Mapping the Photochemistry of European Mid-Latitudes Rivers: An Assessment of Their Ability to Photodegrade Contaminants
Abstract
1. Introduction
2. Results and Discussion
2.1. Mapping the PPRIs Steady-State Concentrations
2.2. Modeling the Photodegradation of Contaminants
3. Materials and Methods
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Daughton, C.G.; Ternes, T.A. Pharmaceuticals and personal care products in the environment: Agents of subtle change? Environ. Health Persp. 1999, 107, 907–938. [Google Scholar] [CrossRef] [PubMed]
- Schwarzenbach, R.P.; Escher, B.I.; Fenner, K.; Hofstetter, T.B.; Johnson, C.A.; von Gunten, U.; Wehrli, B. The challenge of micropollutants in aquatic systems. Science 2006, 313, 1072–1077. [Google Scholar] [CrossRef] [PubMed]
- Pal, A.; Gin, K.Y.-H.; Lin, A.Y.-C.; Reinhard, M. Impacts of emerging organic contaminants on freshwater resources: Review of recent occurrences, sources, fate and effects. Sci. Total Environ. 2010, 408, 6062–6069. [Google Scholar] [CrossRef] [PubMed]
- Luo, Y.; Guo, W.; Ngo, H.H.; Nghiem, L.D.; Hai, F.I.; Zhang, J.; Liang, S.; Wang, X.C. A review on the occurrence of micropollutants in the aquatic environment and their fate and removal during wastewater treatment. Sci. Total Environ. 2014, 473–474, 619–641. [Google Scholar] [CrossRef] [PubMed]
- Lindim, C.; de Zwart, D.; Cousins, I.T.; Kutsarova, S.; Kühne, R.; Schüürmann, G. Exposure and ecotoxicological risk assessment of mixtures of top prescribed pharmaceuticals in Swedish freshwaters. Chemosphere 2019, 220, 344–352. [Google Scholar] [CrossRef]
- Riva, F.; Zuccato, E.; Davoli, E.; Fattore, E.; Castiglioni, S. Risk assessment of a mixture of emerging contaminants in surface water in a highly urbanized area in Italy. J. Hazard. Mater. 2019, 361, 103–110. [Google Scholar] [CrossRef]
- Wang, J.; Wang, S. Removal of pharmaceuticals and personal care products (PPCPs) from wastewater: A review. J. Environ. Manag. 2016, 182, 620–640. [Google Scholar] [CrossRef]
- Comoretto, L.; Arfib, B.; Talva, R.; Chauvelon, P.; Pichaud, M.; Chiron, S.; Höhener, P. Runoff of pesticides from rice fields in the Ile de Camargue (Rhône river delta, France): Field study and modeling. Environ. Pollut. 2008, 151, 486–493. [Google Scholar] [CrossRef]
- Lefrancq, M.; Jadas-Hécart, A.; La Jeunesse, I.; Landry, D.; Payraudeau, S. High frequency monitoring of pesticides in runoff water to improve understanding of their transport and environmental impacts. Sci. Total Environ. 2017, 587–588, 75–86. [Google Scholar] [CrossRef]
- Köck-Schulmeyer, M.; Ginebreda, A.; Postigo, C.; Garrido, T.; Fraile, J.; de Alda, M.L.; Barceló, D. Four-Year advanced monitoring program of polar pesticides in groundwater of Catalonia (NE-Spain). Sci. Total Environ. 2014, 470–471, 1087–1098. [Google Scholar] [CrossRef]
- Reemtsma, T.; Berger, U.; Arp, H.P.H.; Gallard, H.; Knepper, T.P.; Neumann, M.; Quintana, J.B.; Voogt, P. Mind the gap: Persistent and mobile organic compounds - water contaminants that slip through. Environ. Sci. Technol. 2016, 50, 10308–10315. [Google Scholar] [CrossRef] [PubMed]
- Burrows, H.D.; Canle, L.M.; Santaballa, J.A.; Steenken, S. Reaction pathways and mechanisms of photodegradation of pesticides. J. Photochem. Photobiol. B Biol. 2002, 67, 71–108. [Google Scholar] [CrossRef]
- Remucal, C.K. The role of indirect photochemical degradation in the environmental fate of pesticides: A review. Environ. Sci. Process. Impacts 2014, 16, 628–653. [Google Scholar] [CrossRef]
- Vione, D.; Minella, M.; Maurino, V.; Minero, C. Indirect photochemistry in sunlit surface waters: Photoinduced production of reactive transient species. Chem. Eur. J. 2014, 20, 10590–10606. [Google Scholar] [CrossRef] [PubMed]
- Canonica, S.; Laubscher, H.U. Inhibitory effect of dissolved organic matter on triplet-Induced oxidation of aquatic contaminants. Photochem. Photobiol. Sci. 2008, 7, 547–551. [Google Scholar] [CrossRef] [PubMed]
- Wenk, J.; Canonica, S. Phenolic antioxidants inhibit the triplet-induced transformation of anilines and sulfonamide antibiotics in aqueous solution. Environ. Sci. Technol. 2012, 46, 5455–5462. [Google Scholar] [CrossRef]
- Trivella, A.; Stawinoga, M.; Dayan, F.E.; Cantrell, C.L.; Mazellier, P.; Richard, C. Photolysis of natural β-Triketonic herbicides in water. Water Res. 2015, 78, 28–36. [Google Scholar] [CrossRef]
- Katagi, T. Direct photolysis mechanism of pesticides in water. J. Pestic. Sci. 2018, 43, 57–72. [Google Scholar] [CrossRef]
- Bavcon Kralj, M.; Franko, M.; Trebše, P. Photodegradation of organophosphorus insecticides-Investigations of products and their toxicity using gas chromatography-Mass spectrometry and AChE-thermal lens spectrometric bioassay. Chemosphere 2007, 67, 99–107. [Google Scholar] [CrossRef]
- Cermola, M.; DellaGreca, M.; Iesce, M.R.; Previtera, L.; Rubino, M.; Temussi, F.; Brigante, M. Phototransformation of fibrate drugs in aqueous media. Environ. Chem. Lett. 2005, 3, 43–47. [Google Scholar] [CrossRef]
- Katagi, T. Aerobic microbial transformation of pesticides in surface water. J. Pestic. Sci. 2013, 38, 10–26. [Google Scholar] [CrossRef]
- Rahm, S.; Green, N.; Norrgran, J.; Bergman, Å. Hydrolysis of environmental contaminants as an experimental tool for indication of their persistency. Environ. Sci. Technol. 2005, 39, 3128–3133. [Google Scholar] [CrossRef] [PubMed]
- Fenner, K.; Canonica, S.; Wackett, L.P.; Elsner, M. Evaluating pesticide degradation in the environment: Blind spots and emerging opportunities. Science 2013, 341, 752. [Google Scholar] [CrossRef]
- Al Housari, F.; Höhener, P.; Chiron, S. Factors responsible for rapid dissipation of acidic herbicides in the coastal lagoons of the Camargue (Rhône River Delta, France). Sci. Total Environ. 2011, 409, 582–587. [Google Scholar] [CrossRef]
- Zeng, T.; Arnold, W.A. Pesticide photolysis in prairie potholes: Probing photosensitized processes. Environ. Sci. Technol. 2013, 47, 6735–6745. [Google Scholar] [CrossRef]
- Yassine, M.; Fuster, L.; Dévier, M.-H.; Geneste, E.; Pardon, P.; Grélard, A.; Dufourc, E.; Al Iskandarani, M.; Aït-Aïssa, S.; Garric, J.; et al. Photodegradation of novel oral anticoagulants under sunlight irradiation in aqueous matrices. Chemosphere 2018, 193, 329–336. [Google Scholar] [CrossRef]
- Global Environment Monitoring System. Available online: www.GEMStat.org (accessed on 19 September 2018).
- Heberer, T.; Butz, S.; Stan, H.-J. Analysis of phenoxycarboxylic acids and other acidic compounds in tap, ground, surface and sewage water at the low ng/1 level. Int. J. Environ. Anal. Chem. 1995, 58, 43–53. [Google Scholar] [CrossRef]
- Heberer, T.; Stan, H.J. Determination of clofibric acid and N-(phenylsulfonyl)-sarcosine in sewage, river and drinking water. Int. J. Environ. Anal. Chem. 1997, 67, 113–124. [Google Scholar] [CrossRef]
- Buser, H.-R.; Müller, M.D.; Theobald, N. Occurrence of the pharmaceutical drug clofibric acid and the herbicide mecoprop in various Swiss lakes and in the North Sea. Environ. Sci. Technol. 1998, 32, 188–192. [Google Scholar] [CrossRef]
- Ternes, T.A. Occurrence of drugs in German sewage treatment plants and rivers. Water Res. 1998, 32, 3245–3260. [Google Scholar] [CrossRef]
- Weigel, S.; Kuhlmann, J.; Hühnerfuss, H. Drugs and personal care products as ubiquitous pollutants: Occurrence and distribution of clofibric acid, caffeine and DEET in the North Sea. Sci. Total Environ. 2002, 295, 131–141. [Google Scholar] [CrossRef]
- Tixier, C.; Singer, H.P.; Oellers, S.; Müller, S.R. Occurrence and fate of carbamazepine, clofibric acid, diclofenac, ibuprofen, ketoprofen, and naproxen in surface waters. Environ. Sci. Technol. 2003, 37, 1061–1068. [Google Scholar] [CrossRef] [PubMed]
- Vogna, D.; Marotta, R.; Andreozzi, R.; Napolitano, A.; d’Ischia, M. Kinetic and chemical assessment of the UV/H2O2 treatment of antiepileptic drug carbamazepine. Chemosphere 2004, 54, 497–505. [Google Scholar] [CrossRef]
- Carlos, L.; Mártire, D.O.; Gonzalez, M.C.; Gomis, J.; Bernabeu, A.; Amat, A.M.; Arques, A. Photochemical fate of a mixture of emerging pollutants in the presence of humic substances. Water Res. 2012, 46, 4732–4740. [Google Scholar] [CrossRef] [PubMed]
- Vione, D.; Maddigapu, P.R.; De Laurentiis, E.; Minella, M.; Pazzi, M.; Maurino, V.; Minero, C.; Kouras, S.; Richard, C. Modelling the photochemical fate of ibuprofen in surface waters. Water Res. 2011, 45, 6725–6736. [Google Scholar] [CrossRef]
- De Laurentiis, E.; Chiron, S.; Kouras-Hadef, S.; Richard, C.; Minella, M.; Maurino, V.; Minero, C.; Vione, D. Photochemical fate of carbamazepine in surface freshwaters: Laboratory measures and modeling. Environ. Sci. Technol. 2012, 46, 8164–8173. [Google Scholar] [CrossRef]
- Marchetti, G.; Minella, M.; Maurino, V.; Minero, C.; Vione, D. Photochemical transformation of atrazine and formation of photointermediates under conditions relevant to sunlit surface waters: Laboratory measures and modelling. Water Res. 2013, 47, 6211–6222. [Google Scholar] [CrossRef]
- Avetta, P.; Fabbri, D.; Minella, M.; Brigante, M.; Maurino, V.; Minero, C.; Pazzi, M.; Vione, V. Assessing the phototransformation of diclofenac, clofibric acid and naproxen in surface waters: Model predictions and comparison with field data. Water Res. 2016, 105, 383–394. [Google Scholar] [CrossRef]
- McNeill, K.; Canonica, S. Triplet state dissolved organic matter in aquatic photochemistry: Reaction mechanisms, substrate scope, and photophysical properties. Environ. Sci.-Proc. Impacts 2016, 18, 1381–1399. [Google Scholar] [CrossRef]
- Grebel, J.E.; Pignatello, J.J.; William, A.; Mitch, W.A. Sorbic acid as a quantitative probe for the formation, scavenging and steady-state concentrations of the triplet-excited state of organic compounds. Water Res. 2011, 45, 6535–6544. [Google Scholar] [CrossRef]
- QGIS Developmental Team. QGIS Geographic Information System. In Open Source Geospatial Foundation Project; Available online: https://www.osgeo.org (accessed on 3 July 2019).
- Buxton, G.V.; Greenstock, C.L.; Helman, W.P.; Ross, A.B. Critical review of rate constants for reactions of hydrated electrons, hydrogen atoms and hydroxyl radicals (•OH/O−•) in aqueous solution. J. Phys. Chem. Ref. Data 1988, 17, 513–886. [Google Scholar] [CrossRef]
- Mack, J.; Bolton, J.R. Photochemistry of nitrite and nitrate in aqueous solution: A review. J. Photochem. Photobiol. A 1999, 128, 1–13. [Google Scholar] [CrossRef]
- Canonica, S.; Kohn, T.; Mac, M.; Real, F.J.; Wirz, J.; von Gunten, U. Photosensitizer method to determine rate constants for the reaction of carbonate radical with organic compounds. Environ. Sci. Technol. 2005, 39, 9182–9188. [Google Scholar] [CrossRef] [PubMed]
- Yan, S.; Liu, Y.; Lian, L.; Li, R.; Ma, J.; Zhou, H.; Song, W. Photochemical formation of carbonate radical and its reaction with dissolved organic matters. Water Res. 2019, 161, 288–296. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Mabury, S.A. A new method for measuring carbonate radical reactivity toward pesticides. Enviorn. Toxicol. Chem. 2000, 19, 1501–1507. [Google Scholar] [CrossRef]
- Gerecke, A.C.; Canonica, S.; Muller, S.R.; Scharer, M.; Schwarzenbach, R.P. Quantification of dissolved natural organic matter (DOM) mediated phototransformation of phenylurea herbicides in lakes. Environ. Sci. Technol. 2001, 35, 3915–3923. [Google Scholar] [CrossRef]
- Canonica, S.; Hellrung, B.; Müller, P.; Wirz, J. Aqueous oxidation of phenyluera herbicides by triplet aromatic ketones. Environ. Sci. Technol. 2006, 40, 6636–6641. [Google Scholar] [CrossRef]
- Benitez, F.J.; Real, F.J.; Acero, J.L.; Garcia, C. Kinetics of the transformation of phenylurea herbicides during ozonation of natural waters: Rate constants and model predictions. Water Res. 2007, 41, 4073–4084. [Google Scholar] [CrossRef]
- Beck, A.J.; Lam, V.; Henderson, D.E.; Beven, K.J.; Harris, G.L.; Howse, K.R.; Johnny Johnston, A.E.; Jones, K.C. Movement of water and the herbicides atrazine and isoproturon through a large structured clay soil core. J. Contam. Hydrol. 1995, 19, 237–260. [Google Scholar] [CrossRef]
- Williams, R.J.; Brooke, D.N.; Matthiessen, P.; Mills, M.; Turnbull, A.; Harrison, R.M. Pesticide transport to surface waters within an agricultural catchment. J. Inst. Water Environ. Manag. 1995, 9, 72–81. [Google Scholar] [CrossRef]
- Klöppel, H.; Haider, J.; Kördel, W. Herbicides in surface runoff: A rainfall simulation study on small plots in the field. Chemosphere 1994, 28, 649–662. [Google Scholar] [CrossRef]
- Pichon, V.; Chen, L.; Hennion, M.-C. On-Line preconcentration and liquid chromatographic analysis of phenylurea pesticides in environmental water using a silica-based immunosorbent. Anal. Chim. Acta 1995, 311, 429–436. [Google Scholar] [CrossRef]
- Schuelein, J.; Glaessgen, W.E.; Hertkorn, N.; Schroeder, P.; Sandermann, H., Jr.; Kettrup, A. Detection and identification of the herbicide isoproturon and its metabolites in field samples after a heavy rainfall event. Int. J. Environ. Anal. Chem. 1996, 65, 193–202. [Google Scholar] [CrossRef]
- Stangroom, S.J.; Collins, C.D.; Lester, J.N. Sources of organic micropollutants to lowland rivers. Environ. Technol. 1998, 19, 643–666. [Google Scholar] [CrossRef]
- Fingler, S.; Mendaš, G.; Dvoršćak, M.; Stipičević, S.; Vasilić, Ž.; Drevenkar, V. Herbicide micropollutants in surface, ground and drinking waters within and near the area of Zagreb, Croatia. Environ. Sci. Pollut. Res. 2017, 24, 11017–11030. [Google Scholar] [CrossRef]
- Barbosa, M.O.; Ribeiro, A.R.; Ratola, N.; Hain, E.; Homem, V.; Pereira, M.F.R.; Blaney, L.; Silva, A.M.T. Spatial and seasonal occurrence of micropollutants in four Portuguese rivers and a case study for fluorescence excitation-emission matrices. Sci. Total Environ. 2018, 644, 1128–1140. [Google Scholar] [CrossRef]
- Quintana, J.; de la Cal, A.; Boleda, M.R. Monitoring the complex occurrence of pesticides in the Llobregat basin, natural and drinking waters in Barcelona metropolitan area (Catalonia, NE Spain) by a validated multi-residue online analytical method. Sci. Total Environ. 2019, 692, 952–965. [Google Scholar] [CrossRef]
- Knauert, S.; Escher, B.; Singer, H.; Hollender, J.; Knauer, K. Mixture toxicity of three photosystem II inhibitors (atrazine, isoproturon, and diuron) toward photosynthesis of freshwater phytoplankton studied in outdoor mesocosms. Environ. Sci. Technol. 2008, 42, 6424–6430. [Google Scholar] [CrossRef]
- Sigurnjak, M.; Ukić, Š.; Cvetnić, M.; Markić, M.; Stankov, M.N.; Rasulev, B.; Kušić, H.; Božić, A.L.; Rogošić, M.; Bolanča, T. Combined toxicities of binary mixtures of alachlor, chlorfenvinphos, diuron and isoproturon. Chemosphere 2020, 240, 124973. [Google Scholar] [CrossRef]
- Bodrato, M.; Vione, D. APEX (Aqueous Photochemistry of Environmentally occurring Xenobiotics): A free software tool to predict the kinetics of photochemical processes in surface waters. Environ. Sci. Proc. Imp. 2014, 16, 732–740. [Google Scholar] [CrossRef]
- Shchevyev, Y.L.; Bogoyavlensky, N.Y. Calculation of the average flow velocity in rivers. The hydrological basis for water resources management. In Proceedings of the Beijing Symposium on the Hydrological Basis for Water Resources Management, Beijing, China, 23–26 October 1990; IAHS Publ. no. 197. International Association of Hydrological Sciences: Wallingford, UK, 1990; pp. 89–91. [Google Scholar]
- Schulze, K.; Hunger, M.; Döll, P. Simulating river flow velocity on global scale. Adv. Geosci. 2005, 5, 133–136. [Google Scholar] [CrossRef]
- Vione, D.; Scozzaro, A. Photochemistry of surface fresh waters in the framework of climate change. Environ. Sci. Technol. 2019, 53, 7945–7963. [Google Scholar] [CrossRef] [PubMed]
- Worrall, F.; Howden, N.J.K.; Burt, T.P. A method of estimating in-stream residence time of water in rivers. J. Hydrol. 2014, 512, 274–284. [Google Scholar] [CrossRef]
- Carena, L.; Terrenzio, D.; Mosley, L.M.; Toldo, M.; Minella, M.; Vione, D. Photochemical consequences of prolonged hydrological drought: A model assessment of the Lower Lakes of the Murray-Darling Basin (Southern Australia). Chemosphere 2019, 236, 124356. [Google Scholar] [CrossRef]
- National Center for Atmospheric Research (NCAR). 2015. Quick TUV Calculator [WWW Document]. Available online: http://cprm.acom.ucar.edu/Models/TUV/Interactive_TUV/ (accessed on 15 July 2019).
- Chang, E.E.; Chiang, P.C.; Lin, T.F. Development of surrogate organic contaminant parameters for source water quality standards in Taiwan, ROC. Chemosphere 1998, 37, 593–606. [Google Scholar] [CrossRef]
- Minella, M.; De Laurentiis, E.; Buhvestova, O.; Haldna, M.; Kangur, K.; Maurino, V.; Minero, C.; Vione, D. Modelling lake-water photochemistry: Three-Decade assessment of the steady-state concentration of photoreactive transients (OH, CO3− and 3CDOM*) in the surface water of polymictic Lake Peipsi (Estonia/Russia). Chemosphere 2013, 90, 2589–2596. [Google Scholar] [CrossRef]
- Clark, J.B.; Neale, P.; Tzortziou, M.; Cao, F.; Hood, R.R. A mechanistic model of photochemical transformation and degradation of colored dissolved organic matter. Mar. Chem. 2019, 214, 103666. [Google Scholar] [CrossRef]
- Marchisio, A.; Minella, M.; Maurino, V.; Minero, C.; Vione, D. Photogeneration of reactive transient species upon irradiation of natural water samples: Formation quantum yields in different spectral intervals, and implications for the photochemistry of surface waters. Water Res. 2015, 73, 145–156. [Google Scholar] [CrossRef]
- Carena, L.; Puscasu, C.G.; Comis, S.; Sarakha, M.; Vione, D. Environmental photodegradation of emerging contaminants: A re-Examination of the importance of triplet-sensitised processes, based on the use of 4-carboxybenzophenone as proxy for the chromophoric dissolved organic matter. Chemosphere 2019, 237, 124476. [Google Scholar] [CrossRef]
- Huang, Y.; Kong, M.; Westerman, D.; Xu, E.G.; Coffin, S.; Cochran, K.H.; Liu, Y.; Richardson, S.D.; Schlenk, D.; Dionysiou, D.D. Effects of HCO3− on degradation of toxic contaminants of emerging concern by UV/NO3−. Environ. Sci. Technol. 2018, 52, 12697–12707. [Google Scholar] [CrossRef]
- Acero, J.L.; Stemmler, K.; von Gunten, U. Degradation kinetics of atrazine and its degradation products with ozone and OH radicals: A predictive tool for drinking water treatment. Environ. Sci. Technol. 2000, 34, 591–597. [Google Scholar] [CrossRef]
- Larson, R.A.; Zepp, R.G. Reactivity of the carbonate radical with aniline derivatives. Environ. Toxicol. Chem. 1988, 7, 265–274. [Google Scholar] [CrossRef]
- Huber, M.M.; Canonica, S.; Park, G.-Y.; von Gunten, U. Oxidation of pharmaceuticals during ozonation and advanced oxidation processes. Environ. Sci. Technol. 2003, 37, 1016–1024. [Google Scholar] [CrossRef] [PubMed]
- Lam, M.W.; Mabury, S.A. Photodegradation of the pharmaceuticals atorvastatin, carbamazepine, levofloxacin, and sulfamethoxazole in natural waters. Aquat. Sci. 2005, 67, 177–188. [Google Scholar] [CrossRef]
- Hora, P.I.; Novak, P.J.; Arnold, W.A. Photodegradation of pharmaceutical compounds in partially nitritated wastewater during UV irradiation. Environ. Sci. Water Res. Technol. 2019, 5, 897. [Google Scholar] [CrossRef]
Sample Availability: no real samples were used in this modeling study. |

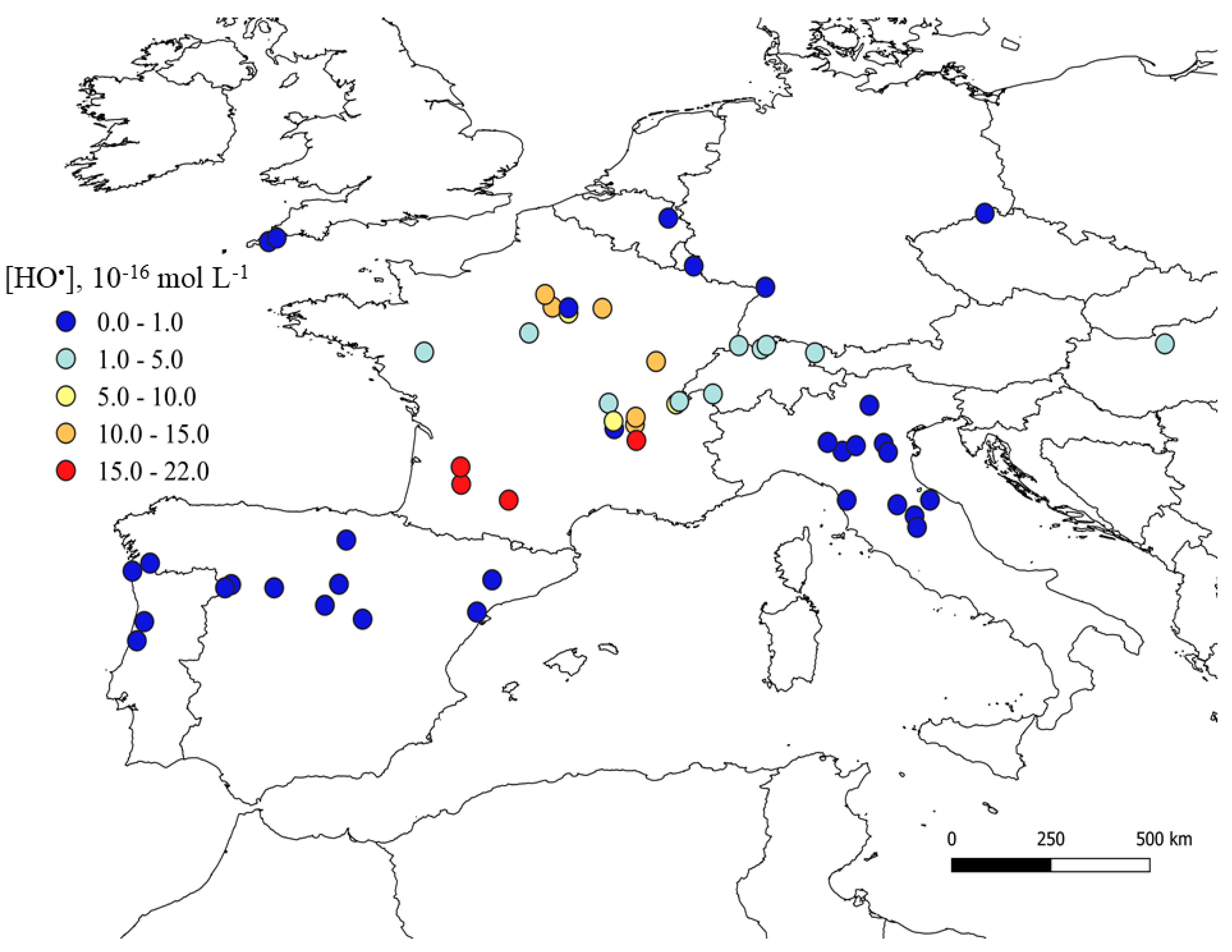
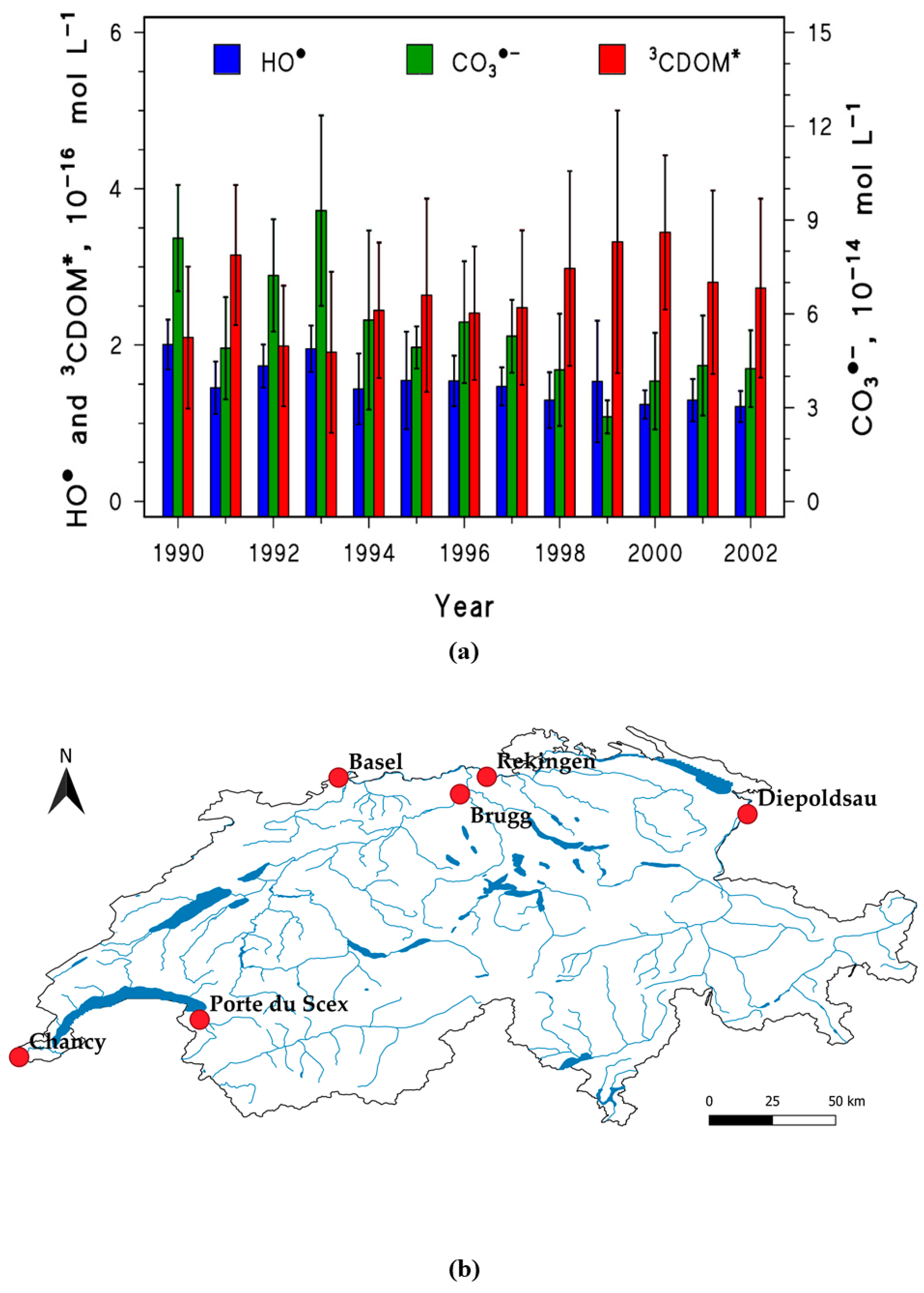
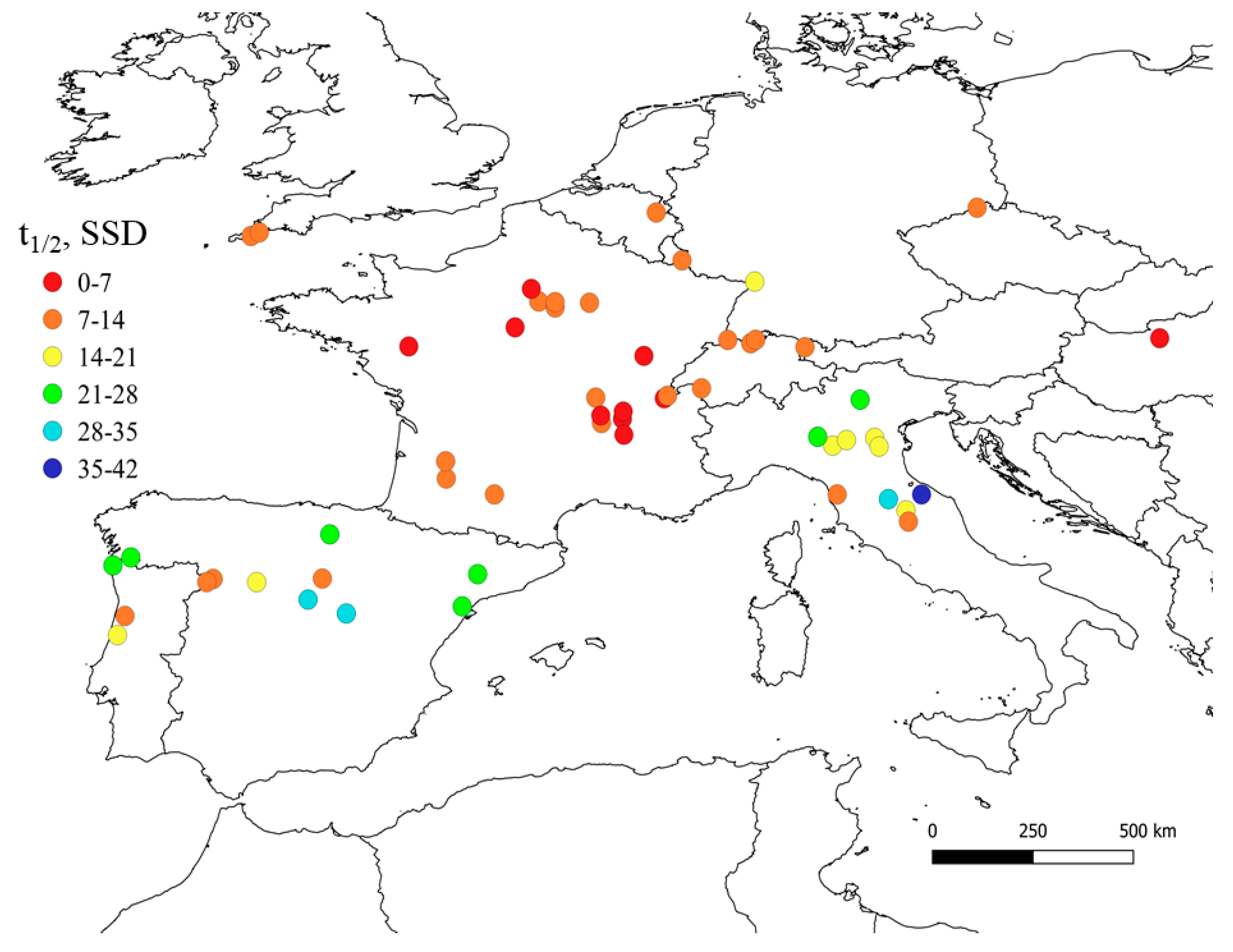

| Compound | Photochemical Half-Life Time, Days | Location | Ref. | |
|---|---|---|---|---|
| Model | Field | |||
| Ibuprofen | 60 ± 10 | 60–115 | Lake Greifensee | [36] |
| Carbamazepine | 110 ± 45 | 140 ± 50 | Lake Greifensee | [37] |
| Atrazine | 17 ± 4 | 20–21 | Chesapeake Bay (1 m depth) | [38] |
| 64 ± 18 | 67–100 | Chesapeake Bay (10 m depth) | ||
| Clofibric acid | 60–120 | 70 | Lake Greifensee | [39] |
| Contaminant | (Units of L mol−1 s−1) | (Unitless) | ||
|---|---|---|---|---|
| ΦC | ||||
| CLO | 1.2 × 1010 [39] | Negligible [39] | φ∙3.6 × 109 [73] | 5.5 × 10−3 [39] |
| IBP | 1.2 × 1010 [36] | 1.2 × 106 [74] | 1.5 × 109 [73] | 0.33 [36] |
| ATZ | 3.0 × 109 [75] | 6.2 × 106 [76] | 7.2 × 108 [73] | 1.6 × 10−2 [38] |
| CBZ | 9.0 × 109 [77,78,79] | 4.2 × 106 [74] | 7.6 × 108 [37] | 7.8 × 10−4 [37] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Carena, L.; Vione, D. Mapping the Photochemistry of European Mid-Latitudes Rivers: An Assessment of Their Ability to Photodegrade Contaminants. Molecules 2020, 25, 424. https://doi.org/10.3390/molecules25020424
Carena L, Vione D. Mapping the Photochemistry of European Mid-Latitudes Rivers: An Assessment of Their Ability to Photodegrade Contaminants. Molecules. 2020; 25(2):424. https://doi.org/10.3390/molecules25020424
Chicago/Turabian StyleCarena, Luca, and Davide Vione. 2020. "Mapping the Photochemistry of European Mid-Latitudes Rivers: An Assessment of Their Ability to Photodegrade Contaminants" Molecules 25, no. 2: 424. https://doi.org/10.3390/molecules25020424
APA StyleCarena, L., & Vione, D. (2020). Mapping the Photochemistry of European Mid-Latitudes Rivers: An Assessment of Their Ability to Photodegrade Contaminants. Molecules, 25(2), 424. https://doi.org/10.3390/molecules25020424







