Assessment of Hepatoprotective Effect of Chokeberry Juice in Rats Treated Chronically with Carbon Tetrachloride
Abstract
1. Introduction
2. Results
2.1. Antioxidant Status Parameters
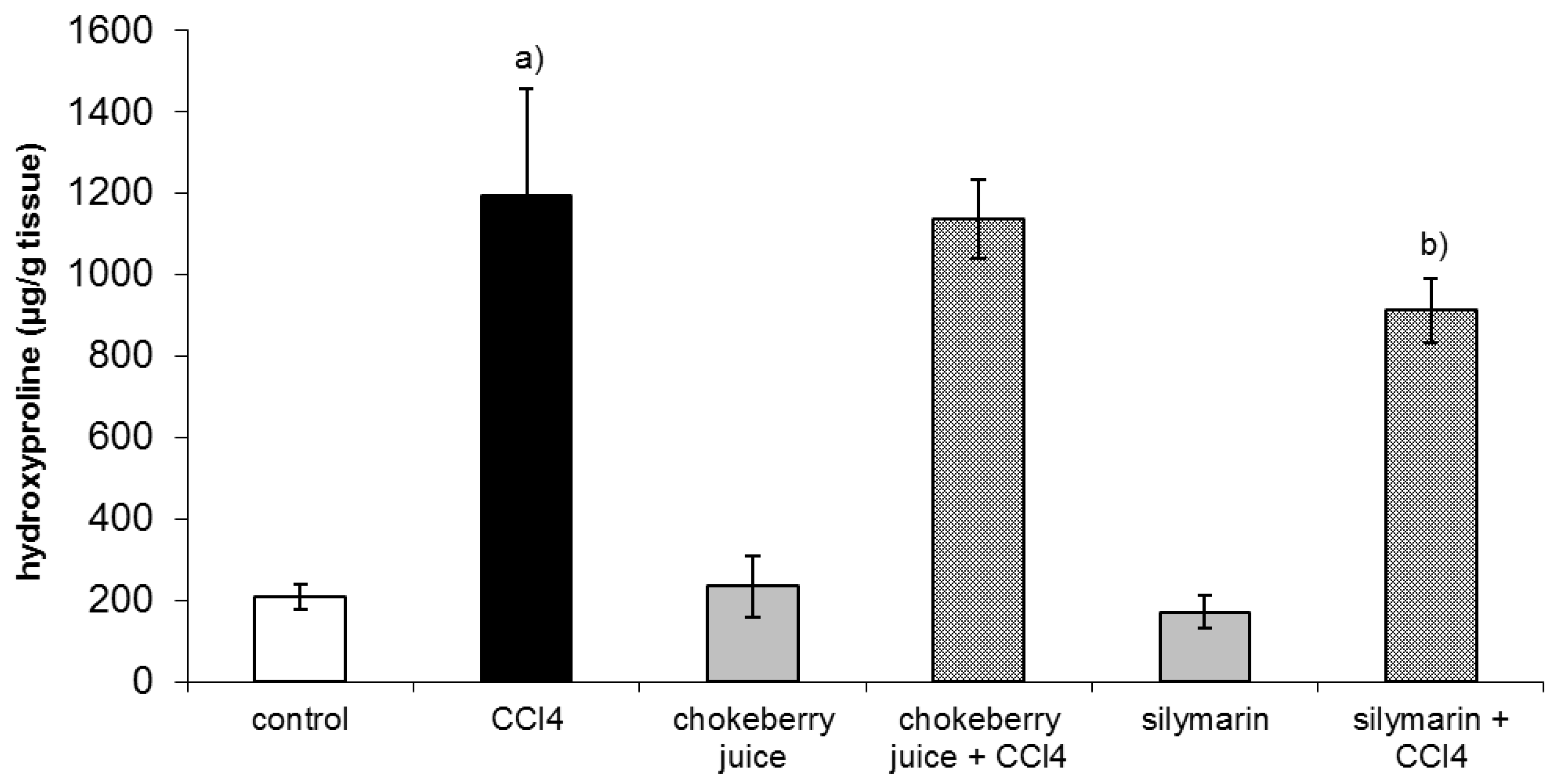
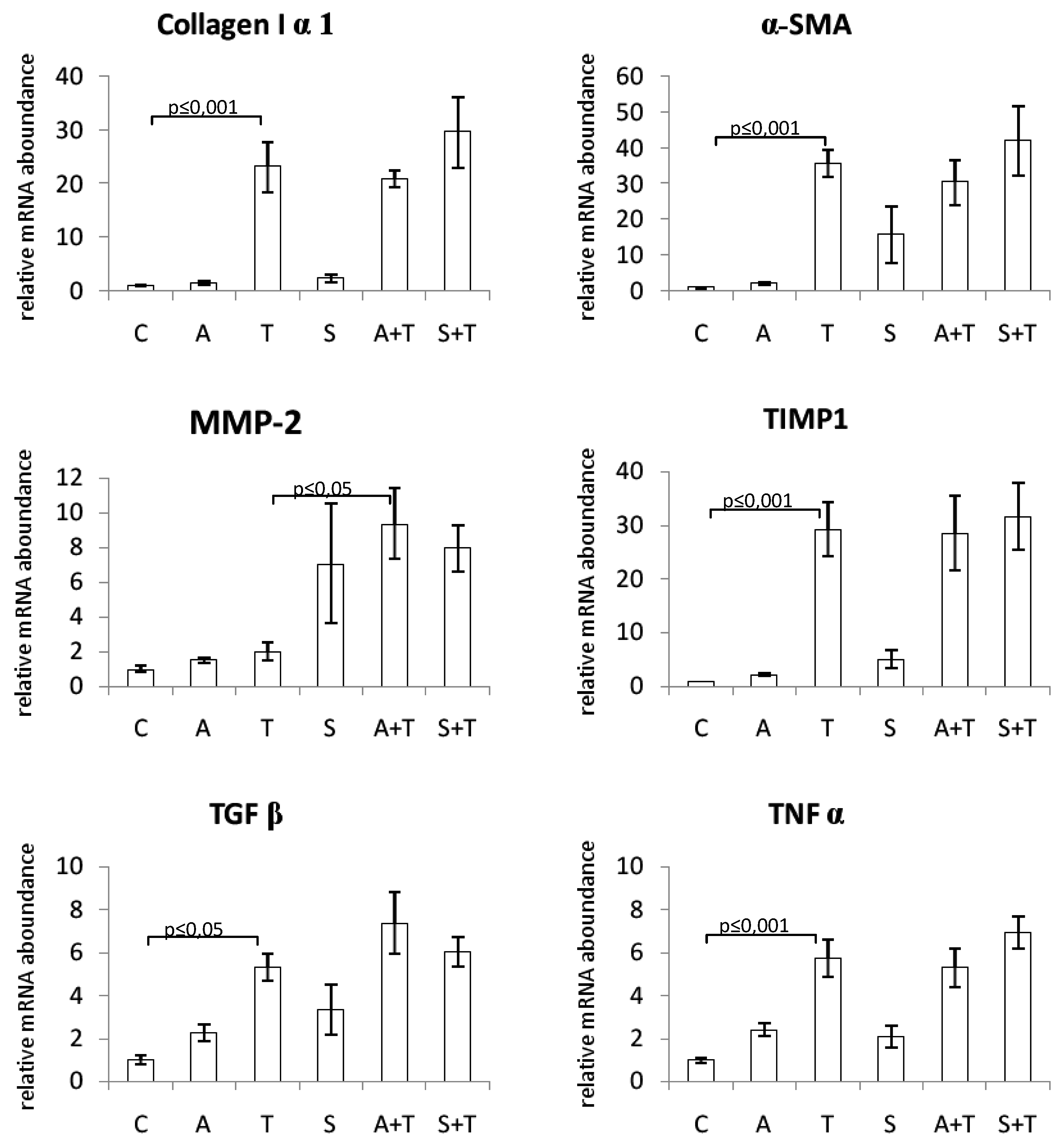
2.2. Clinical Chemistry, Hydroxyproline Concentration, and Gene Expression
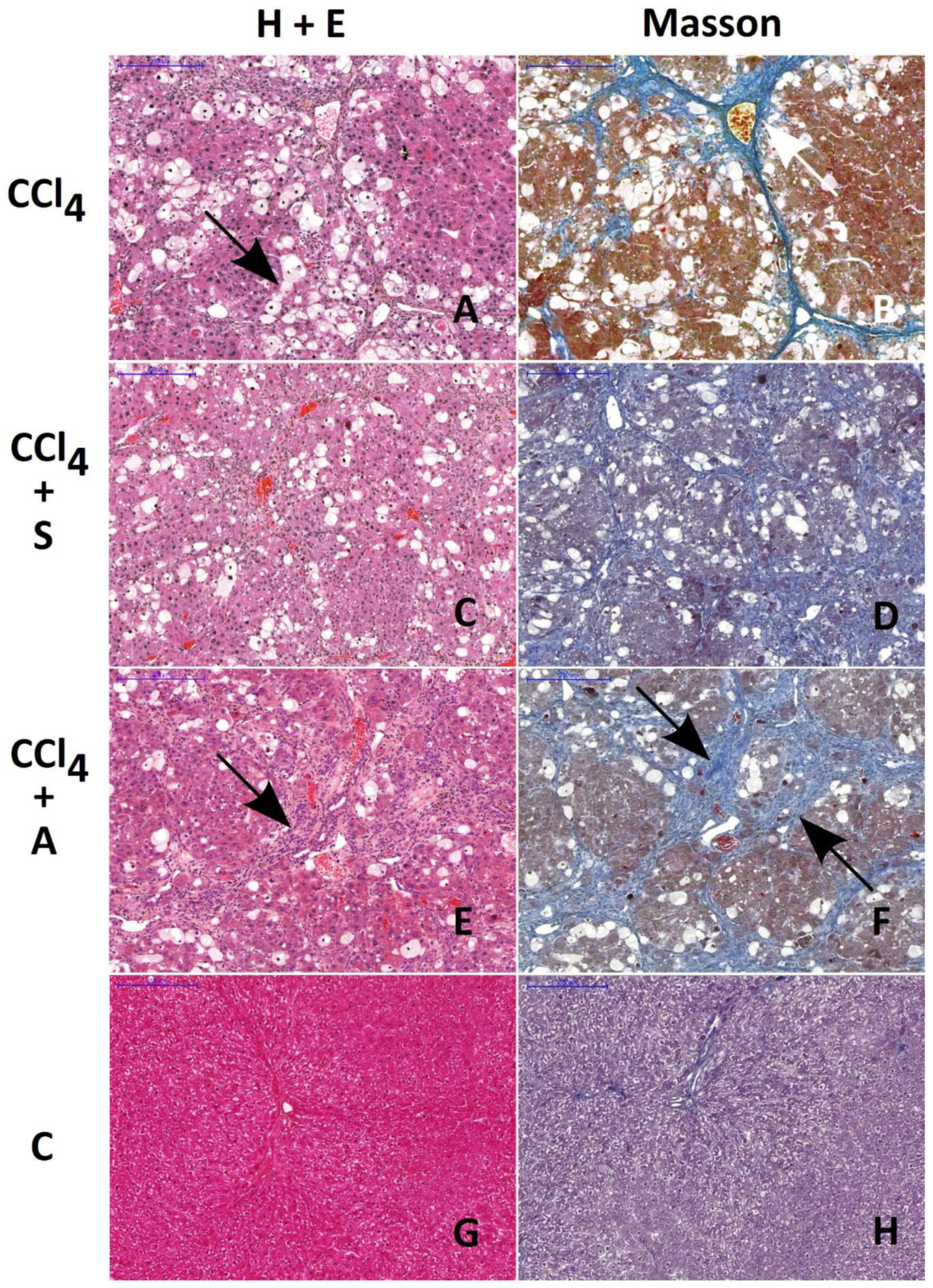
2.3. Morphology and Morphometry
3. Discussion
4. Materials and Methods
4.1. Chemicals
4.2. Experimental Design
4.3. Biochemical Assays
4.4. Real-Time Quantitative PCR (RQ-PCR) Analysis
4.5. Histological Examination
4.6. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Asrani, S.K.; Devarbhavi, H.; Eaton, J.; Kamath, P.S. Burden of liver diseases in the world. J. Hepatol. 2019, 70, 151–171. [Google Scholar] [CrossRef] [PubMed]
- Schuppan, D. Liver fibrosis: Common mechanisms and antifibrotic therapies. Clin. Res. Hepatol. Gastroenterol. 2015, 39 (Suppl. 1), S51–S59. [Google Scholar] [CrossRef]
- Parola, M.; Robino, G. Oxidative stress-related molecules and liver fibrosis. J. Hepatol. 2001, 35, 297–306. [Google Scholar] [CrossRef]
- Gebhardt, R. Oxidative stress, plant-derived antioxidants and liver fibrosis. Planta Med. 2002, 68, 289–296. [Google Scholar] [CrossRef]
- Zhang, X.; Han, X.; Yin, L.; Xu, L.; Qi, Y.; Xu, Y.; Sun, H.; Lin, Y.; Liu, K.; Peng, J. Potent effects of dioscin against liver fibrosis. Sci. Rep. 2015, 5, 9713. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Wang, X.; Han, C.; Xing, G.; Zhou, L.; Li, G.; Niu, Y. Astragaloside IV suppresses collagen production of activated hepatic stellate cells via oxidative stress-mediated p38 MAPK pathway. Free Radic. Biol. Med. 2013, 60, 168–176. [Google Scholar] [CrossRef]
- Huang, Q.; Huang, R.; Zhang, S.; Lin, J.; Wei, L.; He, M.; Zhuo, L.; Lin, X. Protective effect of genistein isolated from Hydrocotyle sibthorpioides on hepatic injury and fibrosis induced by chronic alcohol in rats. Toxicol. Lett. 2013, 217, 102–110. [Google Scholar] [CrossRef]
- Federico, A.; Dallio, M.; Loguercio, C. Silymarin/Silybin and Chronic Liver Disease: A Marriage of Many Years. Molecules 2017, 22, 191. [Google Scholar] [CrossRef]
- Oszmiański, J.; Wojdylo, A. Aronia melanocarpa phenolics and their antioxidant activity. Eur. Food Res. Technol. 2005, 221, 809–813. [Google Scholar] [CrossRef]
- Kulling, S.E.; Rawel, H.M. Chokeberry (Aronia melanocarpa)—A review on the characteristic components and potential health effects. Planta Med. 2008, 74, 1625–1634. [Google Scholar] [CrossRef]
- Kokotkiewicz, A.; Jaremicz, Z.; Luczkiewicz, M. Aronia plants: A review of traditional use, biological activities, and perspectives for modern medicine. J. Med. Food 2010, 13, 255–269. [Google Scholar] [CrossRef] [PubMed]
- Jurikova, T.; Mlcek, J.; Skrovankova, S.; Sumczynski, D.; Sochor, J.; Hlavacova, I.; Snopek, L.; Orsavova, J. Fruits of Black Chokeberry Aronia melanocarpa in the Prevention of Chronic Diseases. Molecules 2017, 22, 944. [Google Scholar] [CrossRef] [PubMed]
- Ho, G.T.; Bräunlich, M.; Austarheim, I.; Wangensteen, H.; Malterud, K.E.; Slimestad, R.; Barsett, H. Immunomodulating activity of Aronia melanocarpa polyphenols. Int. J. Mol. Sci. 2014, 15, 11626–11636. [Google Scholar] [CrossRef] [PubMed]
- Appel, K.; Meiser, P.; Millán, E.; Collado, J.A.; Rose, T.; Gras, C.C.; Carle, R.; Muñoz, E. Chokeberry (Aronia melanocarpa (Michx.) Elliot) concentrate inhibits NF-κB and synergizes with selenium to inhibit the release of pro-inflammatory mediators in macrophages. Fitoterapia 2015, 105, 73–82. [Google Scholar] [CrossRef] [PubMed]
- Martin, D.A.; Taheri, R.; Brand, M.H.; Draghi, A.; Sylvester, F.A.; Bolling, B.W. Anti-inflammatory activity of aronia berry extracts in murine splenocytes. J. Funct. Foods 2014, 8, 68–75. [Google Scholar] [CrossRef]
- Qin, B.; Anderson, R.A. An extract of chokeberry attenuates weight gain and modulates insulin, adipogenic and inflammatory signalling pathways in epididymal adipose tissue of rats fed a fructose-rich diet. Br. J. Nutr. 2012, 108, 581–587. [Google Scholar] [CrossRef]
- Valcheva-Kuzmanova, S.; Kuzmanov, K.; Tancheva, S.; Belcheva, A. Hypoglycemic and hypolipidemic effects of Aronia melanocarpa fruit juice in streptozotocin-induced diabetic rats. Methods Find Exp. Clin. Pharmacol. 2007, 29, 101–105. [Google Scholar] [CrossRef]
- Kim, B.; Park, Y.; Wegner, C.J.; Bolling, B.W.; Lee, J. Polyphenol-rich black chokeberry (Aronia melanocarpa) extract regulates the expression of genes critical for intestinal cholesterol flux in Caco-2 cells. J. Nutr. Biochem. 2013, 24, 1564–1570. [Google Scholar] [CrossRef]
- Kim, N.H.; Jegal, J.; Kim, Y.N.; Heo, J.D.; Rho, J.R.; Yang, M.H.; Jeong, E.J. Chokeberry Extract and Its Active Polyphenols Suppress Adipogenesis in 3T3-L1 Adipocytes and Modulates Fat Accumulation and Insulin Resistance in Diet-Induced Obese Mice. Nutrients 2018, 10. [Google Scholar] [CrossRef]
- Kim, J.H.; Auger, C.; Kurita, I.; Anselm, E.; Rivoarilala, L.O.; Lee, H.J.; Lee, K.W.; Schini-Kerth, V.B. Aronia melanocarpa juice, a rich source of polyphenols, induces endothelium-dependent relaxations in porcine coronary arteries via the redox-sensitive activation of endothelial nitric oxide synthase. Nitric Oxide 2013, 35, 54–64. [Google Scholar] [CrossRef]
- Daskalova, E.; Delchev, S.; Peeva, Y.; Vladimirova-Kitova, L.; Kratchanova, M.; Kratchanov, C.; Denev, P. Antiatherogenic and Cardioprotective Effects of Black Chokeberry (Aronia melanocarpa) Juice in Aging Rats. Evid. Based Complement Alternat. Med. 2015, 2015, 717439. [Google Scholar] [CrossRef] [PubMed]
- Naruszewicz, M.; Laniewska, I.; Millo, B.; Dłuzniewski, M. Combination therapy of statin with flavonoids rich extract from chokeberry fruits enhanced reduction in cardiovascular risk markers in patients after myocardial infraction (MI). Atherosclerosis 2007, 194, e179–e184. [Google Scholar] [CrossRef] [PubMed]
- Lala, G.; Malik, M.; Zhao, C.; He, J.; Kwon, Y.; Giusti, M.M.; Magnuson, B.A. Anthocyanin-rich extracts inhibit multiple biomarkers of colon cancer in rats. Nutr. Cancer 2006, 54, 84–93. [Google Scholar] [CrossRef] [PubMed]
- Ohgami, K.; Ilieva, I.; Shiratori, K.; Koyama, Y.; Jin, X.H.; Yoshida, K.; Kase, S.; Kitaichi, N.; Suzuki, Y.; Tanaka, T.; et al. Anti-inflammatory effects of aronia extract on rat endotoxin-induced uveitis. Invest Ophthalmol Vis. Sci. 2005, 46, 275–281. [Google Scholar] [CrossRef]
- Pawłowicz, P.; Stachowiak, G.; Bielak, A.; Wilczyński, J. Administration of natural anthocyanins derived from chokeberry (aronia melanocarpa) extract in the treatment of oligospermia in males with enhanced autoantibodies to oxidized low density lipoproteins (oLAB). The impact on fructose levels. Ginekol. Pol. 2001, 72, 983–988. [Google Scholar]
- Lee, H.Y.; Weon, J.B.; Jung, Y.S.; Kim, N.Y.; Kim, M.K.; Ma, C.J. Cognitive-Enhancing Effect of Aronia melanocarpa Extract against Memory Impairment Induced by Scopolamine in Mice. Evid. Based Complement. Alternat. Med. 2016, 2016, 6145926. [Google Scholar] [CrossRef]
- Tomić, M.; Ignjatović, Đ.; Tovilović-Kovačević, G.; Krstić-Milošević, D.; Ranković, S.; Popović, T.; Glibetić, M. Reduction of anxiety-like and depression-like behaviors in rats after one month of drinking Aronia melanocarpa berry juice. Food Funct. 2016, 7, 3111–3120. [Google Scholar] [CrossRef]
- Wei, J.; Zhang, G.; Zhang, X.; Xu, D.; Gao, J.; Fan, J.; Zhou, Z. Anthocyanins from Black Chokeberry (Aronia melanocarpa Elliot) Delayed Aging-Related Degenerative Changes of Brain. J. Agric. Food Chem. 2017, 65, 5973–5984. [Google Scholar] [CrossRef]
- Valcheva-Kuzmanova, S.; Borisova, P.; Galunska, B.; Krasnaliev, I.; Belcheva, A. Hepatoprotective effect of the natural fruit juice from Aronia melanocarpa on carbon tetrachloride-induced acute liver damage in rats. Exp. Toxicol. Pathol. 2004, 56, 195–201. [Google Scholar] [CrossRef]
- Kujawska, M.; Ignatowicz, E.; Ewertowska, M.; Oszmianski, J.; Jodynis-Liebert, J. Protective effect of chokeberry on chemical-induced oxidative stress in rat. Hum. Exp. Toxicol. 2011, 30, 199–208. [Google Scholar] [CrossRef]
- Jodynis-Liebert, J.; Adamska, T.; Ewertowska, M.; Bylka, W.; Matławska, I. Aquilegia vulgaris extract attenuates carbon tetrachloride-induced liver fibrosis in rats. Exp. Toxicol. Pathol. 2009, 61, 443–451. [Google Scholar] [CrossRef] [PubMed]
- Boušová, I.; Hájek, J.; Dršata, J.; Skálová, L. Naturally occurring flavonoids as inhibitors of purified cytosolic glutathione S-transferase. Xenobiotica 2012, 42, 872–879. [Google Scholar] [CrossRef] [PubMed]
- Bartholomaeus, A.R.; Bolton, R.; Ahokas, J.T. Inhibition of rat liver cytosolic glutathione S-transferase by silybin. Xenobiotica 1994, 24, 17–24. [Google Scholar] [CrossRef] [PubMed]
- Das, D.; Pemberton, P.W.; Burrows, P.C.; Gordon, C.; Smith, A.; McMahon, R.F.; Warnes, T.W. Antioxidant properties of colchicine in acute carbon tetrachloride induced rat liver injury and its role in the resolution of established cirrhosis. Biochim. Biophys. Acta 2000, 1502, 351–362. [Google Scholar] [CrossRef]
- Domitrović, R.; Jakovac, H.; Tomac, J.; Sain, I. Liver fibrosis in mice induced by carbon tetrachloride and its reversion by luteolin. Toxicol. Appl. Pharmacol. 2009, 241, 311–321. [Google Scholar] [CrossRef]
- Chávez, E.; Reyes-Gordillo, K.; Segovia, J.; Shibayama, M.; Tsutsumi, V.; Vergara, P.; Moreno, M.G.; Muriel, P. Resveratrol prevents fibrosis, NF-kappaB activation and TGF-beta increases induced by chronic CCl4 treatment in rats. J. Appl. Toxicol. 2008, 28, 35–43. [Google Scholar] [CrossRef]
- Mourelle, M.; Muriel, P.; Favari, L.; Franco, T. Prevention of CCl4-induced liver cirrhosis by silymarin. Fundam. Clin. Pharmacol. 1989, 3, 183–191. [Google Scholar] [CrossRef]
- Boigk, G.; Stroedter, L.; Herbst, H.; Waldschmidt, J.; Riecken, E.O.; Schuppan, D. Silymarin retards collagen accumulation in early and advanced biliary fibrosis secondary to complete bile duct obliteration in rats. Hepatology 1997, 26, 643–649. [Google Scholar] [CrossRef]
- Jia, J.D.; Bauer, M.; Cho, J.J.; Ruehl, M.; Milani, S.; Boigk, G.; Riecken, E.O.; Schuppan, D. Antifibrotic effect of silymarin in rat secondary biliary fibrosis is mediated by downregulation of procollagen alpha1(I) and TIMP-1. J. Hepatol. 2001, 35, 392–398. [Google Scholar] [CrossRef]
- Muriel, P.; Moreno, M.G. Effects of silymarin and vitamins E and C on liver damage induced by prolonged biliary obstruction in the rat. Basic Clin. Pharmacol. Toxicol. 2004, 94, 99–104. [Google Scholar] [CrossRef]
- Sidor, A.; Gramza-Michałowska, A. Black Chokeberry Aronia melanocarpa L.-A Qualitative Composition, Phenolic Profile and Antioxidant Potential. Molecules 2019, 24, 3710. [Google Scholar] [CrossRef] [PubMed]
- Dulundu, E.; Ozel, Y.; Topaloglu, U.; Toklu, H.; Ercan, F.; Gedik, N.; Sener, G. Grape seed extract reduces oxidative stress and fibrosis in experimental biliary obstruction. J. Gastroenterol. Hepatol. 2007, 22, 885–892. [Google Scholar] [CrossRef] [PubMed]
- Shin, M.-O.; Yoon, S.; Moon, J.-O. The proanthocyanidins inhibit dimethylnitrosamine-induced liver damage in rats. Arch. Pharm. Res. 2010, 33, 167–173. [Google Scholar] [CrossRef] [PubMed]
- Shin, M.-O.; Moon, J.-O. Effect of dietary supplementation of grape skin and seeds on liver fibrosis induced by dimethylnitrosamine in rats. Nutr. Res. Pract. 2010, 4, 369–374. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Tsukada, S.; Parsons, C.J.; Rippe, R.A. Mechanisms of liver fibrosis. Clin. Chim. Acta 2006, 364, 33–60. [Google Scholar] [CrossRef] [PubMed]
- van de Casteele, M.; Roskams, T.; van der Elst, I.; van Pelt, J.F.; Fevery, J.; Nevens, F. Halofuginone can worsen liver fibrosis in bile duct obstructed rats. Liver Int. 2004, 24, 502–509. [Google Scholar] [CrossRef]
- Sanz, M.J.; Ferrandiz, M.L.; Cejudo, M.; Terencio, M.C.; Gil, B.; Bustos, G.; Ubeda, A.; Gunasegaran, R.; Alcaraz, M.J. Influence of a series of natural flavonoids on free radical generating systems and oxidative stress. Xenobiotica 1994, 24, 689–699. [Google Scholar] [CrossRef]
- Sedlak, J.; Lindsay, R.H. Estimation of total, protein-bound, and nonprotein sulfhydryl groups in tissue with Ellman’s reagent. Anal. Biochem. 1968, 25, 192–205. [Google Scholar] [CrossRef]
- Mohandas, J.; Marshall, J.J.; Duggin, G.G.; Horvath, J.S.; Tiller, D.J. Low activities of glutathione-related enzymes as factors in the genesis of urinary bladder cancer. Cancer Res. 1984, 44, 5086–5091. [Google Scholar]
- Jodynis-Liebert, J.; Murias, M.; Błoszyk, E. Effect of sesquiterpene lactones on antioxidant enzymes and some drug-metabolizing enzymes in rat liver and kidney. Planta Med. 2000, 66, 199–205. [Google Scholar] [CrossRef]
- Chomczynski, P.; Sacchi, N. Single-step method of RNA isolation by acid guanidinium thiocyanate-phenol-chloroform extraction. Anal. Biochem. 1987, 162, 156–159. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds. are not available from the authors. |
| Treatment | LPO | Enzymes | ||||
|---|---|---|---|---|---|---|
| SOD | CAT | GPx | GR | GST | ||
| Controls | 0.21 ± 0.03 | 32.9 ± 4.8 | 4.13 ± 0.33 | 821.0 ± 83.0 | 54.4 ± 6.3 | 903.3 ± 63.9 |
| CCl4 | 0.31 ± 0.06 a | 7.2 ± 1.2 a | 2.12 ±0.34 a | 427.7 ± 48.9 a | 46.0 ± 5.3 | 585.8 ± 62.4 a |
| (T) | (↑48%) | (↓78%) | (↓49%) | (↓48%) | (↓16%) | (↓35%) |
| Chokeberry | 0. 21 ± 0.02 | 29.1 ± 3.1 | 4.83 ± 0.43 a | 882.2 ± 65.3 a | 44.8 ± 7.0 | 755.6 ± 29.4 a |
| juice | — | (↓12%) | (↑17%) | — | (↓18%) | (↓16%) |
| (A) | ||||||
| – | ||||||
| CCl4 + | 0.14 ± 0.03 b | 12.3 ± 2.4 b | 1.79 ± 0.27 | 440.5 ± 41.4 | 54.9 ± 3.7 b | 576.7 ± 52.2 |
| chokeberry | (↓55%) | (↑71%) | (↓16%) | (↑19%) | (↑19%) | — |
| juice | ||||||
| (A + T) | ||||||
| Silymarin | 0.14 ± 0,02 a | 35.0 ± 6.1 | 4.73 ± 0.25a | 978.2 ± 84.1 a | 51.1 ± 7.4 | 820.2 ± 57.3 |
| (S) | (↓33%) | — | (↑15%) | (↑19%) | — | — |
| CCl4 + | 0.16 ± 0.03 b | 19.7 ± 4.7 b | 2.27 ± 0.21 | 535.6 ± 46.0 b | 54.7 ± 3.1 b | 472.4 ± 60.3 b |
| silymarin | (↓48%) | (↑174%) | — | (↑25%) | (↑19%) | (↓19%) |
| (S+T) | ||||||
| Treatment | ||||||
|---|---|---|---|---|---|---|
| Parameters | Controls | CCl4 | Chokeberry | CCl4 + | Silymarin | CCl4 + |
| Juice | Chokeberry | Silymarin | ||||
| (T) | (A) | Juice | (S) | (S + T) | ||
| (A + T) | ||||||
| ALT | 60.1 ± 10.2 | 886 ± 95.7 a | 56.3 ± 9.4 | 838 ± 104 | 49.4 ± 4.5 | 881 ± 89.0 |
| (IU/L) | (↑1374%) | |||||
| AST | 78.7 ± 12.3 | 3401 ± 319a | 86.6 ± 4.0 | 1722 ± 194 b | 76.2 ± 15.7 | 1378 ± 216 b |
| (IU/L) | (↑3766%) | (↓49%) | (↓55%) | |||
| ALP | 190.9 ± 22.7 | 844 ± 84.1 a | 209.0 ± 35.7 | 772 ± 54.6 | 222 ± 16.2 | 820 ± 72.2 |
| (IU/L) | (↑342%) | |||||
| LDH | 183.0 ± 20.0 | 804 ± 96.3 a | 161.7 ± 24.4 | 364 ± 31.5 b | 154 ± 28.0 | 624 ± 55.9 b |
| (IU/L) | (↑339%) | (↓55%) | (↓22%) | |||
| GGT | 10.1 ± 1.0 | 36.9 ± 6.5 a | 8.2 ± 1.2 | 30.2 ± 2.0 b | 8.7 ± 1.0 | 20.3 ± 2.4 b |
| (IU/L) | (↑364%) | (↓18%) | (↓45%) | |||
| SDH | 7.9 ± 1.2 | 415 ± 67.5 a | 6.9 ± 1.4 | 234 ± 36.2 b | 3.9 ± 0.7 | 358 ± 38.3 |
| (IU/L) | (↑5136%) | (↓44%) | ||||
| Bilirubin | 0.32 ± 0.1 | 2.17 ± 0,2 a | 0.38 ± 0.1 | 1.48 ± 0.19 b | 0.29 ± 0.1 | 1.51 ± 0.2 b |
| (mg/dL) | (↑584%) | (↓32%) | (↓30%) | |||
| Cholesterol | 92.7 ± 4.3 | 123 ± 7.3 a | 83.6 ± 5.0 a | 112 ± 4.3 b | 83.8 ± 4.5 a | 109 ± 7.3 b |
| (mg/dL) | (↑33%) | (↓10%) | (↓9%) | (↓10%) | (↓11%) |
| Parameters | Treatment | |||||
|---|---|---|---|---|---|---|
| Controls | CCl4 | Chokeberry | CCl4 + | Silymarin | CCl4 + | |
| Juice | Chokeberry | Silymarin | ||||
| (T) | (A) | Juice | (S) | (S + T) | ||
| (A + T) | ||||||
| Fibrosis | 0.04 ± 0.01 | 0.18 ± 0.03 | 0.04 ± 0.01 | 0.23 ± 0.05 | 0.04 ± 0.01 | 0.09 ± 0.01 a,b |
| Infiltration | - | 4.6 ± 2.2 | - | 14.3 ± 6.6 c | - | 6.2 ± 2.2 |
| Vacuolization | - | 208 ± 23 | - | 65 ± 14 d | - | 179 ± 39 |
| Compounds | |
|---|---|
| Polymeric procyanidins | 224.90 |
| Neochlorogenic acid | 46.40 |
| Chlorogenic acid | 35.40 |
| Quercetin 3-rutinoside | 1.85 |
| Cyanidin 3-galactoside | 1.62 |
| Quercetin 3-galactoside | 1.42 |
| Quercetin 3-glucoside | 1.10 |
| Quercetin 3-vicianoside | 1.10 |
| Quercetin 3-robinobioside | 1.05 |
| Cyanidin 3-arabinoside | 0.66 |
| Cyanidin 3-glucoside | 0.16 |
| Cyanidin 3-xyloside | 0.20 |
| Total | 315.90 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Piotrowska-Kempisty, H.; Nowicki, M.; Jodynis-Liebert, J.; Kurpik, M.; Ewertowska, M.; Adamska, T.; Oszmiański, J.; Kujawska, M. Assessment of Hepatoprotective Effect of Chokeberry Juice in Rats Treated Chronically with Carbon Tetrachloride. Molecules 2020, 25, 1268. https://doi.org/10.3390/molecules25061268
Piotrowska-Kempisty H, Nowicki M, Jodynis-Liebert J, Kurpik M, Ewertowska M, Adamska T, Oszmiański J, Kujawska M. Assessment of Hepatoprotective Effect of Chokeberry Juice in Rats Treated Chronically with Carbon Tetrachloride. Molecules. 2020; 25(6):1268. https://doi.org/10.3390/molecules25061268
Chicago/Turabian StylePiotrowska-Kempisty, Hanna, Michał Nowicki, Jadwiga Jodynis-Liebert, Monika Kurpik, Małgorzata Ewertowska, Teresa Adamska, Jan Oszmiański, and Małgorzata Kujawska. 2020. "Assessment of Hepatoprotective Effect of Chokeberry Juice in Rats Treated Chronically with Carbon Tetrachloride" Molecules 25, no. 6: 1268. https://doi.org/10.3390/molecules25061268
APA StylePiotrowska-Kempisty, H., Nowicki, M., Jodynis-Liebert, J., Kurpik, M., Ewertowska, M., Adamska, T., Oszmiański, J., & Kujawska, M. (2020). Assessment of Hepatoprotective Effect of Chokeberry Juice in Rats Treated Chronically with Carbon Tetrachloride. Molecules, 25(6), 1268. https://doi.org/10.3390/molecules25061268










