3.1.4. Removal of tert-Butoxycarbonyl and Diethyl Phosphonate Ester Protecting Groups
Phosphonopeptide containing N-tert-butoxycarbonyl and O,O-diethyl phosphonate ester was dissolved in 35 %wt HBr in acetic acid (10 equiv) and stirred at room temperature overnight (16–18 h). Dry diethyl ether was added to afford a precipitation. The resulting mixture was stored in the freezer overnight (16–18 h) to result in more precipitation. The solvent was decanted and remaining crude solid was triturated (×3) with dry diethyl ether. The resulting solid was dissolved in dry methanol and propylene oxide was added in excess to afford a crude precipitation, which was recrystallized from the appropriate solvent system to afford the product of interest.
![Molecules 25 01557 i001 Molecules 25 01557 i001]()
1-Aminoethylphosphonic Acid or d/l-fosfalin (
2-DL). To suspension of
N-phenylthiourea (100.0 mmol, 15.2 g) in glacial acetic acid (50 mL), acetaldehyde (130.0 mmol, 7.40 mL) was added dropwise, followed by triphenyl phosphite (100.0 mmol, 27.0 mL). The mixture was stirred at room temperature for 5 mins, then refluxed at 80 °C for 1 h until a clear solution was obtained. A mixture of glacial acetic acid (5 mL) and hydrochloric acid (37%, 50 mL) was added and the reaction was refluxed overnight. The solution was cooled to room temperature and concentrated in vacuo to afford a brown slurry. Absolute ethanol (150 mL) was added while stirring and the resulting off-white solid was collected by filtration and dried in a desiccator containing phosphorus(V) oxide. The crude solid was recrystallized from hot water/ethanol to afford
2-DL as white crystals, as a mixture of enantiomers (12.2 g, 98 mmol, 98%); m.p. 271–274 °C (sublim);
ῡmax/cm
−1 2910 (br OH), 1532 (NH bend), 1143 (P=O), 1035 (P-O-C), 930 (P-OH);
1H NMR (300 MHz, D
2O) δ
H 1.40 (3H, dd,
3JH-P = 14.7 Hz,
3JH-H = 7.2 Hz, C
H3), 3.33 (1H, m, C
H);
13C NMR (75 MHz, D
2O) δ
C 13.5 (d,
2JC-P = 2.6 Hz,
CH
3), 44.7 (d,
1JC-P = 144.2 Hz,
CH);
31P-
1H
decoup NMR (121 MHz, D
2O) δ
P 14.2;
m/z (ESI) calcd for (C
2H
9NO
3P)
+, MH
+: 126.0, found 126.1; CHN (Found: C, 19.45; H, 6.48; N, 11.18. C
2H
8NO
3P requires C, 19.21; H, 6.45; N, 11.20%). (The
1H- and
13C-NMR spectra may be found within the
Supplementary Materials).
![Molecules 25 01557 i002 Molecules 25 01557 i002]()
Diethyl (1-(2,2,2-trifluoroacetamido)ethyl)phosphonate or trifluoroacetyl-d/l-Fos diethyl ester (
8). 1-Aminoethylphosphonic acid (
2-DL) (51.7 mmol, 6.5 g) was added to a mixture of trifluoroacetic acid (65.3 mmol, 5 mL) and trifluoroacetic anhydride (177.4 mmol, 25 mL). The solution was stirred and refluxed at 60 °C for 1 h, then cooled to room temperature and triethyl orthoformate (901.8 mmol, 150 mL) was added dropwise. The solution was refluxed at 110 °C for 2 h, then cooled to room temperature. The solution was concentrated in vacuo to afford a brown solid, which was re-dissolved in DCM and purified by column chromatography using a gradient elution (DCM (100) to DCM/MeOH (95:5)) to give
8 as an off-white solid, a mixture of enantiomers (11.4 g, 41.0 mmol, 80%); m.p. 101–103 °C (sublim) (lit. m.p. 101–102 °C) [
29];
ῡmax/cm
−1 3202 (NH), 1715 (C=O), 1565 (NH bend), 1210 (P=O), 1011 (P-O-C), 968 (P-O-C);
1H NMR (300 MHz, CDCl
3) δ
H 1.24 (3H, t,
3JH-H = 7.2 Hz, OCH
2C
H3), 1.27 (3H, t,
3JH-H = 7.2 Hz, OCH
2C
H3), 1.38 (3H, dd,
3JH-P = 16.5 Hz,
3JH-H = 7.2 Hz, C
H3-2), 4.06 (4H, m, 2 × OC
H2CH
3), 4.39 (1H, m, C
H-1), 8.00 (1H, d,
3JH-H = 6.0 Hz, N
H);
13C NMR (75 MHz, CDCl
3) δ
C 14.8 (
CH
3-2), 16.2 (d,
3JC-P = 2.3 Hz, OCH
2CH
3), 16.3 (d,
3JC-P = 2.3 Hz, OCH
2CH
3), 41.8 (d,
1JC-P = 159.1 Hz,
CH-1), 62.8 (d,
2JC-P = 7.0 Hz, O
CH
2CH
3), 63.2 (d,
2JC-P = 7.1 Hz, O
CH
2CH
3), 115.9 (q,
1JC-F = 285.8 Hz,
CF
3), 156.9 (q,
2JC-F = 5.8 Hz,
C=O);
31P-
1H
decoup NMR (121 MHz, CDCl
3) δ
P 23.0;
19F-
1H
decoup NMR (282 MHz, CDCl
3) δ
P −75.5;
m/z (ESI) calcd for (C
8H
16F
3NO
4P)
+, MH
+: 278.1, found 278.1.
![Molecules 25 01557 i003 Molecules 25 01557 i003]()
Diethyl 1-aminoethylphosphonate or d/l-Fos diethyl ester (9). Diethyl (1-(2,2,2-trifluoroacetamido)ethyl)phosphonate (8) (20.0 mmol, 5.6 g) was dissolved in ethanol (200 ml) and excess sodium borohydride (200.0 mmol, 7.7 g) was added slowly with stirring. The resulting mixture was stirred at room temperature for 1 h, then heated at reflux for 4 h. The mixture was cooled to room temperature and the solvent was removed in vacuo to afford a white solid, which was dissolved in saturated NaHCO3 (96 g/L) (60 mL) with the addition of 10% aqueous K2CO3 (20 mL). The product was extracted into DCM (6 × 30 mL) and dried over MgSO4. The filtrate was concentrated in vacuo to afford a pale yellow liquid and purified by column chromatography using a gradient elution (DCM (100) to DCM/MeOH (90:10)) to afford 9 as a yellow liquid, a mixture of enantiomers (3.5 g, 19.3 mmol, 97%); ῡmax/cm−1 3431 (NH), 1215 ( P=O), 1020 (P-O-C), 967 (P-O-C); 1H NMR (300 MHz, CDCl3) δH 1.26 (6H, t, 3JH-H = 7.2 Hz, 2 × OCH2CH3), 1.34 (3H, dd, 3JH-P = 17.7 Hz, 3JH-H = 7.2 Hz, CH3-2), 1.68 (2H, br, NH2), 3.02–3.12 (1H, m, CH-1), 4.06–4.17 (4H, m, 2 × OCH2CH3); 13C NMR (75 MHz, CDCl3) δC 16.4 (OCH2CH3), 16.5 (OCH2CH3), 17.2 (CH3-2), 44.2 (d, 1JC-P = 148.5 Hz, CH-1), 62.1 (d, 2JC-P = 7.5 Hz, OCH2CH3), 62.1 (d, 2JC-P = 7.5 Hz, OCH2CH3); 31P-1Hdecoup NMR (121 MHz, CDCl3) δP 29.6; HRMS (NSI) calcd for (C6H17NO3P)+, MH+: 204.0760, found 204.0762. LCMS purity >95% (C-18 reversed phase, MeOH–H2O).
![Molecules 25 01557 i004 Molecules 25 01557 i004]()
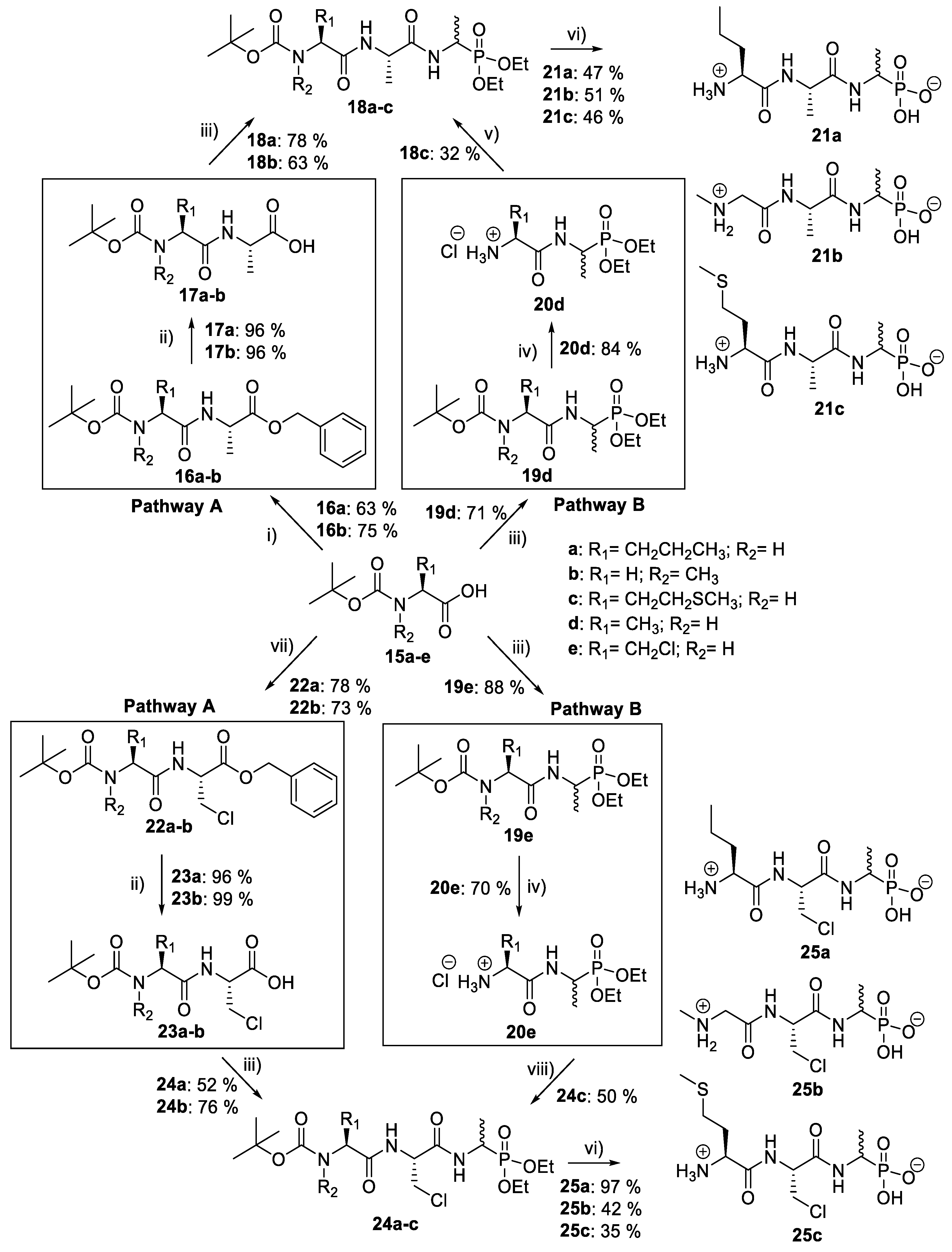
(S)-Benzyl 2-((S)-2-((tert-butoxycarbonyl)amino)pentanamido) propanoate or Boc-l-Nva-l-Ala-OBzl (16a). General peptide coupling method was followed, using Boc-l-Nva-OH (15a) (10.0 mmol, 2.17 g) in dry THF and l-alanine benzyl ester p-tosylic acid (10.0 mmol, 3.52 g) in dry DCM. The yellow crude liquid was purified by column chromatography (40–60 petrol/ethyl acetate (7:3)) to give 16a as an off-white solid (2.40 g, 6.3 mmol, 63%); m.p. 60–63 °C; ῡmax/cm−1 3332 (NH), 1743 (C=O), 1655 (br C=O), 1527 (NH bend), 1245 (C-O), 1162 (C-O); 1H NMR (300 MHz, CDCl3) δH 0.83 (3H, t, 3JH-H = 9.0 Hz, CH3-7″), 1.25–1.31 (2H, m, CH2-7′), 1.34 (3H, d, 3JH-H = 6.0 Hz, CH3-3), 1.36 (9H, s, C(CH3)3), 1.42–1.54 (1H, m, CHa/b-7), 1.64–1.73 (1H, m, CHa/b-7), 4.02 (1H, m, CH-6), 4.54 (1H, pentet, 3JH-H = 6.0 Hz, CH-2), 4.96 (1H, d, 3JH-H = 9.0 Hz, NH-8), 5.07 (1H, d, 2JH-H = 12.0 Hz, OCHa/bAr), 5.12 (1H, d, 2JH-H = 12.0 Hz, OCHa/bAr), 6.56 (1H, d, 3JH-H = 6.0 Hz, NH-4), 7.27 (5H, m, 5 × CHAr); 13C NMR (75 MHz, CDCl3) δC 12.7 (CH3-7″), 17.3 (CH3-3), 17.8 (CH2-7′), 27.3 (C(CH3)3), 33.7 (CH2-7), 47.1 (CH-2), 53.4 (CH-6), 66.1(OCH2Ar), 79.0 (C(CH3)3), 127.1-127.6 (CHAr), 134.3 (CHAr quat.), 154.6 (C=O-9), 170.8 (C=O-5), 171.5 (C=O-1); HRMS (NSI) calcd for (C20H31N2O5)+, MH+: 379.2227, found 379.2222; CHN (Found: C, 63.75; H, 8.37; N, 7.86. C20H30N2O5 requires C, 63.47; H, 7.99; N, 7.40%).
![Molecules 25 01557 i005 Molecules 25 01557 i005]()
(S)-Benzyl 2-(2-((tert-butoxycarbonyl)(methyl)amino)acetamido) propanoate or Boc-Sar-l-Ala-OBzl (16b). General peptide coupling method was followed, using Boc-Sar-OH (15b) (15.0 mmol, 2.84 g) in dry THF and l-alanine benzyl ester p-tosylic acid (15.0 mmol, 5.27 g) in dry DCM. The yellow crude liquid was purified by column chromatography (40–60petrol/ethyl acetate (1:1)) to give 16b as a colourless liquid (3.93 g, 11 mmol, 75%); ῡmax/cm−1 3311 (NH), 1742 (C=O), 1670 (C=O), 1666 (C=O), 1536 (NH bend), 1242 (C-O), 1145 (C-O); 1H NMR (300 MHz, CDCl3) δH 1.35 (3H, t, 3JH-H = 6.0 Hz, CH3-3), 1.39 (9H, s, C(CH3)3), 2.85 (3H, s, CH3-8), 3.72 (1H, d, 2JH-H = 15.0 Hz, CHa/b-6), 3.88 (1H, d, 2JH-H = 15.0 Hz, CHa/b-6), 4.58 (1H, pentet, 3JH-H = 6.0 Hz, CH-2), 5.08 (1H, d, 2JH-H = 12.0 Hz, OCHa/bAr), 5.13 (1H, d, 2JH-H = 12.0 Hz, OCHa/bAr), 6.51 (1H, br, NH-4), 7.25-7.29 (5H, m, 5 × CHAr); 13C NMR (75 MHz, CDCl3) δC 17.5 (CH3-3), 27.3 (C(CH3)3), 34.7 (CH3-8), 47.0 (CH-2), 52.1 (CH2-6), 66.2 (OCH2Ar), 79.8 (C(CH3)3), 127.1–127.6 (CHAr), 134.3 (CHAr quat.), 155.0 (C=O-9), 167.9 (C=O-5), 171.5 (C=O-1); HRMS (NSI) calcd for (C18H27N2O5)+, MH+: 351.1914, found 351.1916. LCMS purity >95% (C-18 reversed phase, MeOH–H2O).
![Molecules 25 01557 i006 Molecules 25 01557 i006]()
Tert-butyl ((2S)-1-((-1-(diethoxyphosphoryl)ethyl)amino)-1-oxopropan-2-yl)carbamate or Boc-l-Ala-d/l-Fos diethyl ester (19d). General peptide coupling method was followed, using Boc-l-Ala-OH (15d) (10.0 mmol, 1.90 g) in dry THF and diethyl 1-aminoethylphosphonate (9) (10.0 mmol, 1.84 g) in dry THF. The pale yellow crude syrup was purified by column chromatography, using 100% DCM and increasing to 95:5 DCM/methanol, to afford 19d as an off-white solid composed of 2 diastereoisomers, Boc-l-Ala-l-Fos diethyl ester and Boc-l-Ala-D-Fos diethyl ester (2.49 g, 7.1 mmol, 71%); m.p. 102–105 °C; ῡmax/cm−1 3280 (NH), 1710 (C=O), 1652 (C=O), 1556 (NH bend), 1229 (P=O), 1173 (C-O), 1013 (P-O-C), 973 (P-O-C); 1H NMR (300 MHz, CDCl3) δH 1.23–1.43 (12H, m, CH3-2, CH3-6, 2 × OCH2CH3), 1.44 (9H, s, C(CH3)3), 4.06–4.23 (5H, m, 2 × OCH2CH3, CH-5), 4.40–4.52 (1H, m, CH-1), 5.12 (0.5H, d, 3JH-H = 1.5 Hz, NH-7), 5.14 (0.5H, d, 3JH-H = 1.5 Hz, NH-7), 6.72 (0.5H, d, 3JH-H = 2.3 Hz, NH-3), 6.74 (0.5H, d, 3JH-H = 2.3 Hz, NH-3); 13C NMR (75 MHz, CDCl3) δC 15.6 (CH3-2), 16.3 (d, 3JC-P = 3.0 Hz, OCH2CH3), 16.4 (d, 3JC-P = 2.3 Hz, OCH2CH3), 16.5 (d, 3JC-P = 3.0 Hz, OCH2CH3), 16.6 (d, 3JC-P = 2.3 Hz, OCH2CH3), 18.4 (CH3-6), 28.3 (C(CH3)3), 40.8 (d, 1JC-P = 156.8 Hz, CH-1), 41.0 (d, 1JC-P = 156.8 Hz, CH-1), 50.0 (CH-5), 62.4 (d, 2JC-P = 6.8 Hz, OCH2CH3), 62.5 (d, 2JC-P = 6.8 Hz, OCH2CH3), 62.6 (d, 2JC-P = 6.8 Hz, OCH2CH3), 62.8 (d, 2JC-P = 6.8 Hz, OCH2CH3), 80.0 (C(CH3)3), 155.2 (C=O-8), 172.1 (C=O-4); 31P-1Hdecoup NMR (121 MHz, CDCl3) δP 25.2; HRMS (NSI) calcd for (C17H35N3O7P)+, MH+: 424.2207, found 424.2200; CHN (Found: C, 48.22; H, 8.58; N, 7.87. C14H29N2O6P requires C, 47.92; H, 8.30; N, 7.95%).
![Molecules 25 01557 i007 Molecules 25 01557 i007]()
(S)-2-((S)-2-((Tert-butoxycarbonyl)amino)pentanamido) propanoic acid or Boc-l-Nva-l-Ala-OH (17a). Deprotection of benzyl ester was followed, using (S)-benzyl 2-((S)-2-((tert-butoxycarbonyl)amino)pentanamido)propanoate (16a) (6.0 mmol, 2.27 g) to afford 17a as a white solid (1.66 g, 5.7 mmol, 96.0%); m.p. 55–58 °C (decomp.); ῡmax/cm−1 3500–3000 (br, OH), 3300 (NH), 1688 (br C=O), 1655 (C=O), 1522 (NH bend), 1245 (C-O), 1164 (C-O); 1H NMR (300 MHz, CDCl3) δH 0.85 (3H, t, 3JH-H = 9.0 Hz, CH3-7″), 1.27–1.31 (5H, m, CH3-3, CH2-7′), 1.39 (9H, s, C(CH3)3), 1.48–1.53 (1H, m, CHa/b-7), 1.67–1.71 (1H, m, CHa/b-7), 4.10 (1H, m, CH-6), 4.50 (1H, m, CH-2), 5.27 (1H, m, NH-8), 6.93 (1H, m, NH-4), 8.87 (1H, br, OH); 13C NMR (75 MHz, CDCl3) δC 13.7 (CH3-7″), 18.0 (CH3-3), 18.8 (CH2-7′), 28.3 (C(CH3)3), 34.5 (CH2-7), 48.1 (CH-2), 54.3 (CH-6), 80.4 (C(CH3)3), 156.0 (C=O-9), 172.5 (C=O-5), 175.5 (C=O-1); HRMS (NSI) calcd for (C13H25N2O5)+, MH+: 289.1758, found 289.1758; CHN (Found: C, 54.18; H, 8.78; N, 9.62. C13H24N2O5 requires C, 54.15; H, 8.39; N, 9.72%).
![Molecules 25 01557 i008 Molecules 25 01557 i008]()
(S)-Benzyl 2-(2-((tert-butoxycarbonyl)(methyl)amino)acetamido) propanoic acid or Boc-Sar-l-Ala-OH (17b). Deprotection of benzyl ester was followed, using (S)-benzyl 2-(2-((tert-butoxycarbonyl)(methyl)amino)acetamido) propanoate (16b) (10.0 mmol, 3.51 g) to afford 17b as a colorless syrup (2.50 g, 9.6 mmol, 96%); ῡmax/cm−1 3301 (NH), 2961 (broad OH), 1736 (C=O), 1664 (br C=O), 1542 (NH bend), 1241 (C-O), 1147 (C-O); 1H NMR (300 MHz, CDCl3) δH 1.36 (3H, t, 3JH-H = 6.0 Hz, CH3-3), 1.39 (9H, s, C(CH3)3), 2.89 (3H, s, CH3-8), 3.72 (1H, d, 2JH-H = 18.0 Hz, CHa/b-6), 3.98 (1H, d, 2JH-H = 18.0 Hz, CHa/b-6), 4.57 (1H, m, CH-2), 6.96 (1H, m, NH-4), 7.26 (1H, br, OH); 13C NMR (75 MHz, CDCl3) δC 17.2 (CH3-3), 27.3 (C(CH3)3), 46.8 (CH-2), 49.6 (CH3-8), 52.1 (CH2-6), 80.6 (C(CH3)3), 155.5 (C=O-9), 168.3 (C=O-5), 174.1 (C=O-1); HRMS (NSI) calcd for (C11H19N2O5)-, MH−: 259.1299, found 259.1295. LCMS purity >95% (C-18 reversed phase, MeOH-H2O).
![Molecules 25 01557 i009 Molecules 25 01557 i009]()
(2S)-1-((1-(Diethoxyphosphoryl)ethyl)amino)-1-oxopropan-2-aminium chloride or l-Ala-d/l-Fos diethyl ester hydrochloride (20d). Deprotection of tert-butoxycarbonyl was followed, using tert-butyl ((2S)-1-((-1-(diethoxyphosphoryl)ethyl)amino)-1-oxopropan-2-yl)carbamate (19d) (6.0 mmol, 2.13 g). The off-white hygroscopic crude solid was washed with petrol to afford 20d as a pale green solid composed of 2 diastereoisomers, l-Ala-l-Fos diethyl ester hydrochloride and l-Ala-D-Fos diethyl ester hydrochloride (1.46 g, 5.1 mmol, 84%); m.p. 60–63 °C; ῡmax/cm−1 2986 (NH+), 1673 (C=O), 1555 (NH bend), 1212(P=O), 1017 (P-O-C), 970 (P-O-C); 1H NMR (300 MHz, CD3OD) δH 1.29-1.44 (9H, m, 2 × OCH2CH3, CH3-2), 1.51 (3H, d, 3JH-H = 6.0 Hz, CH3-6), 3.90-3.98 (1H, m, CH-5), 4.08-4.22 (4H, m, 2 × OCH2CH3), 4.28-4.47 (1H, m, CH-1); 13C NMR (75 MHz, CD3OD) δC 13.7 (CH3-2), 14.0 (CH3-2), 15.4 (2 × OCH2CH3), 16.3 (CH3-6), 41.1 (d, 1JC-P = 158.3 Hz, CH-1), 41.4 (d, 1JC-P = 158.3 Hz, CH-1), 48.8 (CH-5), 48.9 (CH-5), 62.7–63.0 (2 × OCH2CH3), 169.0 (C=O-4); 31P-1Hdecoup NMR (121 MHz, CDCl3) δP 29.0, 29.1; HRMS (NSI) calcd for (C9H22N2O4P)+, M+: 253.1312, found 253.1316. LCMS purity >95% (C-18 reversed phase, MeOH-H2O).
![Molecules 25 01557 i010 Molecules 25 01557 i010]()
Tert-butyl ((2S)-1-(((2S)-1-((1-(diethoxyphosphoryl)ethyl) amino)-1-oxopropan-2-yl)amino)-1-oxopentan-2-yl)carbamate or Boc-l-Nva-l-Ala-d/l-Fos diethyl ester (18a). General peptide coupling method was followed, using (S)-2-((S)-2-((tert-butoxycarbonyl)amino)pentanamido)propanoic acid (17a) (5.0 mmol, 1.45 g) in dry THF and diethyl 1-aminoethylphosphonate (9) (4.8 mmol, 0.87 g) in dry THF. The white crude solid was purified by column chromatography using 100% DCM, increasing to 90:10 DCM/methanol, to afford 18a as a white solid composed of 2 diastereoisomers, Boc-l-Nva-l-Ala-l-Fos diethyl ester and Boc-l-Nva-l-Ala-D-Fos diethyl ester (1.70 g, 3.8 mmol, 78%); m.p. 165–168 °C; ῡmax/cm−1 3267 (NH), 1708 (C=O), 1638 (br C=O), 1537 (NH bend), 1227 (P=O), 1165 (C-O), 1019 (P-O-C), 966 (P-O-C); 1H NMR (300 MHz, CDCl3) δH 0.85 (3H, t, 3JH-H = 9.0 Hz, CH3-10″), 1.18–1.34 (14H, m, 2 × OCH2CH3, CH3-2, CH3-6, CH2-10′), 1.37 (9H, s, C(CH3)3), 1.47–1.54 (1H, m, CHa/b-10), 1.65–1.73 (1H, m, CHa/b-10), 3.98–4.12 (5H, m, 2 × OCH2CH3, CH-9), 4.33–4.44 (1H, m, CH-1), 4.48–4.54 (1H, m, CH-5), 5.19 (0.5H, d, 3JH-H = 6.0 Hz, NH-11), 5.23 (0.5H, d, 3JH-H = 6.0 Hz, NH-11), 6.78 (0.5H, d, 3JH-H = 6.0 Hz, NH-7), 6.87 (0.5H, d, 3JH-H = 6.0 Hz, NH-7), 7.15 (0.5H, d, 3JH-H = 9.0 Hz, NH-3), 7.23 (0.5H, d, 3JH-H = 9.0 Hz, NH-3); 13C NMR (75 MHz, CDCl3) δC 12.7 (CH3-10″), 14.4 (CH3-2), 14.5 (CH3-2), 15.3 (OCH2CH3), 15.4 (OCH2CH3), 15.5 (OCH2CH3), 15.6 (OCH2CH3), 17.6 (CH3-6), 17.7 (CH3-6), 17.8 (CH2-10′), 17.9 (CH2-10′), 27.3 (C(CH3)3), 33.8 (CH2-10), 33.9 (CH2-10), 39.9 (d, 1JP-C = 157.5 Hz, CH-1), 40.0 (d, 1JP-C = 156.8 Hz, CH-1), 47.7 (CH-5), 47.9 (CH-5), 53.5 (CH-9), 53.5 (CH-9), 61.5 (d, 2JC-P = 7.5 Hz, OCH2CH3), 61.6 (d, 2JC-P = 7.5 Hz, OCH2CH3), 61.7 (d, 2JC-P = 7.5 Hz, OCH2CH3), 61.9 (d, 2JC-P = 7.5 Hz, OCH2CH3), 78.9 (C(CH3)3), 154.7 (C=O-12), 170.6 (C=O-4 or C=O-8), 170.7 (C=O-4 or C=O-8), 171.0 (C=O-4 or C=O-8), 171.1 (C=O-4 or C=O-8); 31P-1Hdecoup NMR (121 MHz, CDCl3) δP 25.0, 25.1; HRMS (NSI) calcd for (C19H39N3O7P)+, MH+: 452.2520, found 452.2518; CHN (Found: C, 50.74; H, 8.55; N, 9.51. C19H38N3O7P requires C, 50.54; H, 8.48; N, 9.31%).
![Molecules 25 01557 i011 Molecules 25 01557 i011]()
Tert-butyl (2-(((2S)-1-((1-(diethoxyphosphoryl)ethyl)amino)-1-oxopropan-2-yl)amino)-2-oxoethyl)(methyl)carbamate or Boc-Sar-l-Ala-d/l-Fos (18b). General peptide coupling method was followed, using (S)-benzyl 2-(2-((tert-butoxycarbonyl)(methyl)amino)acetamido)propanoic acid (17b) (6.0 mmol, 1.57 g) in dry THF and diethyl 1-aminoethylphosphonate (9) (6.0 mmol, 1.10 g) in dry THF. The yellow crude liquid was purified by column chromatography, using 100% DCM and increasing to 90:10 DCM/methanol, to afford 18b as a colorless liquid composed of 2 diastereoisomers, Boc-Sar-l-Ala-l-Fos diethyl ester and Boc-Sar-l-Ala-D-Fos diethyl ester (1.60 g, 3.8 mmol, 63%); ῡmax/cm−1 3270 (NH), 1700 (br C=O), 1655 (C=O), 1545 (NH bend), 1225 (P=O), 1149 (C-O), 1018 (P-O-C), 966 (P-O-C); 1H NMR (300 MHz, CDCl3) δH 1.19–1.34 (12H, m, CH3-2, CH3-6, 2 × OCH2CH3), 1.40 (9H, s, C(CH3)3), 2.87 (3H, s, CH3-11), 3.72 (0.5H, d, 2JH-H = 15.0 Hz, CHa/b-9), 3.78 (0.5H, d, 2JH-H = 15.0 Hz, CHa/b-9), 3.81 (0.5H, d, 2JH-H = 15.0 Hz, CHa/b-9), 3.87 (0.5H, d, 2JH-H = 15.0 Hz, CHa/b-9), 4.00–4.11 (4H, m, 2 × OCH2CH3), 4.35-4.43 (1H, m, CH-1), 4.47–4.52 (1H, m, CH-5), 6.67 (1H, d, 3JH-H = 9.0 Hz, NH-7), 6.98 (0.5H, d, 3JH-H = 9.0 Hz, NH-3), 7.15 (0.5H, d, 3JH-H = 9.0 Hz, NH-3); 13C NMR (75 MHz, CDCl3) δC 15.5 (CH3-2), 15.5 (CH3-2), 16.3 (d, 3JP-C = 3.0 Hz, OCH2CH3), 16.4 (d, 3JP-C = 3.0 Hz, OCH2CH3), 18.7 (CH3-6), 28.3 (C(CH3)3), 35.8 (CH3-11), 41.0 (d, 1JP-C = 157.5 Hz, CH-1), 48.5 (CH-5), 53.0 (CH2-9), 62.5 (d, 2JP-C = 6.8 Hz, OCH2CH3), 62.6 (d, 2JP-C = 6.8 Hz, OCH2CH3), 62.7 (d, 2JP-C = 6.8 Hz, OCH2CH3), 62.9 (d, 2JP-C = 6.8 Hz, OCH2CH3), 80.7 (C(CH3)3), 156.0 (C=O-12), 171.5 (C=O-4 or C=O-8), 171.6 (C=O-4 or C=O-8); 31P-1Hdecoup NMR (121 MHz, CDCl3) δP 25.0, 25.1; HRMS (NSI) calcd for (C17H35N3O7P)+, MH+: 424.2207, found 424.2203. LCMS purity >95% (C-18 reversed phase, MeOH–H2O).
![Molecules 25 01557 i012 Molecules 25 01557 i012]()
Tert-butyl ((2S)-1-(((2S)-1-((1-(diethoxyphosphoryl)ethyl) amino)-1-oxopropan-2-yl)amino-4-(methylthio)-1-oxobutan-2-yl)carbamate or Boc-l-Met-l-Ala-d/l-Fos diethyl ester (18c). General peptide coupling method was followed, using Boc-l-Ala-Met-OH (15c) (3.4 mmol, 0.88 g) in dry THF and (2S)-1-((1-(diethoxyphosphoryl)ethyl)amino)-1-oxopropan-2-aminium chloride (20d) (3.4 mmol, 0.97 g) in dry DCM. The yellow crude solid was purified by column chromatography (DCM/MeOH (95:5)) to give 18c as an off-white solid composed of 2 diastereoisomers, Boc-l-Met-l-Ala-l-Fos diethyl ester and Boc-l-Met-l-Ala-D-Fos diethyl ester (0.53 g, 1.1 mmol, 32%); m.p. 172–176 °C; ῡmax/cm−1 3272 (NH), 1708 (C=O), 1673 (C=O), 1637 (C=O), 1530 (NH bend), 1226 (P=O), 1165 (C-O), 1020 (P-O-C), 976 (P-O-C); 1H NMR (300 MHz, CDCl3) δH 1.16–1.36 (12H, m, CH3-2, CH3-6, 2 × OCH2CH3), 1.36 (9H, s, C(CH3)3), 1.82–2.01 (2H, m, CH2-10), 2.04 (3H, s, CH3-10″), 2.49 (2H, dd, 3JH-H = 9.0 Hz, 3.0 Hz, CH2-10′), 4.00–4.12 (4H, m, 2 × OCH2CH3), 4.16–4.26 (1H, m, CH-9), 4.33–4.43 (1H, m, CH-1), 4.45–4.53 (1H, m, CH-5), 5.40 (0.5H, d, 3JH-H = 9.0 Hz, NH-11), 5.44 (0.5H, d, 3JH-H = 6.0 Hz, NH-11), 6.85 (0.5H, d, 3JH-H = 6.0 Hz, NH-7), 6.92 (0.5H, d, 3JH-H = 6.0 Hz, NH-7), 7.07 (0.5H, d, 3JH-H = 9.0 Hz, NH-3), 7.16 (0.5H, d, 3JH-H = 9.0 Hz, NH-3); 13C NMR (75 MHz, CDCl3) δC 14.2 (CH3-2), 14.3 (CH3-2), 14.5 (CH3-10″), 14.6 (CH3-10″), 15.4 (d, 3JC-P = 3.0 Hz, OCH2CH3), 15.4 (d, 3JC-P = 2.3 Hz, OCH2CH3), 15.5 (d, 3JC-P = 3.0 Hz, OCH2CH3), 15.5 (d, 3JC-P = 2.3 Hz, OCH2CH3), 17.7 (CH3-6), 27.3 (C(CH3)3), 29.2 (CH2-10′), 29.3 (CH2-10′), 30.8 (CH2-10), 30.9 (CH2-10), 39.9 (d, 1JC-P = 156.8 Hz, CH-1), 40.0 (d, 1JC-P = 156.8 Hz, CH-1), 47.9 (CH-5), 48.0 (CH-5), 52.6 (CH-9), 61.5 (d, 2JC-P = 6.8 Hz, OCH2CH3), 61.6 (d, 2JC-P = 6.8 Hz, OCH2CH3), 61.7 (d, 2JC-P = 6.8 Hz, OCH2CH3), 61.9 (d, 2JC-P = 6.8 Hz, OCH2CH3), 79.1 (C(CH3)3), 154.6 (C=O-12), 170.3 (C=O-4 or C=O-8), 170.4 (C=O-4 or C=O-8), 170.5 (C=O-4 or C=O-8), 170.6 (C=O-4 or C=O-8); 31P-1Hdecoup NMR (121 MHz, CDCl3) δP 25.0, 25.1; HRMS (NSI) calcd for (C19H39N3O7PS)+, MH+: 484.2241, found 484.2228. LCMS purity >95% (C-18 reversed phase, MeOH–H2O).
![Molecules 25 01557 i013 Molecules 25 01557 i013]()
(1-((S)-2-((S)-2-Aminopentanamido)propanamido)ethyl) phosphonic acid or l-Nva-l-Ala-d/l-Fos (
21a). The
tert-butoxycarbonyl and diethyl ester protecting groups of
tert-butyl ((2
S)-1-(((2
S)-1-((1-(diethoxyphosphoryl)ethyl)amino)-1-oxopropan-2-yl)amino)-1-oxopentan-2-yl)carbamate (
18a) (1.6 mmol, 0.72 g) were removed. The pale green crude solid was recrystallised from hot water/acetone to give
21a as a pale green solid composed of 2 diastereoisomers,
l-Nva-
l-Ala-
l-Fos and
l-Nva-
l-Ala-D-Fos (0.22 g, 0.75 mmol, 47%); m.p. 207–210 °C (decomp.);
ῡmax/cm
−1 3280 (NH
+), 3500-2900 (br OH), 1643 (br C=O), 1552 (NH bend), 1149 (P=O), 1037 (P-O-C), 922 (P-OH);
1H NMR (300 MHz, D
2O) δ
H 0.96 (3H, t,
3JH-H = 7.1 Hz, CH
3-10″), 1.27-1.32 (3H, d,
3JH-H = 6.8 Hz, C
H3-2), 1.40-1.42 (3H, m, C
H3-6), 1.40–1.42 (2H, m, C
H2-10′), 1.88–1.86 (2H, m, C
H2-10), 4.00–4.02 (2H, m, C
H-1, C
H-9), 4.34–4.39 (1H, m, C
H-5);
13C NMR (75 MHz, D
2O) δ
C 13.4 (
CH
3-10″), 16.0 (
CH
3-2), 17.1 (
CH
3-6), 17.2 (
CH
3-6), 18.1 (
CH
2-10′), 18.2 (
CH
2-10′), 33.5 (
CH
2-10), 33.6 (
CH
2-10), 50.5 (
CH-5), 50.8 (
CH-5), 53.5 (
CH-1,
CH-9), 170.4 (
C=O-8), 170.6 (
C=O-8),174.7 (
C=O-4);
31P-
1H
decoup NMR (121 MHz, CDCl
3) δ
P 18.5; HRMS (NSI) calcd for (C
10H
23N
3O
5P)
+, MH
+: 296.1370, found 296.1373. LCMS purity >95% (C-18 reversed phase, MeOH-H
2O). (The LCMS chromatogram and conditions may be found within the
Supplementary Materials).
![Molecules 25 01557 i014 Molecules 25 01557 i014]()
(1-((S)-2-(2-(Methylamino)acetamido)propanamido)ethyl) phosphonic acid or Sar-l-Ala-d/l-Fos (
21b). The
tert-butoxycarbonyl and diethyl ester protecting groups of (1-((
S)-2-((
S)-2-aminopropanamido)propanamido)ethyl)phosphonic acid (
18b) (3.3 mmol, 1.40 g) were removed. The pale green crude solid was recrystallised from hot water/ethanol to give
21b as a pale green solid composed of 2 diastereoisomers, Sar-
l-Ala-
l-Fos and Sar-
l-Ala-D-Fos (0.45 g, 1.7 mmol, 51%); m.p. 241–245 °C (decomp.);
ῡmax/cm
−1 3289 (NH
+), 3500–2900 (br OH), 1632 (br C=O), 1556 (NH bend), 1174 (P=O), 1059 (P-O-C), 919 (P-OH);
1H NMR (300 MHz, D
2O) δ
H 1.14–1.57 (6H, m, C
H3-2, C
H3-6), 2.74 (3H, s, C
H3-11), 3.84–4.07 (3H, m, C
H2-9, C
H-1), 4.32–4.58 (1H, m, C
H-5);
13C NMR (75 MHz, D
2O) δ
C 15.4 (
CH
3-2), 16.8 (
CH
3-6), 32.9 (
CH
3-11), 43.9 (d,
1JP-C = 148.5 Hz,
CH-1), 49.4 (
CH
2-9), 50.0 (
CH-5), 166.0 (
C=O-8), 173.7 (
C=O-4);
31P-
1H
decoup NMR (121 MHz, CDCl
3) δ
P 19.2; HRMS (NSI) calcd for (C
8H
19N
3O
5P)
+, MH
+: 268.1057, found 268.1016; LCMS purity >95% (C-18 reversed phase, MeOH–H
2O). (The LCMS chromatogram and conditions may be found within the
Supplementary Materials).
![Molecules 25 01557 i015 Molecules 25 01557 i015]()
(1-((S)-2-((S)-2-Amino-4-(methylthio)butanamido) propanamido)ethyl)phosphonic acid or l-Met-l-Ala-d/l-Fos (
21c). The
tert-butoxycarbonyl and diethyl ester protecting groups of
tert-butyl ((2
S)-1-(((2
S)-1-((1-(diethoxyphosphoryl)ethyl)amino)-1-oxopropan-2-yl)amino-4-(methylthio)-1-oxobutan-2-yl)carbamate (
18c) (0.9 mmol, 0.43 g) were removed. The green crude solid was recrystallised from hot water/ethanol to give
21c as a pale green solid composed of 2 diastereoisomers,
l-Met-
l-Ala-
l-Fos and
l-Met-
l-Ala-D-Fos (0.13 g, 0.41 mmol, 46%); m.p. 214–217 °C (decomp.);
ῡmax/cm
−1 3263 (NH
+), 2834 (broad OH), 1641 (br C=O), 1552 (NH bend), 1150 (P=O), 1041 (P-O-C), 919 (P-OH);
1H NMR (300 MHz, D
2O) δ
H 1.29–1.33 (3H, m, C
H3-2), 1.42 (3H, d,
3JH-H = 6.0 Hz, C
H3-6), 2.15 (3H, s, C
H3-10″), 2.20 (2H, m, C
H2-10), 2.62 (2H, m, C
H2-10′), 4.05 (1H, m, C
H-1), 4.14 (1H, m, C
H-9), 4.39–4.41 (1H, m, C
H-5);
13C NMR (75 MHz, D
2O) δ
C 16.9 (
CH
3-10″), 18.4 (
CH
3-2), 19.5 (
CH
3-6), 19.6 (
CH
3-6), 32.8 (
CH
2-10′), 33.0 (
CH
2-10′), 30.7 (
CH
2-10), 31.0 (
CH
2-10), 44.4 (
CH-1), 52.9 (
CH-5), 53.0 (
CH-5), 55.0 (
CH-9), 176.1 (
C=O-4,
C=O-8);
31P-
1H
decoup NMR (121 MHz, CDCl
3) δ
P 20.7; HRMS (NSI) calcd for (C
10H
23N
3O
5PS)
+, MH
+: 328.1091, found 328.1094; LCMS purity >95% (C-18 reversed phase, MeOH-H
2O). (The LCMS chromatogram and conditions may be found within the
Supplementary Materials).
![Molecules 25 01557 i016 Molecules 25 01557 i016]()
(S)-Benzyl 2-((tert-butoxycarbonyl)amino)-3-hydroxy propanoate or Boc-l-Ser-OBzl (11). Boc-
l-Serine (
10) (20 mmol, 4.10 g) and 1,8-diazabicyclo [5.4.0]undec-7-ene (DBU) (30 mmol, 4.5 mL) were dissolved in a round-bottom flask containing dry benzene (80 mL), followed by the addition of benzyl bromide (30 mmol, 3.60 mL). Caution: Benzene is a known carcinogen. The solution was stirred overnight at room temperature under nitrogen and later the solvent was removed under reduced pressure to afford an off-white residue. Ethyl acetate (100 mL) was added, the flask contents were sonicated and then washed with 1M HCl (50 mL) and brine (2 × 50 mL). The organic layer was dried over MgSO
4, filtered, concentrated
in vacuo and purified by column chromatography (petrol/ethyl acetate (1:1)) to give
11 as a white solid (5.24 g, 17.7 mmol, 89%); m.p. 61–63 °C (lit. m.p. 59–60 °C) [
30];
ῡmax/cm
−1 3416 (NH, OH), 1756 (C=O), 1666 (C=O), 1522 (NH bend), 1200 (C-O), 1154 (C-O);
1H NMR (300 MHz, CDCl
3) δ
H 1.36 (9H, s, C(C
H3)
3), 2.17 (1H, br, O
H), 3.82 (1H, dd,
2JH-H = 11.1 Hz,
3JH-H = 3.6 Hz, C
Ha/b-3), 3.90 (1H, dd,
2JH-H = 11.1 Hz,
3JH-H = 3.9 Hz, C
Ha/b-3), 4.33 (1H, m, C
H-2), 5.11 (1H, d,
2JH-H = 12.3 Hz, OC
Ha/bAr), 5.16 (1H, d,
2JH-H = 12.3 Hz, OC
Ha/bAr), 5.40 (1H, br, N
H-4), 7.27 (5H, m, 5 × C
HAr);
13C NMR (75 MHz, CDCl
3) δ
C 27.1 (C(
CH
3)
3), 54.7 (
CH-2), 62.3 (
CH
2-3), 66.2 (O
CH
2Ar), 79.1 (
C(CH
3)
3), 127.0 (2 ×
CH
Ar), 127.3 (
CH
Ar), 127.4 (2 ×
CH
Ar), 134.1 (
CH
Ar quat.), 153.0 (
C=O-5), 170.7 (
C=O-1);
m/z (ESI) calcd for (C
15H
21NNaO
5)
+, MNa
+: 318.3, found 318.2. (The
1H-NMR spectrum may be found within the
Supplementary Materials).
![Molecules 25 01557 i017 Molecules 25 01557 i017]()
(R)-Benzyl 2-((tert-butoxycarbonyl)amino)-3-chloropropanoate or Boc-β-Cl-l-Ala-OBzl (
12). (
S)-Benzyl 2-((
tert-butoxycarbonyl)amino)-3-hydroxypropanoate (
11) (15 mmol, 4.43 g) was dissolved in dry DCM (40 mL), followed by the addition of trichloroacetonitrile (30 mmol, 3 ml). The solution was stirred at room temperature for 2 h. To this solution, triphenylphosphine (30 mmol, 7.87 g) in dry DCM (50 mL) was added slowly. The resulting solution was stirred overnight at room temperature under nitrogen; brine (100 mL) was added to quench the reaction. The organic layer was washed with brine (3 × 100 mL), dried over MgSO
4, filtered and concentrated in vacuo to afford an orange residue. The residue was purified by column chromatography (petrol/ethyl acetate (7:3)) to give
12 as a white solid (3.53 g, 11.2 mmol, 75%); m.p. 55–58 °C;
ῡmax/cm
−1 3364 (NH), 1725 (C=O), 1680 (C=O), 1519 (NH bend), 1208 (C-O), 1158 (C-O);
1H NMR (300 MHz, CDCl
3) δ
H 1.38 (9H, s, C(C
H3)
3), 3.78 (1H, dd,
2JH-H = 11.2 Hz,
3JH-H = 3.2 Hz, C
Ha/b-3), 3.92 (1H, dd,
2JH-H = 11.3 Hz,
3JH-H = 3.0 Hz, C
Ha/b-3), 4.67 (1H, m, C
H-2), 5.13 (1H, d,
2JH-H = 12.2 Hz, OC
Ha/bAr), 5.18 (1H, d,
2JH-H = 12.2 Hz, OC
Ha/bAr), 5.37 (1H, d,
3JH-H = 7.5 Hz, N
H-4), 7.29 (5H, m, 5 × C
HAr);
13C NMR (75 MHz, CDCl
3) δ
C 28.3 (C(
CH
3)
3), 45.5 (
CH
2-3), 54.5 (
CH-2), 67.8 (O
CH
2Ar), 80.5 (
C(CH
3)
3), 128.4 (
CH
Ar), 128.6 (
CH
Ar), 128.7 (
CH
Ar), 134.9 (
CH
Ar quat.), 155.0 (
C=O-5), 169.0 (
C=O-1);
m/z (ESI) calcd for (C
15H
20ClNNaO
4)
+, MNa
+: 336.1 (
35Cl), 338.1 (
37Cl), found 336.2 (
35Cl), 338.2 (
37Cl); CHN (Found: C, 57.71; H, 6.46; N, 4.38. C
15H
20ClNO
4 requires C, 57.42; H, 6.42; N, 4.46%). (The mass spectrum may be found within the
Supplementary Materials).
![Molecules 25 01557 i018 Molecules 25 01557 i018]()
(R)-2-((Tert-butoxycarbonyl)amino)-3-chloropropanoic acid or Boc-β-Cl-l-Ala-OH (
13). Deprotection of benzyl ester was followed, using (
R)-benzyl 2-((
tert-butoxycarbonyl)amino)-3-chloropropanoate (
12) (7.0 mmol, 2.20 g) to afford
13 as an off-white solid (1.52 g, 6.78 mmol, 97%); m.p. 125–128 °C (lit. m.p. 127–129 °C) [
15];
ῡmax/cm
−1 3434 (NH), 2973 (br OH), 1752 (C=O), 1735 (C=O), 1519 (NH bend), 1159 (C-O), 1148 (C-O);
1H NMR (300 MHz, CDCl
3) δ
H 1.40 (9H, s, C(C
H3)
3), 3.80 (1H, dd,
2JH-H = 12.0 Hz,
3JH-H = 3.0 Hz, C
Ha/b-3), 3.95 (1H, dd,
2JH-H = 12.0 Hz,
3JH-H = 3.0 Hz, C
Ha/b-3), 4.70 (1H, m, C
H-2), 5.42 (1H, d,
3JH-H = 7.2 Hz, N
H-4), 9.03 (1H, br, O
H);
13C NMR (75 MHz, CDCl
3) δ
C 27.1 (C(
CH
3)
3), 44.0 (
CH
2-3), 53.1 (
CH-2), 79.8 (
C(CH
3)
3), 154.2 (
C=O-5), 172.1 (
C=O-1);
m/z (ESI) calcd for (C
8H
14ClNNaO
4)
+, MNa
+: 246.1 (
35Cl), 248.1 (
37Cl), found 246.1 (
35Cl), 248.1 (
37Cl).
![Molecules 25 01557 i019 Molecules 25 01557 i019]()
(R)-1-(Benzyloxy)-3-chloro-1oxopropan-2-aminium chloride or β-Cl-l-Ala-OBzl hydrochloride (14). Deprotection of tert-butoxycarbonyl was followed, using (R)-benzyl 2-((tert-butoxycarbonyl)amino)-3-chloropropanoate (12) (15 mmol, 4.71 g). The white crude solid was filtered and washed by diethyl ether to give 14 as a white solid (3.47 g, 13.4 mmol, 93%); m.p. 145 °C (sub); ῡmax/cm−1 2841 (NH+), 1750 (C=O), 1231 (C-O); 1H NMR (300 MHz, D2O) δH 4.06 (1H, dd, 2JH-H = 15.0 Hz, 3JH-H = 6.0 Hz, CHa/b-3), 4.20 (1H, dd, 2JH-H = 15.0 Hz, 3JH-H = 6.0 Hz, CHa/b-3), 4.70 (1H, t, 3JH-H = 6.0 Hz, CH-2), 5.29 (1H, d, 2JH-H = 12.0 Hz, OCHa/bAr), 5.37 (1H, d, 2JH-H = 12.0 Hz, OCHa/bAr), 7.42-7.47 (5H, m, 5 × CHAr); 13C NMR (75 MHz, D2O) δC 41.8 (CH2-3), 54.0 (CH-2), 69.1 (OCH2Ar), 128.6–129.1 (CHAr), 134.5 (CHAr quart.), 167.0 (C=O-1); m/z (ESI) calcd for (C10H13ClNO2), M+: 214.1 (35Cl), 216.1 (37Cl), found 214.1 (35Cl), 216.1 (37Cl); CHN (Found: C, 47.16; H, 5.43; N, 5.43. C10H13Cl2NO2∙0.2H2O requires C, 47.34; H, 5.32; N, 5.52%).
![Molecules 25 01557 i020 Molecules 25 01557 i020]()
(R)-Benzyl 2-((S)-2-((tert-butoxycarbonyl)amino)pentanamido)-3-chloropropanoate or Boc-l-Nva-β-chloro-l-Ala-OBzl (22a). General peptide coupling method was followed, using Boc-l-Nva-OH (15a) (6.0 mmol, 1.31 g) in dry THF and (R)-1-(benzyloxy)-3-chloro-1oxopropan-2-aminium chloride (14) (5.4 mmol, 1.36 g) in dry DCM. The yellow crude liquid was purified by column chromatography (40-60 petrol/ethyl acetate (7:3)) to give 22a as a white solid (1.74 g, 4.2 mmol, 78%); m.p. 95–98 °C; ῡmax/cm−1 3327 (NH), 1743 (C=O), 1688 (C=O), 1653 (C=O), 1518 (NH bend), 1206 (C-O), 1169 (C-O); 1H NMR (300 MHz, CDCl3) δH 0.92 (3H, t, 3JH-H = 9.0 Hz, CH3-7″), 1.32-1.43 (2H, m, CH2-7′), 1.45 (9H, s, C(CH3)3), 1.52–1.65 (1H, m, CHa/b-7), 1.75-1.82 (1H, m, CHa/b-7), 3.89 (1H, dd, 2JH-H = 12.0 Hz, 3JH-H = 3.0 Hz, CHa/b-3), 3.99 (1H, dd, 2JH-H = 12.0 Hz, 3JH-H = 3.0 Hz, CHa/b-3), 4.11–4.15 (1H, m, CH-6), 4.96–5.00 (2H, m, CH-2, NH-8), 5.20 (1H, d, 2JH-H = 12.0 Hz, OCHa/bAr), 5.25 (1H, d, 2JH-H = 12.0 Hz, OCHa/bAr), 6.97 (1H, d, 3JH-H = 6.0 Hz, NH-4), 7.33–7.37 (5H, m, 5 x CHAr); 13C NMR (75 MHz, CDCl3) δC 12.7 (CH3-7″), 17.8 (CH2-7′), 27.3 (C(CH3)3), 33.4 (CH2-7), 43.8 (CH2-3), 52.2 (CH-2), 53.4 (CH-6), 67.0 (OCH2Ar), 79.3 (C(CH3)3), 127.4 (CHAr), 127.6 (CHAr), 127.7 (CHAr), 133.8 (CHAr quat.), 154.5 (C=O-9), 167.5 (C=O-1), 171.2 (C=O-5); HRMS (NSI) calcd for (C20H30ClN2O5)+, MH+: 413.1838 (35Cl), 415.1809 (37Cl), found 413.1837 (35Cl), 415.1807 (37Cl); CHN (Found: C, 58.49; H, 7.22; N, 6.81. C20H29ClN2O5 requires C, 58.18; H, 7.08; N, 6.78%).
![Molecules 25 01557 i021 Molecules 25 01557 i021]()
(R)-Benzyl 2-(2-((tert-butoxycarbonyl)(methyl)amino) acetamido)-3-chloropropanoate or Boc-Sar-β-chloro-l-Ala-OBzl (22b). General peptide coupling method was followed, using Boc-Sar-OH (15b) (13.0 mmol, 2.46 g) in dry THF and (R)-1-(benzyloxy)-3-chloro-1oxopropan-2-aminium chloride (14) (13.3 mmol, 3.34 g) in dry DCM. The yellow crude liquid was purified by column chromatography (40–60 petrol/ethyl acetate (7:3)) to give 22b as a light yellow syrup (3.63 g, 9.4 mmol, 73%); ῡmax/cm−1 3302 (NH), 1747 (C=O), 1686 (br C=O), 1522 (NH bend), 1175 (C-O), 1148 (C-O); 1H NMR (300 MHz, CDCl3) δH 1.40 (9H, s, C(CH3)3), 2.87 (3H, s, NCH3-8), 3.80 (1H, d, 2JH-H = 15.0 Hz, CHa/b-6), 3.82 (1H, d, 2JH-H = 15.0 Hz, CHa/b-6), 3.83 (1H, dd, 2JH-H = 12.0 Hz, 3JH-H = 3.0 Hz, CHa/b-3), 3.94 (1H, dd, 2JH-H = 12.0 Hz, 3JH-H = 3.0 Hz, CHa/b-3), 4.91–4.96 (1H, m, CH-2), 5.13 (1H, d, 2JH-H = 12.0 Hz, OCHa/bAr), 5.18 (1H, d, 2JH-H = 12.0 Hz, OCHa/bAr), 6.97 (1H, d, 3JH-H = 6.0 Hz, NH-4), 7.26–7.30 (5H, m, 5 x CHAr); 13C NMR (75 MHz, CDCl3) δC 28.2 (C(CH3)3), 35.6 (NCH3-8), 44.9 (CH2-3), 53.0 (CH2-6), 53.0 (CH-2), 68.0 (OCH2Ar), 81.0 (C(CH3)3), 128.4 (CHAr), 128.6 (CHAr), 128.7 (CHAr), 134.8 (CHAr quat.), 154.5 (C=O-9), 168.4 (C=O-1), 169.4 (C=O-5); HRMS (NSI) calcd for (C18H26ClN2O5)+, MH+: 385.1525 (35Cl), 387.1496 (37Cl), found 385.1527 (35Cl), 387.1498 (37Cl). LCMS purity >95% (C-18 reversed phase, MeOH–H2O).
![Molecules 25 01557 i022 Molecules 25 01557 i022]()
Tert-butyl ((2R)-3-chloro-1-((1-(diethoxyphosphoryl)ethyl) amino)-1-oxopropan-2-yl)carbamate or Boc-β-chloro-l-Ala-d/l-Fos diethyl ester (19e). General peptide coupling method was followed, using (R)-2-((tert-butoxycarbonyl)amino)-3-chloropropanoic acid (13) (6.0 mmol, 1.34 g) in dry THF and diethyl 1-aminoethylphosphonate (9) (6.0 mmol, 1.09 g) in dry THF. The light yellow crude liquid was purified by column chromatography, using 100% petrol and increasing to 100% ethyl acetate, to afford 19e as colorless syrup composed of 2 diastereoisomers, Boc-β-Cl-l-Ala-l-Fos diethyl ester and Boc-β-Cl-l-Ala-D-Fos diethyl ester (2.03 g, 5.2 mmol, 88%); ῡmax/cm−1 3261 (NH), 1713 (C=O), 1670 (C=O), 1517 (NH bend), 1225 (P=O), 1164 (C-O), 1020 (P-O-C), 970 (P-O-C); 1H NMR (300 MHz, CDCl3) δH 1.15–1.46 (9H, m, CH3-2, 2 × OCH2CH3), 1.47 (9H, s, C(CH3)3), 3.74 (1H, dd, 2JH-H = 12.0 Hz, 3JH-H = 6.0 Hz, CHa/b-6), 4.00 (1H, dd, 2JH-H = 12.0 Hz, 3JH-H = 6.0 Hz, CHa/b-6), 4.06–4.22 (4H, m, 2 × OCH2CH3), 4.40–4.56 (2H, m, CH-1, CH-5), 5.46 (0.5H, d, 3JH-H = 6.0 Hz, NH-3 or NH-7), 5.48 (0.5H, d, 3JH-H = 9.0 Hz, NH-3 or NH-7), 7.01 (0.5H, m, NH-3 or NH-7), 7.09 (0.5H, m, NH-3 or NH-7); 13C NMR (75 MHz, CDCl3) δC 15.6 (CH3-2), 15.7 (CH3-2), 16.3 (d, 3JC-P = 1.5 Hz, OCH2CH3), 16.4 (d, 3JC-P = 2.3 Hz, OCH2CH3), 16.4 (d, 3JC-P = 1.5 Hz, OCH2CH3), 16.4 (d, 3JC-P = 2.3 Hz, OCH2CH3), 28.2 (C(CH3)3), 41.2 (d, 1JC-P = 157.5 Hz, CH-1), 41.3 (d, 1JC-P = 157.5 Hz, CH-1), 55.2 (CH2-6), 55.3 (CH-5), 62.6 (d, 2JC-P = 6.8 Hz, OCH2CH3), 62.6 (d, 2JC-P = 6.8 Hz, OCH2CH3), 63.0 (d, 2JC-P = 6.8 Hz, OCH2CH3), 63.0 (d, 2JC-P = 6.8 Hz, OCH2CH3), 80.8 (C(CH3)3), 155.0 (C=O-8), 168.3 (C=O-4); 31P-1Hdecoup NMR (121 MHz, CDCl3) δP 24.7, 24.8; HRMS (NSI) calcd for (C14H28ClN2O6P), MH+: 409.1266 (35Cl), 411.1237 (37Cl), found 409.1258 (35Cl), 411.1231 (37Cl). LCMS purity >95% (C-18 reversed phase, MeOH-H2O).
![Molecules 25 01557 i023 Molecules 25 01557 i023]()
(R)-2-((S)-2-((tert-Butoxycarbonyl)amino)pentanamido)-3-chloropropanoic acid or Boc-l-Nva-β-chloro-l-Ala-OH (23a). Deprotection of benzyl ester was followed, using (R)-benzyl 2-((S)-2-((tert-butoxycarbonyl)amino)pentanamido)-3-chloropropanoate (22a) (5.8 mmol, 2.41 g) to afford 23a as a light yellow solid (1.81 g, 5.61 mmol, 96%); m.p. 60–63 °C; ῡmax/cm−1 3312 (br OH), 2963 (NH), 1655 (br C=O), 1509 (NH bend), 1161 (C-O); 1H NMR (300 MHz, DMSO) δH 0.85 (3H, t, 3JH-H = 9.0 Hz, CH3-7″), 1.24–1.34 (2H, m, CH2-7′), 1.38 (9H. s, C(CH3)3), 1.42–1.52 (1H, m, CHa/b-7), 1.54–1.59 (1H, m, CHa/b-7), 3.34 (1H, br, OH), 3.84 (1H, dd, 2JH-H = 12.0 Hz, 3JH-H = 6.0 Hz, CHa/b-3), 3.91 (1H, dd, 2JH-H = 12.0 Hz, 3JH-H = 6.0 Hz, CHa/b-3), 3.95–4.02 (1H, m, CH-6), 4.62–4.67 (1H, m, CH-2), 6.92 (1H, d, 3JH-H = 9.0 Hz, NH-8), 8.07 (1H, d, 3JH-H = 9.0 Hz, NH-4); 13C NMR (75 MHz, CDCl3) δC 14.1 (CH3-7″), 19.1 (CH2-7′), 28.6 (C(CH3)3), 34.4 (CH2-7), 45.1 (CH2-3), 53.6 (CH-2), 54.5 (CH-6), 78.5 (C(CH3)3), 155.8 (C=O-9), 170.6 (C=O-5), 173.0 (C=O-1); m/z (ESI) calcd for (C13H23ClN2NaO5)+, MNa+: 345.1 (35Cl), 347.1 (37Cl), found 345.2 (35Cl), 347.2 (37Cl); CHN (Found: C, 48.67; H, 7.51; N, 8.42. C13H23ClN2O5 requires C, 48.37; H, 7.18; N, 8.68%).
![Molecules 25 01557 i024 Molecules 25 01557 i024]()
(R)-2-(2-((tert-butoxycarbonyl)(methyl)amino)acetamido)-3-chloropropanoic acid or Boc-Sar-β-chloro-l-Ala-OH (23b). Deprotection of benzyl ester was followed, using (R)-benzyl 2-(2-((tert-butoxycarbonyl)(methyl)amino)acetamido)-3-chloropropanoate (22b) (6.2 mmol, 2.40 g) to afford 23b as an off-white solid (1.81 g, 6.1 mmol, 99%); m.p. 89–91 °C; ῡmax/cm−1 3342 (NH), 2982 (br OH), 1734 (C=O), 1672 (C=O), 1644 (C=O), 1524 (NH bend), 1152 (C-O); 1H NMR (300 MHz, CDCl3) δH 1.47 (9H. s, C(CH3)3), 2.99 (3H, s, NCH3-8), 3.81–4.17 (4H, m, CH2-6, CH2-3), 5.01 (1H, m, CH-2), 7.07 (1H, br, NH-4), 7.45 (1H, br, OH); 13C NMR (75 MHz, CDCl3) δC 28.3 (C(CH3)3), 36.1 (NCH3-8), 44.5 (CH2-3), 53.0 (CH2-6 and CH-2), 81.8 (C(CH3)3), 156.8 (C=O-9), 169.5 (C=O-1 and C=O-5); m/z (ESI) calcd for (C11H19ClN2NaO5)+, MNa+: 317.1 (35Cl), 319.1 (37Cl), found 317.1 (35Cl), 319.1 (37Cl); CHN (Found: C, 43.98; H, 6.69; N, 9.53. C11H19ClN2O5·0.3H2O requires C, 44.02; H, 6.58; N, 9.33%).
![Molecules 25 01557 i025 Molecules 25 01557 i025]()
(2R)-3-Chloro-1-((1-(diethoxyphosphoryl)ethyl)amino)-1-oxopropan-2-aminium chloride or β-Cl-l-Ala-d/l-Fos diethyl ester hydrochloride (20e). Deprotection of tert-butoxycarbonyl was followed, using ((2R)-3-chloro-1-((1-(diethoxyphosphoryl)ethyl)amino)-1-oxopropan-2-yl)carbamate (19e) (6.7 mmol, 2.59 g). The off-white hygroscopic crude solid was washed with petrol to afford 20e as a pale green solid composed of 2 diastereoisomers, β-Cl-l-Ala-l-Fos diethyl ester hydrochloride and β-Cl-l-Ala-D-Fos diethyl ester hydrochloride (1.51 g, 4.7 mmol, 70%); m.p. 129–133 °C (decomp.); ῡmax/cm−1 3204 (NH+), 1687 (C=O), 1562 (NH bend), 1204 (P=O), 1010 (P-O-C), 961 (P-O-C); 1H NMR (300 MHz, D2O) δH 1.28 (3H, t, 3JH-H = 6.0 Hz, OCH2CH3), 1.29 (3H, t, 3JH-H = 6.0 Hz, OCH2CH3), 1.37 (3H, dd, 3JP-H = 18.0 Hz, 3JH-H = 6.0 Hz, CH3-2), 3.92–4.04 (2H, m, CH2-6), 4.07-4.21 (4H, m, 2 × OCH2CH3), 4.38–4.48 (2H, m, CH-1, CH-5); 13C NMR (75 MHz, D2O) δC 13.7 (CH3-2), 14.0 (CH3-2), 15.7 (OCH2CH3), 15.7 (OCH2CH3), 41.7 (d, 1JP-C = 158.3 Hz, CH-1), 42.0 (d, 1JP-C = 157.5 Hz, CH-1), 42.4 (CH2-6), 53.7 (CH-5), 53.8 (CH-5), 64.3 (d, 2JP-C = 6.8 Hz, OCH2CH3), 64.5 (d, 2JP-C = 6.8 Hz, OCH2CH3), 165.7 (C=O-4), 165.8 (C=O-4); 31P-1Hdecoup NMR (121 MHz, CDCl3) δP 26.1, 26.2; HRMS (NSI) calcd for (C9H21ClN2O4P)+, M+: 287.0922 (35Cl), 289.0892 (37Cl), found 287.0922 (35Cl), 289.0890 (37Cl). LCMS purity >95% (C-18 reversed phase, MeOH–H2O).
![Molecules 25 01557 i026 Molecules 25 01557 i026]()
Tert-butyl ((2S)-1-(((2R)-3-chloro-1-((1-(diethoxyphosphoryl)-ethyl)amino)-1-oxopropan-2-yl)amino)-1-oxopentan-2-yl)carbamate or Boc-l-Nva-β-chloro-l-Ala-d/l-Fos diethyl ester (24a). General peptide coupling method was followed, using (R)-2-((S)-2-((tert-butoxycarbonyl)amino)pentanamido)-3-chloropropanoic acid (23a) (1.8 mmol, 0.58 g) in dry THF and diethyl 1-aminoethylphosphonate (9) (1.8 mmol, 0.33 g) in dry THF. The light yellow crude liquid was purified by column chromatography, using ethyl acetate/methanol (96:4), to afford 24a as a white solid composed of 2 diastereoisomers, Boc-l-Nva-β-Cl-l-Ala-l-Fos diethyl ester and Boc-l-Nva-β-Cl-l-Ala-D-Fos diethyl ester (0.45 g, 0.93 mmol, 52%); m.p. 196 °C (decomp); ῡmax/cm−1 3272 (NH), 1709 (C=O), 1680 (C=O), 1644 (C=O), 1530 (NH bend), 1229 (P=O), 1165 (C-O), 1019 (P-O-C), 972 (P-O-C); 1H NMR (300 MHz, CDCl3) δH 0.86 (1.5H, t, 3JH-H = 9.0 Hz, CH3-10″), 0.88 (1.5H, t, 3JH-H = 9.0 Hz, CH3-10″), 1.22–1.34 (11H, m, 2 × OCH2CH3, CH3-2, CH2-10′), 1.38 (9H, s, C(CH3)3), 1.53-1.59 (1H, m, CHa/b-10), 1.70–1.77 (1H, m, CHa/b-10), 3.69 (1H, dd, 2JH-H = 12.0 Hz, 3JH-H = 6.0 Hz, CHa/b-6), 3.78-3.81 (1H, m, CH-9), 3.91 (1H, dd, 2JH-H = 12.0 Hz, 3JH-H = 6.0 Hz, CHa/b-6), 3.97-4.13 (4H, m, 2 x OCH2CH3), 4.35-4.46 (1H, m, CH-1), 4.73-4.79 (1H, m, CH-5), 4.97–5.03 (1H, m, NH-11), 7.01 (0.5H, d, 3JH-H = 9.0 Hz, NH-7), 7.09 (0.5H, d, 3JH-H = 9.0 Hz, NH-7), 7.25 (0.5H, d, 3JH-H = 9.0 Hz, NH-3), 7.33 (0.5H, d, 3JH-H = 9.0 Hz, NH-3); 13C NMR (75 MHz, CDCl3) δC 12.7 (CH3-10″), 14.5 (CH3-2), 15.3 (OCH2CH3), 15.4 (OCH2CH3), 15.5 (OCH2CH3), 15.6 (OCH2CH3), 17.9 (CH2-10′), 18.0 (CH2-10′), 27.0 (C(CH3)3), 27.3 (C(CH3)3), 33.2 (CH2-10), 40.4 (d, 1JC-P = 157.5 Hz, CH-1), 43.4 (CH2-6), 52.6 (CH-5), 52.8 (CH-5), 61.4 (d, 2JC-P = 6.8 Hz, OCH2CH3), 61.6 (d, 2JC-P = 6.8 Hz, OCH2CH3), 61.7 (d, 2JC-P = 6.8 Hz, OCH2CH3), 61.9 (d, 2JC-P = 6.8 Hz, OCH2CH3), 70.5 (CH-9), 79.4 (C(CH3)3), 154.7 (C=O-12), 166.8 (C=O-4), 171.4 (C=O-8); 31P-1Hdecoup NMR (121 MHz, CDCl3) δP 24.9, 25.0; HRMS (NSI) calcd for (C19H38ClN3O7P)+, MH+: 486.2130 (35Cl), 488.2102 (37Cl), found 486.2124 (35Cl), 488.2098 (37Cl); CHN (Found: C, 46.61; H, 7.76; N, 8.31. C19H37ClN3O7P requires C, 46.96; H, 7.67; N, 8.65%).
![Molecules 25 01557 i027 Molecules 25 01557 i027]()
Tert-butyl (2-(((2R)-3-chloro-1-((1-(diethoxyphosphoryl)ethyl) amino)-1-oxopropan-2-yl)amino)-2-oxoethyl)(methyl)carbamate or Boc-Sar-β-chloro-l-Ala-d/l-Fos diethyl ester (24b). General peptide coupling method was followed, using (R)-2-(2-((tert-butoxycarbonyl)(methyl)amino)acetamido)-3-chloropropanoic acid (23b) (5.5 mmol, 1.61 g) in dry THF and diethyl 1-aminoethylphosphonate (9) (6.0 mmol, 1.09 g) in dry THF. The light yellow crude liquid was purified by column chromatography, using DCM/methanol (95:5), to afford 24b as a light yellow syrup composed of 2 diastereoisomers, Boc-Sar-β-Cl-l-Ala-l-Fos diethyl ester and Boc-Sar-β-Cl-l-Ala-D-Fos diethyl ester (1.93 g, 4.21 mmol, 76%); ῡmax/cm−1 3218 (NH), 1690 (C=O), 1665 (br C=O), 1518 (NH bend), 1224 (P=O), 1148 (C-O), 1018 (P-O-C), 967 (P-O-C); 1H NMR (300 MHz, CDCl3) δH 1.11–1.35 (9H, m, 2 × OCH2CH3, CH3-2), 1.41 (9H, s, C(CH3)3), 2.90 (3H, s, CH3-11), 3.70–3.88 (4H, m, CH2-6, CH2-9), 4.02–4.13 (4H, m, 2 × OCH2CH3), 4.36–4.47 (1H, m, CH-1), 4.78–4.82 (1H, m, CH-5), 6.94 (1H, m, NH-7), 7.36 (1H, m, NH-3); 13C NMR (75 MHz, CDCl3) δC 15.2 (CH3-2), 15.6 (CH3-2), 16.3 (OCH2CH3), 16.4 (OCH2CH3), 28.3 (C(CH3)3), 35.9 (CH3-11), 41.2 (d, 1JC-P = 156.8 Hz, CH-1), 44.7 (CH2-6), 53.1 (CH2-9), 53.4 (CH-5), 62.7 (d, 2JC-P = 6.0 Hz, OCH2CH3), 63.0 (d, 2JC-P = 7.5 Hz, OCH2CH3), 81.0 (C(CH3)3), 152.3 (C=O-12), 167.7 (C=O-4 or C=O-8), 169.4 (C=O-4 or C=O-8); 31P-1Hdecoup NMR (121 MHz, CDCl3) δP 24.7, 24.8; HRMS (NSI) calcd for (C16H34ClN3O7P)+, MH+: 480.1637 (35Cl), 482.1608 (37Cl), found 480.1642 (35Cl), 482.1612 (37Cl). LCMS purity >92% (C-18 reversed phase, MeOH-H2O).
![Molecules 25 01557 i028 Molecules 25 01557 i028]()
Tert-butyl ((2S)-1-(((2R)-3-chloro-1-((1-(diethoxyphosphoryl) ethyl)amino)-1-oxopropan-2-yl)amino)-4-(methylthio)-1-oxobutan-2-yl) carbamate or Boc-l-Met-β-Cl-l-Ala-d/l-Fos diethyl ester (24c). General peptide coupling method was followed, using Boc-l-Met-OH (15c) (3.4 mmol, 0.85 g) in dry THF and (2R)-3-chloro-1-((1-(diethoxyphosphoryl)ethyl)amino)-1-oxopropan-2-aminium chloride (20e) (3.4 mmol, 1.10 g) in dry DCM. The yellow crude liquid was purified by column chromatography (DCM/MeOH (95:5)) and recrystallized from diethyl ether/petrol to give 24c as a white solid composed of 2 diastereoisomers, Boc-l-Met-β-Cl-l-Ala-l-Fos diethyl ester and Boc-l-Met-β-Cl-l-Ala-D-Fos diethyl ester (0.88 g, 1.7 mmol, 50%); m.p. 96–99 °C; ῡmax/cm−1 3278 (NH), 1709 (C=O), 1687 (C=O), 1639 (C=O), 1523 (NH bend), 1228 (P=O), 1165 (C-O), 1018 (P-O-C), 970 (P-O-C); 1H NMR (300 MHz, CDCl3) δH 1.17-1.36 (9H, m, CH3-2, 2 × OCH2CH3), 1.38 (9H, s, C(CH3)3), 1.87–2.03 (2H, m, CH2-10), 2.04 (3H, s, CH3-10″), 2.48–2.54 (2H, m, CH2-10′), 3.71 (1H, dd, 2JH-H = 12.0 Hz, 3JH-H = 6.0 Hz, CHa/b-6), 3.88 (1H, dd, 2JH-H = 12.0 Hz, 3JH-H = 6.0 Hz, CHa/b-6), 3.99–4.13 (4H, m, 2 × OCH2CH3), 4.20 (1H, m, CH-9), 4.37–4.47 (1H, m, CH-1), 4.78–4.84 (1H, m, CH-5), 5.39 (0.5H, d, 3JH-H = 6.0 Hz, NH-11), 5.41 (0.5H, d, 3JH-H = 6.0 Hz, NH-11), 7.15 (0.5H, d, 3JH-H = 6.0 Hz, NH-7), 7.24 (0.5H, d, 3JH-H = 6.0 Hz, NH-7), 7.52 (1H, m, NH-3); 13C NMR (75 MHz, CDCl3) δC 14.3 (CH3-2), 14.4 (CH3-2), 14.5 (CH2-10″), 15.3 (OCH2CH3), 15.4 (OCH2CH3), 15.5 (OCH2CH3), 15.6 (OCH2CH3), 27.3 (C(CH3)3), 29.2 (CH2-10′), 29.3 (CH2-10′), 30.2 (CH2-10), 30.4 (CH2-10), 40.3 (d, 1JP-C = 159.0 Hz, CH-1), 43.5 (CH2-6), 43.7 (CH2-6), 52.7 (CH-5), 53.1 (CH-9), 61.6 (d, 2JP-C = 6.8 Hz, OCH2CH3), 61.7 (d, 2JP-C = 6.0 Hz, OCH2CH3), 62.0 (d, 2JP-C = 6.8 Hz, OCH2CH3), 62.1 (d, 2JP-C = 7.5 Hz, OCH2CH3), 79.6 (C(CH3)3), 154.8 (C=O-12), 166.7 (C=O-4), 166.8 (C=O-4), 170.7 (C=O-8), 170.8 (C=O-8); 31P-1Hdecoup NMR (121 MHz, CDCl3) δP 24.5, 24.8; HRMS (NSI) calcd for (C19H3HClN3O7PS)+, MH+: 518.1851 (35Cl), 520.1821 (37Cl), found 518.1842 (35Cl), 520.1814 (37Cl); CHN (Found: C, 44.08; H, 7.47; N, 8.18. C19H37ClN3O7PS requires C, 44.06; H, 7.20; N, 8.11%).
![Molecules 25 01557 i029 Molecules 25 01557 i029]()
(1-((R)-2-((S)-2-Ammoniopentanamido)-3-chloropropanamido) ethyl)phosphonic acid or l-Nva-β-chloro-l-Ala-d/l-Fos (
25a). The
tert-butoxycarbonyl and diethyl ester protecting groups of
tert-butyl ((2
S)-1-(((2
R)-3-chloro-1-((1-(diethoxyphosphoryl)-ethyl)amino)-1-oxopropan-2-yl)amino)-1-oxopentan-2-yl)carbamate (
24a) (2.0 mmol, 0.99 g) were removed. The pale green crude solid was washed with diethyl ether to give
25a as a pale green solid composed of 2 diastereoisomers,
l-Nva-β-Cl-
l-Ala-
l-Fos and
l-Nva-β-Cl-
l-Ala-D-Fos (0.64 g, 1.94 mmol, 97%); m.p. 175 °C (sub);
ῡmax/cm
−1 3294 (NH
+), 3000 (br OH), 1668 (C=O), 1645 (C=O), 1538 (NH bend), 1132 (P=O), 1039 (P-O-C), 921 (P-OH);
1H NMR (300 MHz, D
2O) δ
H 1.01 (3H, t,
3JH-H = 9.0 Hz, C
H3-10″), 1.30–1.37 (3H, br m, C
H3-2), 1.44–1.54 (2H, br m C
H2-10′), 1.90–1.98 (2H, br m, C
H2-10), 3.91–4.15 (4H, br m, C
H2-6, C
H-9, C
H-1), 4.79 (1H, br m, C
H-5);
13C NMR (75 MHz, D
2O) δ
C 12.9 (
CH
3-10″), 15.7 (
CH
3-2), 17.6 (
CH
2-10′), 33.0 (
CH
2-10), 43.3 (
CH
2-6), 53.1 (
CH-1 and
CH-9), 55.0 (
CH-5), 170.4 (
C=O-4 and
C=O-8);
31P-
1H
decoup NMR (121 MHz, CDCl
3) δ
P 18.5; HRMS (NSI) calcd for (C
10H
20ClN
3O
5P)
-, MH
-: 328.0835 (
35Cl), 330.0805 (
37Cl), found 328.0833 (
35Cl), 330.0800 (
37Cl). LCMS purity >95% (C-18 reversed phase, MeOH-H
2O). (The LCMS chromatogram and conditions may be found within the
Supplementary Materials).
![Molecules 25 01557 i030 Molecules 25 01557 i030]()
1-((R)-3-Chloro-2-(2-(methylammonio)acetamido) propanamido)ethyl)phosphonic acid or Sar-β-chloro-l-Ala-d/l-Fos (
25b). The
tert-butoxycarbonyl and diethyl ester protecting groups of
tert-butyl (2-(((2
R)-3-chloro-1-((1-(diethoxyphosphoryl)ethyl)amino)-1-oxopropan-2-yl)amino)-2-oxoethyl)(methyl)carbamate (
24b) (3.8 mmol, 1.74 g) were removed. The pale green crude solid was recrystallised from hot water/ethanol to give
25b as an off-white solid composed of 2 diastereoisomers, Sar-β-Cl-
l-Ala-
l-Fos and Sar-β-Cl-
l-Ala-D-Fos (0.49 g, 1.61 mmol, 42%); m.p. 185-188 °C (decomp.);
ῡmax/cm
−1 3287 (NH
+), 3000 (br OH), 1657 (C=O), 1634 (C=O), 1552 (NH bend), 1172 (P=O), 1054 (P-O-C), 919 (P-OH);
1H NMR (300 MHz, CD
3OD) δ
H 1.24 (3H, dd,
3JH-P = 15.0 Hz,
3JH-H = 6.0 Hz, C
H3-2), 2.74 (3H, s, NC
H3-11), 3.81–3.87 (2H, m, C
H2-6), 3.93–3.94 (2H, m, C
H2-9), 3.97–4.10 (1H, m, C
H-1), 4.79 (1H, m, C
H-5);
13C NMR (75 MHz, CD
3OD) δ
C 15.4 (
CH
3-2), 32.9 (N
CH
3-11), 43.6 (
CH
2-6), 44.1 (d,
1JC-P = 148.5 Hz,
CH-1), 49.5 (
CH
2-9), 54.6 (
CH-5), 166.4 (
C=O-4 or
C=O-9), 166.9 (
C=O-4 or
C=O-9);
31P-
1H
decoup NMR (121 MHz, CDCl
3) δ
P 18.8; HRMS (NSI) calcd for (C
8H
18ClN
3O
5P)
+, MH
+: 302.0667 (
35Cl), 304.0638 (
37Cl), found 302.0670 (
35Cl), 304.0640 (
37Cl). LCMS purity >95% (C-18 reversed phase, MeOH-H
2O). (The LCMS chromatogram and conditions may be found within the
Supplementary Materials).
![Molecules 25 01557 i031 Molecules 25 01557 i031]()
(1-((R)-2-((S)-2-Ammonio-4-(methylthio)butanamido)-3-chloro propanamido)ethyl)phosphonic acid or l-Met-β-Cl-l-Ala-d/l-Fos (
25c). The tert-butoxycarbonyl and diethyl ester protecting groups of tert-butyl ((2S)-1-(((2R)-3-chloro-1-((1-(diethoxyphosphoryl) ethyl)amino)-1-oxopropan-2-yl)amino)-4-(methylthio)-1-oxobutan-2-yl)carbamate (
24c) (1.4 mmol, 0.71 g) were removed. The green crude solid was recrystallised from hot water/ethanol to give
25c as a pale green solid composed of 2 diastereoisomers,
l-Met-β-Cl-
l-Ala-
l-Fos and
l-Met-β-Cl-
l-Ala-D-Fos (0.17 g, 0.48 mmol, 35%); m.p. 175–179 °C (decomp.); ῡ
max/cm
−1 3264 (NH
+), 2829 (broad OH), 1666(C=O), 1641 (C=O), 1546 (NH bend), 1149 (P=O), 1041 (P-O-C), 921 (P-OH);
1H NMR (300 MHz, D
2O) δ
H 1.31 (3H, dd,
3JH-P = 15.0 Hz,
3JH-H = 6.0 Hz, C
H3-2), 2.13 (3H, s, C
H3-10″), 2.18–2.29 (2H, m, C
H2-10), 2.63–2.69 (2H, m, C
H2-10′), 3.89 (1H, dd,
2JH-H = 12.0 Hz,
3JH-H = 6.0 Hz, C
Ha/b-6), 3.97 (1H, dd,
2JH-H = 12.0 Hz,
3JH-H = 6.0 Hz, C
Ha/b-6), 4.01–4.13 (1H, m, C
H-1), 4.22 (1H, br m, C
H-9), 4.75–4.79 (1H, m, C
H-5);
13C NMR (75 MHz, D
2O) δ
C 16.9 (
CH
3-10″), 17.0 (
CH
3-10″), 18.4 (
CH
3-2), 31.1 (
CH
2-10′), 32.9 (
CH
2-10), 46.2 (
CH
2-6), 47.0 (d,
1JC-P = 147.0 Hz,
CH-1), 52.2 (
CH-9), 52.3 (
CH-9), 57.8 (
CH-5), 58.0 (
CH-5), 171.7 (
C=O-4), 171.8 (
C=O-4), 172.3 (
C=O-8);
31P-
1H
decoup NMR (121 MHz, CDCl
3) δ
P 18.7; HRMS (NSI) calcd for (C
10H
21ClN
3O
5PS), MNa
+: 384.0520 (
35Cl), 386.0489 (
37Cl), found 384.0523 (
35Cl), 386.0491 (
37Cl). LCMS purity >95% (C-18 reversed phase, MeOH-H
2O). (The LCMS chromatogram and conditions may be found within the
Supplementary Materials).