Iron(II) Spin Crossover Complexes with 4,4′-Dipyridylethyne—Crystal Structures and Spin Crossover with Hysteresis
Abstract
1. Introduction
2. Results
2.1. Synthesis
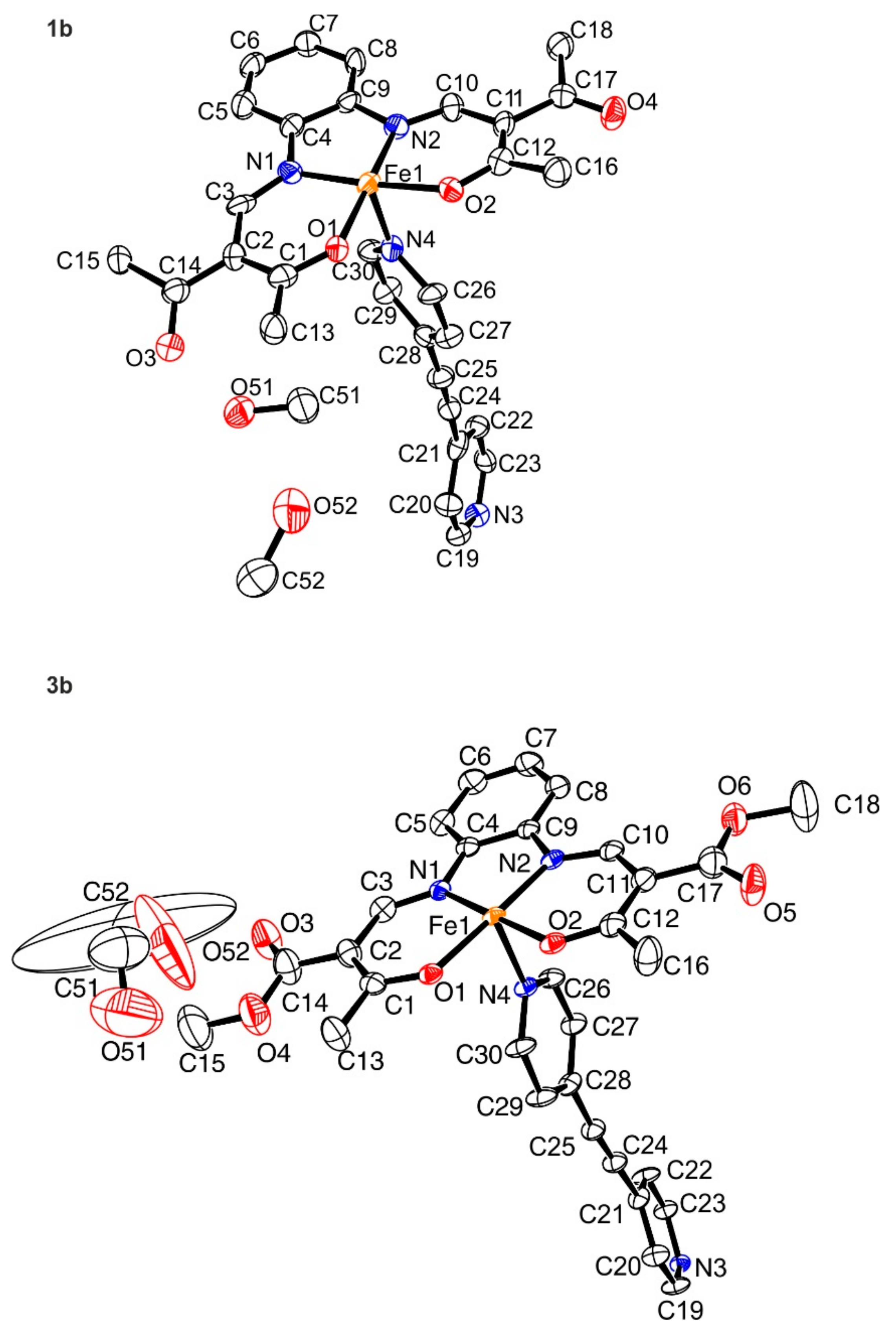
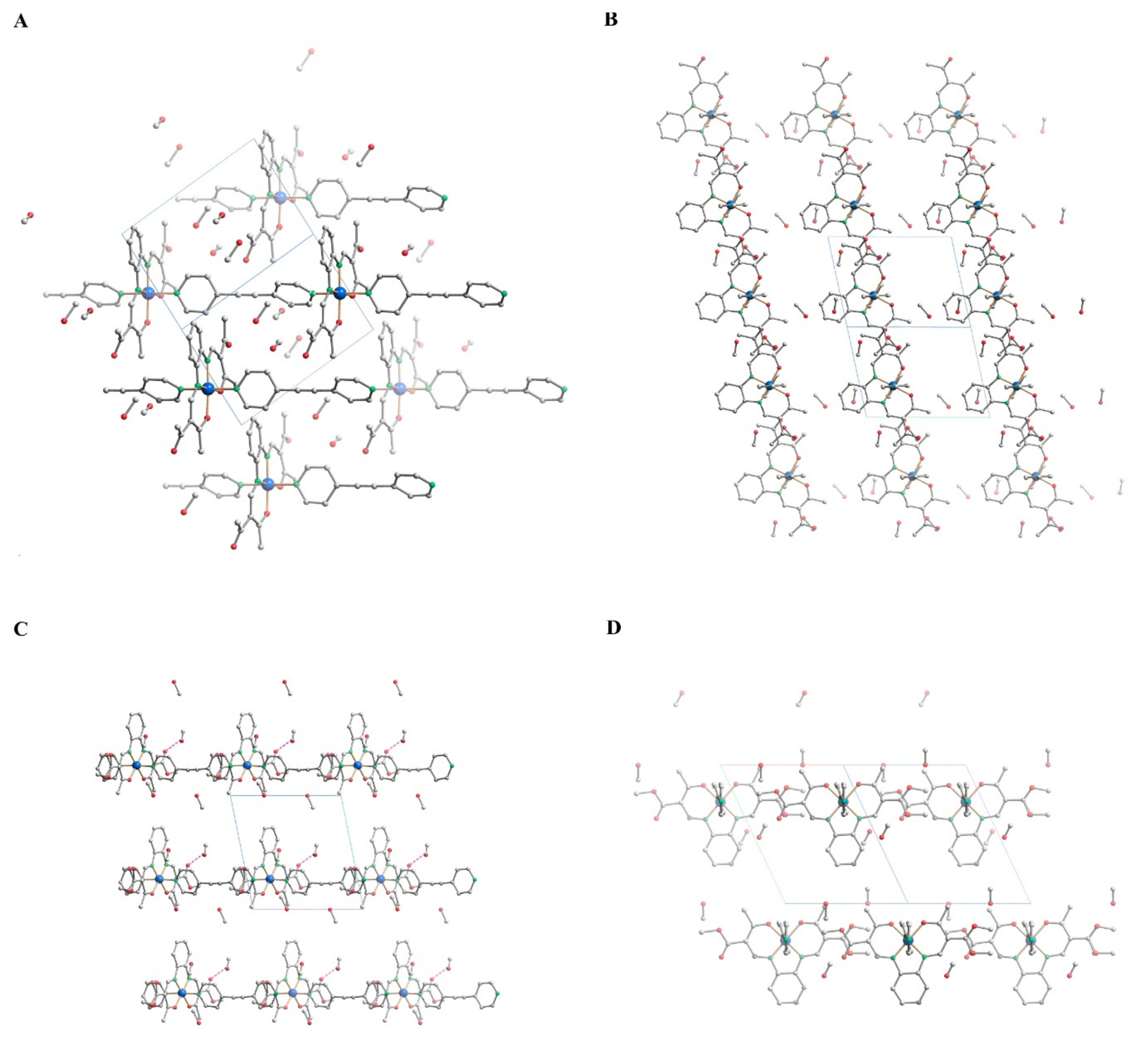
2.2. Crystal Structure Analysis
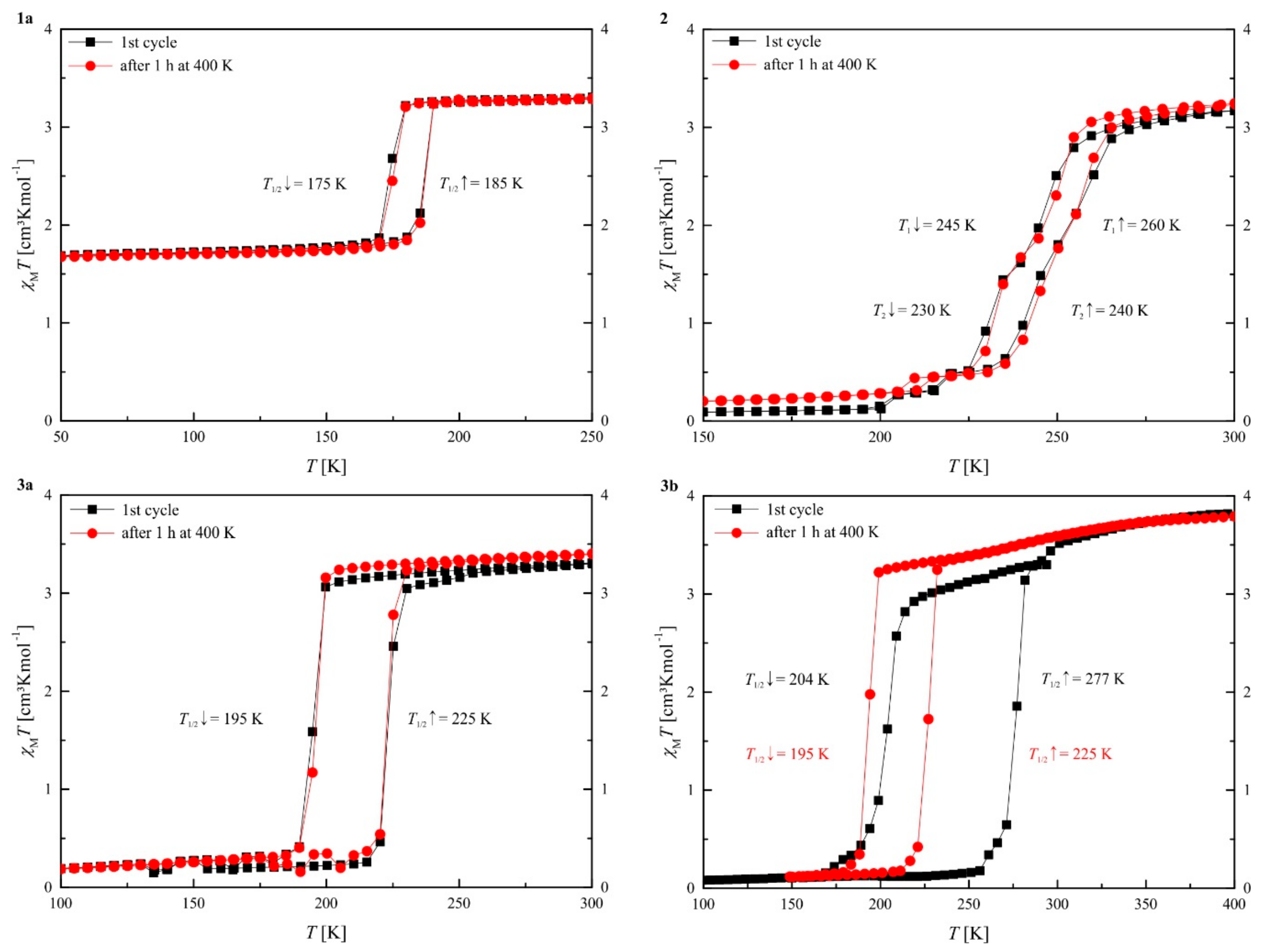
2.3. Magnetism
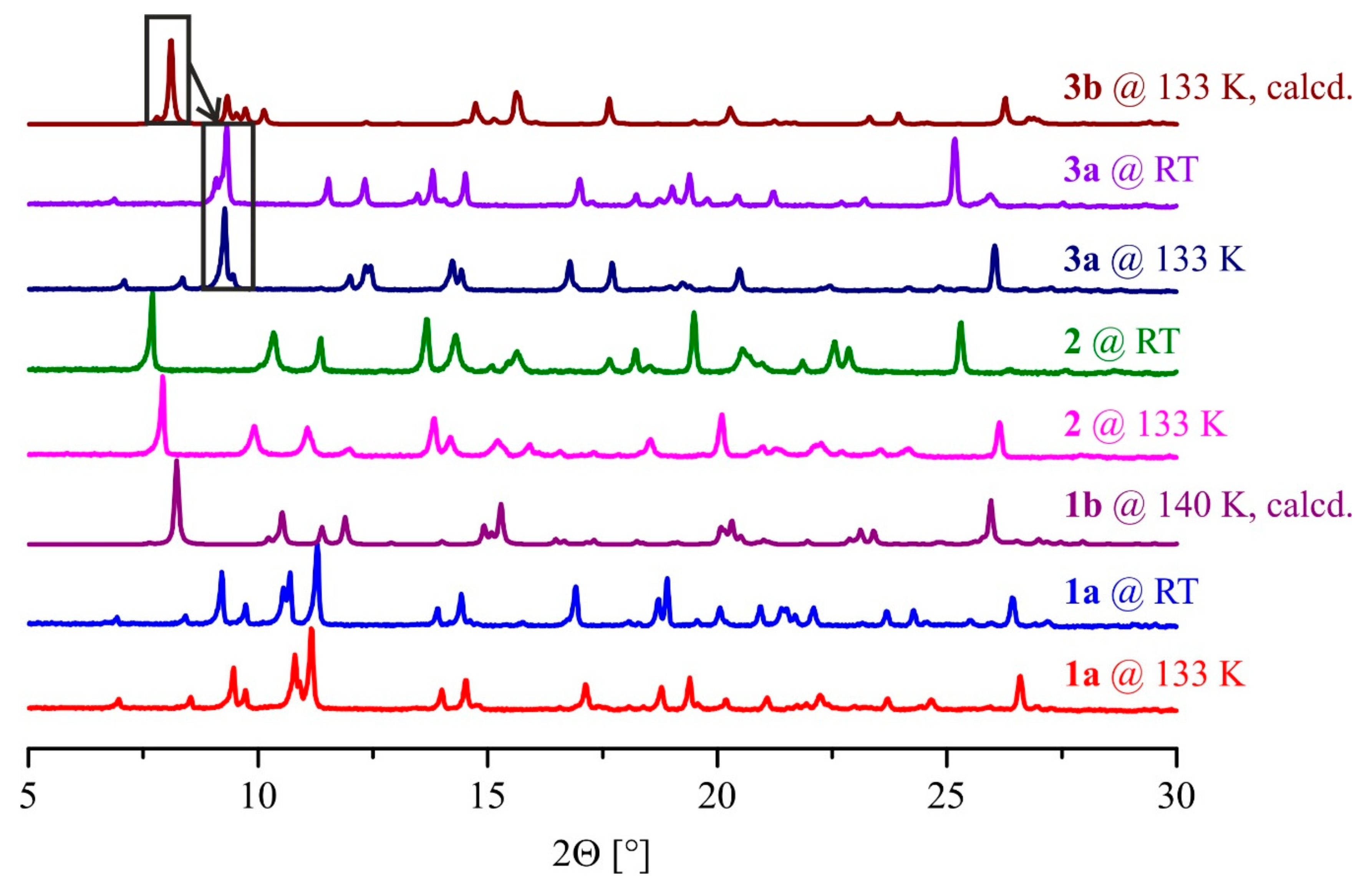
2.4. Powder X-Ray Diffraction Analysis
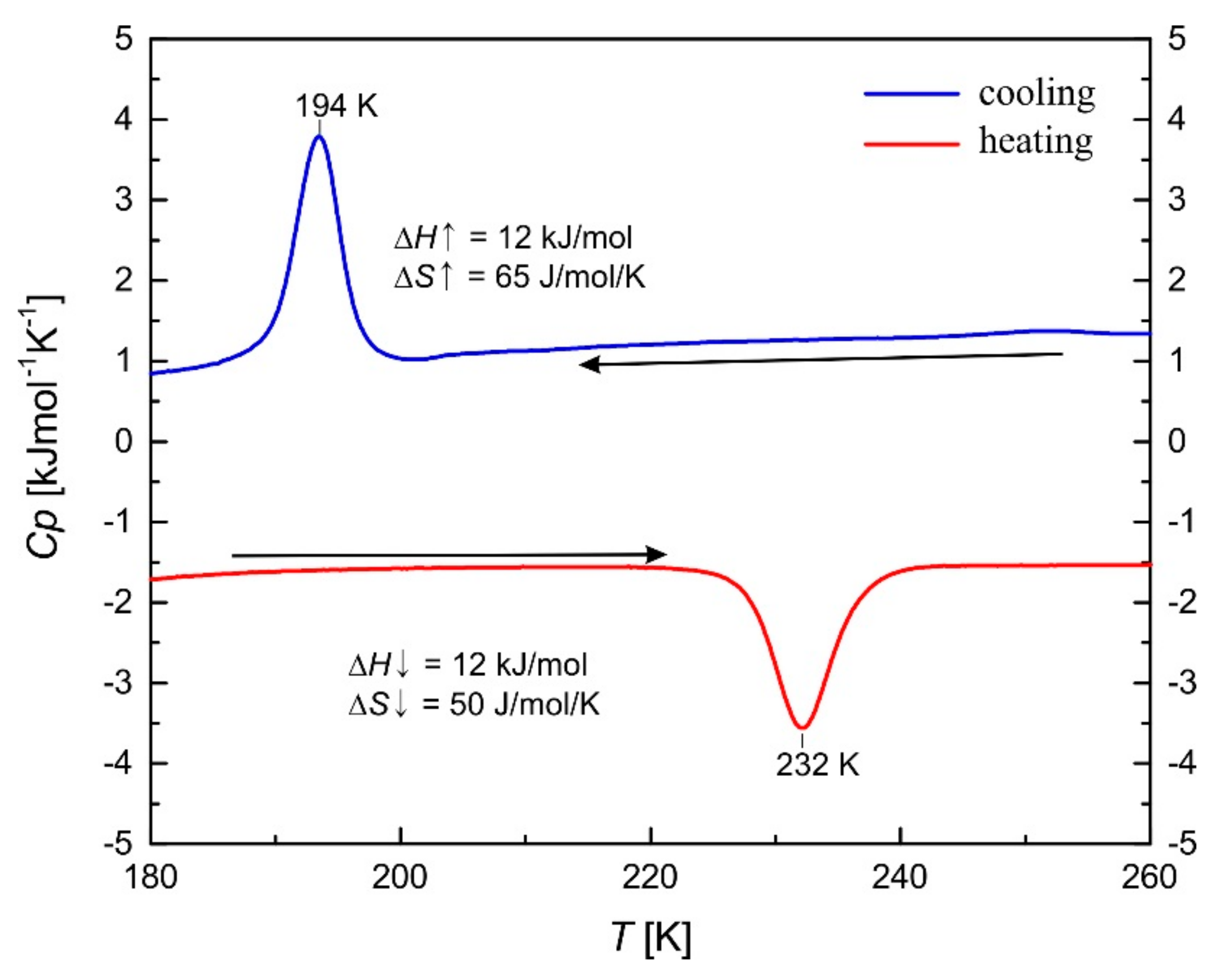
2.5. Thermal Analysis
3. Discussion
4. Materials and Methods
4.1. Experimental
4.2. X-ray Diffraction on Single Crystals
4.3. X-ray Powder Diffraction
4.4. Magnetic Measurements
4.5. Mössbauer Spectrometry
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Coronado, E. Molecular magnetism: From chemical design to spin control in molecules, materials and devices. Nat. Rev. Mater. 2019. [Google Scholar] [CrossRef]
- Boillot, M.-L.; Weber, B. Mononuclear ferrous and ferric complexes. C. R. Chim. 2018, 21, 1196–1208. [Google Scholar] [CrossRef]
- Collet, E.; Guionneau, P. Structural analysis of spin-crossover materials: From molecules to materials. C. R. Chim. 2018, 21, 1133–1151. [Google Scholar] [CrossRef]
- Harding, D.J. An Overview of Spin Crossover Nanoparticles ☆. In Novel Magnetic Nanostructures: Unique Properties and Applications; Rentschler, E., Domracheva, N., Caporali, M., Eds.; Elsevier: Amsterdam, The Netherlands, 2018; pp. 401–426. ISBN 978-0-12-813594-5. [Google Scholar]
- Feltham, H.L.C.; Barltrop, A.S.; Brooker, S. Spin crossover in iron(II) complexes of 3,4,5-tri-substituted-1,2,4-triazole (Rdpt), 3,5-di-substituted-1,2,4-triazolate (dpt −), and related ligands. Coord. Chem. Rev. 2017, 344, 26–53. [Google Scholar] [CrossRef]
- Gaspar, A.B.; Weber, B. Spin Crossover Phenomenon in Coordination Compounds. In Molecular Magnetic Materials; Sieklucka, B., Pinkowicz, D., Eds.; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2017; pp. 231–252. [Google Scholar]
- Senthil Kumar, K.; Ruben, M. Emerging trends in spin crossover (SCO) based functional materials and devices. Coord. Chem. Rev. 2017, 346, 176–205. [Google Scholar] [CrossRef]
- Halcrow, M.A. Spin-Crossover Materials; John Wiley & Sons Ltd.: Chichester, UK, 2013; ISBN 9781118519301. [Google Scholar]
- Gütlich, P.; Goodwin, H.A. (Eds.) Spin Crossover in Transition Metal Compounds I-III; Springer: Berlin/Heidelberg, Germany, 2004; ISBN 3540403949, 3540403965, 9783540403951. [Google Scholar]
- Gütlich, P.; Hauser, A.; Spiering, H. Thermal and Optical Switching of Iron(II) Complexes. Angew. Chem. Int. Ed. 1994, 33, 2024–2054. [Google Scholar] [CrossRef]
- Sato, O.; Tao, J.; Zhang, Y.-Z. Control of Magnetic Properties through External Stimuli. Angew. Chem. Int. Ed. 2007, 46, 2152–2187. [Google Scholar] [CrossRef]
- Nihei, M.; Shiga, T.; Maeda, Y.; Oshio, H. Spin crossover iron(III) complexes: Chemistry of Coordination Space. Coord. Chem. Rev. 2007, 251, 2606–2621. [Google Scholar] [CrossRef]
- Murray, K.S. Advances in Polynuclear Iron(II), Iron(III) and Cobalt(II) Spin-Crossover Compounds. Eur. J. Inorg. Chem. 2008, 2008, 3101–3121. [Google Scholar] [CrossRef]
- Murray, K.S. Recent Advances in Molecular Magnetic Materials. Aust. J. Chem. 2009, 62, 1081–1101. [Google Scholar] [CrossRef]
- Gaspar, A.B.; Seredyuk, M.; Gütlich, P. Spin crossover in iron(II) complexes: Recent advances. J. Mol. Struc. 2009, 924–926, 9–19. [Google Scholar] [CrossRef]
- Brooker, S.; Kitchen, J.A. Nano-magnetic materials: Spin crossover compounds vs. single molecule magnets vs. single chain magnets. Dalton Trans. 2009, 36, 7331–7340. [Google Scholar] [CrossRef] [PubMed]
- Muñoz, M.C.; Real, J.A. Thermo-, piezo-, photo- and chemo-switchable spin crossover iron(II)-metallocyanate based coordination polymers: Special Issue: 39th International Conference on Coordination Chemistry. Coord. Chem. Rev. 2011, 255, 2068–2093. [Google Scholar] [CrossRef]
- Halcrow, M.A. Structure:function relationships in molecular spin-crossover complexes. Chem. Soc. Rev. 2011, 40, 4119–4142. [Google Scholar] [CrossRef] [PubMed]
- Bousseksou, A.; Molnar, G.; Salmon, L.; Nicolazzi, W. Molecular spin crossover phenomenon: Recent achievements and prospects. Chem. Soc. Rev. 2011, 40, 3313–3335. [Google Scholar] [CrossRef]
- Linares, J.; Codjovi, E.; Garcia, Y. Pressure and Temperature Spin Crossover Sensors with Optical Detection. Sensors 2012, 12, 4479–4492. [Google Scholar] [CrossRef]
- Brooker, S. Spin crossover with thermal hysteresis: Practicalities and lessons learnt. Chem. Soc. Rev. 2015, 44, 2880–2892. [Google Scholar] [CrossRef]
- Gütlich, P. Spin Crossover—Quo Vadis? Eur. J. Inorg. Chem. 2013, 2013, 581–591. [Google Scholar] [CrossRef]
- Gütlich, P.; Gaspar, A.B.; Garcia, Y. Spin state switching in iron coordination compounds. Beilstein J. Org. Chem. 2013, 9, 342–391. [Google Scholar] [CrossRef]
- Miller, R.G.; Brooker, S. Reversible quantitative guest sensing via spin crossover of an iron(II) triazole. Chem. Sci. 2016, 7, 2501–2505. [Google Scholar] [CrossRef]
- Lennartson, A.; Southon, P.; Sciortino, N.F.; Kepert, C.J.; Frandsen, C.; Mørup, S.; Piligkos, S.; McKenzie, C.J. Reversible Guest Binding in a Non-Porous Fe(II) Coordination Polymer Host Toggles Spin Crossover. Chem. Eur. J. 2015, 21, 16066–16072. [Google Scholar] [CrossRef] [PubMed]
- Molnár, G.; Salmon, L.; Nicolazzi, W.; Terki, F.; Bousseksou, A. Emerging properties and applications of spin crossover nanomaterials. J. Mater. Chem. C 2014, 2, 1360–1366. [Google Scholar] [CrossRef]
- Kahn, O. Spin-Transition Polymers: From Molecular Materials Toward Memory Devices. Science 1998, 279, 44–48. [Google Scholar] [CrossRef]
- Létard, J.-F.; Guionneau, P.; Goux-Capes, L. Towards Spin Crossover Applications. In Spin Crossover in Transition Metal Compounds I-III; Gütlich, P., Goodwin, H.A., Eds.; Springer: Berlin/Heidelberg, Germany, 2004; pp. 221–249, ISBN 3540403949, 3540403965, 9783540403951. [Google Scholar]
- Galet, A.; Gaspar, A.B.; Muñoz, M.C.; Bukin, G.V.; Levchenko, G.; Real, J.A. Tunable Bistability in a Three-Dimensional Spin-Crossover Sensory- and Memory-Functional Material. Adv. Mater. 2005, 17, 2949–2953. [Google Scholar] [CrossRef]
- Manrique-Juarez, M.D.; Mathieu, F.; Shalabaeva, V.; Cacheux, J.; Rat, S.; Nicu, L.; Leichle, T.; Salmon, L.; Molnar, G.; Bousseksou, A. A Bistable Microelectromechanical System Actuated by Spin-Crossover Molecules. Angew. Chem. Int. Ed. 2017, 56, 8074–8078. [Google Scholar] [CrossRef] [PubMed]
- Niel, V.; Thompson, A.L.; Muñoz, M.C.; Galet, A.; Goeta, A.E.; Real, J.A. Crystalline-State Reaction with Allosteric Effect in Spin-Crossover, Interpenetrated Networks with Magnetic and Optical Bistability. Angew. Chem. Int. Ed. 2003, 42, 3760–3763. [Google Scholar] [CrossRef]
- Dîrtu, M.M.; Neuhausen, C.; Naik, A.D.; Rotaru, A.; Spinu, L.; Garcia, Y. Insights into the Origin of Cooperative Effects in the Spin Transition of [Fe(NH2trz)3](NO3)2. Inorg. Chem. 2010, 49, 5723–5736. [Google Scholar] [CrossRef]
- Weber, B.; Bauer, W.; Obel, J. An Iron(II) Spin-Crossover Complex with a 70 K Wide Thermal Hysteresis Loop. Angew. Chem. Int. Ed. 2008, 47, 10098–10101. [Google Scholar] [CrossRef]
- Weber, B.; Bauer, W.; Pfaffeneder, T.; Dîrtu, M.M.; Naik, A.D.; Rotaru, A.; Garcia, Y. Influence of Hydrogen Bonding on the Hysteresis Width in Iron(II) Spin-Crossover Complexes. Eur. J. Inorg. Chem. 2011, 2011, 3193–3206. [Google Scholar] [CrossRef]
- Bauer, W.; Lochenie, C.; Weber, B. Synthesis and characterization of 1D iron(ii) spin crossover coordination polymers with hysteresis. Dalton Trans. 2014, 43, 1990–1999. [Google Scholar] [CrossRef]
- Halcrow, M.A. Spin-crossover Compounds with Wide Thermal Hysteresis. Chem. Lett. 2014, 43, 1178–1188. [Google Scholar] [CrossRef]
- Kulmaczewski, R.; Olguín, J.; Kitchen, J.A.; Feltham, H.L.C.; Jameson, G.N.L.; Tallon, J.L.; Brooker, S. Remarkable scan rate dependence for a highly constrained dinuclear iron(II) spin crossover complex with a wide thermal hysteresis loop. J. Am. Chem. Soc. 2014, 136, 878–881. [Google Scholar] [CrossRef]
- Seredyuk, M.; Muñoz, M.C.; Castro, M.; Romero-Morcillo, T.; Gaspar, A.B.; Real, J.A. Unprecedented multi-stable spin crossover molecular material with two thermal memory channels. Chem. Eur. J. 2013, 19, 6591–6596. [Google Scholar] [CrossRef] [PubMed]
- Salmon, L.; Catala, L. Spin-crossover nanoparticles and nanocomposite materials. C. R. Chim. 2018, 21, 1230–1269. [Google Scholar] [CrossRef]
- Weber, B. Synthesis of Coordination Polymer Nanoparticles using Self-Assembled Block Copolymers as Template. Chem. Eur. J. 2017, 23, 18093–18100. [Google Scholar] [CrossRef] [PubMed]
- Delgado, T.; Enachescu, C.; Tissot, A.; Hauser, A.; Guénée, L.; Besnard, C. Evidencing size-dependent cooperative effects on spin crossover nanoparticles following their HS→LS relaxation. J. Mater. Chem. C 2018, 6, 12698–12706. [Google Scholar] [CrossRef]
- Bertoni, R.; Lorenc, M.; Cailleau, H.; Tissot, A.; Laisney, J.; Boillot, M.-L.; Stoleriu, L.; Stancu, A.; Enachescu, C.; Collet, E. Elastically driven cooperative response of a molecular material impacted by a laser pulse. Nat. Mater. 2016, 15, 606–610. [Google Scholar] [CrossRef]
- Göbel, C.; Hils, C.; Drechsler, M.; Baabe, D.; Greiner, A.; Schmalz, H.; Weber, B. Confined crystallization of spin crossover nanoparticles in block copolymer micelles. Angew. Chem. Int. Ed. 2020, in press. [Google Scholar] [CrossRef]
- Quintero, C.M.; Félix, G.; Suleimanov, I.; Sánchez Costa, J.; Molnár, G.; Salmon, L.; Nicolazzi, W.; Bousseksou, A. Hybrid spin-crossover nanostructures. Beilstein J. Nanotechnol. 2014, 5, 2230–2239. [Google Scholar] [CrossRef]
- Peng, H.; Tricard, S.; Félix, G.; Molnár, G.; Nicolazzi, W.; Salmon, L.; Bousseksou, A. Re-appearance of cooperativity in ultra-small spin-crossover Fe(pz){Ni(CN)4} nanoparticles. Angew. Chem. Int. Ed. 2014, 53, 10894–10898. [Google Scholar] [CrossRef]
- Lochenie, C.; Schötz, K.; Panzer, F.; Kurz, H.; Maier, B.; Puchtler, F.; Agarwal, S.; Köhler, A.; Weber, B. Spin-Crossover Iron(II) Coordination Polymer with Fluorescent Properties: Correlation between Emission Properties and Spin State. J. Am. Chem. Soc. 2018, 140, 700–709. [Google Scholar] [CrossRef] [PubMed]
- Schlamp, S.; Weber, B.; Naik, A.D.; Garcia, Y. Cooperative spin transition in a lipid layer like system. Chem. Commun. 2011, 47, 7152–7154. [Google Scholar] [CrossRef] [PubMed]
- Luo, Y.-H.; Liu, Q.-L.; Yang, L.-J.; Sun, Y.; Wang, J.-W.; You, C.-Q.; Sun, B.-W. Magnetic observation of above room-temperature spin transition in vesicular nano-spheres. J. Mater. Chem. C 2016, 4, 8061–8069. [Google Scholar] [CrossRef]
- Romero-Morcillo, T.; Seredyuk, M.; Munoz, M.C.; Real, J.A. Meltable Spin Transition Molecular Materials with Tunable Tc and Hysteresis Loop Width. Angew. Chem. Int. Ed. 2015, 54, 14777–14781. [Google Scholar] [CrossRef] [PubMed]
- Ako, A.M.; Alam, M.S.; Rahman, M.; Hill, J.P.; Sanchez-Ballester, N.M.; Ariga, K.; Buth, G.; Anson, C.E.; Powell, A.K. Self-Assembly of a Mononuclear [Fe III (L)(EtOH) 2 ] Complex Bearing an n -Dodecyl Chain on Solid Highly Oriented Pyrolytic Graphite Surfaces. Chem. Eur. J. 2012, 18, 16419–16425. [Google Scholar] [CrossRef] [PubMed]
- Coronado, E.; Giménez-Marqués, M.; Mínguez Espallargas, G.; Rey, F.; Vitórica-Yrezábal, I.J. Spin-crossover modification through selective CO2 sorption. J. Am. Chem. Soc. 2013, 135, 15986–15989. [Google Scholar] [CrossRef] [PubMed]
- Halder, G.J. Guest-Dependent Spin Crossover in a Nanoporous Molecular Framework Material. Science 2002, 298, 1762–1765. [Google Scholar] [CrossRef]
- Lochenie, C.; Bauer, W.; Railliet, A.P.; Schlamp, S.; Garcia, Y.; Weber, B. Large Thermal Hysteresis for Iron(II) Spin Crossover Complexes with N-(Pyrid-4-yl)isonicotinamide. Inorg. Chem. 2014, 53, 11563–11572. [Google Scholar] [CrossRef]
- Weber, B.; Tandon, R.; Himsl, D. Synthesis, Magnetic Properties and X-ray Structure Analysis of a 1-D Chain Iron(II) Spin Crossover Complex with wide Hysteresis. Z. Anorg. Allg. Chem. 2007, 633, 1159–1162. [Google Scholar] [CrossRef]
- Weber, B.; Kaps, E.S.; Desplanches, C.; Létard, J.-F. Quenching the Hysteresis in Single Crystals of a 1D Chain Iron(II) Spin Crossover Complex. Eur. J. Inorg. Chem. 2008, 2008, 2963–2966. [Google Scholar] [CrossRef]
- Nowak, R.; Bauer, W.; Ossiander, T.; Weber, B. Slow Self-Assembly Favors Hysteresis above Room Temperature for an Iron(II) 1D-Chain Spin-Crossover Complex. Eur. J. Inorg. Chem. 2013, 2013, 975–983. [Google Scholar] [CrossRef]
- Pfaffeneder, T.M.; Thallmair, S.; Bauer, W.; Weber, B. Complete and incomplete spin transitions in 1D chain iron(ii) compounds. New J. Chem. 2011, 35, 691–700. [Google Scholar] [CrossRef]
- Bauer, W.; Scherer, W.; Altmannshofer, S.; Weber, B. Two-Step versus One-Step Spin Transitions in Iron(II) 1D Chain Compounds. Eur. J. Inorg. Chem. 2011, 2803–2818. [Google Scholar] [CrossRef]
- Bauer, W.; Pfaffeneder, T.; Achterhold, K.; Weber, B. Complete Two-Step Spin-Transition in a 1D Chain Iron(II) Complex with a 110-K Wide Intermediate Plateau. Eur. J. Inorg. Chem. 2011, 3183–3192. [Google Scholar] [CrossRef]
- Schönfeld, S.; Lochenie, C.; Thoma, P.; Weber, B. 1D iron(ii) spin crossover coordination polymers with 3,3′-azopyridine – kinetic trapping effects and spin transition above room temperature. CrystEngComm 2015, 17, 5389–5395. [Google Scholar] [CrossRef]
- Bauer, W.; Ossiander, T.; Weber, B. Synthesis of iron(II) complexes with asymmetric N2O2 coordinating Schiff base-like ligands and their spin crossover properties. Front. Chem. Sci. Eng. 2018, 12, 400–408. [Google Scholar] [CrossRef]
- Jäger, E.-G.; Häussler, E.; Rudolph, M.; Schneider, A. Struktur-Reaktivitäts-Beziehungen bei koordinativ-ungesättigten Chelatkomplexen. VI. Synthese, Adduktbildung, Redoxpotentiale und photochrome lodderivate von Eisen(II)-Komplexen Schiffscher Basen mit elektronenziehenden Substituenten. Z. Anorg. Allg. Chem. 1985, 525, 67–85. [Google Scholar] [CrossRef]
- Bauer, W.; Ossiander, T.; Weber, B. A Promising New Schiff Base-like Ligand for the Synthesis of Octahedral Iron(II) Spin Crossover Complexes. Z. Naturforsch. B 2010, 2010, 323–328. [Google Scholar] [CrossRef]
- Tanner, M.; Ludi, A. A facile synthesis of 4,4′-dipyridylacetylene. Chimia 1980, 34, 23–24. [Google Scholar]
- Dankhoff, K.; Lochenie, C.; Puchtler, F.; Weber, B. Solvent Influence on the Magnetic Properties of Iron(II) Spin-Crossover Coordination Compounds with 4,4′-Dipyridylethyne as Linker. Eur. J. Inorg. Chem. 2016, 2016, 2136–2143. [Google Scholar] [CrossRef]
- Bauer, W.; Schlamp, S.; Weber, B. A ladder type iron(ii) coordination polymer with cooperative spin transition. Chem. Commun. 2012, 48, 10222. [Google Scholar] [CrossRef] [PubMed]
- Schlamp, S.; Thoma, P.; Weber, B. Influence of the Alkyl Chain Length on the Self-Assembly of Amphiphilic Iron Complexes: An Analysis of X-ray Structures. Chem. Eur. J. 2014, 20, 6462–6473. [Google Scholar] [CrossRef] [PubMed]
- Zappe, L.; Lochenie, C.; Martin, T.; Weber, B. Iron(II) Spin Crossover Polymers of Planar N2O2 Schiff Base Templates and 4,4′-bis(pyridyl)urea Bridges. Chem 2019, 6, 10–18. [Google Scholar] [CrossRef]
- Weihermüller, J.; Schlamp, S.; Milius, W.; Puchtler, F.; Breu, J.; Ramming, P.; Hüttner, S.; Agarwal, S.; Göbel, C.; Hund, M.; et al. Amphiphilic iron(II) spin crossover coordination polymers: Crystal structures and phase transition properties. J. Mater. Chem. C 2019, 7, 1151–1163. [Google Scholar] [CrossRef]
- Weber, B. Spin crossover complexes with N4O2 coordination sphere—The influence of covalent linkers on cooperative interactions. Coord. Chem. Rev. 2009, 253, 2432–2449. [Google Scholar] [CrossRef]
- Weber, B.; Jäger, E.-G. Structure and Magnetic Properties of Iron(II/III) Complexes with N2O22− Coordinating Schiff Base Like Ligands. Eur. J. Inorg. Chem. 2009, 2009, 465–477. [Google Scholar] [CrossRef]
- Miller, R.G.; Narayanaswamy, S.; Tallon, J.L.; Brooker, S. Spin crossover with thermal hysteresis in cobalt(ii) complexes and the importance of scan rate. New J. Chem. 2014, 38, 1932. [Google Scholar] [CrossRef]
- Weihermüller, J.; Schlamp, S.; Dittrich, B.; Weber, B. Kinetic Trapping Effects in Amphiphilic Iron(II) Spin Crossover Compounds. Inorg. Chem. 2019, 58, 1278–1289. [Google Scholar] [CrossRef]
- Bauer, W.; Dîrtu, M.M.; Garcia, Y.; Weber, B. Iron(II) spin transition coordination polymers with a zigzag structure. CrystEngComm 2012, 14, 1223. [Google Scholar] [CrossRef]
- Altomare, A.; Burla, M.C.; Camalli, M.; Cascarano, G.L.; Giacovazzo, C.; Guagliardi, A.; Moliterni, A.G.G.; Polidori, G.; Spagna, R. SIR97: A new tool for crystal structure determination and refinement. J. Appl. Cryst. 1999, 32, 115–119. [Google Scholar] [CrossRef]
- Sheldrick, G. A short history of SHELX. Acta Cryst. A 2008, 64, 112–122. [Google Scholar] [CrossRef] [PubMed]
- Sheldrick, G.M. Crystal structure refinement with SHELXL. Acta Cryst. C 2015, 71, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Johnson, C.K.; Burnett, M.N. ORTEP-III; Oak-Ridge National Laboratory: Oak-Ridge, TN, USA, 1996. [Google Scholar]
- Farrugia, L. ORTEP-3 for Windows—A version of ORTEP-III with a Graphical User Interface (GUI). J. Appl. Cryst. 1997, 30, 565. [Google Scholar] [CrossRef]
- Keller, E. Schakal-99; University of Freiburg: Freiburg, Germany, 1999. [Google Scholar]
- Lagarec, K.; Rancourt, D.G. Recoil, Mössbauer Spectral Analysis Software for Windows 1.0; Department of Physics, University of Ottawa: Ottawa, ON, Canada, 1998. [Google Scholar]
Sample Availability: Samples of the compounds are not available. |
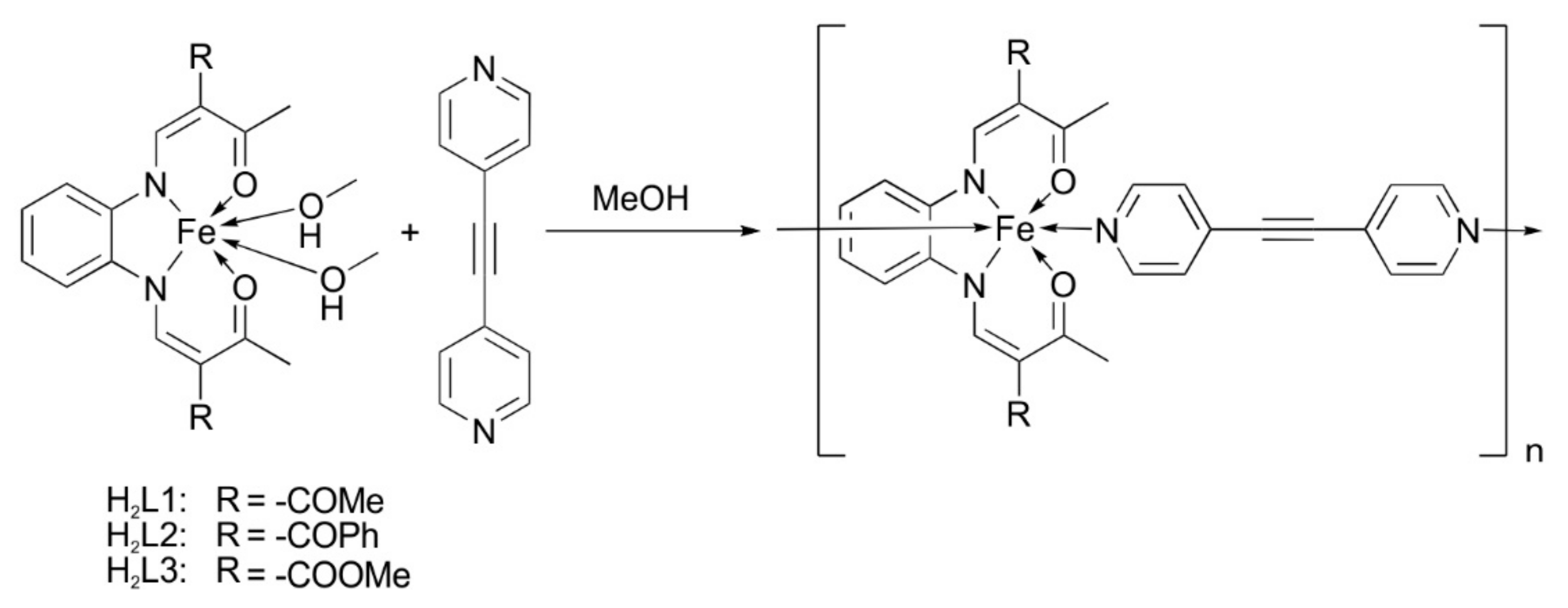
| Compound | Complex | Method |
|---|---|---|
| 1a | [FeL1(bpey)]n | reflux |
| 1b | {[FeL1(bpey)]·2 MeOH}n | slow diffusion |
| 2 | {[FeL2(bpey)]·1 MeOH}n | reflux |
| 3a | {[FeL3(bpey)]·0.25 MeOH}n | reflux |
| 3b | {[FeL3(bpey)]·2 MeOH}n | slow diffusion |
| Compound | S | Fe–Neq | Fe–Oeq | Fe–Nax | Oeq–Fe–Oeq | Nax–Fe–Nax | bpey >C–C≡C | Torsion bpey |
|---|---|---|---|---|---|---|---|---|
| 3b | 0 | 1.898(4) 1.8921(4) | 1.931(3) 1.931(3) | 1.985(3) 1.997(3) | 87.08(11) | 175.18(14) | 174.2(4) 176.9(4) | 17.81 18.44 |
| Compound | D–H⋯A | D–H/Å | H⋯A/Å | D⋯A/Å | D–H⋯A/° |
|---|---|---|---|---|---|
| 3b | O52–H52⋯O3 | 0.84 | 2.33 | 2.84(2) | 119 |
| C26–H26⋯O3 a | 0.95 | 2.50 | 3.150(5) | 126 |
| SCO | T1/2 [K] | χMT (HS) [cm3Kmol−1] | χMT (LS) [cm3Kmol−1] | γHS | |
|---|---|---|---|---|---|
| 1a | Incomplete with hysteresis | ↓ 175 ↑ 185 | 3.29 (250 K) | 1.69 (50 K) | 1 (250 K) 0.51 (50 K) |
| 2 | Stepwise, gradual with hysteresis | ↓ 245 ↑ 260 ↓ 230 ↑ 240 | 3.17 (300 K) | 0.09 (150 K) | 1 (300 K) 0.03 (150 K) |
| Stepwise, gradual with hysteresis a | ↓ 245 ↑ 260 ↓ 230 ↑ 240 | 3.23 (300 K) | 0.20 (150 K) | 1 (300 K) 0.06 (150 K) | |
| 3a | Abrupt with hysteresis | ↓ 195 ↑ 225 | 3.31 (300 K) | 0.19 (100 K) | 1 (300 K) 0.06 (100 K) |
| 3b | Abrupt with hysteresis | ↓ 204 ↑ 277 | 3.79 (400 K) | 0.08 (100 K) | 1 (400 K) 0.02 (100 K) |
| Abrupt with hysteresis a | ↓ 195 ↑ 225 | 3.79 (400 K) | 0.12 (150 K) | 1 (400 K) 0.03 (150 K) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dankhoff, K.; Lochenie, C.; Weber, B. Iron(II) Spin Crossover Complexes with 4,4′-Dipyridylethyne—Crystal Structures and Spin Crossover with Hysteresis. Molecules 2020, 25, 581. https://doi.org/10.3390/molecules25030581
Dankhoff K, Lochenie C, Weber B. Iron(II) Spin Crossover Complexes with 4,4′-Dipyridylethyne—Crystal Structures and Spin Crossover with Hysteresis. Molecules. 2020; 25(3):581. https://doi.org/10.3390/molecules25030581
Chicago/Turabian StyleDankhoff, Katja, Charles Lochenie, and Birgit Weber. 2020. "Iron(II) Spin Crossover Complexes with 4,4′-Dipyridylethyne—Crystal Structures and Spin Crossover with Hysteresis" Molecules 25, no. 3: 581. https://doi.org/10.3390/molecules25030581
APA StyleDankhoff, K., Lochenie, C., & Weber, B. (2020). Iron(II) Spin Crossover Complexes with 4,4′-Dipyridylethyne—Crystal Structures and Spin Crossover with Hysteresis. Molecules, 25(3), 581. https://doi.org/10.3390/molecules25030581







