Characterization of Phenolic Compounds, Vitamin E and Fatty Acids from Monovarietal Virgin Olive Oils of “Picholine marocaine” Cultivar
Abstract
1. Introduction
2. Results and Discussion
3. Materials and Methods
3.1. Chemicals and Reagents
3.2. Sample Collection
3.3. Determination of Acidity
3.4. Phenolic Compounds Extraction
3.5. HPLC-PDA-ESI-MS Analysis of Phenolic Compounds
3.6. NP-HPLC/FLD Analysis of Tocopherols and Tocotrienols
3.7. Fatty Acid Methyl Esters Extraction (FAMEs)
3.8. GC–MS/FID Analysis of FAMEs
3.9. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References and Note
- Moroccan Ministry of Agriculture and Marine Fisheries. Statistiques du Secteur Oléicole Marocain. Available online: http://www.agriculture.gov.ma/pages/rapports-statistiques/campagne-agricole-2015-2016 (accessed on 5 October 2020).
- International Olive Council (IOC). The Olive Grove in Morocco. Off. J. Int. Olive Counc. 2019, 125, 1–89. Available online: www.internationaloliveoil.org (accessed on 12 October 2020).
- Azaizeh, H.; Abu Tayeh, H.N.; Gerchman, Y. Valorisation of Olive Oil Industry Solid Waste and Production of Ethanol and High Value-Added Biomolecules; Elsevier: Amsterdam, The Netherlands, 2020; ISBN 9780128179512. [Google Scholar]
- Azbar, N.; Bayram, A.; Filibeli, A.; Muezzinoglu, A.; Sengul, F.; Ozer, A. A review of waste management options in olive oil production. Crit. Rev. Environ. Sci. Technol. 2004, 34, 209–247. [Google Scholar] [CrossRef]
- Campagne Agricole 2015–2016. Département de L’agriculture-Ministère de L’agriculture, de la pêche Maritime, du Développement Rural et des Eaux et Forêts. Available online: http://www.agriculture.gov.ma/pages/rapports-statistiques/campagne-agricole-2015-2016 (accessed on 13 September 2020).
- Boskou, D. Olive and Olive Oil Bioactive Constituents; Elsevier: Amserdam, The Netherlands, 2015; ISBN 9781630670412. [Google Scholar]
- Bendini, A.; Cerretani, L.; Carrasco-Pancorbo, A.; Gómez-Caravaca, A.M.; Segura-Carretero, A.; Fernández-Gutiérrez, A.; Lercker, G. Phenolic molecules in virgin olive oils: A survey of their sensory properties, health effects, antioxidant activity and analytical methods. An overview of the last decade. Molecules 2007, 12, 1679–1719. [Google Scholar] [CrossRef] [PubMed]
- Rodis, P.S.; Karathanos, V.T.; Mantzavinou, A. Partitioning of olive oil antioxidants between oil and water phases. J. Agric. Food Chem. 2002, 50, 596–601. [Google Scholar] [CrossRef] [PubMed]
- Allaoui, S.; Bennani, M.N.; Ziyat, H.; Qabaqous, O.; Tijani, N.; Ittobane, N.; Barbouchi, M.; Bouymajane, A.; Filali, F.R. Antioxidant and Antimicrobial Activity of Polyphenols Extracted after Adsorption onto Natural Clay “Ghassoul.”. J. Chem. 2020, 2020, 1–6. [Google Scholar] [CrossRef]
- Dehmani, Y.; Ed-Dra, A.; Zennouhi, O.; Bouymajane, A.; Rhazi Filali, F.; Nassiri, L.; Abouarnadasse, S. Chemical characterization and adsorption of oil mill wastewater on Moroccan clay in order to be used in the agricultural field. Heliyon 2020, 6, e03164. [Google Scholar] [CrossRef]
- Klikarová, J.; Rotondo, A.; Cacciola, F.; Česlová, L.; Dugo, P.; Mondello, L.; Rigano, F. The Phenolic Fraction of Italian Extra Virgin Olive Oils: Elucidation Through Combined Liquid Chromatography and NMR Approaches. Food Anal. Methods 2019, 12, 1759–1770. [Google Scholar] [CrossRef]
- Fanali, C.; Posta, S.D.; Dugo, L.; Russo, M.; Gentili, A.; Mondello, L.; De Gara, L. Application of deep eutectic solvents for the extraction of phenolic compounds from extra-virgin olive oil. Electrophoresis 2020, 41, 1752–1759. [Google Scholar] [CrossRef]
- Russo, M.; Bonaccorsi, I.L.; Cacciola, F.; Dugo, L.; De Gara, L.; Dugo, P.; Mondello, L. Distribution of bioactives in entire mill chain from the drupe to the oil and wastes. Nat. Prod. Res. 2020, 1–6. [Google Scholar] [CrossRef]
- Panel, E.; Nda, A. Scientific Opinion on the substantiation of health claims related to polyphenols in olive and protection of LDL particles from oxidative damage (ID 1333, 1638, 1639, 1696, 2865), maintenance of normal blood HDL cholesterol concentrations (ID 1639), mainte. EFSA J. 2011, 9, 1–25. [Google Scholar]
- Gallina-Toschi, T.; Cerretani, L.; Bendini, A.; Bonoli-Carbognin, M.; Lercker, G. Oxidative stability and phenolic content of virgin olive oil: An analytical approach by traditional and high resolution techniques. J. Sep. Sci. 2005, 28, 859–870. [Google Scholar] [CrossRef] [PubMed]
- Tura, D.; Gigliotti, C.; Pedò, S.; Failla, O.; Bassi, D.; Serraiocco, A. Influence of cultivar and site of cultivation on levels of lipophilic and hydrophilic antioxidants in virgin olive oils (Olea Europea L.) and correlations with oxidative stability. Sci. Hortic. 2007, 112, 108–119. [Google Scholar] [CrossRef]
- Aparicio, R.; Morales, M.T. Characterization of Olive Ripeness by Green Aroma Compounds of Virgin Olive Oil. J. Agric. Food Chem. 1998, 46, 1116–1122. [Google Scholar] [CrossRef]
- Bendini, A.; Cerretani, L.; Di Virgilio, F.; Belloni, P.; Bonoli-Carbognin, M.; Lercker, G. Preliminary evaluation of the application of the ftir spectroscopy to control the geographic origin and quality of virgin olive oils. J. Food Qual. 2007, 30, 424–437. [Google Scholar] [CrossRef]
- Opinion, S. Scientific Opinion on Dietary Reference Values for vitamin E as α-tocopherol. EFSA J. 2015, 13, 1–72. [Google Scholar]
- Pérez-Rodrigo, C.; Aranceta, J. Olive Oil: Its Role in the Diet; Elsevier: Amsterdam, The Netherlands, 2016; ISBN 9780123849472. [Google Scholar]
- Ouni, Y.; Taamalli, A.; Gómez-Caravaca, A.M.; Segura-Carretero, A.; Fernández-Gutiérrez, A.; Zarrouk, M. Characterisation and quantification of phenolic compounds of extra-virgin olive oils according to their geographical origin by a rapid and resolutive LC-ESI-TOF MS method. Food Chem. 2011, 127, 1263–1267. [Google Scholar] [CrossRef]
- Ben Brahim, S.; Kelebek, H.; Ammar, S.; Abichou, M.; Bouaziz, M. LC–MS phenolic profiling combined with multivariate analysis as an approach for the characterization of extra virgin olive oils of four rare Tunisian cultivars during ripening. Food Chem. 2017, 229, 9–19. [Google Scholar] [CrossRef]
- Ammar, S.; Kelebek, H.; Zribi, A.; Abichou, M.; Selli, S.; Bouaziz, M. LC-DAD/ESI-MS/MS characterization of phenolic constituents in Tunisian extra-virgin olive oils: Effect of olive leaves addition on chemical composition. Food Res. Int. 2017, 100, 477–485. [Google Scholar] [CrossRef]
- Rotondi, A.; Bendini, A.; Cerretani, L.; Mari, M.; Lercker, G.; Toschi, T.G. Effect of olive ripening degree on the oxidative stability and organoleptic properties of cv. Nostrana di Brisighella extra virgin olive oil. J. Agric. Food Chem. 2004, 52, 3649–3654. [Google Scholar] [CrossRef]
- Klikarová, J.; Česlová, L.; Kalendová, P.; Dugo, P.; Mondello, L.; Cacciola, F. Evaluation of Italian extra virgin olive oils based on the phenolic compounds composition using multivariate statistical methods. Eur. Food Res. Technol. 2020, 246, 1241–1249. [Google Scholar] [CrossRef]
- Commission Regulation (EEC) No 2568/91 on the Characteristics of Olive Oil and Olive-Residue Oil and on the Relevant Methods of Analysis. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=CELEX%3A01991R2568-20151016 (accessed on 15 October 2020).
- Commission Regulation (EU). No 432/2012. Establishing a list of permitted health claims made on foods, other than those referring to the reduction of disease risk and to children’s development and health. OJEU 2012, 136, 1–40. [Google Scholar]
- Regulation (EC). No 1924/2006 of the European Parliament and of the Council of 20 December 2006 on nutrition and health claims made on foods. OJEU 2006, 404, 9–25. [Google Scholar]
- COI. Norme commerciale applicable à l’huile d’olive et à l’huile de grignions d’olive–COI/T.20/Doc. 2001.
- Zarrouk, A.; Martine, L.; Grégoire, S.; Nury, T.; Meddeb, W.; Camus, E.; Badreddine, A.; Durand, P.; Namsi, A.; Yammine, A.; et al. Profile of Fatty Acids, Tocopherols, Phytosterols and Polyphenols in Mediterranean Oils (Argan Oils, Olive Oils, Milk Thistle Seed Oils and Nigella Seed Oil) and Evaluation of their Antioxidant and Cytoprotective Activities. Curr. Pharm. Des. 2019, 25, 1791–1805. [Google Scholar] [CrossRef] [PubMed]
- USD FoodData Central. Available online: https://fdc.nal.usda.gov/ (accessed on 4 September 2020).
- Nevado, J.J.B.; Peñalvo, G.C.; Robledo, V.R. Advantages of using a modified orthogonal sampling configuration originally designed for LC-ESI-MS to couple CE and MS for the determination of antioxidant phenolic compounds found in virgin olive oil. Talanta 2010, 82, 548–554. [Google Scholar] [CrossRef] [PubMed]
- Talhaoui, N.; Taamalli, A.; Gómez-Caravaca, A.M.; Fernández-Gutiérrez, A.; Segura-Carretero, A. Phenolic compounds in olive leaves: Analytical determination, biotic and abiotic influence, and health benefits. Food Res. Int. 2015, 77, 92–108. [Google Scholar] [CrossRef]
- Dierkes, G.; Krieger, S.; Dück, R.; Bongartz, A.; Schmitz, O.J.; Hayen, H. High-performance liquid chromatography-mass spectrometry profiling of phenolic compounds for evaluation of olive oil bitterness and pungency. J. Agric. Food Chem. 2012, 60, 7597–7606. [Google Scholar] [CrossRef] [PubMed]
- Bajoub, A.; Carrasco-Pancorbo, A.; Ajal, E.A.; Ouazzani, N.; Fernández-Gutiérrez, A. Potential of LC-MS phenolic profiling combined with multivariate analysis as an approach for the determination of the geographical origin of north Moroccan virgin olive oils. Food Chem. 2015, 166, 292–300. [Google Scholar] [CrossRef]
- Różańska, A.; Russo, M.; Cacciola, F.; Salafia, F.; Polkowska, Ż.; Dugo, P.; Mondello, L. Concentration of Potentially Bioactive Compounds in Italian Extra Virgin Olive Oils from Various Sources by Using LC-MS and Multivariate Data Analysis. Foods 2020, 9, 1120. [Google Scholar] [CrossRef]
- Bajoub, A.; Hurtado-Fernández, E.; Ajal, E.A.; Ouazzani, N.; Fernández-Gutiérrez, A.; Carrasco-Pancorbo, A. Comprehensive 3-year study of the phenolic profile of Moroccan monovarietal virgin olive oils from the meknès region. J. Agric. Food Chem. 2015, 63, 4376–4385. [Google Scholar] [CrossRef]
- Olmo-García, L.; Bajoub, A.; Benlamaalam, S.; Hurtado-Fernández, E.; Bagur-González, M.G.; Chigr, M.; Mbarki, M.; Fernández-Gutiérrez, A.; Carrasco-Pancorbo, A. Establishing the phenolic composition of olea europaea L. Leaves from cultivars grown in Morocco as a crucial step towards their subsequent exploitation. Molecules 2018, 23, 2524. [Google Scholar] [CrossRef]
- Miho, H.; Moral, J.; López-González, M.; Díez, C.; Priego-Capote, F. The phenolic profile of virgin olive oil is influenced by malaxation conditions and determines the oxidative stability. Food Chem. 2020, 314, 126183. [Google Scholar] [CrossRef] [PubMed]
- El Qarnifa, S.; El Antari, A.; Hafidi, A. Effect of Maturity and Environmental Conditions on Chemical Composition of Olive Oils of Introduced Cultivars in Morocco. J. Food Qual. 2019, 2019, 1854539. [Google Scholar] [CrossRef]
- Dugo, L.; Russo, M.; Cacciola, F.; Mandolfino, F.; Salafia, F.; Vilmercati, A.; Fanali, C.; Casale, M.; De Gara, L.; Dugo, P.; et al. Determination of the Phenol and Tocopherol Content in Italian High-Quality Extra-Virgin Olive Oils by Using LC-MS and Multivariate Data Analysis. Food Anal. Methods 2020, 13, 1027–1041. [Google Scholar] [CrossRef]
- Fanali, C.; Della Posta, S.; Vilmercati, A.; Dugo, L.; Russo, M.; Petitti, T.; Mondello, L.; de Gara, L. Extraction, Analysis, and Antioxidant Activity Evaluation of Phenolic Compounds in Different Italian Extra-Virgin Olive Oils. Molecules 2018, 23, 3249. [Google Scholar] [CrossRef]
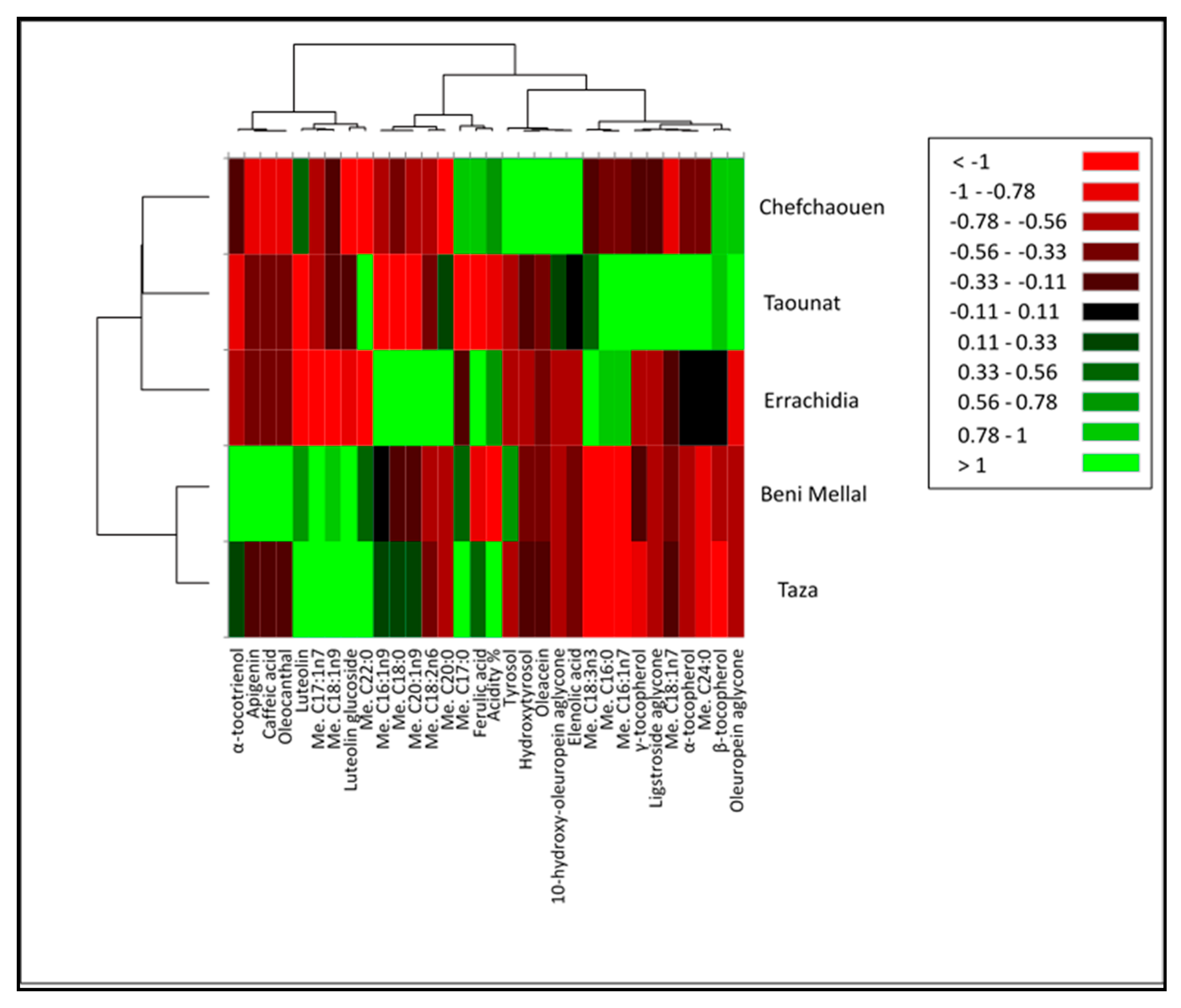
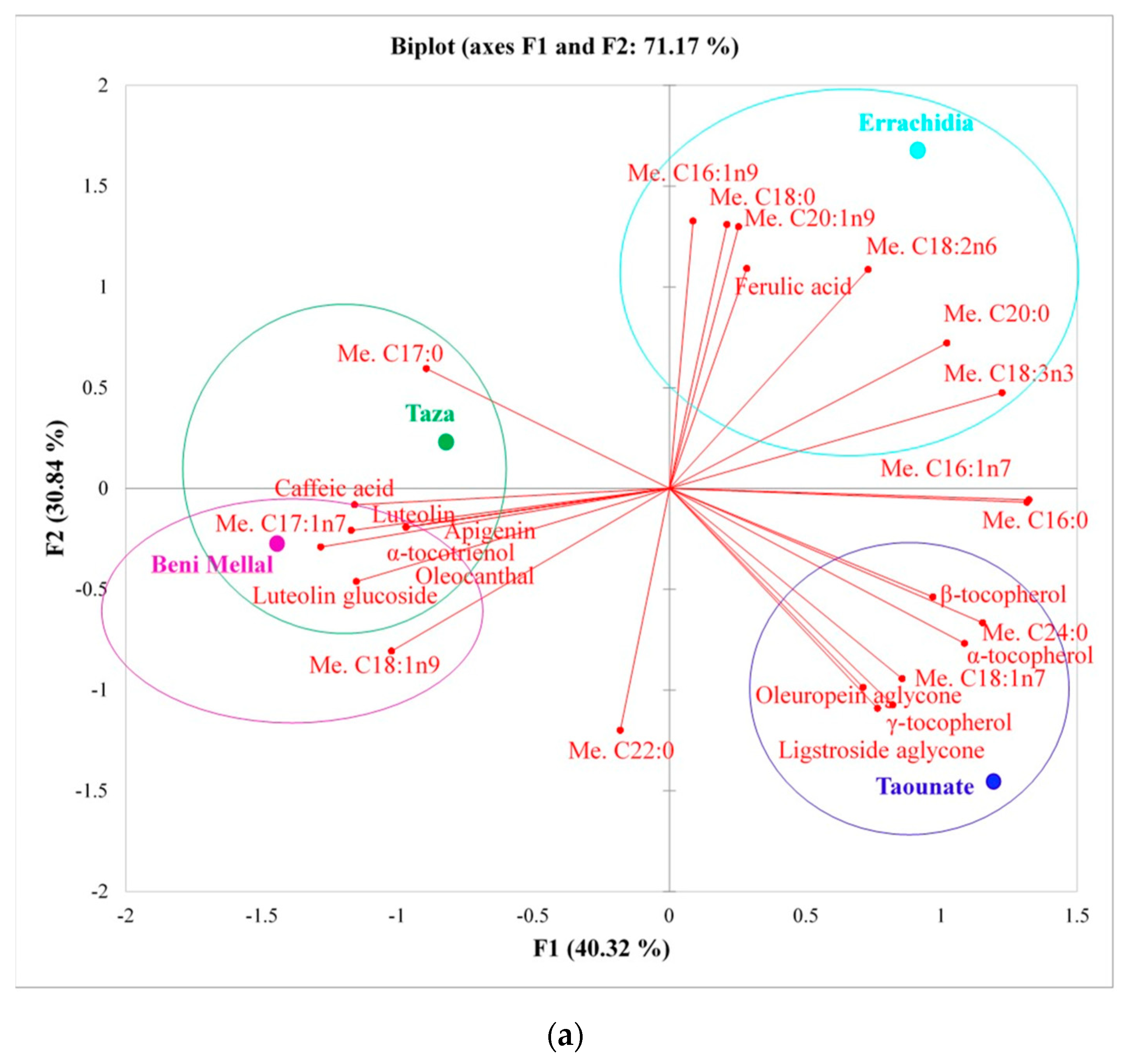
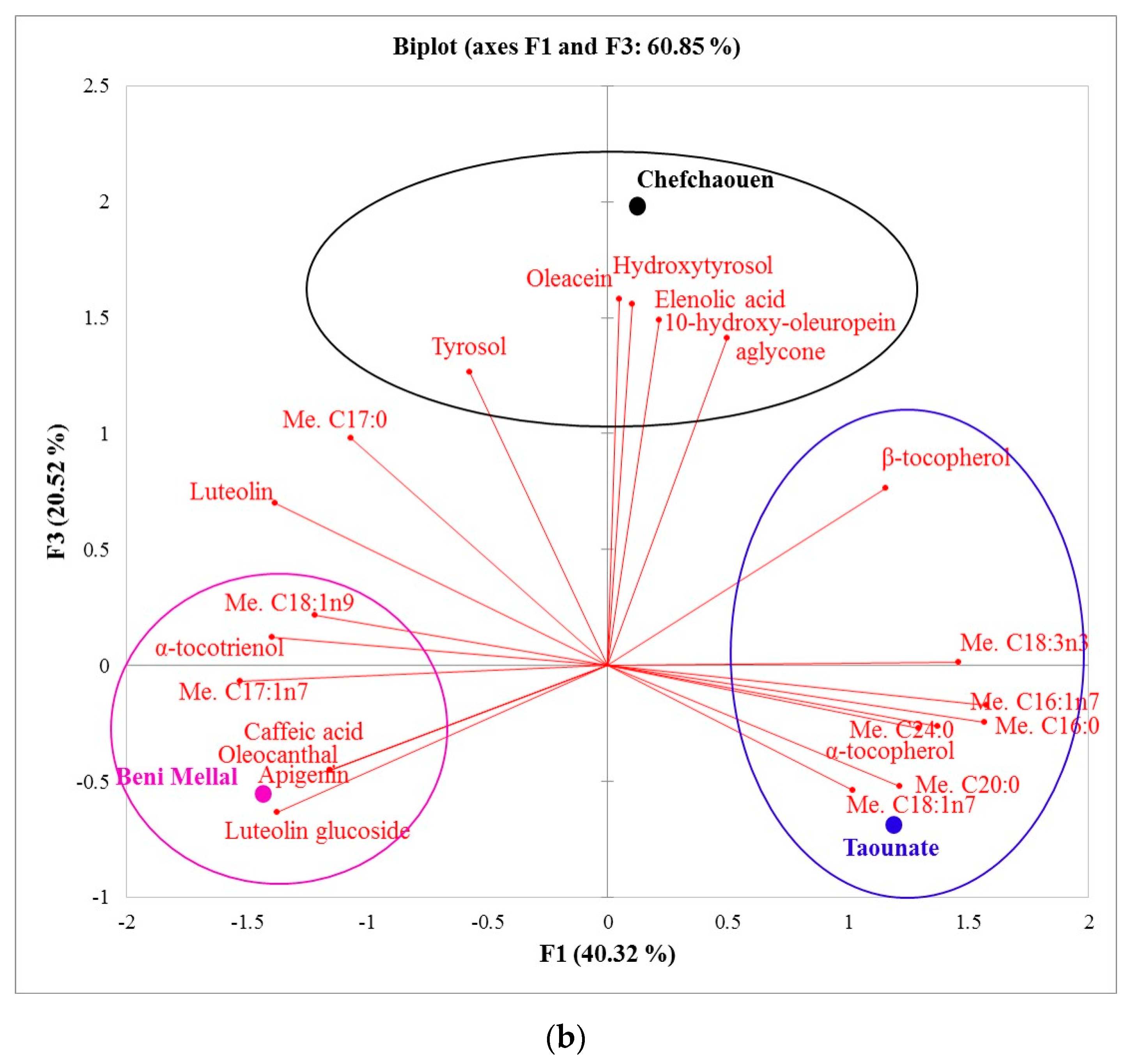
| Compounds\Region (Provinces) | Fes-Meknes (Taounate) | Drâa-Tafilalet (Errachidia) | Tanger-Tetouan-Al Hoceïma (Chefchaouen) | Fes-Meknes (Taza) | Beni Mellal-Khenifra (Beni Mellal) |
|---|---|---|---|---|---|
| α-tocopherol | 100.4 ± 0.14 | 55.8 ± 0.10 | 55.3 ± 0.27 | 38.4 ± 0.10 | 39.9 ± 0.24 |
| α-tocotrienol | <LOQ | <LOQ | 2.92 ± 0.07 | 1.97 ± 0.05 | 7.60 ± 0.04 |
| β-tocopherol | 2.1 ± 0.07 | 1.9 ± 0.07 | 2.1 ± 0.06 | 1.6 ± 0.07 | 1.7 ± 0.05 |
| γ-tocopherol | 5.2 ± 0.06 | 4.2 ± 0.05 | 4.5 ± 0.08 | 4.1 ± 0.10 | 4.3 ± 0.06 |
| δ-tocopherol | <LOD | <LOD | <LOD | <LOD | <LOD |
| Tyrosol | <LOD | <LOD | 300.6 ± 11.26 | <LOD | 186.2 ± 2.58 |
| Hydroxytyrosol | 6.4 ± 0.54 | <LOD | 40.0 ± 1.46 | 2.8 ± 0.15 | 4.6 ± 0.07 |
| Caffeic acid | <LOQ | <LOD | <LOD | <LOD | 3.5 ± 0.06 |
| Ferulic acid | <LOD | 4.4 ± 0.43 | 3.9 ± 0.07 | 2.0 ± 0.03 | 0.8 ± 0.04 |
| 10-hydroxy-oleuropein aglycone a | 25.4 ± 0.09 | 1.2 ± 0.18 | 65.7 ± 3.81 | 0.3 ± 0.00 | 1.4 ± 0.24 |
| Oleocanthal a | <LOD | <LOD | <LOQ | <LOQ | 0.8 ± 0.00 |
| Luteolin | <LOQ | <LOQ | 6.4 ± 0.19 | 7.8 ± 0.72 | 6.3 ± 0.13 |
| Oleacein | 3.1 ± 0.15 | 0.2 ± 0.02 | 33.3 ± 1.91 | 2.3 ± 0.01 | 3.5 ± 0.17 |
| Apigenin | <LOQ | <LOQ | <LOQ | <LOQ | 6.1 ± 0.00 |
| Oleuropein aglycone a | 231.3 ± 4.45 | 1.9 ± 0.00 | 198.3 ± 13.17 | 18.6 ± 0.00 | 20.6 ± 1.24 |
| Luteolin glucoside | 2.5 ± 0.03 | 1.1 ± 0.51 | 1.5 ± 0.03 | 3.9 ± 0.22 | 5.5 ± 1.79 |
| Elenolic acid | 31.3 ± 2.00 | 8.7 ± 0.81 | 85.6 ± 2.67 | 13.7 ± 0.26 | 19.6 ± 1.63 |
| Ligstroside aglycone b | 57.3 ± 2.92 | 0.7 ± 0.04 | 21.4 ± 0.39 | 1.2 ± 0.07 | 3.9 ± 0.19 |
| Acidity% | 1.3% | 2.0% | 2.0% | 2.0% | 1.1% |
| ∑Vitamin E | 107.6 * | 61.9 | 64.8 | 46.1 | 53.5 |
| ∑Phenolic alcohols | 6.4 | <LOD | 340.6 | 2.8 | 190.8 |
| ∑Phenolic acids | 31.3 | 13.1 | 89.5 | 15.8 | 24.0 |
| ∑Secoiridoids | 314.0 | 3.9 | 285.4 | 20.1 | 26.6 |
| ∑Flavonoids | 2.5 | 1.1 | 7.9 | 11.7 | 18.0 |
| ∑Phenols | 354.2 ** | 18.1 | 723.4 ** | 50.4 | 259.4 ** |
| Fatty Acid | Mass Spectral Similarity (%) | Experimental LRI | Tabulated LRI | Taounate (%) | Errachidia (%) | Chefchaouen (%) | Taza (%) | Beni Mellal (%) |
|---|---|---|---|---|---|---|---|---|
| Palmitic acid (C16:0) | 95 | 1600 | 1600 | 9.99 | 9.63 | 8.60 | 7.66 | 7.75 |
| Hypogeic acid (C16:1n9) | 91 | 1605 | 1605 | 0.31 | 0.33 | 0.32 | 0.32 | 0.37 |
| Palmitoleic acid (C16:1n7) | 96 | 1615 | 1616 | 0.65 | 0.61 | 0.47 | 0.32 | 0.33 |
| Margaric acid (C17:0) | 90 | 1694 | 1694 | 0.03 | 0.04 | 0.04 | 0.04 | 0.04 |
| Margaleic acid (C17:1n7) | 91 | 1707 | 1711 | 0.05 | 0.05 | 0.05 | 0.06 | 0.06 |
| Stearic acid (C18:0) | 95 | 1802 | 1801 | 2.08 | 2.95 | 2.42 | 2.35 | 2.49 |
| Oleic acid (C18:1n9) | 91 | 1812 | 1808 | 75.89 | 69.79 | 76.94 | 79.39 | 78.15 |
| Cis-vaccenic acid (C18:1n7) | 96 | 1816 | 1816 | 2.80 | 2.33 | 2.32 | 2.31 | 2.19 |
| linoleic acid (C18:2n6) | 95 | 1839 | 1838 | 6.71 | 12.67 | 7.41 | 6.25 | 7.29 |
| α-linolenic acid (C18:3n3) | 96 | 1883 | 1883 | 0.83 | 0.93 | 0.80 | 0.67 | 0.69 |
| Arachidic acid (C20:0) | 94 | 1999 | 2000 | 0.27 | 0.28 | 0.27 | 0.26 | 0.26 |
| Eicosenoic acid (C20:1n9) | 91 | 2014 | 2015 | 0.26 | 0.31 | 0.28 | 0.28 | 0.30 |
| Behenic acid (C22:0) | 92 | 2199 | 2201 | 0.07 | 0.06 | 0.06 | 0.07 | 0.06 |
| Legnoceric acid (C24:0) | 90 | 2400 | 2400 | 0.04 | 0.03 | 0.03 | 0.02 | 0.03 |
Sample Availability: Samples of the compounds are not available from the authors. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bouymajane, A.; Oulad El Majdoub, Y.; Cacciola, F.; Russo, M.; Salafia, F.; Trozzi, A.; Rhazi Filali, F.; Dugo, P.; Mondello, L. Characterization of Phenolic Compounds, Vitamin E and Fatty Acids from Monovarietal Virgin Olive Oils of “Picholine marocaine” Cultivar. Molecules 2020, 25, 5428. https://doi.org/10.3390/molecules25225428
Bouymajane A, Oulad El Majdoub Y, Cacciola F, Russo M, Salafia F, Trozzi A, Rhazi Filali F, Dugo P, Mondello L. Characterization of Phenolic Compounds, Vitamin E and Fatty Acids from Monovarietal Virgin Olive Oils of “Picholine marocaine” Cultivar. Molecules. 2020; 25(22):5428. https://doi.org/10.3390/molecules25225428
Chicago/Turabian StyleBouymajane, Aziz, Yassine Oulad El Majdoub, Francesco Cacciola, Marina Russo, Fabio Salafia, Alessandra Trozzi, Fouzia Rhazi Filali, Paola Dugo, and Luigi Mondello. 2020. "Characterization of Phenolic Compounds, Vitamin E and Fatty Acids from Monovarietal Virgin Olive Oils of “Picholine marocaine” Cultivar" Molecules 25, no. 22: 5428. https://doi.org/10.3390/molecules25225428
APA StyleBouymajane, A., Oulad El Majdoub, Y., Cacciola, F., Russo, M., Salafia, F., Trozzi, A., Rhazi Filali, F., Dugo, P., & Mondello, L. (2020). Characterization of Phenolic Compounds, Vitamin E and Fatty Acids from Monovarietal Virgin Olive Oils of “Picholine marocaine” Cultivar. Molecules, 25(22), 5428. https://doi.org/10.3390/molecules25225428








