New Substituted Benzoylthiourea Derivatives: From Design to Antimicrobial Applications
Abstract
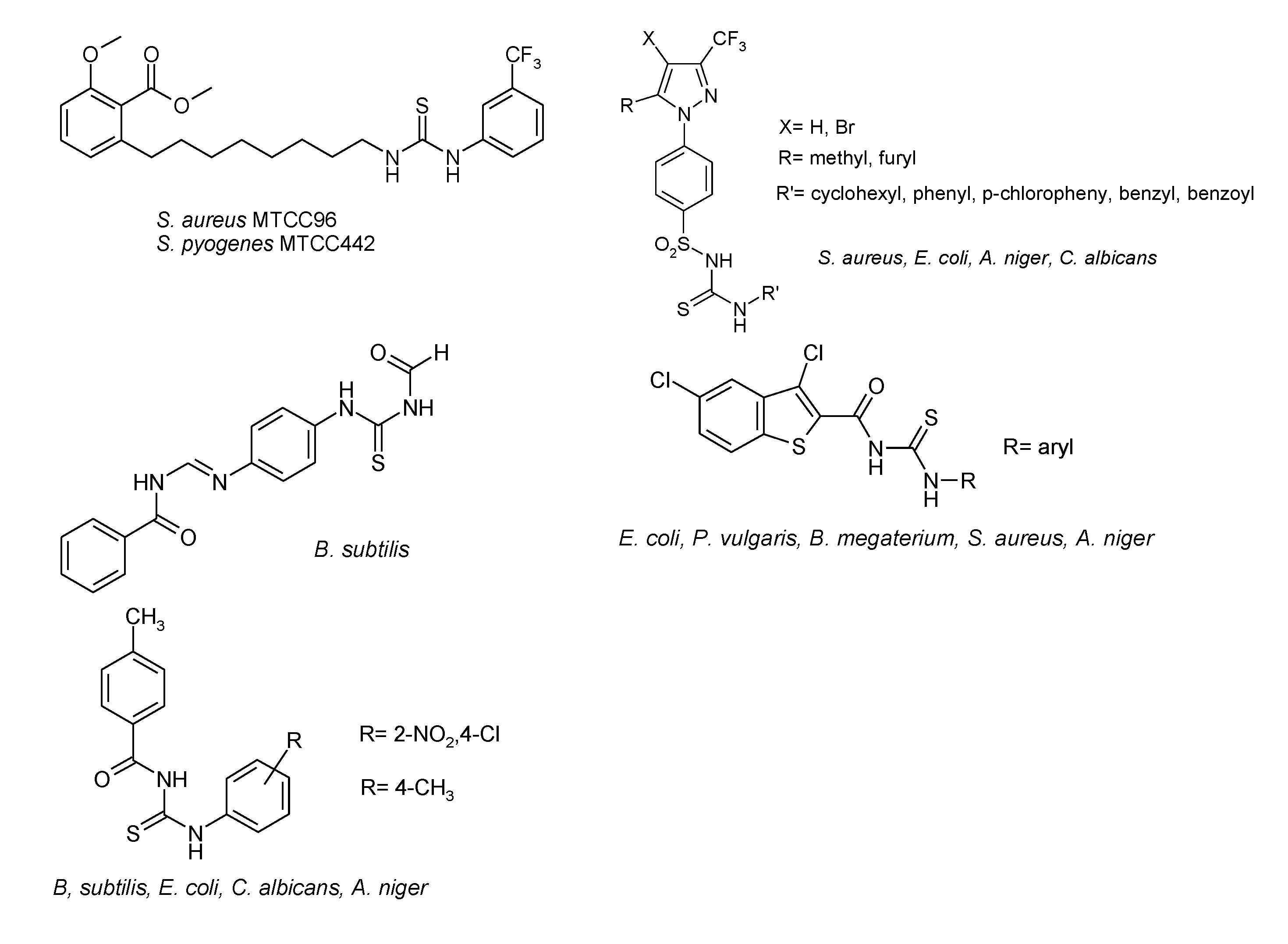
1. Introduction
2. Results
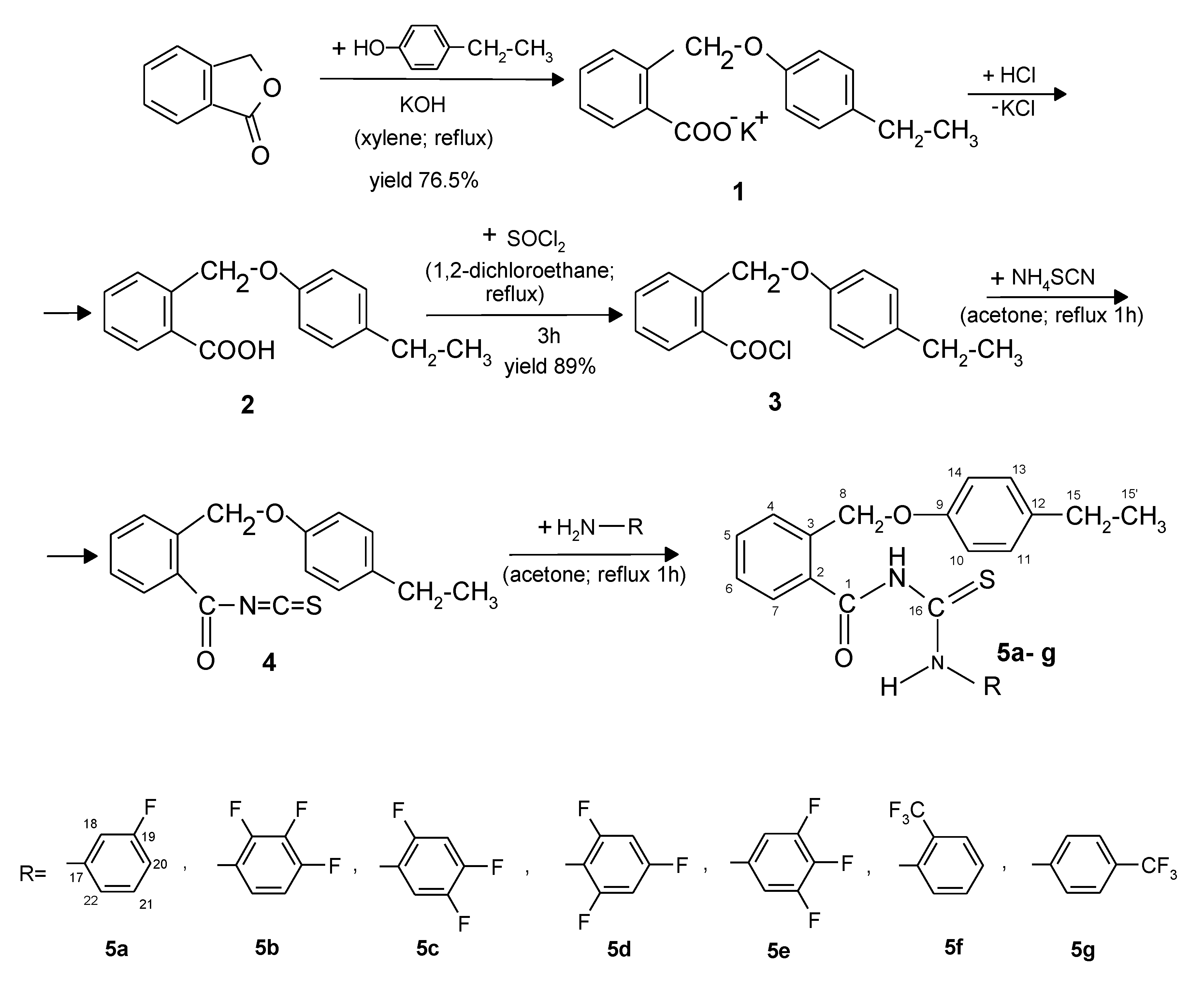
2.1. Chemistry
2.2. Computational Studies
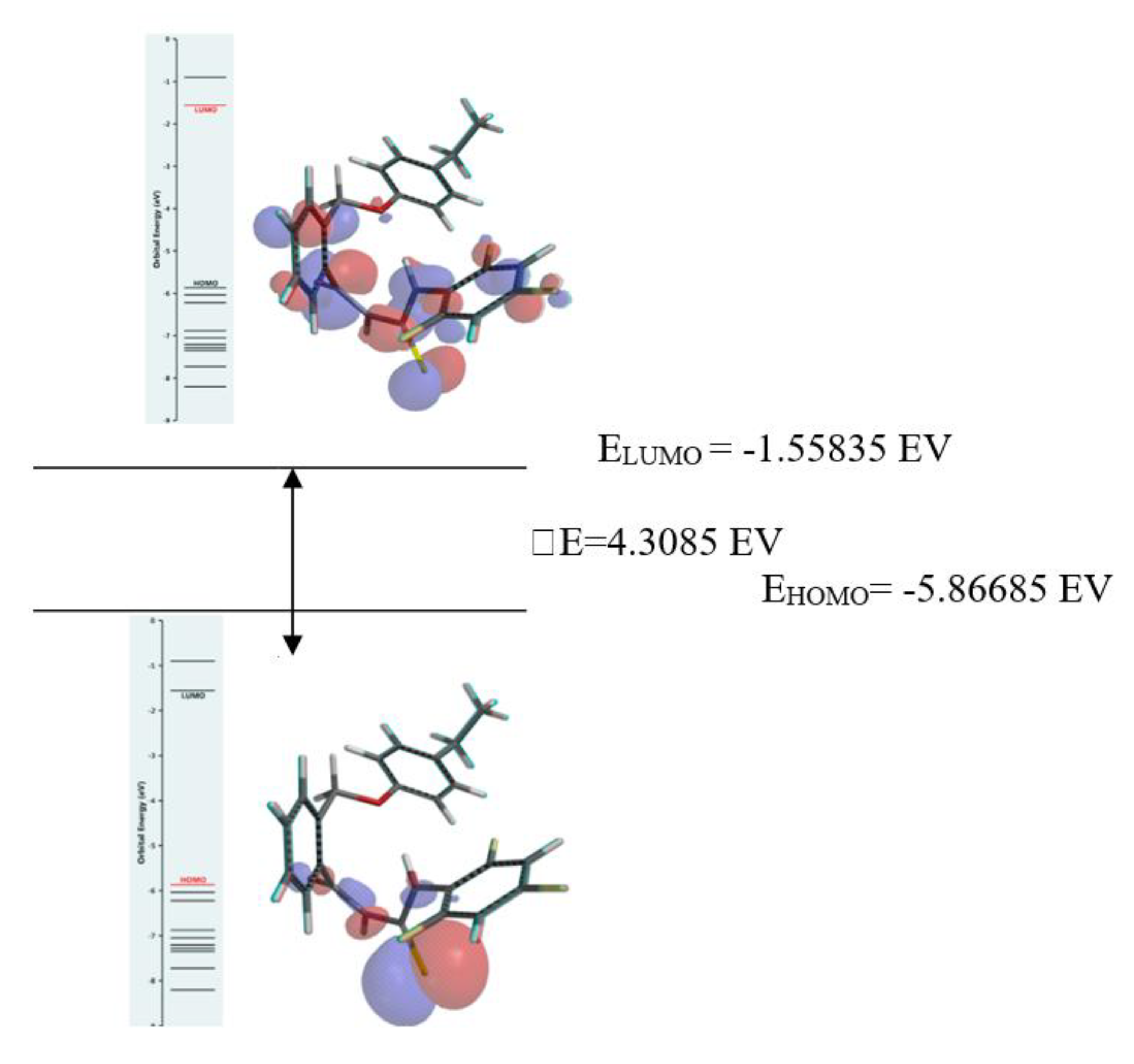
2.2.1. Frontier Molecular Orbitals Analysis
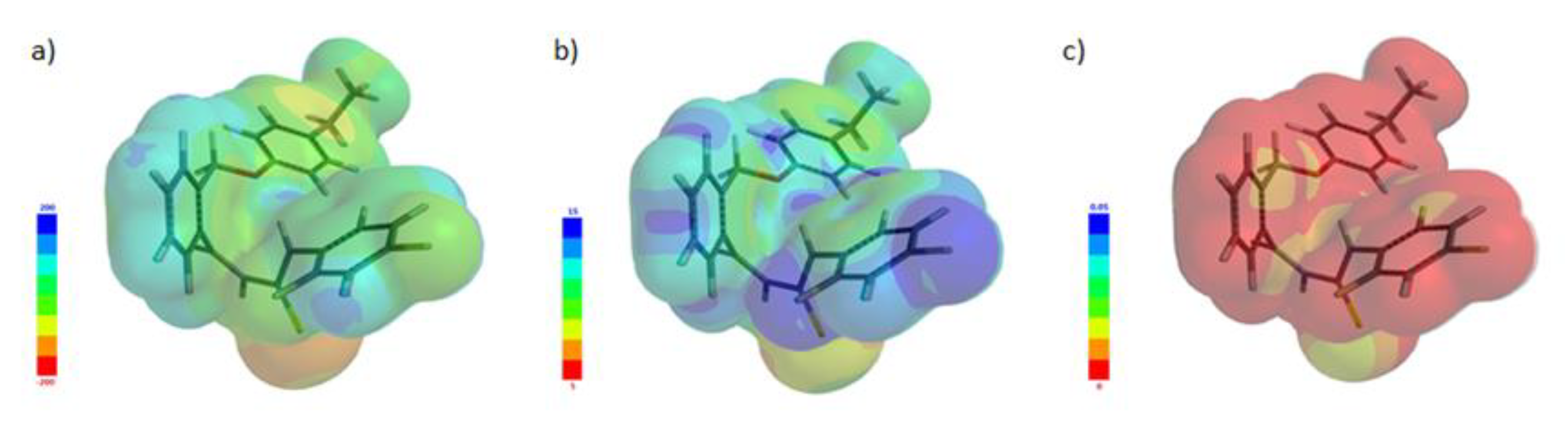
2.2.2. Molecular Electrostatic Potential (MEP)
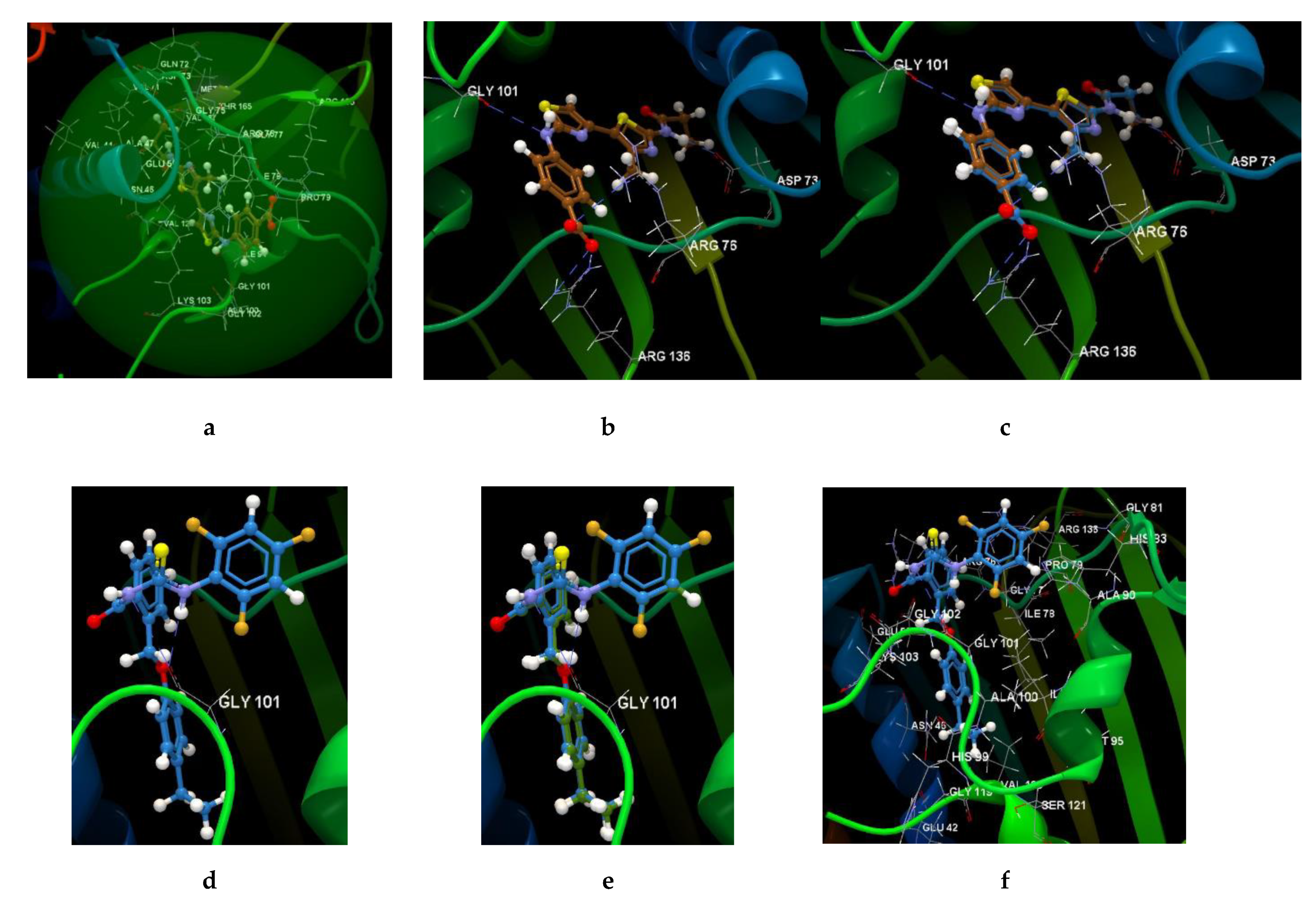
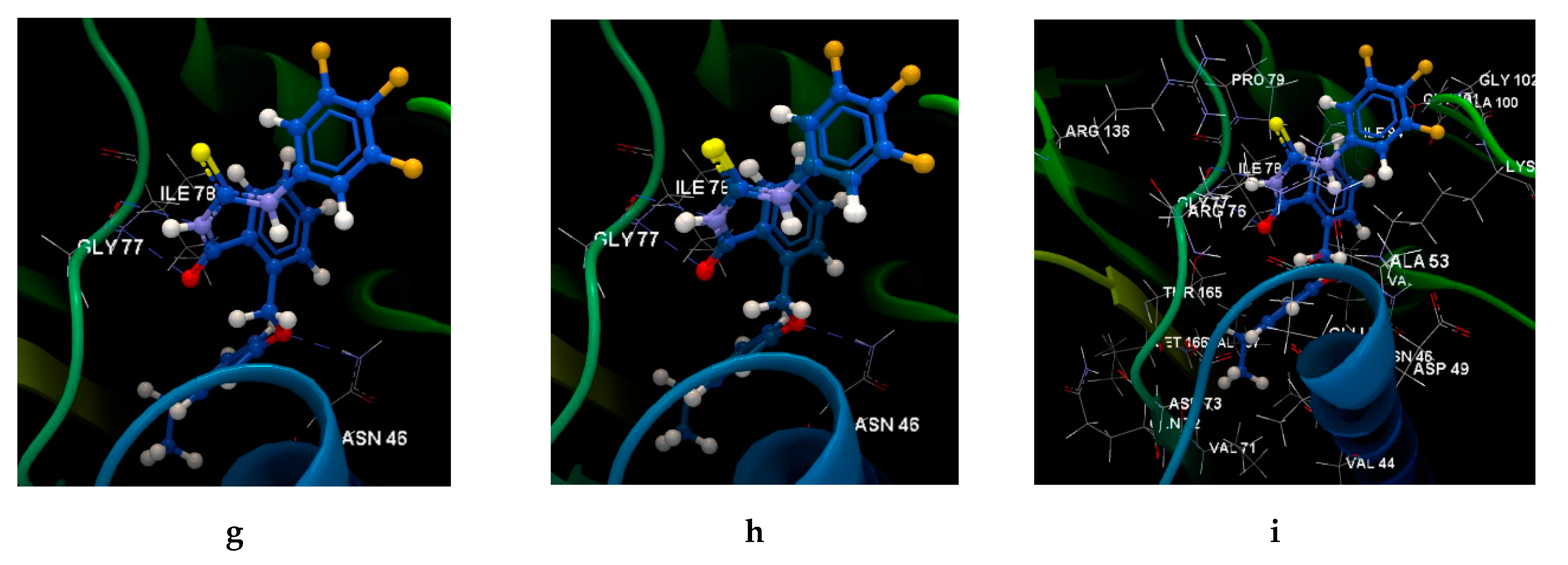
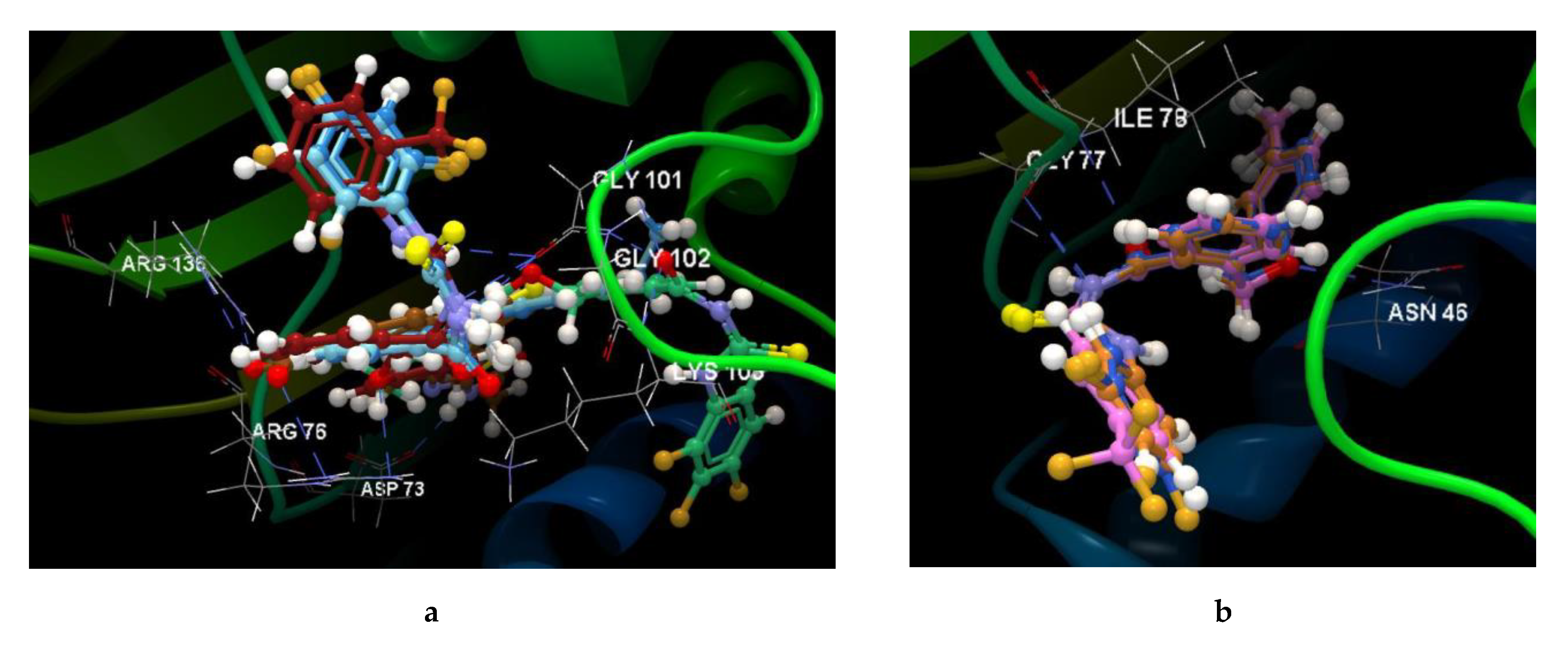
2.2.3. Docking Studies
2.3. Antimicrobial Activity Assay
3. Discussion
4. Materials and Methods
4.1. Chemistry
4.1.1. General Synthesis Procedure of the New Benzoylthiourea Derivatives
2-((4-Ethylphenoxy)methyl)-N-(3-fluorophenylcarbamothioyl)benzamide (5a), 3.18 g white crystals (yield 78%), mp 109–110 °C.
2-((4-Ethylphenoxy)methyl)-N-(2,3,4-trifluorophenylcarbamothioyl)benzamide (5b) 3.37 g white crystals (yield 76%), mp 107–108 °C.
2-((4-Ethylphenoxy)methyl)-N-(2,4,5-trifluorophenylcarbamothioyl)benzamide (5c) 3.24 g white crystals (yield 73%), mp 114–115 °C.
2-((4-Ethylphenoxy)methyl)-N-(2,4,6-trifluorophenylcarbamothioyl)benzamide (5d) 3.20 g white crystals (yield 72%), mp 122–123 °C.
2-((4-Ethylphenoxy)methyl)-N-(3,4,5-trifluorophenylcarbamothioyl)benzamide (5e) 3.33 g light yellow crystals (yield 75%), mp 133–134 °C.
2-((4-Ethylphenoxy)methyl)-N-(2-trifluoromethylphenylcarbamothioyl)benzamide (5f) 3.39 g white crystals (yield 74%); mp 123–124 °C.
2-((4-Ethylphenoxy)methyl)-N-(4-trifluoromethylphenylcarbamothioyl)benzamide (5g) 3.25 g light yellow crystals (yield 71%), mp 138-139 °C.
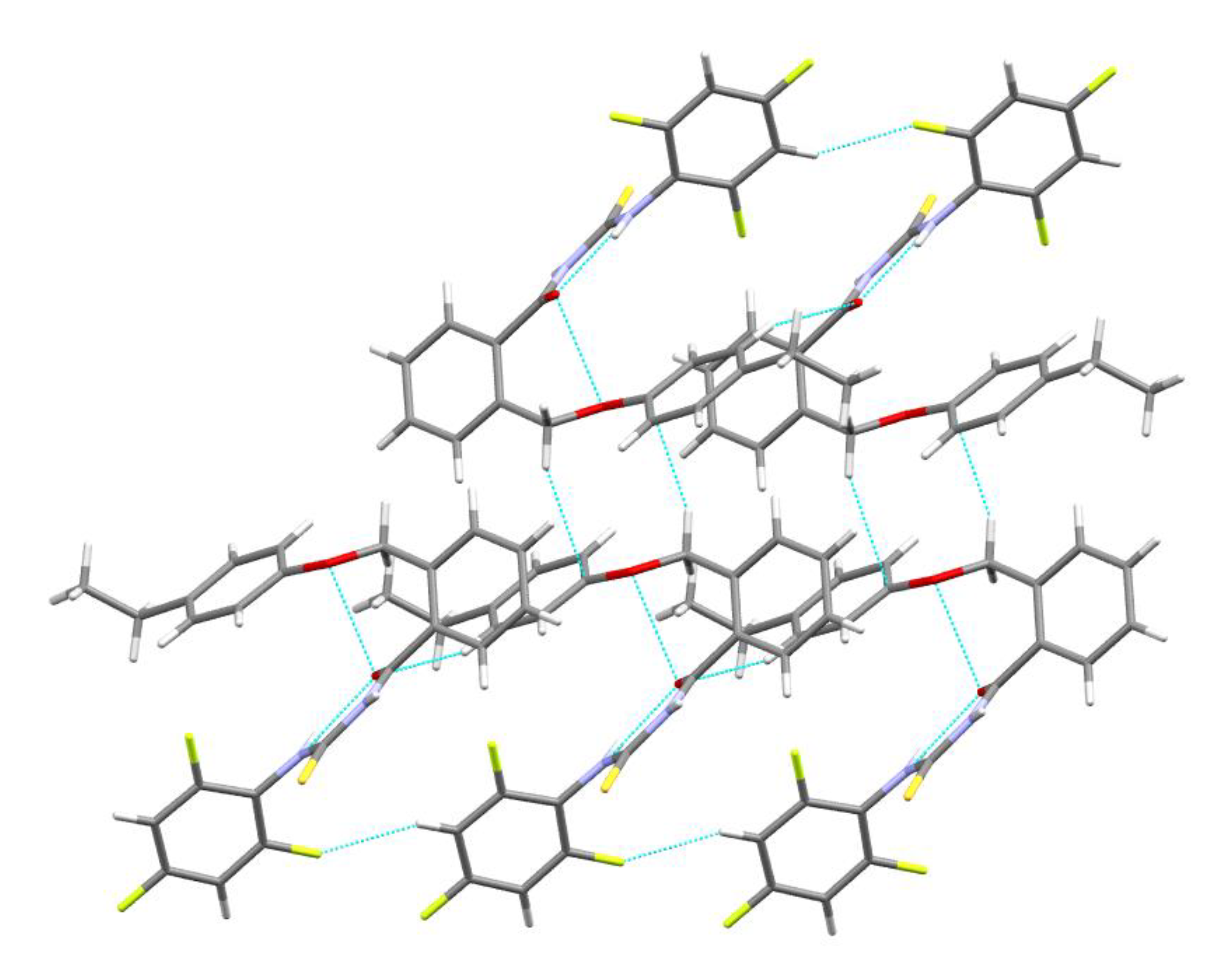
4.1.2. Single Crystal X-ray Diffraction, Crystal Data Collection and Refinement
4.2. Computational Analyses
4.3. In Vitro Assessment of the Antimicrobial Activity of the Newly Synthesized F-Benzoylthiourea Derivatives on Planktonic Microbes (Grown in Suspension)
4.4. Antimicrobial Activity of Newly Synthesized F-Benzoylthiourea Derivatives on Microbial Adherence and Biofilms’ Development on the Inert Substratum
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Vedavathia, P.; Sekhara, C.K.; Rajua, N.C. Synthesis, spectral characterization and antimicrobial activity of thiourea/urea derivatives of amlodipine. Pharm. Lett. 2013, 5, 199–204. [Google Scholar]
- Reddy, V.R.; Raju, N.; Reddy, C.K.; Rao, S. Urea and thiourea derivatives of celecoxib drug: Synthesis and evaluation of antimicrobial activity. IAJPR 2013, 3, 8228–8236. [Google Scholar]
- Mannam, M.R.; Devineni, S.R.; Pavuluri, C.M.; Chamarthi, N.R.; Pal, M. Urea and thiourea derivatives of 3-(trifluoromethyl)-5,6,7,8-tetrahydro-[1,2,4]triazolo[4,3-a]pyrazine: Synthesis, characterization, antimicrobial activity and docking studies. Phosphorus, Sulfur, Silicon Relat. Elements 2019, 194, 922–932. [Google Scholar] [CrossRef]
- Kurt, G.; Sevgi, F.; Mercimek, B. Synthesis, characterization, and antimicrobial activity of new benzoylthiourea ligands. Chem. Pap. 2009, 63, 548–553. [Google Scholar] [CrossRef]
- Thakar, K.; Paghdar, D.; Chovatia, P.; Joshi, H.S. Synthesis of thiourea derivatives bearing the benzo[b]thiophene nucleus as potential antimicrobial agents. J. Serbian Chem. Soc. 2005, 70, 807–815. [Google Scholar] [CrossRef]
- Semwal, A.; Nigam, A.; Gupta, S. Synthesis, characterization and biological evaluation of novel N-p-methylbenzoyl-N’substituted thiourea. AJPLS 2011, 1, 149–155. [Google Scholar]
- Pradhan, A.; Khan, N.; Goyal, H. Antifungal and anti-bacterial Activity of neoteric substituted thiocarbamide. Int. J. Theor. Appl. Sci. 2016, 8, 41–44. [Google Scholar]
- Ghorab, M.M.; El-Gaby, M.; Soliman, A.M.; Alsaid, M.S.; Abdel-Aziz, M.M.; Elaasser, M. Synthesis, docking study and biological evaluation of some new thiourea derivatives bearing benzenesulfonamide moiety. Chem. Cent. J. 2017, 11, 42. [Google Scholar] [CrossRef]
- Krajačić, M.B.; Perić, M.; Smith, K.S.; Schönfeld, Z.I.; Žiher, D.; Fajdetić, A.; Kujundžić, N.; Schönfeld, W.; Landek, G.; Padovan, J.; et al. Synthesis, Structure–Activity Relationship, and Antimalarial Activity of Ureas and Thioureas of 15-Membered Azalides. J. Med. Chem. 2011, 54, 3595–3605. [Google Scholar] [CrossRef]
- El Bissati, K.; Redel, H.; Ting, L.-M.; Lykins, J.D.; McPhillie, M.J.; Upadhya, R.; Woster, P.M.; Yarlett, N.; Kim, K.; Weiss, L.M. Novel Synthetic Polyamines Have Potent Antimalarial Activities in vitro and in vivo by Decreasing Intracellular Spermidine and Spermine Concentrations. Front. Microbiol. 2019, 9, 9. [Google Scholar] [CrossRef]
- Mahajan, A.; Yeh, S.; Nell, M.; Van Rensburg, C.E.; Chibale, K.; Van Rensburg, C.E.J. Synthesis of new 7-chloroquinolinyl thioureas and their biological investigation as potential antimalarial and anticancer agents. Bioorganic Med. Chem. Lett. 2007, 17, 5683–5685. [Google Scholar] [CrossRef]
- Díaz, M.; De Lucio, H.; Moreno, E.; Espuelas, S.; Aydillo, C.; Jimenez-Ruiz, A.; Ángel Toro, M.; Gutiérrez, K.J.; Martinez_Merino, V.; Cornejo, A.; et al. Synthesis and Leishmanicidal Activity of Novel Urea, Thiourea, and Selenourea Derivatives of Diselenides. Antimicrob. Agents Chemother. 2019. [Google Scholar] [CrossRef]
- Elkholy, S.S.; Salem, H.A.; Eweis, M.; Elsabee, M.Z. Synthesis and characterization of some acyl thiourea derivatives of chitosan and their biocidal activities. Int. J. Boil. Macromol. 2014, 70, 199–207. [Google Scholar] [CrossRef]
- Kocyigit-Kaymakcioglu, B.; Celen, A.O.; Tabanca, N.; Ali, A.; Khan, S.; Khan, I.; Wedge, D. Synthesis and Biological Activity of Substituted Urea and Thiourea Derivatives Containing 1,2,4-Triazole Moieties. Molecules 2013, 18, 3562–3576. [Google Scholar] [CrossRef]
- Wang, H.; Zhai, Z.-W.; Shi, Y.-X.; Tan, C.-X.; Weng, J.-Q.; Han, L.; Li, B.-J.; Liu, X. Novel Trifluoromethylpyrazole Acyl Thiourea Derivatives: Synthesis, Antifungal Activity and Docking Study. Lett. Drug Des. Discov. 2019, 16, 785–791. [Google Scholar] [CrossRef]
- Saeed, S.; Rashid, N.; Ali, M.; Hussain, R.; Jones, P.G. Synthesis, spectroscopic characterization, crystal structure and pharmacological properties of some novel thiophene-thiourea core derivatives. Eur. J. Chem. 2010, 1, 221–227. [Google Scholar] [CrossRef]
- Wu, J.; Shi, Q.; Chen, Z.; He, M.; Jin, L.; Hu, D. Synthesis and Bioactivity of Pyrazole Acyl Thiourea Derivatives. Molecules 2012, 17, 5139–5150. [Google Scholar] [CrossRef]
- Ravichandran, V.; Shalini, S.; Kumar, K.S.; Rajak, H.; Agrawal, R.K. Design, Synthesis and Evaluation of Thiourea Derivatives as Antimicrobial and Antiviral Agents. Lett. Drug Des. Discov. 2019, 16, 618–624. [Google Scholar] [CrossRef]
- Uckun, F.M.; Venkatachalam, T.K. Adamantyl thiazole thioureas. US Patent 6,960,606, 1 November 2005. [Google Scholar]
- Küçükgüzel, I.; Tatar, E.; Küçükgüzel, S.G.; Rollas, S.; De De Clercq, E. Synthesis of some novel thiourea derivatives obtained from 5-[(4-aminophenoxy)methyl]-4-alkyl/aryl-2,4-dihydro-3H-1,2,4-triazole-3-thiones and evaluation as antiviral/anti-HIV and anti-tuberculosis agents. Eur. J. Med. Chem. 2008, 43, 381–392. [Google Scholar] [CrossRef]
- Al-Masoudi, N.A. Amino acid derivatives. Part 5. Synthesis and anti-HIV activity of new sebacoyl precursor derived thioureido-amino acid and phthalimide derivatives. Arkivoc 2010, 2010, 185–195. [Google Scholar]
- Galabov, A.S.; Galabov, B.S.; Neykova, N.A. Structure-activity relationship of diphenylthiourea antivirals. J. Med. Chem. 1980, 23, 1048–1051. [Google Scholar] [CrossRef]
- Wyles, D.L.; Kaihara, K.A.; Schooley, R.T. Synergy of a Hepatitis C Virus (HCV) NS4A Antagonist in Combination with HCV Protease and Polymerase Inhibitors. Antimicrob. Agents Chemother. 2008, 52, 1862–1864. [Google Scholar] [CrossRef]
- Reddy, N.S.; Rao, A.S.; Chari, M.A.; Kumar, V.R.; Jyothi, V.; Himabindu, V. Synthesis and anti-bacterial activity of urea and thiourea derivatives at C-8 alkyl chain of anacardic acid mixture isolated from a natural product cashew nutshell liquid (CNSL). Int. J. Org. Chem. 2011, 1, 167–175. [Google Scholar] [CrossRef]
- Faidallah, H.M.; Khan, K.A.; Asiri, A.M. Synthesis and biological evaluation of new 3-trifluoromethylpyrazolessulfonyl-urea and thiourea derivatives as antidiabetic and antimicrobial agents. J. Fluor. Chem. 2011, 132, 131–137. [Google Scholar] [CrossRef]
- Ramadas, K.; Suresh, G.; Janarthanan, N.; Masilamani, S. Antifungal activity of 1,3-disubstituted symmetrical and unsymmetrical thioureas. Pestic. Sci. 1998, 52, 145–151. [Google Scholar] [CrossRef]
- Ke, S. Synthesis and herbicidal activity of N-(o-fluorophenoxyacetyl)thioureas derivatives and related fused heterocyclic compounds. Arkivoc 2006, 2006, 63. [Google Scholar]
- Mei, H.; Han, J.; Fustero, S.; Medio-Simon, M.; Sedgwick, D.M.; Santi, C.; Ruzziconi, R.; Soloshonok, V.A. Fluorine-Containing Drugs Approved by the FDA in 2018. Chemistry 2019, 25, 11797–11819. [Google Scholar] [CrossRef]
- Ma, J.-A.; Cahard, D. Update 1 of: Asymmetric Fluorination, Trifluoromethylation, and Perfluoroalkylation Reactions. Chem. Rev. 2008, 108, PR1–PR43. [Google Scholar] [CrossRef]
- Ismail, F.M.D.; Drew, M.G.B.; Dascombe, M.J. Modulation of drug pharmacokinetics and pharmacodynamics by fluorine substitution. Chim. Oggi. 2009, 27, 18–22. [Google Scholar]
- Filler, R.; Saha, R. Fluorine in medicinal chemistry: A century of progress and a 60-year retrospective of selected highlights. Futur. Med. Chem. 2009, 1, 777–791. [Google Scholar] [CrossRef]
- Patel, N.; Patel, S.; Patel, J.; Patel, J.; Gorgamwala, Y. Synthesis and Antibacterial Activity of Thioureido Amide of Fluoroquinolone. Int. J. Boil. Chem. 2011, 5, 37–45. [Google Scholar] [CrossRef]
- Pathak, A.K.; Chawla, V.; Saraf, S.K. Synthesis of 2-(6’-fluorobenzothiazol-2’-ylamino)-4,6-(disubstituted thioureido)-1,3-pyrimidine derivatives as antimicrobial agents. E-J. Chem. 2011, 8, 240–244. [Google Scholar] [CrossRef]
- Saeed, A.; Shaheen, U.; Hameed, A.; Naqvi, S.H. Synthesis, characterization and antimicrobial activity of some new 1-(fluorobenzoyl)-3-(fluorophenyl)thioureas. J. Fluor. Chem. 2009, 130, 1028–1034. [Google Scholar] [CrossRef]
- Rioton, S.; Pardo, D.G.; Cossy, J. Synthesis of Substituted α-Trifluoromethyl Piperidinic Derivatives. Molecules 2017, 22, 483. [Google Scholar] [CrossRef]
- Zhu, W.; Wang, J.; Wang, S.; Gu, Z.; Aceña, J.L.; Izawa, K.; Liu, H.; Soloshonok, V.A. Recent advances in the trifluoromethylation methodology and new CF3-containing drugs. J. Fluor. Chem. 2014, 167, 37–54. [Google Scholar] [CrossRef]
- Commare, B.; Schmitt, E.; Aribi, F.; Panossian, A.; Vors, J.-P.; Pazenok, S.; Leroux, F. Fluoroalkyl Amino Reagents (FARs): A General Approach towards the Synthesis of Heterocyclic Compounds Bearing Emergent Fluorinated Substituents. Molecules 2017, 22, 977. [Google Scholar] [CrossRef]
- Cybulska, K.; Perk, L.; Booij, J.; Laverman, P.; Rijpkema, M. Huntington’s Disease: A Review of the Known PET Imaging Biomarkers and Targeting Radiotracers. Molecules 2020, 25, 482. [Google Scholar] [CrossRef]
- De La Torre, B.G.; Albericio, F. The Pharmaceutical Industry in 2019. An Analysis of FDA Drug Approvals from the Perspective of Molecules. Molecules 2020, 25, 745. [Google Scholar] [CrossRef]
- Aslam, B.; Wang, W.; Arshad, M.I.; Khurshid, M.; Muzammil, S.; Rasool, M.H.; Nisar, M.A.; Alvi, R.F.; Aslam, M.A.; Qamar, M.U.; et al. Antibiotic resistance: A rundown of a global crisis. Infect. Drug Resist. 2018, 11, 1645–1658. [Google Scholar] [CrossRef]
- Betts, J.W.; Hornsey, M.; La Ragione, R.M. Novel Antibacterials: Alternatives to Traditional Antibiotics. Adv. Microb. Physiol. 2018, 73, 123–169. [Google Scholar]
- Spellberg, B.; Srinivasan, A.; Chambers, H.F. New Societal Approaches to Empowering Antibiotic Stewardship. JAMA 2016, 315, 1229–1230. [Google Scholar] [CrossRef]
- Luepke, K.H.; Mohr, J.F. The Antibiotic Pipeline: Reviving Research and Development and Speeding Drugs to Market. Expert Rev. Anti-infective Ther. 2017, 15, 425–433. [Google Scholar] [CrossRef]
- Limban, C.; Missir, A.V.; Chiriţă, I.C.; Niţulescu, G.M.; Ilie, C.; Căproiu, M.T. Some new 2-(4-ethyl-phenoxymethyl)benzoic acid thioureides: Synthesis and spectral characterisation. Rev.Chim. (Bucharest) 2009, 60, 657–661. [Google Scholar]
- Limban, C.; Missir, A.V.; Chirita, I.C.; Nitulescu, G.M.; Caproiu, M.T.; Chifiriuc, M.C.; Israil, A.M. Synthesis and antimicrobial properties of new 2-((4-ethylphenoxy)methyl)benzoylthioureas. Chem. Pap. 2011, 65, 60–69. [Google Scholar] [CrossRef]
- Saeed, A.; Rafique, H.; Mumtaz, A.; Bolte, M. Ethyl 4-[3-(2-methyl-benzo-yl)thio-ureido]benzoate. Acta Crystallogr. Sect. E Struct. Rep. Online 2009, 65. [Google Scholar] [CrossRef]
- Li, Q.; Yang, C.-L. Synthesis and Crystal Structure of a Compound with Two Conformational Isomers: N-(2-methylbenzoyl)-N′-(4-nitrophenyl)thiourea. J. Chem. Crystallogr. 2008, 38, 927–930. [Google Scholar] [CrossRef]
- Ghose, A.K.; Pritchett, A.; Crippen, G.M. Atomic physicochemical parameters for three dimensional structure directed quantitative structure-activity relationships III: Modeling hydrophobic interactions. J. Comput. Chem. 1988, 9, 80–90. [Google Scholar] [CrossRef]
- Rk, G.B.D.; M., P.R.S.; Yadav, R.; Bps, D.R.G. Synthesis, Characterization, Single Crystal Structural Studies, Antibacterial Activity and DFT Investigations of 2-Chloro-5-Ethoxy-3,6-Bis(Methylamino)-1,4-Benzoquinone. Pharm. Anal. Acta 2015, 6. [Google Scholar] [CrossRef]
- Lasri, J.; Eltayeb, N.E.; Ismail, A.I. Experimental and theoretical study of crystal and molecular structure of 1,2-di(9H-fluoren-9-ylidene)hydrazine. J. Mol. Struct. 2016, 1121, 35–45. [Google Scholar] [CrossRef]
- Sarveswari, S.; Srikanth, A.; Murugan, N.A.; Vijayakumar, V.; Jasinski, J.P.; Beauchesne, H.C.; Jarvis, E.E. Synthesis, characterization of (3E)-1-(6-chloro-2-methyl-4-phenyl quinolin-3-Yl)-3-aryl prop-2-en-1-ones through IR, NMR, single crystal X-ray diffraction and insights into their electronic structure using DFT calculations. Spectrochim. Acta Part A: Mol. Biomol. Spectrosc. 2015, 136, 1010–1017. [Google Scholar] [CrossRef]
- Korb, O.; Stützle, T.; Exner, T.E. Empirical Scoring Functions for Advanced Protein−Ligand Docking with PLANTS. J. Chem. Inf. Model. 2009, 49, 84–96. [Google Scholar] [CrossRef] [PubMed]
- Lipinski, C.; Lombardo, F.; Dominy, B.W.; Feeney, P.J. Experimental and computational approaches to estimate solubility and permeability in drug discovery and development settings. Adv. Drug Deliv. Rev. 2001. [Google Scholar] [CrossRef]
- Cheng, T.; Zhao, Y.; Li, X.; Lin, F.; Xu, Y.; Zhang, X.; Li, Y.; Wang, R.; Lai, L. Computation of Octanol−Water Partition Coefficients by Guiding an Additive Model with Knowledge. J. Chem. Inf. Model. 2007, 47, 2140–2148. [Google Scholar] [CrossRef] [PubMed]
- Limban, C.; Marutescu, L.; Chifiriuc, M.C. Synthesis, Spectroscopic Properties and Antipathogenic Activity of New Thiourea Derivatives. Molecules 2011, 16, 7593–7607. [Google Scholar] [CrossRef]
- Sheldrick, G.M. Foundations of crystallography. Acta Cryst. Section A 2008, 64, 112–122. [Google Scholar] [CrossRef]
- Crystal Structure 4.2: Crystal Structure Analysis Package, Rigaku Corporation (2000-2015). Tokyo, 196-8666, Japan. Available online: https://www.rigaku.com/products/xrd?gclid=EAIaIQobChMI5_Dh4buw6AIVk00YCh0tmw2wEAAYASAAEgIc3PD_BwE (accessed on 23 March 2020).
- Brvar, M.; Perdih, A.; Renko, M.; Anderluh, G.; Turk, D.; Solmajer, T. Structure-Based Discovery of Substituted 4,5′-Bithiazoles as Novel DNA Gyrase Inhibitors. J. Med. Chem. 2012, 55, 6413–6426. [Google Scholar] [CrossRef]
- Jankovic, A.; Erakovic, S.; Ristoscu, C.; Mihailescu, N.; Duta, L.M.; Visan, A.; Stan, G.; Popa, A.C.; Husanu, M.-A.; Luculescu, C.R.; et al. Structural and biological evaluation of lignin addition to simple and silver-doped hydroxyapatite thin films synthesized by matrix-assisted pulsed laser evaporation. J. Mater. Sci. Mater. Electron. 2015, 26. [Google Scholar] [CrossRef]
- Nuta, D.C.; Măruțescu, L.; Missir, A.V.; Morusciag, L.; Chirita, C.; Curutiu, C.; Badiceanu, C.D.; Papacocea, M.T.; Limban, C. In vitro evaluation of the antimicrobial activity of N-phenylcarbamothioyl benzamides against planktonic and adherent microbial cells. Rom. Biotechnol. Lett. 2017, 22, 13163–13169. [Google Scholar]
- Stan, T.; Teodor, E.D.; Gatea, F.; Chifiriuc, M.C.; Lazăr, V. Antioxidant and antifungal activity of Romanian propolis. Rom. Biotechnol. Lett. 2017, 22, 13116–13124. [Google Scholar]
- Bădiceanu, C.D.; Missir, A.V.; Chifiriuc, M.C.; Dracea, O.; Raut, I.; Larion, C.; Ditu, L.M.; Mihaescu, G. Antimicrobial activity of some new 2-thiophene carboxylic acid thioureides. Rom. Biotechnol. Lett. 2010, 15, 5545–5551. [Google Scholar]
- Mihaiescu, D.E.; Cristescu, R.; Dorcioman, G.; Popescu, C.E.; Nita, C.; Socol, G.; Mihailescu, I.N.; Grumezescu, A.M.; Tamas, D.; Enculescu, M.; et al. Functionalized magnetite silica thin films fabricated by MAPLE with antibiofilm properties. Biofabrication 2013. [Google Scholar] [CrossRef] [PubMed]
- Ciobanu, C.S.; Groza, A.; Iconaru, S.L.; Popa, C.L.; Chapon, P.; Chifiriuc, M.C.; Hristu, R.; Stanciu, G.A.; Negrila, C.C.; Ghita, R.V.; et al. Antimicrobial activity evaluation on silver doped hydroxyapatite/polydimethylsiloxane composite layer. BioMed Res. Int. 2015. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds 5a–g are available from the authors. |

| Compound | Atoms | Weight [Daltons] | Flexible Bonds | Lipinski Violations | Hydrogen Donors | Hydrogen Acceptors | LogP ** |
|---|---|---|---|---|---|---|---|
| Co-crystallized * | 41 | 387.46 | 6 | 1 | 2 | 7 | 5.10 |
| 5a | 50 | 408.49 | 6 | 1 | 2 | 4 | 5.52 |
| 5b | 50 | 444.47 | 6 | 1 | 2 | 4 | 5.72 |
| 5c | 50 | 444.47 | 6 | 1 | 2 | 4 | 5.72 |
| 5d | 50 | 444.47 | 6 | 1 | 2 | 4 | 5.72 |
| 5e | 50 | 447.47 | 6 | 1 | 2 | 4 | 5.72 |
| 5f | 53 | 458.50 | 7 | 1 | 2 | 4 | 6.31 |
| 5g | 53 | 458.50 | 7 | 1 | 2 | 4 | 6.31 |
| 5a | 5b | 5c | 5d | 5e | 5f | 5g | |
|---|---|---|---|---|---|---|---|
| E. coli ATCC 25992 | 128 | 128 | >256 | 128 | 128 | >256 | 128 |
| P. aeruginosa ATCC 27853 | 128 | 128 | >256 | >256 | >256 | >256 | >256 |
| S. aureus ATCC 25923 | >128 | >128 | >128 | >128 | >128 | >128 | >128 |
| E. faecalis ATCC 29212 | 128 | >256 | >256 | >256 | >256 | >256 | >256 |
| B. subtilis ATCC 6633 | >128 | >128 | >128 | >128 | >128 | >128 | >128 |
| C. albicans ATCC 10231 | >128 | >128 | >128 | 64 | >128 | >128 | >128 |
| 5a | 5b | 5c | 5d | 5e | 5f | 5g | |
|---|---|---|---|---|---|---|---|
| E. coli ATCC 25992 | A | A | A | A | I | A | I |
| P. aeruginosa ATCC 27853 | A | I | I | I | I | I | I |
| S. aureus ATCC 25923 | I | A | I | A | A | A | I |
| E. faecalis ATCC 29212 | I | I | I | I | I | I | I |
| B. subtilis ATCC 6633 | I | A | I | A | A | I | A |
| C. albicans ATCC 10231 | I | A | A | I | I | I | I |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Limban, C.; Chifiriuc, M.C.; Caproiu, M.T.; Dumitrascu, F.; Ferbinteanu, M.; Pintilie, L.; Stefaniu, A.; Vlad, I.M.; Bleotu, C.; Marutescu, L.G.; et al. New Substituted Benzoylthiourea Derivatives: From Design to Antimicrobial Applications. Molecules 2020, 25, 1478. https://doi.org/10.3390/molecules25071478
Limban C, Chifiriuc MC, Caproiu MT, Dumitrascu F, Ferbinteanu M, Pintilie L, Stefaniu A, Vlad IM, Bleotu C, Marutescu LG, et al. New Substituted Benzoylthiourea Derivatives: From Design to Antimicrobial Applications. Molecules. 2020; 25(7):1478. https://doi.org/10.3390/molecules25071478
Chicago/Turabian StyleLimban, Carmen, Mariana Carmen Chifiriuc, Miron Teodor Caproiu, Florea Dumitrascu, Marilena Ferbinteanu, Lucia Pintilie, Amalia Stefaniu, Ilinca Margareta Vlad, Coralia Bleotu, Luminita Gabriela Marutescu, and et al. 2020. "New Substituted Benzoylthiourea Derivatives: From Design to Antimicrobial Applications" Molecules 25, no. 7: 1478. https://doi.org/10.3390/molecules25071478
APA StyleLimban, C., Chifiriuc, M. C., Caproiu, M. T., Dumitrascu, F., Ferbinteanu, M., Pintilie, L., Stefaniu, A., Vlad, I. M., Bleotu, C., Marutescu, L. G., & Nuta, D. C. (2020). New Substituted Benzoylthiourea Derivatives: From Design to Antimicrobial Applications. Molecules, 25(7), 1478. https://doi.org/10.3390/molecules25071478










