Extraction and Determination of Vitamin K1 in Foods by Ultrasound-Assisted Extraction, SPE, and LC-MS/MS
Abstract
1. Introduction
2. Results and Discussion
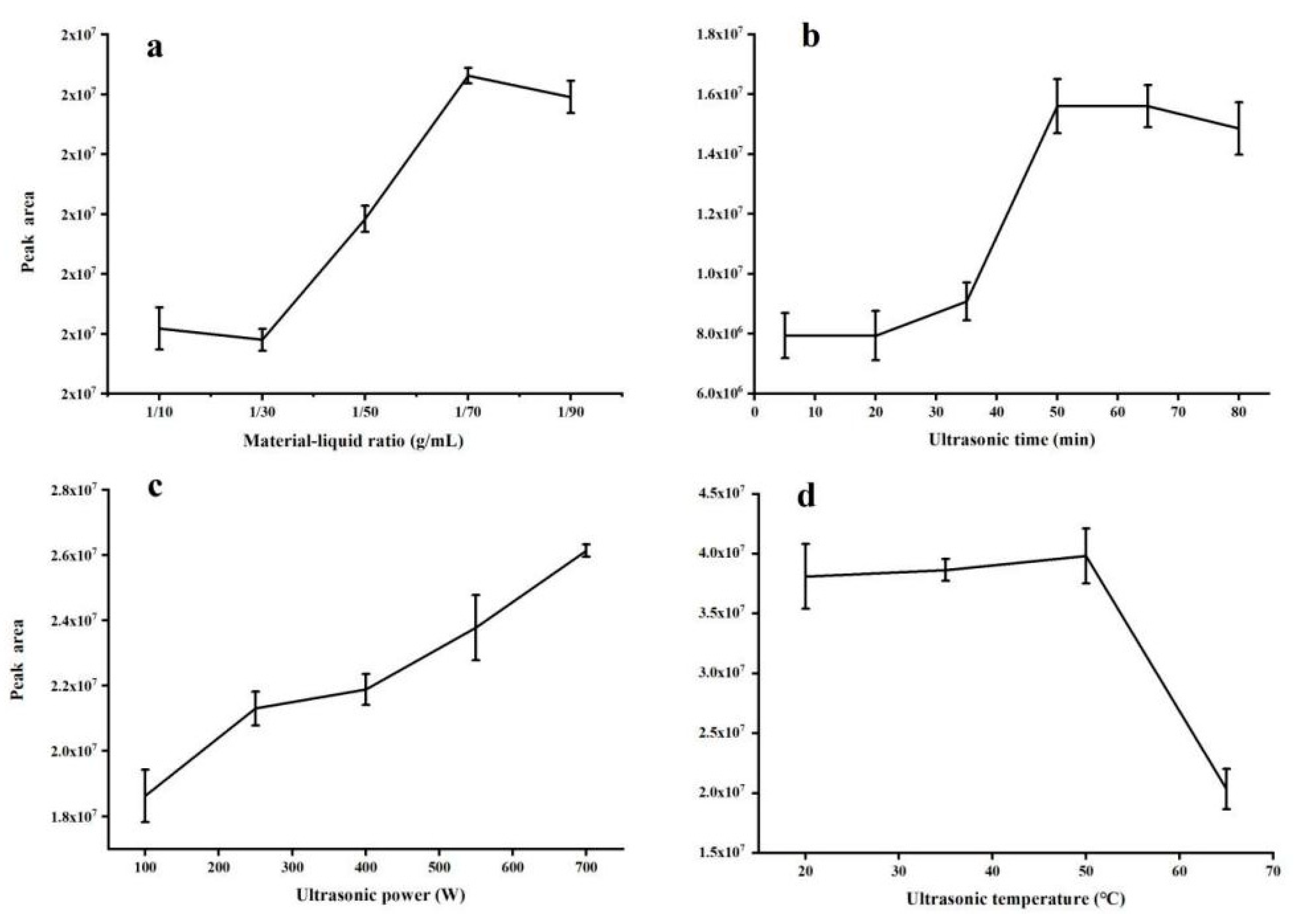
2.1. Optimization of Ultrasound-Assisted Extraction Conditions
2.1.1. Effect of Material-Liquid Ratio
2.1.2. Effect of Ultrasonic Time
2.1.3. Effect of Ultrasonic Power
2.1.4. Effect of Ultrasonic Temperature
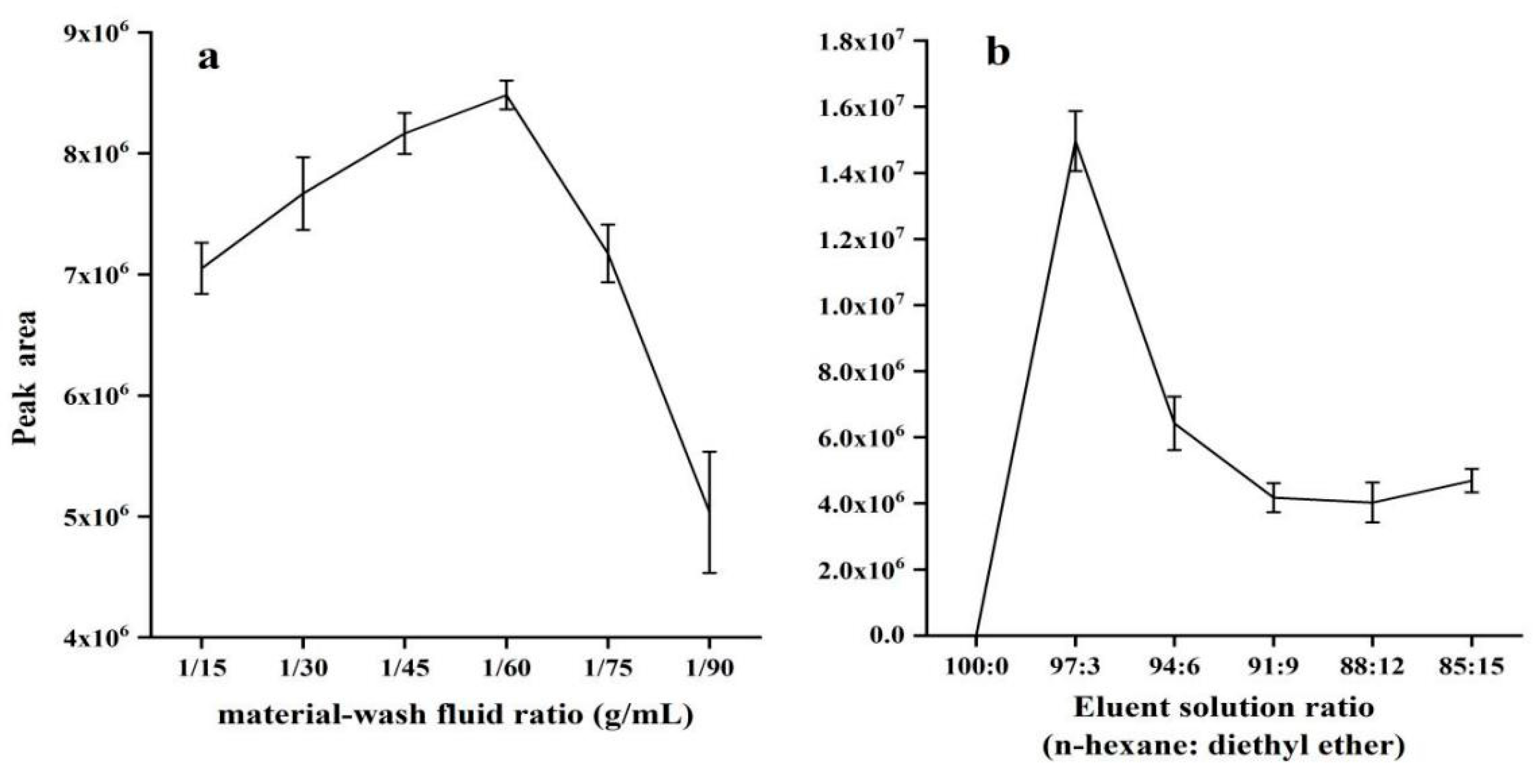
2.2. Optimization of SPE Conditions
2.2.1. Effect of Material-Wash Fluid Ratio
2.2.2. Effect of Elution Solution Ratio
2.3. Optimization of the LC-MS/MS Conditions
2.4. Method Validation
2.4.1. Linearity, Limit of Detection, and Limit of Quantification
2.4.2. Precision and Accuracy
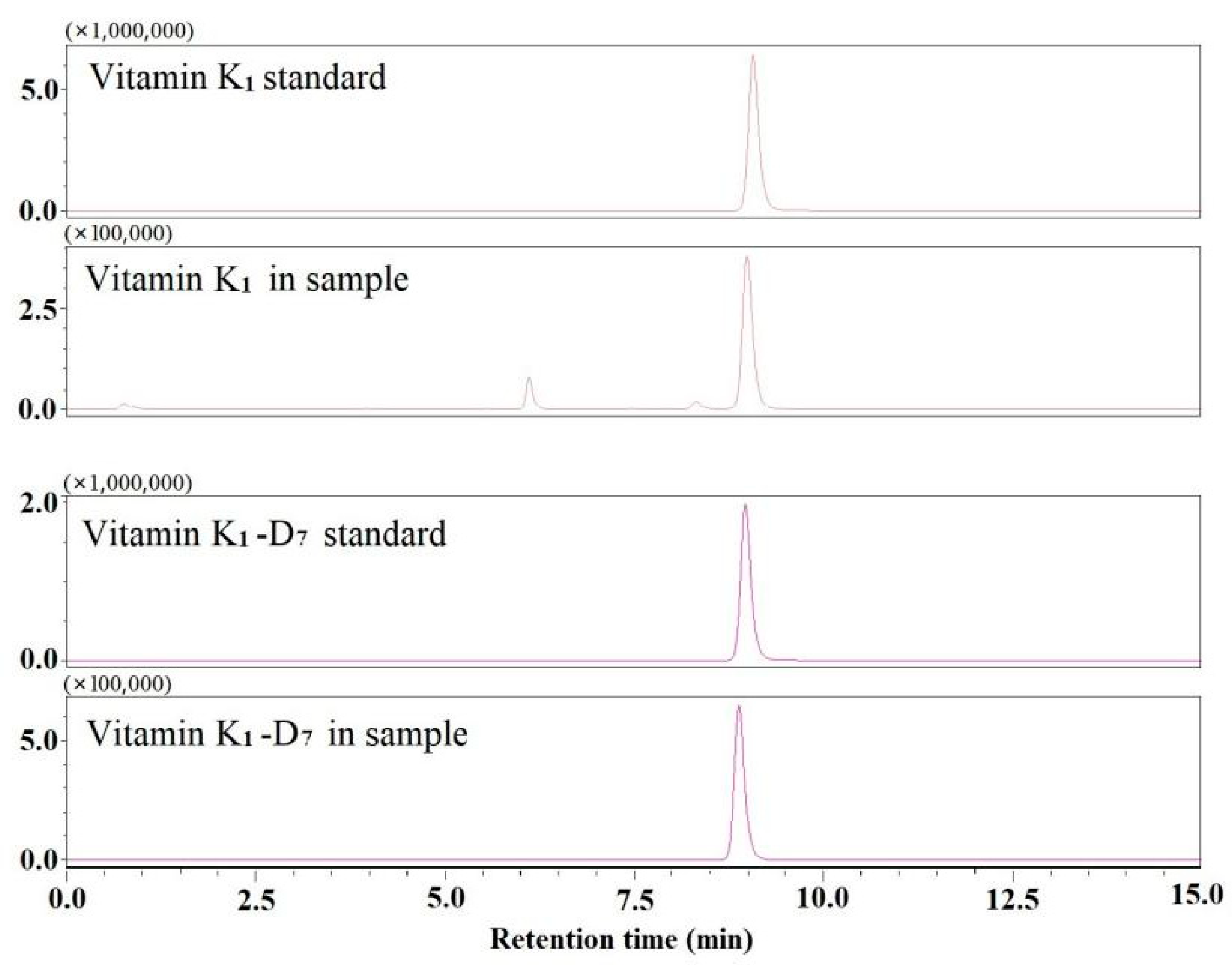
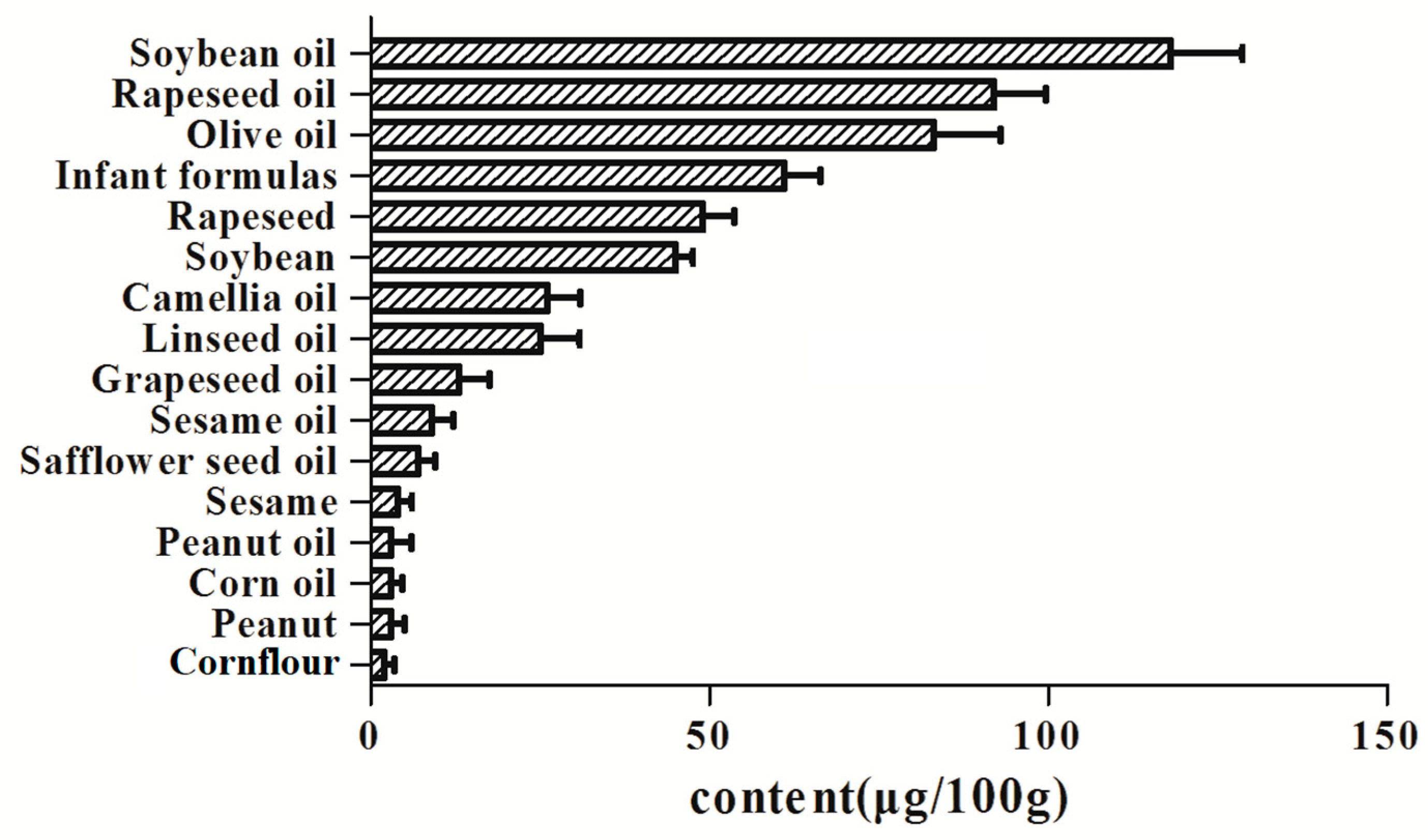
2.5. Real Sample Analysis
3. Materials and Methods
3.1. Materials and Reagents
3.2. Instruments and Equipment
3.3. Preparation of Standard Solutions
3.4. Sample Preparation
3.5. LC-MS/MS Method
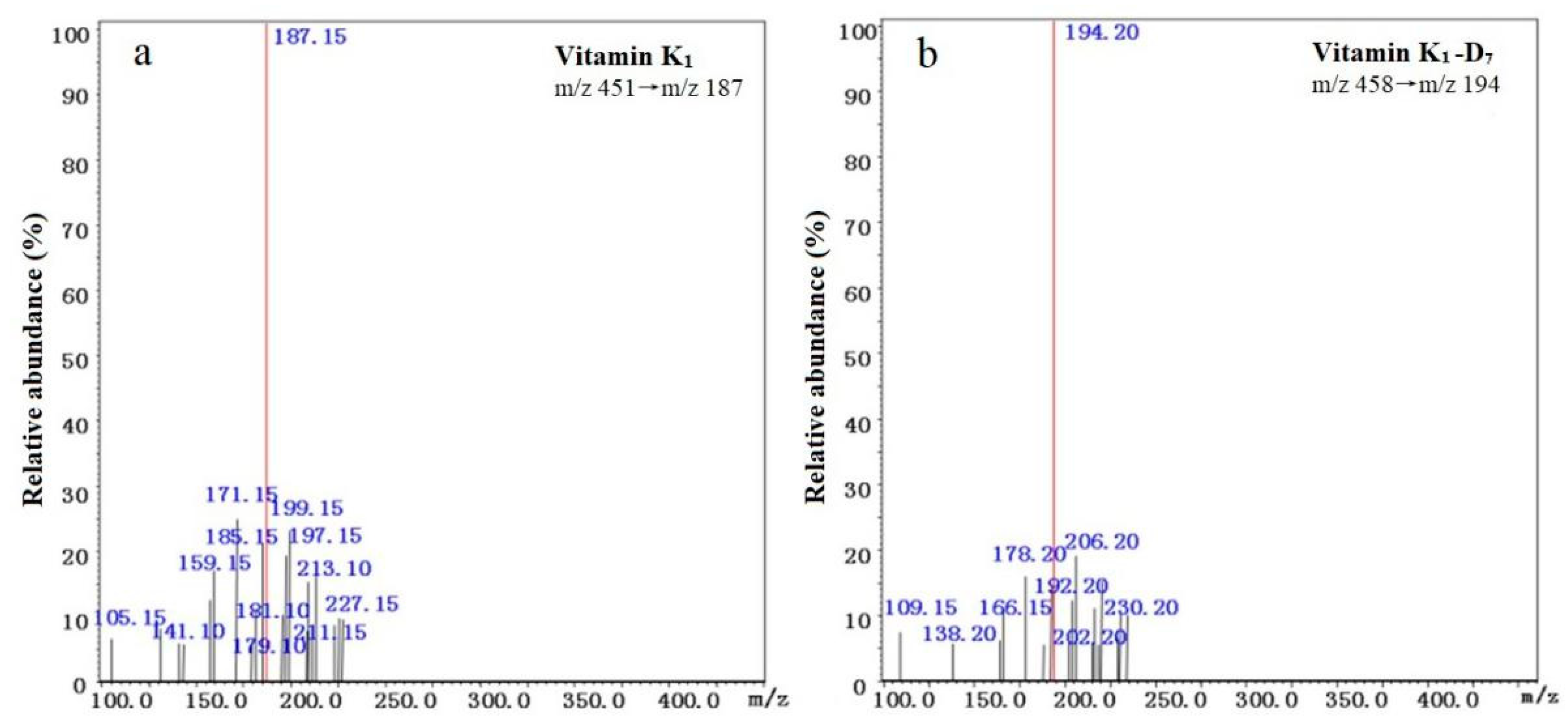
3.6. Peak Identification
3.7. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Schurgers, L.J.; Vermeer, C. Determination of phylloquinone and menaquinones in food—Effect of food matrix on circulating vitamin K concentrations. Haemostasis 2000, 30, 298–307. [Google Scholar] [PubMed]
- Basset, G.J.; Latimer, S.; Fatihi, A.; Soubeyrand, E.; Block, A. Phylloquinone (Vitamin K1): Occurrence, Biosynthesis and Functions. Mini Rev. Med. Chem. 2017, 17, 1028–1038. [Google Scholar] [CrossRef] [PubMed]
- Karl, J.P.; Fu, X.Y.; Dolnikowski, G.G.; Saltzman, E.; Booth, S.L. Quantification of phylloquinone and menaquinones in feces, serum, and food by high-performance liquid chromatography-mass spectrometry. J. Chromatogr. B 2014, 963, 128–133. [Google Scholar] [CrossRef]
- Nannapaneni, N.K.; Jalalpure, S.S.; Muppavarapu, R.; Sirigiri, S.K. A sensitive and rapid UFLC-APCI-MS/MS bioanalytical method for quantification of endogenous and exogenous Vitamin Kl isomers in human plasma: Development, validation and first application to a pharmacokinetic study. Talanta 2017, 164, 233–243. [Google Scholar] [CrossRef]
- Kamao, M.; Suhara, Y.; Tsugawa, N.; Okano, T. Determination of plasma Vitamin K by high-performance liquid chromatography with fluorescence detection using Vitamin K analogs as internal standards. J. Chromatogr. B 2005, 816, 41–48. [Google Scholar] [CrossRef] [PubMed]
- Mizuta, T.; Ozaki, I. Hepatocellular carcinoma and vitamin K. Vitam. Horm. 2008, 78, 435–442. [Google Scholar] [CrossRef]
- Brandenburg, V.M.; Schurgers, L.J.; Kaesler, N.; Pusche, K.; van Gorp, R.H.; Leftheriotis, G.; Reinartz, S.; Koos, R.; Kruger, T. Prevention of vasculopathy by vitamin K supplementation: Can we turn fiction into fact? Atherosclerosis 2015, 240, 10–16. [Google Scholar] [CrossRef]
- Villa, J.K.D.; Diaz, M.A.N.; Pizziolo, V.R.; Martino, H.S.D. Effect of vitamin K in bone metabolism and vascular calcification: A review of mechanisms of action and evidences. Crit. Rev. Food Sci. 2017, 57, 3959–3970. [Google Scholar] [CrossRef]
- Riphagen, I.J.; van der Molen, J.C.; van Faassen, M.; Navis, G.; de Borst, M.H.; Muskiet, F.A.J.; de Jong, W.H.A.; Bakker, S.J.L.; Kema, I.P. Measurement of Plasma Vitamin K1 (Phylloquinone) and K2 (Menaquinones-4 and -7) using HPLC-Tandem Mass Spectrometry. Clin. Chem. Lab. Med. 2016, 54, 1201–1210. [Google Scholar] [CrossRef]
- Chen, W.X.; Guo, Y.F. Therapeutic effect of vitamin K1 combined with meropenem on the treatment of pertussis syndrome. Exp. Med. 2019, 18, 642–646. [Google Scholar] [CrossRef]
- Booth, S.L.; Suttie, J.W. Dietary intake and adequacy of vitamin K. J. Nutr. 1998, 128, 785–788. [Google Scholar] [CrossRef] [PubMed]
- Claussen, F.A.; Taylor, M.L.; Breeze, M.L.; Liu, K. Measurement of Vitamin K-1 in Commercial Canola Cultivars from Growing Locations in North and South America Using High-Performance Liquid Chromatography Tandem Mass Spectrometry. J. Agric. Food Chem. 2015, 63, 1076–1081. [Google Scholar] [CrossRef] [PubMed]
- Kamao, M.; Suhara, Y.; Tsugawa, N.; Uwano, M.; Yamaguchi, N.; Uenish, K.; Ishida, H.; Sasaki, S.; Okano, T. Vitamin K content of foods and dietary vitamin K intake in Japanese young women. J. Nutr. Sci. Vitaminol. 2007, 53, 464–470. [Google Scholar] [CrossRef]
- Cook, K.K.; Mitchell, G.V.; Grundel, E.; Rader, J.I. HPLC analysis for trans-vitamin K1 and dihydro-vitamin K1 in margarines and margarine-like products using the C30 stationary phase. Food Chem. 1999, 67, 79–88. [Google Scholar] [CrossRef]
- Gentili, A.; Miccheli, A.; Tomai, P.; Baldassarre, M.E.; Curini, R.; Perez-Fernandez, V. Liquid chromatography-tandem mass spectrometry method for the determination of vitamin K homologues in human milk after overnight cold saponification. J. Food Compos. Anal. 2016, 47, 21–30. [Google Scholar] [CrossRef]
- Shao, Y.; Zhai, L.F.; Wang, H.X.; Chai, P.H. Improvement research on the determination of vitamin K1 in milk using high performance liquid chromatography. China Dairy Ind. 2016, 44, 57–58, 64. [Google Scholar] [CrossRef]
- Japelt, R.B.; Jakobsen, J. Analysis of vitamin K-1 in fruits and vegetables using accelerated solvent extraction and liquid chromatography tandem mass spectrometry with atmospheric pressure chemical ionization. Food Chem. 2016, 192, 402–408. [Google Scholar] [CrossRef]
- Andersen, I.B.; Brasen, C.L.; Madsen, J.S.; Schmedes, A. Quantitation of vitamin K1 in serum using online SPE-LC-MS/MS and the challenges of working with vitamin K. J Chromatogr. B 2019, 1117, 41–48. [Google Scholar] [CrossRef]
- Hennion, M.C. Solid-phase extraction: Method development, sorbents, and coupling with liquid chromatography. J. Chromatogr. A 1999, 856, 3–54. [Google Scholar] [CrossRef]
- Zhang, L.X.; Wang, S.J.; Yang, R.N.; Mao, J.; Jiang, J.; Wang, X.P.; Zhang, W.; Zhang, Q.; Li, P.W. Simultaneous determination of tocopherols, carotenoids and phytosterols in edible vegetable oil by ultrasound-assisted saponification, LLE and LC-MS/MS. Food Chem. 2019, 289, 313–319. [Google Scholar] [CrossRef]
- Wang, D.D.; Zhang, L.X.; Xu, Y.Q.; Qi, X.; Wang, X.F.; Wang, X.P.; Zhang, Q.; Li, P.W. Optimization of an Ultrasound-Assisted Extraction for Simultaneous Determination of Antioxidants in Sesame with Response Surface Methodology. Antioxidants 2019, 8, 321. [Google Scholar] [CrossRef]
- Koivu-Tikkanen, T.J.; Ollilainen, V.; Piironen, V.I. Determination of Phylloquinone and Menaquinones in Animal Products with Fluorescence Detection after Postcolumn Reduction with Metallic Zinc. J. Agric. Food Chem. 2000, 48, 6325–6331. [Google Scholar] [CrossRef] [PubMed]
- Piironen, V.; Koivu, T.; Tammisalo, O.; Mattila, P. Determination of phylloquinone in oils, margarines and butter by high-performance liquid chromatography with electrochemical detection. Food Chem. 1997, 59, 473–480. [Google Scholar] [CrossRef]
- Tyśkiewicz, K.; Gieysztor, R.; Maziarczyk, I.; Hodurek, P.; Rój, E.; Skalicka-Woźniak, K. Supercritical Fluid Chromatography with Photodiode Array Detection in the Determination of Fat-Soluble Vitamins in Hemp Seed Oil and Waste Fish Oil. Molecules 2018, 23, 1131. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, S.; Kishikawa, N.; Nakashima, K.; Kuroda, N. Determination of vitamin K homologues by high-performance liquid chromatography with on-line photoreactor and peroxyoxalate chemiluminescence detection. Anal. Chim. Acta 2007, 591, 148–154. [Google Scholar] [CrossRef] [PubMed]
- Górska, R.M. Chapter 5-Methods for assessment of Vitamin K, in Harrington, D. Lab. Assess. Vitam. Status 2019, 107–147. [Google Scholar] [CrossRef]
- Lacina, O.; Urbanova, J.; Poustka, J.; Hajslova, J. Identification/quantification of multiple pesticide residues in food plants by ultra-high-performance liquid chromatography-time-of-flight mass spectrometry. J. Chromatogr. A 2010, 1217, 648–659. [Google Scholar] [CrossRef]
- Kruve, A.; Kunnapas, A.; Herodes, K.; Leito, I. Matrix effects in pesticide multi-residue analysis by liquid chromatography-mass spectrometry. J. Chromatogr. A 2008, 1187, 58–66. [Google Scholar] [CrossRef]
- Hiemstra, M.; de Kok, A. Comprehensive multi-residue method for the target analysis of pesticides in crops using liquid chromatography-tandem mass spectrometry. J. Chromatogr. A 2007, 1154, 3–25. [Google Scholar] [CrossRef]
- Huang, B.F.; Wang, Z.; Yao, J.H.; Ke, X.; Xu, J.J.; Pan, X.D.; Xu, X.M.; Lu, M.L.; Ren, Y.P. Quantitative analysis of vitamin K-1 in fruits and vegetables by isotope dilution LC-MS/MS. Anal. Methods 2016, 8, 5707–5711. [Google Scholar] [CrossRef]
- Gao, Z.H.; Ackman, R.G. Determination of Vitamin-K-1 in Canola-Oils by High-Performance Liquid-Chromatography with Menaquinone-4 as an Internal Standard. Food Res. Int. 1995, 28, 61–69. [Google Scholar] [CrossRef]
- Tarvainen, M.; Fabritius, M.; Yang, B. Determination of vitamin K composition of fermented food. Food Chem. 2019, 275, 515–522. [Google Scholar] [CrossRef] [PubMed]
- pokkanta, p.; sookwong, p.; tanamg, m.; setchaiyan, s.; boontakham, p.; mahatheeranont, s. simultaneous determination of tocols, γ-oryzanols, phytosterols, squalene, cholecalciferol and phylloquinone in rice bran and vegetable oil samples. Food Chem. 2019, 271, 630–638. [Google Scholar] [CrossRef] [PubMed]
- Rathi, D.N.; Liew, C.Y.; Fairulnizal, M.N.M.; Isameyah, D.; Barknowitz, G. Fat-Soluble Vitamin and Carotenoid Analysis in Cooking Oils by Ultra-Performance Convergence Chromatography. Food Anal. Methods 2017, 10, 1087–1096. [Google Scholar] [CrossRef]
- Rebufa, C.; Artaud, J. Analytical Determination of Phylloquinone (Vitamin K1) in Olive Oils. Comparison with Other Vegetable Oils. Eur. J. Lipid Sci. Technol. 2018, 120, 1700527. [Google Scholar] [CrossRef]
- Woollard, D.C.; Indyk, H.E.; Fong, B.Y.; Cook, K.K. Determination of vitamin K-1 isomers in foods by liquid chromatography with C-30 bonded-phase column. J. AOAC Int. 2002, 85, 682–691. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are not available from the authors. |
| Compound | Calibration Curve | Linear Range (µg/kg) | R2 | LOD (µg/kg) | LOQ (µg/kg) |
|---|---|---|---|---|---|
| Vitamin K1 | Y = 1.4831X − 0.1171 | 10–500 | 0.9988 | 0.05 | 0.16 |
| Compound | Spiked Concentration (μg/kg) | Intra-Day Precisions (RSD%, n = 5) | Inter-Day Precisions (RSD%, n = 5) | Recovery (%, n = 3) |
|---|---|---|---|---|
| Vitamin K1 | 10 | 3.7 | 6.2 | 93.2/119.1/107.1 |
| 50 | 1.6 | 4.3 | 91.0/89.3/89.1 | |
| 100 | 5.4 | 7.2 | 80.9/82.8/81.5 |
| No | Sample | Sample Pretreatment | Determination Technique | Column | LOQ | Recovery (%) | Analysis Time (min) | Literatures |
|---|---|---|---|---|---|---|---|---|
| 1 | Corn oil | Enzymatic digestion and extraction | HPLC-Fluorescence | YMC C30 Wilmington (250 × 4.6 mm, 3 μm) | - | 82~100 | 30 | [14] |
| 2 | Spinach, peas, apples, et. al | ASE-SPE | HPLC-MS/MS (APCI) | Phenomenex Kinetex PFP (100 × 2.1 mm, 2.6 μm) | 0.5 μg/100 g | 90~120 | 20 | [17] |
| 3 | Fruits and vegetables | LLE-SPE | LC-MS/MS (ESI) | ZORBAX Eclipse plus C18 (50× 2.1 mm, 1.8 μm) | 0.004 mg/kg | 84~115.6 | 8 | [30] |
| 4 | Rapeseed or Canola oil | Enzymatic hydrolysis and SPE | HPLC-Fluorescence | PartiSphere Whatman C18 (150 × 4.6 mm, 6 μm) | - | 80.8~95.4 | 30 | [31] |
| 5 | Fermented food | SPE | HPLC-MS/MS (APCI) | Phenomenex Kinetex C18 (100 × 2.1 mm, 1.7 µm) | 0.385 ng/g | 99.8 | 10 | [32] |
| 6 | Rice bran and vegetable oil | LLE | HPLC-DAD-FLD | Phenomenex Kinetex PFP (250 × 4.6 mm, 5 μm) | 0.915 μg/mL | 96.0~102.9 | 30 | [33] |
| 7 | Cooking oils | LLE | UPC2 | HSS C18 SB (100 × 3.0 mm, 1.8 μm) | 0.98 μg/mL | 94.24 ± 0.56 | 8 | [34] |
| 8 | Fat-containing foods | UAE-SPE | HPLC-MS/MS (ESI) | ZORBAX SB-C18 (50 × 2.1 mm, 1.8 μm) | 0.16 μg/kg | 80.9~119.1 | 15 | This study |
| Compounds | Scan Mode | RT (min) | Parent Ions (m/z) | Product Ions (m/z) | Collision Energy (eV) |
|---|---|---|---|---|---|
| Vitamin K1 | [M + H]+ | 9.058 | 451 | 227/187 * | 23/25 |
| Vitamin K1-D7 | [M + H]+ | 8.980 | 458 | 194* | 18 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xu, Y.; Zhang, L.; Yang, R.; Yu, X.; Yu, L.; Ma, F.; Li, H.; Wang, X.; Li, P. Extraction and Determination of Vitamin K1 in Foods by Ultrasound-Assisted Extraction, SPE, and LC-MS/MS. Molecules 2020, 25, 839. https://doi.org/10.3390/molecules25040839
Xu Y, Zhang L, Yang R, Yu X, Yu L, Ma F, Li H, Wang X, Li P. Extraction and Determination of Vitamin K1 in Foods by Ultrasound-Assisted Extraction, SPE, and LC-MS/MS. Molecules. 2020; 25(4):839. https://doi.org/10.3390/molecules25040839
Chicago/Turabian StyleXu, Yueqing, Liangxiao Zhang, Ruinan Yang, Xu Yu, Li Yu, Fei Ma, Hui Li, Xiupin Wang, and Peiwu Li. 2020. "Extraction and Determination of Vitamin K1 in Foods by Ultrasound-Assisted Extraction, SPE, and LC-MS/MS" Molecules 25, no. 4: 839. https://doi.org/10.3390/molecules25040839
APA StyleXu, Y., Zhang, L., Yang, R., Yu, X., Yu, L., Ma, F., Li, H., Wang, X., & Li, P. (2020). Extraction and Determination of Vitamin K1 in Foods by Ultrasound-Assisted Extraction, SPE, and LC-MS/MS. Molecules, 25(4), 839. https://doi.org/10.3390/molecules25040839









