1. Introduction
Alkyl glucosides and their derivatives are environmentally friendly compounds that are used as industrially important non-ionic surfactants with high surface activity [
1]. Alkyl glucosides are biodegradable and have excellent foaming control, wetting, detergent and emulsifying properties. The structures of alkyl glucosides or alkyl polyglucosides consist of aliphatic alcohols and glucose unit(s) obtained from biomass feedstocks. Industrially, it is difficult to produce pure alkyl mono-glucosides. As a result, complex mixture of alkyl-mono-, di-, tri- and oligo-glucosides are produced. These mixtures are generally called alkyl polyglucosides [
2]. The products are characterised by the length of the alkyl chain and the average number of glucose units linked to it, which is referred to as the degree of polymerisation (DP).
The enzymatic synthesis of alkyl glucosides occurs through a process called transglycosylation [
3]. In the transglycosylation approach, activated glycosyl donors and an alcohol (as glycosyl acceptor) are used to generate a new glycosidic bond and water acts a competing nucleophile. A number of alkyl glucosides have been produced through enzymatic synthesis. These include, among others, methyl, ethyl, proponyl, butanyl, hexanyl and octanyl-
d-glucosides [
3,
4,
5]. Bhatia et al. [
6] reported that β-glucosidase is one of the most important enzymes responsible for synthesising alkyl glucosides. Cyclodextrin glycosyltransferase is also known to synthesise alkyl glucosides [
7].
Bhatia et al. [
6] reported that the glycoside hydrolase family 1 (GH1) β-glucosidase enzyme, with an (α/β)
8 barrel structure, displayed transglycosylation activity. The authors also suggested that xylanase/cellulase (PDB 2HIS), a family 26 lichenase (PDB 2CIP), β-glucosidase (PDB 1UG6), Cel7A (PDB 4C4C), arabinofuranosidase (PDB 2VRQ) and E-82 xylanase (PDB 2D24) are glycosyl hydrolase enzymes, which also display transglycosylation properties. The glycoside hydrolase family 5 is one of the biggest families in the CAZy database (
http://www.cazy.org). Most GH5 enzymes have been reported as hydrolases, which break down different carbohydrates and their conjugates molecules [
8,
9].
Among the GH5 enzymes, cellulases are a group of well-studied enzymes which generally hydrolyse cellulose. Cellulases consists of endoglucanases (EG, EC 3.2.1.4), exoglucanases/cellobiohydrolases (CBHI, EC 3.2.1.91 and CBHII, EC 3.2.1.176) and β-glucosidases (BGLs EC 3.2.1.21). GH5 exoglucanases hydrolyse cellulose from its chain ends through a retaining mechanism, producing short cellobiosides as products [
8]. Under favourable conditions, some exoglucanases can also perform trans-glycosylation reactions [
8]. To date, there have been known studies demonstrating transglycosylation reactions with respect to alkyl glucosides with the exoglucanases from GH5 enzymes (
http://www.cazy.org/GH5_38.html; [
9]).
In the current study, we present a novel oligomeric enzyme from GH5_38, Exg-D, exhibiting both hydrolase and trans-glycosylase activities. The dimeric and monomeric oligomers of the enzyme were purified to homogeneity through affinity and size exclusion chromatographic techniques. The pure oligomers were biochemically and structurally characterised. The transglycosylation activity of Exg-D was used to successfully synthesise methyl-, ethyl- and propyl-cellobiosides using p-nitrophenyl cellobioside as a glycosyl donor and several primary alcohols as acceptors.
3. Discussion
Exg-D is a member of GH family 5 subfamily 38, which includes 27 carbohydrate active enzymes identified to date. Twenty of these enzymes are of bacterial origin (including Exg-D: GenBank ID AMO13174), while seven were unclassified at the time of the current study. Exg-D was sourced from the termite hindgut via metagenomic techniques by Rashamuse et al. [
10]. The protein has about 379 amino acid residues, with 21 amino acids constituting the transmembrane signalling of the protein. Tamura et al. [
11] demonstrated that the
Bacteroides ovatus had a GH 16 enzyme that was localised on the membrane and its function was to reduce the length of mixed-linkage-β-(1,3)-(1,4)-glucan to di- or trisaccharides that were transported to the periplasmic region. This shows that the transmembrane signalling sequence of the Exg-D is common among the gut bacterial species and GH enzymes.
The overexpression of Exg-D was optimised using 2× YT liquid Luria-Bertani (LB) medium with 100 µg/mL ampicillin. The results revealed that Exg-D was expressed successfully in T7
E. coli cells and the concentration levels were also above 1.8 mg/mL (30 µM). After expression, Exg-D was purified with a Ni
2+-charged IMAC column as the enzyme was tagged with polyhistidine. To achieve homogeneity of the pure enzyme, Exg-D samples were further purified with a G75 Sephadex gel filtration column. IMAC columns are widely used to purify polyhistidine-tagged proteins, while gel filtration columns are used to purify proteins and also to demonstrate the quaternary oligomeric state (monomeric or dimeric) of proteins in solution [
11,
12,
13]. Interestingly, the gel filtration results demonstrated that Exg-D exists as both a dimer and monomer in solution (
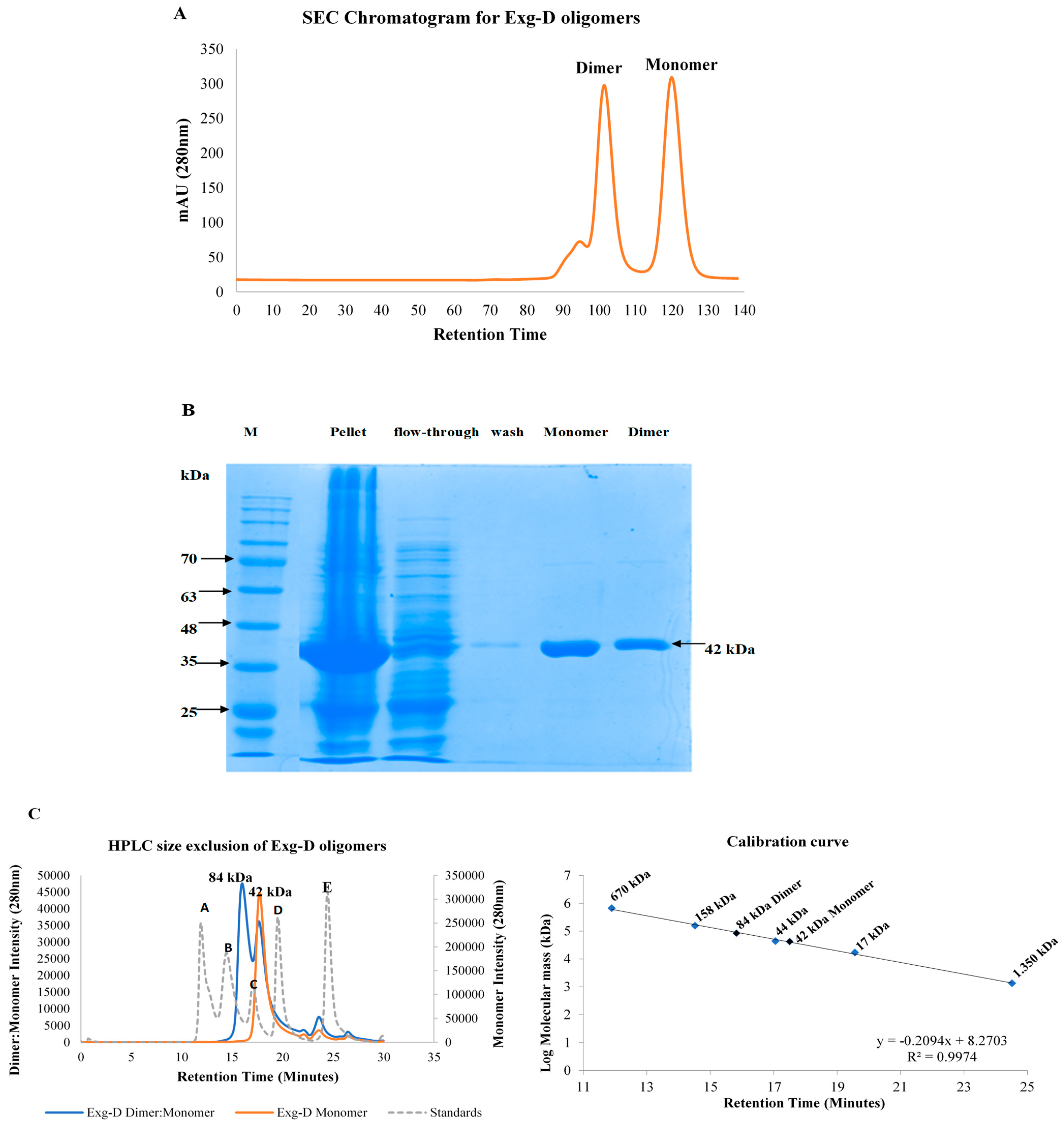
Figure 1).
HPLC size exclusion confirmed that the Exg-D exists as a dimer and a monomer, with molecular masses of 84 kDa and 42 kDa, respectively. The dimer and monomer were easily collected into two different fractions because there was a significant difference in the retention times of the two peaks representing these Exg-D oligomers. Further studies probing the nature of the dimer and the monomer revealed that the dimer fraction always consisted of the dimer–monomer combination. This combination was concentration dependent—during high concentrations, the equilibrium shifted to form more of the dimer and vice versa. In contrast, the collected monomeric fraction from G75 gel filtration did not form the dimer–monomer combination (
Figure 1C). The GH enzymes generally occur in more than one form; for instance, the bacterial GH 67 α-glucuronidases and the GH 52 β-xylosidase or fungal GH 30_7 exist as dimeric or monomeric oligomers in solution [
12,
13,
14].
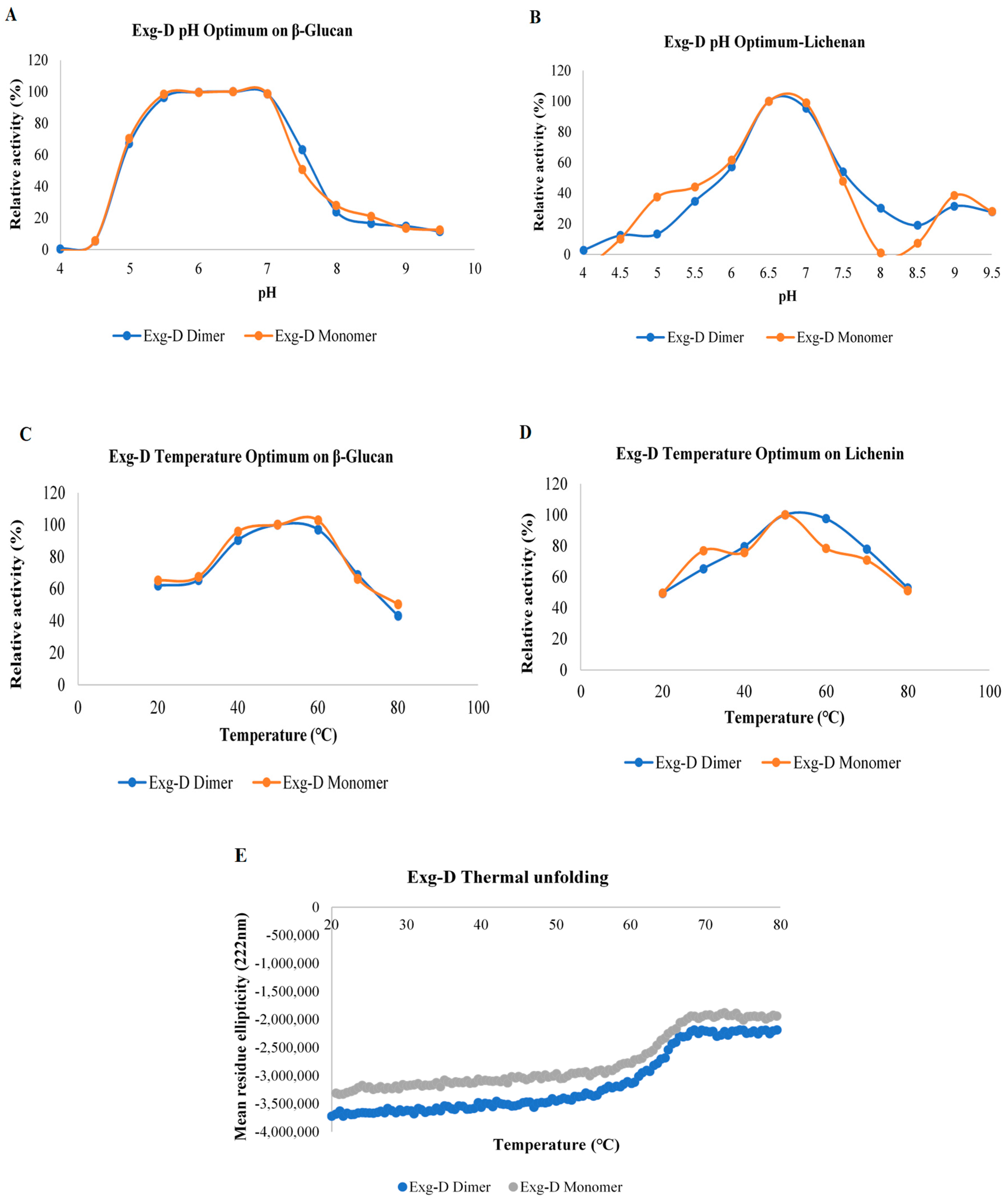
The specific activity of Exg-D revealed that both monomeric and dimeric forms of the enzyme were active. The dimer displayed a higher activity of about two activity units (compared to the monomer) on the tested substrates, except for pachyman and curdlan. The active monomer of the Exg-D enzyme was found to be novel, as several studies have generally shown that dimeric oligomers of the GH enzymes are active, while their monomers are not active or display very low activity [
13,
15,
16,
17]. The specific activity results were supported by biochemical characterisation and kinetic studies, which showed that the dimeric and monomeric species of Exg-D displayed the same pH and temperature optima. Both Exg-D enzyme oligomers demonstrated a broad pH and temperature optima range on β-glucan. The kinetics studies of the Exg-D also showed that it had the capacity to break down β-glucan (a
Km of 7.9 mg/mL and a
kcat of 117.2 s
−1) compared to lichenin (a
Km of 21.5 mg/mL and a
kcat 70.0 s
−1). The result confirmed the novelty of Exg-D, which has active monomeric and dimeric forms.
Aspeborg et al. [
9] demonstrated that enzymes belonging to GH5_38 are not well characterised; out of 27 proteins in this subfamily, only 3 are partially characterised. Thus, the significance of the present study among other things was to biochemically and structurally characterise Exg-D classified under GH5_38 (GenBank ID: AMO13174.1). The GH16 elongating β-transglycosylase from
Paecilomyces thermophila (PtBgt16A), which also performed hydrolysis of mixed linkages, showed a pH optimum of 5.5 and a temperature optimum of 60 °C [
18]. Additionally, a recent study showed the GH5A enzyme from
Talaromyces leycettanus JCM12802 to have a high specific activity on a mixed-linkage substrate [
19]. The enzyme showed a pH optimum of 3 and a temperature optimum of 75 °C. In contrast, Exg-D showed a broad pH optimum from 5.5 to 7 and a temperature optimum from 40 to 60 °C. The broad pH and temperature optima of Exg-D enzyme make it a potential candidate for the hydrolysis of lichenin- or β-glucan-containing biomass.
The oligosaccharide hydrolysis product patterns showed a novel mechanism by which Exg-D enzyme was employed to break down the β-(1,3)-(1,4)-mixed-linkage oligosaccharides. The 3
1-cellotriosyl-glucose (G4G4G3G) was effectively hydrolysed by the enzyme, while the enzyme showed no activity on the 3
2-glucosyl-cellobiose (G3G4G). The findings suggest that at least two β-1,4-glycosidic bonds must be joined to a β-1,3-glycosidic bond for hydrolysis to occur. Tamura et al. [
11] proposed that GH16 enzymes hydrolyse mixed-linkage oligosaccharides at the β-1,4-glycosidic bond which follows a β-1,3-glycosidic bond; hence, the GH16 enzymes were able to hydrolyse 3
2-glucosyl-cellobiose (G3G4G). Additionally, the GH16 enzymes were shown to be prolific hydrolysers of laminari-oligosaccharides [
11,
18]. In contrast, Exg-D showed lost activity on laminaripentaose. This distinction between GH16 enzymes and Exg-D emphasised the novelty of the mechanism employed by Exg-D during the hydrolysis of mixed-linkage oligosaccharides/substrates. In addition, Exg-D was also highly active on cello-oligosaccharides, and it employed the well-studied processive mechanism of exoglucanases [
19].
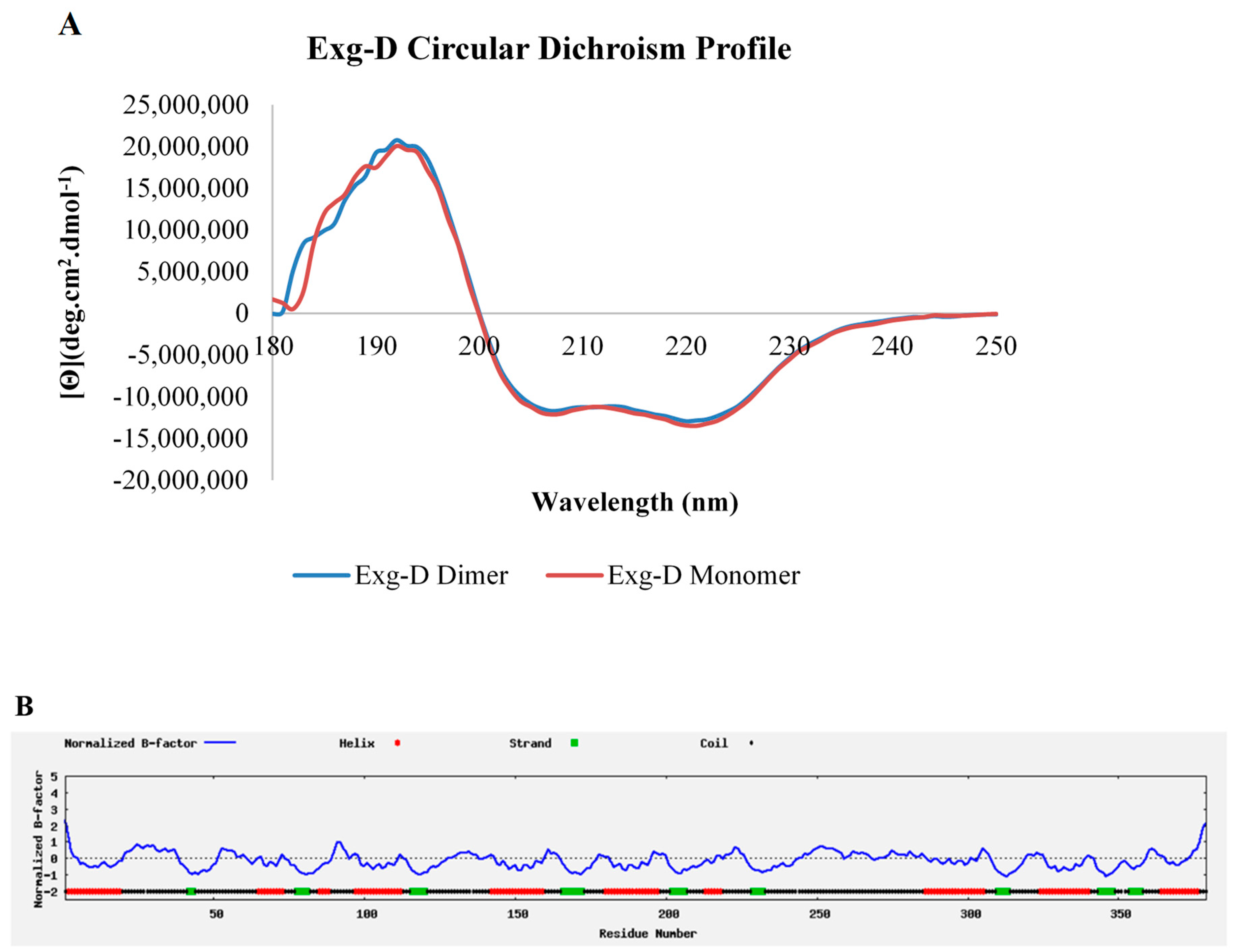
Exoglucanases are known for hydrolysing their substrates in a processive manner, meaning that the enzyme moves along the backbone of cellulose or glucan, releasing cellobiose [
9,
18,
19]. The processivity of the exoglucanases is linked to their tunnel-like-cleft active site, as demonstrated by Qin et al., [
18]. Annamalai et al. [
20] also argued that CBH I and CBH II from GH7 and GH6 were highly processive due to the topology of the tunnel-like active sites they possess. The current study structurally characterised a termite-hindgut-bacterial-metagenome-derived exoglucanase, Exg-D. The secondary structure of Exg-D consisted of eight α-helices and eight β-strands which were joined by coils. The modelled 3D structure of the Exg-D enzyme resembled the GH5 structure reported by Davies and Sinnott, [
8], which is a classical TIM barrel (β/α). The surface topology of Exg-D demonstrated that the catalytic site of the modelled 3D structure of Exg-D was a tunnel-like cleft. Thus, it allowed the enzyme to hydrolyse its substrate in a processive manner, or possibly to perform trans-glycosylation [
21,
22].
Exg-D performed the transglycosylation activity as hypothesised based on its structural elements. In the present study, Exg-D synthesised methyl-, ethyl- and propyl-cellobiosides. Several other GH enzymes have also been reported to act as trans-glycosylases, for example β-glucosidase, and cellobiose phosphorylase can produce alkyl glucosides [
5,
21,
23,
24,
25]. To the best of our knowledge, this is the first study demonstrating that GH5_38 exo-glucanases (Exg-D) can perform transglycosylation. Most enzymes hydrolyse polysaccharides or disaccharides to monosaccharides (particularly glucose) first and then use the produced monosaccharides as glycosyl donors, while alcohol is used as an acceptor [
26,
27]. In contrast, Exg-D hydrolysed
pNPC, releasing cellobiose and
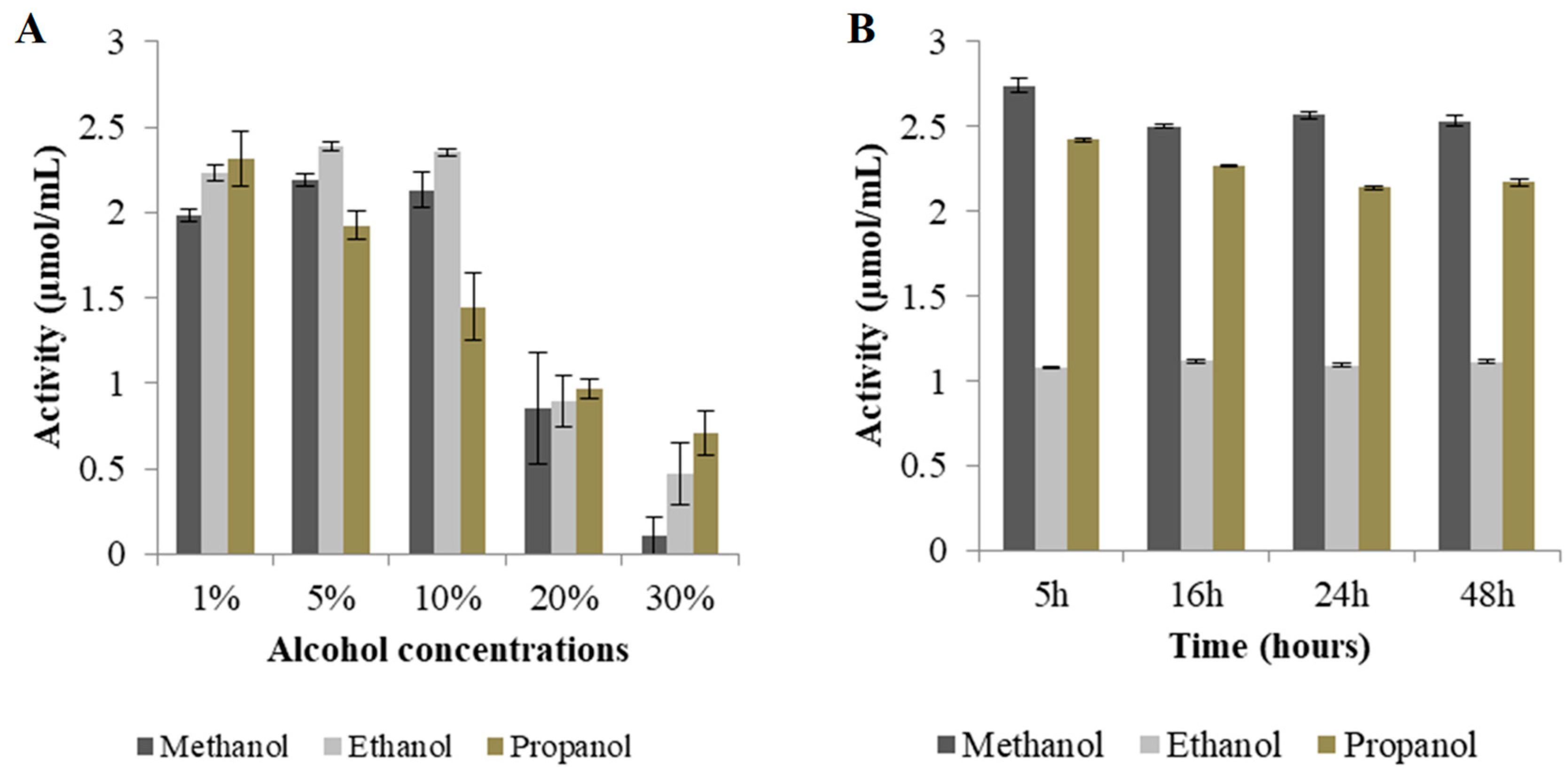
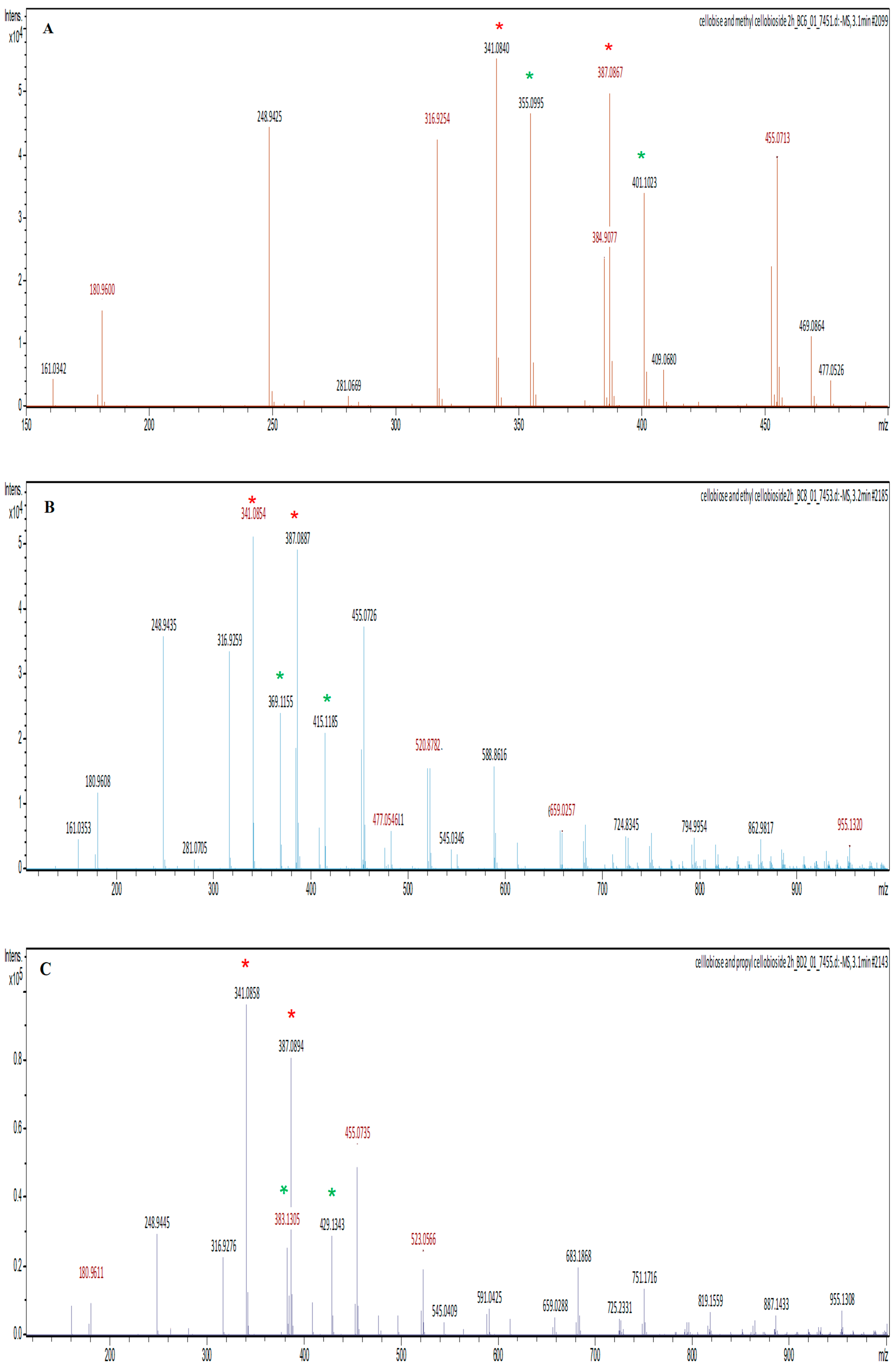
p-nitrophenol. Subsequent to the hydrolysis process, the enzyme donated cellobiose to the alcohols. The ESI results showed that the transfer of the cellobiose to methanol resulted in a higher yield of alkyl cellobiosides compared to when longer chain alcohols were used. The enzyme also synthesised appreciable yields of ethyl cellobioside followed by propyl cellobiose. These results suggest that Exg-D can be used only for the synthesis of these short alkyl cellobiosides, as the enzyme lost its trans-glycosylase activity in the presence of higher/longer alcohol (hexanol) acceptors. These results suggest that the transfer of cellobiose to alcohol molecules with a longer chain requires more energy.
4. Experimental Section
4.1. Protein Sequence Analysis, Cloning and Transformation
The amino acid sequence of the exoglucanase (Exg-D) was recovered from the NCBI GenBank (sequence ID: AMO13174.1), followed by analysis for a possible transmembrane signal peptide sequence using the SignalP 4.1 Server. This showed that the Exg-D sequence contains a transmembrane signal with about 21 amino acids. The Exg-D sequence without the transmembrane signal was sent to GenScript (Piscataway, New Jersey. USA) to cloning the Exg-D gene into a pET-11a plasmid. The pET-11a constructs harbouring the Exg-D gene were used to transform Escherichia coli T7 competent cells. The E. coli T7 competent cells were transformed with 50 ng (2 µL) of pET 11a construct containing the Exg-D gene insert, mixed gently and incubated on ice for 30 min. Subsequently, cells were heat-shocked for 45 s at 42 °C. The cells were then placed on the ice for 2 min and SOC media (20 g/L Tryptone, 5 g/L Yeast Extract, 4.8 g/L MgSO4, 3.603 g/L dextrose, 0.5g/L NaCl and 0.186 g/L KC) was added to the transformed cells and controls. The cells in the SOC medium were allowed to recover by incubation in a shaker incubator (New Braunswick™ Excella®, Hamburg, Germany) with a speed of 225 rpm for 1 h at 37 °C. The transformed cells of about 10 µL were streaked onto 2× YT agar plates (16 g/L tryptone, 10 g/L teast extract, 5 g/L NaCl, 15 g/L agar) containing 100 µg/mL ampicillin and incubated at 37 °C overnight (16 h).
4.2. Heterologous Overexpression of Exg-D
The overexpression of Exg-D was optimised and performed by selecting a single colony of transformed E. coli cells. For overexpression of Exg-D, the medium (a 2× YT (16 g/L tryptone, 10 g/L yeast extract, 5 g/L NaCl) supplemented with 100 µg/mL ampicillin) was incubated at 37 °C in a shaking incubator until the cells reached mid-log phase (OD600 = 0.6). The overexpression of Exg-D was induced by adding 1 mM isopropyl-β-D-1-thiogalactopyranoside (IPTG) into 500 mL of 2× YT medium containing cells, and the cells were incubated at 30 °C in a standing incubator shaking at 150 rpm for 20 h. The cells were harvested by centrifugation (5000× g) for 20 min at 4 °C. The cell pellet was resuspended in the lysis buffers (20 mM NaH2PO4, 0.5 M NaCl, 1 mM CaCl2 and 20 mM imidazole, pH 7.0), lysed through pulse sonication (50 Hz) on ice and centrifuged at 5000× g for 20 min to separate the Exg-D soluble protein fraction from the cell debris.
4.3. Protein Purification of Exg-D
Purification of Exg-D soluble protein fraction was achieved by immobilised metal ion affinity chromatography (IMAC) charged with nickel ion (Ni
2+), followed by size exclusion chromatography (SEC). The crude protein obtained from lysed
E. coli cells was loaded onto the nickel-charged affinity column (GE Healthcare) using an AKTA-Prime fast pressure liquid chromatography (FPLC) purification system. The IMAC column was equilibrated with the 20 mM sodium phosphate equilibration buffer (20 mM of sodium monobasic phosphate, 20 mM sodium dibasic phosphate, 500 mM NaCl and 20 mM imidazole at pH 7). Subsequently, the column was washed with the equilibration buffer until the absorbance values at 280 nm of the effluent reached baseline to ensure that all unbound proteins were washed from the column. The poly-histidine-tagged Exg-D was eluted from the IMAC column using an elution buffer (20 mM of sodium monobasic phosphate, 20 mM sodium dibasic phosphate, 500 mM NaCl and 300 mM Imidazole at pH 7). Exg-D eluted from the IMAC column was purified further using a SEC column (HiLoad™ 16/600 Superdex™ 75pg) connected to an AKTA-Prime system. The SEC column was equilibrated at a flow rate of 0.5 mL/min with 20 mM sodium phosphate buffer containing 500 mM NaCl at pH 7. Exg-D eluted from IMAC column was loaded on the AKTA-Prime system. The two protein peaks showing the highest absorbance values at 280 nm were collected separately and tested for Exg-D activity with
p-nitrophenol-β-
d-cellobioside (a yellow colour change indicated activity). Exg-D was dialysed with dialyses tubes with a 10 kDa molecular weight cut-off in a 20 mM sodium phosphate buffer at pH 7.0 and stored on ice or 4 °C. The level of purity of Exg-D was analysed via 12% SDS-PAGE [
28].
4.4. Protein Concentration Determination
The concentration of purified Exg-D was determined using the absorbance values of the monomer or dimer at 280 nm and the extinction coefficient of the protein (74 499 M
−1 cm
−1). A Jasco (V-630 UV-VIS) spectrophotometer was used to measure the concentration of the monomeric or dimeric oligomers Exg-D in a glass cuvette. All measurements were performed in triplicate and the purified protein was subsequently determined using the Beer–Lambert law:
where
A is the absorbance value at 280 nm,
ε is the molar extinction coefficient of Exg-D at 280 nm (M
−1 cm
−1),
c is the concentration of the protein in M and
l is the path length of the cuvette (1 cm). The molar extinction coefficient of Exg-D was calculated using the extinction coefficients of tryptophan (W), tyrosine (Y) and cysteine (C) residues [
29]:
Absorbance at 280 nm of the protein dilution series was determined by fitting a linear regression line to the absorbance values of Exg-D. All readings were corrected for buffer (20 mM sodium phosphate at pH 7.5).
4.5. Analysis of the Oligomeric State of Exg-D in Solution
The dimer and monomer oligomers of Exg-D were further investigated via high-performance liquid chromatography (HPLC: Shimadzu, Duisburg, Germany) and AKTA-Prime (FLPC), respectively. The two peaks that were collected separately from the SEC column were analysed with gel filtration HPLC, using a TSK-Gel S3000WXL column (Tosoh, Tokyo, Japan) at 20 °C. The mobile phase was 20 mM sodium phosphate buffer (pH 7.0) containing 500 mM NaCl, using an isocratic flow rate of 0.2 mL/min for 30 min, and the protein was detected at 280 nm. Aliquots of 20 µL of 1 mg/mg to 5 mg/mg Exg-D enzyme were loaded onto the HPLC system to elucidate the oligomeric nature of this protein.
4.6. Specific Activity Determination
The specific activity of dimer and monomer oligomers of the Exg-D was tested by using β-1,3-glycosidic linked substrates (pachyman and curdlan from Megazyme), β-1,4-glycosidic linked substrates (carboxymethylcellulose and Avicel PH-101) and mixed-linkage β-(1,3)-(1,4)- substrates (lichenin and β-glucan from Megazyme). The reaction was initiated by mixing 1% (
w/v) of the substrate with Exg-D (10 µg/mg substrate) in 20 mM sodium phosphate buffer (pH 7) at 37 °C for 30 min. Exg-D activity was determined using a modified 3,5-dinitrosalicylic acid (DNS) method [
30]. The modified procedure was performed as described previously [
31]. Glucose was used as a suitable standard.
Exg-D activity was also tested using
p-nitrophenol-β-D-cellobioside (
pNPC) purchased from Sigma. The reaction was initiated by mixing the 4 mM
pNPC with Exg-D (10 µg/mg substrate) in 20 mM sodium phosphate buffer (pH 7) at 37 °C for 10 min. The para-nitrophenol released by the enzyme action was measured as described by Malgas et al. [
32].
4.7. Biochemical Properties and Kinetic Studies
To determine the pH optima of the dimeric or monomeric Exg-D, the enzyme with substrate (10 µg/mg substrate) were incubated in 20 mM citric acid–sodium phosphate buffer at pH 4, 4.5, 5, 5.5; 20 mM sodium phosphate at pH 6, 6.5, 7; and 20 mM Tris-HCl buffer at pH 7.5, 8, 8.5, 9 and 9.5, containing 1% (w/v) lichenin and β-glucan, respectively. Temperature optima were also determined by dissolving the 1% (w/v) lichenin and β-glucan in 20 mM sodium phosphate buffer at temperatures ranging between 20 and 80 °C. The DNS method was used to determine the total reducing sugars released by enzymatic activity.
The thermostabilities of the dimeric and monomeric Exg-D were assessed continuously from 20 to 80 °C with a circular dichroism (CD) spectropolarimeter (Jasco 1500). To detect the thermal unfolding due to increasing temperature (20 and 80 °C), CD spectral measurements were recorded with a spectral bandwidth of 5 nm, a data pitch of 1 nm and a quartz cuvette with a path length of 2 mm. The spectra were recorded as an average of seven scans at a scan speed of 100 nm/min in triplicate. The protein concentration used was 10 μM in 5 mM sodium phosphate at pH 7 to minimise the signal to noise ratio. The spectra were converted from θ (m.deg) to ΘMRE using the following equation:
where Θ is the mean residual ellipticity (deg.cm
2.dmol
−1), θ is the measured ellipticity (mdeg),
c is the protein concentration (mM), n is the number of residues in the protein and
l is the path length (cm).
The enzyme kinetics studies of monomeric Exg-D were performed by varying the concentrations (2 to 17.5 mg/mL) of the β-glucan and lichenin dissolved in 20 mM sodium phosphate (pH 7). The Vmax, and the Km values of Exg-D were measured under steady-state/zero-order conditions and the kcat was measured from first-order conditions. The reactions were performed at 37 °C for 30 min, heated at 100 °C to terminate the reaction and the total reducing sugars were quantified using DNS assays. Kaleidagraph and Excel-solver were used to determine the Vmax and Km values, while kcat values were derived from the slopes of pseudo-first-order kinetics.
4.8. Oligosaccharide Hydrolysis Product Patterns
Oligosaccharides were used to study the hydrolysis product patterns of Exg-D and also to establish which of the following bonds: (1) β-1,3-D-glycosidic bonds, (2) β-1,4-D-glycosidic bonds or (3) mixed-linkage β-(1,3)-(1,4)-D-glycosidic bonds were hydrolysed by these enzymes. The reaction was initiated by mixing Exg-D (10 µg/mg substrate) with 5 mg/mL of the oligosaccharides, and the reaction was carried out for 2 h to allow the substrate to be completely converted to products. The reaction was quenched by heating at 100 °C. Laminaripentose was used to study hydrolysis of β-1,3-D-glycosidic bonds by Exg-D. Cellotriitol, cellotetraitol, cellopentaitol, cellopentaose and cellohexaitol were used to study β-1,4-D-glycosidic bond enzymatic activity. The mixed-linked 32-β-D-glucosyl-cellobiose and 31-β-D-cellotriosyl-glucose substrates were used to investigate the order of bonds and the number of β-1,4-D-glycosidic bond required for hydrolysis/cleavage to occur. The cello-oligosaccharides with the suffix “itol” were reduced with borohydride and all the oligosaccharides were purchased from Megazyme. The hydrolysis patterns were detected using thin-layer chromatography (TLC).
4.9. Thin-Layer Chromatography
Thin-layer chromatography was used to identify and confirm the oligosaccharide hydrolysis products of Exg-D. The reaction products were analysed using Silica Gel 60G F254 HPTLC plates (Merck, Darmstadt, Germany). Plates were developed twice with n-butanol:acetic acid:water (2:1:1, v/v/v). To detect the oligosaccharides, plates were briefly submerged in methanol containing 5% (v/v) sulphuric acid and 0.3% (w/v) α-naphthol (Molisch’s reagent). Plates were air-dried and heated at 120 °C for 10 min to develop the chromatogram.
4.10. Secondary Structure and 3D Modelling of Exg-D
The secondary structures of the dimeric and monomeric Exg-D were determined with a far-UV CD spectropolarimeter. The CD spectral measurements were performed from 180 to 250 nm at 20 °C using a Jasco-1500 spectropolarimeter as described above (
Section 2.4). The Exg-D protein concentration used was 5 μM in 5 mM sodium phosphate at pH 7 to minimise the signal to noise ratio. The spectra were converted from θ (m.deg) to ΘMRE was calculated using Equation (3). The CD spectroscopic data were further quantitatively analysed using the Dichroweb server using Contin-LL (Provencher & Glockner Method) to determine the secondary structural content of the Exg-D [
33].
Secondary structural characterisation of Exg-D was performed using the I-TASSER server. The full amino acid sequence of Exg-D was submitted to the I-TASSER server (and was assigned ID S505239) for modelling of the secondary and 3D structure of Exg-D according to References [
34,
35].
4.11. Alkyl Cellobioside Synthesis Assays
The alkyl cellobioside synthesis assays (through transglycosylation) were investigated using
pNPC as a substrate. The standard reaction mixture contained 5% (
v/v) alcohol (methanol, ethanol or propanol), 4 mM
pNPC dissolved in a 50 mM sodium citrate buffer (pH 5.5) and 10 µg of Exg-D. The reaction was measured as described in
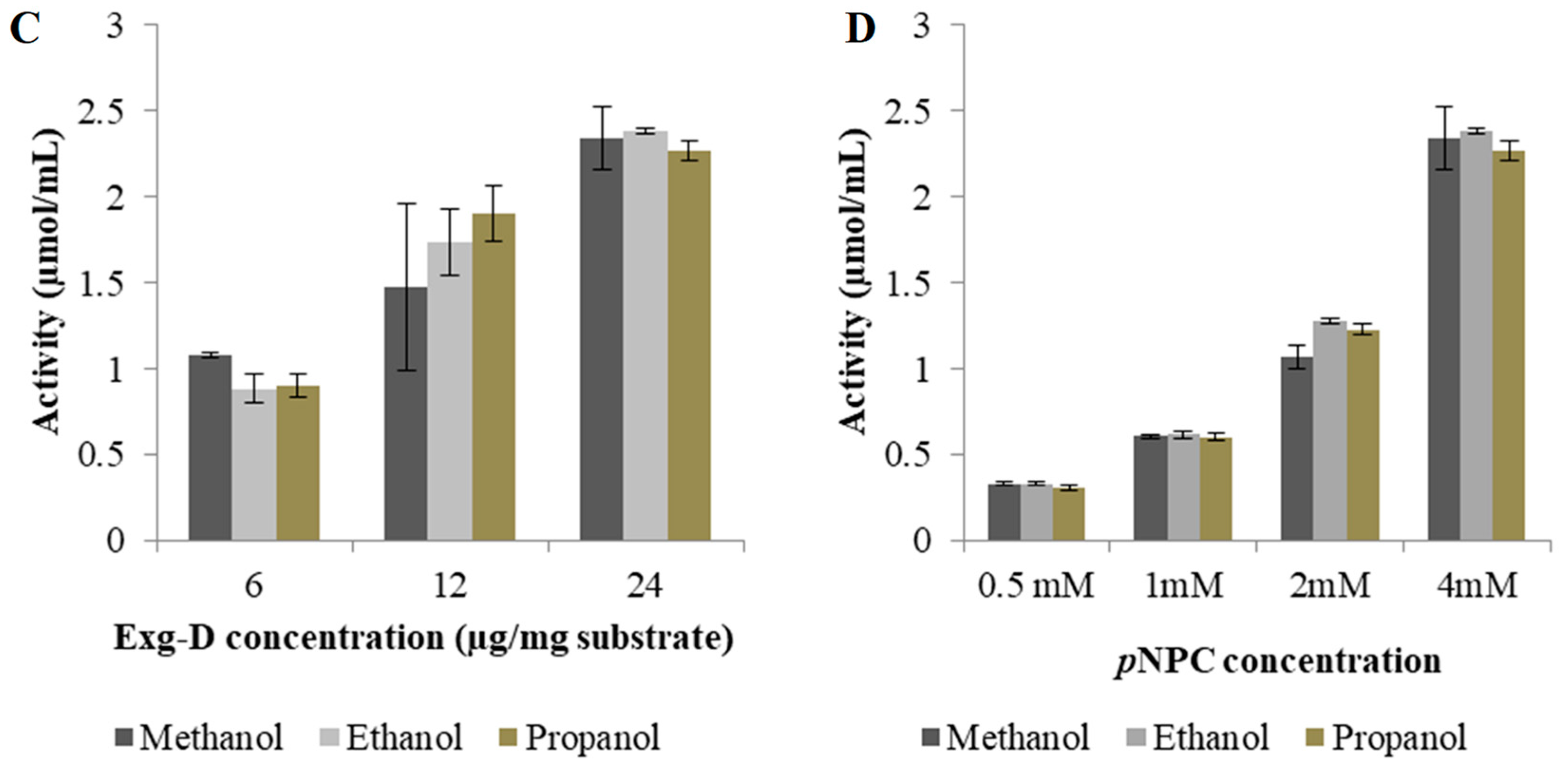
Section 2.6. The optimal conditions for producing the alkyl cellobiosides were investigated by varying: (1) the reaction time from 1 to 6 h, (2) alcohol concentration from 1% to 30% (
v/v), (3) enzyme concentration from 6 µg/mg to 24 µg/mg substrate and (4) substrate concentration from 0.5 to 4 mM. The alkyl cellobiosides produced during optimisation were measured qualitatively and quantitatively by TLC, LC-MS and by the
p-nitrophenol released from the reaction, respectively.
Liquid chromatography–mass spectrometry (LC-MS) was performed in the negative mode to identify and quantify alkyl cellobiosides. LC-MS used mass to charge ratio for identification of the alkyl cellobiosides and electrospray ionisation (ESI) profiles were obtained for quantification. The instrument used was an Ultimate 3000 Thermo-Scientific UHPLC with a Fortis UHPLC C18 column (1.7 μm, 2.1 × 50 mm) and detection was performed by Bruker Compact QqTOF operated in the negative electrospray ionisation mode scanning from 100–1000 daltons. The mobile phase was 50:50 water: acetonitrile with 0.1% (v/v) formic acid. The injection volume was 5 μL of ~2.5–20 mM sample solution dissolved in sodium acetate buffer. The masses of the samples were analysed and quantified with Thermo Scientific™ Dionex™ Chromeleon™ 7.2 Chromatography Data System (CDS) software.