Blue Emitting Star-Shaped and Octasilsesquioxane-Based Polyanions Bearing Boron Clusters. Photophysical and Thermal Properties †
Abstract
1. Introduction
2. Results and Discussion
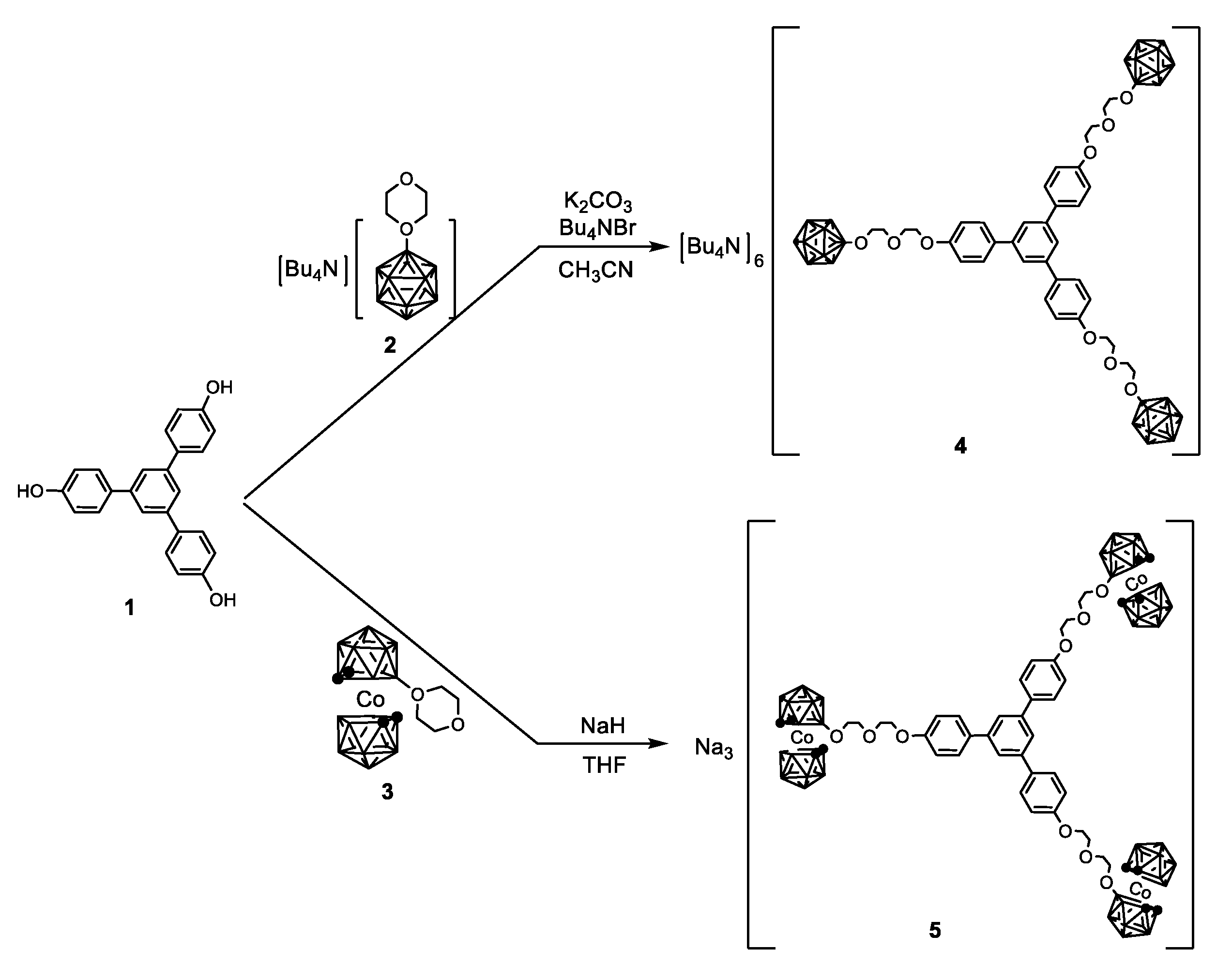
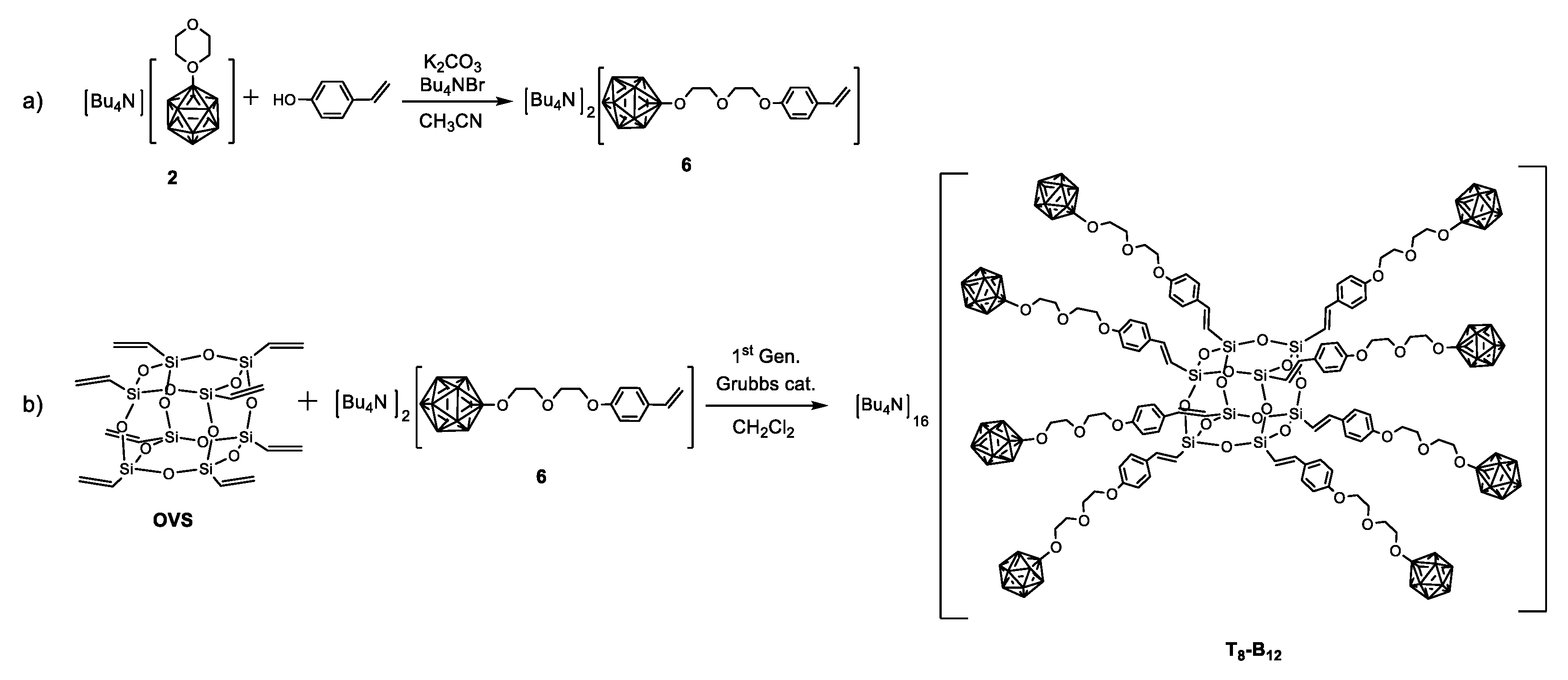
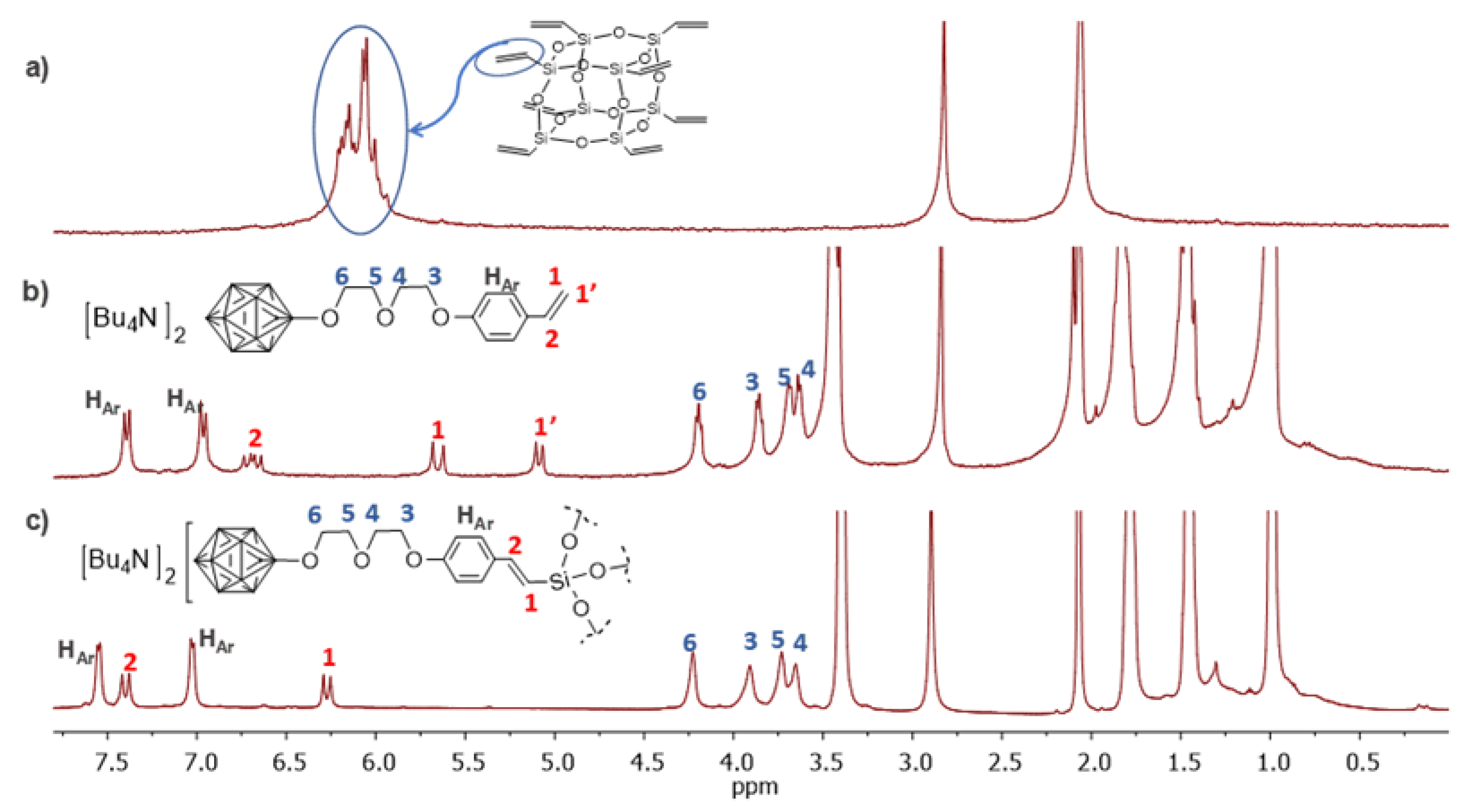
2.1. Synthesis and Characterisation
2.2. Photophysical Properties
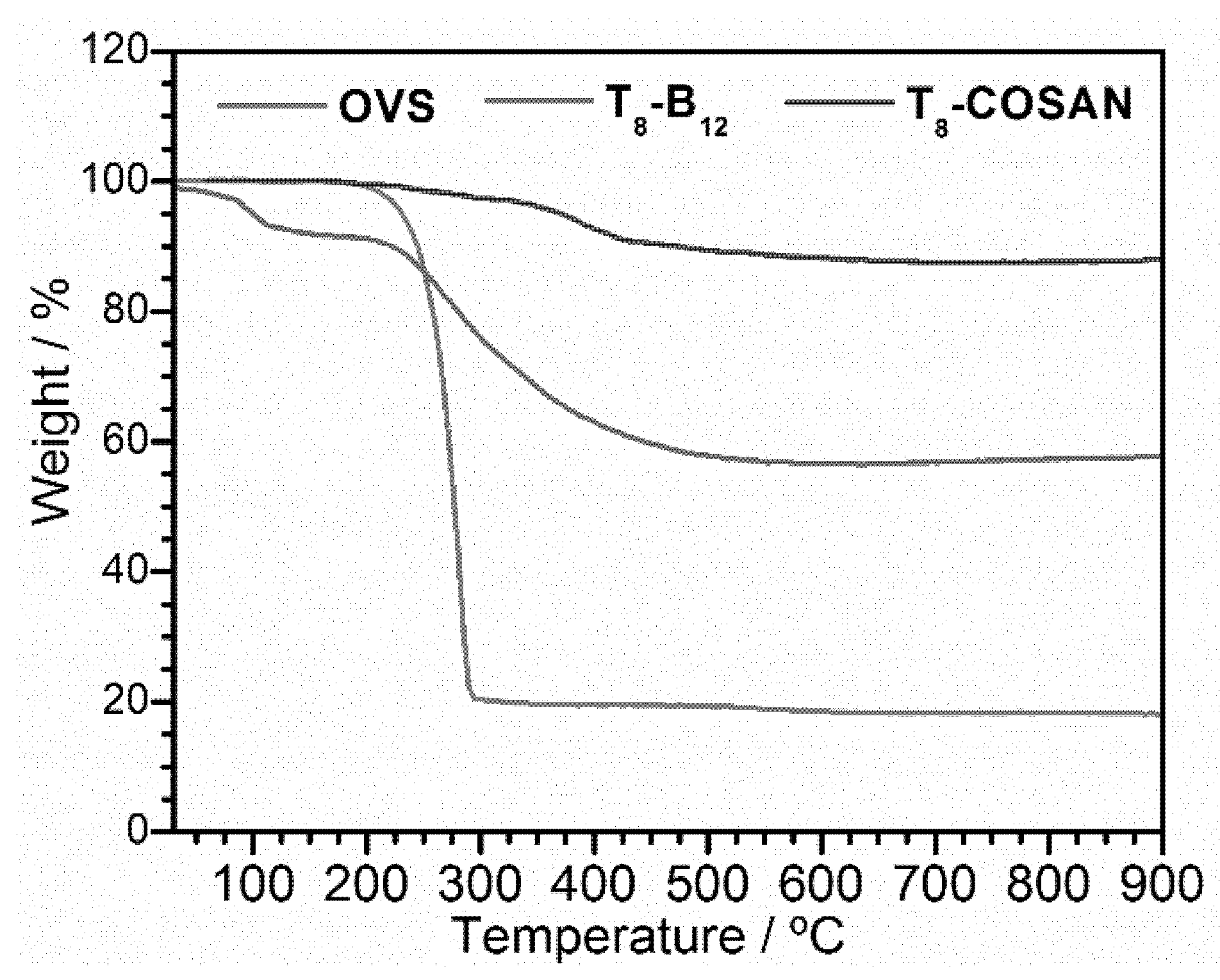
2.3. Thermal Stability of Anionic-Boron Clusters Containing T8
3. Materials and Methods
3.1. Materials
3.2. Instrumentation
3.3. Synthesis of Derivatives 4, 5, 6, and T8-B12
3.3.1. Synthesis of 4
3.3.2. Synthesis of 5
3.3.3. Synthesis of 6
3.3.4. Synthesis of T8-B12
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Núñez, R.; Tarrès, M.; Ferrer-Ugalde, A.; Fabrizi de Biani, F.; Teixidor, F. Electrochemistry and Photoluminescence of Icosahedral Carboranes, Boranes, Metallacarboranes, and Their Derivatives. Chem. Rev. 2016, 116, 14307–14378. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, S.; Thilagar, P. Boron clusters in luminescent materials. Chem. Commun. 2016, 52, 1070–1093. [Google Scholar] [CrossRef] [PubMed]
- Poater, J.; Solà, M.; Viñas, C.; Teixidor, F. π Aromaticity and Three-Dimensional Aromaticity: Two sides of the Same Coin? Angew. Chem. Int. Ed. 2014, 53, 12191–12195. [Google Scholar] [CrossRef] [PubMed]
- Poater, J.; Solà, M.; Viñas, C.; Teixidor, F. Back Cover: A Simple Link between Hydrocarbon and Borohydride Chemistries. Chem. Eur. J. 2013, 19, 4372. [Google Scholar] [CrossRef]
- Kahlert, J.; Stammler, H.-G.; Neumann, B.; Harder, R.A.; Weber, L.; Fox, M.A. Crystal Structures of the Carborane Dianions [1,4-(PhCB10H10C)2C6H4]2− and [1,4-(PhCB10H10C)2C6F4]2− and the Stabilizing Role of the para-Phenylene Unit on 2 n+3 Skeletal Electron Clusters. Angew. Chem. 2014, 126, 3776–3779. [Google Scholar] [CrossRef]
- Sillanpaa, R.; Kivekas, R.; Teixidor, F.; Viñas, C.; Núñez, R. 1-Diisopropylphosphino-2-phenyl-1,2-dicarba-closo-dodecaborane(12). Acta Crystallogr. 1996, 52, 2223–2225. [Google Scholar] [CrossRef]
- Cabrera-Gonzlez, J.; Ferrer-Ugalde, A.; Bhattacharyya, S.; Chaari, M.; Teixidor, F.; Gierschner, J.; Núñez, R. Fluorescent carborane-vinylstilbene functionalised octasilsesquioxanes: Synthesis, structural, thermal and photophysical properties. J. Mater. Chem. C 2017, 5, 10211–10219. [Google Scholar] [CrossRef]
- Ferrer-Ugalde, A.; Juárez-Pérez, E.J.; Teixidor, F.; Viñas, C.; Núñez, R. Synthesis, Characterization, and Thermal Behavior of Carboranyl–Styrene Decorated Octasilsesquioxanes: Influence of the Carborane Clusters on Photoluminescence. Chem. Eur. J. 2013, 19, 17021–17030. [Google Scholar] [CrossRef]
- González-Campo, A.; Ferrer-Ugalde, A.; Viñas, C.; Teixidor, F.; Sillanpää, R.; Rodríguez-Romero, J.; Santillan, R.; Farfán, N.; Núñez, R. A Versatile Methodology for the Controlled Synthesis of Photoluminescent High-Boron-Content Dendrimers. Chem. Eur. J. 2013, 19, 6299–6312. [Google Scholar] [CrossRef]
- Scholz, M.; Hey-Hawkins, E. Carbaboranes as Pharmacophores: Properties, Synthesis, and Application Strategies. Chem. Rev. 2011, 111, 7035–7062. [Google Scholar] [CrossRef]
- Issa, F.; Kassiou, M.; Rendina, L.M. Boron in Drug Discovery: Carboranes as Unique Pharmacophores in Biologically Active Compounds. Chem. Rev. 2011, 111, 5701–5722. [Google Scholar] [CrossRef] [PubMed]
- Řezáčová, P.; Pokorná, J.; Brynda, J.; Kožíšek, M.; Cígler, P.; Lepšík, M.; Fanfrlík, J.; Řezáč, J.; Grantz Šašková, K.; Sieglová, I.; et al. Design of HIV Protease Inhibitors Based on Inorganic Polyhedral Metallacarboranes. J. Med. Chem. 2009, 52, 7132–7141. [Google Scholar] [CrossRef] [PubMed]
- Sivaev, I.B.; Bregadze, V.V. Polyhedral Boranes for Medical Applications: Current Status and Perspectives. Eur. J. Inorg. Chem. 2009, 2009, 1433–1450. [Google Scholar] [CrossRef]
- Kožíšek, M.; Cígler, P.; Lepšík, M.; Fanfrlík, J.; Řezáčová, P.; Brynda, J.; Pokorná, J.; Plešek, J.; Grüner, B.; Grantz Šašková, K.; et al. Inorganic Polyhedral Metallacarborane Inhibitors of HIV Protease: A New Approach to Overcoming Antiviral Resistance. J. Med. Chem. 2008, 51, 4839–4843. [Google Scholar] [CrossRef]
- Cígler, P.; Kožíšek, M.; Řezáčová, P.; Brynda, J.; Otwinowski, Z.; Pokorná, J.; Plešek, J.; Grüner, B.; Dolečková-Marešová, L.; Máša, M.; et al. From nonpeptide toward noncarbon protease inhibitors: Metallacarboranes as specific and potent inhibitors of HIV protease. PNAS 2005, 102, 15394–15399. [Google Scholar] [CrossRef]
- Valliant, J.F.; Guenther, K.J.; King, A.S.; Morel, P.; Schaffer, P.; Sogbein, O.O.; Stephenson, K.A. The medicinal chemistry of carboranes. Coord. Chem. Rev. 2002, 232, 173–230. [Google Scholar] [CrossRef]
- Hawthorne, M.F.; Maderna, A. Applications of Radiolabeled Boron Clusters to the Diagnosis and Treatment of Cancer. (Chem. Rev. 1999, 99, 3421−3434. Published on the Web Nov. 13, 1999). Chem. Rev. 2000, 100, 1165. [Google Scholar] [CrossRef]
- Wu, X.; Guo, J.; Jia, W.; Zhao, J.; Jia, D.; Shan, H. Highly-efficient solid-state emission of tethered anthracene-o-carborane dyads and their visco- and thermo-chromic luminescence properties. Dyes and Pigments 2019, 162, 855–862. [Google Scholar] [CrossRef]
- Wu, X.; Guo, J.; Quan, Y.; Jia, W.; Jia, D.; Chen, Y.; Xie, Z. Cage carbon-substitute does matter for aggregation-induced emission features of o-carborane-functionalized anthracene triads. J. Mater. Chem. C 2018, 6, 4140–4149. [Google Scholar] [CrossRef]
- Wu, X.; Guo, J.; Cao, Y.; Zhao, J.; Jia, W.; Chen, Y.; Jia, D. Mechanically triggered reversible stepwise tricolor switching and thermochromism of anthracene-o-carborane dyad. Chem. Sci. 2018, 9, 5270–5277. [Google Scholar] [CrossRef]
- Tu, D.; Leong, P.; Guo, S.; Yan, H.; Lu, C.; Zhao, Q. Highly Emissive Organic Single-Molecule White Emitters by Engineering o-Carborane-Based Luminophores. Angew. Chem. Int. Ed. 2017, 56, 11370–11374. [Google Scholar] [CrossRef] [PubMed]
- Naito, H.; Nishino, K.; Morisaki, Y.; Tanaka, K.; Chujo, Y. Highly-efficient solid-state emissions of anthracene–o-carborane dyads with various substituents and their thermochromic luminescence properties. J. Mater. Chem. C 2017, 5, 10047–10054. [Google Scholar] [CrossRef]
- Naito, H.; Nishino, K.; Morisaki, Y.; Tanaka, K.; Chujo, Y. Solid-State Emission of the Anthracene-o-Carborane Dyad from the Twisted-Intramolecular Charge Transfer in the Crystalline State. Angew. Chem. Int. Ed. 2017, 56, 254–259. [Google Scholar] [CrossRef] [PubMed]
- Tu, D.; Leong, P.; Li, Z.; Hu, R.; Shi, C.; Zhang, K.Y.; Yan, H.; Zhao, Q. A carborane-triggered metastable charge transfer state leading to spontaneous recovery of mechanochromic luminescence. Chem. Commun. 2016, 52, 12494–12497. [Google Scholar] [CrossRef]
- Kim, Y.; Park, S.; Lee, Y.H.; Jung, J.; Yoo, S.; Lee, M.H. Homoleptic Tris-Cyclometalated Iridium Complexes with Substituted o-Carboranes: Green Phosphorescent Emitters for Highly Efficient Solution-Processed Organic Light-Emitting Diodes. Inorg. Chem. 2016, 55, 909–917. [Google Scholar] [CrossRef]
- Furue, R.; Nishimoto, T.; Park, I.S.; Lee, J.; Yasuda, T. Aggregation-Induced Delayed Fluorescence Based on Donor/Acceptor-Tethered Janus Carborane Triads: Unique Photophysical Properties of Nondoped OLEDs. Angew. Chem. Int. Ed. 2016, 55, 7171–7175. [Google Scholar] [CrossRef]
- Kahlert, J.; Bohling, L.; Brockhinke, A.; Stammler, H.-G.; Neumann, B.; Rendina, L.M.; Low, P.J.; Weber, L.; Fox, M.A. Syntheses and reductions of C-dimesitylboryl-1,2-dicarba-closo-dodecaboranes. Dalton Trans. 2015, 44, 9766–9781. [Google Scholar] [CrossRef]
- Naito, H.; Morisaki, Y.; Chujo, Y. o-Carborane-Based Anthracene: A Variety of Emission Behaviors. Angew. Chem. Int. Ed. 2015, 54, 5084–5087. [Google Scholar] [CrossRef]
- Zhu, L.; Lv, W.; Liu, S.; Yan, H.; Zhao, Q.; Huang, W. Carborane enhanced two-photon absorption of tribranched fluorophores for fluorescence microscopy imaging. Chem. Commun. 2013, 49, 10638–10640. [Google Scholar] [CrossRef]
- Wee, K.-R.; Cho, Y.-J.; Jeong, S.; Kwon, S.; Lee, J.-D.; Suh, I.-H.; Kang, S.O. Carborane-Based Optoelectronically Active Organic Molecules: Wide Band Gap Host Materials for Blue Phosphorescence. J. Am. Chem. Soc. 2012, 134, 17982–17990. [Google Scholar] [CrossRef]
- Chaari, M.; Kelemen, Z.; Choquesillo-Lazarte, D.; Gaztelumendi, N.; Teixidor, F.; Viñas, C.; Nogués, C.; Núñez, R. Efficient blue light emitting materials based on m-carborane–anthracene dyads. Structure, photophysics and bioimaging studies. Biomater. Sci. 2019, 7, 5324–5337. [Google Scholar] [CrossRef] [PubMed]
- Bellomo, C.; Chaari, M.; Cabrera-González, J.; Blangetti, M.; Lombardi, C.; Deagostino, A.; Viñas, C.; Gaztelumendi, N.; Nogués, C.; Nuñez, R.; et al. Carborane-BODIPY Dyads: New Photoluminescent Materials through an Efficient Heck Coupling. Chem. Eur. J. 2018, 24, 15622–15630. [Google Scholar] [CrossRef] [PubMed]
- Chaari, M.; Cabrera-González, J.; Kelemen, Z.; Viñas, C.; Ferrer-Ugalde, A.; Choquesillo-Lazarte, D.; Ben Salah, A.; Teixidor, F.; Núñez, R. Luminescence properties of carborane-containing distyrylaromatic systems. J. Organomet. Chem. 2018, 865, 206–213. [Google Scholar] [CrossRef]
- Chaari, M.; Kelemen, Z.; Planas, J.G.; Teixidor, F.; Choquesillo-Lazarte, D.; Ben Salah, A.; Viñas, C.; Núñez, R. Photoluminescence in m-carborane–anthracene triads: A combined experimental and computational study. J. Mater. Chem. C 2018, 6, 11336–11347. [Google Scholar] [CrossRef]
- Ferrer-Ugalde, A.; Cabrera-González, J.; Juárez-Pérez, E.J.; Teixidor, F.; Pérez-Inestrosa, E.; Montenegro, J.M.; Sillanpää, R.; Haukka, M.; Núñez, R. Carborane-stilbene dyads: The influence of substituents and cluster isomers on photoluminescence properties. Dalton Trans. 2017, 46, 2091–2104. [Google Scholar] [CrossRef]
- Cabrera-González, J.; Bhattacharyya, S.; Milián-Medina, B.; Teixidor, F.; Farfán, N.; Arcos-Ramos, R.; Vargas-Reyes, V.; Gierschner, J.; Núñez, R. Tetrakis{[(p-dodecacarboranyl)methyl]stilbenyl}ethylene: A Luminescent Tetraphenylethylene (TPE) Core System. Eur. J. Inorg. Chem. 2017, 2017, 4575–4580. [Google Scholar] [CrossRef]
- Cabrera-González, J.; Viñas, C.; Haukka, M.; Bhattacharyya, S.; Gierschner, J.; Núñez, R. Photoluminescence in Carborane–Stilbene Triads: A Structural, Spectroscopic, and Computational Study. Chem. Eur. J. 2016, 22, 13588–13598. [Google Scholar] [CrossRef]
- Ferrer-Ugalde, A.; González-Campo, A.; Viñas, C.; Rodríguez-Romero, J.; Santillan, R.; Farfán, N.; Sillanpää, R.; Sousa-Pedrares, A.; Núñez, R.; Teixidor, F. Fluorescence of New o-Carborane Compounds with Different Fluorophores: Can it be Tuned? Chem. Eur. J. 2014, 20, 9940–9951. [Google Scholar] [CrossRef]
- Ferrer-Ugalde, A.; Juárez-Pérez, E.J.; Teixidor, F.; Viñas, C.; Sillanpää, R.; Pérez-Inestrosa, E.; Núñez, R. Synthesis and Characterization of New Fluorescent Styrene-Containing Carborane Derivatives: The Singular Quenching Role of a Phenyl Substituent. Chem. Eur. J. 2012, 18, 544–553. [Google Scholar] [CrossRef]
- Tarrès, M.; Arderiu, V.S.; Zaulet, A.; Viñas, C.; Fabrizi de Biani, F.; Teixidor, F. How to get the desired reduction voltage in a single framework! Metallacarborane as an optimal probe for sequential voltage tuning. Dalton Trans. 2015, 44, 11690–11695. [Google Scholar] [CrossRef]
- Lupu, M.; Zaulet, A.; Teixidor, F.; Ruiz, E.; Viñas, C. Negatively Charged Metallacarborane Redox Couples with Both Members Stable to Air. Chem. Eur. J. 2015, 21, 6888–6897. [Google Scholar] [CrossRef] [PubMed]
- Pepiol, A.; Teixidor, F.; Sillanpää, R.; Lupu, M.; Viñas, C. Stepwise Sequential Redox Potential Modulation Possible on a Single Platform. Angew. Chem. Int. Ed. 2011, 50, 12491–12495. [Google Scholar] [CrossRef] [PubMed]
- Cabrera-González, J.; Sánchez-Arderiu, V.; Viñas, C.; Parella, T.; Teixidor, F.; Núñez, R. Redox-Active Metallacarborane-Decorated Octasilsesquioxanes. Electrochemical and Thermal Properties. Inorg. Chem. 2016, 55, 11630–11634. [Google Scholar] [CrossRef] [PubMed]
- Cabrera-González, J.; Cabana, L.; Ballesteros, B.; Tobias, G.; Núñez, R. Highly Dispersible and Stable Anionic Boron Cluster–Graphene Oxide Nanohybrids. Chem. Eur. J. 2016, 22, 5096–5101. [Google Scholar] [CrossRef]
- Grüner, B.; Rais, J.; Selucký, P.; Lucaníková, M. Recent Progress in Extraction Agents Based on Cobalt Bis(Dicarbollides) for Partitioning of Radionuclides from High-Level Nuclear Waste. In Boron Science: New Technologies and Applications; Hosmane, N.S., Ed.; Taylor & Francis: Bosa Roca, FL, USA, 2012; pp. 463–490. [Google Scholar]
- Masalles, C.; Llop, J.; Viñas, C.; Teixidor, F. Extraordinary Overoxidation Resistance Increase in Self-Doped Polypyrroles by Using Non-conventional Low Charge-Density Anions. Adv. Mater. 2002, 14, 826–829. [Google Scholar] [CrossRef]
- Fernández-Alvarez, R.; Ďorďovič, V.; Uchman, M.; Matějíček, P. Amphiphiles without Head-and-Tail Design: Nanostructures Based on the Self-Assembly of Anionic Boron Cluster Compounds. Langmuir 2018, 34, 3541–3554. [Google Scholar] [CrossRef]
- Gassin, P.-M.; Girard, L.; Martin-Gassin, G.; Brusselle, D.; Jonchère, A.; Diat, O.; Viñas, C.; Teixidor, F.; Bauduin, P. Surface Activity and Molecular Organization of Metallacarboranes at the Air–Water Interface Revealed by Nonlinear Optics. Langmuir 2015, 31, 2297–2303. [Google Scholar] [CrossRef]
- Uchman, M.; Ďorďovič, V.; Tošner, Z.; Matějíček, P. Classical Amphiphilic Behavior of Nonclassical Amphiphiles: A Comparison of Metallacarborane Self-Assembly with SDS Micellization. Angew. Chem. Int. Ed. 2015, 54, 14113–14117. [Google Scholar] [CrossRef]
- Verdiá-Báguena, C.; Alcaraz, A.; Aguilella, V.M.; Cioran, A.M.; Tachikawa, S.; Nakamura, H.; Teixidor, F.; Viñas, C. Amphiphilic COSAN and I2-COSAN crossing synthetic lipid membranes: Planar bilayers and liposomes. Chem. Commun. 2014, 50, 6700–6703. [Google Scholar] [CrossRef]
- Brusselle, D.; Bauduin, P.; Girard, L.; Zaulet, A.; Viñas, C.; Teixidor, F.; Ly, I.; Diat, O. Lyotropic Lamellar Phase Formed from Monolayered θ-Shaped Carborane-Cage Amphiphiles. Angew. Chem. Int. Ed. 2013, 52, 12114–12118. [Google Scholar] [CrossRef]
- Rak, J.; Dejlová, B.; Lampová, H.; Kaplánek, R.; Matějíček, P.; Cígler, P.; Král, V. On the Solubility and Lipophilicity of Metallacarborane Pharmacophores. Mol. Pharm. 2013, 10, 1751–1759. [Google Scholar] [CrossRef] [PubMed]
- Bauduin, P.; Prevost, S.; Farràs, P.; Teixidor, F.; Diat, O.; Zemb, T. A Theta-Shaped Amphiphilic Cobaltabisdicarbollide Anion: Transition From Monolayer Vesicles to Micelles. Angew. Chem. Int. Ed. 2011, 50, 5298–5300. [Google Scholar] [CrossRef] [PubMed]
- Fuentes, I.; García-Mendiola, T.; Sato, S.; Pita, M.; Nakamura, H.; Lorenzo, E.; Teixidor, F.; Marques, F.; Viñas, C. Metallacarboranes on the Road to Anticancer Therapies: Cellular Uptake, DNA Interaction, and Biological Evaluation of Cobaltabisdicarbollide [COSAN]−. Chem. Eur. J. 2018, 24, 17239–17254. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Y.; Liu, W.; Chen, Y.; Jiang, H.; Yan, H.; Kosenko, I.; Chekulaeva, L.; Sivaev, I.; Bregadze, V.; Wang, X. A Highly Potent Antibacterial Agent Targeting Methicillin-Resistant Staphylococcus aureus Based on Cobalt Bis(1,2-Dicarbollide) Alkoxy Derivative. Organometallics 2017, 36, 3484–3490. [Google Scholar] [CrossRef]
- Goszczyński, T.M.; Fink, K.; Kowalski, K.; Leśnikowski, Z.J.; Boratyński, J. Interactions of Boron Clusters and their Derivatives with Serum Albumin. Sci. Rep. 2017, 7, 9800. [Google Scholar] [CrossRef]
- García-Mendiola, T.; Bayon-Pizarro, V.; Zaulet, A.; Fuentes, I.; Pariente, F.; Teixidor, F.; Viñas, C.; Lorenzo, E. Metallacarboranes as tunable redox potential electrochemical indicators for screening of gene mutation. Chem. Sci. 2016, 7, 5786–5797. [Google Scholar] [CrossRef]
- Leśnikowski, Z.J. Challenges and Opportunities for the Application of Boron Clusters in Drug Design. J. Med. Chem. 2016, 59, 7738–7758. [Google Scholar] [CrossRef]
- Tarrés, M.; Canetta, E.; Paul, E.; Forbes, J.; Azzouni, K.; Viñas, C.; Teixidor, F.; Harwood, A.J. Biological interaction of living cells with COSAN-based synthetic vesicles. Sci. Rep. 2015, 5, 7804. [Google Scholar] [CrossRef]
- Viñas, C. The uniqueness of boron as a novel challenging element for drugs in pharmacology, medicine and for smart biomaterials. Future Med. Chem. 2013, 5, 617–619. [Google Scholar] [CrossRef]
- Sivaev, I.B.; Bregadze, V.I.; Sjöberg, S. Chemistry of closo-Dodecaborate Anion [B 12 H 12] 2-: A Review. Collect. Czech. Chem. Commun. 2002, 67, 679–727. [Google Scholar] [CrossRef]
- Karki, K.; Gabel, D.; Roccatano, D. Structure and Dynamics of Dodecaborate Clusters in Water. Inorg. Chem. 2012, 51, 4894–4896. [Google Scholar] [CrossRef] [PubMed]
- Assaf, K.I.; Ural, M.S.; Pan, F.; Georgiev, T.; Simova, S.; Rissanen, K.; Gabel, D.; Nau, W.M. Water Structure Recovery in Chaotropic Anion Recognition: High-Affinity Binding of Dodecaborate Clusters to γ-Cyclodextrin. Angew. Chem. Int. Ed. 2015, 54, 6852–6856. [Google Scholar] [CrossRef] [PubMed]
- Viñas, C.; Tarrès, M.; González-Cardoso, P.; Farràs, P.; Bauduin, P.; Teixidor, F. Surfactant behaviour of metallacarboranes. A study based on the electrolysis of water. Dalton Trans. 2014, 43, 5062–5068. [Google Scholar] [CrossRef] [PubMed]
- Tachikawa, S.; Miyoshi, T.; Koganei, H.; El-Zaria, M.E.; Viñas, C.; Suzuki, M.; Ono, K.; Nakamura, H. Spermidinium closo-dodecaborate-encapsulating liposomes as efficient boron delivery vehicles for neutron capture therapy. Chem. Commun. 2014, 50, 12325–12328. [Google Scholar] [CrossRef] [PubMed]
- Ishii, S.; Sato, S.; Asami, H.; Hasegawa, T.; Kohno, J.-Y.; Nakamura, H. Design of S–S bond containing maleimide-conjugated closo-dodecaborate (SSMID): Identification of unique modification sites on albumin and investigation of intracellular uptake. Org. Biomol. Chem. 2019, 17, 5496–5499. [Google Scholar] [CrossRef]
- Ishii, S.; Nakamura, H. Synthesis and biological evaluation of closo-dodecaborate ibuprofen conjugate (DIC) as a new boron agent for neutron capture therapy. J. Organomet. Chem. 2018, 865, 178–182. [Google Scholar] [CrossRef]
- Awad, D.; Bartok, M.; Mostaghimi, F.; Schrader, I.; Sudumbrekar, N.; Schaffran, T.; Jenne, C.; Eriksson, J.; Winterhalter, M.; Fritz, J.; et al. Halogenated Dodecaborate Clusters as Agents to Trigger Release of Liposomal Contents. ChemPlusChem 2015, 80, 656–664. [Google Scholar] [CrossRef]
- Gabel, D.; Awad, D.; Schaffran, T.; Radovan, D.; Dărăban, D.; Damian, L.; Winterhalter, M.; Karlsson, G.; Edwards, K. The Anionic Boron Cluster (B12H11SH)2− as a Means To Trigger Release of Liposome Contents. ChemMedChem 2007, 2, 51–53. [Google Scholar] [CrossRef]
- Lee, M.W.; Farha, O.K.; Hawthorne, M.F.; Hansch, C.H. Alkoxy derivatives of dodecaborate: Discrete nanomolecular ions with tunable pseudometallic properties. Angew. Chem. 2007, 119, 3078–3082. [Google Scholar] [CrossRef]
- Joshi, M.; Ghanty, T.K. Hybrid Organic–Inorganic Functionalized Dodecaboranes and Their Potential Role in Lithium and Magnesium Ion Batteries. J. Phys. Chem. C 2018, 122, 27947–27954. [Google Scholar] [CrossRef]
- Vishnoi, P.; Kaleeswaran, D.; Murugavel, R. 1,3,5-Triphenylbenzene: A versatile photoluminescent chemo-sensor platform and supramolecular building block. RSC Adv. 2018, 8, 17535–17550. [Google Scholar] [CrossRef]
- Wu, Q.; Hook, A.; Wang, S. A Blue Luminescent Starburst Molecule and Its Orange Luminescent Trinuclear PdII Complex: 1,3,5-tris(7-azaindol-1-yl)benzene (tabH) and [Pd(tab)2Cl4]. Angew. Chem. Int. Ed. 2000, 39, 3933–3935. [Google Scholar] [CrossRef]
- Dash, B.P.; Satapathy, R.; Bode, B.P.; Reidl, C.T.; Sawicki, J.W.; Mason, A.J.; Maguire, J.A.; Hosmane, N.S. “Click” Chemistry-Mediated Phenylene-Cored Carborane Dendrimers. Organometallics 2012, 31, 2931–2935. [Google Scholar] [CrossRef]
- Dash, B.P.; Satapathy, R.; Gaillard, E.R.; Norton, K.M.; Maguire, J.A.; Chug, N.; Hosmane, N.S. Enhanced π-Conjugation and Emission via Icosahedral Carboranes: Synthetic and Spectroscopic Investigation. Inorg. Chem. 2011, 50, 5485–5493. [Google Scholar] [CrossRef] [PubMed]
- Juárez-Pérez, E.J.; Viñas, C.; Teixidor, F.; Santillan, R.; Farfán, N.; Abreu, A.; Yépez, R.; Núñez, R. Polyanionic Aryl Ether Metallodendrimers Based on Cobaltabisdicarbollide Derivatives. Photoluminescent Properties. Macromolecules 2010, 43, 150–159. [Google Scholar] [CrossRef]
- Zhou, H.; Ye, Q.; Xu, J. Polyhedral oligomeric silsesquioxane-based hybrid materials and their applications. Mater. Chem. Front. 2017, 1, 212–230. [Google Scholar] [CrossRef]
- Laine, R.M.; Roll, M.F. Polyhedral Phenylsilsesquioxanes. Macromolecules 2011, 44, 1073–1109. [Google Scholar] [CrossRef]
- Hartmann-Thompson, C. Applications of Polyhedral Oligomeric Silsesquioxanes; Springer: Midland, TX, USA, 2011. [Google Scholar]
- Wang, M.; Chi, H.; KS, J.; Wang, F. Progress in the Synthesis of Bifunctionalized Polyhedral Oligomeric Silsesquioxane. Polymers 2019, 11, 2098. [Google Scholar] [CrossRef]
- Maegawa, T.; Miyashita, O.; Irie, Y.; Imoto, H.; Naka, K. Synthesis and properties of polyimides containing hexaisobutyl-substituted T8 cages in their main chains. RSC Adv. 2016, 6, 31751–31757. [Google Scholar] [CrossRef]
- Ro, H.W.; Soles, C.L. Silsesquioxanes in nanoscale patterning applications. Mater. Today 2011, 14, 20–33. [Google Scholar] [CrossRef]
- Xia, S.; Yang, Y.; Zhu, W.; Lü, C. Quaternized polyhedral oligomeric silsesquioxanes stabilized Pd nanoparticles as efficient nanocatalysts for reduction reaction. Colloids Surf., A 2020, 585, 124110. [Google Scholar] [CrossRef]
- Ghanbari, H.; Cousins, B.G.; Seifalian, A.M. A Nanocage for Nanomedicine: Polyhedral Oligomeric Silsesquioxane (POSS). Macromol. Rapid Commun. 2011, 32, 1032–1046. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Kong, J.; Wang, F.; He, C. Polyhedral oligomeric silsesquioxanes (POSSs): An important building block for organic optoelectronic materials. J. Mater. Chem. C 2017, 5, 5283–5298. [Google Scholar] [CrossRef]
- Bahrami, M.; Furgal, J.C.; Hashemi, H.; Ehsani, M.; Jahani, Y.; Goodson, T.; Kieffer, J.; Laine, R.M. Synthesis and Characterization of Nanobuilding Blocks [o-RStyrPhSiO1.5]10,12 (R = Me, MeO, NBoc, and CN). Unexpected Photophysical Properties Arising from Apparent Asymmetric Cage Functionalization as Supported by Modeling Studies. J. Phys. Chem. C 2015, 119, 15846–15858. [Google Scholar] [CrossRef]
- Zhang, T.; Wang, J.; Zhou, M.; Ma, L.; Yin, G.; Chen, G.; Li, Q. Influence of polyhedral oligomeric silsesquioxanes (POSS) on blue light-emitting materials for OLED. Tetrahedron 2014, 70, 2478–2486. [Google Scholar] [CrossRef]
- Sulaiman, S.; Bhaskar, A.; Zhang, J.; Guda, R.; Goodson, T.; Laine, R.M. Molecules with Perfect Cubic Symmetry as Nanobuilding Blocks for 3-D Assemblies. Elaboration of Octavinylsilsesquioxane. Unusual Luminescence Shifts May Indicate Extended Conjugation Involving the Silsesquioxane Core. Chem. Mater. 2008, 20, 5563–5573. [Google Scholar] [CrossRef]
- Cheng, G.; Vautravers, N.R.; Morris, R.E.; Cole-Hamilton, D.J. Synthesis of functional cubes from octavinylsilsesquioxane (OVS). Org. Biomol. Chem. 2008, 6, 4662–4667. [Google Scholar] [CrossRef]
- Itami, Y.; Marciniec, B.; Kubicki, M. Functionalization of Octavinylsilsesquioxane by Ruthenium-Catalyzed Silylative Coupling versus Cross-Metathesis. Chem. Eur. J. 2004, 10, 1239–1248. [Google Scholar] [CrossRef]
- Anisimov, A.A.; Ol’shevskaya, V.A.; Novikov, R.A.; Korlyukov, A.A.; Buzin, M.I.; Shchegolikhina, O.I.; Kalinin, V.N.; Muzafarov, A.M. Polyfunctional carboranyl substituted octasilsesquioxane: Synthesis and characterization. J. Organomet. Chem. 2016, 822, 1–4. [Google Scholar] [CrossRef]
- Farràs, P.; Teixidor, F.; Kivekäs, R.; Sillanpää, R.; Viñas, C.; Grüner, B.; Cisarova, I. Metallacarboranes as Building Blocks for Polyanionic Polyarmed Aryl-Ether Materials. Inorg. Chem. 2008, 47, 9497–9508. [Google Scholar] [CrossRef]
- Sivaev, I.B.; Kulikova, N.Y.; Nizhnik, E.A.; Vichuzhanin, M.V.; Starikova, Z.A.; Semioshkin, A.A.; Bregadze, V.I. Practical synthesis of 1,4-dioxane derivative of the closo-dodecaborate anion and its ring opening with acetylenic alkoxides. J. Organomet. Chem. 2008, 693, 519–525. [Google Scholar] [CrossRef]
- Teixidor, F.; Pedrajas, J.; Rojo, I.; Viñas, C.; Kivekäs, R.; Sillanpää, R.; Sivaev, I.; Bregadze, V.; Sjöberg, S. Chameleonic Capacity of [3,3‘-Co(1,2-C2B9H11)2]- in Coordination. Generation of the Highly Uncommon S(thioether)−Na Bond. Organometallics 2003, 22, 3414–3423. [Google Scholar] [CrossRef]
- Chaari, M.; Gaztelumendi, N.; Cabrera-González, J.; Peixoto-Moledo, P.; Viñas, C.; Xochitiotzi-Flores, E.; Farfán, N.; Ben Salah, A.; Nogués, C.; Núñez, R. Fluorescent BODIPY-anionic boron cluster conjugates as potential agents for cell tracking. Bioconjugate Chem. 2018, 29, 1763–1773. [Google Scholar] [CrossRef] [PubMed]
- Cabrera-González, J.; Muñoz Flores, B.M.; Viñas, C.; Chávez-Reyes, A.; Dias, H.V.R.; Jiménez Pérez, V.M.; Núñez, R. Organotin dyes bearing anionic boron clusters as cell-staining fluorescent probes. Chem. Eur. J. 2018, 24, 5601–5612. [Google Scholar]
- Dash, B.P.; Satapathy, R.; Maguire, J.A.; Hosmane, N.S. Facile Synthetic Routes to Phenylene and Triazine Core Based Dendritic Cobaltabisdicarbollides. Organometallics 2010, 29, 5230–5235. [Google Scholar] [CrossRef]
- Semioshkin, A.A.; Las’kova, Y.N.; Zhidkova, O.B.; Bregadze, V.I. Synthesis of new building blocks based on the closo-dodecaborate anion. Russ. Chem. Bull. 2008, 57, 1996–1998. [Google Scholar] [CrossRef]
- Semioshkin, A.; Tsaryova, O.; Zhidkova, O.; Bregadze, V.; Wöhrle, D. Reactions of oxonium derivatives of (B12H12)2- with phenoles, and synthesis and photochemical properties of a phthalocyanine containing four (B12H12)2- groups. J. Porphyr. Phthalocyanines 2006, 10, 1293–1300. [Google Scholar] [CrossRef]
- Prikaznov, A.V.; Las’kova, Y.N.; Semioshkin, A.A.; Sivaev, I.B.; Kisin, A.V.; Bregadze, V.I. Synthesis of boron-containing tyrosine derivatives based on the closo-decaborate and closo-dodecaborate anions. Russ. Chem. Bull. 2011, 60, 2550–2554. [Google Scholar] [CrossRef]
- Grubbs, R.H.; O’Leary, D.J. Handbook of Metathesis: Applications in Organic Synthesis; John Wiley & Sons: Weinheim, Germany, 2015. [Google Scholar]
- Cioran, A.M.; Teixidor, F.; Viñas, C. The effect of a paramagnetic metal ion within a molecule: Comparison of the structurally identical paramagnetic [3,3-Fe(1,2-C2B9H11)2]- with the diamagnetic [3,3-Co(1,2-C2B9H11)2]- sandwich complexes. Dalton Trans. 2015, 44, 2809–2818. [Google Scholar] [CrossRef]
- Kukhta, N.A.; Volyniuk, D.; Peciulyte, L.; Ostrauskaite, J.; Juska, G.; Grazulevicius, J.V. Structure–property relationships of star-shaped blue-emitting charge-transporting 1,3,5-triphenylbenzene derivatives. Dyes Pigment. 2015, 117, 122–132. [Google Scholar] [CrossRef]
- Khotina, I.A.; Lepnev, L.S.; Burenkova, N.S.; Valetsky, P.M.; Vitukhnovsky, A.G. Phenylene dendrimers and novel hyperbranched polyphenylenes as light emissive materials for blue OLEDs. J. Lumin. 2004, 110, 232–238. [Google Scholar] [CrossRef]
- Núñez, R.; Juárez-Pérez, E.J.; Teixidor, F.; Santillan, R.; Farfán, N.; Abreu, A.; Yépez, R.; Viñas, C. Decorating Poly(alkyl aryl-ether) Dendrimers with Metallacarboranes. Inorg. Chem. 2010, 49, 9993–10000. [Google Scholar] [CrossRef] [PubMed]
- Rojo, I.; Teixidor, F.; Viñas, C.; Kivekäs, R.; Sillanpää, R. Relevance of the Electronegativity of Boron in η5-Coordinating Ligands: Regioselective Monoalkylation and Monoarylation in Cobaltabisdicarbollide [3,3′-Co(1,2-C2B9H11)2]- Clusters. Chem. Eur. J. 2003, 9, 4311–4323. [Google Scholar] [CrossRef] [PubMed]
- André, P.; Cheng, G.; Ruseckas, A.; van Mourik, T.; Früchtl, H.; Crayston, J.A.; Morris, R.E.; Cole-Hamilton, D.; Samuel, I.D.W. Hybrid Dendritic Molecules with Confined Chromophore Architecture to Tune Fluorescence Efficiency. J. Phys. Chem. B 2008, 112, 16382–16392. [Google Scholar] [CrossRef] [PubMed]
- Furgal, J.C.; Jung, J.H.; Goodson, T.; Laine, R.M. Analyzing Structure–Photophysical Property Relationships for Isolated T8, T10, and T12 Stilbenevinylsilsesquioxanes. J. Am. Chem. Soc. 2013, 135, 12259–12269. [Google Scholar] [CrossRef]
- Niemczyk, A.; Dziubek, K.; Sacher-Majewska, B.; Czaja, K.; Czech-Polak, J.; Oliwa, R.; Lenża, J.; Szołyga, M. Thermal Stability and Flame Retardancy of Polypropylene Composites Containing Siloxane-Silsesquioxane Resins. Polymers 2018, 10, 1019. [Google Scholar] [CrossRef] [PubMed]
- Cordes, D.B.; Lickiss, P.D.; Rataboul, F. Recent Developments in the Chemistry of Cubic Polyhedral Oligosilsesquioxanes. Chem. Rev. 2010, 110, 2081–2173. [Google Scholar] [CrossRef]
- Yang, D.; Zhang, W.; Yao, R.; Jiang, B. Thermal stability enhancement mechanism of poly(dimethylsiloxane) composite by incorporating octavinyl polyhedral oligomeric silsesquioxanes. Polym. Degrad. Stab. 2013, 98, 109–114. [Google Scholar] [CrossRef]
- Lakowicz, J.R. Principles of Fluorescence Spectroscopy; Springer US: Baltimore, MD, USA, 2007. [Google Scholar]
- Brouwer, A.M. Standards for photoluminescence quantum yield measurements in solution (IUPAC Technical Report). Pure Appl. Chem. 2011, 83, 2213–2228. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are available from the authors. |
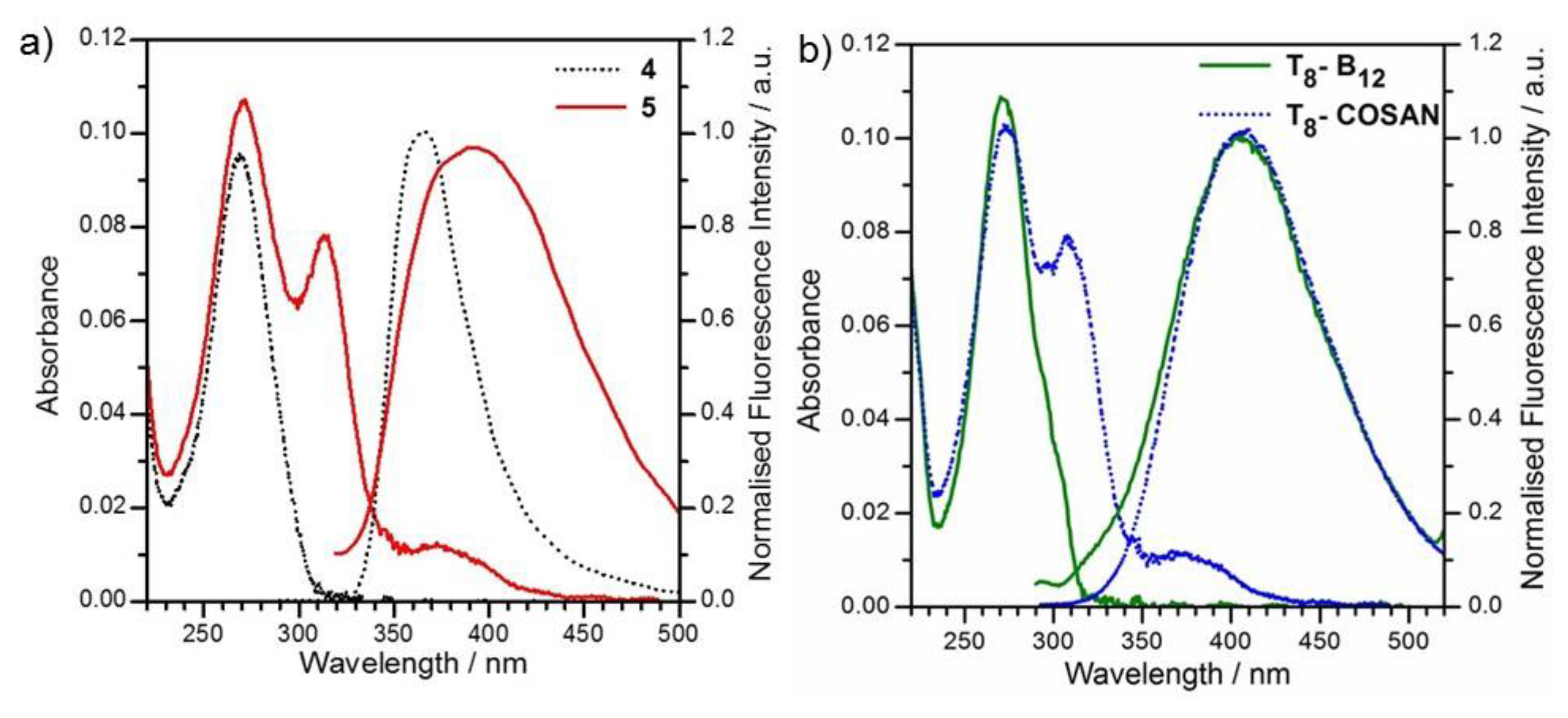
| Compounds | λabs/nm | ε/M−1 cm−1 | λem (λexc)/nm | ϕFa | Stokes Shift (nm) |
|---|---|---|---|---|---|
| 4 | 269 | 68000 | 369 (269) | 0.128 | 100 |
| 5 | 272 | 83000 | 394 (272) | 0.008 | 122 |
| T8-B12 | 270 | 186000 | 405 (270) | 0.014 | 136 |
| T8-COSAN | 272 | 233000 | 406 (272) | 0.018 | 138 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cabrera-González, J.; Chaari, M.; Teixidor, F.; Viñas, C.; Núñez, R. Blue Emitting Star-Shaped and Octasilsesquioxane-Based Polyanions Bearing Boron Clusters. Photophysical and Thermal Properties. Molecules 2020, 25, 1210. https://doi.org/10.3390/molecules25051210
Cabrera-González J, Chaari M, Teixidor F, Viñas C, Núñez R. Blue Emitting Star-Shaped and Octasilsesquioxane-Based Polyanions Bearing Boron Clusters. Photophysical and Thermal Properties. Molecules. 2020; 25(5):1210. https://doi.org/10.3390/molecules25051210
Chicago/Turabian StyleCabrera-González, Justo, Mahdi Chaari, Francesc Teixidor, Clara Viñas, and Rosario Núñez. 2020. "Blue Emitting Star-Shaped and Octasilsesquioxane-Based Polyanions Bearing Boron Clusters. Photophysical and Thermal Properties" Molecules 25, no. 5: 1210. https://doi.org/10.3390/molecules25051210
APA StyleCabrera-González, J., Chaari, M., Teixidor, F., Viñas, C., & Núñez, R. (2020). Blue Emitting Star-Shaped and Octasilsesquioxane-Based Polyanions Bearing Boron Clusters. Photophysical and Thermal Properties. Molecules, 25(5), 1210. https://doi.org/10.3390/molecules25051210







