Butein Promotes Lineage Commitment of Bone Marrow-Derived Stem Cells into Osteoblasts via Modulating ERK1/2 Signaling Pathways
Abstract
1. Introduction
2. Results
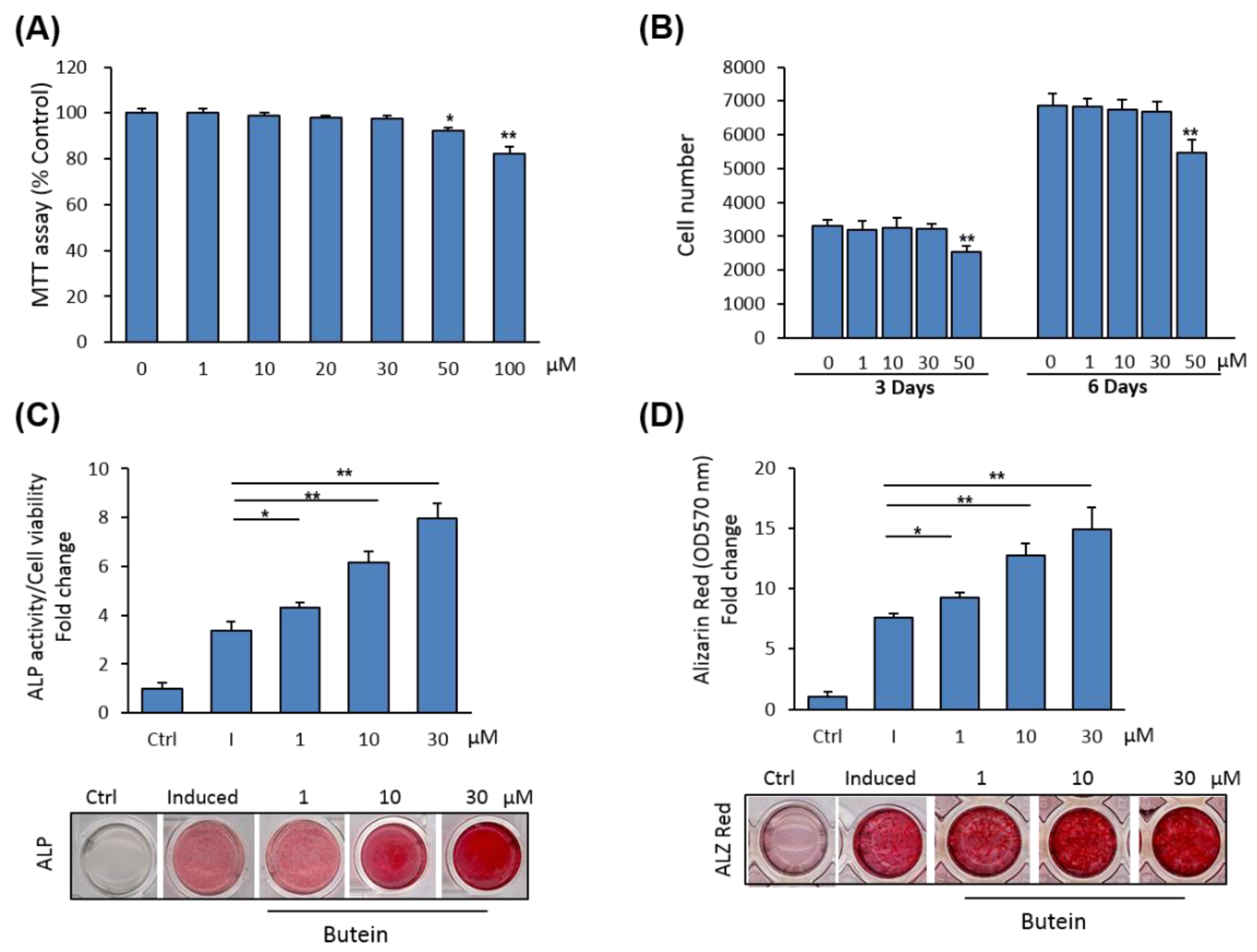
2.1. Butein Induces Osteoblast Differentiation of mBMSCs
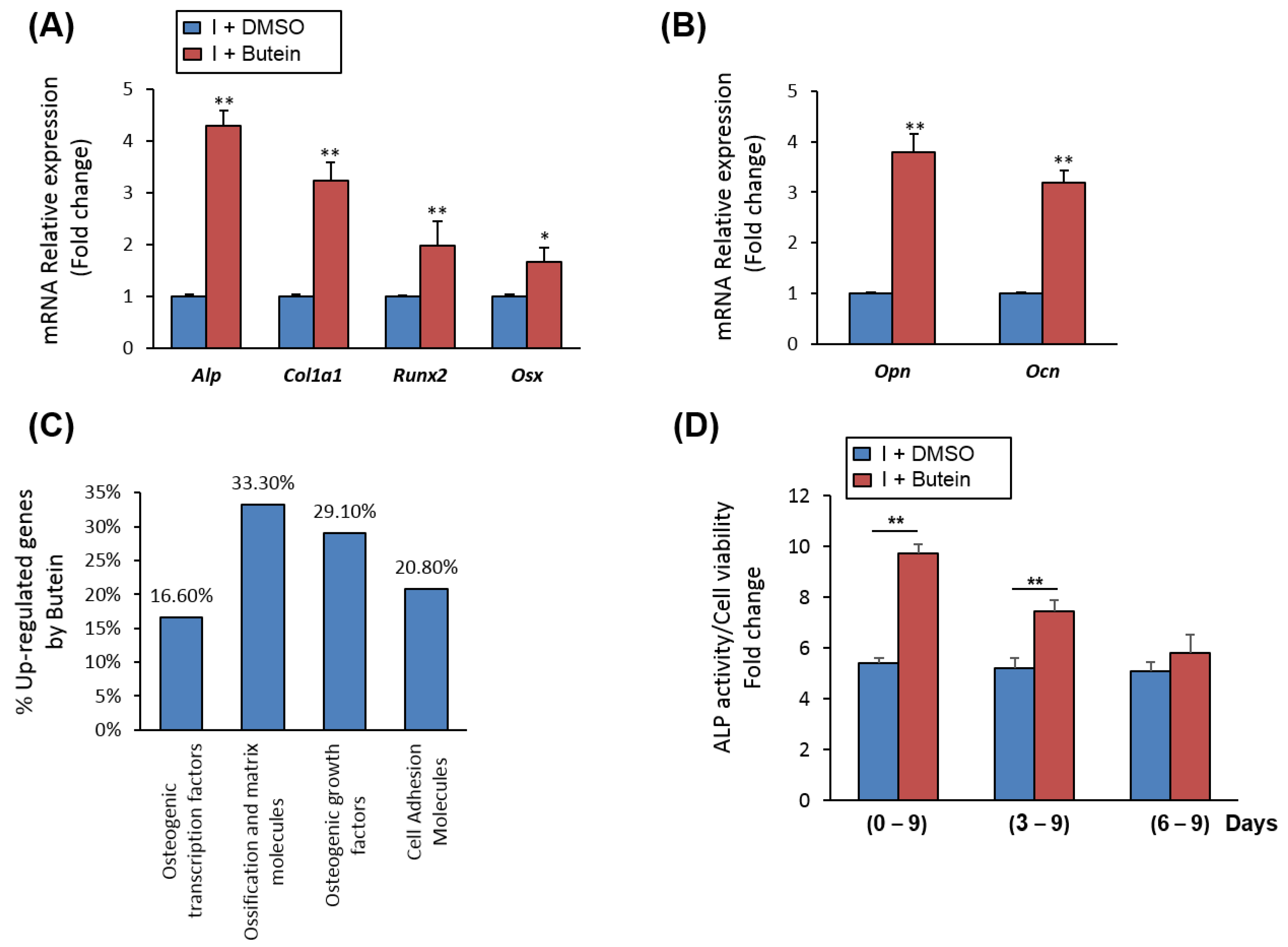
2.2. Butein Upregulates the mRNA Expression of Osteoblast-Related Genes in mBMSCs
2.3. Butein Promotes Osteogenesis of mBMSCs at Early Commitment Stage
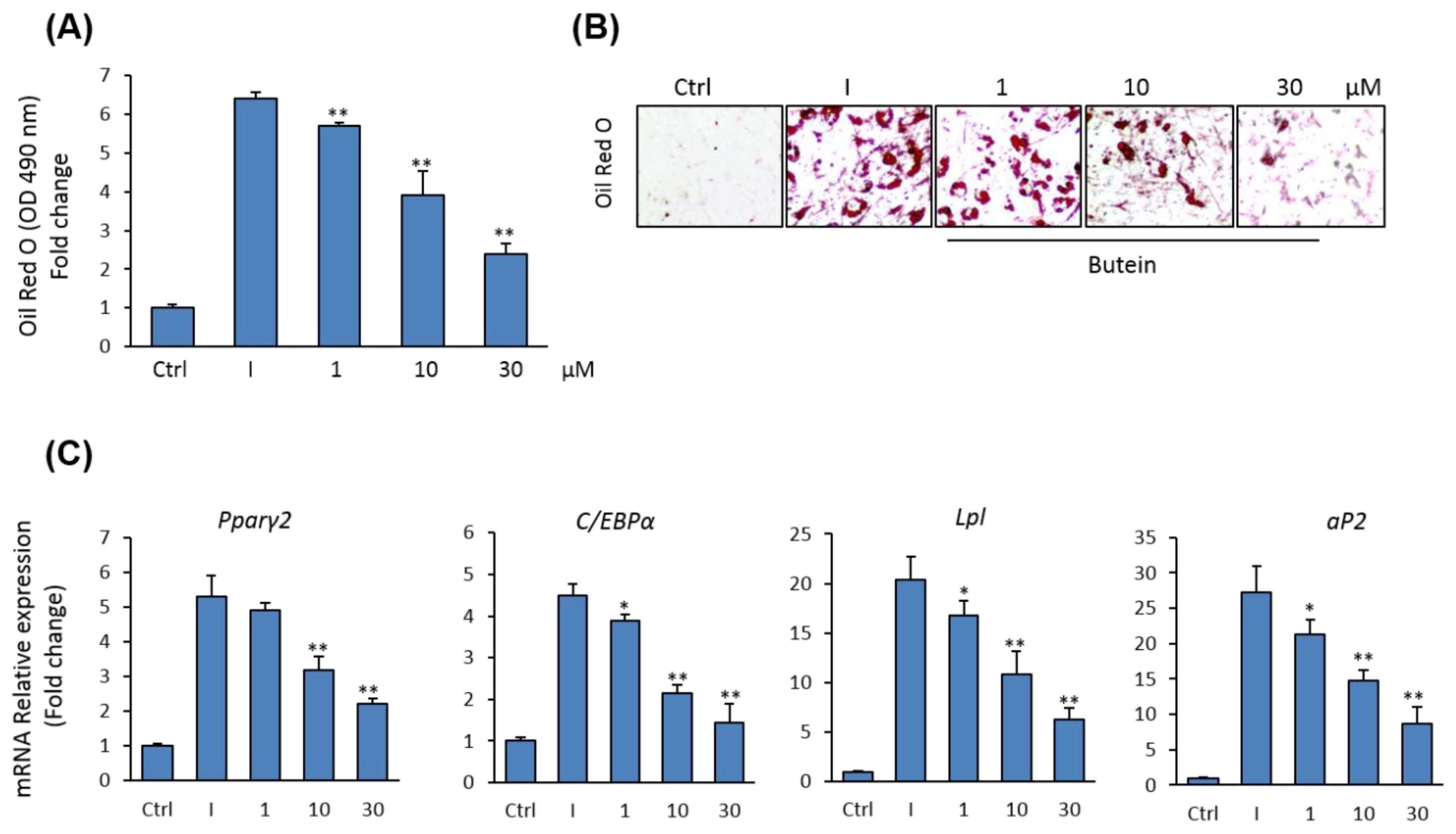
2.4. Butein Suppresses the Differentiation of mBMSCs into Adipocytes
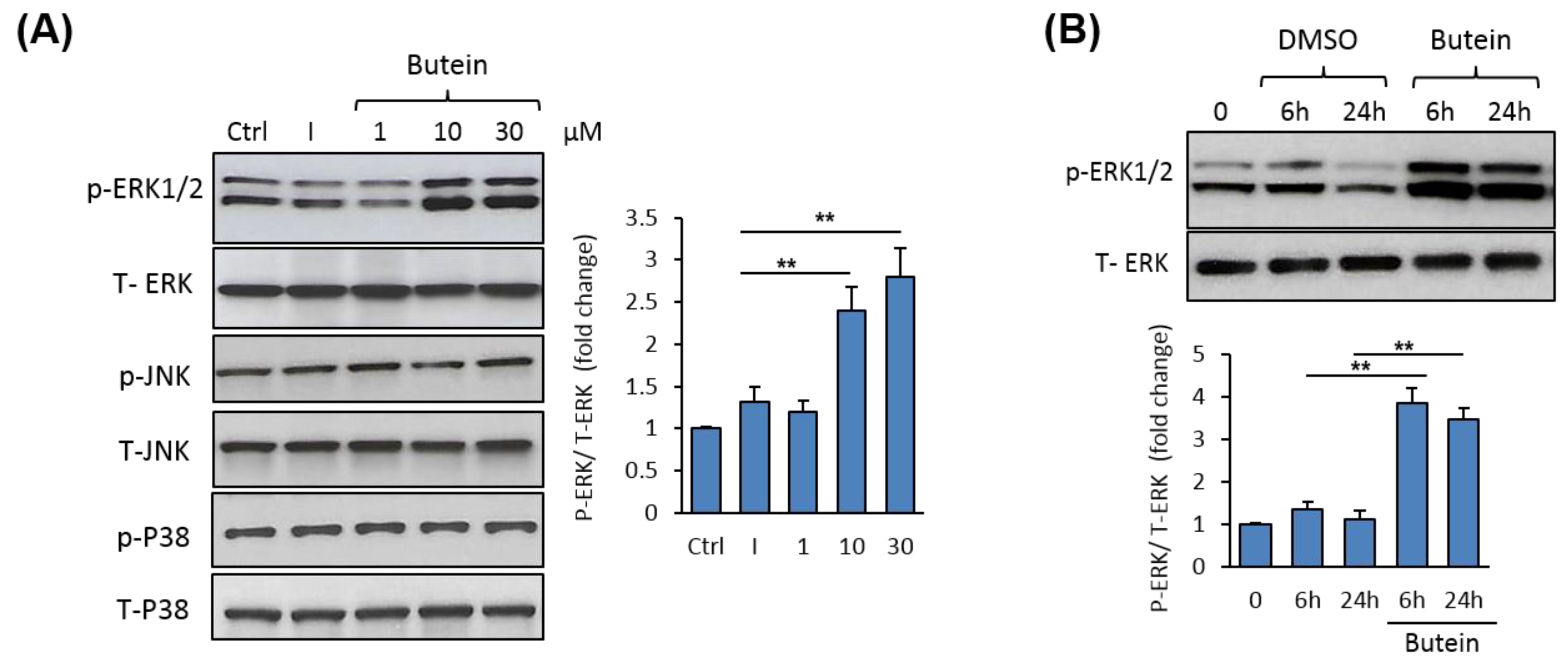
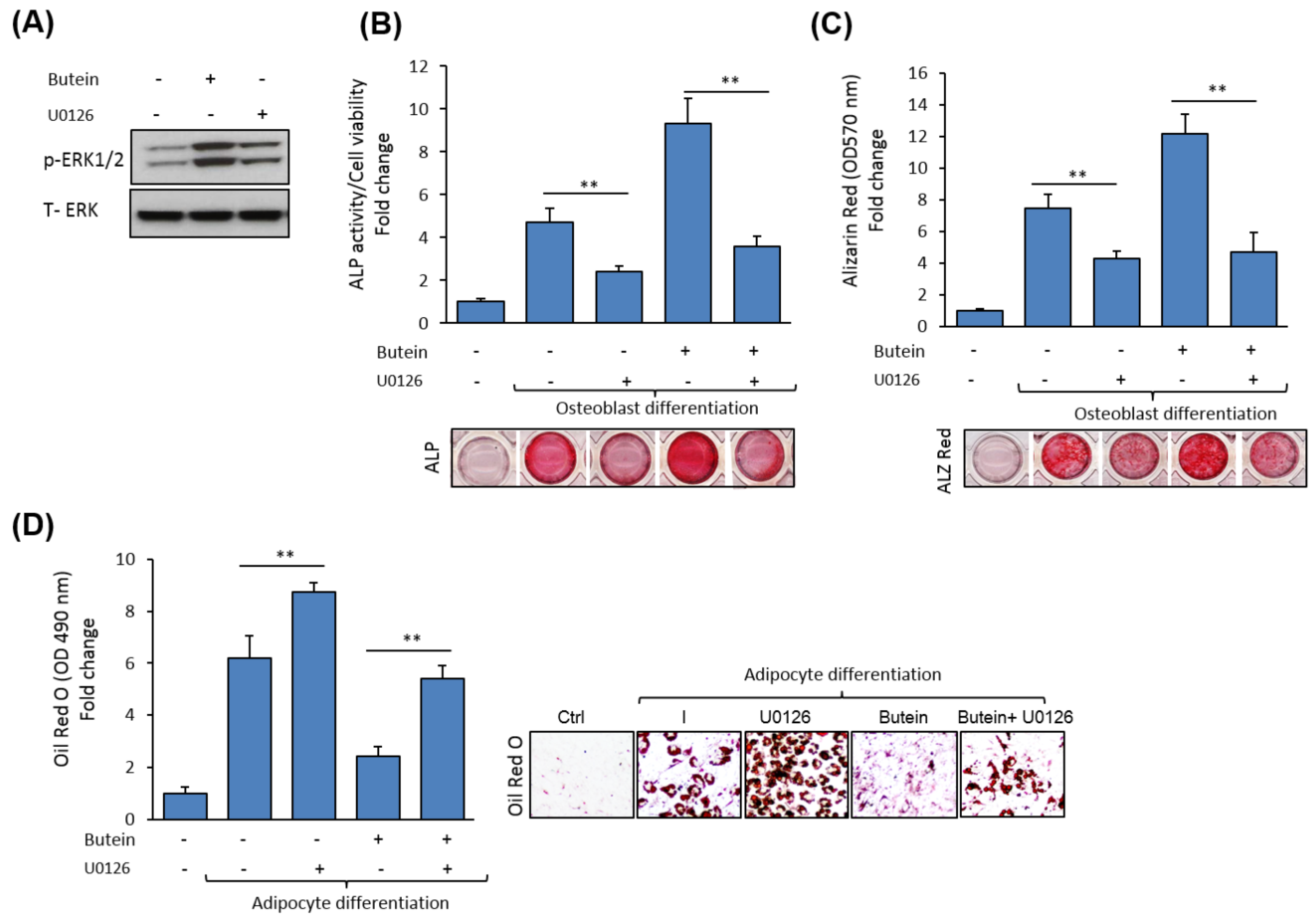
2.5. Butein Activates ERK1/2 Signaling Pathway during Osteogenesis.
2.6. Butein Stimulates mBMSCs Differentiation into Osteoblast Versus Adipocyte in ERK1/2-Dependent Pathway
2.7. Butein Induces the Differentiation of Human (h) BMSCs into Osteoblasts
3. Discussion
4. Materials and Methods
4.1. Cell Cultures and Reagents
4.2. Cell Toxicity Assay
4.3. Cell Proliferation Study
4.4. Osteoblast Differentiation
4.5. Adipocyte Differentiation
4.6. Alkaline Phosphatase (ALP) Activity Assay
4.7. Cytochemical Staining
4.7.1. Alkaline Phosphatase Staining
4.7.2. Alizarin Red S Staining and Quantification
4.7.3. Oil Red O Staining and Quantification
4.8. Western Blot Analysis
4.9. RNA Extraction and Real-Time PCR Analysis
4.10. PCR Array Analysis
4.11. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Goltzman, D. The Aging Skeleton. Adv. Exp. Med. Biol. 2019, 1164, 153–160. [Google Scholar] [PubMed]
- Paspaliaris, V.; Kolios, G. Stem cells in Osteoporosis: From Biology to New Therapeutic Approaches. Stem Cells Int. 2019, 2019, 1730978. [Google Scholar] [CrossRef] [PubMed]
- Kenkre, J.S.; Bassett, J. The bone remodelling cycle. Ann. Clin. Biochem. 2018, 55, 308–327. [Google Scholar] [CrossRef] [PubMed]
- Kassem, M.; Abdallah, B.M.; Saeed, H. Osteoblastic cells: Differentiation and trans-differentiation. ArchBiochemBiophys 2008, 473, 183–187. [Google Scholar] [CrossRef] [PubMed]
- Amarasekara, D.S.; Yun, H.; Kim, S.; Lee, N.; Kim, H.; Rho, J. Regulation of Osteoclast Differentiation by Cytokine Networks. Immune Netw. 2018, 18, 8. [Google Scholar] [CrossRef]
- Cummings, S.R.; Martin, J.S.; McClung, M.R.; Siris, E.S.; Eastell, R.; Reid, I.R.; Delmas, P.; Zoog, H.B.; Austin, M.; Wang, A.; et al. Denosumab for Prevention of Fractures in Postmenopausal Women with Osteoporosis. New Engl. J. Med. 2009, 361, 756–765. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Xiao, Z.; Quarles, L.D.; Li, W. Osteoporosis: Mechanism, Molecular Target, and Current Status on Drug Development. Curr. Med. Chem. 2020. [Google Scholar] [CrossRef] [PubMed]
- Valverde, P. Pharmacotherapies to manage bone loss-associated diseases: A quest for the perfect benefit-to-risk ratio. CurrMedChem 2008, 15, 284–304. [Google Scholar] [CrossRef]
- Weinerman, S.; Usera, G.L. Antiresorptive Therapies for Osteoporosis. Oral Maxillofac. Surg. Clin. N. Am. 2015, 27, 555–560. [Google Scholar] [CrossRef]
- Weinerman, S.; Usera, G.L. Indications and Outcomes of Osteoporosis and Bone Modulation Therapies. Oral Maxillofac. Surg. Clin. N. Am. 2015, 27, 567–571. [Google Scholar] [CrossRef]
- Bhattacharyya, S.; Pal, S.; Chattopadhyay, N. Targeted inhibition of sclerostin for post-menopausal osteoporosis therapy: A critical assessment of the mechanism of action. Eur. J. Pharmacol. 2018, 826, 39–47. [Google Scholar] [CrossRef] [PubMed]
- Komori, T. Regulation of osteoblast differentiation by Runx2. Adv. Exp. Med. Biol. 2010, 658, 43–49. [Google Scholar] [PubMed]
- Abdallah, B.M.; Jafari, A.; Zaher, W.; Qiu, W.; Kassem, M. Skeletal (stromal) stem cells: An update on intracellular signaling pathways controlling osteoblast differentiation. Bone 2015, 70, 28–36. [Google Scholar] [CrossRef] [PubMed]
- Tang, C.H.; Yang, R.S.; Chien, M.Y.; Chen, C.C.; Fu, W.M. Enhancement of bone morphogenetic protein-2 expression and bone formation by coumarin derivatives via p38 and ERK-dependent pathway in osteoblasts. Eur. J. Pharmacol. 2008, 579, 40–49. [Google Scholar] [CrossRef]
- Shah, M.; Kola, B.; Bataveljic, A.; Arnett, T.R.; Viollet, B.; Saxon, L.; Korbonits, M.; Chenu, C. AMP-activated protein kinase (AMPK) activation regulates in vitro bone formation and bone mass. Bone 2010, 47, 309–319. [Google Scholar] [CrossRef]
- Cargnello, M.; Roux, P.P. Activation and function of the MAPKs and their substrates, the MAPK-activated protein kinases. Microbiol. Mol. Biol. Rev. 2011, 75, 50–83. [Google Scholar] [CrossRef]
- Franceschi, R.T.; Ge, C.; Xiao, G.; Roca, H.; Jiang, D. Transcriptional regulation of osteoblasts. Ann. N. Y. Acad. Sci. 2007, 1116, 196–207. [Google Scholar] [CrossRef]
- Kanno, T.; Takahashi, T.; Tsujisawa, T.; Ariyoshi, W.; Nishihara, T. Mechanical stress-mediated Runx2 activation is dependent on Ras/ERK1/2 MAPK signaling in osteoblasts. J. Cell. Biochem. 2007, 101, 1266–1277. [Google Scholar] [CrossRef]
- Greenblatt, M.B.; Shim, J.H.; Glimcher, L.H. Mitogen-activated protein kinase pathways in osteoblasts. Annu. Rev. Cell Dev. Biol. 2013, 29, 63–79. [Google Scholar] [CrossRef]
- Zhang, W.; Shen, X.; Wan, C.; Zhao, Q.; Zhang, L.; Zhou, Q.; Deng, L. Effects of insulin and insulin-like growth factor 1 on osteoblast proliferation and differentiation: Differential signalling via Akt and ERK. Cell Biochem. Funct. 2012, 30, 297–302. [Google Scholar] [CrossRef]
- Kim, S.J.; Kim, S.Y.; Kwon, C.H.; Kim, Y.K. Differential effect of FGF and PDGF on cell proliferation and migration in osteoblastic cells. Growth Factors 2007, 25, 77–86. [Google Scholar] [CrossRef] [PubMed]
- Chan, S.C.; Chang, Y.S.; Wang, J.P.; Chen, S.C.; Kuo, S.C. Three new flavonoids and antiallergic, anti-inflammatory constituents from the heartwood of Dalbergia odorifera. Planta Med. 1998, 64, 153–158. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.H.; Kim, M.; Chang, K.H.; Hong, C.Y.; Na, C.S.; Dong, M.S.; Lee, D.; Lee, M.Y. Antiplatelet effects of Rhus verniciflua Stokes heartwood and its active constituents-fisetin, butein, and sulfuretin-in rats. J. Med. Food 2015, 18, 21–30. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.D.; Choe, J.W.; Lee, B.J.; Kang, M.H.; Joo, M.K.; Kim, J.H.; Yeon, J.E.; Park, J.J.; Kim, J.S.; Bak, Y.T. Butein effects in colitis and interleukin-6/signal transducer and activator of transcription 3 expression. World J. Gastroenterol. 2015, 21, 465–474. [Google Scholar] [CrossRef]
- Lee, S.H.; Seo, G.S.; Sohn, D.H. Inhibition of lipopolysaccharide-induced expression of inducible nitric oxide synthase by butein in RAW 264.7 cells. Biochem. Biophys. Res. Commun. 2004, 323, 125–132. [Google Scholar] [CrossRef]
- Cioce, M.; Canino, C.; Pulito, C.; Muti, P.; Strano, S.; Blandino, G. Butein impairs the protumorigenic activity of malignant pleural mesothelioma cells. Cell Cycle 2012, 11, 132–140. [Google Scholar] [CrossRef]
- Sung, J.; Lee, J. Anti-Inflammatory Activity of Butein and Luteolin through Suppression of NFkappaB Activation and Induction of Heme Oxygenase-1. J. Med. Food 2015, 18, 557–564. [Google Scholar] [CrossRef]
- Lai, Y.-W.; Wang, S.-W.; Chang, C.-H.; Liu, S.-C.; Chen, Y.-J.; Chi, C.-W.; Chiu, L.-P.; Chen, S.-S.; Chiu, A.W.; Chung, C.-H. Butein inhibits metastatic behavior in mouse melanoma cells through VEGF expression and translation-dependent signaling pathway regulation. BMC Complement. Altern. Med. 2015, 15, 445. [Google Scholar] [CrossRef]
- Samoszuk, M.; Tan, J.; Chorn, G. The chalcone butein from Rhus verniciflua Stokes inhibits clonogenic growth of human breast cancer cells co-cultured with fibroblasts. BMC Complement. Altern. Med. 2005, 5, 5. [Google Scholar] [CrossRef]
- Song, N.J.; Yoon, H.J.; Kim, K.H.; Jung, S.R.; Jang, W.S.; Seo, C.R.; Lee, Y.M.; Kweon, D.H.; Hong, J.W.; Lee, J.S.; et al. Butein is a novel anti-adipogenic compound. J. Lipid Res. 2013, 54, 1385–1396. [Google Scholar] [CrossRef]
- Song, Z.; Shanmugam, M.K.; Yu, H.; Sethi, G. Butein and Its Role in Chronic Diseases. Adv. Exp. Med. Biol. 2016, 928, 419–433. [Google Scholar] [PubMed]
- Jeong, G.S.; Lee, D.S.; Song, M.Y.; Park, B.H.; Kang, D.G.; Lee, H.S.; Kwon, K.B.; Kim, Y.C. Butein from Rhus verniciflua protects pancreatic beta cells against cytokine-induced toxicity mediated by inhibition of nitric oxide formation. Biol. Pharm. Bull. 2011, 34, 97–102. [Google Scholar] [CrossRef] [PubMed]
- Padmavathi, G.; Roy, N.K.; Bordoloi, D.; Arfuso, F.; Mishra, S.; Sethi, G.; Bishayee, A.; Kunnumakkara, A.B. Butein in health and disease: A comprehensive review. Phytomed. Int. J. Phytother. Phytopharm. 2017, 25, 118–127. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.H.; Ho, Y.J.; Lin, J.F.; Yeh, C.W.; Kao, S.H.; Hsu, L.S. Butein inhibits the proliferation of breast cancer cells through generation of reactive oxygen species and modulation of ERK and p38 activities. Mol. Med. Rep. 2012, 6, 1126–1132. [Google Scholar] [CrossRef]
- Padmavathi, G.; Rathnakaram, S.R.; Monisha, J.; Bordoloi, D.; Roy, N.K.; Kunnumakkara, A.B. Potential of butein, a tetrahydroxychalcone to obliterate cancer. Phytomed. Int. J. Phytother. Phytopharm. 2015, 22, 1163–1171. [Google Scholar] [CrossRef]
- Ma, C.Y.; Ji, W.T.; Chueh, F.S.; Yang, J.S.; Chen, P.Y.; Yu, C.C.; Chung, J.G. Butein inhibits the migration and invasion of SK-HEP-1 human hepatocarcinoma cells through suppressing the ERK, JNK, p38, and uPA signaling multiple pathways. J. Agric. Food Chem. 2011, 59, 9032–9038. [Google Scholar] [CrossRef]
- Sung, B.; Cho, S.G.; Liu, M.; Aggarwal, B.B. Butein, a tetrahydroxychalcone, suppresses cancer-induced osteoclastogenesis through inhibition of receptor activator of nuclear factor-kappaB ligand signaling. Int. J. Cancer 2011, 129, 2062–2072. [Google Scholar] [CrossRef]
- Zheng, W.; Zhang, H.; Jin, Y.; Wang, Q.; Chen, L.; Feng, Z.; Chen, H.; Wu, Y. Butein inhibits IL-1beta-induced inflammatory response in human osteoarthritis chondrocytes and slows the progression of osteoarthritis in mice. Int. Immunopharmacol. 2017, 42, 1–10. [Google Scholar] [CrossRef]
- Abdallah, B.M.; Kassem, M. New factors controlling the balance between osteoblastogenesis and adipogenesis. Bone 2012, 50, 540–545. [Google Scholar] [CrossRef]
- Jayasooriya, R.; Molagoda, I.M.N.; Park, C.; Jeong, J.W.; Choi, Y.H.; Moon, D.O.; Kim, M.O.; Kim, G.Y. Molecular chemotherapeutic potential of butein: A concise review. Food Chem. Toxicol. Int. J. Publ. Br. Ind. Biol. Res. Assoc. 2018, 112, 1–10. [Google Scholar] [CrossRef]
- Moon, D.O.; Kim, M.O.; Choi, Y.H.; Hyun, J.W.; Chang, W.Y.; Kim, G.Y. Butein induces G(2)/M phase arrest and apoptosis in human hepatoma cancer cells through ROS generation. Cancer Lett. 2010, 288, 204–213. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.H.; Yeh, C.W.; Lo, H.C.; Su, S.L.; Hseu, Y.C.; Hsu, L.S. Generation of reactive oxygen species mediates butein-induced apoptosis in neuroblastoma cells. Oncol. Rep. 2012, 27, 1233–1237. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Lee, Y.; Eun, J.S.; Bae, E.J. Inhibition of adipocyte inflammation and macrophage chemotaxis by butein. Eur. J. Pharmacol. 2014, 738, 40–48. [Google Scholar] [CrossRef] [PubMed]
- Benzler, J.; Ganjam, G.K.; Pretz, D.; Oelkrug, R.; Koch, C.E.; Legler, K.; Stohr, S.; Culmsee, C.; Williams, L.M.; Tups, A. Central inhibition of IKKbeta/NF-kappaB signaling attenuates high-fat diet-induced obesity and glucose intolerance. Diabetes 2015, 64, 2015–2027. [Google Scholar] [CrossRef]
- Lim, S.S.; Jung, S.H.; Ji, J.; Shin, K.H.; Keum, S.R. Synthesis of flavonoids and their effects on aldose reductase and sorbitol accumulation in streptozotocin-induced diabetic rat tissues. J. Pharm. Pharmacol. 2001, 53, 653–668. [Google Scholar] [CrossRef]
- Lee, E.H.; Song, D.G.; Lee, J.Y.; Pan, C.H.; Um, B.H.; Jung, S.H. Inhibitory effect of the compounds isolated from Rhus verniciflua on aldose reductase and advanced glycation endproducts. Biol. Pharm. Bull. 2008, 31, 1626–1630. [Google Scholar] [CrossRef]
- Lee, S.H.; Nan, J.X.; Zhao, Y.Z.; Woo, S.W.; Park, E.J.; Kang, T.H.; Seo, G.S.; Kim, Y.C.; Sohn, D.H. The chalcone butein from Rhus verniciflua shows antifibrogenic activity. Planta Med. 2003, 69, 990–994. [Google Scholar]
- Siersbaek, M.S.; Jafari, A.; Zaher, W.; Chen, L.; Kassem, M. Identification of a small molecule kinase inhibitor that enhances osteoblastic differentiation and in vivo bone formation of human skeletal (mesenchymal) stem cells through regulation of BMP signaling. Bone 2011, 48, 104–123. [Google Scholar]
- Abdallah, B.M.; Jensen, C.H.; Gutierrez, G.; Leslie, R.G.; Jensen, T.G.; Kassem, M. Regulation of human skeletal stem cells differentiation by Dlk1/Pref-1. J. Bone Miner. Res. 2004, 19, 841–852. [Google Scholar] [CrossRef]
- Stechschulte, L.A.; Lecka-Czernik, B. Reciprocal regulation of PPARgamma and RUNX2 activities in marrow mesenchymal stem cells: Fine balance between p38 MAPK and Protein Phosphatase 5. Curr. Mol. Biol. Rep. 2017, 3, 107–113. [Google Scholar] [CrossRef]
- Wanachewin, O.; Boonmaleerat, K.; Pothacharoen, P.; Reutrakul, V.; Kongtawelert, P. Sesamin stimulates osteoblast differentiation through p38 and ERK1/2 MAPK signaling pathways. BMC Complement. Altern. Med. 2012, 12, 71. [Google Scholar] [CrossRef] [PubMed]
- Ge, C.; Cawthorn, W.P.; Li, Y.; Zhao, G.; Macdougald, O.A.; Franceschi, R.T. Reciprocal Control of Osteogenic and Adipogenic Differentiation by ERK/MAP Kinase Phosphorylation of Runx2 and PPARgamma Transcription Factors. J. Cell. Physiol. 2016, 231, 587–596. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.A.; Kim, J.H.; Wang, Y.; Sul, H.S. Pref-1 (preadipocyte factor 1) activates the MEK/extracellular signal-regulated kinase pathway to inhibit adipocyte differentiation. MolCell Biol 2007, 27, 2294–2308. [Google Scholar] [CrossRef] [PubMed]
- Khatiwala, C.B.; Kim, P.D.; Peyton, S.R.; Putnam, A.J. ECM compliance regulates osteogenesis by influencing MAPK signaling downstream of RhoA and ROCK. J. Bone Miner. Res. 2009, 24, 886–898. [Google Scholar] [CrossRef]
- Abdallah, B.M.; Alzahrani, A.M.; Kassem, M. Secreted Clusterin protein inhibits osteoblast differentiation of bone marrow mesenchymal stem cells by suppressing ERK1/2 signaling pathway. Bone 2018, 110, 221–229. [Google Scholar] [CrossRef]
- McBeath, R.; Pirone, D.M.; Nelson, C.M.; Bhadriraju, K.; Chen, C.S. Cell shape, cytoskeletal tension, and RhoA regulate stem cell lineage commitment. Dev. Cell 2004, 6, 483–495. [Google Scholar] [CrossRef]
- Wang, Z.; Ka, S.O.; Lee, Y.; Park, B.H.; Bae, E.J. Butein induction of HO-1 by p38 MAPK/Nrf2 pathway in adipocytes attenuates high-fat diet induced adipose hypertrophy in mice. Eur. J. Pharmacol. 2017, 799, 201–210. [Google Scholar] [CrossRef]
- Yang, J.; Sung, J.; Kim, Y.; Jeong, H.S.; Lee, J. Inhibitory Effects of Butein on Adipogenesis through Upregulation of the Nrf2/HO-1 Pathway in 3T3-L1 Adipocytes. Prev. Nutr. Food Sci. 2017, 22, 306–311. [Google Scholar] [CrossRef]
- Song, N.J.; Choi, S.; Rajbhandari, P.; Chang, S.H.; Kim, S.; Vergnes, L.; Kwon, S.M.; Yoon, J.H.; Lee, S.; Ku, J.M.; et al. Prdm4 induction by the small molecule butein promotes white adipose tissue browning. Nat. Chem. Biol. 2016, 12, 479–481. [Google Scholar] [CrossRef]
- Sun, S.; Choi, Y.-H.; Na, C.-S.; Lee, D.; Yoo, H.H.; Hong, C.Y.; Ahn, B.Y.; Dong, M.-S. Estrogenic activity of a Rhus verniciflua extract and its major components. J. Funct. Foods 2014, 11, 250–260. [Google Scholar] [CrossRef]
- Cepeda, S.B.; Sandoval, M.J.; Crescitelli, M.C.; Rauschemberger, M.B.; Massheimer, V.L. The isoflavone genistein enhances osteoblastogenesis: Signaling pathways involved. J. Physiol. Biochem. 2020, 76, 99–110. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Xia, L.; Zhou, Y.; Xu, Y.; Jiang, X. Icariin induces osteogenic differentiation of bone mesenchymal stem cells in a MAPK-dependent manner. Cell Prolif. 2015, 48, 375–384. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.J.; Liu, L.Q.; Bo, T.; Li, S.J.; Zhu, Z.; Cui, R.R.; Mao, D.A. Puerarin Suppress Apoptosis of Human Osteoblasts via ERK Signaling Pathway. Int. J. Endocrinol. 2013, 2013, 786574. [Google Scholar] [CrossRef] [PubMed]
- Abdallah, B.M.; Alzahrani, A.M.; Abdel-Moneim, A.M.; Ditzel, N.; Kassem, M. A simple and reliable protocol for long-term culture of murine bone marrow stromal (mesenchymal) stem cells that retained their in vitro and in vivo stemness in long-term culture. Biol. Proced. Online 2019, 21, 3. [Google Scholar] [CrossRef]
- Abdallah, B.M.; Ali, E.M. 5′-hydroxy Auraptene stimulates osteoblast differentiation of bone marrow-derived mesenchymal stem cells via a BMP-dependent mechanism. J. Biomed. Sci. 2019, 26, 51. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are available from the authors. |

| Gene Name | Gene Symbol | Fold Change |
|---|---|---|
| Ossification and Matrix Molecules | ||
| Alkaline phosphatase, liver/bone/kidney | Alpl | 6.2 |
| Bone gamma carboxyglutamate protein | Bglap | 5.8 |
| Biglycan | Bgn | 3.5 |
| Collagen type I alpha 1 | Col1a1 | 5.6 |
| Collagen type I alpha 2 | Col1a2 | 4.2 |
| Collagen type V alpha 1 | Col5a1 | 2.4 |
| FMS-like tyrosine kinase 1 | Flt1 | 3.6 |
| Secreted phosphoprotein 1 (Osteopontin) | Spp1 | 4.3 |
| Cell Adhesion Molecules | ||
| Fibronectin 1 | Fn1 | 2.1 |
| Integrin beta 1 (fibronectin receptor beta) | Itgb1 | 2.3 |
| Integrin alpha 2 | Itga2 | 3.4 |
| Integrin alpha 2b | Itga2b | 2.2 |
| Integrin alpha 3 | Itga3 | 2.5 |
| Osteogenic Growth Factors | ||
| Fibroblast growth factor receptor 2 | Fgfr2 | 3.6 |
| Growth differentiation factor 10 | Gdf10 | 2.8 |
| Insulin-like growth factor 1 | Igf1 | 3.4 |
| Insulin-like growth factor I receptor | Igf1r | 3.5 |
| Platelet-derived growth factor alpha | Pdgfa | 2.9 |
| Vascular endothelial growth factor-A | Vegf-a | 5.3 |
| Vascular endothelial growth factor-B | Vegf-b | 3.6 |
| Osteogenic Transcription Factors | ||
| Distal-less homeobox 5 | Dlx5 | 4.6 |
| Runt-related transcription factor 2 | Runx2 | 3.4 |
| Sp7 transcription factor 7 | Sp7 | 5.2 |
| Twist gene homolog 1 | Twist1 | 3.7 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abdallah, B.M.; Ali, E.M. Butein Promotes Lineage Commitment of Bone Marrow-Derived Stem Cells into Osteoblasts via Modulating ERK1/2 Signaling Pathways. Molecules 2020, 25, 1885. https://doi.org/10.3390/molecules25081885
Abdallah BM, Ali EM. Butein Promotes Lineage Commitment of Bone Marrow-Derived Stem Cells into Osteoblasts via Modulating ERK1/2 Signaling Pathways. Molecules. 2020; 25(8):1885. https://doi.org/10.3390/molecules25081885
Chicago/Turabian StyleAbdallah, Basem M., and Enas M. Ali. 2020. "Butein Promotes Lineage Commitment of Bone Marrow-Derived Stem Cells into Osteoblasts via Modulating ERK1/2 Signaling Pathways" Molecules 25, no. 8: 1885. https://doi.org/10.3390/molecules25081885
APA StyleAbdallah, B. M., & Ali, E. M. (2020). Butein Promotes Lineage Commitment of Bone Marrow-Derived Stem Cells into Osteoblasts via Modulating ERK1/2 Signaling Pathways. Molecules, 25(8), 1885. https://doi.org/10.3390/molecules25081885





