A Review of Cytotoxic Plants of the Indian Subcontinent and a Broad-Spectrum Analysis of Their Bioactive Compounds
Abstract
1. Introduction
2. Methodology
2.1. Search Strategy
2.2. Selection of Studies for Inclusion in the Review
2.3. Data Extraction
3. Cytotoxic Plants of Indian Subcontinent
3.1. Acanthaceae
3.2. Actinidiaceae
3.3. Amaryllidaceae
3.4. Anacardiaceae
3.5. Annonaceae
3.6. Apocynaceae
3.7. Araceae
3.8. Aristolochiaceae
3.9. Asphodelaceae
3.10. Aspleniaceae
3.11. Asteraceae
3.12. Berberidaceae
3.13. Bignoniaceae
3.14. Boraginaceae
3.15. Bromeliaceae
3.16. Caesalpiniaceae
3.17. Compositae
3.18. Dilleniaceae
3.19. Dipsacaceae
3.20. Ebenaceae
3.21. Ericaceae
3.22. Euphorbiaceae
3.23. Fabaceae
3.24. Gramineae
3.25. Hypericaceae
3.26. Labiatae
3.27. Lamiaceae
3.28. Malvaceae
3.29. Meliaceae
3.30. Menispermaceae
3.31. Moraceae
3.32. Myristicaceae
3.33. Myrtaceae
3.34. Nyctaginaceae
3.35. Oleaceae
3.36. Oxalidaceae
3.37. Phyllanthacae
3.38. Plumbaginaceae
3.39. Picrorhiza
3.40. Pinaceae
3.41. Piperaceae
3.42. Poaceae
3.43. Polygonaceae
3.44. Primulaceae
3.45. Ranunculaceae
3.46. Rosaceae
3.47. Rubiaceae
3.48. Salicaceae
3.49. Sapotaceae
3.50. Saururaceae
3.51. Scrophulariaceae
3.52. Solanaceae
3.53. Sterculiaceae
3.54. Thymelaeaceae
3.55. Verbenaceae
3.56. Vitaceae
3.57. Zingiberaceae
4. Reported Cytotoxic Constituents: Therapeutic Perspective and Future Directions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| A549 | Human Lung Adenocarcinoma |
| AGS | Gastric Adenocarcinoma |
| AMPK | AMP Activated Protein Kinase |
| CACO | Human Colon Carcinoma |
| CCK | Cell Counting Kit |
| CDK | Cyclin Dependent Kinase |
| DAPI | 4′,6-Diamidino-2-Phenylindole |
| DCFH-DA | Dichlorodihydrofluorescein diacetate |
| DLA/DAL | Dalton’s Lymphoma Ascites/Dalton’s Ascites Lymphoma |
| DTP | Developmental Therapeutic Program |
| GSH | Glutathione |
| H1299 | Human Non-Small Cell Lung Cancer |
| HCT/HT | Human Colorectal Adenocarcinoma |
| HeLa | Henrietta Lacks Cervical Cancer |
| Hep | Human Epithelial |
| HL | Human Caucasian Promyelocytic Leukemia |
| IC50/EC50 | Concentration Required to Inhibit/Destroy 50% Cells |
| NF-kB | Nuclear Factor-Kappa B |
| MCF | Human Breast Adenocarcinoma |
| MCL | Myeloid Cell Leukemia |
| MDA-MB | M. D. Anderson Metastasis Breast Cancer |
| MMP | Matrix Metallopeptidase |
| MST | Mean Survival Time |
| MTT | 3-[4,5-Dimethylthiazol-2-yl]-2,5-diphenyltetrazolium bromide |
| OSA | Osteosarcoma |
| PARP | PolyADP-ribosepolymerase |
| PC | Prostate Cancer |
| PKC | Protein Kinase C |
| SRB | Sulforhodamine B |
| STAT | Signal Transducer and Activator of Transcription |
| T47D | Ductal Epithelial Breast Tumor |
| TBE | Trypan Blue Exclusion |
| THP | Human Acute Monocytic Leukemia |
| VEGF | Vascular Endothelial Growth Factor |
| Vero | Verda Reno |
| WM | Malignant Melanoma |
| WST | Water Soluble Tetrazolium |
| XTT | Cell Proliferation Kit II |
References
- Iqbal, J.; Abbasi, B.H.; Mahmood, T.; Kanwal, S.; Ali, B.; Shah, S.A.; Khalil, A.T. Plant-derived anticancer agents: A green anticancer approach. Asian Pac. J. Trop. Biomed. 2017, 7, 1129–1150. [Google Scholar] [CrossRef]
- Krishnamurthi, K. 17-screening of natural products for anticancer and antidiabetic properties. Cancer 2007, 3, 69–75. [Google Scholar]
- Thirumal, M.; Kishore, G.; Prithika, R.; Das, S.; Nithya, G. In vitro anticancer activity of Tecoma stans (L) ethanolic leaf extract on human breast cancer cell line (MCF-7). Intl. J. Pharma Bio Sci. 2012, 2, 488–493. [Google Scholar]
- Zyad, A.; Leouifoudi, I.; Tilaoui, M.; Mouse, H.A.; Khouchani, M.; Jaafari, A. Natural products as cytotoxic agents in chemotherapy against cancer. Cytotoxicity 2018, 65–88. [Google Scholar] [CrossRef]
- Merina, N.; Chandra, K.J.; Jibon, K. Medicinal plants with potential anticancer activities: A review. Int. Res. J. Pharm. 2012, 3, 26–30. [Google Scholar]
- Tilaoui, M.; Mouse, H.A.; Jaafari, A.; Zyad, A. Comparative phytochemical analysis of essential oils from different biological parts of Artemisia herba alba and their cytotoxic effect on cancer cells. PLoS ONE 2015, 10, e0131799. [Google Scholar] [CrossRef]
- Shukla, Y.; Pal, S.K. Dietary cancer chemoprevention: An overview. Int. J. Hum. Genet. 2004, 4, 265–276. [Google Scholar] [CrossRef]
- Latosińska, J.N.; Latosińska, M. Anticancer drug discovery—From serendipity to rational design. Drug Discov. 2013, 35–74. [Google Scholar] [CrossRef]
- Cragg, G.M.; Newman, D.J. Plants as a source of anti-cancer agents. J. Ethnopharmacol. 2005, 100, 72–79. [Google Scholar] [CrossRef]
- Rengasamy, G.; Venkataraman, A.; Veeraraghavan, V.P.; Jainu, M. Cytotoxic and apoptotic potential of Myristica fragrans Houtt. (mace) extract on human oral epidermal carcinoma KB cell lines. Braz. J. Pharm. Sci. 2018, 54, 54. [Google Scholar] [CrossRef]
- Verdine, G.L. The combinatorial chemistry of nature. Nature 1996, 384, 11–13. [Google Scholar]
- Demain, A.L.; Vaishnav, P. Natural products for cancer chemotherapy. Microb. Biotechnol. 2011, 4, 687–699. [Google Scholar] [CrossRef]
- Kerr, J.F.R.; Wyllie, A.H.; Currie, A.R. Apoptosis: A basic biological phenomenon with wideranging implications in tissue kinetics. Br. J. Cancer 1972, 26, 239–257. [Google Scholar] [CrossRef] [PubMed]
- Hücre, B.T.B.F.K.; Aktiviteleri, H.S. Cytotoxic activities of certain medicinal plants on different cancer cell lines. Turk. J. Pharm. Sci. 2017, 14, 222–230. [Google Scholar]
- Garg, A.; Darokar, M.P.; Sundaresan, V.; Faridi, U.; Luqman, S.; Khanuja, S. Anticancer activity of some medicinal plants from high altitude evergreen elements of Indian Western Ghats. J. Res. Educ. Indian Med. 2007, 13, 1–6. [Google Scholar]
- Umadevi, M.; Kumar, K.S.; Bhowmik, D.; Duraivel, S. Traditionally used anticancer herbs in India. J. Med. Plants Stud. 2013, 1, 56–74. [Google Scholar]
- Manglani, N.; Vaishnava, S.; Dhamodaran, P.; Sawarkar, H. In vitro and in vivo anticancer activity of leaf extract of Barleria grandiflora. Int. J. Pharm. Pharm. Res. 2014, 6, 70–72. [Google Scholar]
- Kumar, A.S.G.; Javvadi, R.K.; Kumar, V.K.; Reddy, M.E.; Reddy, V.Y.; Harshavardhan, G.; Akbar, M. Effect of methanolic extract of Adenanthera pavonina Linn on Dalton’s ascitic lymphoma. Indian J. Res. Pharm. Biotech. 2013, 1, 138. [Google Scholar]
- Trease, G.; Evans, W. Text book of Pharmacognosy; Bailliare Tindall: London, UK, 1983; pp. 193–336. [Google Scholar]
- Mazumder, K.; Siwu, E.R.; Nozaki, S.; Watanabe, Y.; Tanaka, K.; Fukase, K. Ursolic acid derivatives from Bangladeshi medicinal plant, Saurauja roxburghii: Isolation and cytotoxic activity against A431 and C6 glioma cell lines. Phytochem. Lett. 2011, 4, 287–291. [Google Scholar] [CrossRef]
- Mazumder, K.; Tanaka, K.; Fukase, K. Cytotoxic activity of ursolic acid derivatives obtained by isolation and oxidative derivatization. Molecules 2013, 18, 8929–8944. [Google Scholar] [CrossRef]
- Talib, W.H.; Mahasneh, A.M. Antiproliferative activity of plant extracts used against cancer in traditional medicine. Sci. Pharm. 2010, 78, 33–45. [Google Scholar] [CrossRef] [PubMed]
- Artun, F.T.; Karagoz, A.; Ozcan, G.; Melikoglu, G.; Anil, S.; Kultur, S.; Sutlupinar, N. In vitro anticancer and cytotoxic activities of some plant extracts on HeLa and Vero cell lines. JBUON: Off. J. Balk. Union Oncol. 2016, 21, 720–725. [Google Scholar] [CrossRef]
- Akter, R.; Uddin, S.N.; Grice, I.D.; Tiralongo, E. Cytotoxic activity screening of Bangladeshi medicinal plant extracts. J. Nat. Med. 2013, 68, 246–252. [Google Scholar] [CrossRef] [PubMed]
- Weerapreeyakul, N.; Junhom, C.; Barusrux, S.; Thitimetharoch, T. Induction of apoptosis in human hepatocellular carcinoma cells by extracts of Lannea coromandelica (Houtt.) Merr. and Diospyros castanea (Craib) Fletcher. Chin. Med. 2016, 11, 19. [Google Scholar] [CrossRef] [PubMed]
- Yun, X.-J.; Shu, H.-M.; Chen, G.-Y.; Ji, M.-H.; Ding, J.-Y. Chemical constituents from barks of Lannea coromandelica. Chin. Herb. Med. 2014, 6, 65–69. [Google Scholar] [CrossRef]
- Gavamukulya, Y.; Abou-Elella, F.; Wamunyokoli, F.; El-Shemy, H.A. Phytochemical screening, anti-oxidant activity and in vitro anticancer potential of ethanolic and water leaves extracts of Annona muricata (Graviola). Asian Pac. J. Trop. Med. 2014, 7, S355–S363. [Google Scholar] [CrossRef]
- Sumithra, P.; Gricilda Shoba, F.; Vimala, G.; Sathya, J.; Sankar, V.; Saraswathi, R. Anticancer activity of Annona squamosa and Manilkara zapota flower extract against MCF-7 cell line. DerPharm. Sin. 2014, 5, 98–100. [Google Scholar]
- Dantu, A.S.; Shankarguru, P.; Ramya, D.D.; Vedha, H.B. Evaluation of in vitro anticancer activity of hydroalcoholic extract of Tabernaemontana divaricata. Asian J. Pharm. Clin. Res. 2012, 5, 59–61. [Google Scholar]
- Bao, M.-F.; Yan, J.-M.; Cheng, G.-G.; Li, X.-Y.; Liu, Y.-P.; Li, Y.; Cai, X.-H.; Luo, X.-D. Cytotoxic indole alkaloids from Tabernaemontana divaricata. J. Nat. Prod. 2013, 76, 1406–1412. [Google Scholar] [CrossRef]
- Abdel-Salam, I.M.; Awadein, N.E.-S.; Ashour, M. Cytotoxicity of Luffa cylindrica (L.) M. Roem. extract against circulating cancer stem cells in hepatocellular carcinoma. J. Ethnopharmacol. 2019, 229, 89–96. [Google Scholar] [CrossRef]
- Abdel-Salam, I.M.; Ashmawy, A.M.; Hilal, A.M.; Eldahshan, O.A.; Ashour, M. Chemical composition of aqueous ethanol extract of Luffa cylindrica leaves and its effect on representation of caspase-8, caspase-3, and the proliferation marker Ki67 in intrinsic molecular subtypes of breast cancer in vitro. Chem. Biodivers. 2018, 15, e1800045. [Google Scholar] [CrossRef] [PubMed]
- Akindele, A.; Wani, Z.; Mahajan, G.; Sharma, S.; Aigbe, F.R.; Satti, N.; Adeyemi, O.O.; Mondhe, D.M. Anticancer activity of Aristolochia ringens Vahl. (Aristolochiaceae). J. Tradit. Complement. Med. 2014, 5, 35–41. [Google Scholar] [CrossRef] [PubMed]
- Daoudi, A.; El Yoube, A.; Bagrel, D.; Aarab, L. In vitro anticancer activity of some plants used in Moroccan traditional medicine. J. Med. Plants Res. 2013, 7, 1182–1189. [Google Scholar]
- Shalabi, M.; Khilo, K.; Zakaria, M.M.; Elsebaei, M.G.; Abdo, W.; Awadin, W. Anticancer activity of Aloe vera and Calligonum comosum extracts separetely on hepatocellular carcinoma cells. Asian Pac. J. Trop. Biomed. 2015, 5, 375–381. [Google Scholar] [CrossRef]
- Jarial, R.; Thakur, S.; Sakinah, M.; Zularisam, A.; Sharad, A.; Kanwar, S.; Singh, L. Potent anticancer, antioxidant and antibacterial activities of isolated flavonoids from Asplenium nidus. J. King Saud Univ.-Sci. 2018, 30, 185–192. [Google Scholar] [CrossRef]
- Jirangkul, P.; Srisawat, P.; Punyaratabandhu, T.; Songpattanaslip, T.; Mungthin, M. Cytotoxic effect of artemisinin and its derivatives on human osteosarcoma cell lines. J. Med. Assoc. Thail. = Chotmaihet thangphaet 2014, 97, 215–221. [Google Scholar]
- Hosoya, K.; Murahari, S.; Laio, A.; London, C.A.; Couto, C.G.; Kisseberth, W.C. Biological activity of dihydroartemisinin in canine osteosarcoma cell lines. Am. J. Veter- Res. 2008, 69, 519–526. [Google Scholar] [CrossRef]
- Efferth, T.; Herrmann, F.; Tahrani, A.; Wink, M. Cytotoxic activity of secondary metabolites derived from Artemisia annua L. towards cancer cells in comparison to its designated active constituent artemisinin. Phytomedicine 2011, 18, 959–969. [Google Scholar] [CrossRef]
- Isani, G.; Bertocchi, M.; Andreani, G.; Farruggia, G.; Cappadone, C.; Salaroli, R.; Forni, M.; Bernardini, C. Cytotoxic effects of Artemisia annua L. and pure artemisinin on the D-17 canine osteosarcoma cell line. Oxidative Med. Cell. Longev. 2019, 2019, 1615758–1615759. [Google Scholar] [CrossRef]
- Sundararajan, P.; Dey, A.; Smith, A.; Doss, A.G.; Rajappan, M.; Natarajan, S. Studies of anticancer and antipyretic activity of Bidens pilosa whole plant. Afr. Health Sci. 2006, 6, 27–30. [Google Scholar]
- Saranya, K.; Manivasagan, V.; Kanakadurga, R.; Mohan Babu, V.P.; Ramesh Babu, N.G. A survey on anticancer properties of Indian medicinal plants – A broad spectrum analysis. Int. J. Pharm. Sci. Res. 2019, 10, 3635–3640. [Google Scholar]
- Abu-Rish, E.Y.; Kasabri, V.; Hudaib, M.; Mashalla, S.H.; Alalawi, L.H.; Tawaha, K.; Mohammad, M.K.; Mohamed, Y.S.; Bustanji, Y. Evaluation of antiproliferative activity of some traditional anticancer herbal remedies from Jordan. Trop. J. Pharm. Res. 2016, 15, 469. [Google Scholar] [CrossRef]
- Choi, J.-M.; Lee, E.-O.; Lee, H.-J.; Kim, K.-H.; Ahn, K.-S.; Shim, B.-S.; Kim, N.-I.; Song, M.-C.; Baek, N.-I.; Kim, S.-H. Identification of campesterol from Chrysanthemum coronarium L. and its antiangiogenic activities. Phytother. Res. 2007, 21, 954–959. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Chashoo, G.; Saxena, A.K.; Pandey, A.K. Parthenium hysterophorus: A probable source of anticancer, antioxidant and anti-HIV agents. BioMed Res. Int. 2013, 2013, 1–11. [Google Scholar] [CrossRef]
- Rosangkima, G.; Jagetia, G. In vitro anticancer screening of medicinal plants of Mizoram State, India, against Dalton’s lymphoma, MCF-7 and HELA cells. Int. J. Recent Sci. Res. 2015, 6, 5648–5653. [Google Scholar]
- Serasanambati, M.; Chilakapati, S.R.; Manikonda, P.K.; Kanala, J.R. Anticancer activity of methanolic extract of Berberis aristata in MCF-7 human breast cancer cell lines. Int. J. Life Sci. Biotech. Pharma Res. 2015, 4, 31–35. [Google Scholar]
- Gaidhani, S.; Singh, A.; Kumari, S.; Lavekar, G.; Juvekar, A.; Sen, S.; Padhi, M. Evaluation of some plant extracts for standardization and anticancer activity. Indian. J. Tradit. Know. 2013, 12, 682–687. [Google Scholar]
- Jayachandran, P.R.; Suriya, K.; Subbaiya, R.; Ponmurugan, P. Antioxidant and cytotoxic activity of Tecoma stans against lung cancer cell line (A549). Braz. J. Pharm. Sci. 2017, 53, 53. [Google Scholar] [CrossRef]
- Zhu, J.; Viñas, R.; Smith, E.E. In vitro evaluation of human liver cancer cells and the potential cytotoxicity of Tecoma stans (Bignoniaceae) and Brickellia cavanillesi (Asteraceae) both single and in combination. Toxicol. Environ. Chem. 2008, 90, 801–808. [Google Scholar] [CrossRef]
- Marzouk, M.S.; Gamal-Eldeen, A.; Mohamed, M.; El-Sayed, M. Anti-proliferative and antioxidant constituents from Tecoma stans. Zeitschrift für Naturforschung C 2007, 61, 783–791. [Google Scholar]
- Rahman, A.; Sahabjada; Akhtar, J. Evaluation of anticancer activity of Cordia dichotoma leaves against a human prostate carcinoma cell line, PC3. J. Tradit. Complement. Med. 2016, 7, 315–321. [Google Scholar] [CrossRef] [PubMed]
- Lowe, H.I.; Toyang, N.J.; Watson, C.; Badal, S.; Bahado-Singh, P.; Bryant, J. In vitro anticancer activity of the crude extract and two dicinnamate isolates from the Jamaican ball moss (Tillandsia Recurvata L.). Am. Int. J. Contemp. Res. 2013, 3, 93–96. [Google Scholar] [PubMed]
- Bukke, A.N.; Hadi, F.N.; Babu, K.S.; Shankar, P.C. In vitro studies data on anticancer activity of Caesalpinia sappan L. heartwood and leaf extracts on MCF7 and A549 cell lines. Data Brief 2018, 19, 868–877. [Google Scholar] [CrossRef] [PubMed]
- Al-Snafi, A.E. The medical benefit of Gnaphalium luteoalbum —A review. IOSR J. Pharm. 2019, 9, 40–44. [Google Scholar]
- Prasad, S.; Rosangkima, G.; Rongpi, T. Role of glutathione and glutathione-related enzymes in the antitumor activity of Dillenia pentagyna in Dalton’s lymphoma-bearing mice. Int. J. Cancer Res. 2008, 4, 92–102. [Google Scholar] [CrossRef]
- Alex, A.T.; Nawagamuwa, N.H.; Joseph, A.; Rao, J.V.; Mathew, J.A.; Udupa, N. In vitro anticancer and antioxidant activity of different fractions of Diospyros peregrina unripe fruit extract. Free. Radicals Antioxidants 2012, 2, 45–49. [Google Scholar] [CrossRef]
- Munro, B.R.; Vuong, Q.; Chalmers, A.C.; Goldsmith, C.; Bowyer, M.C.; Scarlett, C.J. Phytochemical, antioxidant and anti-cancer properties of Euphorbia tirucalli methanolic and aqueous extracts. Antioxidants 2015, 4, 647–661. [Google Scholar] [CrossRef]
- Balasubramanian, K.; Padma, P.R. Anticancer activity of Zea mays leaf extracts on oxidative stress-induced Hep2 Cells. J. Acupunct. Meridian Stud. 2013, 6, 149–158. [Google Scholar] [CrossRef][Green Version]
- Kim, S.L.; Snook, M.E.; Lee, J.O. Radical scavenging activity and cytotoxicity of maysin (C-glycosylflavone) isolated from silks of Zea mays L. Korean J. Crop. Sci. 2003, 48, 392–396. [Google Scholar]
- Muniyandi, K.; George, E.; Mudili, V.; Kalagatur, N.K.; Anthuvan, A.J.; Krishna, K.; Thangaraj, P.; Natarajan, G. Antioxidant and anticancer activities of Plectranthus stocksii Hook. f. leaf and stem extracts. Agric. Nat. Resour. 2017, 51, 63–73. [Google Scholar] [CrossRef]
- Jawad, A.; Balayeshwanth, R.V.; Rami, A.; Waleed, R.; Hatem, S.; Nathan, W.L.; Alzeer, J.; Vummidi, B.R.; Arafeh, R.; Rimawi, W.; et al. The influence of extraction solvents on the anticancer activities of Palestinian medicinal plants. J. Med. Plants Res. 2014, 8, 408–415. [Google Scholar] [CrossRef]
- Karthikeyan, K.; Ravichandran, P.; Govindasamy, S. Chemopreventive effect of Ocimum sanctum on DMBA-induced hamster buccal pouch carcinogenesis. Oral Oncol. 1999, 35, 112–119. [Google Scholar] [CrossRef]
- Godhwani, S.; Godhwani, J.; Was, D. Ocimum sanctum — A preliminary study evaluating its immunoregulatory profile in albino rats. J. Ethnopharmacol. 1988, 24, 193–198. [Google Scholar] [CrossRef]
- Mediratta, P.K.; Dewan, V.; Bhattacharya, S.K.; Gupta, V.S.; Maiti, P.C.; Sen, P. Effect of Ocimum sanctum Linn. on humoral immune responses. Indian J. Med. Res. 1988, 87, 384–386. [Google Scholar]
- Mandal, S.; Chatterjee, A. Seminar on research in Ayurveda and Siddha; CCRAS: New Delhi, India, 1994; pp. 58–59. [Google Scholar]
- Alam, P.; Al-Yousef, H.M.; Siddiqui, N.A.; Alhowiriny, T.A.; Alqasoumi, S.I.; Amina, M.; Hassan, W.H.B.; Abdelaziz, S.; Abdalla, R.H. Anticancer activity and concurrent analysis of ursolic acid, β-sitosterol and lupeol in three different Hibiscus species (aerial parts) by validated HPTLC method. Saudi Pharm. J. 2018, 26, 1060–1067. [Google Scholar] [CrossRef]
- Kim, D.K.; Baek, J.H.; Kang, C.M.; Yoo, M.A.; Sung, J.W.; Kim, D.K.; Chung, H.Y.; Kim, N.D.; Choi, Y.H.; Lee, S.H. Apoptotic activity of ursolic acid may correlate with the inhibition of initiation of DNA replication. Int. J. Cancer 2000, 87, 629–636. [Google Scholar] [CrossRef]
- Choi, Y.; Kong, K.; Kim, Y.-A.; Jung, K.-O.; Kil, J.-H.; Rhee, S.-H.; Park, K.-Y. Induction of Bax and activation of caspases during β-sitosterol-mediated apoptosis in human colon cancer cells. Int. J. Oncol. 2003, 23, 1657–1662. [Google Scholar] [CrossRef]
- Gallo, M.B.; Sarachine, M.J. Biological activities of lupeol. Int. J. Biomed. Pharm. Sci. 2009, 3, 46–66. [Google Scholar]
- Amer, H.; Helmy, W.A.; Taie, H.A. In vitro antitumor and antiviral activities of seeds and leaves Neem (Azadirachta indica) extracts. Int. J. Acad. Res. 2010, 2, 47–51. [Google Scholar]
- Jafari, S.; Saeidnia, S.; Hajimehdipoor, H.; Reza, S.A.M.; Faramarzi, M.A.; Hadjiakhoondi, A.; Khanavi, M. Cytotoxic evaluation of Melia azedarach in comparison with, Azadirachta indica and its phytochemical investigation. DARU J. Pharm. Sci. 2013, 21, 37. [Google Scholar] [CrossRef]
- Takagi, M.; Tachi, Y.; Zhang, J.; Shinozaki, T.; Ishii, K.; Kikuchi, T.; Ukiya, M.; Banno, N.; Tokuda, H.; Akihisa, T. Cytotoxic and melanogenesis-inhibitory activities of limonoids from the leaves of Azadirachta indica (Neem). Chem. Biodivers. 2014, 11, 451–468. [Google Scholar] [CrossRef] [PubMed]
- Akihisa, T. ChemInform abstract: Limonoids from Azadirachta indica var. siamensis extracts and their cytotoxic and melanogenesis-inhibitory activities. ChemInform 2014, 45, 505–531. [Google Scholar] [CrossRef]
- Kikuchi, T.; Ishii, K.; Noto, T.; Takahashi, A.; Tabata, K.; Suzuki, T.; Akihisa, T. Cytotoxic and apoptosis-inducing activities of limonoids from the seeds of Azadirachta indica (Neem). J. Nat. Prod. 2011, 74, 866–870. [Google Scholar] [CrossRef] [PubMed]
- Kitdamrongtham, W.; Ishii, K.; Ebina, K.; Zhang, J.; Ukiya, M.; Koike, K.; Akazawa, H.; Manosroi, A.; Manosroi, J.; Akihisa, T. ChemInform abstract: Limonoids and flavonoids from the flowers of Azadirachta indica var. siamensis, and their melanogenesis-inhibitory and cytotoxic activities. ChemInform 2014, 45, 73–84. [Google Scholar] [CrossRef]
- Chen, J.; Chen, J.; Sun, Y.; Yan, Y.; Kong, L.-M.; Li, Y.; Qiu, M. Cytotoxic triterpenoids from Azadirachta indica. Planta Medica 2011, 77, 1844–1847. [Google Scholar] [CrossRef]
- Thakkar, K.; Prasad, A.; Nayak, J.; Iyer, S.; Kumar, S. Antioxidant and in vitro cytotoxic activity of extracts of aerial parts of Cocculus hirsutus (L) using cell line cultures (breast cell line). J. Phytopharmacol. 2014, 3, 395–399. [Google Scholar]
- Thavamani, B.S.; Mathew, M.; Dhanabal, S.P. Cocculus hirsutus: Molecular docking to identify suitable targets for hepatocellular carcinoma by in silico technique. Pharmacogn. Mag. 2016, 12, S350–S352. [Google Scholar] [CrossRef][Green Version]
- Patel, R.M.; Patel, S.K. Cytotoxic activity of methanolic extract of Artocarpus heterophyllus against A549, Hela and MCF-7 cell lines. J. Appl. Pharm. Sci. 2011, 1, 167–171. [Google Scholar]
- Arung, E.T.; Yoshikawa, K.; Nakagawa, T.; Kondo, R. Isoprenoid-substituted flavonoids from wood of Artocarpus heterophyllus on B16 melanoma cells: Cytotoxicity and structural criteria. Fitoterapia 2010, 81, 120–123. [Google Scholar] [CrossRef]
- Arung, E.T.; Wicaksono, B.D.; Handoko, Y.A.; Kusuma, I.W.; Nakagawa, T.; Yulia, D.; Sandra, F. Cytotoxic effect of artocarpin on T47D cells. J. Nat. Med. 2010, 64, 423–429. [Google Scholar] [CrossRef]
- Arung, E.T.; Nakagawa, T.; Kondo, R. Structure–activity relationship of prenyl-substituted polyphenols from Artocarpus heterophyllus as Inhibitors of melanin biosynthesis in cultured melanoma cells. Chem. Biodivers. 2007, 4, 2166–2171. [Google Scholar] [CrossRef] [PubMed]
- Yen, G.; Chen, C.-S.; Chang, W.-T.; Wu, M.-F.; Cheng, F.-T.; Shiau, D.-K.; Hsu, C.-L. Antioxidant activity and anticancer effect of ethanolic and aqueous extracts of the roots of Ficus beecheyana and their phenolic components. J. Food Drug Anal. 2018, 26, 182–192. [Google Scholar] [CrossRef]
- Khan, A.; Anand, V.; Badrinarayanan, V.; Thirunethiran, K.; Natarajan, P. In vitro antioxidant and cytotoxicity analysis of leaves of Ficus racemosa. Free. Radic. Antioxid. 2016, 7, 8–12. [Google Scholar] [CrossRef]
- Sukhramani, P.S.; Vidyasagar, G.; Patel, P.M. In vitro screening of Ficus racemosa for anticancer activity. Res. J. Pharmacog. Phytochem. 2013, 5, 119–122. [Google Scholar]
- Gavhane, D.S.; Moregaonkar, S.D.; Mhase, A.K. Cytotoxic and anticancer activity of Ficus racemosa fruit extract on MCF7 human breast cancer cell line by SRB method. J. Anim. Res. 2016, 6, 43–47. [Google Scholar] [CrossRef]
- Qadir, M.I.; Ali, M.; Ibrahim, Z. Anticancer activity of Morus nigra leaves extract. Bangladesh J. Pharmacol. 2014, 9, 496–497. [Google Scholar] [CrossRef]
- Souza, G.R.; Oliveira-Júnior, R.G.; Diniz, T.C.; Branco, A.; Lima-Saraiva, S.R.G.; Guimarães, A.L.; Oliveira, A.P.; Pacheco, A.G.M.; Silva, M.G.; Moraes-Filho, M.O.; et al. Assessment of the antibacterial, cytotoxic and antioxidant activities of Morus nigra L. (Moraceae). Braz. J. Boil. 2017, 78, 248–254. [Google Scholar] [CrossRef]
- Piaru, S.P.; Mahmud, R.; Majid, A.M.S.A.; Ismail, S.; Nin Man, C. Chemical composition, antioxidant and cytotoxicity activities of the essential oils of Myristica fragrans and Morinda citrifolia. J. Sci. Food Agric. 2012, 92, 593–597. [Google Scholar] [CrossRef]
- Duan, L.; Tao, H.-W.; Hao, X.-J.; Gu, Q.-Q.; Zhu, W. Cytotoxic and antioxidative phenolic compounds from the traditional chinese medicinal plant, Myristica fragrans. Planta Medica 2009, 75, 1241–1245. [Google Scholar] [CrossRef]
- Yadav, S.S.; Meshram, G.; Shinde, D.; Patil, R.; Manohar, S.M.; Upadhye, M.V. Antibacterial and anticancer activity of bioactive fraction of Syzygium cumini L. seeds. HAYATI J. Biosci. 2011, 18, 118–122. [Google Scholar] [CrossRef]
- Nazif, N.M. The anthocyanin components and cytotoxic activity of Syzygium cumini (L.) fruits growing in Egypt. Nat. Prod. Sci. 2007, 13, 135–139. [Google Scholar]
- Banerjee, J.; Narendhirakannan, R. Phytochemical analyses, antibacterial, in vitro antioxidant and cytotoxic activities of ethanolic extract of Syzygium cumini (L.) seed extract. Int. J. Pharm. Sci. Res. 2011, 2, 1799–1806. [Google Scholar]
- Kale, D.K.C.; Mukundan, U. Cytotoxicity against tumor cell lines of a purified mirabilis antiviral protein isolated from root of Mirabilis jalapa. World J. Pharm. Res. 2015, 4, 1696–1710. [Google Scholar]
- Xu, J.-J.; Qing, C.; Lv, Y.-P.; Liu, Y.-M.; Liu, Y.; Chen, Y.-G. Cytotoxic rotenoids from Mirabilis jalapa. Chem. Nat. Compd. 2010, 46, 792–794. [Google Scholar] [CrossRef]
- Fares, R.; Bazzi, S.; Baydoun, S.E.; Abdel-Massih, R.M. The antioxidant and anti-proliferative activity of the Lebanese Olea europaea extract. Plant. Foods Hum. Nutr. 2011, 66, 58–63. [Google Scholar] [CrossRef]
- Nair, M.S.; Soren, K.; Singh, V.; Boro, B. Anticancer activity of fruit and leaf extracts of Averrhoa bilimbi on MCF-7 human breast cancer cell lines: A preliminary study. Austin J. Pharmacol. Ther. 2016, 4, 1082. [Google Scholar]
- Ahmed, Q.U.; Alhassan, A. Averrhoa bilimbi Linn.: A review of its ethnomedicinal uses, phytochemistry, and pharmacology. J. Pharm. Bioallied Sci. 2016, 8, 265–271. [Google Scholar] [CrossRef]
- Sumalatha, D. Antioxidant and antitumor activity of Phyllanthus emblica in colon cancer cell lines. Int. J. Curr. Microbiol. Appl. Sci. 2013, 2, 189–195. [Google Scholar]
- Qi, W.; Li, Y.; Hua, L.; Wang, K.; Gao, K. Cytotoxicity and structure activity relationships of phytosterol from Phyllanthus emblica. Fitoterapia 2013, 84, 252–256. [Google Scholar] [CrossRef]
- Desouky, S.; Ryu, S.Y.; Kim, Y.-K. A new cytotoxic acylated apigenin glucoside from Phyllanthus emblica L. Nat. Prod. Res. 2008, 22, 91–95. [Google Scholar] [CrossRef]
- Medini, F.; Bourgou, S.; Lalancette, K.; Snoussi, M.; Mkadmini, K.; Coté, I.; Abdelly, C.; Legault, J.; Ksouri, R. Phytochemical analysis, antioxidant, anti-inflammatory, and anticancer activities of the halophyte Limonium densiflorum extracts on human cell lines and murine macrophages. S. Afr. J. Bot. 2015, 99, 158–164. [Google Scholar] [CrossRef]
- Rajkumar, V.; Guha, G.; Kumar, R.A. Antioxidant and anti-neoplastic activities of Picrorhiza kurroa extracts. Food Chem. Toxicol. 2011, 49, 363–369. [Google Scholar] [CrossRef] [PubMed]
- Masood, M. Picrorhiza kurroa: An ethnopharmacologically important plant species of Himalayan region. Pure Appl. Boil. 2015, 4, 407–417. [Google Scholar] [CrossRef]
- Shashi, B.; Jaswant, S.; Madhusudana, R.J.; Kumar, S.A.; Nabi, Q.G. A novel lignan composition from Cedrus deodara induces apoptosis and early nitric oxide generation in human leukemia Molt-4 and HL-60 cells. Nitric Oxide 2006, 14, 72–88. [Google Scholar] [CrossRef]
- Sunila, E.; Kuttan, G. Immunomodulatory and antitumor activity of Piper longum Linn. and piperine. J. Ethnopharmacol. 2004, 90, 339–346. [Google Scholar] [CrossRef]
- Longato, G.; Rizzo, L.; Sousa, I.D.O.; Tinti, S.; Possenti, A.; Figueira, G.; Ruiz, A.L.T.G.; Foglio, M.; De Carvalho, J. In vitro and in vivo anticancer activity of extracts, fractions, and eupomatenoid-5 obtained from Piper regnellii leaves. Planta Med. 2011, 77, 1482–1488. [Google Scholar] [CrossRef]
- Alothman, E.A.; Awaad, A.S.; Al-Qurayn, N.A.; Al-Kanhal, H.F.; El-Meligy, R.M.; Zain, Y.M.; Alasmary, F.A.; Alqasoumi, S.I. Anticancer effect of Cenchrus ciliaris L. Saudi Pharm. J. 2018, 26, 952–955. [Google Scholar] [CrossRef]
- Awaad, A.S.; Al Qurain, N.A.; Alkanhal, H.F.; El-Meligy, R.M.; Al-Asamary, F.A. Cenchrus ciliaris L. as an Anticancer Agent. U.S. Patent application no. 15/882,929, 2019. [Google Scholar]
- Badria, F.A.; Ameen, M.; Akl, M.R. Evaluation of cytotoxic compounds from Calligonum comosum L. growing in Egypt. Zeitschrift für Naturforschung C 2007, 62, 656–660. [Google Scholar] [CrossRef]
- Vinh, L.B.; Nguyet, N.T.M.; Yang, S.Y.; Kim, J.H.; Van Thanh, N.; Cuong, N.X.; Nam, N.H.; Van Minh, C.; Hwang, I.; Kim, Y.H. Cytotoxic triterpene saponins from the mangrove Aegiceras corniculatum. Nat. Prod. Res. 2017, 33, 628–634. [Google Scholar] [CrossRef]
- Ding, L.; Dahse, H.-M.; Hertweck, C. Cytotoxic alkaloids from Fusarium incarnatum associated with the mangrove tree Aegiceras corniculatum. J. Nat. Prod. 2012, 75, 617–621. [Google Scholar] [CrossRef]
- Ramírez-Macías, I.; Marín, C.; Díaz, J.G.; Rosales, M.J.; Gutierrez-Sanchez, R.; Sánchez-Moreno, M. Leishmanicidal activity of nine novel flavonoids from Delphinium staphisagria. Sci. World J. 2012, 2012, 203646. [Google Scholar] [CrossRef][Green Version]
- Bura, F.T.; Firuzja, R.A.; Nemati, F. Cytotoxic effect of the flower and leaf bud extract of Crataegus microphylla C. Koch on HeLa cell line. IIOAB J. 2016, 7, 214–218. [Google Scholar]
- Abdel-Hameed, E.-S.S.; Bazaid, S.A.; Hagag, H.A. Chemical characterization of Rosa damascena Miller var. trigintipetala Dieck essential oil and its in vitro genotoxic and cytotoxic properties. J. Essent. Oil Res. 2016, 28, 121–129. [Google Scholar] [CrossRef]
- Khairunnisa, K.; Karthik, D. Evaluation of in vitro apoptosis induction, cytotoxic activity of Hymenodictyon excelsum (Roxb) Wall in Dalton’s lymphoma ascites (DLA) and Lung fibroblast-Mouse L929 cell lines. J. Appl. Pharm. Sci. 2014, 4, 11–17. [Google Scholar]
- Pandey, K.; Sharma, P.; Dudhe, R. Anticancer activity of Parthenium hysterophorus Linn. and Oldenlandia corymbosa Lam. by SRB Method. Open Acces Sci. Rep. 2012, 1. [Google Scholar]
- Gezici, S.; Sekeroglu, N.; Kijjoa, A. In vitro anticancer activity and antioxidant properties of essential oils from Populus alba L. and Rosmarinus officinalis L. from South Eastern Anatolia of Turkey. Indian J. Pharm. Educ. Res. 2017, 51, 498–503. [Google Scholar] [CrossRef]
- Awasare, S.; Bhujbal, S.; Nanda, R. In vitro cytotoxic activity of novel oleanane type of triterpenoid saponin from stem bark of Manilkara zapota Linn. Asian J. Pharm. Clin. Res. 2012, 5, 183–188. [Google Scholar]
- Alaklabi, A.; Arif, I.A.; Ahamed, A.; Kumar, R.S.; Idhayadhulla, A. Evaluation of antioxidant and anticancer activities of chemical constituents of the Saururus chinensis root extracts. Saudi J. Boil. Sci. 2017, 25, 1387–1392. [Google Scholar] [CrossRef]
- Lee, Y.-K.; Seo, C.-S.; Lee, C.-S.; Lee, K.-S.; Kang, S.-J.; Jahng, Y.; Chang, H.W.; Son, J.K. Inhibition of DNA topoisomerases I and II and cytotoxicity by lignans from Saururus chinensis. Arch. Pharmacal Res. 2009, 32, 1409–1415. [Google Scholar] [CrossRef]
- Afifi, M.S.; Ahmed, M.M.; Pezzuto, J.M.; Kinghornt, A.D. Cytotoxic flavonolignans and flavones from Verbascum sinaiticum leaves. Phytochemistry 1993, 34, 839–841. [Google Scholar] [CrossRef]
- Shokrzadeh, M.; Azadbakht, M.; Ahangar, N.; Hashemi, A.; Saravi, S.S.S. Cytotoxicity of hydro-alcoholic extracts of Cucurbita pepo and Solanum nigrum on HepG2 and CT26 cancer cell lines. Pharmacogn. Mag. 2010, 6, 176–179. [Google Scholar] [CrossRef] [PubMed]
- Patel, S.; Gheewala, N.; Suthar, A.; Shah, A. In vitro cytotoxicity activity of Solanum nigrum extract against HeLa cell line and Vero cell line. Int. J. Pharm. Pharm. Sci. 2009, 1, 38–46. [Google Scholar]
- Maqsood, M.; Qureshi, R.; Ikram, M.; Ahmad, M.S.; Jabeen, B.; Asi, M.R.; Khan, J.A.; Ali, S.; Lilge, L. In vitro anticancer activities of Withania coagulans against HeLa, MCF-7, RD, RG2, and INS-1 cancer cells and phytochemical analysis. Integr. Med. Res. 2018, 7, 184–191. [Google Scholar] [CrossRef] [PubMed]
- Shaikh, R.; Pund, M.; Dawane, A.; Iliyas, S. Evaluation of anticancer, antioxidant, and possible anti-inflammatory properties of selected medicinal plants used in Indian traditional medication. J. Tradit. Complement. Med. 2014, 4, 253–257. [Google Scholar] [CrossRef] [PubMed]
- Bean, M.F.; Antoun, M.; Abramson, D.; Chang, C.-J.; McLaughlin, J.L.; Cassady, J.M. Cucurbitacin B and Isocucurbitacin B: Cytotoxic components of Helicteres isora. J. Nat. Prod. 1985, 48, 500. [Google Scholar] [CrossRef]
- Hegde, K.; Fathima Jazeela, M.; Vijetha Poojary, K.; Satish, S. Anticancer potentials of the plant Aquilaria malaccensis leaves. Indian J. Pharm. Pharmacol. 2018, 5, 135–140. [Google Scholar] [CrossRef]
- Ibrahim, A.; Al-Rawi, S.; Majid, A.A.; Rahman, N.A.; Salah, K.A.-; Ab Kadir, M. Separation and fractionation of Aquilaria Malaccensis oil using supercritical fluid extraction and tthe cytotoxic properties of the extracted oil. Procedia Food Sci. 2011, 1, 1953–1959. [Google Scholar] [CrossRef]
- Adam, A.; Tajuddin, S.; Sudmoon, R.; Chaveerach, A.; Abdullah, U.; Mahat, M.; Mohamed, R. Chemical constituents and toxicity effects of leaves from several agarwood tree species (Aquilaria). J. Trop. For. Sci. 2018, 30, 342–353. [Google Scholar] [CrossRef]
- Chacko, T.; Menon, A.; Nair, S.V.; AlSuhaibani, E.; Nair, C.K.K. Cytotoxic and antitumor activity of the extract of Clerodendron infortunatum: A mechanistic study. Am. J. Phytomed. Clin. Therapeut. 2015, 2, 145–158. [Google Scholar]
- Ghagane, S.C.; Puranik, S.I.; Kumbar, V.M.; Nerli, R.B.; Jalalpure, S.S.; Hiremath, M.B.; Neelagund, S.; Aladakatti, R. In vitro antioxidant and anticancer activity of Leea indica leaf extracts on human prostate cancer cell lines. Integr. Med. Res. 2017, 6, 79–87. [Google Scholar] [CrossRef]
- Handoussa, H.; Hanafi, R.; Eddiasty, I.; El-Gendy, M.; El Khatib, A.; Linscheid, M.W.; Mahran, L.; Ayoub, N. Anti-inflammatory and cytotoxic activities of dietary phenolics isolated from Corchorus olitorius and Vitis vinifera. J. Funct. Foods 2013, 5, 1204–1216. [Google Scholar] [CrossRef]
- Nirmala, J.G.; Akila, S.; Narendhirakannan, R.; Chatterjee, S. Vitis vinifera peel polyphenols stabilized gold nanoparticles induce cytotoxicity and apoptotic cell death in A431 skin cancer cell lines. Adv. Powder Technol. 2017, 28, 1170–1184. [Google Scholar] [CrossRef]
- Srivastava, P.; Srivastava, A. In vitro anticancer activity of ethanolic extract of Curcumin longa (Turmeric) in HEp-2 cell lines. Int. J. Eng. Res. General Sci. 2015, 3, 495–508. [Google Scholar]
- Ramsewak, R.; DeWitt, D.; Nair, M. Cytotoxicity, antioxidant and anti-inflammatory activities of Curcumins I–III from Curcuma longa. Phytomedicine 2000, 7, 303–308. [Google Scholar] [CrossRef]
- Kuttan, R.; Bhanumathy, P.; Nirmala, K.; George, M. Potential anticancer activity of turmeric (Curcuma longa). Cancer Lett. 1985, 29, 197–202. [Google Scholar] [CrossRef]
- Tyagi, A.K.; Prasad, S.; Yuan, W.; Li, S.; Aggarwal, B.B. Identification of a novel compound (β-sesquiphellandrene) from turmeric (Curcuma longa) with anticancer potential: Comparison with curcumin. Investig. New Drugs 2015, 33, 1175–1186. [Google Scholar] [CrossRef]
- Atsumi, T.; Murakami, Y.; Shibuya, K.; Tonosaki, K.; Fujisawa, S. Induction of cytotoxicity and apoptosis and inhibition of cyclooxygenase-2 gene expression, by curcumin and its analog, α-diisoeugenol. Anticancer Res. 2005, 25, 4029–4036. [Google Scholar] [PubMed]
- Kim, J.S.; Lee, S.I.; Park, H.W.; Yang, J.H.; Shin, T.-Y.; Kim, Y.-C.; Baek, N.-I.; Kim, S.-H.; Choi, S.U.; Kwon, B.-M.; et al. Cytotoxic components from the dried rhizomes of Zingiber officinale Roscoe. Arch. Pharmacal Res. 2008, 31, 415–418. [Google Scholar] [CrossRef] [PubMed]
- Jeena, K.; Liju, V.B.; Kuttan, R. Antitumor and cytotoxic activity of ginger essential oil (Zingiber officinale Roscoe). Int. J. Pharm. Pharm. Sci. 2015, 7, 341–344. [Google Scholar]
- Kurapati, K.R.V.; Samikkannu, T.; Kadiyala, D.B.; Zainulabedin, S.M.; Gandhi, N.; Sathaye, S.S.; Indap, M.A.; Boukli, N.; Rodriguez, J.W.; Nair, M. Combinatorial cytotoxic effects of Curcuma longa and Zingiber officinale on the PC-3M prostate cancer cell line. J. Basic Clin. Physiol. Pharmacol. 2012, 23, 139–146. [Google Scholar] [CrossRef] [PubMed]
- Monga, M.; Sausville, E. Developmental Therapeutics Program at the NCI: Molecular target and drug discovery process. Leukeumia 2002, 16, 520–526. [Google Scholar] [CrossRef]
- Jaman, S.; Sayeed, A. Ellagic acid, sulforaphane, and ursolic acid in the prevention and therapy of breast cancer: Current evidence and future perspectives. Breast Cancer 2018, 25, 517–528. [Google Scholar] [CrossRef]
- Manayi, A.; Nikan, M.; Nobakht-Haghighi, N.; Abdollahi, M.; Haghighid, N.N. Advances in the anticancer value of the ursolic acid through nanodelivery. Curr. Med. Chem. 2019, 25, 4866–4875. [Google Scholar] [CrossRef]
- Efferth, T. From ancient herb to modern drug: Artemisia annua and artemisinin for cancer therapy. Semin. Cancer Boil. 2017, 46, 65–83. [Google Scholar] [CrossRef]
- Jia, Y.; Tong, X.; Fan, J. Effect of brazilin on apoptosis and autophagy of tongue cancer Tca8113 cells and its molecular mechanism. J. South. Med. Univ. 2019, 39, 351–356. [Google Scholar]
- Zhang, T.; He, J.; Zhang, S.; Wang, G.; Ma, E.; Wang, J.; Yang, X.; Zheng, Y.-W.; Zhang, J. Brazilin induces T24 cell death through c-Fos and GADD45β independently regulated genes and pathways. IUBMB Life 2018, 70, 1101–1110. [Google Scholar] [CrossRef]
- Bin Sayeed, M.S.; Ameen, S.S. Beta-Sitosterol: A promising but orphan nutraceutical to fight against cancer. Nutr. Cancer 2015, 67, 1216–1222. [Google Scholar] [CrossRef]
- Tsai, F.-S.; Lin, L.-W.; Wu, C.-R. Lupeol and its role in chronic diseases. In Drug Discovery from Mother Nature; Springer Science and Business Media: Basel, Switzerland, 2016; Volume 929, pp. 145–175. [Google Scholar]
- Devassy, J.G.; Nwachukwu, I.; Jones, P.J. Curcumin and cancer: Barriers to obtaining a health claim. Nutr. Rev. 2015, 73, 155–165. [Google Scholar] [CrossRef]
- Yang, C.S.; Wang, H. Cancer preventive activities of tea catechins. Molecules 2016, 21, 1679. [Google Scholar] [CrossRef]
- Jiang, M.; Zhu, M.; Wang, L.; Yu, S. Antitumor effects and associated molecular mechanisms of myricetin. Biomed. Pharmacother. 2019, 120, 109506. [Google Scholar] [CrossRef]
- Devi, K.P.; Rajavel, T.; Habtemariam, S.; Nabavi, S.F.; Nabavi, S.M. Molecular mechanisms underlying anticancer effects of myricetin. Life Sci. 2015, 142, 19–25. [Google Scholar] [CrossRef] [PubMed]
- Rauf, A.; Imran, M.; Khan, I.A.; Ur-Rehman, M.-; Gilani, S.A.; Mehmood, Z.; Mubarak, M.S. Anticancer potential of quercetin: A comprehensive review. Phytotherapy Res. 2018, 32, 2109–2130. [Google Scholar] [CrossRef]
- Reyes-Farias, M.; Carrasco-Pozo, C. The Anticancer effect of Quercetin: Molecular implications in cancer metabolism. Int. J. Mol. Sci. 2019, 20, 3177. [Google Scholar] [CrossRef] [PubMed]
- Rupani, R.; Chavez, A. Medicinal plants with traditional use: Ethnobotany in the Indian subcontinent. Clin. Dermatol. 2018, 36, 306–309. [Google Scholar] [CrossRef]
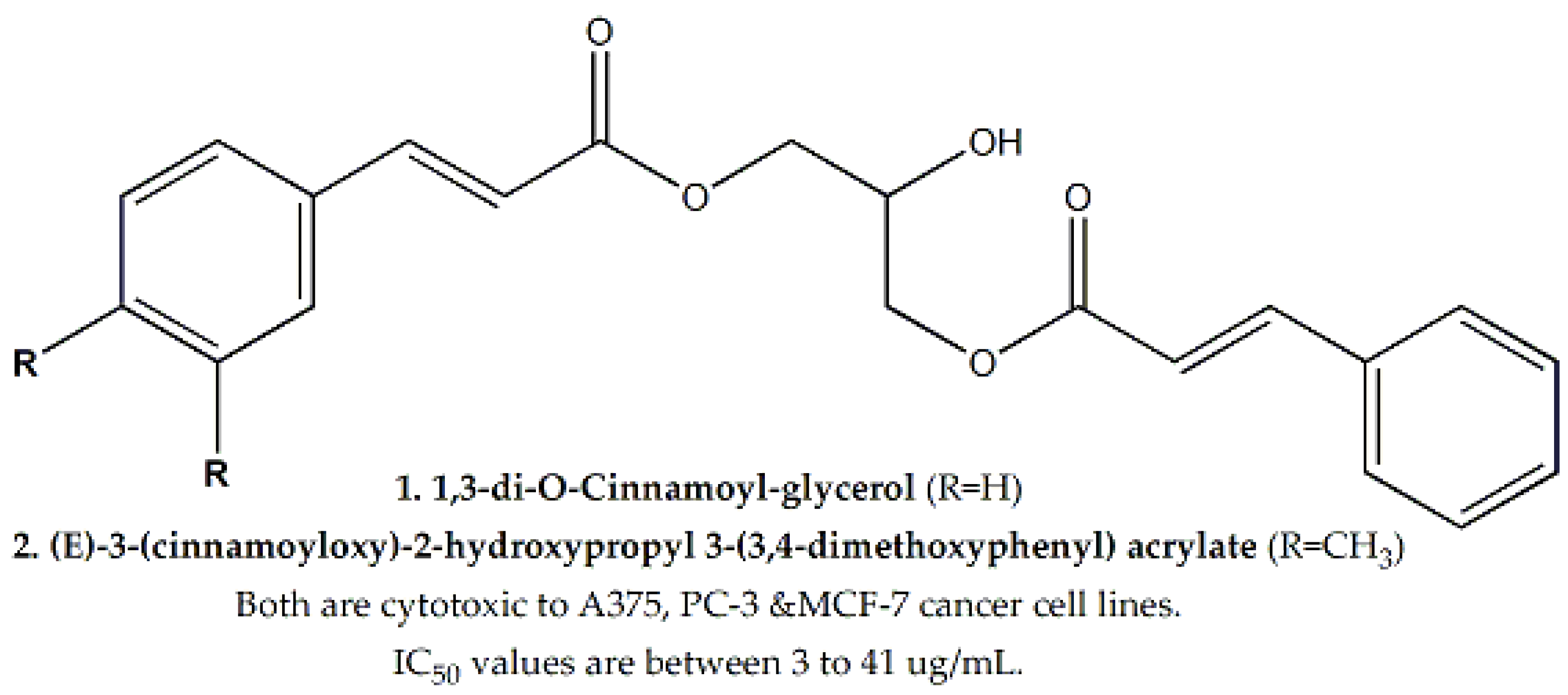
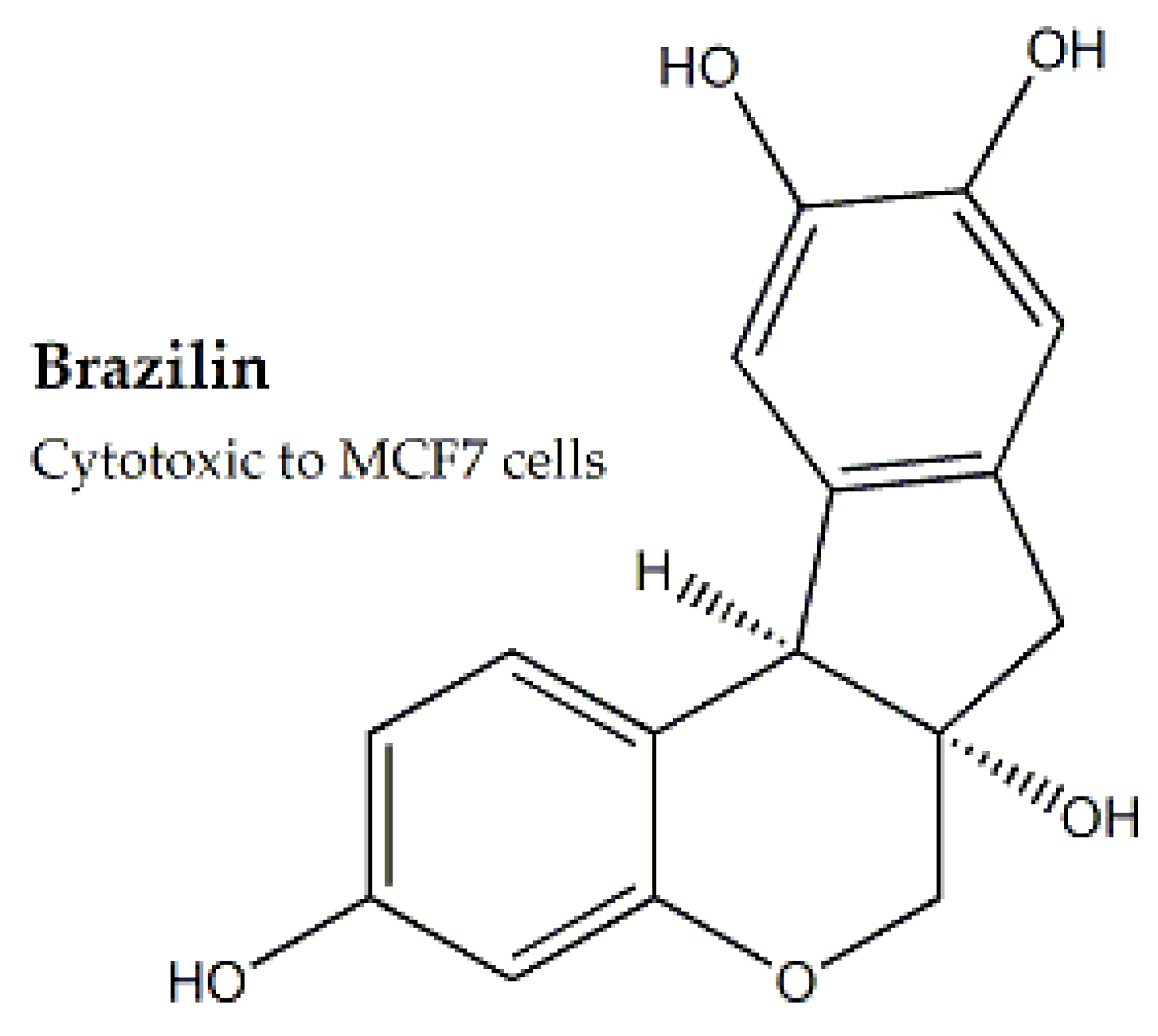
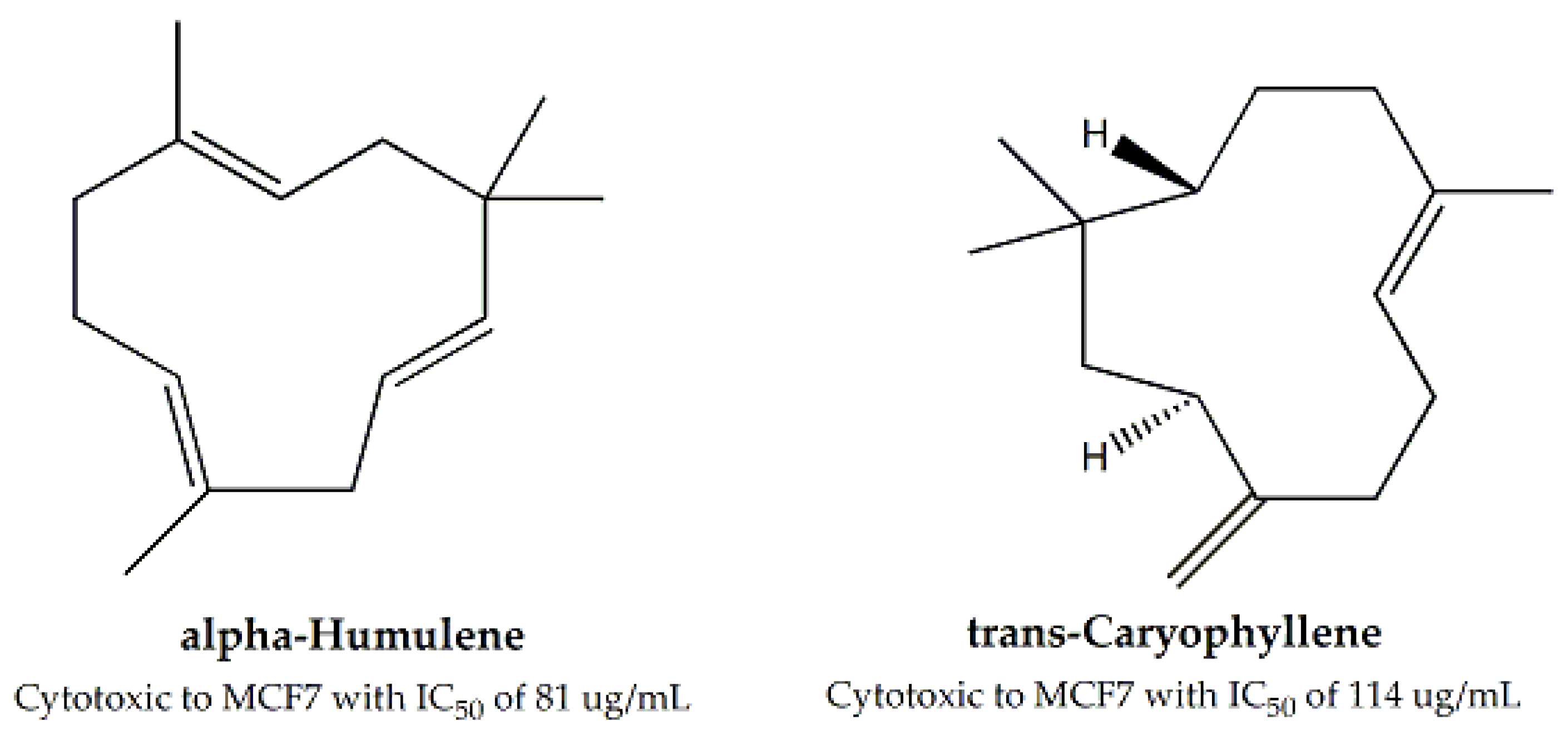
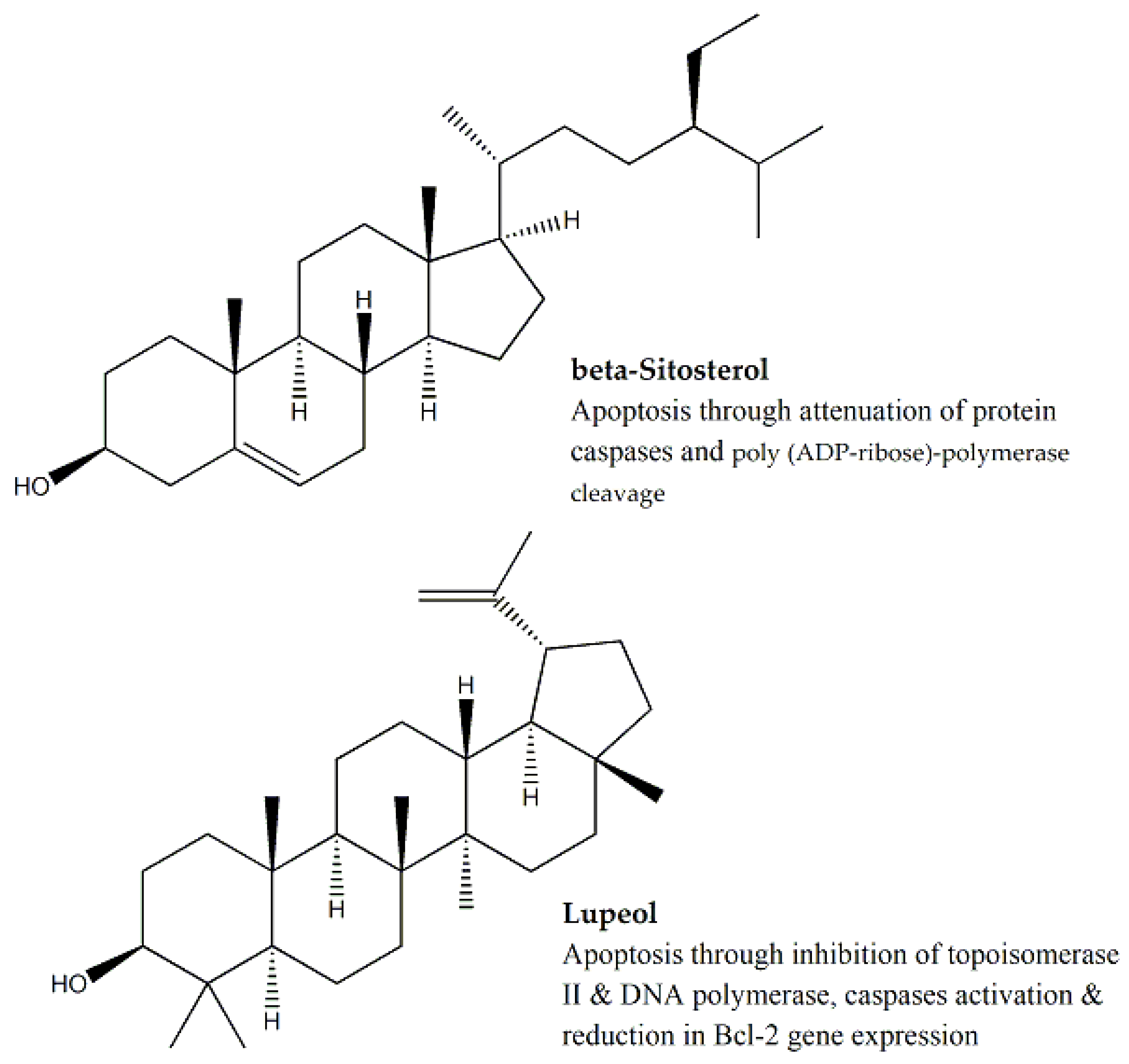
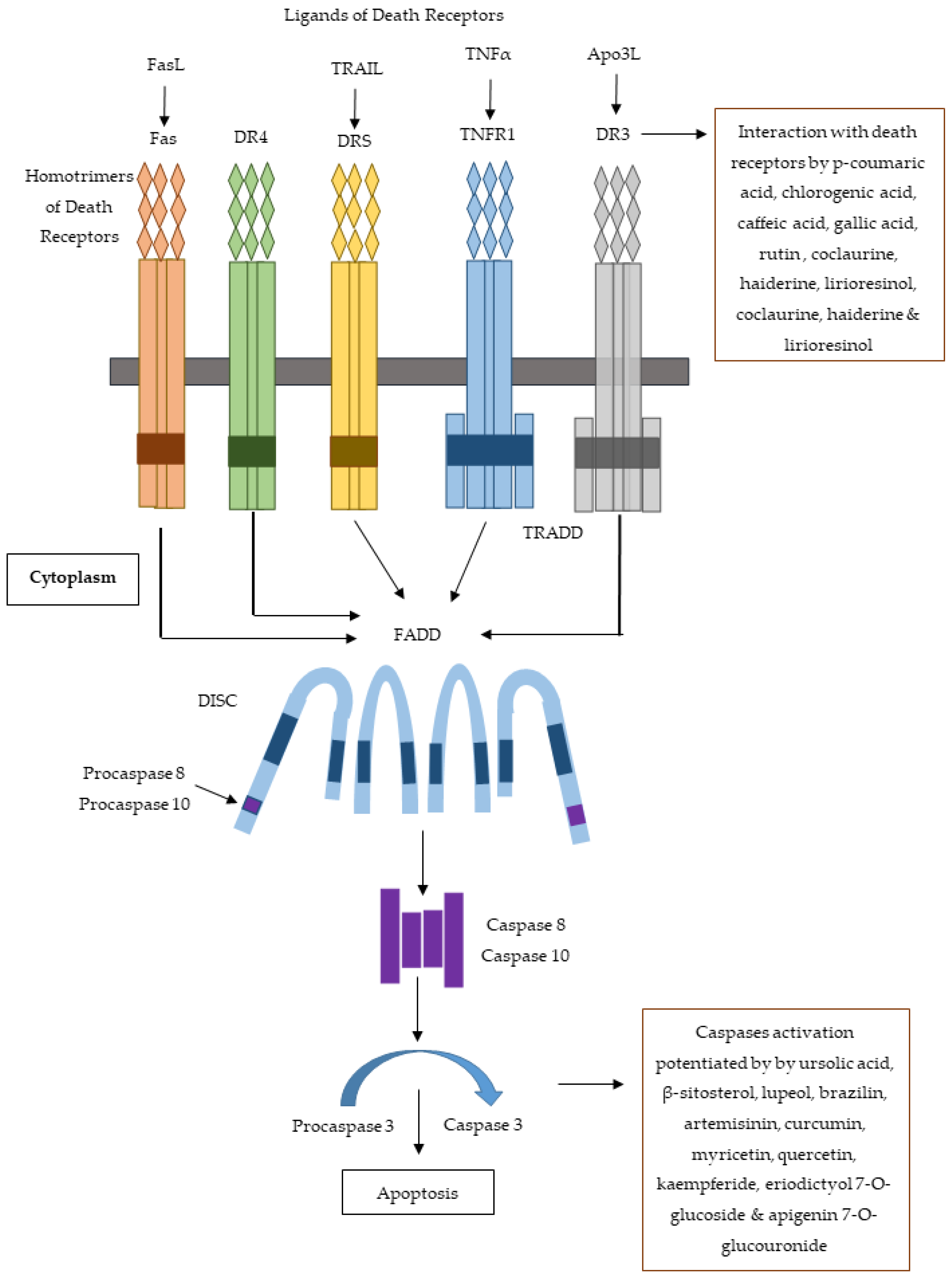
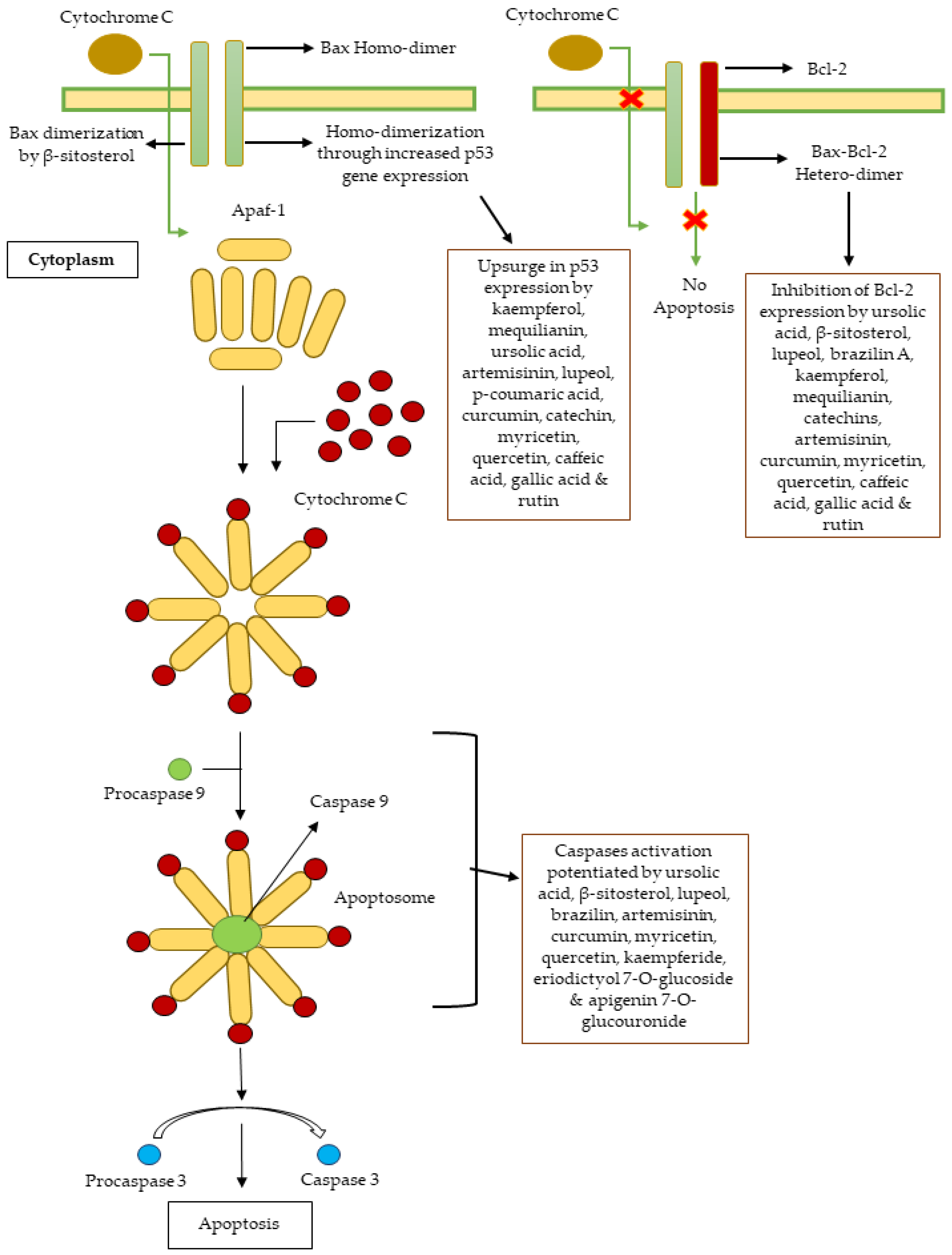
| Family | Genus and Species | Parts Used | Study Methods | Cell Line | IC50 (μg/mL) | Bioactive Compounds | IC50 (μg/mL) for Bioactive Compound | Mechanism of Cytotoxicity | Reference |
|---|---|---|---|---|---|---|---|---|---|
| Acanthaceae | Barleria grandiflora | Leaves | In vivo DAL induced ascitic tumor model in mice | DLA, A-549 and Vero | - | - | - | Upsurge in ascitic fluid | [17,18] |
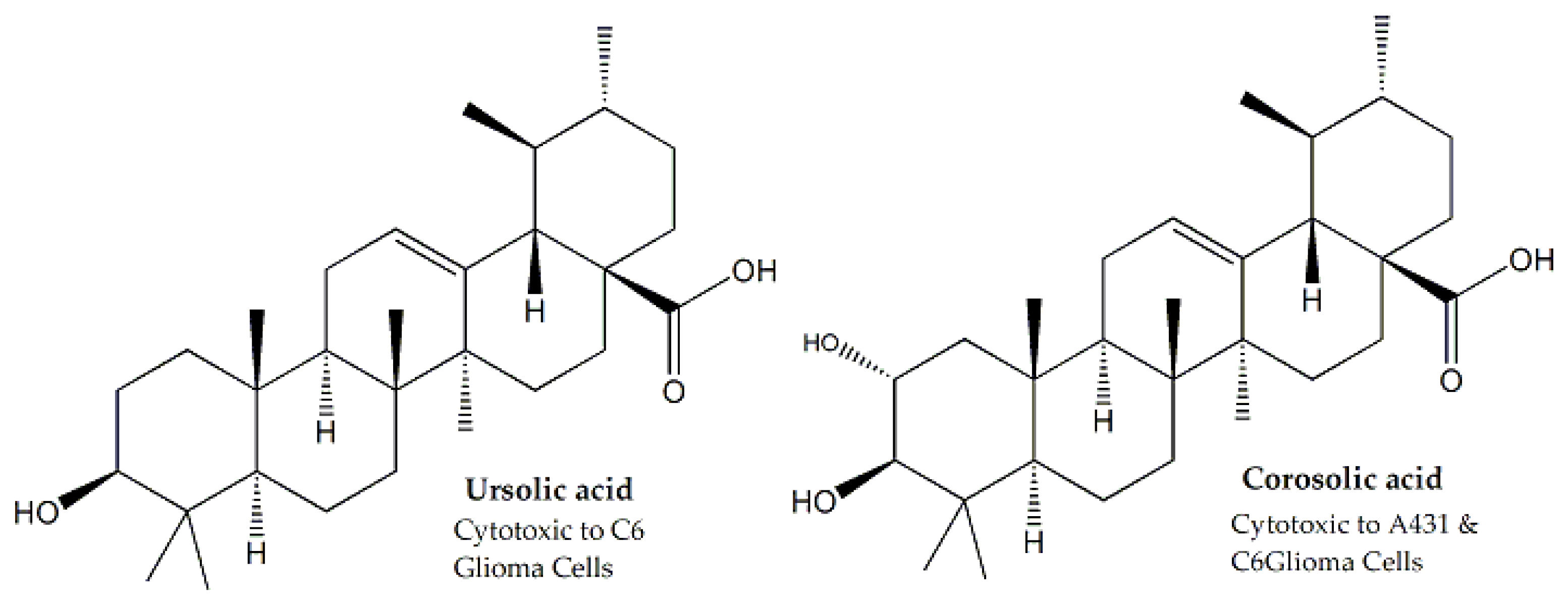
| Actinidiaceae | Saurauja roxburghii | Leaves | MTT assay | A431 and C6 rat glioma | - | - | - | Interaction with caspases 3 and 9 topoisomerase I and p21WAF1 cell-cycle regulator | [20,21] |
| Amaryllidaceae | Narcissus tazetta | Aerial parts, Flowers | MTT assay | MCF-7 and Hep-2 | - | - | - | - | [22] |
| Anacardiaceae | Cotinus coggygria | Leaves | MTT assay | HeLa and Vero | 293 for HeLa | - | - | - | [23] |
| Lannea coromandelica | Barks, Leaves | MTT assay | AGS, HT-29, MCF-7 and MDA-MB-231 | 90, 520, 270 and 160, respectively | 2-Palmitoylglycerol, myricadiol, pyrogallol, isovanillin etc. | - | Apoptosis and DNA fragmentation | [24,25,26] | |
| Annonaceae | Annona muricata | Leaves | MTT assay | EACC, MDA and SKBR3 | 335, 248 and 202, respectively | - | - | - | [27] |
| Annona squamosa | Flowers | MTT assay | MCF-7 and Vero | 6.9 and 75, respectively | Eupafolin, apigenin, rhamnetin etc. | - | - | [28] | |
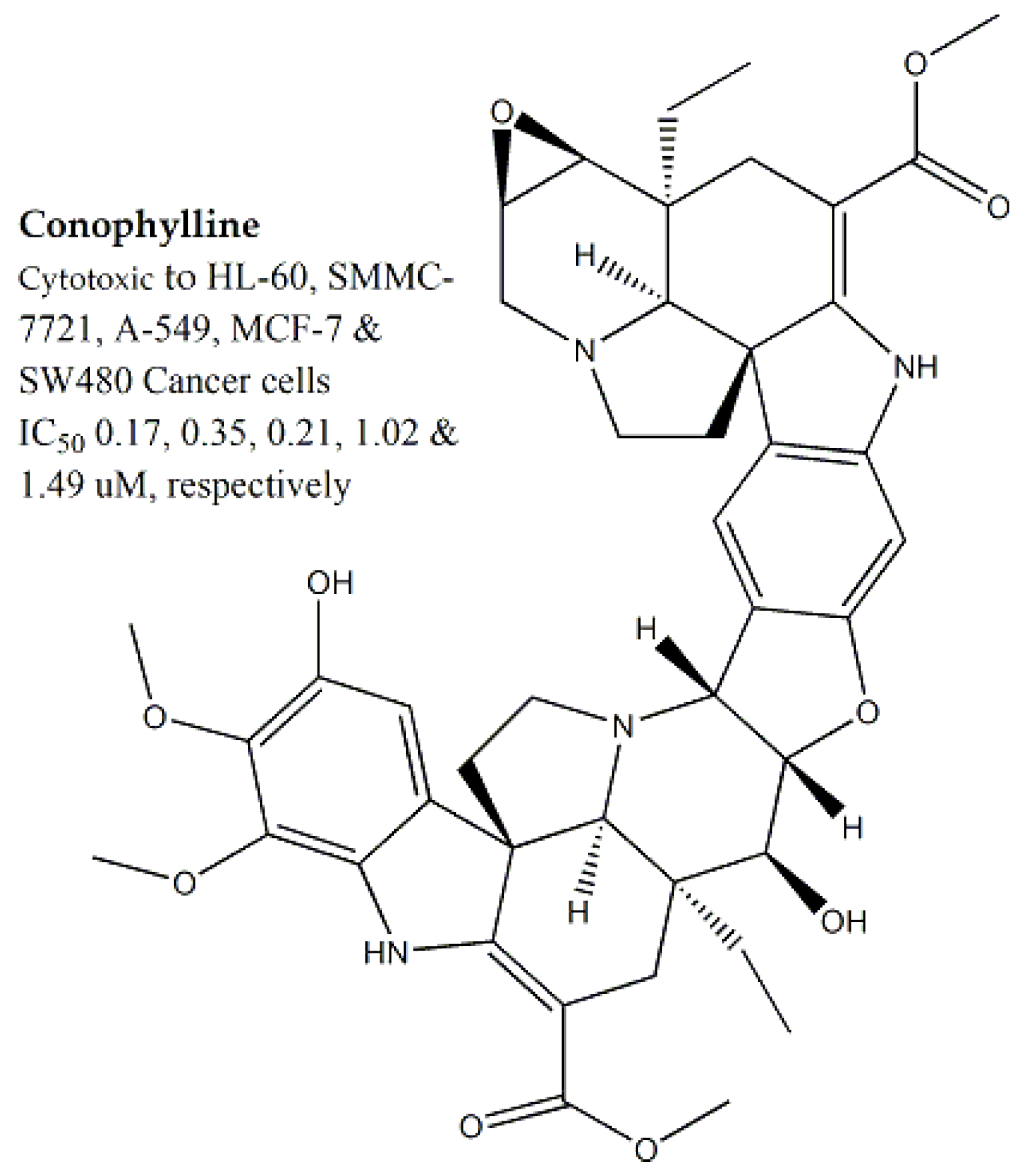
| Apocynaceae | Tabernaemontana divaricata | Flowers | MTT assay | HL-60, SMMC-7721, A-549, MCF-7 and SW480, NIH3T3 and HeLa | 35.3 and >100 for NIH3T3 and HeLa, respectively | Ervachinine, cononitarine, conofoline, conophylline and cisplatin | Ranging between 0.17 to 1.49 for conophylline | - | [29,30] |
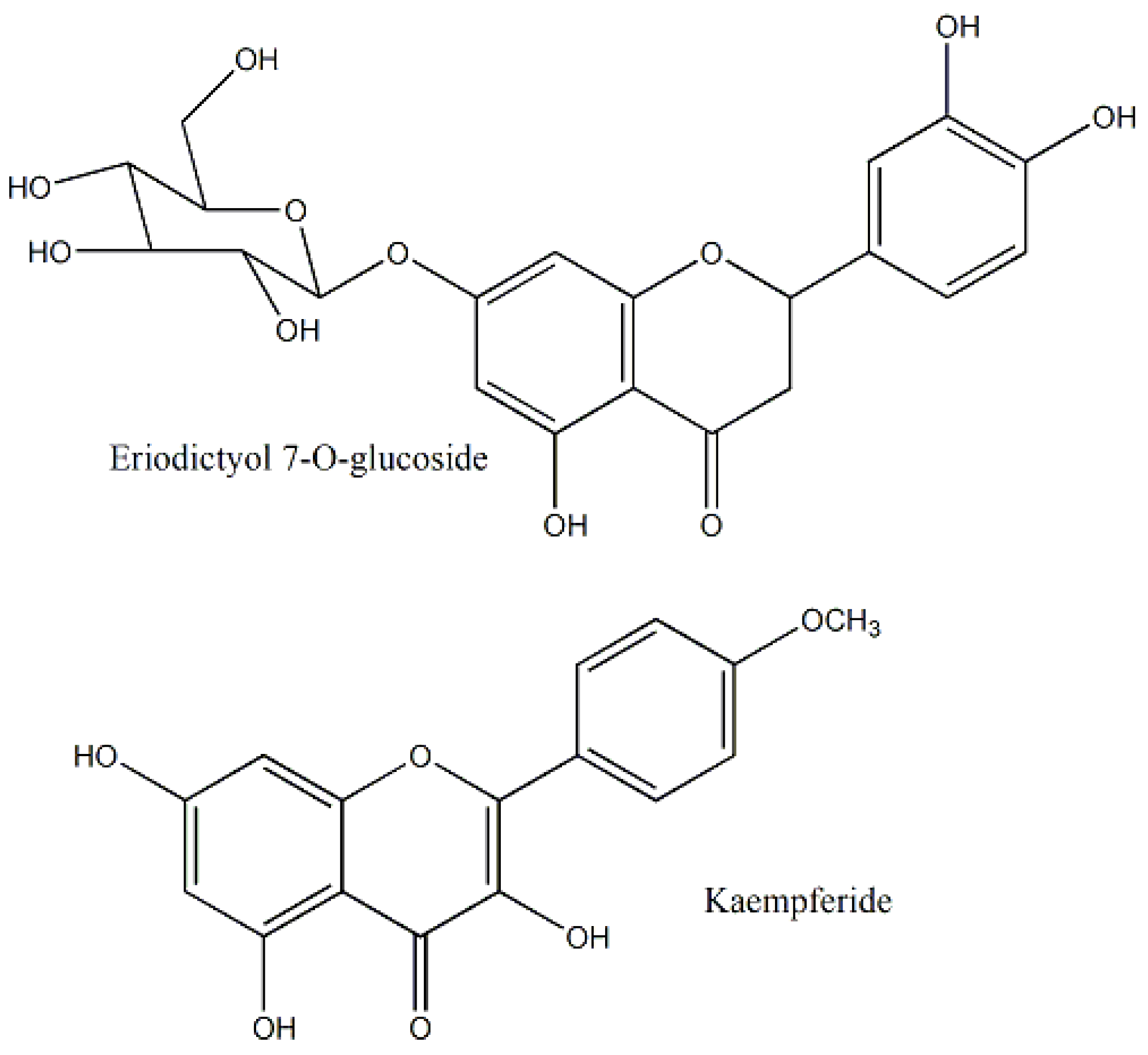
| Araceae | Luffa cylindrica | Aerial Parts | MTT assay | MCF-7 and Hep-2, BT-474, MDA-MB-231 and HepG2 | 329.2 | Kaempferide, eriodictyol 7-O-glucoside and apigenin 7-O-glucouronide | - | Interaction with caspases 3 and 8 | [22,31,32] |
| Aristolochiaceae | Aristolochia ringens | Roots | In vivo S 180 induced ascetic and solid tumor model in mice | HeLa, A-431, A-549, PC-3, HCT-116 and THP-1 | Ranging between 3 to 24 | - | - | - | [33] |
| Aristolochia longa | Roots | MTT assay | MCF-7, HT-29, H5-6 and N2A | - | - | - | - | [34] | |
| Asphodelaceae | Aloe vera | Whole Plant | MTT assay | HepG2 | 10.5 | - | - | Increased p53 and reduced Bcl-2 gene expression | [35] |
| Aspleniaceae | Asplenium nidus | Whole Plant | MTT assay | HepG2 and HeLa | - | Gliricidin-7-O- hexoside | About 507 | - | [36] |
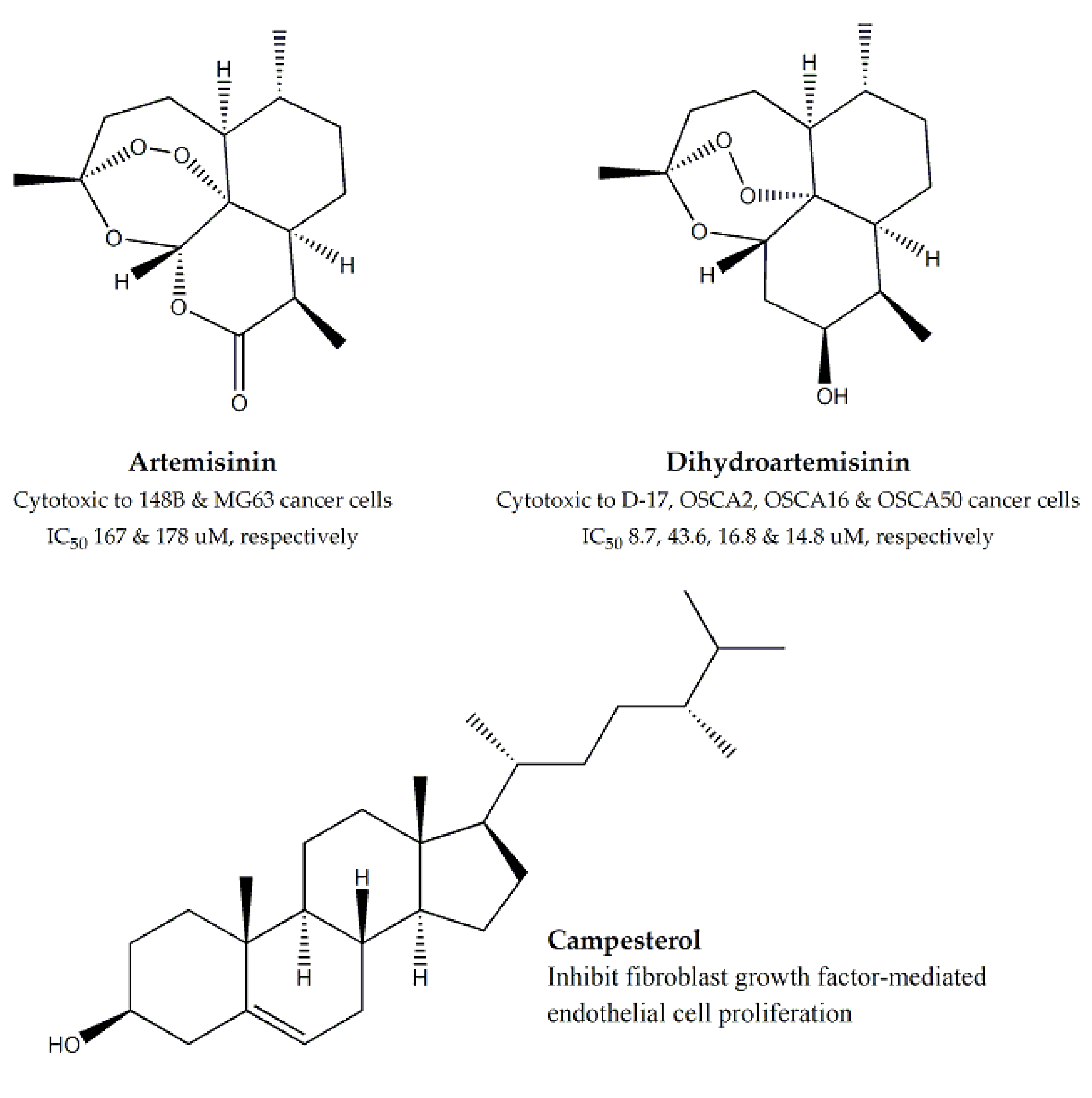
| Asteraceae | Artemisia annua | Aerial Parts | MTT assay | 148B, MG63, D-17, OSCA2, OSCA16 and OSCA50 | 167 and 178 for 148B and MG63, respectively | Dihydro- artemisinin | Ranging between 8.7 to 43.6 | - | [37,38,39,40] |
| Bidens pilosa | Whole Plant | MTT and Comet assays | HeLa and KB | 88.6 and 372.4, respectively | - | - | - | [41] | |
| Centaurea antiochia | Aerial Parts | MTT assay | Vero and HeLa | - | - | - | - | [23] | |
| Centaurea nerimaniae | Aerial Parts | MTT assay | Vero and HeLa | - | - | - | - | [23,42] | |
| Chrysanthemum coronarium | Aerial Parts | In vitro MTT assay and in vivo chorioallantoic membrane (CAM) model | MCF-7, T47D, CACO-2, HRT18, A375.S2 and WM1361A | 75.7, 79.8 and 138.5 for WM1361A, T47D and MCF-7 | Campesterol | - | Inhibition of fibroblast growth factor-mediated cell proliferation | [43,44] | |
| Inula viscosa | Flowers | MTT assay | MCF-7 and Hep-2 | 15.78 for MCF-7 | - | - | - | [22] | |
| Parthenium hysterophorus | Leaves | MTT and SRB assays | DU-145, THP-1, MCF-7 and K562 | - | - | - | - | [42,45] | |
| Phagnalon rupestre | Aerial Parts | MTT assay | MCF-7 and Hep-2 | 93.3 and 97, respectively | - | - | - | [22] | |
| Scorzonera tomentosa | Aerial Parts | MTT assay | Vero and HeLa | 987 and 195, respectively | - | - | - | [23] | |
| Senecio scandens | Leaves | MTT and Clonogenic assays | DL, MCF-7 and HeLa | 27, 24 and 18, respectively | - | - | - | [46] | |
| Berberidaceae | Berberis aristata | Roots, Stem | MTT assay | MCF-7, DWD, Hop62 and A2780 | 71 and 220 for MCF-7 and DWD | - | - | - | [47,48] |
| Bignoniaceae | Tecoma stans | Leaves, Flowers | MTT assay | A549 and HepG2 | - | Rutin, luteolin, diosmetin, skytanthine, etc. | - | - | [49,50,51] |
| Boraginaceae | Cordia dichotoma | Leaves | MTT, DCFH-DA and DAPI staining assays | PC3 | 74.5 | - | - | apoptosis, nuclear condensation and ROS production | [52] |
| Bromeliaceae | Tillandsia recurvata | Whole Plant | WST-1 assay | A375, MCF-7 and PC-3 | 1, 40.5 and 6, respectively | 1,3-di-O-Cinnamoyl-glycerol and (E)-3-(cinnamoyloxy)-2-hydroxypropyl 3-(3,4-dimethoxyphenyl) acrylate | Ranging between 3 to 41 | - | [53] |
| Caesalpiniaceae | Caesalpinia sappan | Heartwood, Leaves | MTT assay | MCF-7 and A-549 | - | Brazilin A | - | Reduction of Bcl-2 apoptotic inhibitor | [54] |
| Saraca asoca | Leaves | MTT assay | AGS | 20 | - | - | - | [24] | |
| Compositae | Gnaphalium luteoalbum | Leaves | MTT assay | VERO, NIH3T3, AGS, HT-29, MCF-7 and MDA-MB-231 | 980 and 340 for AGS and MCF-7, respectively | Apigenin, luteolin, jaceosidin, gnaphalin, etc. | - | - | [24,55] |
| Dilleniaceae | Dillenia pentagyna | Stem Bark | In vivo DAL induced ascitic tumor model in mice | DL, MCF-7 and HeLa | 25.8, 41.6 and 76.8, respectively | - | - | Reduction in glutathione level | [46,56] |
| Dillenia indica | Leaves | MTT assay | MCF-7 and MDA-MB-231 | 340 and 540, respectively | - | - | - | [24] | |
| Dipsacaceae | Pterocephalus pulverulentus | Aerial Parts | MTT assay | MCF-7 and Hep-2 | - | - | - | - | [22] |
| Ebenaceae | Diospyros peregrina | Leaves, Fruits | MTT assay | MCF-7, HepG2 and MDA-MB-231 | 7, 64.4 and 33, respectively | - | - | - | [24,57] |
| Ericaceae | Arbutus andrachne | Aerial Parts | MTT assay | MCF-7, T47D, CACO-2, HRT-18, A375.S2 and WM1361A | - | - | - | - | [43] |
| Euphorbiaceae | Croton caudatus | Leaves | MTT and Clonogenic assays | DL, MCF-7 and HeLa | 29.7 for DL | - | - | - | [46] |
| Euphorbia tirucalli | Leaves, Stem | In vitro cell viability study with CCK-8 | MiaPaCa-2 | - | - | - | - | [58] | |
| Fabaceae | Adenanthera pavonina | Stem Bark | In vivo DAL induced ascitic tumor model in mice | DAL | - | - | - | - | [18] |
| Ononis hirta | Aerial Parts | MTT assay | MCF-7, Hep-2 and Vero | 28, 48.8 and 41.9, respectively | - | - | - | [22] | |
| Ononis sicula | Aerial Parts | MTT assay | MCF-7, Hep-2 and Vero | 66, 75.3 and 79.5, respectively | - | - | - | [22] | |
| Gramineae | Zea mays | Leaves | MTT and SRB assays | Hep-2, A549, SK-OV-3, SK-MEL-2, XF-489 and HCT-15 | - | Maysin | Ranging between 32 to 62 | - | [59,60] |
| Hypericaceae | Hypericum kotschyanum | Aerial Parts | MTT assay | HeLa and Vero | 507 and 367, respectively | - | - | - | [23] |
| Labiatae | Salvia pinardi | Aerial Parts | MTT assay | MCF-7 and Hep-2 | 85.5 for MCF-7 | - | - | - | [22] |
| Lamiaceae | Lavandula angustifolia | Flowers | MTT assay | MCF-7 and Hep-2 | - | - | - | - | [22] |
| Nepeta italica | Aerial Parts | MTT assay | HeLa and Vero | 980 and >1000, respectively | - | - | - | [23] | |
| Plectranthus stocksii | Leaves, Stem | MTT assay | RAW264.7, Caco-2 and MCF-7 | 9, 36.1 and 48.9, respectively | - | - | - | [61] | |
| Salvia officinalis | Leaves, Stem | “Alamar Blue” resazurin reduction assays | MCF-7, B16F10 and HeLa | Ranging between 14 – 36 | α-Humulene and trans-caryophyllene | For MCF-7, 81 and 114, respectively | - | [62] | |
| Ocimum sanctum | Leaves | In vivo S 180 induced mice model | DMBA-induced hamster and S-180 induced mice | - | - | - | Regulation of humoral immunity and stimulation of cell mediated immunity | [63,64,65,66] | |
| Origanum sipyleum | Aerial Parts | MTT assay | HeLa and Vero | >1000 | - | - | - | [23] | |
| Salvia hypargeia | Aerial Parts | MTT assay | Vero and HeLa | > 1000 | - | - | - | [23] | |
| Teucrium polium | Aerial Parts | MTT assay | MCF-7, Hep-2, T47D, CACO-2, HRT18, A375.S2 and WM1361A | - | - | - | - | [22,43] | |
| Teucrium sandrasicum | Aerial Parts | MTT assay | HeLa and Vero | 513 and 593, respectively | - | - | - | [23] | |
| Malvaceae | Hibiscus calyphyllus | Aerial Parts | MTT assay | HepG2 and MCF-7 | 14.5 and 25.1, respectively | Ursolic acid, β-sitosterol and lupeol | - | Interaction with caspases 3 and 9, topoisomerase I, Bcl-2, Bax, DNA polymerase and poly (ADP-ribose)-polymerase | [67,68,69,70] |
| Hibiscus deflersii | Aerial Parts | MTT assay | HepG2 and MCF-7 | 14.4 and 11.1, respectively | Ursolic acid, β-sitosterol and lupeol | - | Interaction with caspases 3 and 9, topoisomerase I, Bcl-2, Bax, DNA polymerase and poly (ADP-ribose)-polymerase | [67,68,69,70] | |
| Hibiscus micranthus | Aerial Parts | MTT assay | HepG2 and MCF-7 | 27.6 and 24.1, respectively | Ursolic acid, β-sitosterol and lupeol | - | Interaction with caspases 3 and 9, topoisomerase I, Bcl-2, Bax, DNA polymerase and poly (ADP-ribose)-polymerase | [67,68,69,70] | |
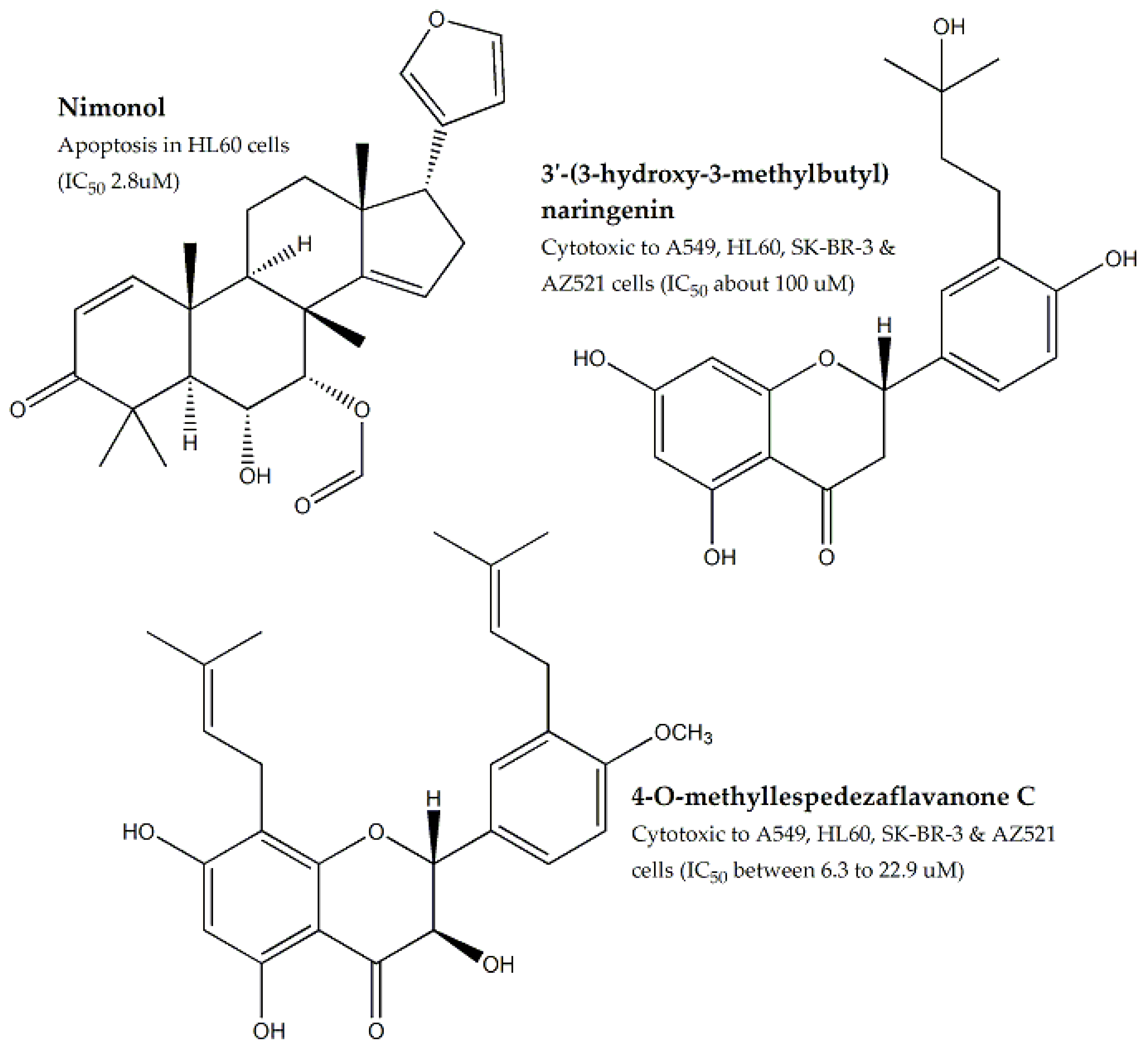
| Meliaceae | Azadirachta indica | Leaves, Seeds | MTT assay | HT-29, A-549, MCF-7, HepG-2, MDBK, EAC, HL60, SK-BR-3, AZ521, SW-480, and SMMC7721 | Ranging between 83.5 to 212.2 | Nimonol, 3′-(3-hydroxy-3-methyl-butyl)naringenin, 4′-O-methyl- lespedezaflavanone C, triterpenoids, limonoids etc. | Ranging between 4.2 to 100 | - | [71,72,73,74,75,76,77] |
| Menispermaceae | Cocculus hirsutus | Aerial Parts | MTT assay | MCF-7 | 39.1 | Coclaurine, haiderine, lirioresinol etc. | - | Interactions with Aurora kinase, c-Kit, FGF, NF-kB, Bcl-xL and VEGF | [78,79] |
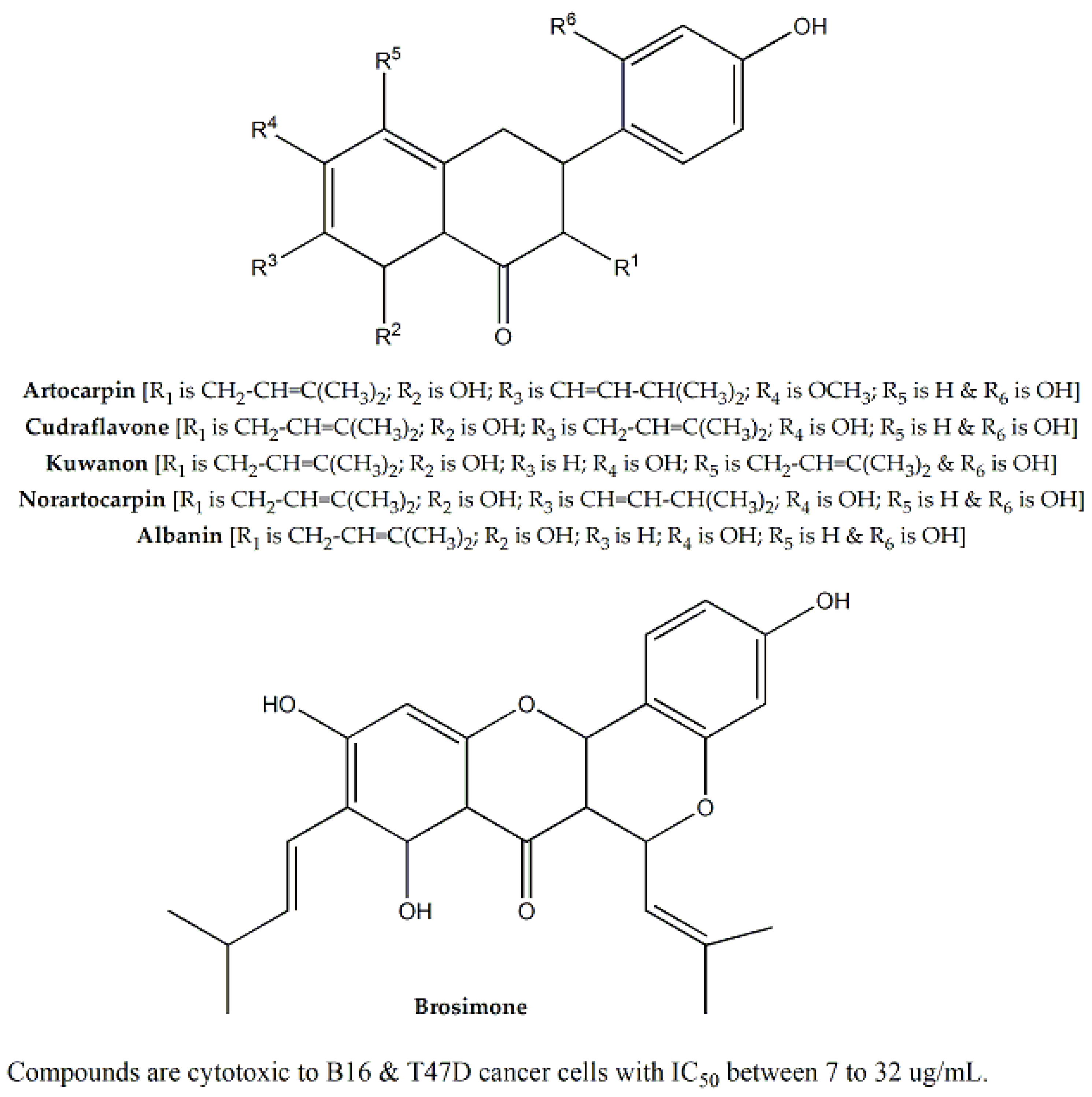
| Moraceae | Artocarpus heterophyllus | Seeds | SRB assay and MTT assays | HEK293, A549, HeLa, MCF-7, B16 and T47D | 35.3 for A549 | Noratocarpin, cudraflavone, artocarpin, brosimone, kuwanon, albanin, etc. | Ranging between 7 to 32 | Inhibition of melanin biosynthesis | [80,81,82,83] |
| Ficus beecheyana | Roots | MTT assay | AGS, SW-872, HL-60 and HepG2 | - | p-Coumaric acid, chlorogenic acid, caffeic acid, gallic acid, rutin etc. | - | Interaction with Fas, Fas-L, p53, Bcl-2 and caspases (3,8,9) proteins | [84] | |
| Ficus carica | Fruits, Leaves | “Alamar Blue” resazurin reduction assays | MCF-7, B16F10 and HeLa | 440, 880 and >1000, respectively | - | - | - | [62] | |
| Ficus racemosa | Fruits | SRB assay | MCF-7, DLA, HL-60, HepG2, NCI-H23 and HEK-293T | 80, 175, 276.9 and 363 for MCF-7, DLA, HL-60 and HepG2, respectively | - | - | - | [85,86,87] | |
| Morus nigra | Leaves | MTT assay | OVCAR-8, SF-295, HCT-116 and HeLa | - | - | - | - | [88,89] | |
| Myristicaceae | Myristica fragrans | Barks | MTT assay | KB, K-562, HCT-116 and MCF-7 | 75 for KB | 38 Essential oils | Ranging between 2.11 – 78.15 | Apoptosis through Bcl-2 inhibition | [90,91] |
| Myrtaceae | Syzygium cumini | Seeds | MTS cell proliferation assay | A2780, MCF-7, PC3 and H460 | 49, 110, 140 and 165, respectively | Pelargonidin-3-O- glucoside, cyanidin-3-O-malonyl glucoside, delphenidin-3-O- glucoside, ellagitannins etc. | - | - | [92,93,94] |
| Nyctaginaceae | Mirabilis jalapa | Aerial Parts, Roots, Stem | MTT assay | HeLa, Raji, A549, HCT 116, Vero MCF-7 and Hep-2 | - | Rotenoids and mirabilis antiviral protein (MAP) | For MAP, 150, 175, 200 against HCT116, MCF-7 and A549, respectively | - | [22,95,96] |
| Oleaceae | Jasminum sambac | Flowers, Leaves | MTT assay | MCF-7 and HEP-2 | 7 for MCF-7 | - | - | - | [22,24] |
| Olea europaea | Leaves | MTT assay | MCF-7, B16F10, HeLa and Vero | 43, 170, 440 and >1000, respectively | Interaction with Bcl-2, Bax, and p53 proteins | [23,62,97] | |||
| Syringa vulgaris | Fruits | MTT assay | MCF-7 and HeLa | - | - | - | - | [22] | |
| Oxalidaceae | Averrhoa bilimbi | Fruits, Leaves | MTT assay | MCF-7 | 154.9 and 668 for fruits and leaves | Nonanal, tricosane, squalene, malonic acid, etc. | - | - | [98,99] |
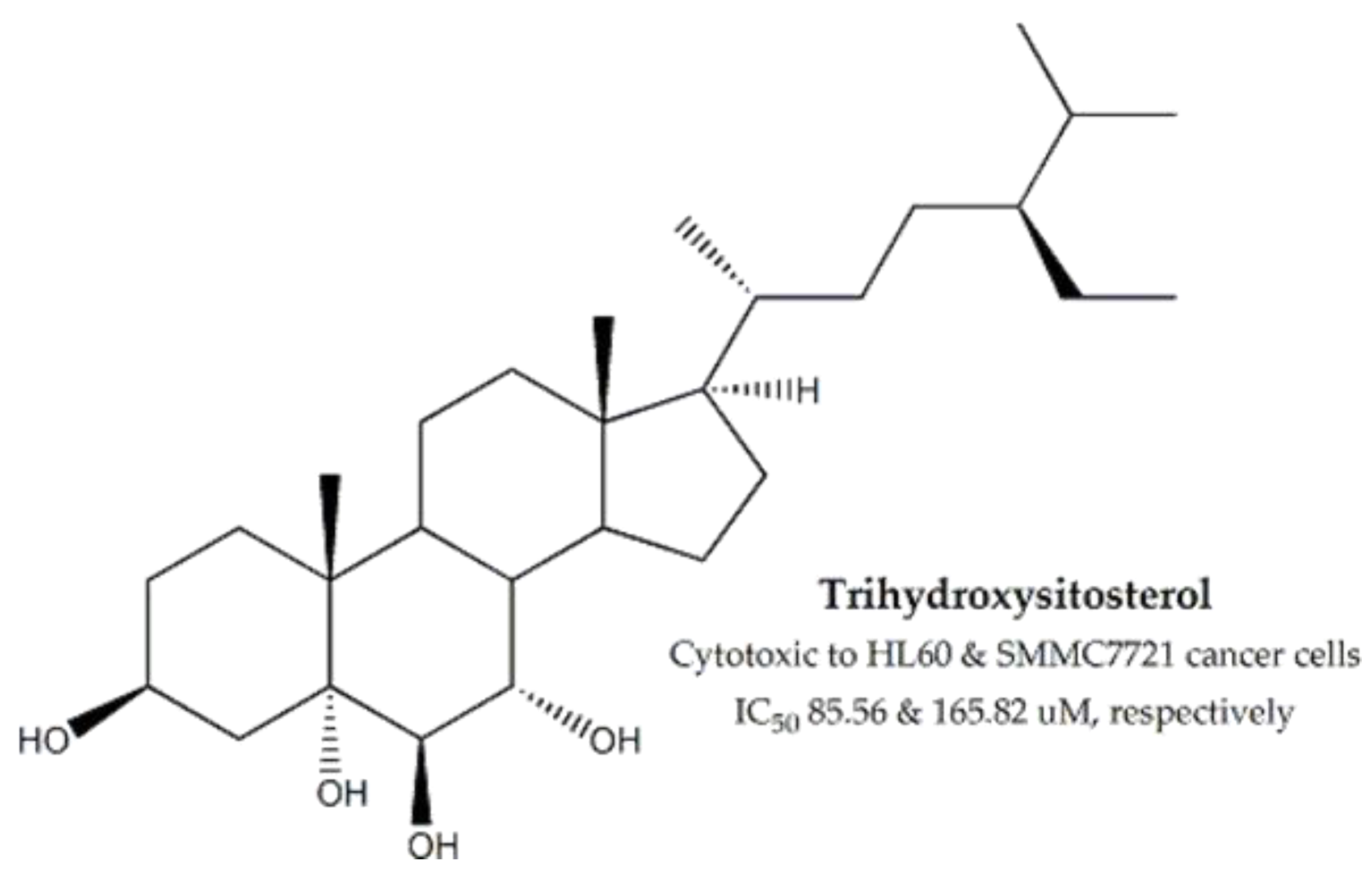
| Phyllanthacae | Phyllanthus emblica | Fruits, Leaves | MTT assay | HT-29, HL-60 and SMMC-7721 | 35 for HT-29 | Trihydroxy- sitosterol | 85.56 and 165.82 for HL-60 and SMMC-7721, respectively | - | [100,101,102] |
| Plumbaginaceae | Limonium densiflorum | Shoots | Resazurin reduction assays | A-549 and DLD-1 | 29 and 85, respectively | Myricetin, isorhamnetin and trans- 3-hydroxy- cinnamic acid | - | - | [103] |
| Picrorhiza | Picrorhiza kurroa | Rhizomes | XTT and SRB assays | Hep3B, MDA-MB-43 and PC-3 | 32.6, 19 and 63.9, respectively | Curcubitacin | - | Apoptosis | [104,105] |
| Pinaceae | Cedrus deodara | Woods | SRB assay | MIA-PA-CA-2, PC3, A-2780 and Molt-4 | 74, 77, 63 and 15, respectively | - | - | Interaction with caspase 3,8 and 9 proteins | [48,106] |
| Piperaceae | Piper longum | Fruits | In vitro SRB assay, in vivo DLA and EAC induced ascites tumor model in mice | Colo-205, DLA and EAC | 18 for Colo-205 | Piperine | - | Apoptosis | [48,107] |
| Piper regnellii | Leaves | MTT assay | UACC-62, 786–0, MCF-7, NCI-H460, OVCAR-3, PC-3, HT-29 and K-562 | - | Eupomatenoid-5 | - | - | [108] | |
| Poaceae | Cenchrus ciliaris | Aerial Parts, Roots | MTT assay | A-549, CACO, HCT-116, HeLa, HepG2, MCF-7 and PC3 | Ranging between 9 to 27.2 | - | - | - | [109,110] |
| Polygonaceae | Calligonum comosum | Whole Plant | MTT assay | HepG2 | 9.6 | Catechin, kaempferol, mequilianin, etc. | - | Increased p53 and reduced Bcl-2 gene expression | [35,111] |
| Primulaceae | Aegiceras corniculatum | Fruits | MTT assay | VERO, NIH3T3, AGS HT-29, MCF-7 and MDA-MB-231 | 150, 97, 0.5, 998, 91 and 461, respectively | Aegicoroside A, sakurasosaponin, fusarine, fusamine etc. | - | - | [24,112,113] |
| Ranunculaceae | Delphinium staphisagria | Seeds | MTT assay | MCF7, HT29, N2A, H5-6 and VCREMS | 41.9, 41.1 and 14.5 for N2A, H5-6 and VCREMS, respectively | Astragalin, paeonoside, petiolaroside, etc. | - | - | [34,114] |
| Rosaceae | Crataegus microphylla | Leaves, Flowers | MTT assay | HeLa and Vero | 576 for HeLa | - | - | - | [23,115] |
| Rosa damascena | Flowers, Seeds | MTT assay | MCF-7, Hep-2, HeLa and Vero | 265 for HeLa | Nerol, geraniol, β-citronellol, linalool, nonadecane and phenylethyl alcohol | - | - | [22,23,116] | |
| Rubiaceae | Hymenodictyon excelsum | Barks, Woods | MTT assay | VERO, NIH3T3, AGS HT-29, MCF-7 and MDA-MB-231 | 230, 70, 90, 160, 80 and 440, respectively | - | - | DNA fragmentation and apoptosis | [24,117] |
| Oldenlandia corymbosa | Leaves | MTT assay | K562 | 114.4 | - | - | Apoptosis | [118] | |
| Salicaceae | Populus alba | Flowers | MTT assay | Hep-2, A549, H1299 and MCF-7 | 12.05, 10.53 and 28.16 for A549, H1299 and MCF-7 | - | - | - | [22,119] |
| Sapotaceae | Manilkara zapota | Flowers | MTT assay | MCF-7, HT-29 and HL-60 | 12.5 | Lupanes, oleananes, ursanes, manilkoraside, etc. | For manilkoraside, 64 and 24 against HT-29 and HL-60, respectively | DNA fragmentation | [28,120] |
| Saururaceae | Saururus chinensis | Roots | MTT assay | MCF-7, HT-29 and HepG2 | 91.2 | Aristolactram, dihydroguaiatric acid, sauchinone, saucerneol D, manassantin A and B, saucerneol F etc. | Ranging between 10-16 | Inhibition of DNA topoisomerase I and II | [121,122] |
| Scrophulariaceae | Verbascum sinaiticum | Aerial Parts, Flowers | MTT assay | MCF-7 and Hep-2 | Sinaiticin, hydrocarpin and flavonolignans | For hydrocarpin and sinaiticin, 1.2 and 7.7 against P-388 | - | [22,123] | |
| Solanaceae | Solanum khasianum | Fruits | MTT and Clonogenic assays | DL, MCF-7 and HeLa | 27.4, 71.2 and 62.5, respectively | - | - | - | [46] |
| Solanum nigrum | Fruits | MTT and Clonogenic assays | HeLa, Vero, HepG2 and CT26 | 265, 6.9, 56.4 and 77.6, respectively | - | - | - | [124,125] | |
| Withania coagulans | Fruits, Roots and Leaves | Presto Blue cell viability assay | HeLa, MCF-7 and RD | Ranging between 0.7 to 6.7 | Myricetin, quercetin, gallic acid, etc. | - | - | [126] | |
| Withania somnifera | Roots | SRB assay | PC3, A-549, A-2780 and K-562 | 52, 46, 79 and 41, respectively | - | - | - | [48] | |
| Sterculiaceae | Helicteres isora | Whole Plant | MTT assay | HeLa-B75, HL-60, HEP-3B and PN-15 | - | Cucurbitacin B and isocucurbitacin B | - | - | [127,128] |
| Thymelaeaceae | Aquilaria malaccensis | Leaves | In vitro TBE and MTT assay, in vivo EAC induced ascites model | HCT116, DLA and EAC | 4, 72 and 79, respectively | Benzaldehyde, pinene, octanol, germacrene, hexadecanal, etc. | - | - | [129,130,131] |
| Verbenaceae | Clerodendrum viscosum | Leaves | MTT assay | VERO, NIH3T3, AGS, HT-29, MCF-7 and MDA-MB-231 | 50 and 880 for MCF-7 and HT-29 | - | - | Apoptosis | [24] |
| Clerodendron infortunatum | Roots | DLA induced ascites tumor model in mice | DLA | - | - | - | Apoptosis through interaction with Bax, Bcl-2, caspases 8 and 10 proteins | [132] | |
| Vitaceae | Leea indica | Leaves | MTT assay | DU-145 and PC-3 | 529.4 and 677.1, respectively | - | - | - | [133] |
| Vitis vinifera | Stem | “Alamar Blue” resazurin reduction assays | MCF-7, B16F10 and HeLa | 62, 137 and 336, respectively | Quercetin 3-O-β-D-4C1 galactoside and quercetin 3-O-β-D-4C1 glucuronide | - | Apoptosis | [62,134,135] | |
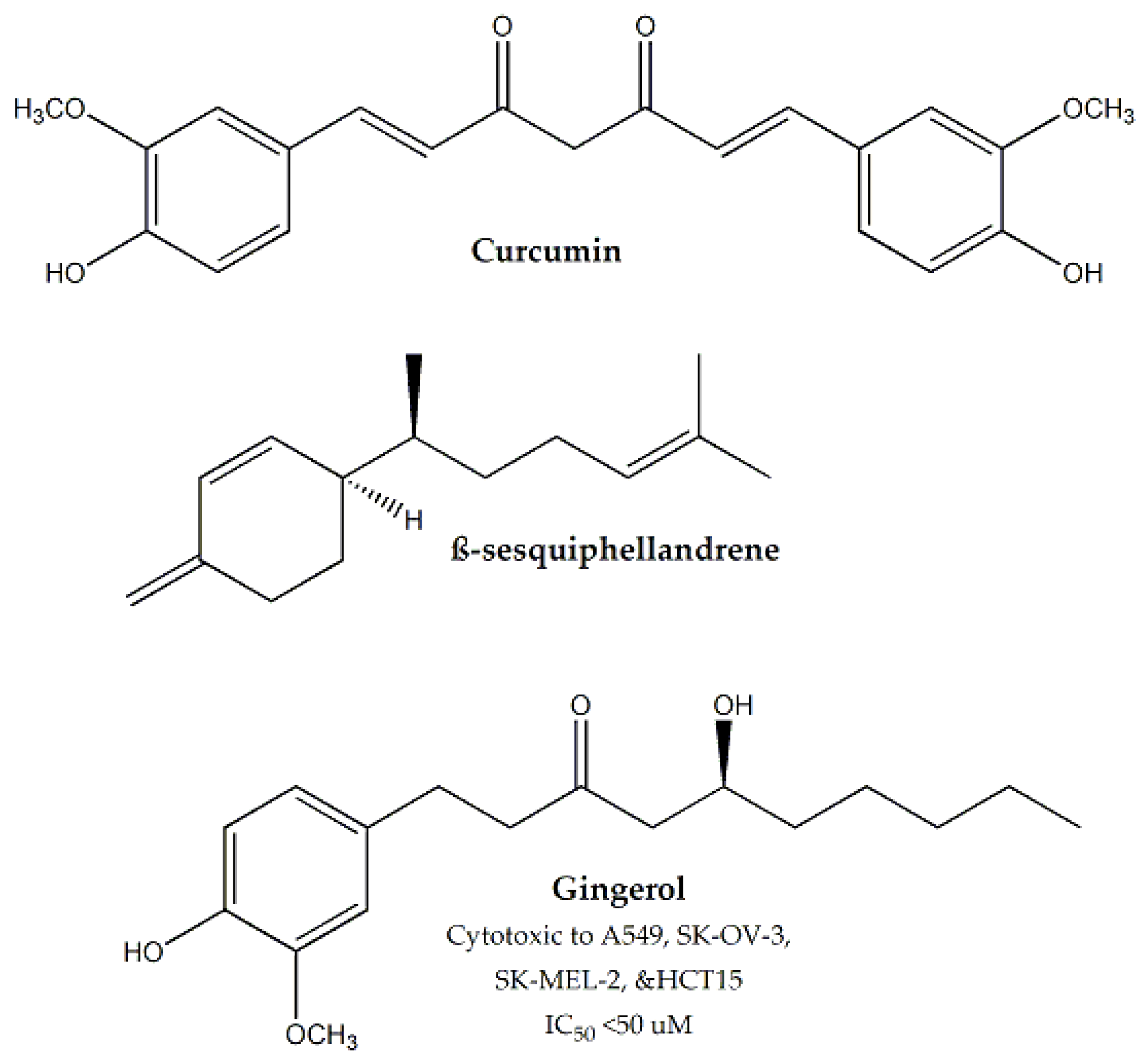
| Zingiberaceae | Curcuma longa | Rhizomes | MTT assay | Hep-2 | - | Curcumin, β-sesquiphellandrene | - | Apoptosis | [136,137,138,139,140] |
| Zingiber officinale | Rhizomes | In vitro MTT assay, in vivo DLA and EAC induced ascites model | DLA, EAC, A549, SK-OV-3, SK-MEL-2, PC-3M and HCT15 | - | Gingerol | < 50 | - | [140,141] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mazumder, K.; Biswas, B.; Raja, I.M.; Fukase, K. A Review of Cytotoxic Plants of the Indian Subcontinent and a Broad-Spectrum Analysis of Their Bioactive Compounds. Molecules 2020, 25, 1904. https://doi.org/10.3390/molecules25081904
Mazumder K, Biswas B, Raja IM, Fukase K. A Review of Cytotoxic Plants of the Indian Subcontinent and a Broad-Spectrum Analysis of Their Bioactive Compounds. Molecules. 2020; 25(8):1904. https://doi.org/10.3390/molecules25081904
Chicago/Turabian StyleMazumder, Kishor, Biswajit Biswas, Iqbal Mahmud Raja, and Koichi Fukase. 2020. "A Review of Cytotoxic Plants of the Indian Subcontinent and a Broad-Spectrum Analysis of Their Bioactive Compounds" Molecules 25, no. 8: 1904. https://doi.org/10.3390/molecules25081904
APA StyleMazumder, K., Biswas, B., Raja, I. M., & Fukase, K. (2020). A Review of Cytotoxic Plants of the Indian Subcontinent and a Broad-Spectrum Analysis of Their Bioactive Compounds. Molecules, 25(8), 1904. https://doi.org/10.3390/molecules25081904






