A One-Pot Three-Component Synthesis and Investigation of the In Vitro Mechanistic Anticancer Activity of Highly Functionalized Spirooxindole-Pyrrolidine Heterocyclic Hybrids
Abstract
1. Introduction
2. Results and Discussion
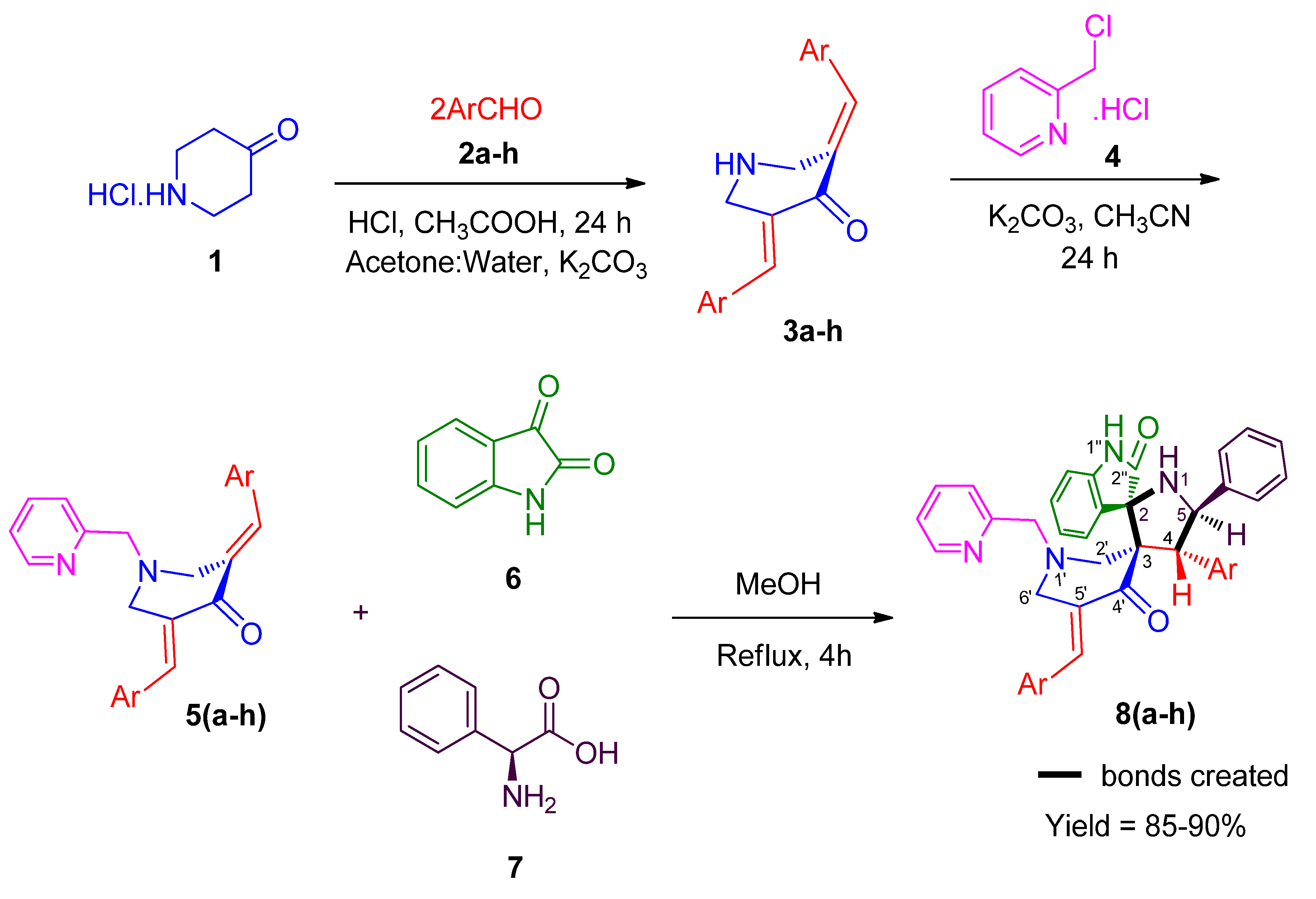
2.1. Chemistry
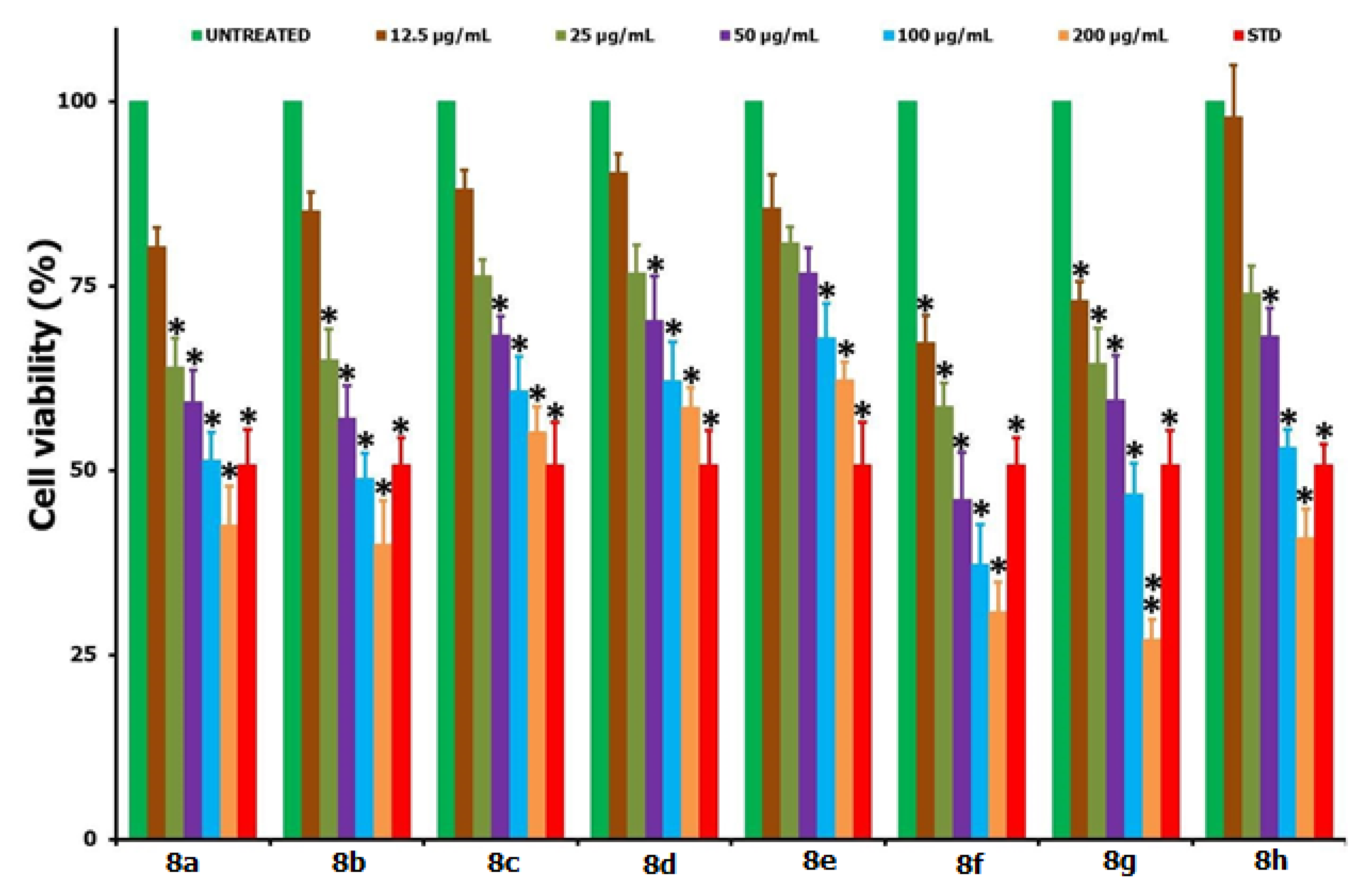
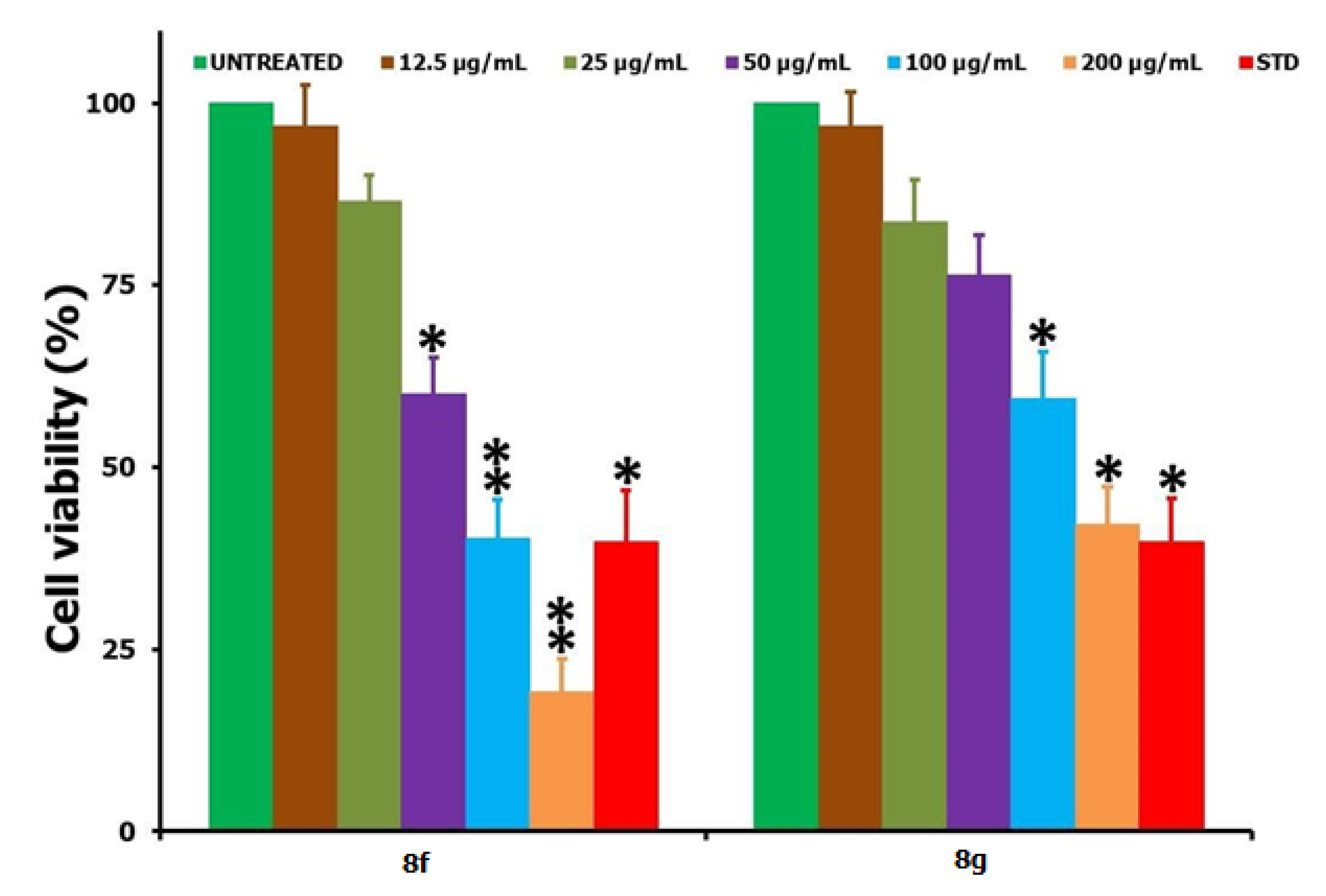
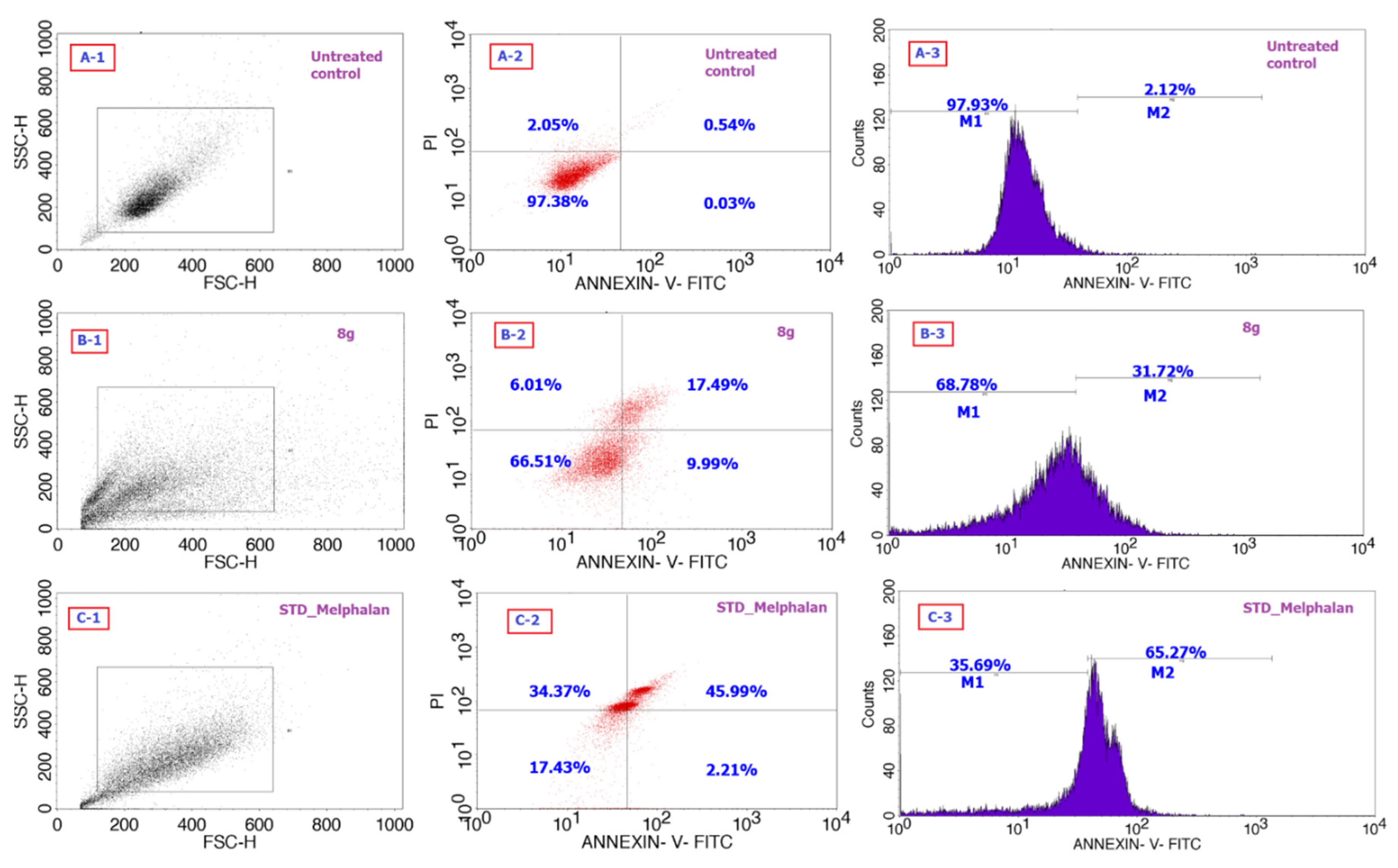
2.2. Biology
3. Materials and Methods
3.1. 4-(aryl)-5-phenylpyrrolo(spiro[2.3″]-oxindole)-spiro[3.3′]-5′-(2-arylmethylidene)-1′-N-(pyridinylmethyl)piperidin-4′-one 8 (a-h)
3.2. 4,5-Diphenylpyrrolo(spiro[2.3″]-oxindole)-spiro-[3.3′]-5′-(phenylmethylidene)-1′-N-(pyridinylmethyl)piperidin-4′-one (8a)
3.3. 4-(2-Methylphenyl)-5-phenylpyrrolo(spiro[2.3″]-oxindole)spiro[3.3′]-5′-(2-methylphenyl-methylidene)-1′-N-(pyridinylmethyl)piperidin-4′-one (8b)
3.4. 4-(2-Methoxyphenyl)-5-phenylpyrrolo(spiro[2.3″]-oxindole)spiro[3.3′]-5′-(2-methoxy-phenylmethylidene) 1′-N-(pyridinylmethyl)piperidin-4′-one (8c)
3.5. 4-(3-Nitrophenyl)-5-phenylpyrrolo(spiro[2.3″]-oxindole)spiro[3.3′]-5′-(3-nitrophenyl methylidene)-1′-N-(pyridinylmethyl)piperidin-4′-one (8d)
3.6. 4-(4-Methylphenyl)-5-phenylpyrrolo(spiro[2.3″]-oxindole)spiro[3.3′]-5′-(4-methylphenyl-methylidene)-1′-N-(pyridinylmethyl)piperidin-4′-one (8e)
3.7. 4-(4-Chlorophenyl)-5-phenylpyrrolo(spiro[2.3″]-oxindole)spiro[3.3′]-5′-(4-chlorophenyl-methylidene)-1′-N-(pyridinylmethyl)piperidin-4′-one (8f)
3.8. 4-(4-Fluorophenyl)-5-phenylpyrrolo(spiro[2.3″]-oxindole)spiro[3.3′]-5′-(4-fluorophenyl-methylidene)-1′-N-(pyridinylmethyl)piperidin-4′-one (8g)
3.9. 4-(2,4-Dichlorophenyl)-5-phenylpyrrolo(spiro[2.3″]-oxindole)spiro[3.3′]-5′-(2,4-dichloro-phenylmethylidene)-1′-N-(pyridinylmethyl)piperidin-4′-one (8h)
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Irigaray, P.; Belpomme, D. Basic properties and molecular mechanisms of exogenous chemical carcinogens. Carcinog. 2009, 31, 135–148. [Google Scholar]
- Gupta, A.; Kumar, B.S.; Negi, A.S. Current status on development of steroids as anticancer agents. J. Steroid Biochem. Mol. Biol. 2013, 137, 242–270. [Google Scholar] [PubMed]
- Choi, S.K. Synthetic Multivalent Molecules: Concepts and Biomedical Applications; John Wiley & Sons: New York City, NY, USA, 1807. [Google Scholar]
- Nakagawa-Goto, K.; Nakamura, S.; Bastow, K.F.; Nyarko, A.K.; Peng, C.-Y.; Lee, F.-Y.; Lee, K.-H.; Lee, F.-C. Antitumor agents. 256. Conjugation of paclitaxel with other antitumor agents: Evaluation of novel conjugates as cytotoxic agents. Bioorg. Med. Chem. Lett. 2007, 17, 2894–2898. [Google Scholar] [PubMed]
- Mohammadi, M.R.; Nojoomi, A.; Mozafari, M.; Dubnika, A.; Inayathullah, M.; Rajadas, J. Nanomaterials engineering for drug delivery: a hybridization approach. J. Mater. Chem. B 2017, 5, 3995–4018. [Google Scholar]
- Tatsuzaki, J.; Taniguchi, M.; Bastow, K.F.; Nakagawa-Goto, K.; Morris-Natschke, S.L.; Itokawa, H.; Baba, K.; Lee, K.H. Antitumor Agents 255: Novel Glycyrrhetinic Acid-Dehydrozingerone Conjugates as Cytotoxic Agents. Bioorg. Med. Chem. 2007, 15, 6193–6199. [Google Scholar]
- Nakagawa-Goto, K.; Yamada, K.; Nakamura, S.; Chen, T.-H.; Chiang, P.-C.; Bastow, K.F.; Wang, S.C.; Spohn, B.; Hung, M.C.; Lee, F.Y.; et al. Antitumor agents. 258. Syntheses and evaluation of dietary antioxidant – taxoid conjugates as novel cytotoxic agents. Bioorg. Med. Chem. Lett. 2007, 17, 5204–5209. [Google Scholar]
- Shi, B.; Zhang, Z.; Jin, Q.; Wang, Z.; Tang, J.; Xu, G.; Zhu, T.; Gong, X.-Q.; Tang, X.; Zhao, C. Selective tracking of ovarian-cancer-specific γ-glutamyltranspeptidase using a ratiometric two-photon fluorescent probe. J. Mater. Chem. B 2018, 6, 7439–7443. [Google Scholar]
- Samala, G.; Devi, P.B.; Nallangi, R.; Sridevi, J.P.; Saxena, S.; Yogeeswari, P.; Sriram, D. Development of novel tetrahydrothieno[2–c]pyridine-3-carboxamide based Mycobacterium tuberculosis pantothenate synthetase inhibitors: Molecular hybridization from known antimycobacterial leads. Bioorg. Med. Chem. 2014, 22, 1938–1947. [Google Scholar]
- Sang, Z.; Li, Y.; Qiang, X.; Xiao, G.; Liu, Q.; Tan, Z.; Deng, Y. Multifunctional scutellarin-rivastigmine hybrids with cholinergic, antioxidant, biometal chelating and neuroprotective properties for the treatment of Alzheimer’s disease. Bioorg. Med. Chem. 2015, 23, 668–680. [Google Scholar]
- Kamiński, K.; Rapacz, A.; Łuszczki, J.J.; Latacz, G.; Obniska, J.; Kieć-Kononowicz, K.; Filipek, B. Design, synthesis and biological evaluation of new hybrid anticonvulsants derived from N-benzyl-2-(2,5-dioxopyrrolidin-1-yl)propanamide and 2-(2,5-dioxopyrrolidin-1-yl)butanamide derivatives. Bioorg. Med. Chem. 2015, 23, 2548–2561. [Google Scholar]
- Yang, X.; Qian, Y. A NIR facile, cell-compatible fluorescent sensor for glutathione based on Michael addition induced cascade spirolactam opening and its application in hepatocellular carcinoma. J. Mater. Chem. B 2018, 6, 7486–7494. [Google Scholar]
- Liang, C.; Zhang, X.; Wang, Z.; Wang, W.; Yang, M.-S.; Dong, X. Organic/inorganic nanohybrids rejuvenate photodynamic cancer therapy. J. Mater. Chem. B 2020, 8, 4748–4763. [Google Scholar]
- Wan, Z.-Y.; Tao, Y.; Wang, Y.-F.; Mao, T.-Q.; Yin, H.; Chen, F.-E.; Piao, H.-R.; Clercq, E.D.; Daelemans, D.; Pannecouque, C. Hybrid chemistry. Part 4: Discovery of etravirine-VRX-480773 hybrids as potent HIV-1 non-nucleoside reverse transcriptase inhibitors. Bioorg. Med. Chem. 2015, 23, 4248–4255. [Google Scholar]
- Zhao, Y.; Su, J.; Goto, M.; Morris-Natschke, S.L.; Li, Y.; Zhao, Q.-S.; Yao, Z.-J.; Lee, K.-H. Dual-Functional abeo-Taxane Derivatives Destabilizing Microtubule Equilibrium and Inhibiting NF-κB Activation. J. Med. Chem. 2013, 56, 4749–4757. [Google Scholar]
- Zhang, Y.; Wei, B.-W.; Lin, H.; Zhang, L.; Liu, J.-X.; Luo, H.-Q.; Fan, X.-L. “On water” direct catalytic vinylogous Henry (nitroaldol) reactions of isatins for the efficient synthesis of isoxazole substituted 3-hydroxyindolin-2-ones. Green Chem. 2015, 17, 3266–3270. [Google Scholar]
- AlSalhi, M.S.; Elangovan, K.; Ranjitsingh, A.J.A.; Murali, P.; Devanesan, S. Synthesis of silver nanoparticles using plant derived 4-N-methyl benzoic acid and evaluation of antimicrobial, antioxidant and antitumor activity. Saudi J. Biol. Sci. 2019, 26, 970–978. [Google Scholar]
- Devanesan, S.; Ponmurugan, K.; AlSalhi, M.S.; Dhabi, N.A.A. Cytotoxic and Antimicrobial Efficacy of Silver Nanoparticles Synthesized Using a Traditional Phytoproduct, Asafoetida Gum. Int. J. Nanomed. 2020, 15, 4351–4362. [Google Scholar]
- MacLean, D.; Schullek, J.R.; Murphy, M.M.; Ni, Z.-J.; Gordon, E.M.; Gallop, M.A. Encoded combinatorial chemistry: Synthesis and screening of a library of highly functionalized pyrrolidines. Proc. Natl. Acad. Sci. USA 1997, 94, 2805–2810. [Google Scholar]
- Hanessian, S.; Bayrakdarian, M. Pyrrolidine as a cogwheel-like scaffold for the deployment of diverse functionality through cycloaddition reactions of metallo-1,3-dipoles in aqueous media. Bioorganic Med. Chem. Lett. 2000, 10, 427–431. [Google Scholar]
- Hanessian, S.; Bayrakdarian, M. Solution- and solid-phase asymmetric synthesis of substituted N-hydroxypyrrolidine dicarboxylic acids. Tetrahedron Lett. 2002, 43, 9441–9444. [Google Scholar]
- Lo, M.M.-C.; Neumann, C.S.; Nagayama, S.; Perlstein, E.O.; Schreiber, S.L. A Library of Spirooxindoles Based on a Stereoselective Three-Component Coupling Reaction. J. Am. Chem. Soc. 2004, 126, 16077–16086. [Google Scholar] [CrossRef]
- Han, W.-Y.; Zhao, J.-Q.; Zuo, J.; Xu, X.-Y.; Zhang, X.-M.; Yuan, W.-C. Recent Advances of α-Isothiocyanato Compounds in the Catalytic Asymmetric Reaction. Adv. Synth. Catal. 2015, 357, 3007–3031. [Google Scholar] [CrossRef]
- Zhou, J.; Yu, J.-S.; Zhou, F.; Liu, Y. A Journey in the Catalytic Synthesis of 3-Substituted 3-Aminooxindoles. Synlett 2015, 26, 2491–2504. [Google Scholar] [CrossRef]
- Das, U.; Pati, H.N.; Sakagami, H.; Hashimoto, K.; Kawase, M.; Balzarini, J.; De Clercq, E.; Dimmock, J.R. 3,5-Bis(benzylidene)-1-[3-(2-hydroxyethylthio)propanoyl]piperidin-4-ones: A Novel Cluster of Potent Tumor-Selective Cytotoxins. J. Med. Chem. 2011, 54, 3445–3449. [Google Scholar] [CrossRef][Green Version]
- Dimmock, J.R.; Maniyan, P.P.; Ramanan, N.P.; Adil, J.N.; Narasimhan, L.M.; Gordon, A.Z.; Quail, J.W.; Oloo, E.O.; Kraatz, H.B.; Prisciak, J.S.; et al. A conformational and structure-activity relationship study of cytotoxic 3, 5-bis (arylidene)-4-piperidones and related N-acryloyl analogues. J. Med. Chem. 2001, 44, 586–593. [Google Scholar] [CrossRef]
- Al-Thamili, D.M.; Almansour, A.I.; Arumugam, N.; Mohammad, F.; Kumar, R.S. Functionalized N-Pyridinylmethyl Engrafted Bisarylmethylidenepyridinones as Anticancer Agents. Processes 2020, 8, 1154. [Google Scholar] [CrossRef]
- Al-thamili, D.M.; Almansour, A.I.; Arumugam, N.; Kansız, S.; Dege, N.; Soliman, S.M.; Azam, M.; Kumar, R.S. Highly functionalized N-1-(2-pyridinylmethyl)-3,5-bis[(E)-arylmethylidene]tetrahydro-4(1H)-pyridinones: Synthesis, characterization, crystal structure and DFT studies. J. Mol. Struct. 2020, 1222. [Google Scholar] [CrossRef]



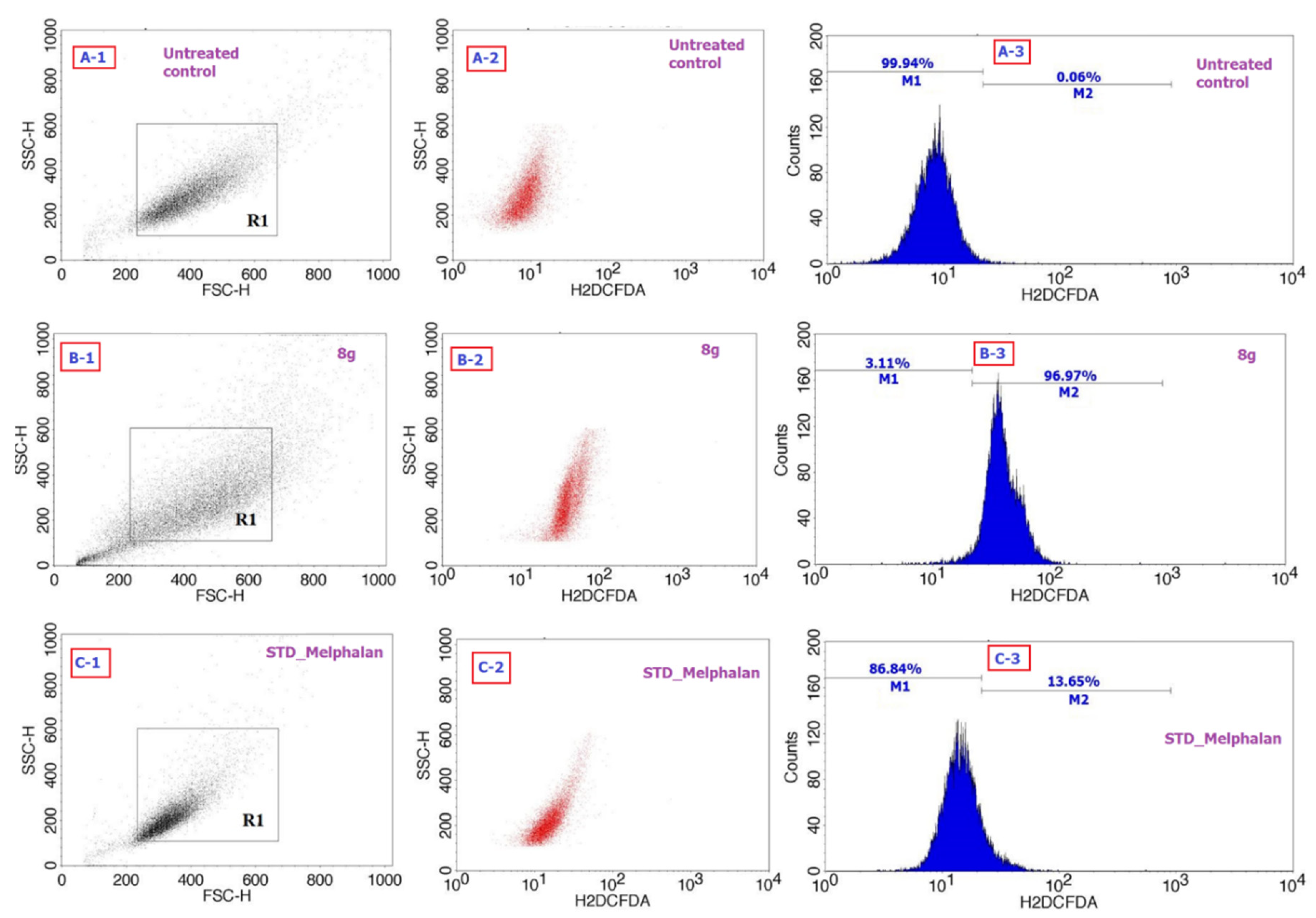

| Entry | Compound | IC50 Values (µg/mL) HepG2 Cells | IC50 Values (µg/mL) L929 Cells | |
|---|---|---|---|---|
| 24 h Treatment | 48 h Treatment | 48 h Treatment | ||
| 1 |  | 105.78 ± 2.1 | 27.43 ± 4.1 | NA |
| 2 | 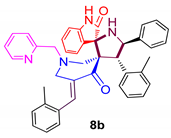 | 91.5 ± 2.1 | 31.21 ± 5.3 | NA |
| 3 | 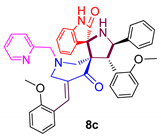 | 274.61 ± 2.1 | 52.14 ± 6.7 | NA |
| 4 | 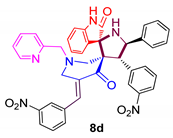 | 352.86 ± 2.1 | 53.04 ± 5.5 | NA |
| 5 | 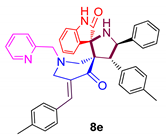 | 897.42 ± 2.1 | 62.25 ± 5.8 | NA |
| 6 | 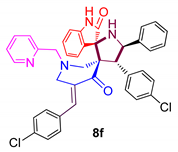 | 43.46 ± 2.1 | 25.82 ± 3.2 | 72.12 ± 3.3 |
| 7 | 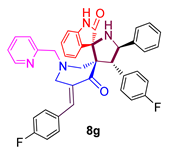 | 65.18 ± 2.1 | 12.79 ± 2.6 | 153.89 ± 4.2 |
| 8 | 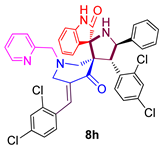 | 120.76 ± 2.1 | 41.32 ± 3.2 | NA |
Sample Availability: Samples of the compounds 8 (a–h) are available from the authors. | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kumar, R.S.; M. Al-thamili, D.; Almansour, A.I.; Arumugam, N.; Mohammad, F. A One-Pot Three-Component Synthesis and Investigation of the In Vitro Mechanistic Anticancer Activity of Highly Functionalized Spirooxindole-Pyrrolidine Heterocyclic Hybrids. Molecules 2020, 25, 5581. https://doi.org/10.3390/molecules25235581
Kumar RS, M. Al-thamili D, Almansour AI, Arumugam N, Mohammad F. A One-Pot Three-Component Synthesis and Investigation of the In Vitro Mechanistic Anticancer Activity of Highly Functionalized Spirooxindole-Pyrrolidine Heterocyclic Hybrids. Molecules. 2020; 25(23):5581. https://doi.org/10.3390/molecules25235581
Chicago/Turabian StyleKumar, Raju Suresh, Dhaifallah M. Al-thamili, Abdulrahman I. Almansour, Natarajan Arumugam, and Faruq Mohammad. 2020. "A One-Pot Three-Component Synthesis and Investigation of the In Vitro Mechanistic Anticancer Activity of Highly Functionalized Spirooxindole-Pyrrolidine Heterocyclic Hybrids" Molecules 25, no. 23: 5581. https://doi.org/10.3390/molecules25235581
APA StyleKumar, R. S., M. Al-thamili, D., Almansour, A. I., Arumugam, N., & Mohammad, F. (2020). A One-Pot Three-Component Synthesis and Investigation of the In Vitro Mechanistic Anticancer Activity of Highly Functionalized Spirooxindole-Pyrrolidine Heterocyclic Hybrids. Molecules, 25(23), 5581. https://doi.org/10.3390/molecules25235581






