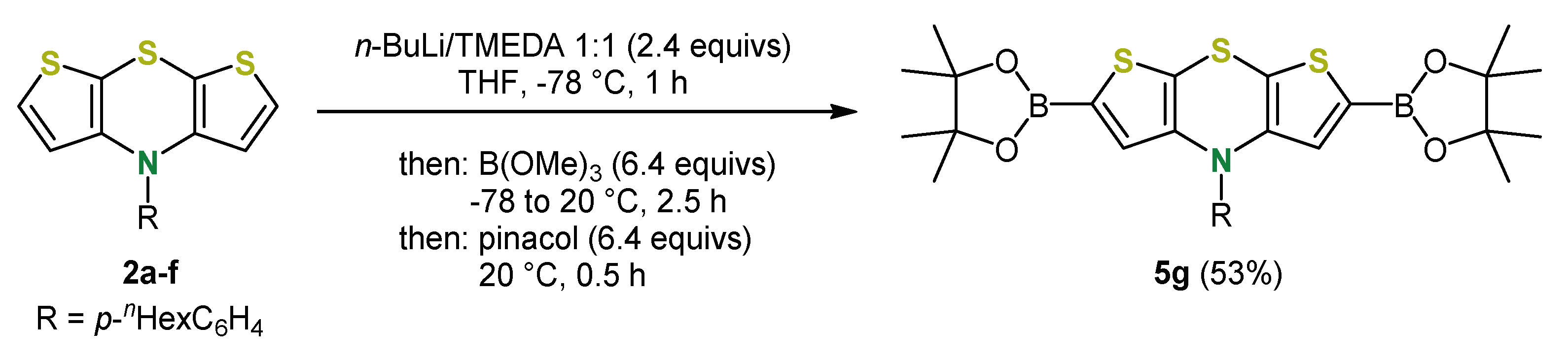
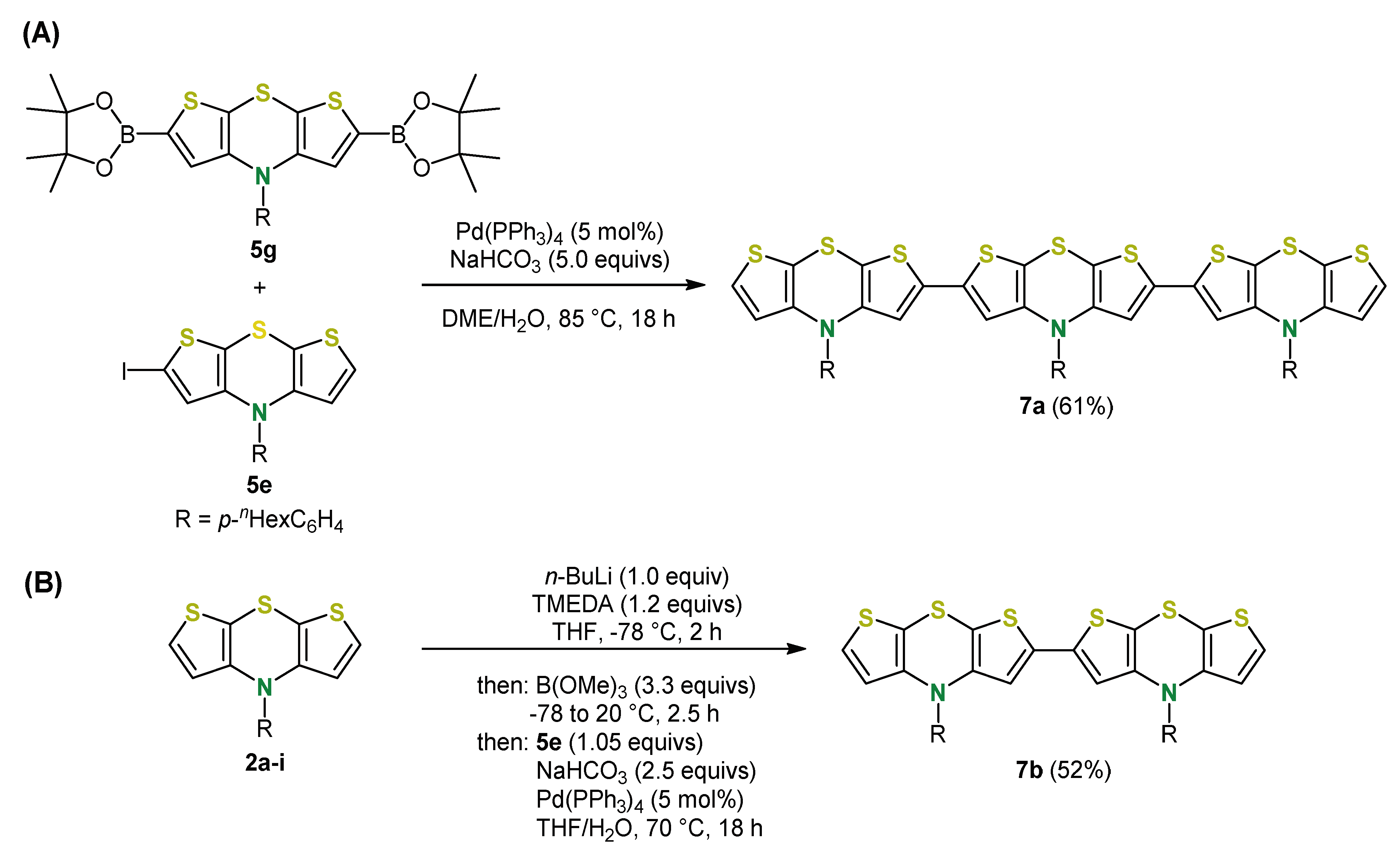
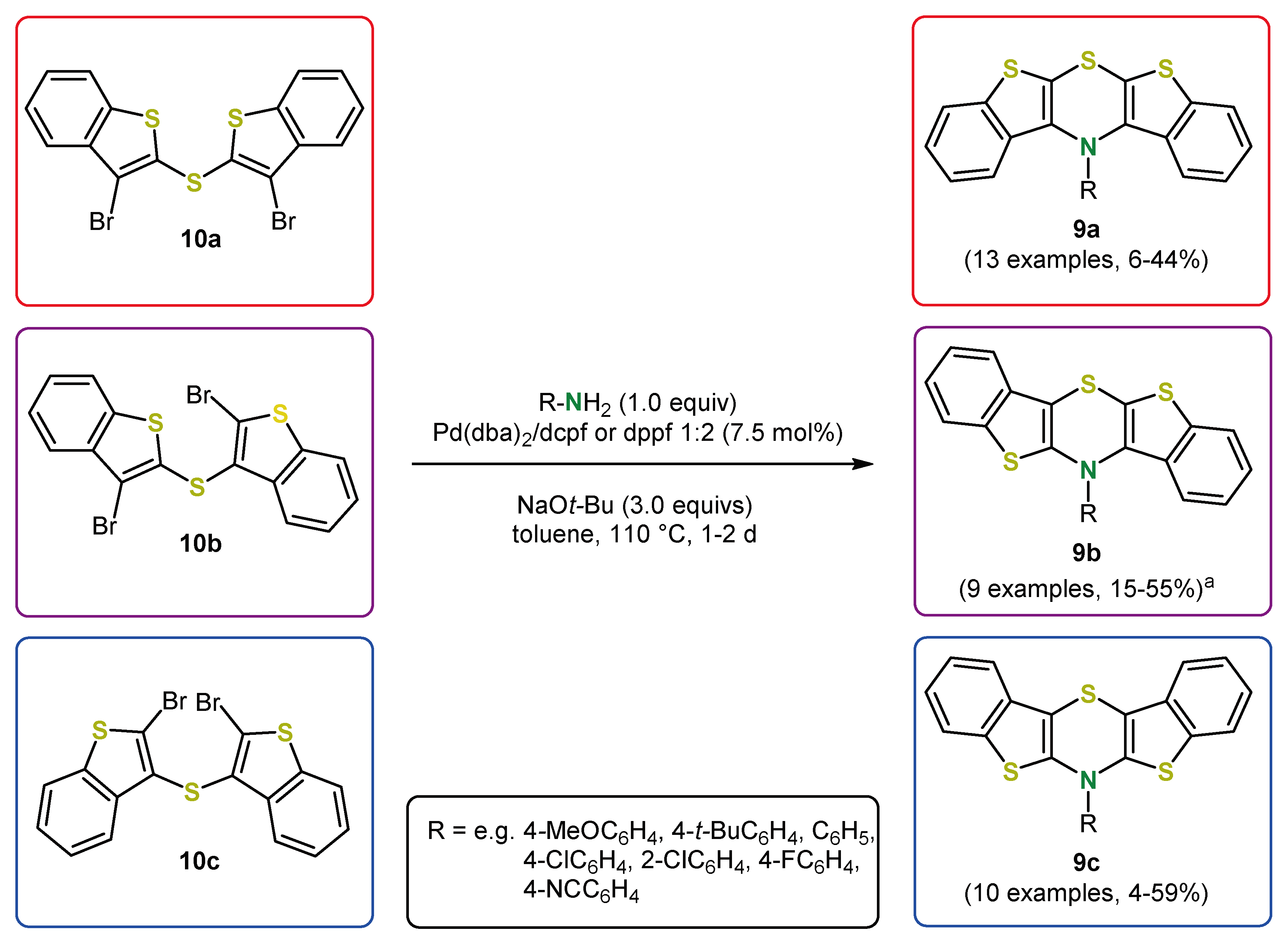
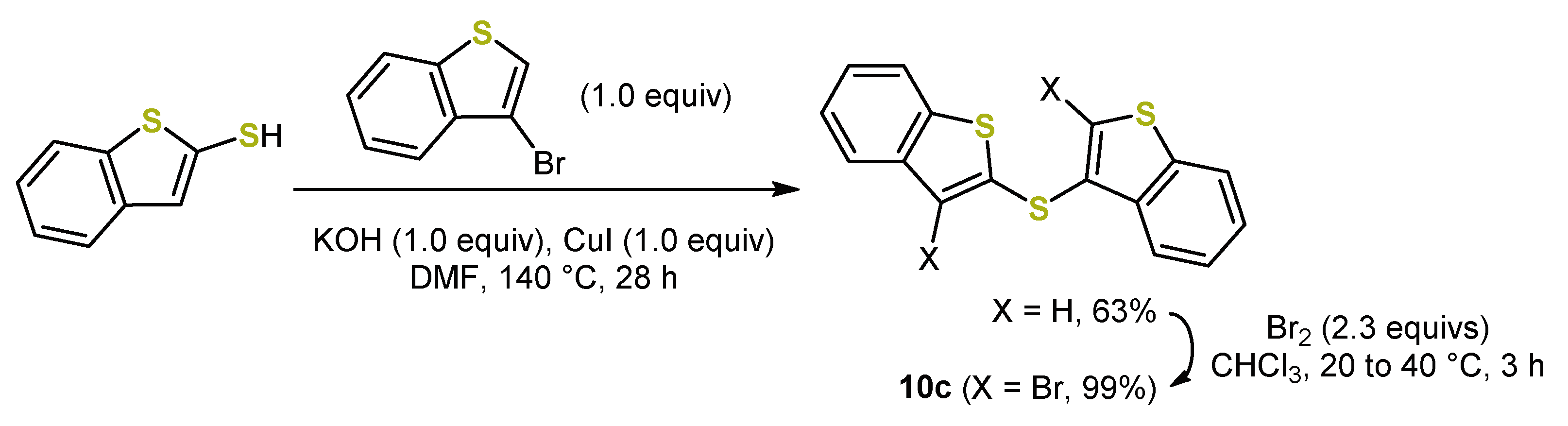
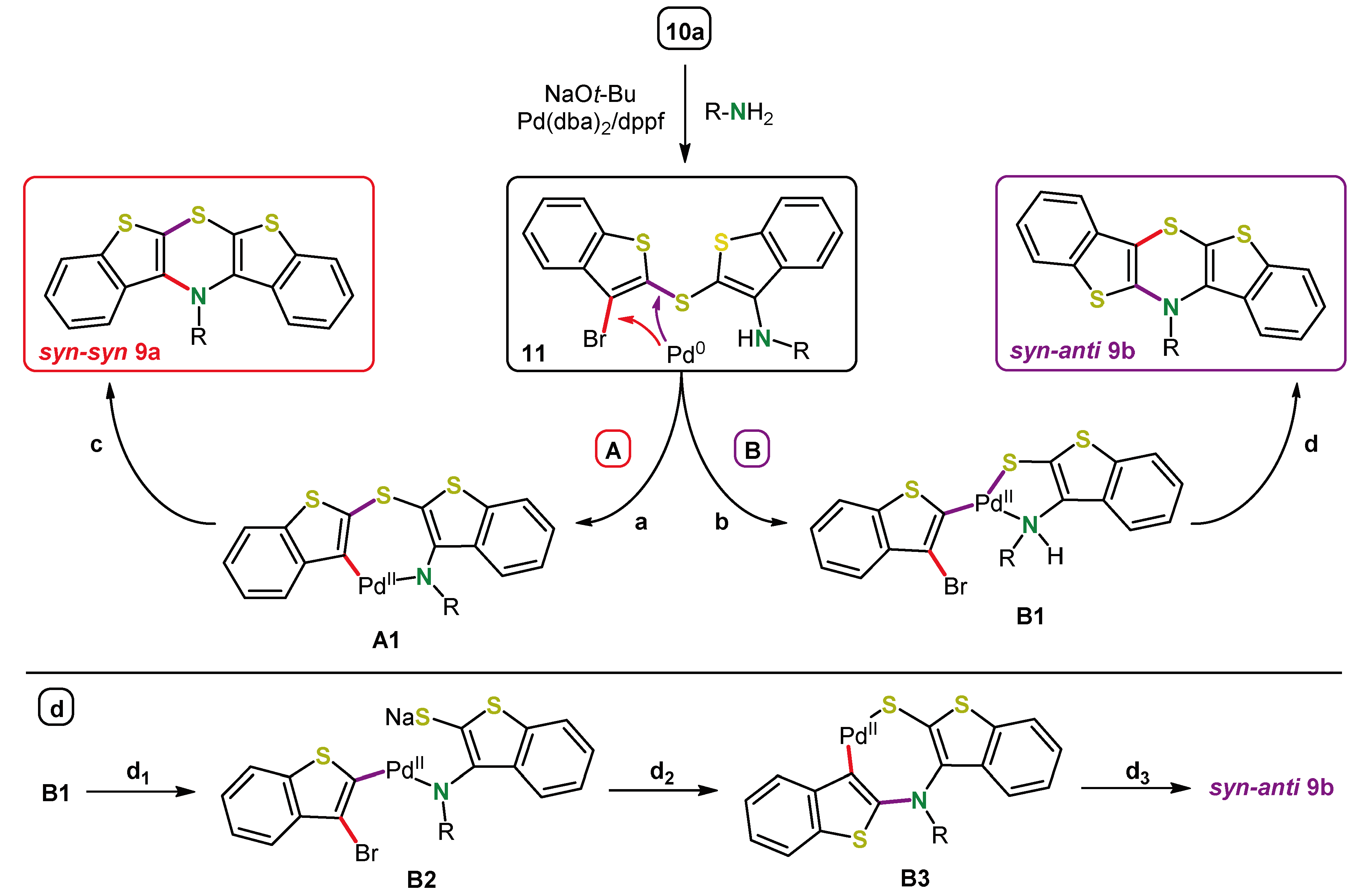
5. Expansion of the π-System by Anellation: Bis[1]benzothieno[1,4]thiazines
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Xiang, D.; Wang, X.; Jia, C.; Lee, T.; Guo, X. Molecular-Scale Electronics: From Concept to Function. Chem. Rev. 2016, 116, 4318–4440. [Google Scholar] [CrossRef] [PubMed]
- Thejo Kalyani, N.; Dhoble, S.J. Organic light emitting diodes: Energy saving lighting technology—A review. Renew. Sustain. Energy Rev. 2012, 16, 2696–2723. [Google Scholar] [CrossRef]
- Torsi, L.; Magliulo, M.; Manoli, K.; Palazzo, G. Organic field-effect transistor sensors: A tutorial review. Chem. Soc. Rev. 2013, 42, 8612–8628. [Google Scholar] [CrossRef] [PubMed]
- Wu, W.; Liu, Y.; Zhu, D. π-Conjugated molecules with fused rings for organic field-effect transistors: Design, synthesis and applications. Chem. Soc. Rev. 2010, 39, 1489–1502. [Google Scholar] [CrossRef] [PubMed]
- Cheng, P.; Li, G.; Zhan, X.; Yang, Y. Next-generation organic photovoltaics based on non-fullerene acceptors. Nat. Photonics 2018, 12, 131–142. [Google Scholar] [CrossRef]
- Hagfeldt, A.; Boschloo, G.; Sun, L.; Kloo, L.; Pettersson, H. Dye-Sensitized Solar Cells. Chem. Rev. 2010, 110, 6595–6663. [Google Scholar] [CrossRef]
- Levi, L.; Müller, T.J.J. Multicomponent syntheses of functional chromophores. Chem. Soc. Rev. 2016, 45, 2825–2846. [Google Scholar] [CrossRef]
- Fuse, S.; Sugiyama, S.; Maitani, M.; Wada, Y.; Ogomi, Y.; Hayase, S.; Katoh, R.; Kaiho, T.; Takahashi, T. Elucidating the Structure-Property Relationships of Donor-π-Acceptor Dyes for Dye-Sensitized Solar Cells (DSSCs) through Rapid Library Synthesis by a One-Pot Procedure. Chem. Eur. J. 2014, 20, 10685–10694. [Google Scholar] [CrossRef]
- Poriel, C.; Rault-Berthelot, J.; Thiery, S.; Quinton, C.; Jeannin, O.; Biapo, U.; Tondelier, D.; Geffroy, B. 9H-Quinolino[3,2,1-k]phenothiazine: A New Electron-Rich Fragment for Organic Electronics. Chem. Eur. J. 2016, 22, 17930–17935. [Google Scholar] [CrossRef]
- Hauck, M.; Stolte, M.; Schönhaber, J.; Kuball, H.-G.; Müller, T.J.J. Synthesis, Electronic, and Electro-Optical Properties of Emissive Solvatochromic Phenothiazinyl Merocyanine Dyes. Chem. Eur. J. 2011, 17, 9984–9998. [Google Scholar] [CrossRef]
- Ahn, Y.; Jang, D.E.; Cha, Y.B.; Kim, M.; Ahn, K.H.; Kim, Y.C. Electroluminescence characteristics of a new green-emitting phenylphenothiazine derivative with phenylbenzimidazole substituent. Bull. Korean Chem. Soc. 2013, 34, 107–111. [Google Scholar] [CrossRef]
- Onoabedje, E.A.; Egu, S.A.; Ezeokonkwo, M.A.; Okoro, U.C. Highlights of molecular structures and applications of phenothiazine & phenoxazine polycycles. J. Mol. Struct. 2019, 1175, 956–962. [Google Scholar] [CrossRef]
- Schaper, K.; Müller, T.J.J. Thiophene Syntheses by Ring Forming Multicomponent Reactions. Top. Curr. Chem. 2018, 376, 38. [Google Scholar] [CrossRef] [PubMed]
- Barbarella, G.; Melucci, M.; Sotgiu, G. The Versatile Thiophene: An Overview of Recent Research on Thiophene-Based Materials. Adv. Mater. 2005, 17, 1581–1593. [Google Scholar] [CrossRef]
- Varga, B.; Csonka, Á.; Csonka, A.; Molnar, J.; Amaral, L.; Spengler, G. Possible Biological and Clinical Applications of Phenothiazines. Anticancer Res. 2017, 37, 5983–5993. [Google Scholar]
- Aitken, R.A.; Aitken, K.M. 1,4-Thiazines and their Benzo Derivatives. In Comprehensive Heterocyclic Chemistry III; Katritzky, A.R., Ramsden, A.C., Scriven, E.F.V., Taylor, R.J.K., Eds.; Elsevier: Amsterdam, The Netherlands, 2008; Volume 8, p. 607. [Google Scholar]
- Michaelis, L. Semiquinones, the Intermediate Steps of Reversible Organic Oxidation-Reduction. Chem. Rev. 1935, 16, 243–286. [Google Scholar] [CrossRef]
- Pereţeanu, I.S.; Müller, T.J.J. Synthesis and electronic properties of 3,7-dianilino substituted N-hexyl phenothiazines. Org. Biomol. Chem. 2013, 11, 5127–5135. [Google Scholar] [CrossRef]
- Park, Y.; Kim, B.; Lee, C.; Hyun, A.; Jang, S.; Lee, J.-H.; Gal, Y.-S.; Kim, T.H.; Kim, K.-S.; Park, J. Highly Efficient New Hole Injection Materials for OLEDs Based on Dimeric Phenothiazine and Phenoxazine Derivatives. J. Phys. Chem. C 2011, 115, 4843–4850. [Google Scholar] [CrossRef]
- Salunke, J.K.; Wong, F.L.; Feron, K.; Manzhos, S.; Lo, M.F.; Shinde, D.; Patil, A.; Lee, C.S.; Roy, V.A.L.; Sonar, P.; et al. Phenothiazine and carbazole substituted pyrene based electroluminescent organic semiconductors for OLED devices. J. Mater. Chem. C 2016, 4, 1009–1018. [Google Scholar] [CrossRef]
- Qu, B.; Chen, Z.; Liu, Y.; Cao, H.; Xu, S.; Cao, S.; Lan, Z.; Wang, Z.; Gong, Q. Orange and red emitting OLEDs based on phenothiazine polymers. J. Phys. D Appl. Phys. 2006, 39, 2680–2683. [Google Scholar] [CrossRef]
- Urselmann, D.; Deilhof, K.; Mayer, B.; Müller, T.J.J. Thiophene-forming one-pot synthesis of three thienyl-bridged oligophenothiazines and their electronic properties. Beilstein J. Org. Chem. 2016, 12, 2055–2064. [Google Scholar] [CrossRef] [PubMed]
- Kim, G.; Yeom, H.R.; Cho, S.; Seo, J.H.; Kim, J.Y.; Yang, C. Easily Attainable Phenothiazine-Based Polymers for Polymer Solar Cells: Advantage of Insertion of S,S-dioxides into its Polymer for Inverted Structure Solar Cells. Macromolecules 2012, 45, 1847–1857. [Google Scholar] [CrossRef]
- Tian, H.; Yang, X.; Chen, R.; Pan, Y.; Li, L.; Hagfeldt, A.; Sun, L. Phenothiazine derivatives for efficient organic dye-sensitized solar cells. Chem. Commun. 2007, 3741–3743. [Google Scholar] [CrossRef] [PubMed]
- Weng, Y.-L.; Li, Y.-C.; Chen, C.-P.; Chang, Y.J. Effect of intermolecular interaction with phenothiazine core on inverted organic photovoltaics by using different acceptor moiety. Dyes Pigment. 2017, 146, 374–385. [Google Scholar] [CrossRef]
- Meyer, T.; Ogermann, D.; Pankrath, A.; Kleinermanns, K.; Müller, T.J.J. Phenothiazinyl Rhodanylidene Merocyanines for Dye-Sensitized Solar Cells. J. Org. Chem. 2012, 77, 3704–3715. [Google Scholar] [CrossRef]
- Meyer, T.; Müller, T.J.J. Consecutive Three-Component Synthesis of Donor-Substituted Merocyanines by a One-Pot Suzuki–Knoevenagel Condensation Sequence. Org. Mater. 2020, 2, 64–70. [Google Scholar] [CrossRef]
- Sailer, M.; Nonnenmacher, M.; Oeser, T.; Müller, T.J.J. Synthesis and Electronic Properties of 3-Acceptor-Substituted and 3,7-Bisacceptor-Substituted Phenothiazines. Eur. J. Org. Chem. 2006, 2006, 423–435. [Google Scholar] [CrossRef]
- Mayer, L.; May, L.; Müller, T.J.J. The interplay of conformations and electronic properties in N-aryl phenothiazines. Org. Chem. Front. 2020. [Google Scholar] [CrossRef]
- May, L.; Müller, T.J.J. Electron-Rich Phenothiazine Congeners and Beyond: Synthesis and Electronic Properties of Isomeric Dithieno[1,4]thiazines. Chem. Eur. J. 2020. [Google Scholar] [CrossRef]
- Grol, C.J.; Faber, J.S. Dithieno-1,4-thiazines. Part I. Recl. Trav. Chim. Pay-B 1970, 89, 68–73. [Google Scholar] [CrossRef]
- Grol, C.J. Dithieno- and thienobenzothiazines. J. Heterocycl. Chem. 1974, 11, 953–958. [Google Scholar] [CrossRef]
- Grol, C.J. Synthesis of a dithieno[2,3-b:3′,2′-e][1,4]thiazine and of di-3-thienylamine. J. Chem. Soc. Perkin Trans. 1975, 1, 1234. [Google Scholar] [CrossRef]
- Dahl, T.; Tornøe, C.W.; Bang-Andersen, B.; Nielsen, P.; Jørgensen, M. Palladium-Catalyzed Three-Component Approach to Promazine with Formation of One Carbon–Sulfur and Two Carbon–Nitrogen Bonds. Angew. Chem. Int. Ed. 2008, 47, 1726–1728. [Google Scholar] [CrossRef] [PubMed]
- Dostert, C.; Wanstrath, C.; Frank, W.; Müller, T.J.J. 4H-Dithieno[2,3-b:3′,2′-e][1,4]thiazines—Synthesis and electronic properties of a novel class of electron rich redox systems. Chem. Commun. 2012, 48, 7271–7273. [Google Scholar] [CrossRef] [PubMed]
- Dostert, C.; Czajkowski, D.; Müller, T.J.J. 2,6-Difunctionalization of N-Substituted Dithienothiazines via Dilithiation. Synlett 2014, 25, 371–374. [Google Scholar] [CrossRef]
- Nau, J.; Schneeweis, A.P.W.; Müller, T.J.J. Dithienothiazine dimers, trimers and polymers – novel electron-rich donors with red-shifted luminescence. Mater. Chem. Front. 2020, 4, 621–630. [Google Scholar] [CrossRef]
- Miyasaka, M.; Rajca, A. Synthesis of Dithieno[2,3-b:3′,2′-d]thiophenesBuilding Blocks for Cross-Conjugated β-Oligothiophenes. J. Org. Chem. 2006, 71, 3264–3266. [Google Scholar] [CrossRef]
- Allared, F.; Hellberg, J.; Remonen, T. A convenient and improved synthesis of dithieno[3,2-b:2′,3′-d]thiophene. Tetrahedron Lett. 2002, 43, 1553–1554. [Google Scholar] [CrossRef]
- Schneeweis, A.; Neidlinger, A.; Reiss, G.J.; Frank, W.; Heinze, K.; Müller, T.J.J. Radical cation and dication of a 4H-dithieno[2,3-b:3′,2′-e][1,4]-thiazine. Org. Chem. Front. 2017, 4, 839–846. [Google Scholar] [CrossRef]
- Janssen, M.J.; De Jong, F. Synthesis, oxidation, and electronic spectra of four dithienothiophenes. J. Org. Chem. 1971, 36, 1645–1648. [Google Scholar] [CrossRef]
- Chen, M.-C.; Chiang, Y.-J.; Kim, C.; Guo, Y.-J.; Chen, S.-Y.; Liang, Y.-J.; Huang, Y.-W.; Hu, T.-S.; Lee, G.-H.; Facchetti, A.; et al. One-pot [1+1+1] synthesis of dithieno[2,3-b:3′,2′-d]thiophene (DTT) and their functionalized derivatives for organic thin-film transistors. Chem. Commun. 2009, 1846–1848. [Google Scholar] [CrossRef] [PubMed]
- Bejan, A.; Shova, S.; Damaceanu, M.-D.; Simionescu, B.C.; Marin, L. Structure-Directed Functional Properties of Phenothiazine Brominated Dyes: Morphology and Photophysical and Electrochemical Properties. Cryst. Growth Des. 2016, 16, 3716–3730. [Google Scholar] [CrossRef]
- Dostert, C.; Müller, T.J.J. A one-pot dilithiation–lithium–zinc exchange–Negishi coupling approach to 2,6-di(hetero)aryl substituted dithienothiazines – a novel class of electronically fine-tunable redox systems. Org. Chem. Front. 2015, 2, 481–491. [Google Scholar] [CrossRef][Green Version]
- May, L.; Müller, T.J.J. Widely Electronically Tunable 2,6-Disubstituted Dithieno[1,4]thiazines—Electron-Rich Fluorophores up to Intense NIR Emission. Chem. Eur. J. 2020. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Wei, H.-x.; Schlosser, M. The Simultaneous In-Situ Generation of Aldehydes and Phosphorus Ylides: A Convenient Multi-Step One-Pot Olefination Protocol. Eur. J. Org. Chem. 1999, 1999, 3263–3268. [Google Scholar] [CrossRef]
- May, L.; Daniel, S.; Müller, T.J.J. Diversity-oriented approach to functional thiophene dyes by Suzuki coupling-lithiation one-pot sequences. Org. Chem. Front. 2020, 7, 329–339. [Google Scholar] [CrossRef]
- Sailer, M.; Franz, A.W.; Müller, T.J.J. Synthesis and Electronic Properties of Monodisperse Oligophenothiazines. Chem. Eur. J. 2008, 14, 2602–2614. [Google Scholar] [CrossRef]
- Schneeweis, A.P.W.; Hauer, S.T.; Lopez, D.A.; von Dressler, B.; Reiss, G.J.; Müller, T.J.J. Game of Isomers: Bifurcation in the Catalytic Formation of Bis[1]benzothieno[1,4]thiazines with Conformation-Dependent Electronic Properties. J. Org. Chem. 2019, 84, 5582–5595. [Google Scholar] [CrossRef]
- Schneeweis, A.P.W.; Hauer, S.T.; Reiss, G.J.; Müller, T.J.J. Bis[1]benzothieno[1,4]thiazines: Planarity, Enhanced Redox Activity and Luminescence by Thieno-Expansion of Phenothiazine. Chem. Eur. J. 2019, 25, 3582–3590. [Google Scholar] [CrossRef]
- Hong, G.; Antaris, A.L.; Dai, H. Near-infrared fluorophores for biomedical imaging. Nat. Biomed. Eng. 2017, 1, 0010. [Google Scholar] [CrossRef]

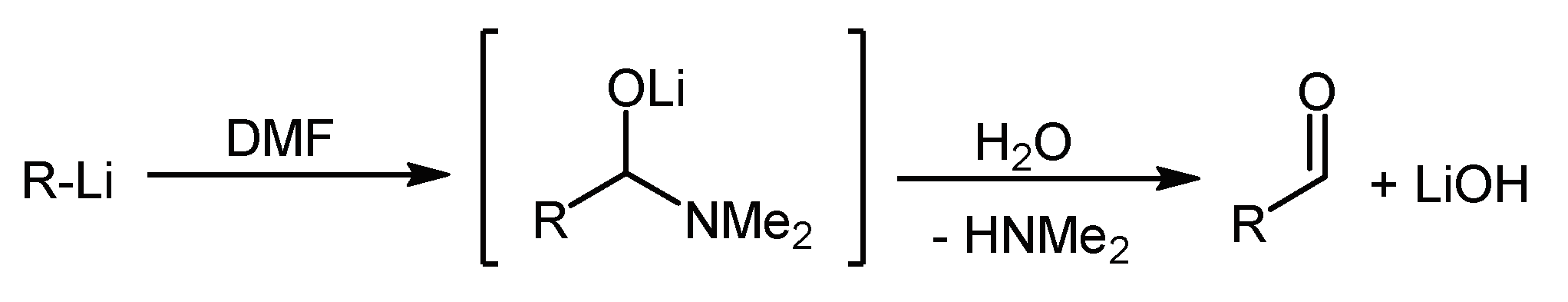
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
May, L.; Müller, T.J.J. Dithieno[1,4]thiazines and Bis[1]benzothieno[1,4]thiazines—Organometallic Synthesis and Functionalization of Electron Density Enriched Congeners of Phenothiazine. Molecules 2020, 25, 2180. https://doi.org/10.3390/molecules25092180
May L, Müller TJJ. Dithieno[1,4]thiazines and Bis[1]benzothieno[1,4]thiazines—Organometallic Synthesis and Functionalization of Electron Density Enriched Congeners of Phenothiazine. Molecules. 2020; 25(9):2180. https://doi.org/10.3390/molecules25092180
Chicago/Turabian StyleMay, Lars, and Thomas J. J. Müller. 2020. "Dithieno[1,4]thiazines and Bis[1]benzothieno[1,4]thiazines—Organometallic Synthesis and Functionalization of Electron Density Enriched Congeners of Phenothiazine" Molecules 25, no. 9: 2180. https://doi.org/10.3390/molecules25092180
APA StyleMay, L., & Müller, T. J. J. (2020). Dithieno[1,4]thiazines and Bis[1]benzothieno[1,4]thiazines—Organometallic Synthesis and Functionalization of Electron Density Enriched Congeners of Phenothiazine. Molecules, 25(9), 2180. https://doi.org/10.3390/molecules25092180






