Rosmarinic Acid as a Candidate in a Phenotypic Profiling Cardio-/Cytotoxicity Cell Model Induced by Doxorubicin
Abstract
1. Introduction
2. Results
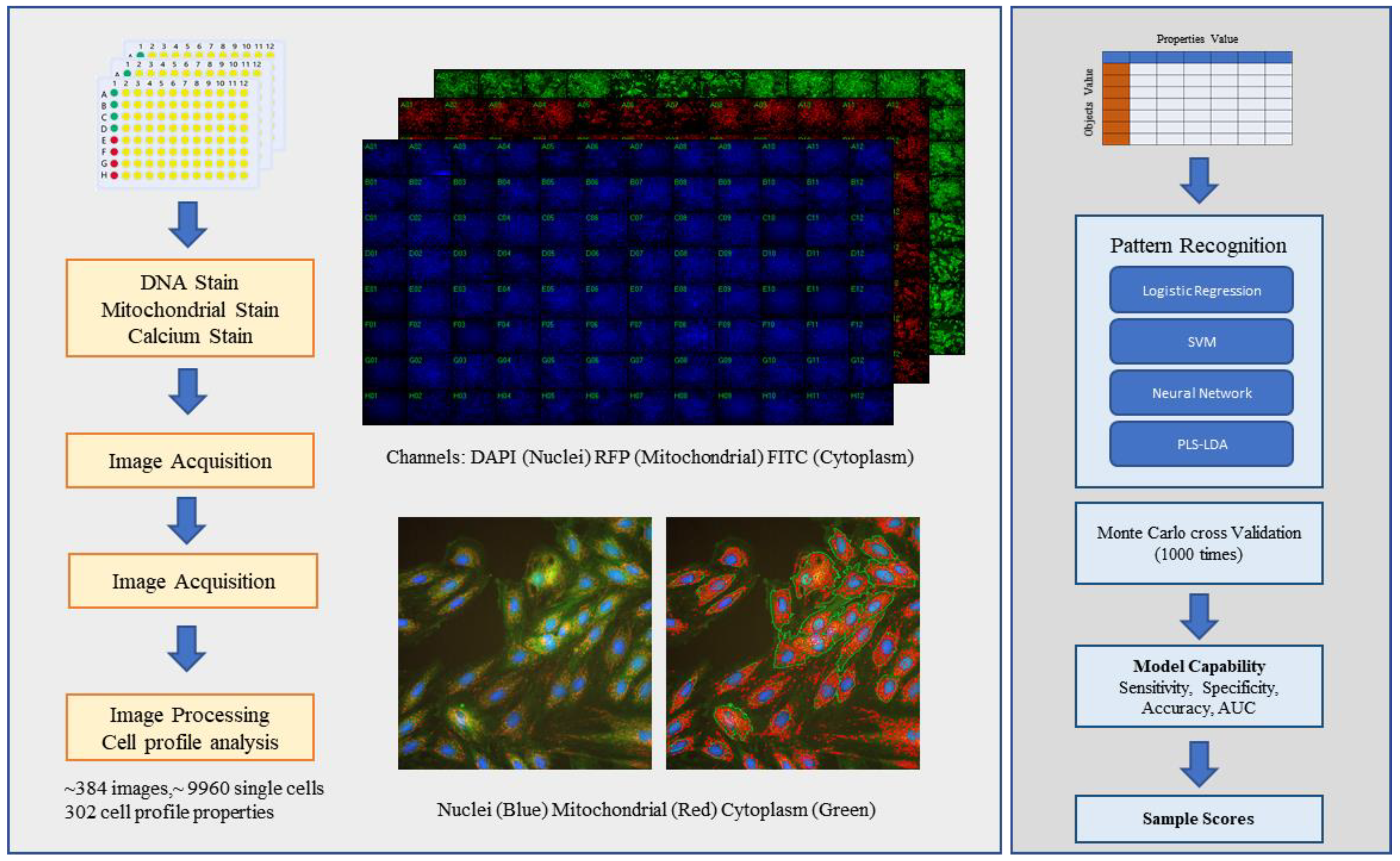
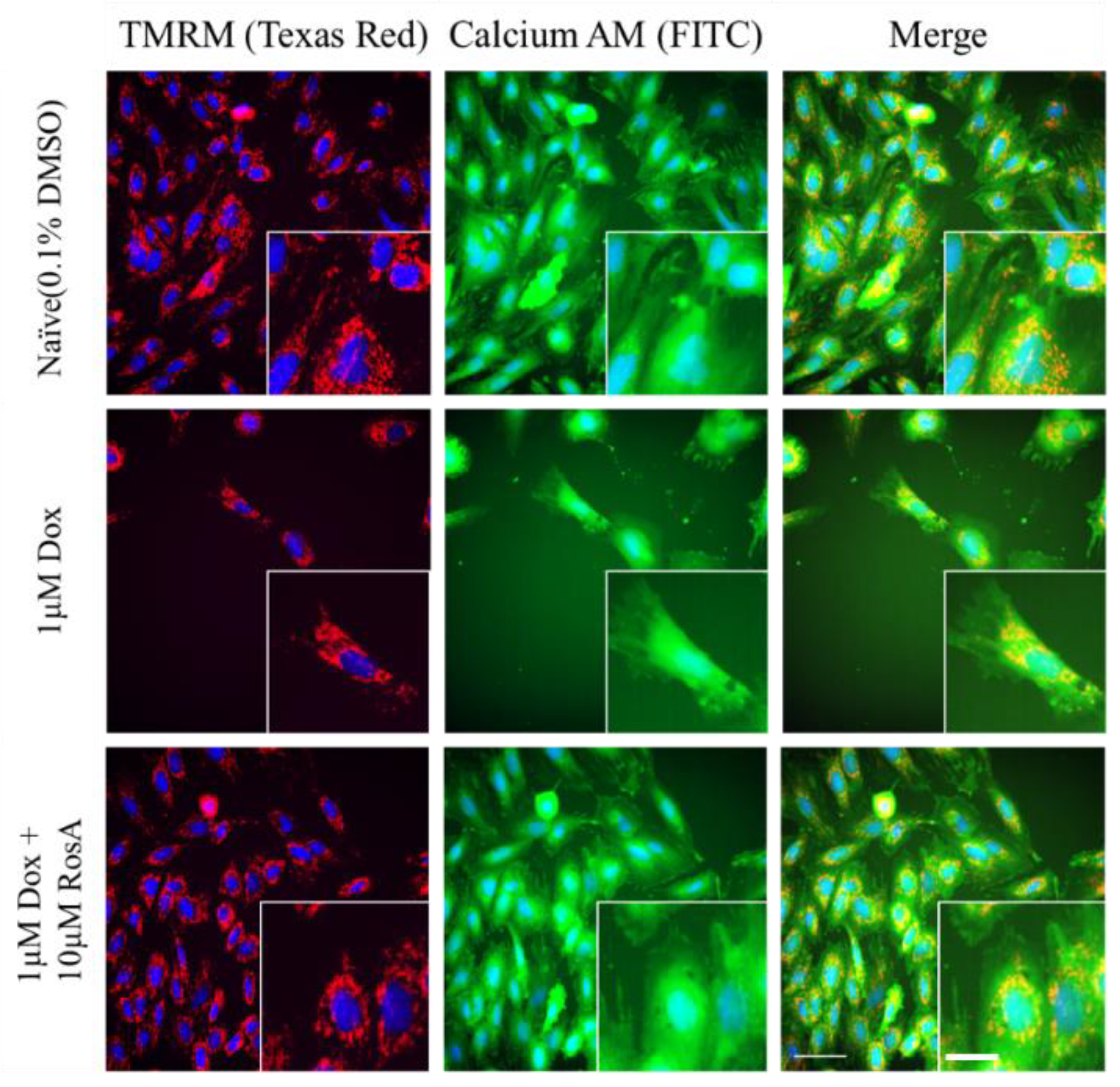
2.1. Using a High-Content Assay to Establish a Cardiotoxicity H9C2 Cell Model
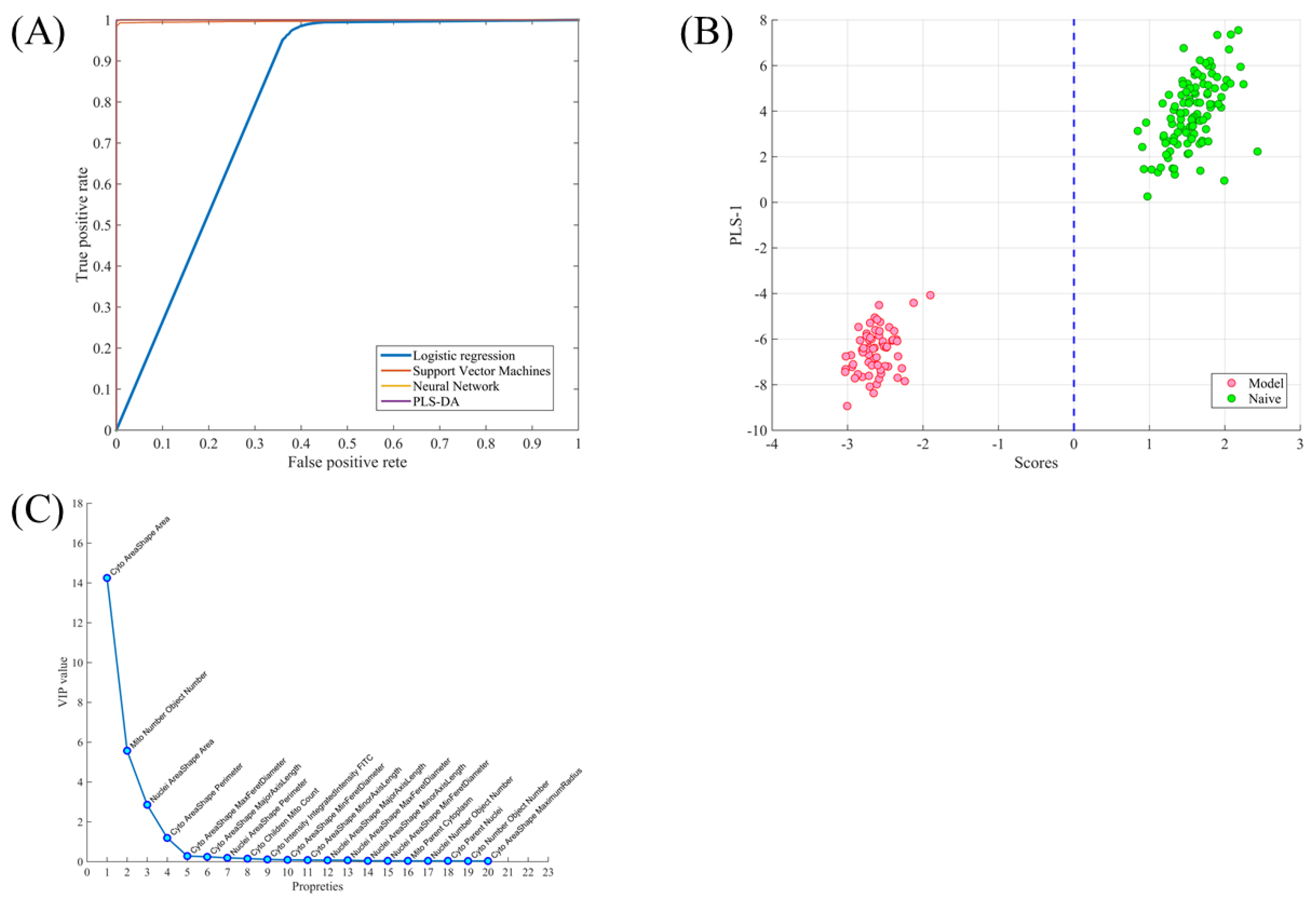
2.2. Using Cell Morphological Parameters for Pattern Recognition
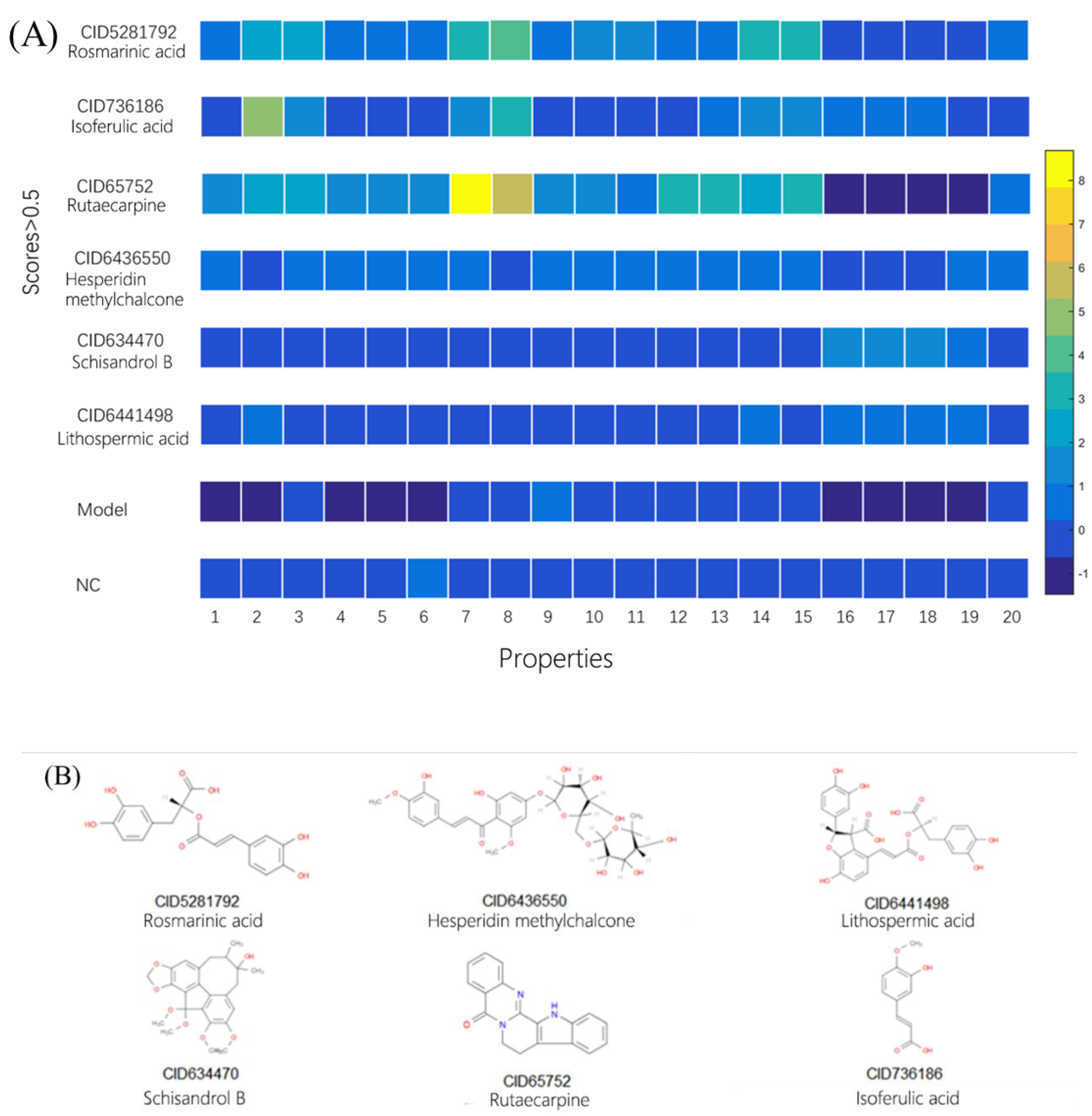
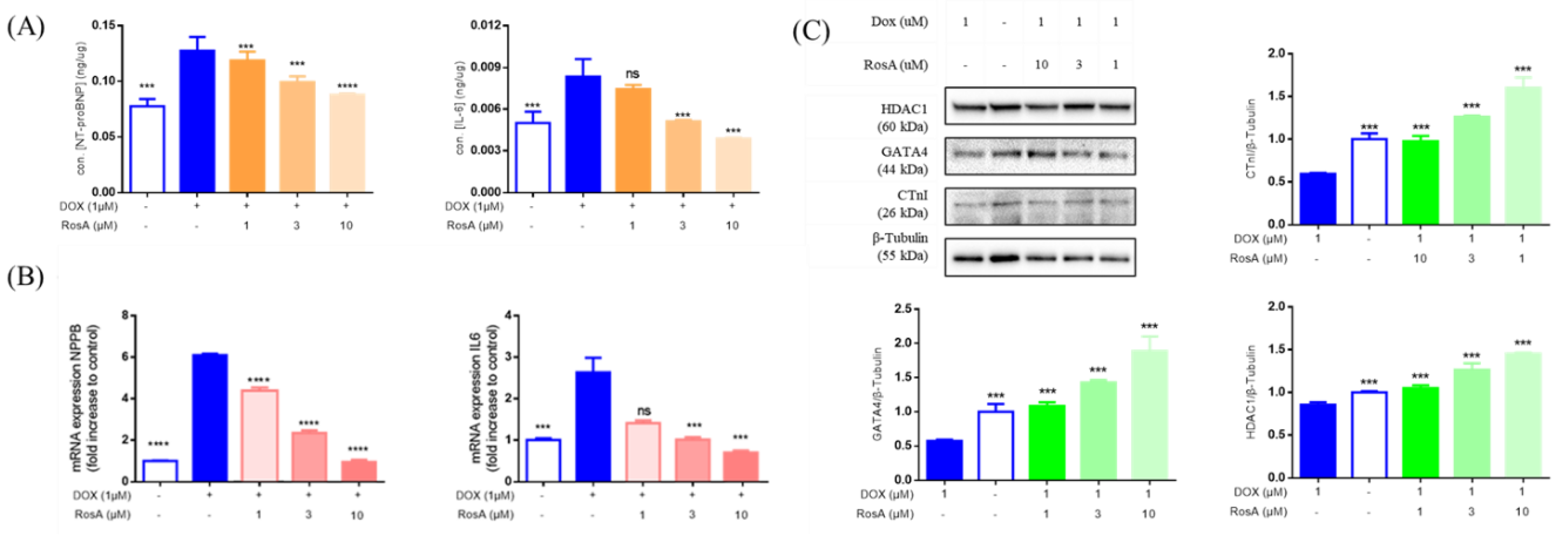
2.3. Using Morphology Pattern Recognition to Assess the Cardioprotection of Natural Compounds
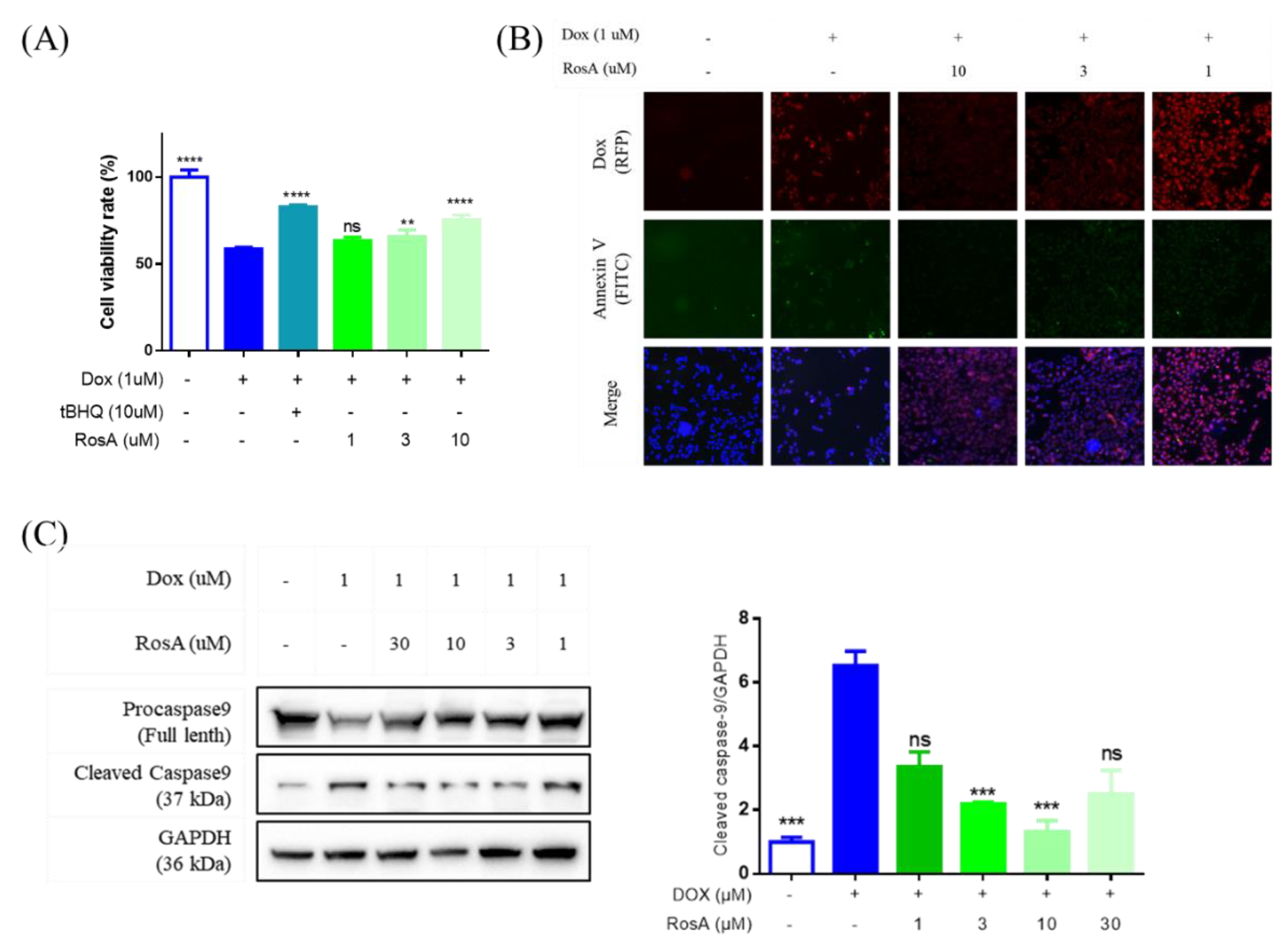
2.4. RosA Protects AC16 Cells Against Dox-Induced Cell Apoptosis
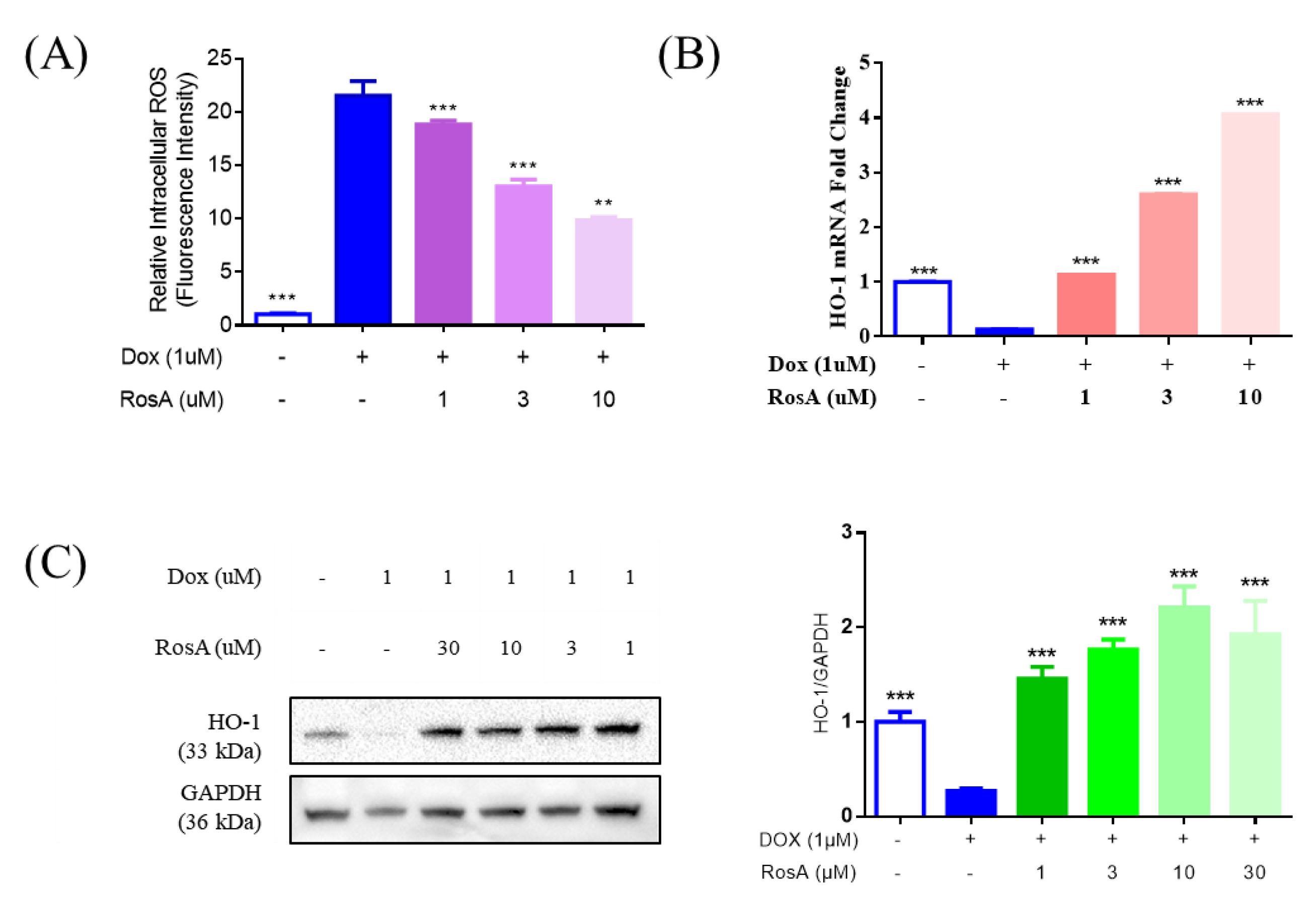
2.5. Pretreatment with RosA Results in Suppressed ROS and the Generation of HO-1
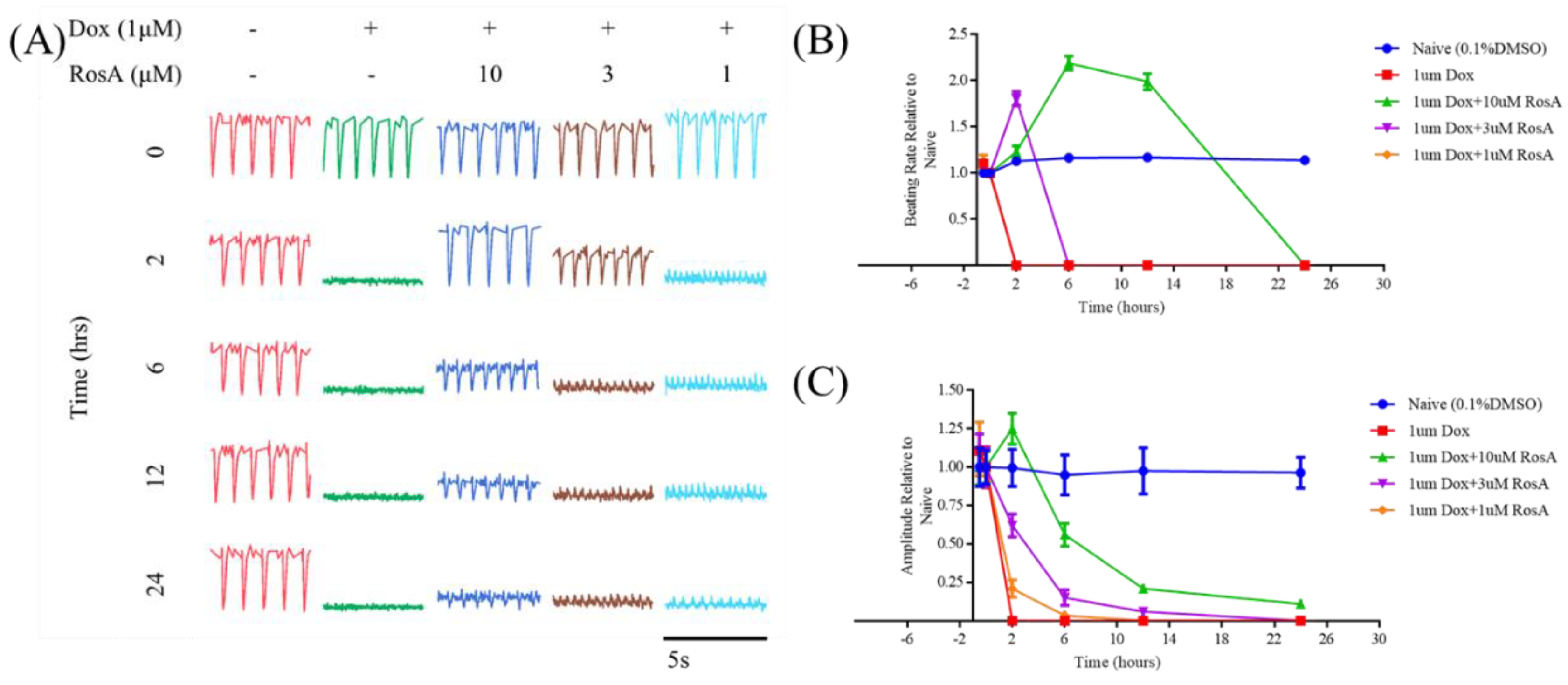
2.6. Pretreatment with RosA Impairs hiPS-CMs Muscle Function
3. Discussion
4. Materials and Methods
4.1. Chemicals and Reagents
4.2. Cell Cultures
4.3. Live-Cell Image Acquisition
4.4. Cell Viability Assay
4.5. Annexin V-FITC Staining Assay
4.6. Ros Generation Determined by FACS
4.7. ELISA Analysis
4.8. RNA Extraction and Quantitative PCR
4.9. Western Blotting
4.10. Cheminformatics, Image Quantification, Pattern Recognition, and Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Carver, J.R.; Shapiro, C.L.; Ng, A. American Society of Clinical Oncology Clinical Evidence Review on the Ongoing Care of Adult Cancer Survivors: Cardiac and Pulmonary Late Effects. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2007, 25, 3991–4008. [Google Scholar] [CrossRef] [PubMed]
- Silber, J.H.; Cnaan, A.; Clark, B.J. Enalapril to prevent cardiac function decline in long-term survivors of pediatric cancer exposed to anthracyclines. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2004, 22, 820–828. [Google Scholar] [CrossRef] [PubMed]
- Bloom, M.W.; Hamo, C.E.; Cardinale, D. Cancer Therapy-Related Cardiac Dysfunction and Heart Failure: Part 1: Definitions, Pathophysiology, Risk Factors, and Imaging. Circ. Heart Fail. 2016, 9, e002661. [Google Scholar] [CrossRef] [PubMed]
- Rahman, A.M.; Yusuf, S.W.; Ewer, M.S. Anthracycline-induced cardiotoxicity and the cardiac-sparing effect of liposomal formulation. Int. J. Nanomed. 2007, 2, 567–583. [Google Scholar]
- Bagheri, F.; Khori, V.; Alizadeh, A.M. Reactive oxygen species-mediated cardiac-reperfusion injury: Mechanisms and therapies. Life Sci. 2016, 43–55. [Google Scholar] [CrossRef]
- Moris, D.; Spartalis, M.; Tzatzaki, E. The role of reactive oxygen species in myocardial redox signaling and regulation. Ann. Transl. Med. 2017, 5, 324. [Google Scholar] [CrossRef]
- Teske, A.J.; Linschoten, M.; Kamphuis, J.A.M. Cardio-oncology: An overview on outpatient management and future developments. Neth. Heart J. 2018, 26, 521–532. [Google Scholar] [CrossRef]
- Hao, E.; Mukhopadhyay, P.; Cao, Z. Cannabidiol Protects against Doxorubicin-Induced Cardiomyopathy by Modulating Mitochondrial Function and Biogenesis. Mol. Med. 2015, 21, 38–45. [Google Scholar] [CrossRef]
- Hamo, C.E.; Bloom, M.W.; Cardinale, D. Cancer Therapy-Related Cardiac Dysfunction and Heart Failure: Part 2: Prevention, Treatment, Guidelines, and Future Directions. Circ. Heart Fail. 2016, 9, e002843. [Google Scholar] [CrossRef]
- Das, S.; Santani, D.D.; Dhalla, N.S. Experimental evidence for the cardioprotective effects of red wine. Exp. Clin. Cardiol. 2007, 12, 5–10. [Google Scholar]
- Samarghandian, S.; Borji, A.; Farkhondeh, T. Evaluation of Antidiabetic Activity of Carnosol (Phenolic Diterpene in Rosemary) in Streptozotocin-Induced Diabetic Rats. Cardiovasc. Hematol. Disord. Drug Targets 2017, 17, 11–17. [Google Scholar] [CrossRef] [PubMed]
- Javidanpour, S.; Dianat, M.; Badavi, M. The inhibitory effect of rosmarinic acid on overexpression of NCX1 and stretch- induced arrhythmias after acute myocardial infarction in rats. Biomed. Pharmacother. 2018, 102, 884–893. [Google Scholar] [CrossRef] [PubMed]
- Cai, X.; Yang, F.; Zhu, L. Rosmarinic Acid, the Main Effective Constituent of Orthosiphon stamineus, Inhibits Intestinal Epithelial Apoptosis via Regulation of the Nrf2 Pathway in Mice. Molecules 2019, 24, 3027. [Google Scholar] [CrossRef]
- Prasannarong, M.; Saengsirisuwan, V.; Surapongchai, J. Rosmarinic acid improves hypertension and skeletal muscle glucose transport in angiotensin II-treated rats. BMC Complement. Altern. Med. 2019, 19, 165. [Google Scholar] [CrossRef] [PubMed]
- Chlopcikova, S.; Psotova, J.; Miketova, P. Chemoprotective effect of plant phenolics against anthracycline-induced toxicity on rat cardiomyocytes. Part II. caffeic, chlorogenic and rosmarinic acids. Phytother. Res. PTR 2004, 18, 408–413. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.S.; Kim, H.R.; Woo, E.R. Inhibitory effects of rosmarinic acid on adriamycin-induced apoptosis in H9c2 cardiac muscle cells by inhibiting reactive oxygen species and the activations of c-Jun N-terminal kinase and extracellular signal-regulated kinase. Biochem. Pharmacol. 2005, 70, 1066–1078. [Google Scholar] [CrossRef]
- Blanchet, L.; Smeitink, J.A.; Van Emst-De Vries, S.E. Quantifying small molecule phenotypic effects using mitochondrial morpho-functional fingerprinting and machine learning. Sci. Rep. 2015, 5, 8035. [Google Scholar] [CrossRef]
- Pejin, B.; Jovanovic, K.K.; Mojovic, M. New and Highly Potent Antitumor Natural Products from Marine-Derived Fungi: Covering the Period from 2003 to 2012. Curr. Top. Med. Chem. 2013, 13, 2745–2766. [Google Scholar] [CrossRef]
- Caicedo, J.C.; Cooper, S.; Heigwer, F. Data-analysis strategies for image-based cell profiling. Nat. Methods 2017, 14, 849–863. [Google Scholar] [CrossRef]
- Feng, Y.; Mitchison, T.J.; Bender, A. Multi-parameter phenotypic profiling: Using cellular effects to characterize small-molecule compounds. Nat. Rev. Drug Discov. 2009, 8, 567–578. [Google Scholar] [CrossRef]
- Carpenter, A.E.; Jones, T.R.; Lamprecht, M.R. CellProfiler: Image analysis software for identifying and quantifying cell phenotypes. Genome Biol. 2006, 7, R100. [Google Scholar] [CrossRef] [PubMed]
- Li, H.-D.; Xu, Q.-S.; Liang, Y.-Z. libPLS: An integrated library for partial least squares regression and linear discriminant analysis. Chemom. Intell. Lab. Syst. 2018, 176, 34–43. [Google Scholar] [CrossRef]
- Suykens, J.A.K.; Vandewalle, J. Least Squares Support Vector Machine Classifiers. Neural Process. Lett. 1999, 9, 293–300. [Google Scholar] [CrossRef]
- Chong, I.; Jun, C. Performance of some variable selection methods when multicollinearity is present. Chemom. Intell. Lab. Syst. 2005, 78, 103–112. [Google Scholar] [CrossRef]
- Ogawa, T.; De Bold, A.J. Brain natriuretic Peptide production and secretion in inflammation. J. Transplant. 2012, 2012, 962347. [Google Scholar] [CrossRef] [PubMed]
- Lu, D.F.; Yao, Y.; Su, Z.Z. Downregulation of HDAC1 is involved in the cardiomyocyte differentiation from mesenchymal stem cells in a myocardial microenvironment. PLoS ONE 2014, 9, e93222. [Google Scholar] [CrossRef]
- Pan, B.; Quan, J.; Liu, L. Epigallocatechin gallate reverses cTnI-low expression-induced age-related heart diastolic dysfunction through histone acetylation modification. J. Cell. Mol. Med. 2017, 21, 2481–2490. [Google Scholar] [CrossRef]
- Stefanovic, S.; Barnett, P.; Van Duijvenboden, K. GATA-dependent regulatory switches establish atrioventricular canal specificity during heart development. Nat. Commun. 2014, 5, 3680. [Google Scholar] [CrossRef]
- Serkova, N.J.; Standiford, T.J.; Stringer, K.A. The emerging field of quantitative blood metabolomics for biomarker discovery in critical illnesses. Am. J. Respir. Crit. Care Med. 2011, 184, 647–655. [Google Scholar] [CrossRef]
- Peng, J.Y.; Lin, C.C.; Chen, Y.J. Automatic morphological subtyping reveals new roles of caspases in mitochondrial dynamics. PLoS Comput. Biol. 2011, 7, e1002212. [Google Scholar] [CrossRef]
- Ahmad, T.; Aggarwal, K.; Pattnaik, B. Computational classification of mitochondrial shapes reflects stress and redox state. Cell Death Dis. 2013, 4, e461. [Google Scholar] [CrossRef] [PubMed]
- Lupertz, R.; Watjen, W.; Kahl, R. Dose- and time-dependent effects of doxorubicin on cytotoxicity, cell cycle and apoptotic cell death in human colon cancer cells. Toxicology 2010, 271, 115–121. [Google Scholar] [CrossRef] [PubMed]
- Coelho, A.R.; Martins, T.R.; Couto, R. Berberine-induced cardioprotection and Sirt3 modulation in doxorubicin-treated H9c2 cardiomyoblasts. Biochim. Biophys. Acta Mol. Basis Dis. 2017, 1863, 2904–2923. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Zhang, B. Doxorubicin induces cardiotoxicity through upregulation of death receptors mediated apoptosis in cardiomyocytes. Sci. Rep. 2017, 7, 44735. [Google Scholar] [CrossRef] [PubMed]
- Stockwell, D.R.B.; Peterson, A.T. Effects of sample size on accuracy of species distribution models. Ecol. Model. 2002, 148, 1–13. [Google Scholar] [CrossRef]
- Han, J.; Wang, D.; Ye, L. Rosmarinic Acid Protects against Inflammation and Cardiomyocyte Apoptosis during Myocardial Ischemia/Reperfusion Injury by Activating Peroxisome Proliferator-Activated Receptor Gamma. Front. Pharmacol. 2017, 8, 456. [Google Scholar] [CrossRef]
- Zhang, X.; Zhu, J.X.; Ma, Z.G. Rosmarinic acid alleviates cardiomyocyte apoptosis via cardiac fibroblast in doxorubicin-induced cardiotoxicity. Int. J. Biol. Sci. 2019, 15, 556–567. [Google Scholar] [CrossRef]
- Davidson, M.M.; Nesti, C.; Palenzuela, L. Novel cell lines derived from adult human ventricular cardiomyocytes. J. Mol. Cell. Cardiol. 2005, 39, 133–147. [Google Scholar] [CrossRef]
- Colatsky, T.; Fermini, B.; Gintant, G. The Comprehensive in Vitro Proarrhythmia Assay (CiPA) initiative—Update on progress. J. Pharmacol. Toxicol. Methods 2016, 81, 15–20. [Google Scholar] [CrossRef]
- Markousis-Mavrogenis, G.; Tromp, J.; Ouwerkerk, W. The clinical significance of interleukin-6 in heart failure: Results from the BIOSTAT-CHF study. Eur. J. Heart Fail. 2019, 21, 965–973. [Google Scholar] [CrossRef]
| Gene | Sequence (5′‒3′) | Product Length (bp) |
|---|---|---|
| HO-1 F | ACTGCGTTCCTGCTCAACAT | 133 |
| HO-1 R | GGGCAGAATCTTGCACTTTGT | |
| IL6 F | GGCACTGGCAGAAAACAACC | 90 |
| IL6 R | ACCAGGCAAGTCTCCTCATTG | |
| NPPB F | CTTTCCTGGGAGGTCGTTCC | 86 |
| NPPB R | GTTGCGCTGCTCCTGTAAC | |
| GAPDH F | CTTTGTCAAGCTCATTTCCTGG | 133 |
| GAPDH R | TCTTCCTCTTGTGCTCTTGC |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, Q.; Li, J.; Peng, S.; Zhang, Y.; Qiao, Y. Rosmarinic Acid as a Candidate in a Phenotypic Profiling Cardio-/Cytotoxicity Cell Model Induced by Doxorubicin. Molecules 2020, 25, 836. https://doi.org/10.3390/molecules25040836
Zhang Q, Li J, Peng S, Zhang Y, Qiao Y. Rosmarinic Acid as a Candidate in a Phenotypic Profiling Cardio-/Cytotoxicity Cell Model Induced by Doxorubicin. Molecules. 2020; 25(4):836. https://doi.org/10.3390/molecules25040836
Chicago/Turabian StyleZhang, Qiao, Jing Li, Sha Peng, Yanling Zhang, and Yanjiang Qiao. 2020. "Rosmarinic Acid as a Candidate in a Phenotypic Profiling Cardio-/Cytotoxicity Cell Model Induced by Doxorubicin" Molecules 25, no. 4: 836. https://doi.org/10.3390/molecules25040836
APA StyleZhang, Q., Li, J., Peng, S., Zhang, Y., & Qiao, Y. (2020). Rosmarinic Acid as a Candidate in a Phenotypic Profiling Cardio-/Cytotoxicity Cell Model Induced by Doxorubicin. Molecules, 25(4), 836. https://doi.org/10.3390/molecules25040836





