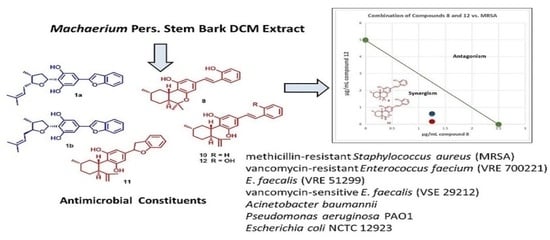
Antimicrobial Constituents from Machaerium Pers.: Inhibitory Activities and Synergism of Machaeriols and Machaeridiols against Methicillin-Resistant Staphylococcus aureus, Vancomycin-Resistant Enterococcus faecium, and Permeabilized Gram-Negative Pathogens
Abstract
1. Introduction
2. Results and Discussion
2.1. Botanical Identity of Machaerium sp. (Rimachi 12161)
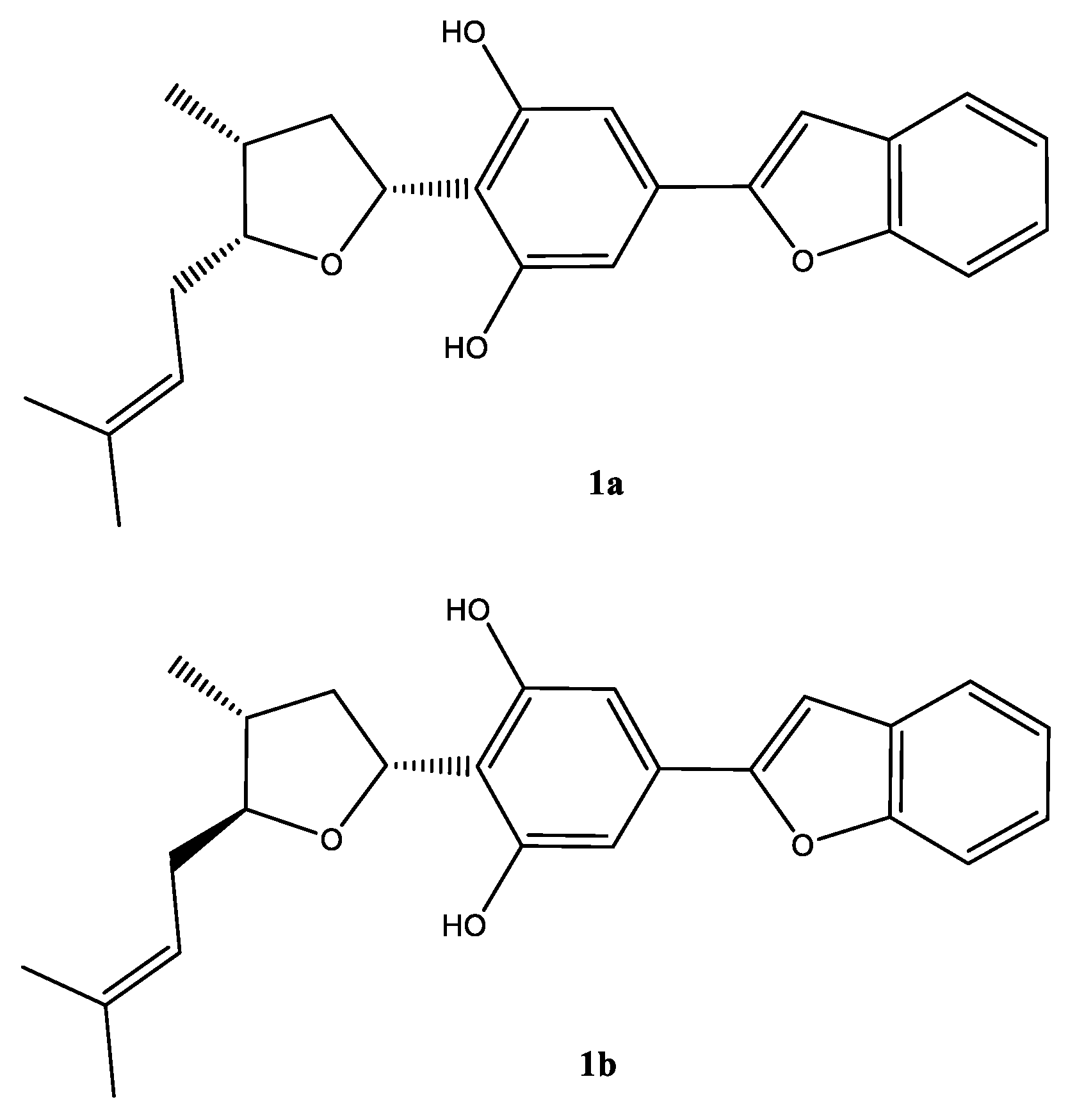
2.2. Phytochemical Constituents
2.3. Antimicrobial Activity against Gram-Positive Species and Fungi
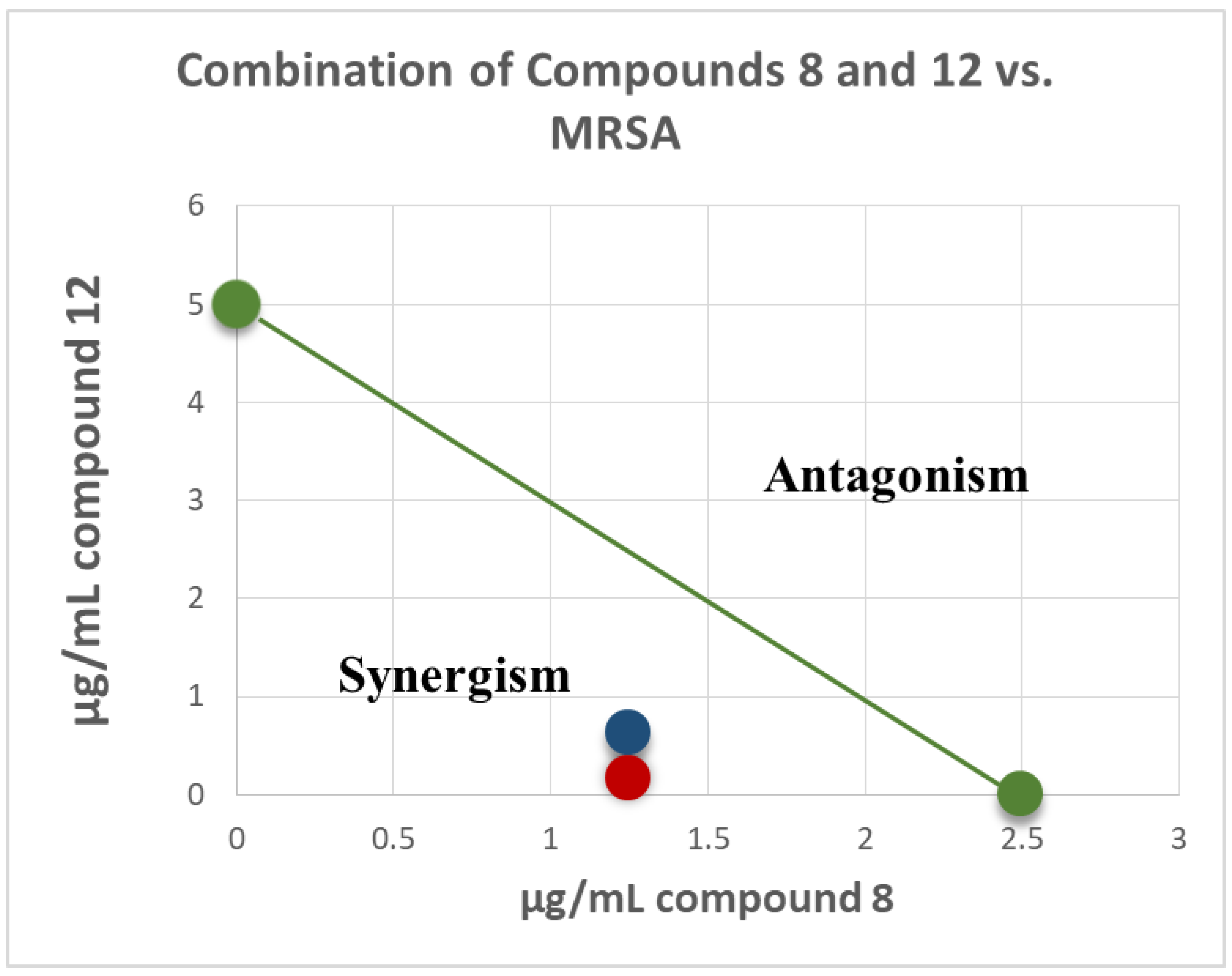
2.4. Antimicrobial Combination Studies
2.5. Antimicrobial Activity against Gram-Negative Species
3. Materials and Methods
3.1. General Experimental Procedures
3.2. Plant Material
3.3. Extraction and Isolation of Compounds from Stem Bark and Leaves
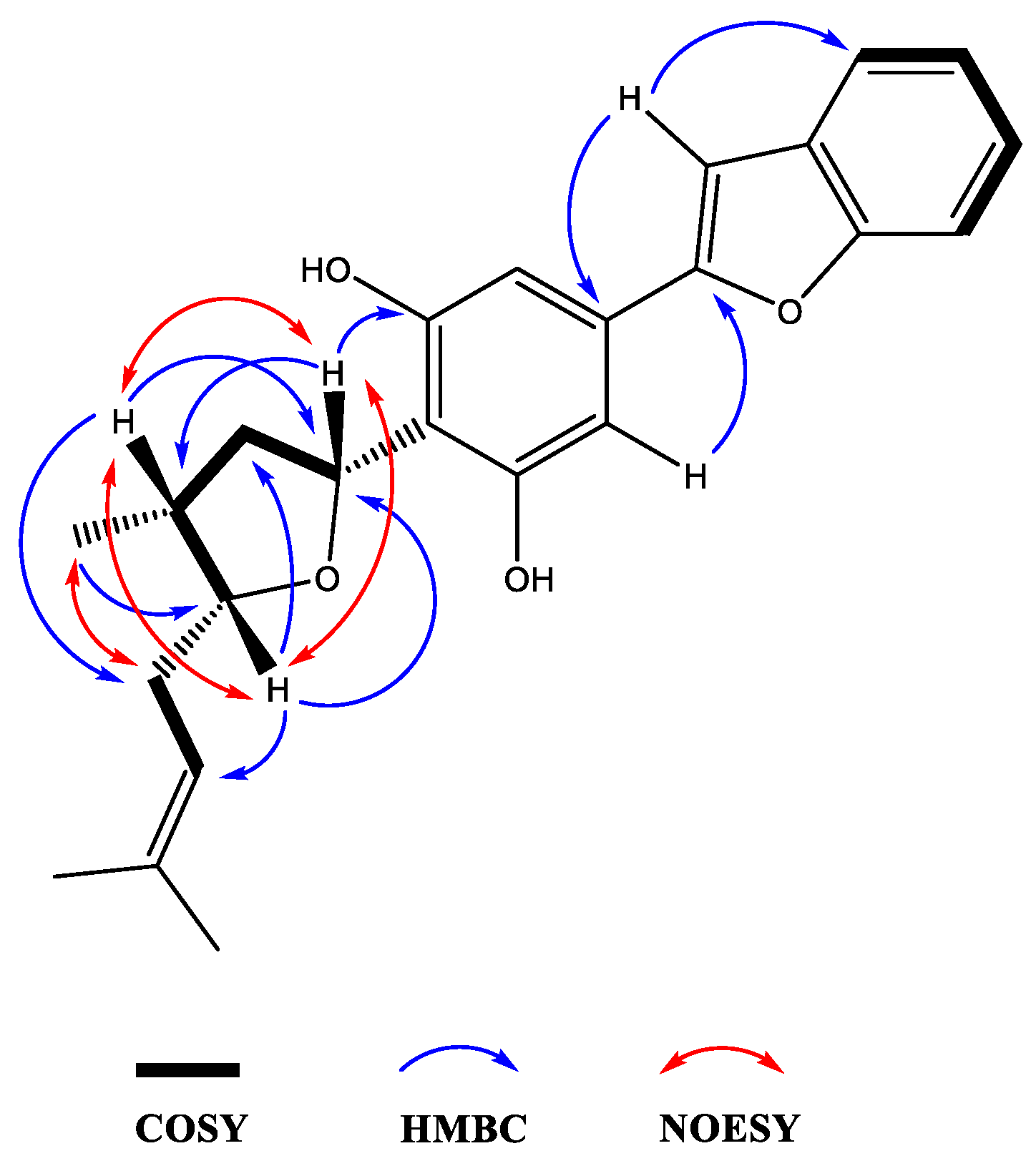
3.4. Machaerifurogerol (1a) and 5-epi-Machaerifurogerol (1b)
3.5. Identification of Compounds 6–8 and 10–12 by LC–MS
3.6. Antimicrobial Assays
3.7. Antimicrobial Combination Study by Checkerboard Method
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Mabberley, D.J. Mabberley’s Plant-Book: A Portable Dictionary of Plants, Their Classification and Uses; Cambridge University Press: Cambridge, UK, 2017. [Google Scholar]
- Amen, Y.M.; Marzouk, A.M.; Zaghloul, M.G.; Afifi, M.S. The genus Machaerium (Fabaceae): Taxonomy, phytochemistry, traditional uses and biological activities. Natl. Prod. Res. 2015, 29, 1388–1405. [Google Scholar] [CrossRef] [PubMed]
- Joly, L.G.; Guerra, S.; Septimo, R.; Solis, P.N.; Correa, M.; Gupta, M.; Levy, S.; Sandberg, F. Ethnobotanical inventory of medicinal plants used by the Guaymi Indians in Western Panama. Part I. J. Ethnopharmacol. 1987, 20, 145–171. [Google Scholar] [CrossRef]
- Polhill, R.M.; Raven, P.H. Advances in Legume Systematics, Part 1; Royal Botanic Gardens: Kew, UK, 1981. [Google Scholar]
- Muhammad, I.; Li, X.-C.; Dunbar, D.C.; ElSohly, M.A.; Khan, I.A. Antimalarial (+)-trans-hexahydrodibenzopyran derivatives from Machaerium multiflorum. J. Nat. Prod. 2001, 64, 1322–1325. [Google Scholar] [CrossRef] [PubMed]
- Muhammad, I.; Li, X.-C.; Jacob, M.R.; Tekwani, B.L.; Dunbar, D.C.; Ferreira, D. Antimicrobial and Antiparasitic (+)-trans-Hexahydrodibenzopyrans and Analogues from Machaerium multiflorum. J. Nat. Prod. 2003, 66, 804–809. [Google Scholar] [CrossRef] [PubMed]
- Filardi, F.L.R.; Lima, H.C.D. The diversity of Machaerium (Leguminosae: Papilionoideae) in the Atlantic Forest: Three new species, nomenclatural updates, and a revised key. Syst. Bot. 2014, 39, 145–159. [Google Scholar] [CrossRef]
- David, M.Z.; Daum, R.S. Community-associated methicillin-resistant Staphylococcus aureus: Epidemiology and clinical consequences of an emerging epidemic. Clin. Microbiol. Rev. 2010, 23, 616–687. [Google Scholar] [CrossRef]
- Salgado, C.D. The risk of developing a vancomycin-resistant Enterococcus bloodstream infection for colonized patients. Am. J. Infect. Control 2008, 36, S175. [Google Scholar] [CrossRef]
- Lozano, P.; Klitgaard, B.B. The genus Machaerium (Leguminosae: Papilionoideae: Dalbergieae) in Ecuador. Brittonia 2006, 58, 124–150. [Google Scholar] [CrossRef]
- Piccinelli, A.L.; Campo Fernandez, M.; Cuesta-Rubio, O.; Márquez Hernández, I.; De Simone, F.; Rastrelli, L. Isoflavonoids isolated from Cuban propolis. J. Agric. Food Chem. 2005, 53, 9010–9016. [Google Scholar] [CrossRef] [PubMed]
- Kurosawa, K.; Ollis, W.D.; Redman, B.T.; Sutherland, I.O.; Gottlieb, O.R. Vestitol and vesticarpan, isoflavonoids from Machaerium vestitum. Phytochemistry 1978, 17, 1413–1415. [Google Scholar] [CrossRef]
- Norden, C.W.; Wentzel, H.; Keleti, E. Comparison of techniques for measurement of in vitro antibiotic synergism. J. Infect. Dis. 1979, 140, 629–633. [Google Scholar] [CrossRef] [PubMed]
- Renneberg, J. Definitions of antibacterial interactions in animal infection models. J. Antimicrob. Chemother. 1993, 31 (Suppl. D), 167–175. [Google Scholar] [CrossRef] [PubMed]
- Ilias, M.; Samoylenko, V.; Gillium, V.D. Preparation of Pre-Coated Rp-Rotors and Universal Chromatorotors, Chromatographic Separation Devices and Methods for Centrifugal Preparative Chromatography. Google Patents US20140224740A1, 14 August 2014. [Google Scholar]
- Wikler, M.A. Methods for dilution antimicrobial susceptibility tests for bacteria that grow aerobically: Approved standard. CLSI (NCCLS) 2006, 26, M7–A7. [Google Scholar]
- Michael, A.; Vishnu, C.; Ana, E.; Mahmoud, A.; Linda, L.; Frank, C. NCCLS, Reference Method for Broth Dilution Antifungal Susceptibility Testing of Yeasts; Approved Standard, M27-A2; CLSI: Wayne, PA, USA, 2002. [Google Scholar]
- NCCLS. Susceptibility Testing of Mycobacteria, Nocardiae, and Other Aerobic Actinomycetes; Approved Standard; NCCLS: Wayne, PA, USA, 2003. [Google Scholar]
- Bock, L.; Hind, C.; Sutton, J.; Wand, M. Growth media and assay plate material can impact on the effectiveness of cationic biocides and antibiotics against different bacterial species. Lett. Appl. Microbiol. 2018, 66, 368–377. [Google Scholar] [CrossRef] [PubMed]
- Toyota, M.; Shimamura, T.; Ishii, H.; Renner, M.; Braggins, J.; Asakawa, Y. New bibenzyl cannabinoid from the New Zealand liverwort Radula marginata. Chem. Pharm. Bull. 2002, 50, 1390–1392. [Google Scholar] [CrossRef] [PubMed]
- Chittiboyina, A.G.; Reddy, C.R.; Watkins, E.B.; Avery, M.A. First synthesis of antimalarial Machaeriols A and B. Tetrahedron Lett. 2004, 45, 1689–1691. [Google Scholar] [CrossRef]
- Huang, Q.; Wang, Q.; Zheng, J.; Zhang, J.; Pan, X.; She, X. A general route to 5, 6-seco-hexahydrodibenzopyrans and analogues: First total synthesis of (+)-Machaeridiol B and (+)-Machaeriol B. Tetrahedron 2007, 63, 1014–1021. [Google Scholar] [CrossRef]
- Wang, Q.; Huang, Q.; Chen, B.; Lu, J.; Wang, H.; She, X.; Pan, X. Total Synthesis of (+)-Machaeriol D with a Key Regio-and Stereoselective SN2′ Reaction. Angew. Chem. Int. Ed. 2006, 45, 3651–3653. [Google Scholar] [CrossRef] [PubMed]
- Xia, L.; Lee, Y.R. A short total synthesis for biologically interesting (+)-and (−)-machaeriol A. Synlett 2008, 2008, 1643–1646. [Google Scholar]
- Thapa, D.; Lee, J.S.; Heo, S.W.; Lee, Y.R.; Kang, K.W.; Kwak, M.K.; Choi, H.G.; Kim, J.A. Novel hexahydrocannabinol analogs as potential anti-cancer agents inhibit cell proliferation and tumor angiogenesis. Eur. J. Pharmacol. 2011, 650, 64–71. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds 1–8 and 10–12 are available from the authors. |

| Position | 1a (Major Epimer) | 1b (Minor Epimer) | HMBC | ||
|---|---|---|---|---|---|
| δc a (J in Hz) | δHb (J in Hz) | δc a (J in Hz) | δHb (J in Hz) | ||
| 1 | 35.2 CH | 2.4 (m) | 35.4 CH | 2.4 (m) | 5, 2, 11 |
| 2 | 41.8 CH2 | 2.72 (m), 1.59 (m) | 42.0 CH2 | 2.39 (m), 1.58 (m) | 5, 11 |
| 3 | 77.7 CH | 5.32 (dd, 12, 7.6) | 77.4 CH | 5.59 (dd, 12, 8.0) | 2, 1’, 2’, 6’ |
| 5 | 83.1 CH | 4.00 (m) | 83.3 CH | 4.21 (m) | 11, 7 |
| 6 | 29.9 CH2 | 1.24 (br s), 2.4 (m) | 29.4 CH2 | 1.24 (br s), 2.4 (m) | 7, 8, 5 |
| 7 | 120.0 CH | 5.18 (t, 7.2) | 120.0 CH | 5.18 (t, 7.2) | 9, 10 |
| 8 | 134.5 C | 134.4 CH | |||
| 9 | 18.2 CH3 | 1.66 (s) | 18.3 CH3 | 1.66 (s) | 7, 8, 10 |
| 10 | 26.1 CH3 | 1.72 (s) | 26.1 CH3 | 1.72 (s) | 7, 8, 9 |
| 11 | 15.5 CH3 | 1.02 (d, 7.2) | 13.9 CH3 | 1.03 (d, 7.2) | 1, 2, 5 |
| 1’ | 113.3 C | 114.0 C | |||
| 2’ | 155.3 C | 155.3 C | |||
| 3’ | 104.9 CH | 6.83 (br s) | 104.9 CH | 6.83 (br s) | 5’, 4’, 1’ |
| 4’ | 130.6 C | 130.8 C | |||
| 5’ | 104.9 CH | 6.83 (br s) | 104.9 CH | 6.83 (br s) | 3’, 4’, 1’, 7’ |
| 6’ | 155.4 C | 155.4 C | |||
| 7’ | 154.9 C | 154.9 C | |||
| 8’ | 101.7 CH | 6.89 (br s) | 101.6 CH | 6.89 (br s) | 4’, 7’ |
| 9’ | 129.5 C | 129.4 C | |||
| 10’ | 155.5 C | 155.5 C | |||
| 11’ | 111.3 CH | 7.45 (d, 7.6) | 111.3 CH | 7.45 (d, 7.6) | 12’, 13’, 10’ |
| 12’ | 124.4 CH | 7.22 (m) | 124.4 CH | 7.22 (m) | 13’, 11’, 10’ |
| 13’ | 123.1 CH | 7.18 (m) | 123.1 CH | 7.18 (m) | 11’, 9’ |
| 14’ | 121.1 CH | 7.52 (d, 7.2) | 121.1 CH | 7.52 (d, 7.2) | 8’, 10’, 13’ |
| C. glabrata | C. krusei | C. neoformans | S. aureus | MRSA | VRE 29212 b | VRE 51299 c | VRE 700221 d | |
|---|---|---|---|---|---|---|---|---|
| DCM-1-8 | >20 | >20 | >20 | 6.09 | 5.64 | - | - | - |
| DCM-10-15 | >20 | >20 | 4.26 | <0.8 | 0.81 | 11.41 | 1.89 | 3.47 |
| DCM-12 | 6.07 | 12.58 | <0.8 | 1.95 | 3 | 5.68 | 1.65 | 2.95 |
| DCM-25-32 | <0.8 | 5.35 | <0.8 | <0.8 | <0.8 | 3.70 | 1.34 | <0.8 |
| DCM-56 | >20 | >20 | >20 | >20 | >20 | - | - | 2.94 |
| DCM-1-8-A-31 | >20 | >20 | >20 | 9.91 | >20 | - | - | - |
| DCM-1-8-A-41 | >20 | >20 | 4.41 | 4.51 | 10.62 | 5.41 | 8.52 | 4.52 |
| DCM-1-8-A-63 | >20 | >20 | >20 | 4.29 | 4.55 | 11.39 | 14.49 | 9.97 |
| DCM-1-8-A-167 | >20 | >20 | >20 | 4.5 | 4.41 | - | - | - |
| MRSA BAA-1708 | MRSA BAA-1717 | MRSA 33591 | MRSA BAA-1696 | MRSA XEN31 | |
|---|---|---|---|---|---|
| Compound | IC50/MIC/MBC | IC50/MIC/MBC | IC50/MIC/MBC | IC50/MIC/MBC | IC50/MIC/MBC |
| 6 | 10.61/-/- | I1.93/-/- | _ | 11.87/-/- | 3.50/-/- |
| 7 | 5.36/-/- | 3.29/-/- | NT | 14.64/-/- | 4.95/20/20 |
| 8 | 0.69/1.25/1.25 | 0.72/1.25/10 | NT | 0.71/2.5/2.5 | 0.46/1.25/2.5 |
| 10 | 1.03/2.5/5 | 1.03/2.5/5 | NT | 1.07/2.5/2.5 | 1.01/2.5/5 |
| 11 | 0.38/1.25/1.25 | 0.38/1.25/2.5 | 0.71/1.25/1.25 | 0.43/1.25/5 | 0.33/0.63/1.25 |
| 12 | 1.52/5/5 | 0.38/2.5/5 | 1.57/2.5/5 | 1.40/5/10 | 1.64/5/10 |
| 10 + 11 | 0.34/1.25/1.25 | 0.39/1.25/1.25 | 0.61/1.25/10 | 0.41/1.25/10 | 0.32/0.63/1.25 |
| Ciprofloxacin | -/-/- | 0.14/0.63/1.25 | 0.04/0.16/0.31 | 6.17/-/- | 0.10/0.31/0.63 |
| Vancomycin | >20/>20/>20 | 0.73/1.25/>20 | 0.47/1.25/5.0 | 0.37/0.62/>20 | NT |
| Methicillin | 2.2/50/50 | - | - | 2.54/50/50 | 0.38/1.56/3.13 |
| Cefotaxime | 0.35/0.63/0.63 | 0.35/0.63/0.63 | NT | 2.47/12.5/25 | 0.29/0.78/3.13 |
| Enterococcus faecalis ATCC 29212 a | Enterococcus faecalis ATCC 51299 b | Enterococcus faecium ATCC 700221 c | |
|---|---|---|---|
| Compound | IC50/MIC/MBC | IC50/MIC/MBC | IC50/MIC/MBC |
| 6 | 3.51/-/- | 18.79/-/- | 0.55/1.25/10 |
| 8 | 1.16/2.5/2.5 | 1.02/1.25/5 | 0.48/1.25/2.5 |
| 12 | 2.99/5/10 | 2.96/5/10 | 1.91/2.5/5 |
| 10 + 11 | 0.72/1.25/5 | 0.70/1.25/5 | 0.49/1.25/2.5 |
| Ciprofloxacin | 0.25/0.78/6.25 | 0.22/0.39/6.25 | >20/>20/>20 |
| Vancomycin | 0.73/1.25/>20 | 3.8/10/>20 | >20/>20/>20 |
| Methicillin | 15.3/25/50 | 14.2/50.0/50.0 | >20/>20/>20 |
| Compound | MRSA 1708 | MRSA 1717 | MRSA 33591 | MRSA 1696 | MRS XEN31 | Ef 29212 b | Ef 51299 c | Ef 700221 d |
|---|---|---|---|---|---|---|---|---|
| 7 | 20 | >10 | NT | 10 | 20 | NT | NT | NT |
| 8 | 2.5 | 2.5 | 2.5 | 2.5 | 1.25 | 2.5 | 2.5 | 1.25 |
| 12 | 5 | 5 | 5 | 5 | 5 | 5 | >5 | 5 |
| 11 | 20 | 20 | NT | 10 | 10 | NT | NT | NT |
| 8 + 12 (+2.5 µg/mL) | 0.625 (↓4X) | 0.625 (↓4X) | 1.25 (↓2X) | 0.625 (↓4X) | NT | 1.25 (↓2X) | 1.25 (↓2X) | 0.625 (↓2X) |
| 12 + 8 (+1.25 µg/mL) | 0.156 (↓32X) | 0.625 (↓8X) | 1.25 (↓4X) | NT | NT | 2.5 (↓2X) | 2.5 (↓2X) | NT |
| 12 + 8 (+0.625 µg/mL) | NT | NT | NT | 2.5 (↓2X) | NT | NT | NT | 1.25 (↓4X) |
| 11 + 7 (+2.5 µg/mL) | 5.0 (↓4X) | 10.0 (↓2X) | NT | 5.0 (↓2X) | 2.5 (↓4X) | NT | NT | NT |
| DAPG e | >1.25 | NT | NT | NT | NT | NT | NT | >1.25 |
| 8 + DAPG (+0.63 µg/mL) | 2.5 (=) | NT | NT | NT | NT | NT | NT | 1.25 (=) |
| 12 + DAPG (+0.63 µg/mL) | >5 (=) | NT | NT | NT | NT | NT | NT | 5 (=) |
| Methicillin | 50 | - | - | 50 | 1.56 | 25 | 50.0 | >20 |
| Vancomycin | >20 | 1.25 | 1.25 | 0.62 | NT | 1.25 | 10 | >20 |
| Klebsiella pneumoniae NCTC13368 | Klebsiella pneumoniae M6 | Acinetobacter baumannii AYE | Acinetobacter baumannii ATCC17978 | Pseudomonas aeruginosa PAO1 | Pseudomonas aeruginosa NCTC13437 | Escherichia coli NCTC12923 | |
|---|---|---|---|---|---|---|---|
| Compound | MIC | MIC | MIC | MIC | MIC | MIC | MIC |
| 6 + 7 +PMBN | >64 | >64 | >64 | >64 | 32 | 64 | >64 |
| 10 + 11 +PMBN | >64 | >64 | 8 | 0.5 | 2 | 64 | 2 |
| 11 + PMBN | >64 | >64 | 2 | 1 | 4 | 64 | 2 |
| 12 + PMBN | >64 | >64 | 4 | 2 | 16 | 64 | 2 |
| 8 + PMBN | >64 | >64 | 8 | 2 | 2 | 8 | 2 |
| Ciprofloxacin a | 0.5 | ≤0.125 | 128 | 0.5 | 0.5 | 64 | ≤0.125 |
| Ceftazidime a | >128 | 0.25 | >128 | <0.5 | 1 | >128 | 0.5 |
| Gentamicin a | 4 | 0.25 | >512 | 0.5 | 4 | 256 | 1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Muhammad, I.; Jacob, M.R.; Ibrahim, M.A.; Raman, V.; Kumarihamy, M.; Wang, M.; Al-Adhami, T.; Hind, C.; Clifford, M.; Martin, B.; et al. Antimicrobial Constituents from Machaerium Pers.: Inhibitory Activities and Synergism of Machaeriols and Machaeridiols against Methicillin-Resistant Staphylococcus aureus, Vancomycin-Resistant Enterococcus faecium, and Permeabilized Gram-Negative Pathogens. Molecules 2020, 25, 6000. https://doi.org/10.3390/molecules25246000
Muhammad I, Jacob MR, Ibrahim MA, Raman V, Kumarihamy M, Wang M, Al-Adhami T, Hind C, Clifford M, Martin B, et al. Antimicrobial Constituents from Machaerium Pers.: Inhibitory Activities and Synergism of Machaeriols and Machaeridiols against Methicillin-Resistant Staphylococcus aureus, Vancomycin-Resistant Enterococcus faecium, and Permeabilized Gram-Negative Pathogens. Molecules. 2020; 25(24):6000. https://doi.org/10.3390/molecules25246000
Chicago/Turabian StyleMuhammad, Ilias, Melissa R. Jacob, Mohamed A. Ibrahim, Vijayasankar Raman, Mallika Kumarihamy, Mei Wang, Taha Al-Adhami, Charlotte Hind, Melanie Clifford, Bethany Martin, and et al. 2020. "Antimicrobial Constituents from Machaerium Pers.: Inhibitory Activities and Synergism of Machaeriols and Machaeridiols against Methicillin-Resistant Staphylococcus aureus, Vancomycin-Resistant Enterococcus faecium, and Permeabilized Gram-Negative Pathogens" Molecules 25, no. 24: 6000. https://doi.org/10.3390/molecules25246000
APA StyleMuhammad, I., Jacob, M. R., Ibrahim, M. A., Raman, V., Kumarihamy, M., Wang, M., Al-Adhami, T., Hind, C., Clifford, M., Martin, B., Zhao, J., Sutton, J. M., & Rahman, K. M. (2020). Antimicrobial Constituents from Machaerium Pers.: Inhibitory Activities and Synergism of Machaeriols and Machaeridiols against Methicillin-Resistant Staphylococcus aureus, Vancomycin-Resistant Enterococcus faecium, and Permeabilized Gram-Negative Pathogens. Molecules, 25(24), 6000. https://doi.org/10.3390/molecules25246000