Kinetic Isotope Effects and Hydrogen Tunnelling in PCET Oxidations of Ascorbate: New Insights into Aqueous Chemistry?
Abstract
1. Introduction
2. Kinetic Isotope Effects and Hydrogen Tunnelling in Proton-Coupled Electron Transfer Reactions
2.1. Proton-Coupled Electron Transfer Reactions
2.2. Kinetic Isotope Effects in PCET Reactions
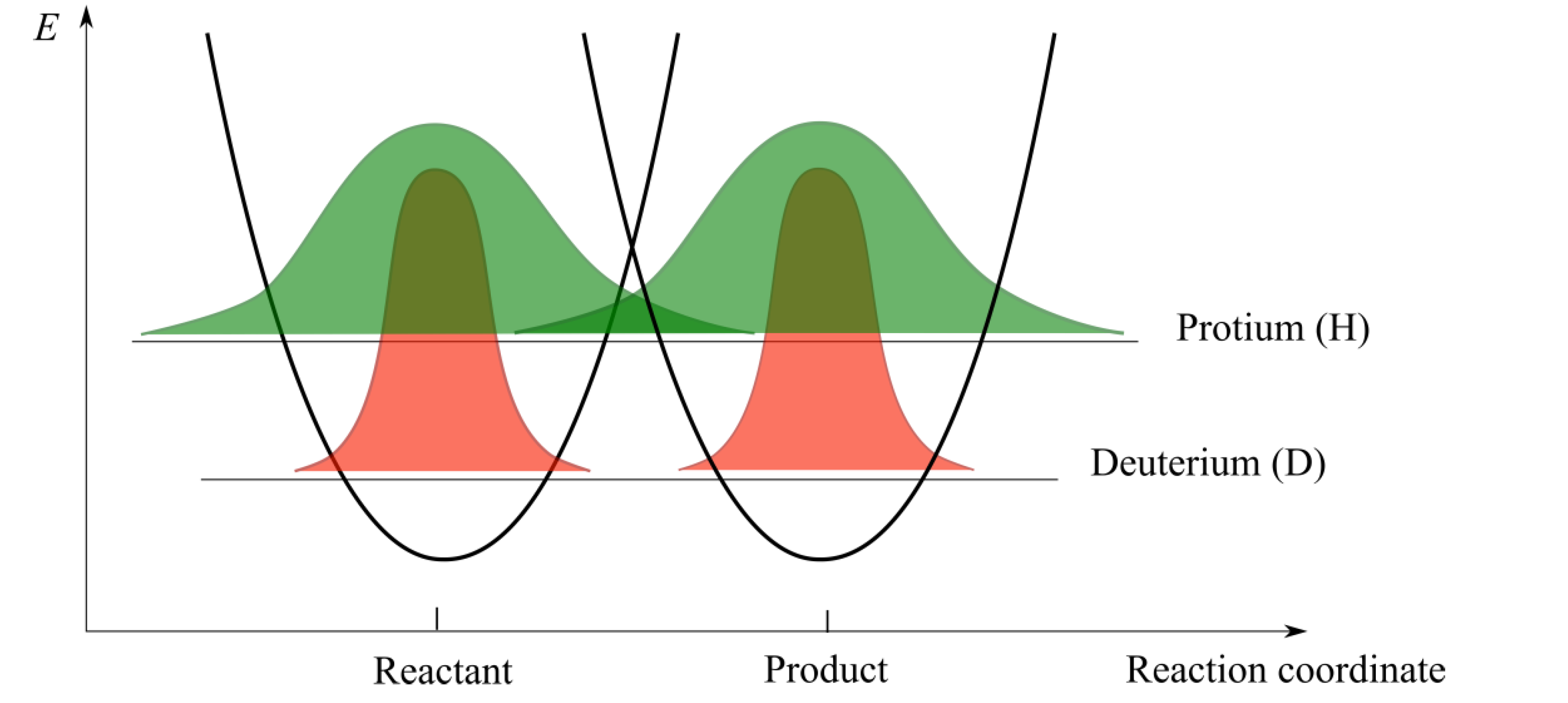
2.3. Hydrogen Tunnelling
3. Kinetic Isotope Effects and Hydrogen Tunnelling in Ascorbate PCET Oxidations
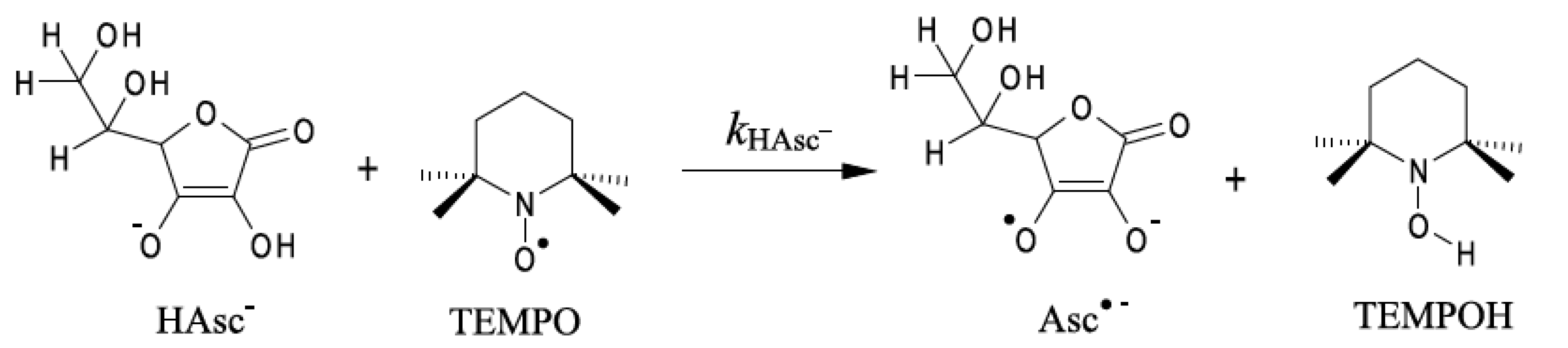
3.1. Reaction of Ascorbate with TEMPO Radical in the Mixed Water-Organic Cosolvent Systems
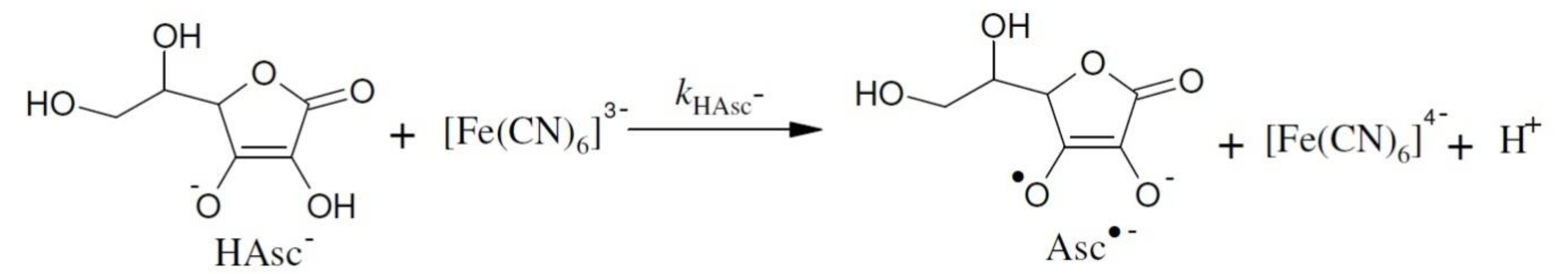
3.2. Reaction of Ascorbate with Hexacyanoferrate(III) in the Mixed Water-Organic Cosolvent Systems
3.3. Reaction of Ascorbate with Hexacyanoferrate(III) in More Concentrated Aqueous Solutions of the Salts
3.4. Reaction of Ascorbate with Ferricinium Ions in Aqueous Solutions of Sodium Chloride
3.5. Reaction of Ascorbate with Hexacyanoferrate(III) in Fairly Diluted Aqueous Solutions
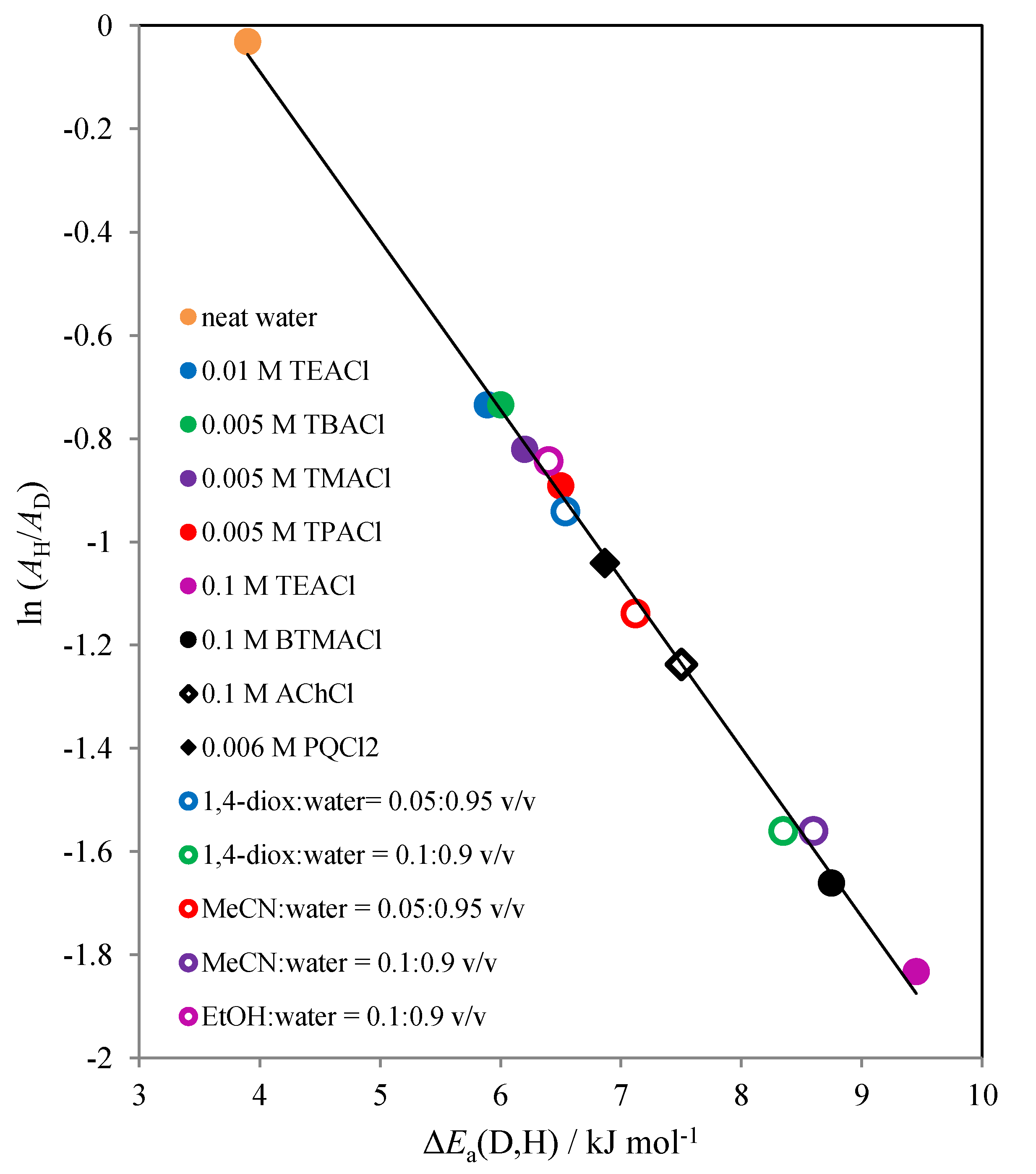
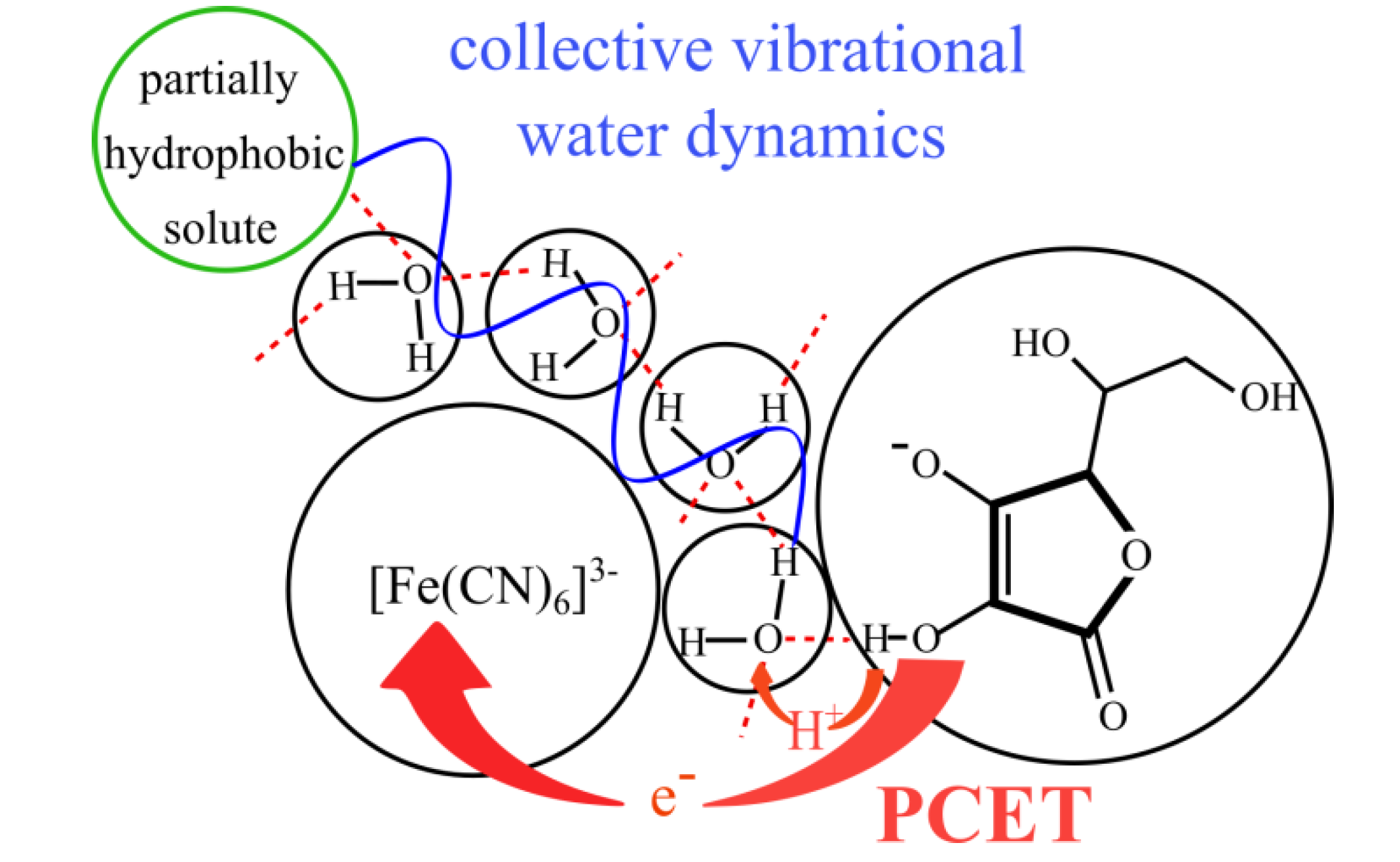
3.5.1. Reaction of Ascorbate with Hexacyanoferrate(III) in Fairly Diluted Aqueous Solutions: Hydrogen Tunnelling and the Vibrational Water Dynamics Connected?
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Melander, L.; Saunders, W.H. Reaction Rates of Isotopic Molecules; Krieger, Publ. Comp.: Malabar, FL, USA, 1987. [Google Scholar]
- Vogel, P.; Houk, K.N. Organic Chemistry: Theory, Reactivity and Mechanisms in Modern Synthesis; Wiley-VCH: Hoboken, NJ, USA, 2019. [Google Scholar]
- Bell, R.P. The tunnel effect in chemistry; Chapman and Hall: London, UK, 1980. [Google Scholar]
- HydrogenTransfer Reactions: Reductions and Beyond; Guillena, G., Ramon, D.J., Eds.; Springer: Berlin/Heidelberg, Germany; New York, NY, USA, 2016. [Google Scholar]
- Romesberg, F.E.; Schowen, R.L. Isotope Effects and Quantum Tunneling in Enzyme-Catalyzed Hydrogen Transfer. Part I. The Experimental Basis. In Advances in Physical Organic Chemistry; Richard, J.P., Ed.; Elsevier Academic Press: London, UK, 2004; pp. 27–77. [Google Scholar]
- Sen, A.; Kohen, A. Quantum Effects in Enzyme Kinetics. In Quantum Tunnelling in Enzyme-Catalysed Reactions; Scrutton, N.S., Allemann, R.K., Eds.; Royal Society of Chemistry: Cambridge, UK, 2009; pp. 164–181. [Google Scholar]
- Klinman, J.P.; Offenbacher, A.R. Understanding Biological Hydrogen Transfer Through the Lens of Temperature Dependent Kinetic Isotope Effects. Acc. Chem. Res. 2018, 51, 1966–1974. [Google Scholar] [CrossRef] [PubMed]
- Layfield, J.P.; Hammes-Schiffer, S. Hydrogen Tunneling in Enzymes and Biomimetic Models. Chem. Rev. 2014, 114, 3466–3494. [Google Scholar] [CrossRef] [PubMed]
- Truhlar, D.G. Tunneling in enzymatic and nonenzymatic hydrogen transfer reactions. J. Phys. Org. Chem. 2010, 23, 660–676. [Google Scholar] [CrossRef]
- Weinberg, D.R.; Gagliardi, C.J.; Hull, J.F.; Murphy, C.F.; Kent, C.A.; Westlake, B.C.; Paul, A.; Ess, D.H.; McCafferty, D.G.; Meyer, T.J. Proton-Coupled Electron Transfer. Chem. Rev. 2012, 112, 4016–4093. [Google Scholar] [CrossRef] [PubMed]
- Warren, J.J.; Tronic, T.A.; Mayer, J.M. Thermochemistry of Proton-Coupled Electron Transfer Reagents and its Implications. Chem. Rev. 2010, 110, 6961–7001. [Google Scholar] [CrossRef] [PubMed]
- Darcy, J.W.; Koronkiewicz, B.; Parada, G.A.; Mayer, J.M. A Continuum of Proton-Coupled Electron Transfer Reactivity. Acc. Chem. Res. 2018, 51, 2391–2399. [Google Scholar] [CrossRef] [PubMed]
- Hammes-Schiffer, S. Proton-Coupled Electron Transfer: Moving Together and Charging Forward. J. Am. Chem. Soc. 2015, 137, 8860–8871. [Google Scholar] [CrossRef]
- Hammes-Schiffer, S. Controlling Electrons and Protons through Theory: Molecular Electrocatalysts to Nanoparticles. Acc. Chem. Res. 2018, 51, 1975–1983. [Google Scholar] [CrossRef]
- Parada, G.A.; Goldsmith, Z.K.; Kolmar, S.; Pettersson Rimgard, B.; Mercado, B.Q.; Hammarström, L.; Hammes-Schiffer, S.; Mayer, J.M. Concerted proton-electron transfer reactions in the Marcus inverted region. Science 2019, 364, 471–475. [Google Scholar] [CrossRef]
- Slakman, B.L.; West, R.H. Kinetic solvent effects in organic reactions. J. Phys. Org. Chem. 2018, 32, e3904. [Google Scholar] [CrossRef]
- Sajenko, I.; Pilepić, V.; Jakobušić Brala, C.; Uršić, S. Solvent Dependence of the Kinetic Isotope Effect in the Reaction of Ascorbate with the 2,2,6,6-Tetramethylpiperidine-1-oxyl Radical: Tunnelling in a Small Molecule Reaction. J. Phys. Chem. A 2010, 114, 3423–3430. [Google Scholar] [CrossRef] [PubMed]
- Sajenko, I.; Pilepić, V.; Uršić, S. Modulating Hydrogen Tunnelling in Ascorbate Proton-Coupled Electron Transfers. Z. Phys. Chem. 2011, 225, 805–820. [Google Scholar] [CrossRef]
- Karković, A.; Brala, C.J.; Pilepić, V.; Uršić, S. Solvent-induced hydrogen tunnelling in ascorbate proton-coupled electron transfers. Tetr. Lett. 2011, 52, 1757–1761. [Google Scholar] [CrossRef]
- Jakobušić Brala, C.; Pilepić, V.; Sajenko, I.; Karković, A.; Uršić, S. Ions Can Move a Proton-Coupled Electron-Transfer Reaction into Tunneling Regime. Helv. Chim. Acta 2011, 94, 1718–1731. [Google Scholar] [CrossRef]
- Jakobušić Brala, C.; Karković, A.; Sajenko, I.; Pilepić, V.; Uršić, S. Sizeable Increase of Kinetic Isotope Effects and Tunnelling in Coupled Electron–Proton Transfers in Presence of the Quaternary Ions. PCET Processes and Hydrogen Tunnelling as a “Probe” for Structuring and Dynamical Phenomena in Water Solution. Z. Phys. Chem. 2012, 226, 29–46. [Google Scholar]
- Jakobušić Brala, C.; Karković, A.; Klepac, K.; Vučinović, A.M.; Pilepić, V.; Uršić, S. Small Molecule Tunnelling Systems: Variation of Isotope Effects. Z. Phys. Chem. 2011, 225, 821–841. [Google Scholar] [CrossRef]
- Karković Marković, A.; Jakobušić Brala, C.; Pilepić, V.; Uršić, S. Hydrogen Tunnelling as a Probe of the Involvement of Water Vibrational Dynamics in Aqueous Chemistry? Molecules 2020, 25, 172. [Google Scholar] [CrossRef]
- Ramasesha, K.; De Marco, L.; Mandal, A.; Tokmakoff, A. Water vibrations have strongly mixed intra- and intermolecular character. Nat. Chem. 2013, 5, 935–940. [Google Scholar] [CrossRef]
- De Marco, L.; Fournier, J.A.; Thämer, M.; Carpenter, W.; Tokmakoff, A. Anharmonic exciton dynamics and energy dissipation in liquid water from two-dimensional infrared spectroscopy. J. Chem. Phys. 2016, 145, 094501. [Google Scholar] [CrossRef]
- Fournier, J.A.; Carpenter, W.; De Marco, L.; Tokmakoff, A. Interplay of Ion–Water and Water–Water Interactions within the Hydration Shells of Nitrate and Carbonate Directly Probed with 2D IR Spectroscopy. J. Am. Chem. Soc. 2016, 138, 9634–9645. [Google Scholar] [CrossRef]
- Carpenter, W.B.; Fournier, J.A.; Biswas, R.; Voth, G.A.; Tokmakoff, A. Delocalization and Stretch-Bend Mixing of the HOH Bend in Liquid Water. J. Chem. Phys. 2017, 147, 084503. [Google Scholar] [CrossRef] [PubMed]
- Auer, B.M.; Skinner, J.L. IR and Raman Spectra of Liquid Water: Theory and Interpretation. J. Chem. Phys. 2008, 128, 224511. [Google Scholar] [CrossRef] [PubMed]
- Paarmann, A.; Hayashi, T.; Mukamel, S.; Miller, R.J.D. Nonlinear Response of Vibrational Excitons: Simulating the Two-Dimensional Infrared Spectrum of Liquid Water. J. Chem. Phys. 2009, 130, 204110. [Google Scholar] [CrossRef] [PubMed]
- Falvo, C.; Palmieri, B.; Mukamel, S. Coherent Infrared Multidimensional Spectra of the OH Stretching Band in Liquid Water Simulated by Direct Nonlinear Exciton Propagation. J. Chem. Phys. 2009, 130, 184501. [Google Scholar] [CrossRef]
- Kraemer, D.; Cowan, M.L.; Paarmann, A.; Huse, N.; Nibbering, E.T.J.; Elsaesser, T.; Dwayne Miller, R.J. Temperature Dependence of the Two-Dimensional Infrared Spectrum of Liquid H2O. Proc. Natl. Acad. Sci. USA 2008, 105, 437–442. [Google Scholar] [CrossRef]
- Imoto, S.; Xantheas, S.S.; Saito, S. Ultrafast Dynamics of Liquid Water: Frequency Fluctuations of the OH Stretch and the HOH Bend. J. Chem. Phys. 2013, 139, 044503. [Google Scholar] [CrossRef]
- Nagata, Y.; Yoshimune, S.; Hsieh, C.S.; Hunger, J.; Bonn, M. Ultrafast Vibrational Dynamics of Water Disentangled by Reverse Nonequilibrium Ab Initio Molecular Dynamics Simulations. Phys. Rev. X 2015, 5, 1–11. [Google Scholar]
- Cukier, R.I.; Nocera, D.G. Proton-coupled electron transfer. Annu. Rev. Phys. Chem. 1998, 49, 337–369. [Google Scholar] [CrossRef]
- Hammes-Schiffer, S.; Stuchebrukhov, A.A. Theory of Coupled Electron and Proton Transfer Reactions. Chem. Rev. 2010, 110, 6939–6960. [Google Scholar] [CrossRef]
- Nocera, D.G. The Artificial Leaf. Acc. Chem. Res. 2012, 45, 767–776. [Google Scholar] [CrossRef]
- Migliore, A.; Polizzi, N.F.; Therien, M.J.; Beratan, D.N. Biochemistry and Theory of Proton-Coupled Electron Transfer. Chem. Rev. 2014, 114, 3381–3465. [Google Scholar] [CrossRef] [PubMed]
- Gentry, E.C.; Knowles, R.R. Synthetic Applications of Proton-Coupled Electron Transfer. Acc. Chem. Res. 2016, 49, 1546–1556. [Google Scholar] [CrossRef] [PubMed]
- Galano, A.; Mazzone, G.; Alvarez-Diduk, R.; Marino, T.; Alvarez-Idaboy, J.R.; Russo, N. Food Antioxidants: Chemical Insights at the Molecular Level. Annu. Rev. Food Sci. Technol. 2016, 7, 335–352. [Google Scholar] [CrossRef] [PubMed]
- Kaila, V.R.I.; Verkhovsky, M.I.; Wikström, M. Proton-Coupled Electron Transfer in Cytochrome Oxidase. Chem. Rev. 2010, 110, 7062–7081. [Google Scholar] [CrossRef]
- Li, P.; Soudackov, A.V.; Hammes-Schiffer, S. Fundamental Insights into Proton-Coupled Electron Transfer in Soybean Lipoxygenase from Quantum Mechanical/Molecular Mechanical Free Energy Simulations. J. Am. Chem. Soc. 2018, 140, 3068–3076. [Google Scholar] [CrossRef]
- Cheng, T.; Shen, D.X.; Meng, M.; Mallick, S.; Cao, L.; Patmore, N.J.; Zhang, H.L.; Zou, S.F.; Chen, H.W.; Qin, Y.; et al. Efficient electron transfer across hydrogen bond interfaces by proton-coupled and -uncoupled pathways. Nat. Commun. 2019, 10, 1531. [Google Scholar] [CrossRef]
- Pannwitz, A.; Wenger, O.S. Proton-coupled multi-electron transfer and its relevance for artificial photosynthesis and photoredox catalysis. Chem. Commun. 2019, 55, 4004–4014. [Google Scholar] [CrossRef]
- Li, W.; Li, F.; Yang, H.; Wu, X.; Zhang, P.; Shan, Y.; Sun, L. A bio-inspired coordination polymer as outstanding water oxidation catalyst via second coordination sphere engineering. Nat. Commun. 2019, 10, 5074. [Google Scholar] [CrossRef]
- Meyer, T.J.; Sheridan, M.V.; Sherman, B.D. Mechanisms of molecular water oxidation in solution and on oxide surfaces. Chem. Soc. Rev. 2017, 46, 6148–6169. [Google Scholar] [CrossRef]
- Goldsmith, Z.K.; Soudackov, A.V.; Hammes-Schiffer, S. Theoretical analysis of the inverted region in photoinduced proton-coupled electron transfer. Faraday Discuss. 2019, 216, 363–378. [Google Scholar] [CrossRef]
- Dempsey, J.L. How a highly driven reaction hits the brakes. Science 2019, 364, 436–437. [Google Scholar] [CrossRef] [PubMed]
- Miller, J.L. Inverted kinetics seen in concerted charge transfer. Phys. Today 2019, 72, 16–19. [Google Scholar] [CrossRef]
- Markle, T.F.; Darcy, J.W.; Mayer, J.M. A new strategy to efficiently cleave and form C–H bonds using proton-coupled electron transfer. Sci. Adv. 2018, 4, eaat5776. [Google Scholar] [CrossRef] [PubMed]
- Qiu, G.; Knowles, R.R. Rate–Driving Force Relationships in the Multisite Proton-Coupled Electron Transfer Activation of Ketones. J. Am. Chem. Soc. 2019, 141, 2721–2730. [Google Scholar] [CrossRef] [PubMed]
- Zheng, S.; Gutiérrez-Bonet, Á.; Molander, G.A. Merging Photoredox PCET with Ni-Catalyzed Cross-Coupling: Cascade Amidoarylation of Unactivated Olefins. Chem 2019, 5, 339–352. [Google Scholar] [CrossRef]
- Qiu, G.; Knowles, R.R. Understanding Chemoselectivity in Proton-Coupled Electron Transfer: A Kinetic Study of Amide and Thiol Activation. J. Am. Chem. Soc. 2019, 141, 16574–16578. [Google Scholar] [CrossRef]
- Ashida, Y.; Arashiba, K.; Nakajima, K.; Nishibayashi, Y. Molybdenum-catalysed ammonia production with samarium diiodide and alcohols or water. Nature 2019, 568, 536–540. [Google Scholar] [CrossRef]
- Hammes-Schiffer, S.; Soudackov, A.V. Proton-Coupled Electron Transfer in Solution, Proteins, and Electrochemistry. J. Phys. Chem. B 2008, 112, 14108–14123. [Google Scholar] [CrossRef]
- Allemann, R.K.; Scrutton, N.S. Quantum Tunnelling in Enzyme-catalysed Reactions; Royal Society of Chemistry: Cambridge, UK, 2009; ISBN 978-0-85404-122-0. [Google Scholar]
- Maldonado-Domínguez, M.; Bím, D.; Fučíík, R.; Čurík, R.; Srnec, M. Reactive mode composition factor analysis of transition states: The case of coupled electron–proton transfers. Phys. Chem. Chem. Phys. 2019, 21, 24912–24918. [Google Scholar]
- Mayer, J.M. Understanding Hydrogen Atom Transfer: From Bond Strengths to Marcus Theory. Acc. Chem. Res. 2011, 44, 36–46. [Google Scholar] [CrossRef]
- Pu, J.; Gao, J.; Truhlar, D.G. Multidimensional Tunneling, Recrossing, and the Transmission Coefficient for Enzymatic Reactions. Chem. Rev. 2006, 106, 3140–3169. [Google Scholar] [CrossRef]
- Liu, T.; Tyburski, R.; Wang, S.; Fernández-Terán, R.; Ott, S.; Hammarström, L. Elucidating Proton-Coupled Electron Transfer Mechanisms of Metal Hydrides with Free Energy- and Pressure-Dependent Kinetics. J. Am. Chem. Soc. 2019, 141, 17245–17259. [Google Scholar] [CrossRef]
- Cape, J.L.; Bowman, M.K.; Kramer, D.M. Reaction Intermediates of Quinol Oxidation in a Photoactivatable System that Mimics Electron Transfer in the Cytochrome bc 1 Complex. J. Am. Chem. Soc. 2005, 127, 4208–4215. [Google Scholar] [CrossRef] [PubMed]
- Brunton, G.; Griller, D.; Barclay, L.R.C.; Ingold, K.U. Kinetic applications of electron paramagnetic resonance spectroscopy. 26. Quantum-mechanical tunneling in the isomerization of sterically hindered aryl radicals. J. Am. Chem. Soc. 1976, 98, 6803–6811. [Google Scholar] [CrossRef]
- Huynh, M.H.V.; Meyer, T.J. Colossal kinetic isotope effects in proton-coupled electron transfer. Proc. Natl. Acad. Sci. USA 2004, 101, 13138–13141. [Google Scholar] [CrossRef] [PubMed]
- Huynh, M.H.V.; Meyer, T.J. Proton-Coupled Electron Transfer from Phosphorus: A P−H/P−D Kinetic Isotope Effect of 178.4. Angew. Chem. Int. Ed. 2002, 41, 1395–1398. [Google Scholar] [CrossRef]
- Knapp, M.J.; Rickert, K.; Klinman, J.P. Temperature-Dependent Isotope Effects in Soybean Lipoxygenase-1: Correlating Hydrogen Tunneling with Protein Dynamics. J. Am. Chem. Soc. 2002, 124, 3865–3874. [Google Scholar] [CrossRef]
- Hu, S.; Soudackov, A.V.; Hammes-Schiffer, S.; Klinman, J.P. Enhanced Rigidification within a Double Mutant of Soybean Lipoxygenase Provides Experimental Support for Vibronically Nonadiabatic Proton-Coupled Electron Transfer Models. ACS Catal. 2017, 7, 3569–3574. [Google Scholar] [CrossRef]
- Soudackov, A.V.; Hammes-Schiffer, S. Proton-coupled electron transfer reactions: Analytical rate constants and case study of kinetic isotope effects in lipoxygenase. Faraday Discuss. 2016, 195, 171–189. [Google Scholar] [CrossRef]
- Zhang, Y.; de La Harpe, K.; Kohl, F.R.; Kohler, B. Isotopic substitution affects excited state branching in a DNA duplex in aqueous solution. Chem. Commun. 2019, 55, 4174–4177. [Google Scholar] [CrossRef]
- Goldsmith, Z.K.; Lam, Y.C.; Soudackov, A.V.; Hammes-Schiffer, S. Proton Discharge on a Gold Electrode from Triethylammonium in Acetonitrile: Theoretical Modeling of Potential-Dependent Kinetic Isotope Effects. J. Am. Chem. Soc. 2019, 141, 1084–1090. [Google Scholar] [CrossRef] [PubMed]
- Klinman, J.P.; Kohen, A. Hydrogen Tunneling Links Protein Dynamics to Enzyme Catalysis. Ann. Rev. Biochem. 2013, 82, 471–496. [Google Scholar] [CrossRef] [PubMed]
- Hay, S.; Scrutton, N.S. Good vibrations in enzyme-catalysed reactions. Nat. Chem. 2012, 4, 161–168. [Google Scholar] [CrossRef] [PubMed]
- Wu, A.; Mader, E.A.; Datta, A.; Hrovat, D.A.; Borden, W.T.; Mayer, J.M. Nitroxyl Radical Plus Hydroxylamine Pseudo Self-Exchange Reactions: Tunneling in Hydrogen Atom Transfer. J. Am. Chem. Soc. 2009, 131, 11985–11997. [Google Scholar] [CrossRef]
- Kandathil, S.M.; Driscoll, M.D.; Dunn, R.V.; Scrutton, N.S.; Hay, S. Proton tunnelling and promoting vibrations during the oxidation of ascorbate by ferricyanide? Phys. Chem. Chem. Phys. 2014, 16, 2256–2259. [Google Scholar] [CrossRef]
- Edwards, S.J.; Soudackov, A.V.; Hammes-Schiffer, S. Analysis of Kinetic Isotope Effects for Proton-Coupled Electron Transfer Reactions †. J. Phys. Chem. A 2009, 113, 2117–2126. [Google Scholar] [CrossRef]
- Ludlow, M.K.; Soudackov, A.V.; Hammes-Schiffer, S. Theoretical Analysis of the Unusual Temperature Dependence of the Kinetic Isotope Effect in Quinol Oxidation. J. Am. Chem. Soc. 2009, 131, 7094–7102. [Google Scholar] [CrossRef]
- Hydrogen-Transfer Reactions; Hynes, J.T., Klinman, J.P., Limbach, H.H., Schowen, R.L., Eds.; Wiley-VCH: Weinheim, Germany; Hoboken, NJ, USA, 2007. [Google Scholar]
- Nagel, Z.D.; Klinman, J.P. Update 1 of: Tunneling and Dynamics in Enzymatic Hydride Transfer. Chem. Rev. 2010, 110, PR41–PR67. [Google Scholar] [CrossRef]
- Johannissen, L.O.; Hay, S.; Scrutton, N.S. Nuclear quantum tunnelling in enzymatic reactions—An enzymologist’s perspective. Phys. Chem. Chem. Phys. 2015, 17, 30775–30782. [Google Scholar] [CrossRef]
- Kohen, A. Role of Dynamics in Enzyme Catalysis: Substantial versus Semantic Controversies. Acc. Chem. Res. 2015, 48, 466–473. [Google Scholar] [CrossRef]
- Hay, S.; Pudney, C.R.; Sutcliffe, M.J.; Scrutton, N.S. Solvent as a Probe of Active Site Motion and Chemistry during the Hydrogen Tunnelling Reaction in Morphinone Reductase. ChemPhysChem 2008, 9, 1875–1881. [Google Scholar] [CrossRef] [PubMed]
- Horitani, M.; Offenbacher, A.R.; Carr, C.A.M.; Yu, T.; Hoeke, V.; Cutsail, G.E.; Hammes-Schiffer, S.; Klinman, J.P.; Hoffman, B.M. 13C ENDOR Spectroscopy of Lipoxygenase–Substrate Complexes Reveals the Structural Basis for C–H Activation by Tunneling. J. Am. Chem. Soc. 2017, 139, 1984–1997. [Google Scholar] [CrossRef] [PubMed]
- Ley, D.; Gerbig, D.; Schreiner, P.R. Tunnelling control of chemical reactions – the organic chemist’s perspective. Org. Biomol. Chem. 2012, 10, 3781–3790. [Google Scholar] [CrossRef] [PubMed]
- Dix, E.J.; Herman, M.S.; Goodman, J.L. The 1,2-hydrogen rearrangement of methylchlorocarbene: Contribution of quantum mechanical tunneling. J. Am. Chem. Soc. 1993, 115, 10424–10425. [Google Scholar] [CrossRef]
- Raven, E.L.; Lad, L.; Sharp, K.H.; Mewies, M.; Moody, P.C. Defining substrate specificity and catalytic mechanism in ascorbate peroxidase. Biochem. Soc. Symp. 2004, 27–38. [Google Scholar]
- Njus, D.; Wigle, M.; Kelley, P.M.; Kipp, B.H.; Schlegel, H.B. Mechanism of Ascorbic Acid Oxidation by Cytochrome b561. Biochemistry 2001, 40, 11905–11911. [Google Scholar] [CrossRef]
- Frauenfelder, H.; Chen, G.; Berendzen, J.; Fenimore, P.W.; Jansson, H.; McMahon, B.H.; Stroe, I.R.; Swenson, J.; Young, R.D. A unified model of protein dynamics. Proc. Natl. Acad. Sci. USA 2009, 106, 5129–5134. [Google Scholar] [CrossRef]
| Solvent | KIE | ΔH‡/kJ mol−1 | ΔS‡/JK−1 mol−1 | ΔΔH‡/kJ mol−1 | ΔΔS‡/JK−1 mol−1 | Ah/Ad |
|---|---|---|---|---|---|---|
| Water | 24.2 (0.6) | 31.0 (0.4) | −134 (2) | 9.0 (0.6) | 4 (3) | 0.6 (0.2) |
| Water a | 21.9 (0.2) | 33.7 (0.4) | −124 (2) | 7.0 (0.4) | −2 (2) | 1.34 (0.15) |
| 1,4-diox:water b | 31.1 (1.1) | 23.9 (0.2) | −151 (1) | 8.2 (0.4) | −1 (1) | 1.2 (0.2) |
| MeCN:water b | 25.4 (0.3) | 31.1 (0.1) | −131 (1) | 8.4 (0.4) | 2 (2) | 0.82 (0.1) |
| MeCN:water a,b | 23.3 (0.1) | 29.4 (0.3) | −133 (1) | 11.6 (0.5) | 13 (1) | 0.22 (0.03) |
| Organic Cosolvent:Water = 1:1 v/v | KIE | ΔH‡/kJ mol−1 | ΔS‡/JK−1 mol−1 | ΔΔH‡/kJ mol−1 | ΔΔS‡/JK−1 mol−1 | Ah/Ad |
|---|---|---|---|---|---|---|
| MeCN:water | 8.25 (0.09) | 24.8 (0.3) | −137.3 (1.0) | 6.9 (0.4) | 5.9 (1.5) | 0.49 (0.09) |
| MeCN:water a | 6.55 (0.05) | |||||
| MeCN:water b | 6.70 (0.27) | |||||
| MeCN:water c | 5.86 (0.17) | 28.7 (0.2) | −104.0 (0.7) | 9.1 (0.3) | 16.1 (0.9) | 0.14 (0.02) |
| MeCN:water d | 7.81 (0.17) | |||||
| 1,4-diox:water | 7.84 (0.20) | 37.6 (0.2) | −99.0 (0.7) | 12.7 (0.4) | 25.6 (1.4) | 0.046 (0.008) |
| 1,4-diox:water e | 5.75 (0.14) | 35.3 (0.2) | −80.8 (0.8) | 10.9 (0.3) | 22.1 (1.0) | 0.07 (0.01) |
| 1,4-diox:water c | 4.39 (0.13) | 35.5 (0.3) | −76.5 (1.2) | 8.6 (0.6) | 16.5 (2.0) | 0.14 (0.03) |
| EtOH:water | 7.90 (0.22) | 26.4 (0.2) | −136.0 (0.7) | 11.9 (0.4) | 22.7 (1.2) | 0.065 (0.010) |
| Acetone:water | 8.59 (0.13) |
| Salt Added | KIE | ΔH‡/kJ mol−1 | ΔS‡/JK−1 mol−1 | ΔΔH‡/kJ mol−1 | ΔΔS‡/JK−1 mol−1 | Ah/Ad |
|---|---|---|---|---|---|---|
| KCl 0.5 M | 3.37 (0.07) | 20.3 (0.2) | −121.0 (0.8) | 1.8 (0.4) | −4.0 (1.2) | 1.61 (0.25) |
| NaCl 0.5 M | 3.78 (0.07) | 21.1 (0.4) | −121.2 (1.3) | 1.3 (0.6) | −6.9 (2.1) | 2.29 (0.60) |
| NaCl 1.0 M | 3.31 (0.07) | 21.1 (0.2) | −118.3 (0.6) | 1.5 (0.4) | −4.7 (1.3) | 1.77 (0.29) |
| Na-acetate 0.5 M | 3.77 (0.12) | |||||
| LiCl 0.5 M | 3.93 (0.18) | |||||
| TEACl 0.5 M | 9.49 (0.12) | 21.6 (0.3) | −131.0 (1.0) | 11.1 (0.5) | 19.0 (2.2) | 0.10 (0.02) |
| TEACl 1.0 M | 10.08 (0.07) | |||||
| TMACl 0.5 M | 6.48 (0.08) | 15.9 (0.2) | −141.0 (1.0) | 6.9 (0.3) | 8.0 (1.4) | 0.35 (0.06) |
| TMACl 1.0 M | 6.79 (0.13) | |||||
| BTMACl 0.5 M | 7.53 (0.11) | 15.7 (0.2) | −141.8 (0.6) | 8.6 (0.2) | 12.3 (0.8) | 0.23 (0.02) |
| BTMACl 1.0 M | 8.01 (0.19) |
| KIE | ΔH‡/kJ mol−1 | ΔS‡/JK−1 mol−1 | ΔΔH‡/kJ mol−1 | ΔΔS‡/JK−1 mol−1 | Ah/Ad | |
|---|---|---|---|---|---|---|
| Water | 1.91 (0.02) | 45.0 (0.2) | −26.0 (0.7) | 2.1 (0.3) | 1.8 (0.9) | 0.80 (0.09) |
| 0.5 M NaCl | 2.01 (0.03) | 45.6 (0.1) | −32.7 (0.4) | 3.4 (0.4) | 5.5 (1.5) | 0.52 (0.10) |
| MeCN:water a | 1.48 (0.03) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Karković Marković, A.; Jakobušić Brala, C.; Pilepić, V.; Uršić, S. Kinetic Isotope Effects and Hydrogen Tunnelling in PCET Oxidations of Ascorbate: New Insights into Aqueous Chemistry? Molecules 2020, 25, 1443. https://doi.org/10.3390/molecules25061443
Karković Marković A, Jakobušić Brala C, Pilepić V, Uršić S. Kinetic Isotope Effects and Hydrogen Tunnelling in PCET Oxidations of Ascorbate: New Insights into Aqueous Chemistry? Molecules. 2020; 25(6):1443. https://doi.org/10.3390/molecules25061443
Chicago/Turabian StyleKarković Marković, Ana, Cvijeta Jakobušić Brala, Viktor Pilepić, and Stanko Uršić. 2020. "Kinetic Isotope Effects and Hydrogen Tunnelling in PCET Oxidations of Ascorbate: New Insights into Aqueous Chemistry?" Molecules 25, no. 6: 1443. https://doi.org/10.3390/molecules25061443
APA StyleKarković Marković, A., Jakobušić Brala, C., Pilepić, V., & Uršić, S. (2020). Kinetic Isotope Effects and Hydrogen Tunnelling in PCET Oxidations of Ascorbate: New Insights into Aqueous Chemistry? Molecules, 25(6), 1443. https://doi.org/10.3390/molecules25061443






