Preparation, Spectroscopic Characterization, Theoretical Investigations, and In Vitro Anticancer Activity of Cd(II), Ni(II), Zn(II), and Cu(II) Complexes of 4(3H)-Quinazolinone-Derived Schiff Base
Abstract
1. Introduction
2. Results and Discussion
2.1. 1H-NMR Spectral Analysis
2.2. FT-IR Spectral Studies
2.3. UV–Visible Spectroscopy
2.4. Mass Spectral Studies
2.5. Thermal Analysis
X-ray Diffraction (XRD) Analysis
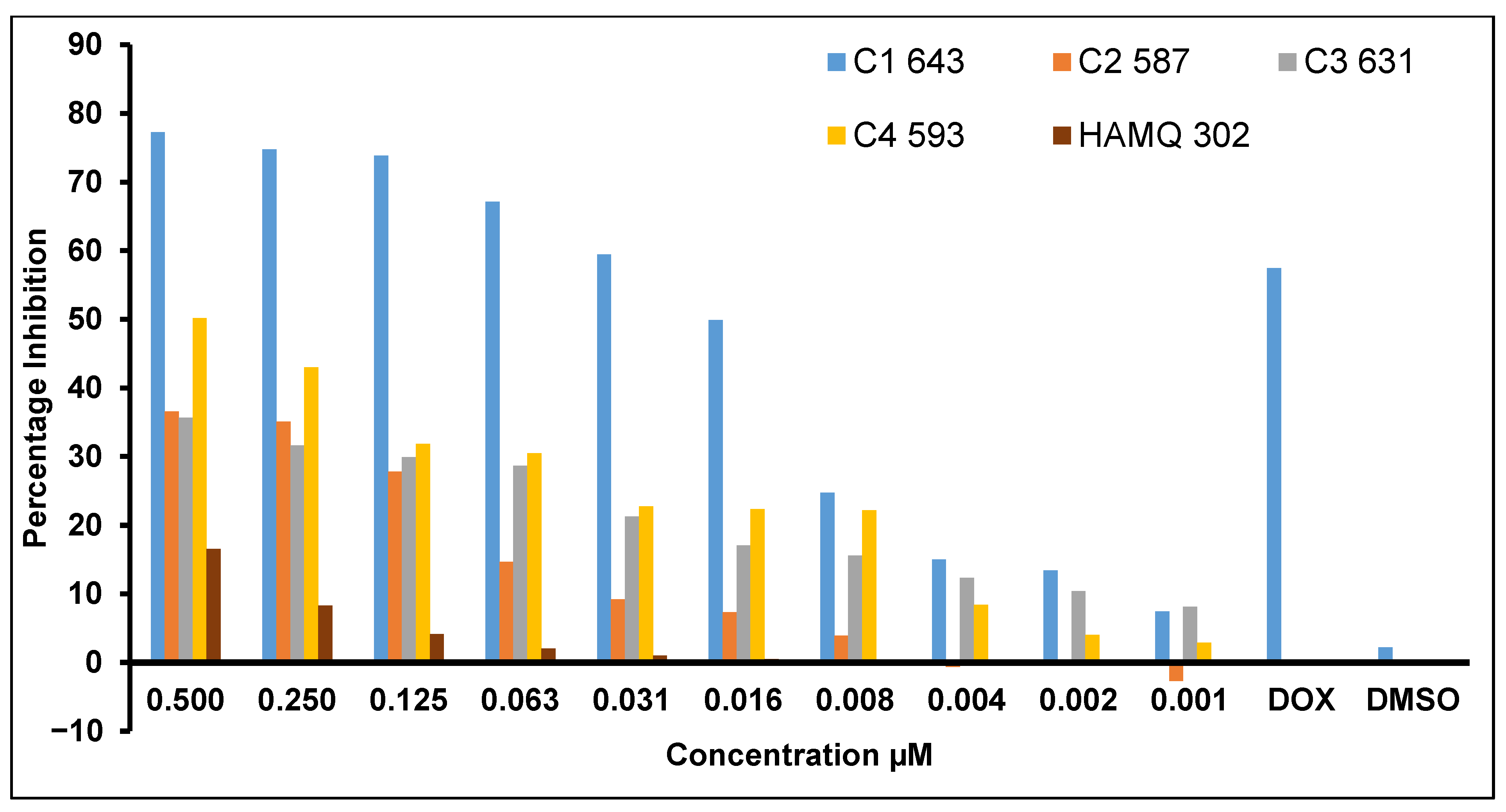
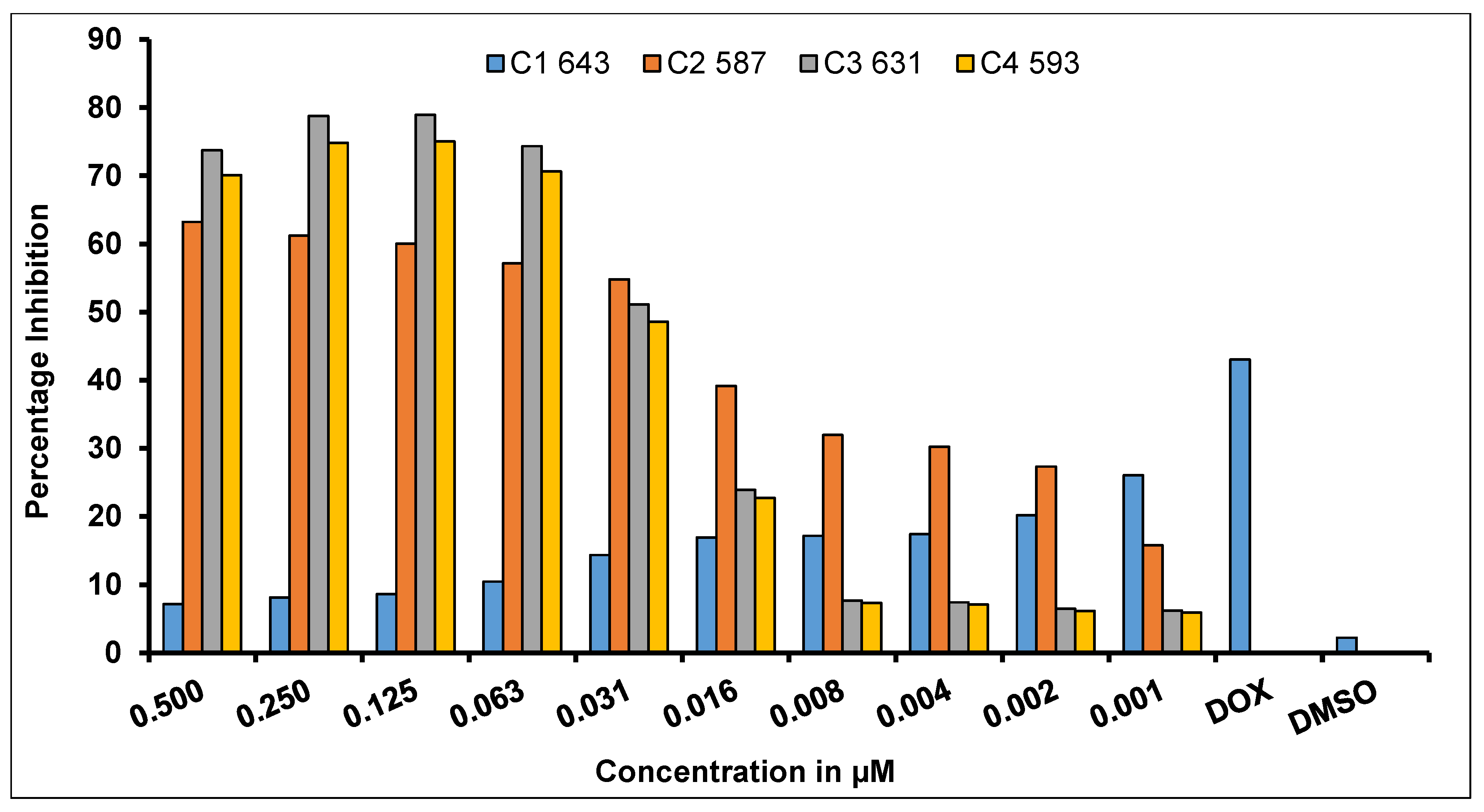
2.6. In Vitro Anticancer Activity Evaluation Using Sulforhodamine-B (SRB) Assay
2.7. Determination of the Descriptors Related to Chemical Reactivity
3. Experimental
3.1. Materials and Methods
3.2. Computational Details
3.3. Chemical Synthesis
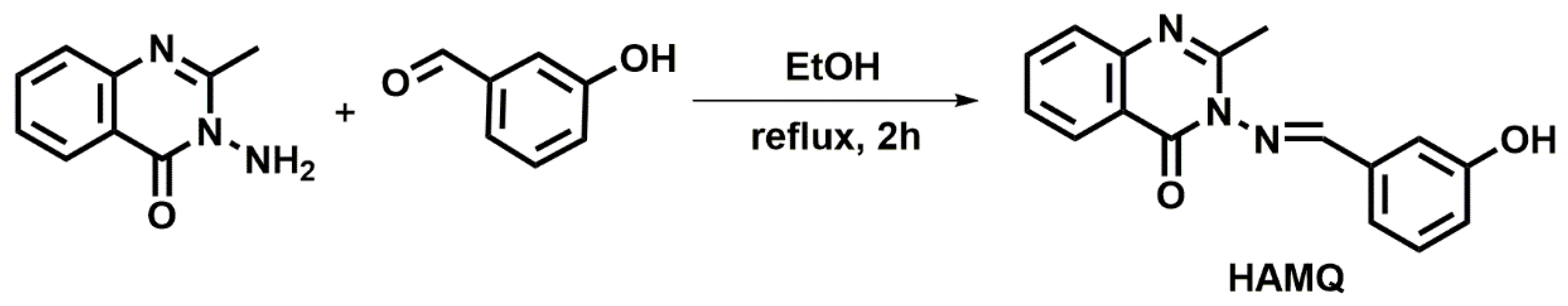
3.3.1. Synthesis of 3-[[(E)-(3-Hydroxyphenyl)methylidene]amino]-2-methyl- quinazolin-4(3H)-one (HAMQ)
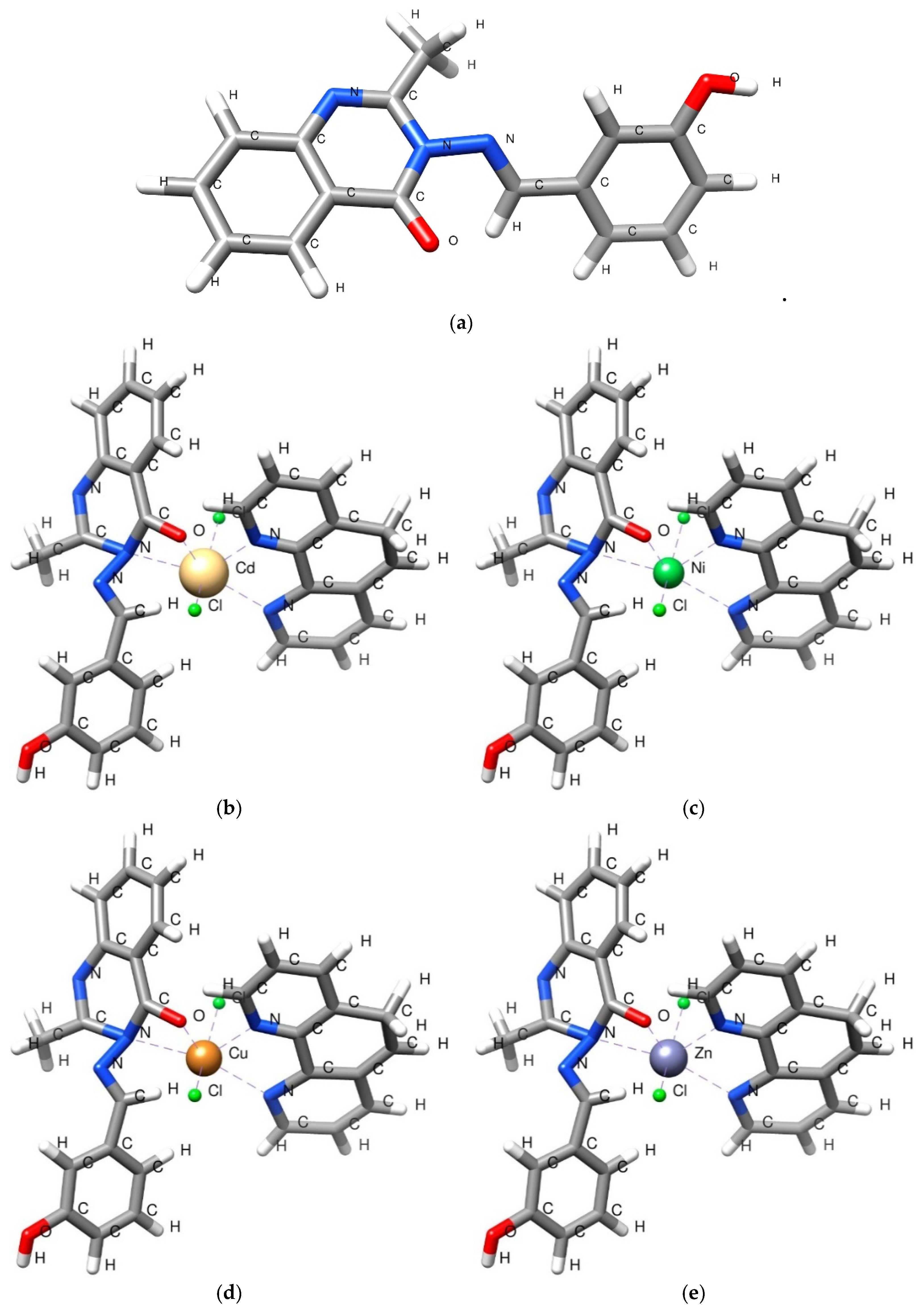
3.3.2. Synthesis of Metal Complexes (C1–C4)
3.3.3. In Vitro Antiproliferative Activity against Human Cancer Cell Lines
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Misra, R.; Acharya, S.; Sahoo, S.K. Cancer nanotechnology: Application of nanotechnology in cancer therapy. Drug Discov. Today 2010, 15, 842–850. [Google Scholar] [CrossRef] [PubMed]
- Masoud, V.; Pagès, G. Targeted therapies in breast cancer: New challenges to fight against resistance. World J. Clin. Oncol. 2017, 8, 120–134. [Google Scholar] [CrossRef] [PubMed]
- Shaili, E. Platinum anticancer drugs and photochemotherapeutic agents: Recent advances and future developments. Sci. Prog. 2014, 97, 20–40. [Google Scholar] [CrossRef] [PubMed]
- King, A.P.; Gellineau, H.A.; Ahn, J.E.; MacMillan, S.N.; Wilson, J.J. Bis(thiosemicarbazone) Complexes of Cobalt(III). Synthesis, Characterization, and Anticancer Potential. Inorg. Chem. 2017, 56, 6609–6623. [Google Scholar] [CrossRef]
- Ambika, S.; Manojkumar, Y.; Arunachalam, S.; Gowdhami, B.; Sundaram, K.K.M.; Solomon, R.V.; Venuvanalingam, P.; Akbarsha, M.A.; Sundararaman, M. Biomolecular Interaction, Anti-Cancer and Anti-Angiogenic Properties of Cobalt(III) Schiff Base Complexes. Sci Rep. 2019, 9, 2721. [Google Scholar] [CrossRef]
- Miller, M.; Tshuva, E.Y. Racemic vs. enantiopure inert Ti(IV) complex of a single diaminotetrakis(phenolato) ligand in anticancer activity toward human drug-sensitive and -resistant cancer cell lines. RSC Adv. 2018, 8, 39731. [Google Scholar] [CrossRef]
- Ashok, U.P.; Kollur, S.P.; Arun, B.P.; Sanjay, C.; Suresh, K.S.; Anil, N.; Baburao, H.V.; Markad, D.; Castro, J.O.; Frau, J.; et al. In vitro anticancer activity of 4(3H)-quinazolinone derived Schiff base and its Cu(II), Zn(II) and Cd(II) complexes: Preparation, X-ray structural, spectral characterization and theoretical investigations. Inorg. Chim. Acta 2020, 511, 119846. [Google Scholar] [CrossRef]
- Sivaramakarthikeyan, R.; Iniyaval, S.; Saravanan, V.; Lim, W.-M.; Mai, C.-W.; Ramalingan, C. Molecular Hybrids Integrated with Benzimidazole and Pyrazole Structural Motifs: Design, Synthesis, Biological Evaluation, and Molecular Docking Studies. ACS Omega 2020, 5, 10089–10098. [Google Scholar] [CrossRef]
- Mohamed, G.G. Metal complexes of antibiotic drugs. Studies on dicluxacillin complexes of FeII, FeIII, CoII, NiII and CuII. Spectrochim. Acta A 2001, 57, 1643–1648. [Google Scholar] [CrossRef]
- Panchal, P.K.; Patel, M.N. Synthesis, Structural Characterization, and Antibacterial Studies of Some Mixed-Ligand First Row d-Transition Metal Complexes. Synth. React. Inorg. Met. Chem. 2004, 34, 1277–1289. [Google Scholar] [CrossRef]
- Larson, E.J.; Pecoraro, V.L. The peroxide-dependent mu.2-O bond formation of manganese complex [Mn(IV)SALPN(O)]2. J. Am. Chem. Soc. 1991, 113, 3810–3818. [Google Scholar] [CrossRef]
- Masoud, M.S.; Amira, M.F.; Ramadan, A.M.; El-Ashry, G.M. Synthesis and characterization of some pyrimidine, purine, amino acid and mixed ligand complexes. Spectrochim. Acta A 2008, 69, 230–238. [Google Scholar] [CrossRef] [PubMed]
- Shiri, -Y.Z.; Yaftian, M.R. Anion Control Selectivity of Neutral N4-Type Schiff Base Extractants towards Transition Metal Ions, Iran. J. Chem. Chem. Eng. 2010, 29, 11–17. [Google Scholar]
- Kumar, R.; Mani, G. Exhibition of the Brønsted acid–base character of a Schiff base in palladium(ii) complex formation: Lithium complexation, fluxional properties and catalysis of Suzuki reactions in water. Dalton Trans. 2015, 44, 6896–6908. [Google Scholar] [CrossRef] [PubMed]
- Puthilibai, G.; Vasudhevan, S.; Rani, S.K.; Rajagopal, G. Synthesis, spectroscopic characterization, electrochemical behaviour and antibacterial activity of Ru(III) complexes of 2-[(4-N,N′-dimethylaminophenylimino)-methyl]-4-halophenol. Spectrochim. Acta A 2009, 72, 796–800. [Google Scholar] [CrossRef]
- Kulkarni, A.D.; Bagihalli, G.B.; Patil, S.A.; Badami, P.S. Synthesis, characterization, electrochemical and in-vitro antimicrobial studies of Co(II), Ni(II), and Cu(II) complexes with Schiff bases of formyl coumarin derivatives. J. Coord. Chem. 2009, 62, 3060–3072. [Google Scholar] [CrossRef]
- Gupta, K.; Sutar, A.K. Catalytic Activities of Schiff Base Transition Metal Complexes. Coord. Chem. Rev. 2008, 252, 1420–1450. [Google Scholar] [CrossRef]
- Shivakumar, L.; Shivaprasad, K.; Revanasiddappa, H.D. Synthesis, spectroscopic characterization, antimicrobial, DNA binding and oxidative-induced DNA cleavage activities: New oxovanadium(IV) complexes of 2-(2-hydroxybenzylideneamino)isoindoline-1,3-dione. Spectrochim. Acta A 2012, 97, 659–666. [Google Scholar] [CrossRef]
- Bansode, P.; Patil, P.; Choudhari, P.; Bhatia, M.; Birajdar, A.; Somasundaram, I.; Rashinkar, G. Anticancer activity and molecular docking studies of ferrocene tethered ionic liquids. J. Mol. Liq. 2019, 290, 111182. [Google Scholar] [CrossRef]
- Bansode, P.; Anantacharya, R.; Dhanavade, M.; Kamble, S.; Barale, S.; Sonawane, K.; Satyanarayan, N.D.; Rashinkar, G. Evaluation of drug candidature: In silico ADMET, binding interactions with CDK7 and normal cell line studies of potentially anti-breast cancer enamidines. Comput. Biol. Chem. 2019, 83, 107124. [Google Scholar] [CrossRef]
- Bansode, P.A.; Patil, P.V.; Birajdar, A.R.; Somasundaram, I.; Bachute, M.T.; Rashinkar, G. Anticancer, Antioxidant and Antiangiogenic Activities of Nanoparticles of Bioactive Dietary Nutraceuticals. ChemistrySelect 2019, 4, 13792–13796. [Google Scholar] [CrossRef]
- Bagihalli, G.B.; Avaji, P.G.; Patil, S.A.; Badami, P.S. Synthesis, spectral characterization, in vitro antibacterial, antifungal and cytotoxic activities of Co(II), Ni(II) and Cu(II) complexes with 1,2,4-triazole Schiff bases. Eur. J. Med. Chem. 2008, 43, 2639–2649. [Google Scholar] [CrossRef] [PubMed]
- Skehan, P.; Storeng, R.; Scudiero, D.; Monks, A.; McMahon, J.; Vistica, D.; Warren, J.T.; Bokesch, H.; Kenney, S.; Boyd, M.R. New colorimetric cytotoxicity assay for anticancer-drug screening. J. Natl. Cancer Inst. 1990, 82, 1107–1112. [Google Scholar] [CrossRef] [PubMed]
- Sakthivel, A.; Thangagiri, B.; Raman, N.; Joseph, J.; Guda, R.; Kasula, M.; Mitu, L. Spectroscopic, SOD, anticancer, antimicrobial, molecular docking and DNA binding properties of bioactive VO(IV), Cu(II), Zn(II), Co(II), Mn(II) and Ni(II) complexes obtained from 3-(2-hydroxy-3-methoxybenzylidene)pentane-2,4-dione. J. Biomol. Struct. Dyn. 2020, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Flores-Holguín, N.; Frau, J.; Glossman-Mitnik, D. Chemical Reactivity Properties, Drug-Likeness Features and Bioactivity Scores of the Cholecystokinin Peptide Hormone. Comput. Mol. Biosci. 2019, 9, 41–47. [Google Scholar] [CrossRef][Green Version]
- Flores-Holguín, N.; Frau, J.; Glossman-Mitnik, D. Cheminformatics and Its Applications; Amalia, S., Ed.; IntechOpen: Rijeka, Croatia, 2019; pp. 1–8. [Google Scholar]
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Scalmani, G.; Barone, V.; Mennucci, B.; Petersson, G.A. Gaussian 09Revision E.01 2016; Gaussian Inc.: Wallingford, CT, USA, 2016. [Google Scholar]
- Peverati, R.; Truhlar, D.G. Screened-exchange density functionals with broad accuracy for chemistry and solid-state physics. Phys. Chem. Chem. Phys. 2012, 14, 16187–16191. [Google Scholar] [CrossRef]
- Parr, R.; Yang, W. Density-Functional Theory of Atoms and Molecules; Oxford University Press: New York, NY, USA, 1989. [Google Scholar] [CrossRef]
- Geerlings, P.; de Proft, F.; Langenaeker, W. Conceptual Density Functional Theory. Chem. Rev. 2003, 103, 1793–1874. [Google Scholar] [CrossRef]
- Gázquez, J.; Cedillo, A.; Vela, A. Electrodonating and Electroaccepting Powers. J. Phys. Chem. A 2007, 111, 1966–1970. [Google Scholar] [CrossRef]
- Chattaraj, P.; Chakraborty, A.; Giri, S. Net Electrophilicity. J. Phys. Chem. A 2009, 113, 10068–10074. [Google Scholar] [CrossRef]
- Frau, J.; Glossman-Mitnik, D. Molecular Reactivity and Absorption Properties of Melanoidin Blue-G1 through Conceptual DFT. Molecules 2018, 23, 559. [Google Scholar] [CrossRef]
- Frau, J.; Glossman-Mitnik, D. Conceptual DFT study of the local chemical reactivity of the dilysyldipyrrolones A and B intermediate melanoidins. Theor. Chem. Acc. 2018, 137, 1210. [Google Scholar] [CrossRef]
- Frau, J.; Glossman-Mitnik, D. Conceptual DFT Study of the Local Chemical Reactivity of the Colored BISARG Melanoidin and Its Protonated Derivative. Front. Chem. 2018, 6, 136. [Google Scholar] [CrossRef] [PubMed]
- Frau, J.; Glossman-Mitnik, D. Molecular Reactivity of some Maillard Reaction Products Studied through Conceptual DFT. Contemp. Chem. 2018, 1, 1–14. [Google Scholar]
- Frau, J.; Glossman-Mitnik, D. Computational study of the chemical reactivity of the Blue-M1 intermediate melanoidin. Comput. Theor. Chem. 2018, 1134, 22–29. [Google Scholar] [CrossRef]
- Frau, J.; Glossman-Mitnik, D. Chemical Reactivity Theory Applied to the Calculation of the Local Reactivity Descriptors of a Colored Maillard Reaction Product. Chem. Sci. Int. J. 2018, 22, 1–14. [Google Scholar] [CrossRef]
- Frau, J.; Glossman-Mitnik, D. Blue M2: An intermediate melanoidin studied via conceptual DFT. J. Mol. Model 2018, 24, 1–13. [Google Scholar] [CrossRef]
- Frau, J.; Flores-Holguín, N.; Glossman-Mitnik, D. Chemical Reactivity Properties, pKa Values, AGEs Inhibitor Abilities and Bioactivity Scores of the Mirabamides A–H Peptides of Marine Origin Studied by Means of Conceptual DFT. Mar. Drugs 2018, 16, 302. [Google Scholar] [CrossRef]
- Weigend, F.; Ahlrichs, R. Balanced basis sets of split valence, triple zeta valence and quadruple zeta valence quality for H to Rn: Design and assessment of accuracy. Phys. Chem. Chem. Phys. 2005, 7, 3297–3305. [Google Scholar] [CrossRef]
- Weigend, F. Accurate Coulomb-fitting basis sets for H to Rn. Phys. Chem. Chem. Phys. 2006, 8, 1057–1065. [Google Scholar] [CrossRef]
- Marenich, A.; Cramer, C.; Truhlar, D. Universal Solvation Model Based on Solute Electron Density and on a Continuum Model of the Solvent Defined by the Bulk Dielectric Constant and Atomic Surface Tensions. J. Phys. Chem. B 2009, 113, 6378–6396. [Google Scholar] [CrossRef]
- Yanai, T.; Tew, D.P.; Handy, N.C. A new hybrid exchange–correlation functional using the Coulomb-attenuating method (CAM-B3LYP). Chem. Phys. Lett. 2004, 393, 51–57. [Google Scholar] [CrossRef]
Sample Availability: Not available. |
| Compound | Lattice Constants (A˚) | Angles (˚) | FoM | Volume (Å) | Crystallite Size (Å) |
|---|---|---|---|---|---|
| C1 | a = 4.8 b = 14.1 c = 15.8 | α = 55.11 β = 82.97 ϓ = 41.62 | 1.89 | 1069.34 | 570.62 |
| C2 | a = 5.3 b = 7.4 c = 18.4 | α = 56.55 β = 85.65 ϓ = 90.48 | 9.95 | 721.65 | 432.81 |
| C3 | a = 4.9 b = 9.4 c = 23.3 | α = 48.20 β = 61 ϓ = 58.99 | 3.76 | 1073.20 | 312.51 |
| C4 | a = 2.5 b = 7.3 c = 13.7 | α = 50.80 β = 67.40 ϓ = 69.06 | 2.23 | 250.03 | 1336.94 |
| Concentration (µg/mL) | IC50 (µM) | ||||
|---|---|---|---|---|---|
| C1 (MW 643) | C2 (MW 587) | C3 (MW 631) | C4 (MW 593) | HAMQ (MW 302) | |
| 0.5 | 77.26 | 36.58 | 35.64 | 50.17 | 16.56 |
| 0.250 | 74.78 | 35.11 | 31.65 | 42.98 | 8.28 |
| 0.125 | 73.83 | 27.8 | 29.9 | 31.86 | 4.14 |
| 0.063 | 67.12 | 14.69 | 28.64 | 30.5 | 2.07 |
| 0.031 | 59.49 | 9.23 | 21.3 | 22.73 | 1.03 |
| 0.016 | 49.91 | 7.31 | 17.03 | 22.38 | 0.52 |
| 0.008 | 24.72 | 3.88 | 15.59 | 22.17 | 0.26 |
| 0.004 | 150.1 | −0.62 | 12.34 | 8.42 | 0.13 |
| 0.002 | 13.43 | 0.27 | 10.42 | 4.05 | 0.06 |
| 0.001 | 7.43 | −2.69 | 8.14 | 2.86 | 0.03 |
| Doxorubicin | 18.40 | ||||
| Compound | HOMO | LUMO | SOMO | H–L gap | ΔSL | |||
|---|---|---|---|---|---|---|---|---|
| HAMQ | −6.16 | −2.21 | −2.25 | 3.95 | 0.01 | 0.02 | 0.02 | 0.04 |
| C1 | −6.08 | −2.57 | −2.57 | 3.51 | 0.06 | 0.00 | 0.06 | 0.00 |
| C2 | −6.16 | −2.62 | −2.61 | 3.54 | 0.02 | 0.00 | 0.02 | 0.01 |
| C3 | −5.89 | −2.69 | −2.61 | 3.28 | 0.02 | 0.01 | 0.06 | 0.08 |
| C4 | −6.05 | −2.69 | −2.67 | 3.36 | 0.06 | 0.01 | 0.06 | 0.02 |
| Compound | ||||||
|---|---|---|---|---|---|---|
| HAMQ | 4.19 | 3.95 | 2.22 | 6.78 | 2.59 | 9.37 |
| C1 | 4.32 | 3.52 | 2.66 | 7.70 | 3.38 | 11.08 |
| C2 | 4.39 | 3.54 | 2.72 | 7.85 | 3.46 | 11.31 |
| C3 | 4.29 | 3.20 | 2.87 | 8.08 | 3.79 | 11.88 |
| C4 | 4.39 | 3.54 | 2.72 | 7.85 | 3.46 | 11.31 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ashok, U.P.; Kollur, S.P.; Anil, N.; Arun, B.P.; Jadhav, S.N.; Sarsamkar, S.; Helavi, V.B.; Srinivasan, A.; Kaulage, S.; Veerapur, R.; et al. Preparation, Spectroscopic Characterization, Theoretical Investigations, and In Vitro Anticancer Activity of Cd(II), Ni(II), Zn(II), and Cu(II) Complexes of 4(3H)-Quinazolinone-Derived Schiff Base. Molecules 2020, 25, 5973. https://doi.org/10.3390/molecules25245973
Ashok UP, Kollur SP, Anil N, Arun BP, Jadhav SN, Sarsamkar S, Helavi VB, Srinivasan A, Kaulage S, Veerapur R, et al. Preparation, Spectroscopic Characterization, Theoretical Investigations, and In Vitro Anticancer Activity of Cd(II), Ni(II), Zn(II), and Cu(II) Complexes of 4(3H)-Quinazolinone-Derived Schiff Base. Molecules. 2020; 25(24):5973. https://doi.org/10.3390/molecules25245973
Chicago/Turabian StyleAshok, Ubale Panchsheela, Shiva Prasad Kollur, Nishad Anil, Bansode Prakash Arun, Sanjay Namdev Jadhav, Sanjay Sarsamkar, Vasant Baburao Helavi, Asha Srinivasan, Sandeep Kaulage, Ravindra Veerapur, and et al. 2020. "Preparation, Spectroscopic Characterization, Theoretical Investigations, and In Vitro Anticancer Activity of Cd(II), Ni(II), Zn(II), and Cu(II) Complexes of 4(3H)-Quinazolinone-Derived Schiff Base" Molecules 25, no. 24: 5973. https://doi.org/10.3390/molecules25245973
APA StyleAshok, U. P., Kollur, S. P., Anil, N., Arun, B. P., Jadhav, S. N., Sarsamkar, S., Helavi, V. B., Srinivasan, A., Kaulage, S., Veerapur, R., Al-Rashed, S., Syed, A., Ortega-Castro, J., Frau, J., Flores-Holguín, N., & Glossman-Mitnik, D. (2020). Preparation, Spectroscopic Characterization, Theoretical Investigations, and In Vitro Anticancer Activity of Cd(II), Ni(II), Zn(II), and Cu(II) Complexes of 4(3H)-Quinazolinone-Derived Schiff Base. Molecules, 25(24), 5973. https://doi.org/10.3390/molecules25245973








