The Cannabis Terpenes
Abstract
1. Introduction
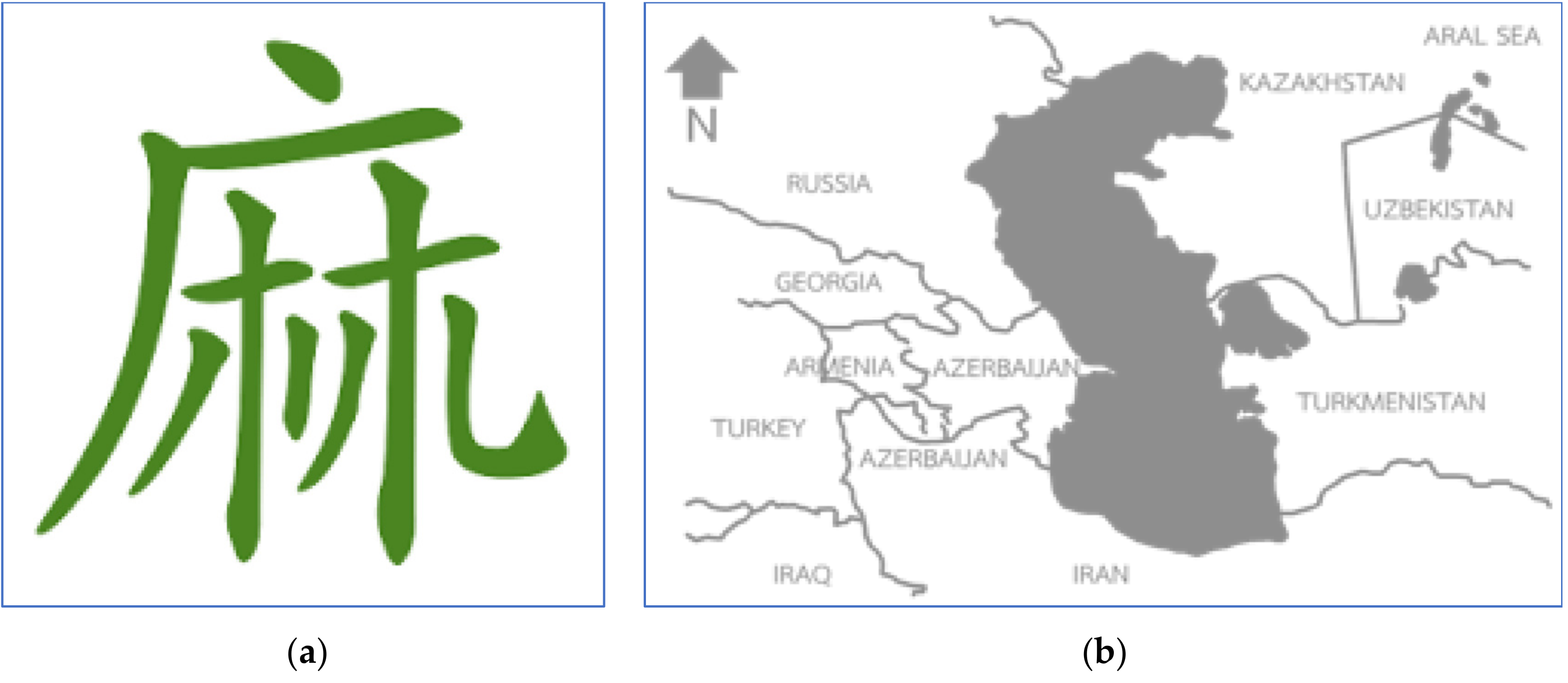
2. The Cannabis Discovery and Its Importance as a Source of Terpene
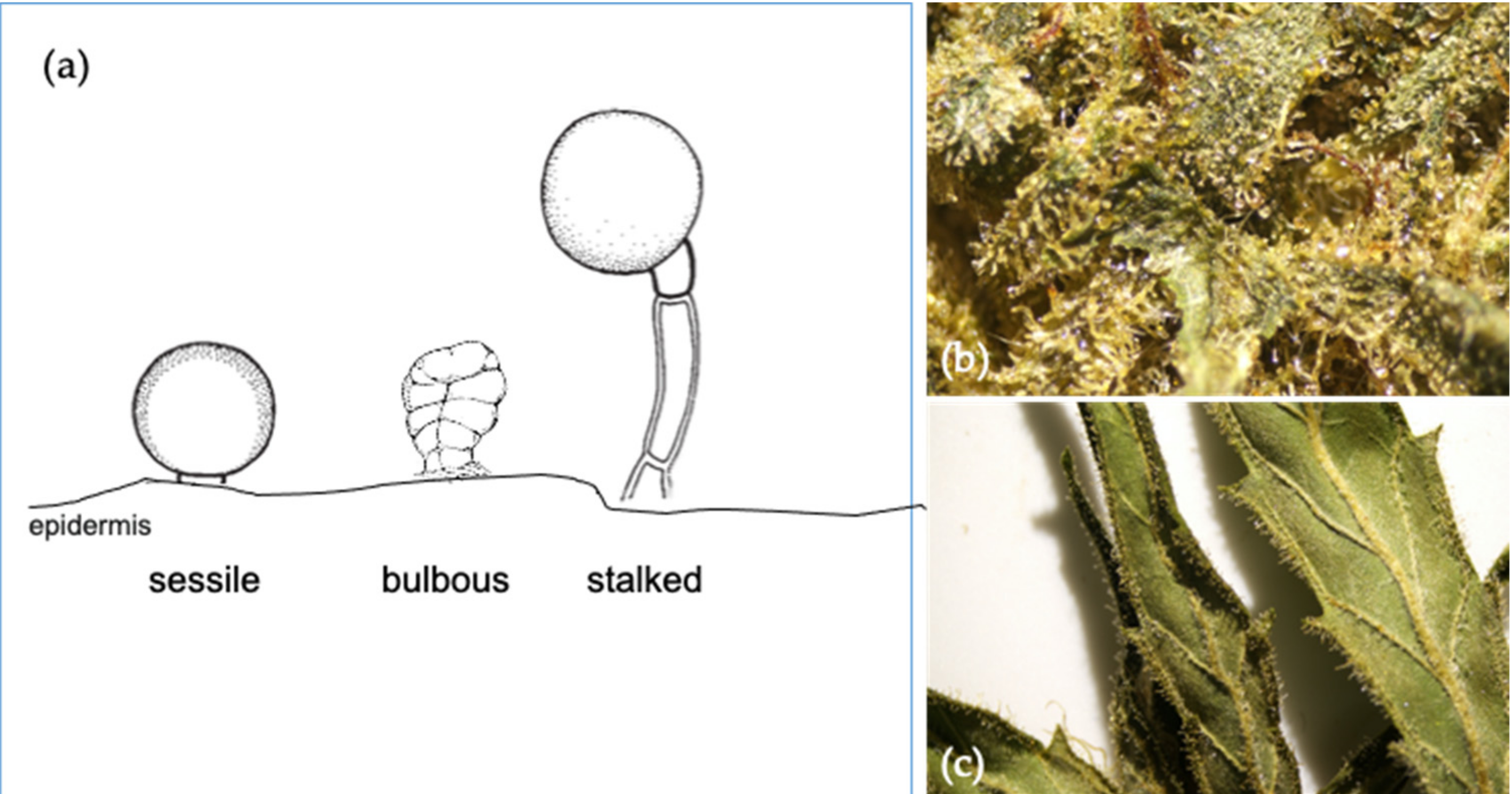
3. Taxonomy and Localization of the Cannabis Terpenes
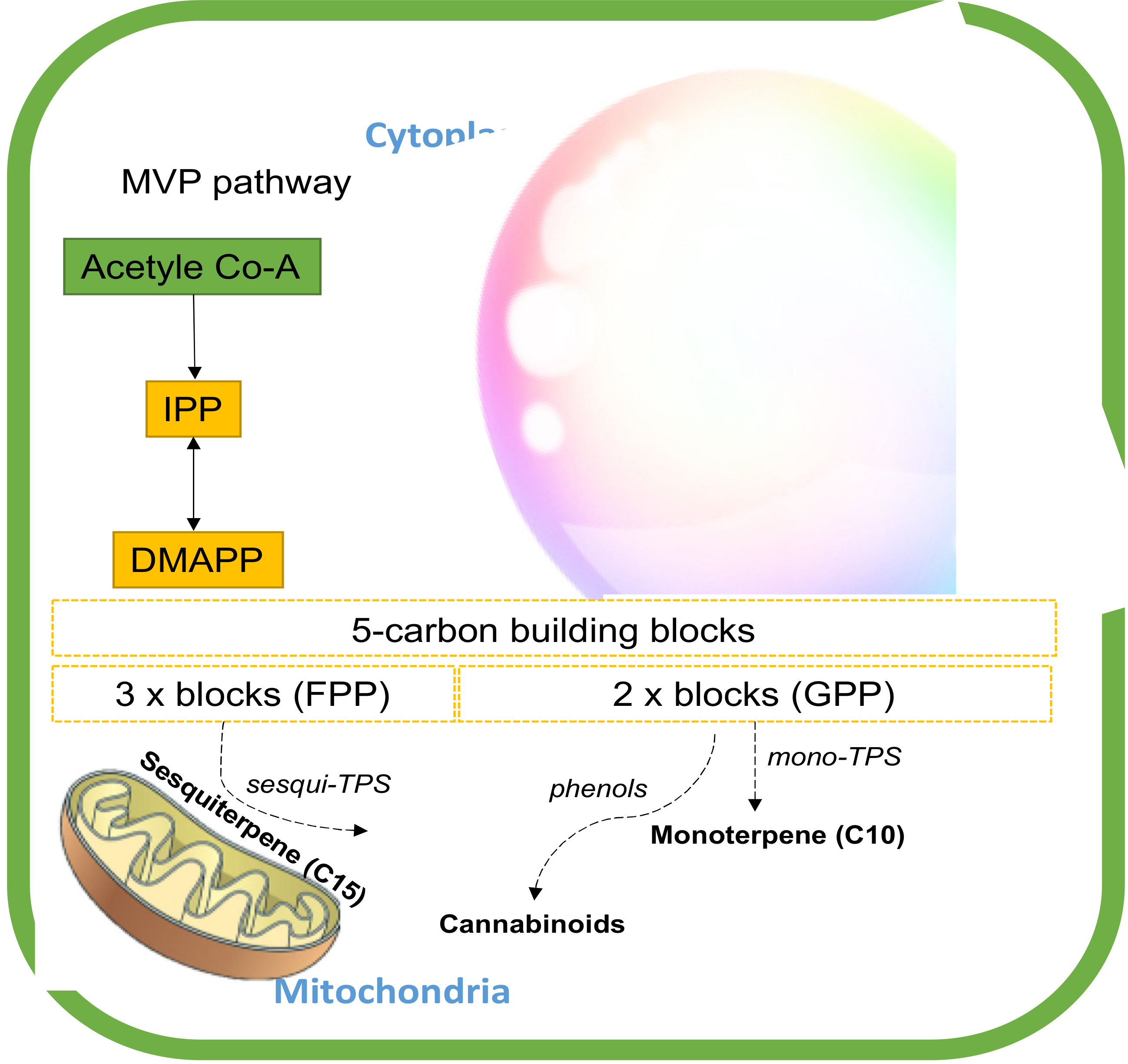
4. Terpene Biosynthesis in Cannabis
4.1. Cannabis Monoterpene
4.2. Cannabis Sesquiterpenes
5. The Cannabis Chemovars
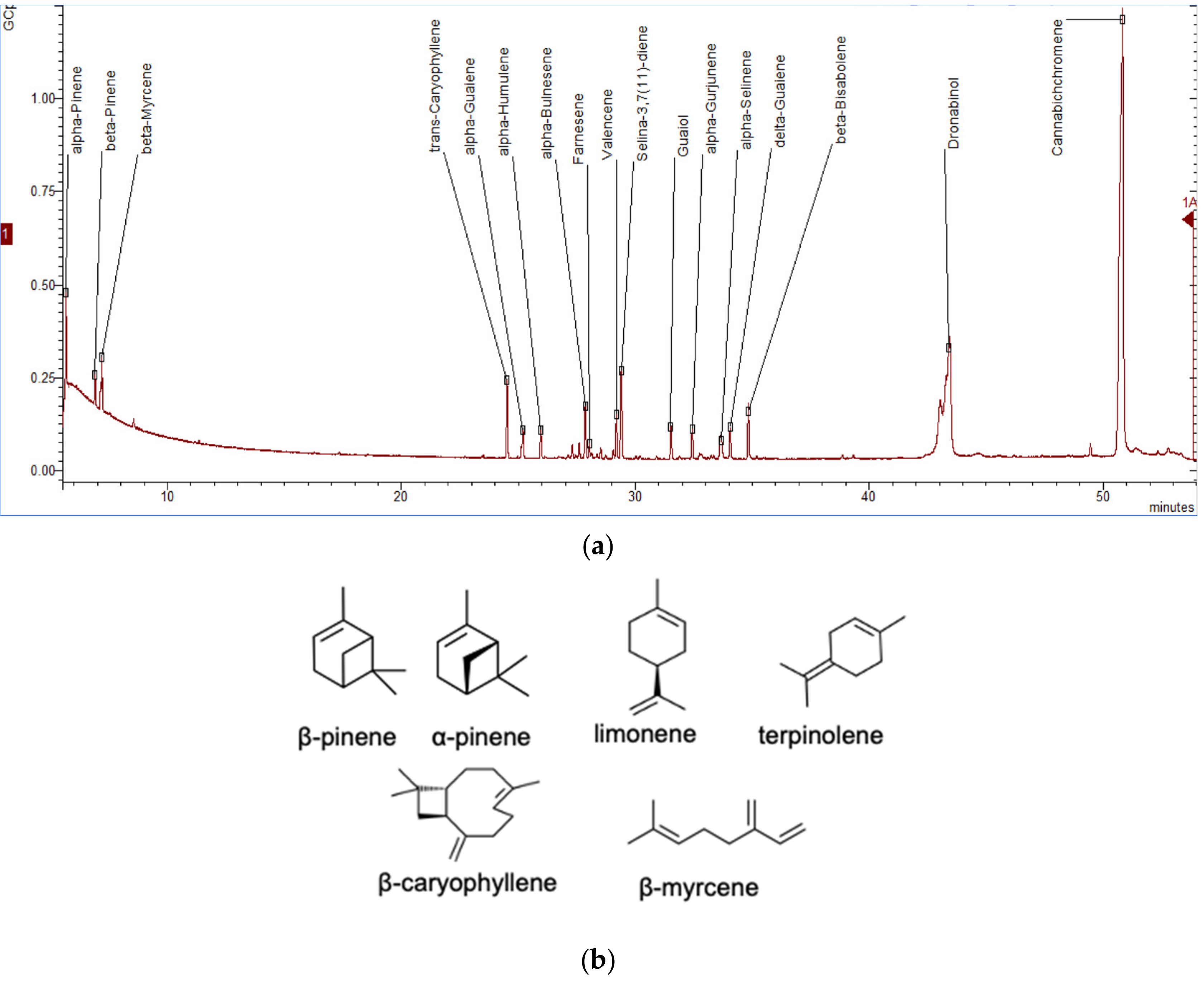
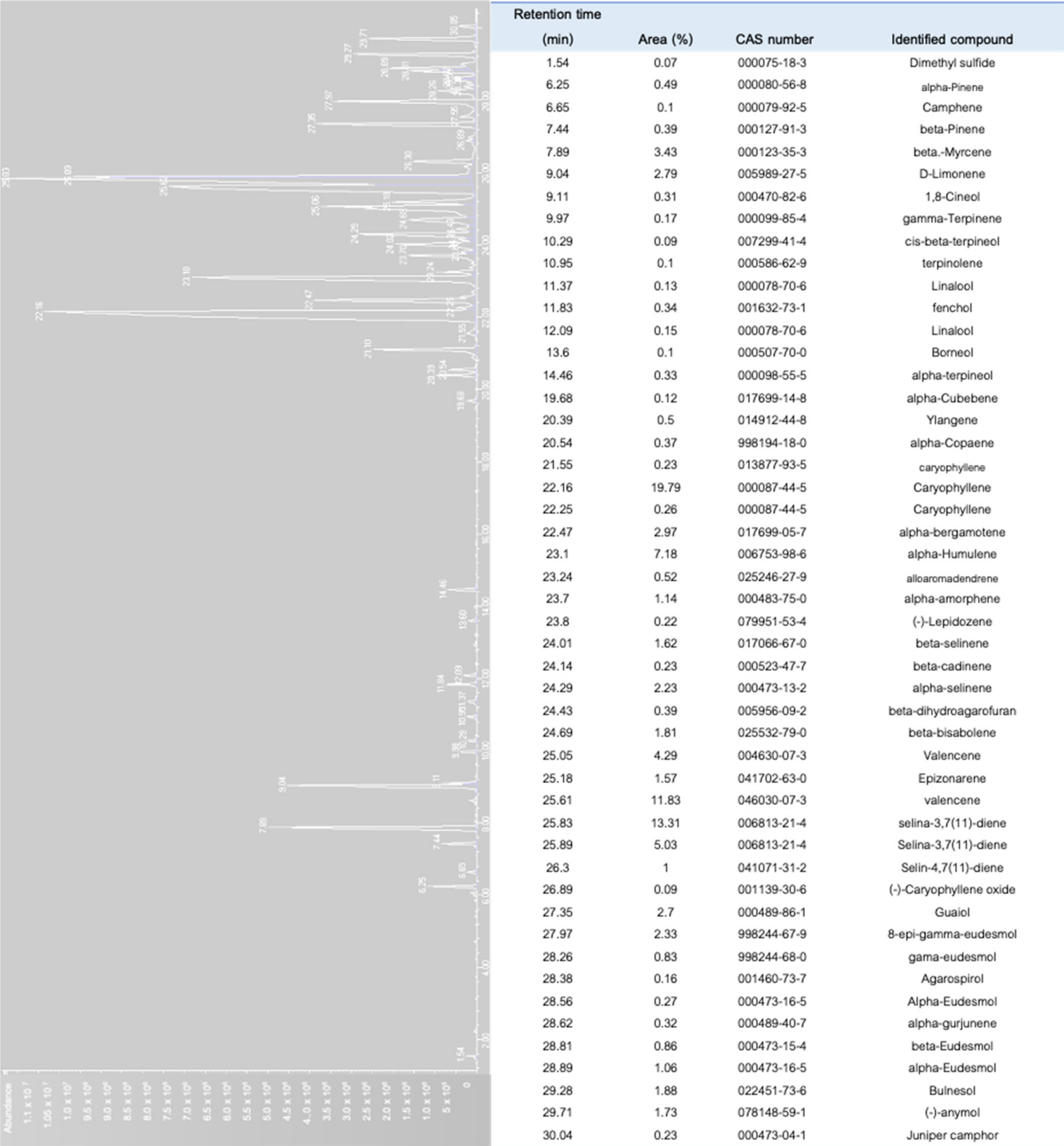
6. Separation of Cannabis Terpenes and Industrial Importance
7. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Cheng, C.; Zang, G.; Zhao, L.; Gao, C.; Tang, Q.; Chen, J.; Guo, X.; Peng, D.; Su, J. A rapid shoot regeneration protocol from the cotyledons of hemp (Cannabis sativa L.). Ind. Crop. Prod. 2016, 83, 61–65. [Google Scholar] [CrossRef]
- Weinberger, A.H.; Zhu, J.; Levin, J.; Barrington-Trimis, J.L.; Copeland, J.; Wyka, K.; Kim, J.H.; Goodwin, R.D. Cannabis use among US adults with anxiety from 2008 to 2017: The role of state-level cannabis legalization. Drug Alcohol Depend. 2020, 214, 108163. [Google Scholar] [CrossRef] [PubMed]
- Rabiee, R.; Lundin, A.; Agardh, E.; Hensing, G.; Allebeck, P.; Danielsson, A.-K. Cannabis use and the risk of anxiety and depression in women: A comparison of three Swedish cohorts. Drug Alcohol Depend. 2020, 216, 108332. [Google Scholar] [CrossRef] [PubMed]
- Lloyd, S.L.; Lopez-Quintero, C.; Striley, C.W. Sex differences in driving under the influence of cannabis: The role of medical and recreational cannabis use. Addict. Behav. 2020, 110, 106525. [Google Scholar] [CrossRef] [PubMed]
- Turna, J.; Balodis, I.; Munn, C.; Van Ameringen, M.; Busse, J.; MacKillop, J. Overlapping patterns of recreational and medical cannabis use in a large community sample of cannabis users. Compr. Psychiatry 2020, 102, 152188. [Google Scholar] [CrossRef] [PubMed]
- Theparat, C. New Rule Makes It Legal to Grow Hemp. Available online: https://www.bangkokpost.com/thailand/general/1845714/new-rule-makes-it-legal-to-grow-hemp (accessed on 2 October 2020).
- Koltai, H.; Namdar, D. Cannabis Phytomolecule ‘Entourage’: From Domestication to Medical Use. Trends Plant Sci. 2020, 25, 976. [Google Scholar] [CrossRef] [PubMed]
- Booth, J.K.; Bohlmann, J. Terpenes in Cannabis sativa—From plant genome to humans. Plant Sci. 2019, 284, 67. [Google Scholar] [CrossRef]
- Clarke, R.; Merlin, M.D. History of Cannabis Use for Fiber. In Cannabis; University of California Press: Berkeley, CA, USA, 2019. [Google Scholar]
- Fleming, M.; Clarke, R. Physical evidence for the antiquity of Cannabis sativa L. J. Int. Hemp Assoc. 1998, 5, 80–95. [Google Scholar]
- Li, H.-L. An archaeological and historical account of cannabis in China. Econ. Bot. 1973, 28, 437–448. [Google Scholar] [CrossRef]
- Li, H.-L. The origin and use of cannabis in Eastern Asia linguistic-cultural implications. Econ. Bot. 1974, 28, 293–301. [Google Scholar] [CrossRef]
- Hillig, K.W. A chemotaxonomic analysis of terpenoid variation in Cannabis. Biochem. Syst. Ecol. 2004, 32, 875–891. [Google Scholar] [CrossRef]
- Aluko, R.E. Chapter 7—Hemp Seed (Cannabis sativa L.) Proteins: Composition, Structure, Enzymatic Modification, and Functional or Bioactive Properties. In Sustainable Protein Sources; Nadathur, S.R., Wanasundara, J.P.D., Scanlin, L., Eds.; Academic Press: San Diego, CA, USA, 2017. [Google Scholar] [CrossRef]
- Abel, E.L. Cannabis in the Ancient World. In Marihuana; Springer: Berlin/Heidelberg, Germany, 1980; pp. 3–35. [Google Scholar]
- Bonini, S.A.; Premoli, M.; Tambaro, S.; Kumar, A.; Maccarinelli, G.; Memo, M.; Mastinu, A. Cannabis sativa: A comprehensive ethnopharmacological review of a medicinal plant with a long history. J. Ethnopharmacol. 2018, 227, 300. [Google Scholar] [CrossRef] [PubMed]
- Mediavilla, V.; Leupin, M.; Keller, A. Influence of the growth stage of industrial hemp on the yield formation in relation to certain fibre quality traits. Ind. Crop. Prod. 2001, 13, 49–56. [Google Scholar] [CrossRef]
- Cosentino, S.L.; Riggi, E.; Testa, G.; Scordia, D.; Copani, V. Evaluation of European developed fibre hemp genotypes (Cannabis sativa L.) in semi-arid Mediterranean environment. Ind. Crop. Prod. 2013, 50, 312–324. [Google Scholar] [CrossRef]
- Bewley-Taylor, D.; Blickman, T.; Jelsma, M. The Rise and Decline of Cannabis Prohibition, the History of Cannabis in the UN Drug Control System and Options for Reform; Transnational Institute (TNI): Amsterdam, The Netherlands, 2014. [Google Scholar]
- Gaoni, Y.; Mechoulam, R. Isolation, structure, and partial synthesis of an active constituent of Hashish. J. Am. Chem. Soc. 1964, 86, 1646–1647. [Google Scholar] [CrossRef]
- Mudr, P.; Et, S.; Facultatis, M. Compounds. In Acta Universitatis Palackianae Olomucensis-TOM.35; Facultatis, M., Ed.; Palacký University Olomouc: Olomouc, Czech Republic, 1964. [Google Scholar]
- Adams, R.; Pease, D.C.; Cain, C.K.; Baker, B.R.; Clark, J.H.; Wolff, H.; Wearn, R.B. Conversion of cannabidiol to a product with marihuana activity. A type reaction for synthesis of analogous substances. conversion of cannabidiol to cannabinol. J. Am. Chem. Soc. 1940, 62, 2245–2246. [Google Scholar] [CrossRef]
- Devane, W.A.; Dysarz, F.A., 3rd; Johnson, M.R.; Melvin, L.S.; Howlett, A.C. Determination and characterization of a cannabinoid receptor in rat brain. Mol. Pharmacol. 1988, 34, 605–613. [Google Scholar] [PubMed]
- Devane, A.W.; Hanus, L.; Breuer, A.; Pertwee, R.G.; Stevenson, A.L.; Griffin, G.; Gibson, D.; Mandelbaum, A.; Etinger, A.; Mechoulam, R. Isolation and structure of a brain constituent that binds to the cannabinoid receptor. Science 1992, 258, 1946–1949. [Google Scholar] [CrossRef]
- Simonsen, J.L.; Todd, A.R. 32. Cannabis indica. Part X. The essential oil from Egyptian hashish. J. Chem. Soc. 1942, 10, 188. [Google Scholar] [CrossRef]
- Hanuš, L.O.; Hod, Y. Terpenes/Terpenoids in Cannabis: Are They Important? Med. Cannabis Cannabinoids 2020, 3, 25–60. [Google Scholar] [CrossRef]
- Ben-Shabat, S.; Fride, E.; Sheskin, T.; Tamiri, T.; Rhee, M.-H.; Vogel, Z.; Bisogno, T.; De Petrocellis, L.; Di Marzo, V.; Mechoulam, R. An entourage effect: Inactive endogenous fatty acid glycerol esters enhance 2-arachidonoyl-glycerol cannabinoid activity. Eur. J. Pharmacol. 1998, 353, 23–31. [Google Scholar] [CrossRef]
- Russo, E.B. Taming THC: Potential cannabis synergy and phytocannabinoid-terpenoid entourage effects. Br. J. Pharmacol. 2011, 163, 1344–1364. [Google Scholar] [CrossRef]
- Gallily, R.; Yekhtin, Z.; Hanuš, L.O. The Anti-Inflammatory Properties of Terpenoids from Cannabis. Cannabis Cannabinoid Res. 2018, 3, 282. [Google Scholar] [CrossRef] [PubMed]
- Baron, E.P. Medicinal properties of cannabinoids, terpenes, and flavonoids in cannabis, and benefits in migraine, headache, and pain: An update on current evidence and cannabis science. Headache J. Head Face Pain 2018, 58, 1139. [Google Scholar] [CrossRef] [PubMed]
- Lewis, M.A.; Russo, E.B.; Smith, K.M. Pharmacological foundations of cannabis chemovars. Planta Med. 2017, 84, 225. [Google Scholar] [CrossRef] [PubMed]
- Horne, M.R.L. 5B-Bast fibres: Hemp cultivation and production. In Handbook of Natural Fibres, 2nd ed.; Kozłowski, R.M., Mackiewicz-Talarczyk, M., Eds.; Woodhead Publishing: Cambridge, UK, 2020. [Google Scholar] [CrossRef]
- Réquilé, S.; Le Duigou, A.; Bourmaud, A.; Baley, C. Peeling experiments for hemp retting characterization targeting biocomposites. Ind. Crop. Prod. 2018, 123, 573. [Google Scholar] [CrossRef]
- Anderson, L.C. Leaf variation among cannabis species from a controlled garden. Bot. Mus. Leafl. Harv. Univ. 1980, 28, 61–69. [Google Scholar]
- Schultes, R.E.; Klein, W.M.; Plowman, T.; Lockwood, T.E. 35. In Cannabis: An Example of Taxonomic Neglect; Walter de Gruyter GmbH: Berlin, Germany, 1975; Volume 23, pp. 21–38. [Google Scholar]
- Cai, C.; Yu, W.; Wang, C.; Liu, L.; Li, F.; Tan, Z. Green extraction of cannabidiol from industrial hemp (Cannabis sativa L.) using deep eutectic solvents coupled with further enrichment and recovery by macroporous resin. J. Mol. Liq. 2019, 287, 110957. [Google Scholar] [CrossRef]
- Wiebelhaus, N.; Hamblin, D.; Kreitals, N.M.; Almirall, J.R. Differentiation of marijuana headspace volatiles from other plants and hemp products using capillary microextraction of volatiles (CMV) coupled to gas-chromatography-mass spectrometry (GC-MS). Forensic Chem. 2016, 2, 1–8. [Google Scholar] [CrossRef]
- Hammond, C.T.; Mahlberg, P.G. Morphogenesis of capitate glandular hairs of Cannabis sativa (Cannabaceae). Am. J. Bot. 1977, 64, 1023–1031. [Google Scholar] [CrossRef]
- Potter, D. The Propagation, Characterisation and Optimisation of Cannabis sativa L. as a Phytopharmaceutical; King’s College London: London, UK, 2009. [Google Scholar]
- Livingston, S.J.; Quilichini, T.D.; Booth, J.K.; Wong, D.C.J.; Rensing, K.H.; Laflamme-Yonkman, J.; Castellarin, S.D.; Bohlmann, J.; Page, J.E.; Samuels, A.L. Cannabis glandular trichomes alter morphology and metabolite content during flower maturation. Plant J. 2019, 101, 37. [Google Scholar] [CrossRef] [PubMed]
- Gupta, A.K.; Jain, A.; Roy, P.; Singh, R. Pharmacological evaluation of Cannabis indica for their aphrodisiac potential. Int. J. Ayurvedic Med. 2020, 11, 399. [Google Scholar] [CrossRef]
- Ahmed, A.; Shapiro, D.; Su, J.; Nelson, L.P. Vaping Cannabis Butane Hash Oil Leads to Severe Acute Respiratory Distress Syndrome—A case of EVALI in a teenager with hypertrophic cardiomyopathy. J. Intensiv. Care Med. 2020. [Google Scholar] [CrossRef] [PubMed]
- Stephens, D.; Patel, J.K.; Angelo, D.; Frunzi, J. Cannabis butane hash oil dabbing induced lung injury mimicking atypical pneumonia. Cureus 2020, 12, e7033. [Google Scholar] [CrossRef] [PubMed]
- Schwachtje, J.; Baldwin, I.T. Why does herbivore attack reconfigure primary metabolism? Plant Physiol. 2008, 146, 845–851. [Google Scholar] [CrossRef] [PubMed]
- Sommano, S. Effect of Food Processing on Bioactive Compounds. In Advances in Food Science and Nutrition, 2nd ed.; Visakh, M.P., Iturriaga, L.B., Ribotta, P.D., Eds.; Scrivener Publishing LLC: Beverly, MA, USA, 2013; pp. 361–390. [Google Scholar] [CrossRef]
- Sommano, S.R.; Caffin, N.; Kerven, G. Screening for antioxidant activity, phenolic content, and flavonoids from Australian native food plants. Int. J. Food Prop. 2012, 16, 1394–1406. [Google Scholar] [CrossRef]
- Sunanta, P.; Chung, H.-H.; Kunasakdakul, K.; Ruksiriwanich, W.; Jantrawut, P.; Hongsibsong, S.; Sommano, S.R. Genomic relationship and physiochemical properties among raw materials used for Thai black garlic processing. Food Sci. Nutr. 2020, 8, 4534–4545. [Google Scholar] [CrossRef]
- Wisetkomolmat, J.; Suppakittpaisarn, P.; Sommano, S.R. Detergent plants of northern Thailand: Potential sources of natural saponins. Resources 2019, 8, 10. [Google Scholar] [CrossRef]
- Tangpao, T.; Chung, H.-H.; Sommano, S.R. Aromatic profiles of essential oils from five commonly used Thai basils. Foods 2018, 7, 175. [Google Scholar] [CrossRef]
- Ross, S.A.; ElSohly, M.A. The volatile oil composition of fresh and air-dried buds of Cannabis sativa. J. Nat. Prod. 1996, 59, 49–51. [Google Scholar] [CrossRef]
- Turner, C.E.; ElSohly, M.A.; Boeren, E.G. Constituents of Cannabis sativa L. XVII. A review of the natural constituents. J. Nat. Prod. 1980, 43, 169–234. [Google Scholar] [CrossRef] [PubMed]
- Wanas, A.S.; Radwan, M.M.; Chandra, S.; Lata, H.; Mehmedic, Z.; Ali, A.; Baser, K.; Demirci, B.; ElSohly, M.A. Chemical composition of volatile oils of fresh and air-dried buds of cannabis chemovars, their insecticidal and repellent activities. Nat. Prod. Commun. 2020, 15, 1934578X20926729. [Google Scholar] [CrossRef]
- Rice, S.; Koziel, J.A. Characterizing the smell of marijuana by odor impact of volatile compounds: An application of simultaneous chemical and sensory analysis. PLoS ONE 2015, 10, e0144160. [Google Scholar] [CrossRef]
- Ternelli, M.; Brighenti, V.; Anceschi, L.; Poto, M.; Bertelli, D.; Licata, M.; Pellati, F. Innovative methods for the preparation of medical cannabis oils with a high content of both cannabinoids and terpenes. J. Pharm. Biomed. Anal. 2020, 186, 113296. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.-T.; Ashworth, K.; Wiedinmyer, C.; Ortega, J.; Harley, P.C.; Rasool, Q.Z.; Vizuete, W. Ambient measurements of monoterpenes near Cannabis cultivation facilities in Denver, Colorado. Atmos. Environ. 2020, 232, 117510. [Google Scholar] [CrossRef]
- Abdollahi, M.; Sefidkon, F.; Calagari, M.; Mousavi, A.; Mahomoodally, M.F. Impact of four hemp (Cannabis sativa L.) varieties and stage of plant growth on yield and composition of essential oils. Ind. Crop. Prod. 2020, 155, 112793. [Google Scholar] [CrossRef]
- Sriwichai, T.; Junmahasathien, T.; Sookwong, P.; Potapohn, N.; Sommano, S.R. Evaluation of the optimum harvesting maturity of makhwaen fruit for the perfumery industry. Agriculture 2019, 9, 78. [Google Scholar] [CrossRef]
- Booth, J.K.; Page, J.E.; Bohlmann, J. Terpene synthases from Cannabis sativa. PLoS ONE 2017, 12, e0173911. [Google Scholar] [CrossRef]
- Nagegowda, D.A.; Gupta, P. Advances in biosynthesis, regulation, and metabolic engineering of plant specialized terpenoids. Plant Sci. 2020, 294, 110457. [Google Scholar] [CrossRef]
- Chen, F.; Tholl, D.; Bohlmann, J.; Pichersky, E. The family of terpene synthases in plants: A mid-size family of genes for specialized metabolism that is highly diversified throughout the kingdom. Plant J. 2011, 66, 212–229. [Google Scholar] [CrossRef]
- Fellermeier, M.; Eisenreich, W.; Bacher, A.; Zenk, M.H. Biosynthesis of cannabinoids Incorporation experiments with 13C-labeled glucoses. JBIC J. Biol. Inorg. Chem. 2001, 268, 1596–1604. [Google Scholar] [CrossRef] [PubMed]
- Aliferis, K.A.; Bernard-Perron, D. Cannabinomics: Application of metabolomics in cannabis (Cannabis sativa L.) research and development. Front. Plant Sci. 2020, 11, 11. [Google Scholar] [CrossRef] [PubMed]
- Masoud, A.N.; Doorenbos, N.J. Mississippi-Grown Cannabis sativa L. III: Cannabinoid and cannabinoid acid content. J. Pharm. Sci. 1973, 62, 313–315. [Google Scholar] [CrossRef]
- Grijó, D.R.; Osorio, I.A.V.; Cardozo-Filho, L. Supercritical extraction strategies using CO2 and ethanol to obtain cannabinoid compounds from Cannabis hybrid flowers. J. CO2 Util. 2018, 28, 174. [Google Scholar] [CrossRef]
- Miyazawa, M.; Yamafuji, C. Inhibition of acetylcholinesterase activity by bicyclic monoterpenoids. J. Agric. Food Chem. 2005, 53, 1765–1768. [Google Scholar] [CrossRef] [PubMed]
- Gaggiotti, S.; Palmieri, S.; Pelle, F.D.; Sergi, M.; Cichelli, A.; Mascini, M.; Compagnone, D. Piezoelectric peptide-hpDNA based electronic nose for the detection of terpenes; Evaluation of the aroma profile in different Cannabis sativa L. (hemp) samples. Sens. Actuators B Chem. 2020, 308, 127697. [Google Scholar] [CrossRef]
- Sriwichai, T.; Sookwong, P.; Siddiqui, M.W.; Sommano, S.R. Aromatic profiling of Zanthoxylum myriacanthum (makwhaen) essential oils from dried fruits using different initial drying techniques. Ind. Crop. Prod. 2019, 133, 284. [Google Scholar] [CrossRef]
- Maayah, Z.H.; Takahara, S.; Ferdaoussi, M.; Dyck, J.R. The molecular mechanisms that underpin the biological benefits of full-spectrum cannabis extract in the treatment of neuropathic pain and inflammation. Biochim. Biophys. Acta (BBA) Mol. Basis Dis. 2020, 1866, 165771. [Google Scholar] [CrossRef]
- Rao, V.S.N.; Menezes, A.M.S.; Viana, G.S.B. Effect of myrcene on nociception in mice. J. Pharm. Pharmacol. 1990, 42, 877–878. [Google Scholar] [CrossRef]
- Meschler, J. Thujone exhibits low affinity for cannabinoid receptors but fails to evoke cannabimimetic responses. Pharmacol. Biochem. Behav. 1999, 62, 473–480. [Google Scholar] [CrossRef]
- Gilbert, A.N.; DiVerdi, J.A. Consumer perceptions of strain differences in Cannabis aroma. PLoS ONE 2018, 13, e0192247. [Google Scholar] [CrossRef] [PubMed]
- Brenneisen, R. Chemistry and Analysis of Phytocannabinoids and Other Cannabis Constituents. In Marijuana and the Cannabinoids; ElSohly, M.A., Ed.; Humana Press: Totowa, NJ, USA, 2007. [Google Scholar] [CrossRef]
- Fischedick, J.T.; Hazekamp, A.; Erkelens, T.; Choi, Y.H.; Verpoorte, R. Metabolic fingerprinting of Cannabis sativa L., cannabinoids and terpenoids for chemotaxonomic and drug standardization purposes. Phytochemistry 2010, 71, 2058–2073. [Google Scholar] [CrossRef] [PubMed]
- Brown, A.K.; Xia, Z.; Bulloch, P.; Idowu, I.; Francisco, O.; Stetefeld, J.; Stout, J.; Zimmer, J.; Marvin, C.; Letcher, R.J.; et al. Validated quantitative cannabis profiling for Canadian regulatory compliance—Cannabinoids, aflatoxins, and terpenes. Anal. Chim. Acta 2019, 1088, 79. [Google Scholar] [CrossRef] [PubMed]
- Shapira, A.; Berman, P.; Futoran, K.; Guberman, O.; Meiri, D. Tandem Mass Spectrometric Quantification of 93 terpenoids in cannabis using static headspace injections. Anal. Chem. 2019, 91, 11425. [Google Scholar] [CrossRef]
- Rice, S.; Koziel, J.A. The relationship between chemical concentration and odor activity value explains the inconsistency in making a comprehensive surrogate scent training tool representative of illicit drugs. Forensic Sci. Int. 2015, 257, 257–270. [Google Scholar] [CrossRef]
- Kabir, A.; Holness, H.; Furton, K.G.; Almirall, J.R. Recent advances in micro-sample preparation with forensic applications. TrAC Trends Anal. Chem. 2013, 45, 264–279. [Google Scholar] [CrossRef]
- Calvi, L.; Pentimalli, D.; Panseri, S.; Giupponi, L.; Gelmini, F.; Beretta, G.; Vitali, D.; Bruno, M.; Zilio, E.; Pavlovic, R.; et al. Comprehensive quality evaluation of medical Cannabis sativa L. inflorescence and macerated oils based on HS-SPME coupled to GC–MS and LC-HRMS (q-exactive orbitrap®) approach. J. Pharm. Biomed. Anal. 2018, 150, 208. [Google Scholar] [CrossRef]
- Koltai, H.; Poulin, P.; Namdar, D. Promoting cannabis products to pharmaceutical drugs. Eur. J. Pharm. Sci. 2019, 132, 118. [Google Scholar] [CrossRef]
- Koby, M. How Terpenes Could Revolutionize the Cannabis Industry as We Know It in Innovators; Entrepreneur Media, Inc.: Irvine, CA, USA, 2020. [Google Scholar]
- King, J.W. The relationship between cannabis/hemp use in foods and processing methodology. Curr. Opin. Food Sci. 2019, 28, 32. [Google Scholar] [CrossRef]
- Charlebois, S.; Somogyi, S.; Sterling, B. Cannabis-infused food and Canadian consumers’ willingness to consider “recreational” cannabis as a food ingredient. Trends Food Sci. Technol. 2018, 74, 112. [Google Scholar] [CrossRef]
- Khan, R.S.; Grigor, J.V.; Winger, R.; Win, A. Functional food product development—Opportunities and challenges for food manufacturers. Trends Food Sci. Technol. 2013, 30, 27–37. [Google Scholar] [CrossRef]
- Benelli, G.; Pavela, R.; Petrelli, R.; Cappellacci, L.; Santini, G.; Fiorini, D.; Sut, S.; Dall’Acqua, S.; Canale, A.; Maggi, F. The essential oil from industrial hemp (Cannabis sativa L.) by-products as an effective tool for insect pest management in organic crops. Ind. Crop. Prod. 2018, 122, 308. [Google Scholar] [CrossRef]
- Hanif, M.A.; Nawaz, H.; Naz, S.; Mukhtar, R.; Rashid, N.; Bhatti, I.A.; Saleem, M. Raman spectroscopy for the characterization of different fractions of hemp essential oil extracted at 130 °C using steam distillation method. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2017, 182, 168. [Google Scholar] [CrossRef] [PubMed]
- Fiorini, D.; Scortichini, S.; Bonacucina, G.; Greco, N.G.; Mazzara, E.; Petrelli, R.; Torresi, J.; Maggi, F.; Cespi, M. Cannabidiol-enriched hemp essential oil obtained by an optimized microwave-assisted extraction using a central composite design. Ind. Crop. Prod. 2020, 154, 112688. [Google Scholar] [CrossRef]
- Blake, A.; Nahtigal, I. The evolving landscape of cannabis edibles. Curr. Opin. Food Sci. 2019, 28, 25. [Google Scholar] [CrossRef]
- Baldino, L.; Scognamiglio, M.; Reverchon, E. Supercritical fluid technologies applied to the extraction of compounds of industrial interest from Cannabis sativa L. and to their pharmaceutical formulations: A review. J. Supercrit. Fluids 2020, 165, 104960. [Google Scholar] [CrossRef]
- Naz, S.; Hanif, M.A.; Bhatti, H.N.; Ansari, T.M. Impact of supercritical fluid extraction and traditional distillation on the isolation of aromatic compounds from Cannabis indica and Cannabis sativa. J. Essent. Oil Bear. Plants 2017, 20, 175. [Google Scholar] [CrossRef]


| Characteristics | Cannabis (Marijuana) | Hemp |
|---|---|---|
| Genus | Cannabis sativa L. | Cannabis sativa L. |
| Sub variety | Indica | sativa |
| Utilized organs | leaves, flowers, stems and seeds containing trichomes | stem |
| Level of psychoactive THC | High (>1%/DW) | Low |
| Medicinal CBD | Can be high | Can be high |
| Leaf | Broad, darker leaf color | Thinner and greenish |
| Content of terpene (Rosin) | High (gluey) | Low |
| Cannabis Family (commercial)* | Stains (Commercial Names) | Seed Types | Level of Cannabinoid THC (Max = 5) | Chemotypic Catagories 1 | Descriptive Sensorial Categories 1 | |||||
| (i) β-myrcene | (ii) α- and β-pinene | (iii) β-caryophyllene and Limonene | (iv) β-caryophyllene | (v) Terpinolene | (i) Earthy, Woody and Herbal | (ii) Citrus, Lemon, Sweet and Pungent | ||||
| Afghani Kush | Banana Blaze® | F | 3 |  | ||||||
| Auto Banana Blaze® | F, A | 5 |  | |||||||
| Master Kush | F | 3 |  | |||||||
| Night Queen® | F | 4 |  | |||||||
| Blue family | Auto Blue Berry® | F, A | 3 |  | ||||||
| Auto Black Berry Kush® | F, A | 4 |  | |||||||
| Blue Auto Mazar® | F, A | 4 |  |  | ||||||
| CBD rich | CBD Charlotte’s Angel® | F | 1 |  |  | |||||
| CBD Skunk Haze® | F | 2 |  |  | ||||||
| Classics | C-vibez® | F | 5 |  | ||||||
| Mokum’s Tulip® | F | 4 |  | |||||||
| Auto Ultimate® | F, A | 4 |  |  | ||||||
| Think Fast® | F | 3 |  | |||||||
| Auto Cinderella Jack® | F, A | 5 |  |  | ||||||
| Outlaw Amnesia® | F | 4 |  |  | ||||||
| Auto Xtreme® | F, A | 4 |  |  | ||||||
| Auto White Widow® | F, A | 4 |  |  | ||||||
| Dutch outdoor | Frisian Dew® | F | 2 |  |  | |||||
| Purple N0. 1® | R, F | 2 |  |  | ||||||
| Auto Durban Poison® | F, A | 2 |  |  | ||||||
| High altitude | Snow Bud® | F | 2 |  |  | |||||
| Orange family | Passion Fruit® | F | 4 |  |  | |||||
| US special | Sugar Bomb Punch® | F | 5 |  |  | |||||
| Kerosene Krash® | F | 5 |  | |||||||
| Meringue® | F | 5 |  | |||||||
| Hifi 4G® | F | 4 |  |  | ||||||
| Auto lemon Kix® | F, A | 5 |  |  | ||||||
| Bubba island Kush® | F | 4 |  | |||||||
| Auto Glueberry O.G.® | F, A | 4 |  |  | ||||||
 hop;
hop;  pine;
pine;  lime;
lime;  spice and
spice and  orange peel were according to Russo [28].
orange peel were according to Russo [28].Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sommano, S.R.; Chittasupho, C.; Ruksiriwanich, W.; Jantrawut, P. The Cannabis Terpenes. Molecules 2020, 25, 5792. https://doi.org/10.3390/molecules25245792
Sommano SR, Chittasupho C, Ruksiriwanich W, Jantrawut P. The Cannabis Terpenes. Molecules. 2020; 25(24):5792. https://doi.org/10.3390/molecules25245792
Chicago/Turabian StyleSommano, Sarana Rose, Chuda Chittasupho, Warintorn Ruksiriwanich, and Pensak Jantrawut. 2020. "The Cannabis Terpenes" Molecules 25, no. 24: 5792. https://doi.org/10.3390/molecules25245792
APA StyleSommano, S. R., Chittasupho, C., Ruksiriwanich, W., & Jantrawut, P. (2020). The Cannabis Terpenes. Molecules, 25(24), 5792. https://doi.org/10.3390/molecules25245792










