Structural Study on Hypochlorous Acid-Mediated Chlorination of Betanin and Its Decarboxylated Derivatives from an Anti-Inflammatory Beta vulgaris L. Extract
Abstract
1. Introduction
2. Results and Discussion
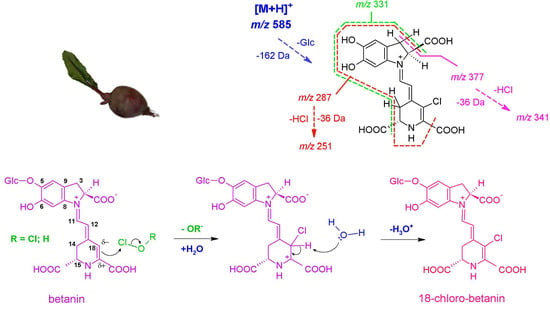
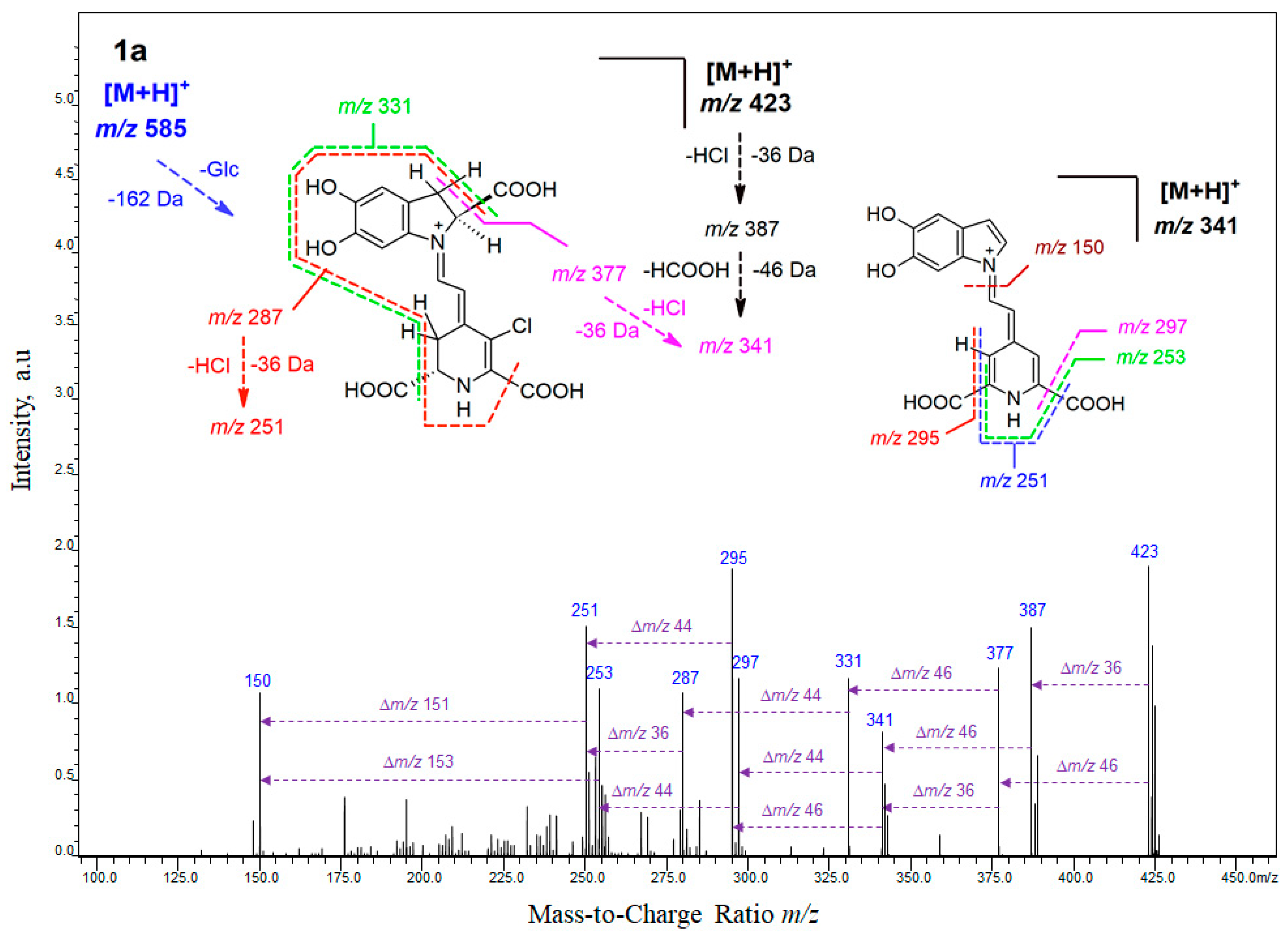
2.1. High-Resolution Mass Spectrometric Study on Mono-Chlorinated Betanin and Its Decarboxylated Derivatives
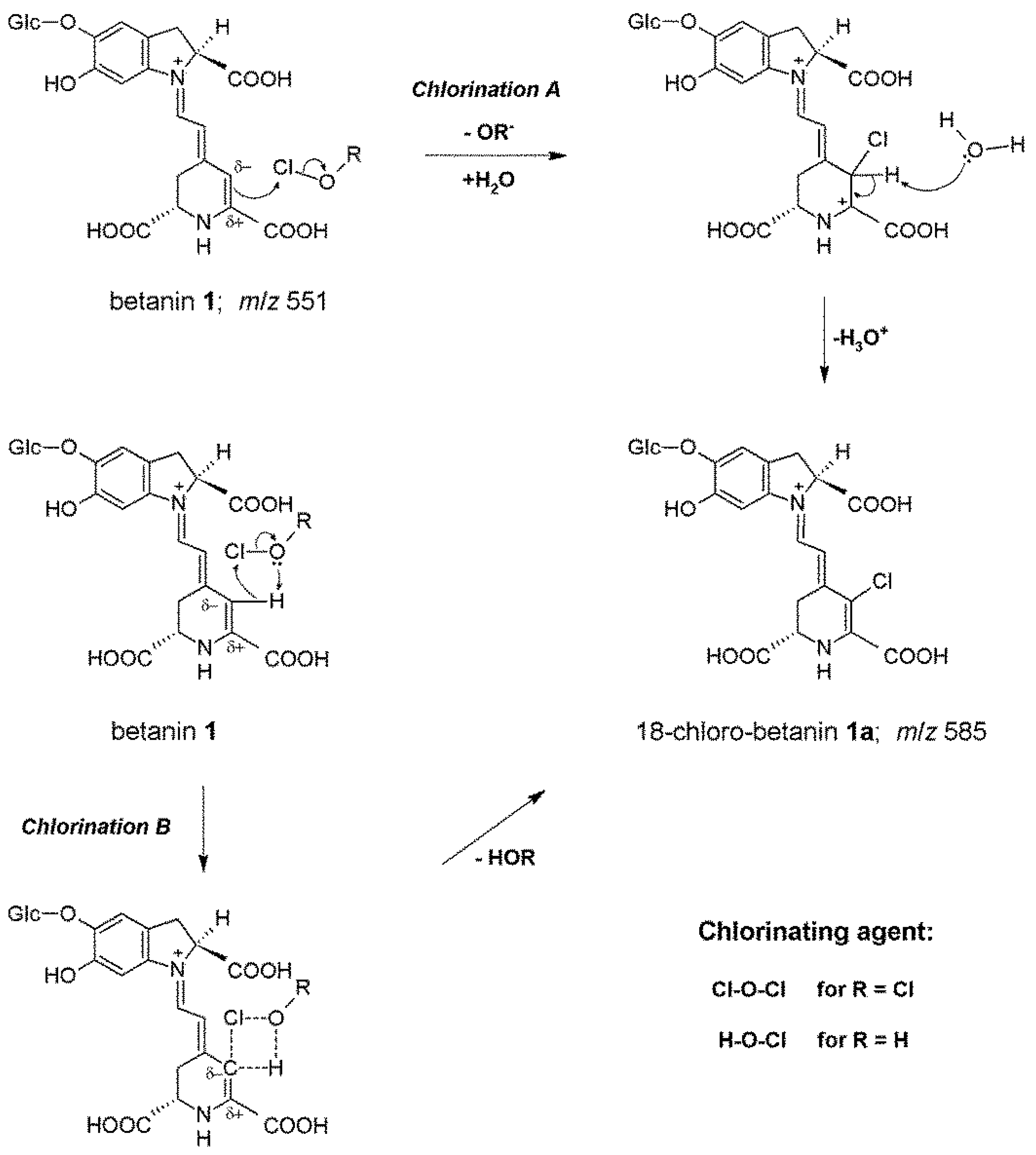
2.2. NMR Structural Elucidation of 18-Chloro-Betanin and Its Decarboxylated Derivatives
3. Materials and Methods
3.1. Reagents
3.2. Preparation of Betalains from Beta vulgaris L. Extracts
3.3. Chromatographic and Mass Spectrometric Determination of the Identity of the Generated Chlorinated Betacyanins in the Chemical (NaOCl) and Enzymatic Systems (MPO)
3.4. Preparative Chlorination of Betanin and Its Decarboxylated Derivatives
3.5. Chromatographic Analysis by LC-DAD-ESI-MS/MS System
3.6. NMR Experiments
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Khan, M.I.; Giridhar, P. Plant betalains: Chemistry and biochemistry. Phytochemistry 2015, 117, 267–295. [Google Scholar] [CrossRef] [PubMed]
- Azeredo, H.M.C. Betalains: Properties, sources, applications, and stability—A review. Int. J. Food Sci. Technol. 2009, 44, 2365–2376. [Google Scholar] [CrossRef]
- Gandía-Herrero, F.; Escribano, J.; García-Carmona, F. Biological activities of plant pigments betalains. Crit. Rev. Food Sci. 2016, 56, 937–945. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.I. Plant betalains: Safety, antioxidant activity, clinical efficacy, and bioavailability. Compr. Rev. Food Sci. Food Saf. 2015, 15, 316–330. [Google Scholar] [CrossRef]
- Wybraniec, S.; Starzak, K.; Pietrzkowski, Z. Chlorination of betacyanins in several hypochlorous acid systems. J. Agric. Food Chem. 2016, 64, 2865–2874. [Google Scholar] [CrossRef] [PubMed]
- Wybraniec, S.; Starzak, K.; Szneler, E.; Pietrzkowski, Z. Separation of chlorinated diastereomers of decarboxy-betacyanins in myeloperoxidase catalyzed chlorinated Beta vulgaris L. extract. J. Chromatogr. B 2016, 20, 1036–1037. [Google Scholar] [CrossRef] [PubMed]
- Pietrzkowski, Z.; Nemzer, B.; Michałowski, T.; Wybraniec, S. Influence of betalain-rich extract on reduction of discomfort associated with osteoarthritis. New Med. 2010, 1, 12–17. [Google Scholar]
- Pietrzkowski, Z.; Argumedo, R.; Shu, C.; Nemzer, B.; Wybraniec, S.; Reyes-Izquierdo, T. Betalain-rich red beet concentrate improves reduced knee discomfort and joint function: A double blind, placebo-controlled pilot clinical study. Nutr. Diet. Suppl. 2014, 6, 9–13. [Google Scholar] [CrossRef]
- Kettle, A.J.; Albrett, A.M.; Chapman, A.L.; Dickerhof, N.; Forbes, L.V.; Khalilova, I.; Turner, R. Measuring chlorine bleach in biology and medicine. Biochim. Biophys. Acta Gen. Subj. 2014, 1840, 781–793. [Google Scholar] [CrossRef]
- Klebanoff, S.J. Myeloperoxidase: Friend and foe. J. Leukoc. Biol. 2005, 7, 598–625. [Google Scholar] [CrossRef]
- Zhang, R.; Song, B.; Yuan, J. Bioanalytical methods for hypochlorous acid detection: Recent advances and challenges. Trends Anal. Chem. 2018, 99, 1–33. [Google Scholar] [CrossRef]
- Albrich, J.M.; McCarthy, C.A.; Hurst, J.K. Biological reactivity of hypochlorous acid: Implications for microbicidal mechanisms of leukocyte myeloperoxidase. Proc. Natl. Acad. Sci. USA 1981, 78, 210–214. [Google Scholar] [CrossRef] [PubMed]
- Dennis, W.H.; Olivieri, V.O., Jr.; Krusé, C.W. Reaction of uracil with hypochlorous acid. Water Res. 1979, 13, 357–362. [Google Scholar] [CrossRef]
- Visser, M.C.M.; Winterbourn, C.C. Oxidative damage to fibronectin. I. The effects of the neutrophil myeloperoxidase system and HOCl. Arch. Biochem. Biophys. 1991, 285, 53–59. [Google Scholar] [CrossRef]
- Carr, A.C.; Vissers, M.C.M.; Domigan, N.M.; Winterbourn, C.C. Modification of red cell membrane lipids by hypochlorous acid and haemolysis by preformed lipid chlorohydrins. Redox Rep. 1997, 3, 236–271. [Google Scholar] [CrossRef]
- Nemzer, B.; Pietrzkowski, Z.; Spórna, A.; Stalica, P.; Thresher, W.; Michałowski, T.; Wybraniec, S. Betalainic and nutritional profiles of pigment-enriched red beet root (Beta vulgaris L.) dried extracts. Food Chem. 2011, 127, 42–53. [Google Scholar] [CrossRef]
- Sivey, J.D.; McCullough, C.E.; Roberts, A.L. Chlorine monoxide (Cl2O) and molecular chlorine (Cl2) as active chlorinating agents in reaction of dimethenamid with aqueous free chlorine. Environ. Sci. Technol. 2010, 44, 3357–3362. [Google Scholar] [CrossRef]
- Agrawal, S.; Ingle, N.; Maity, U.; Jasra, R.V.; Munshi, P. Effect of Aqueous HCl with Dissolved Chlorine on Certain Corrosion-Resistant Polymers. ACS Omega 2018, 3, 6692–6702. [Google Scholar] [CrossRef]
- Lau, S.S.; Reber, K.P.; Roberts, A.L. Aqueous Chlorination Kinetics of Cyclic Alkenes-Is HOCl the Only Chlorinating Agent that Matters? Environ. Sci. Technol. 2019, 53, 11133–11141. [Google Scholar] [CrossRef]
- Swain, C.G.; Crist, D.R. Mechanisms of chlorination by hypochlorous acid. The last of chlorinium ion, Cl+. J. Am. Chem. Soc. 1972, 94, 3195–3200. [Google Scholar] [CrossRef]
- Strack, D.; Steglich, W.; Wray, V. Betalains. In Methods in Plant Biochemistry; Dey, P.M., Harborne, J.B., Waterman, P.G., Eds.; Academic Press: London, UK, 1993; Volume 8, pp. 421–450. [Google Scholar]
- Wybraniec, S. Formation of decarboxylated betacyanins in heated purified betacyanin fractions from red beet root (Beta vulgaris L.) monitored by LC−MS/MS. J. Agric. Food Chem. 2005, 59, 3483–3487. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are not available from the authors. |

| No. | Compound | λmax [nm] | m/z [M + H]+ | m/z from MS/MS of [M + H]+ |
|---|---|---|---|---|
| 1 | betanin | 538 | 551 | 389 |
| 1a | 18-chloro-betanin | 522 | 585 | 423 |
| 2 | 17-decarboxy-betanin | 505 | 507 | 345 |
| 2a | 18-chloro-17-decarboxy-betanin | 524 | 541 | 379 |
| 3 | 15-decarboxy-betanin | 528 | 507 | 345 |
| 3a | 18-chloro-15-decarboxy-betanin | 514 | 541 | 379 |
| 4 | 2-decarboxy-betanin | 533 | 507 | 345 |
| 4a | 18-chloro-2-decarboxy-betanin | 525 | 541 | 379 |
| 5 | betanidin | 540 | 389 | 345 |
| 5a | 18-chloro-betanidin | 528 | 423 | 377 |
| 6/6’ | 2,17-bidecarboxy-betanin/-isobetanin | 507 | 463 | 301 |
| 6a | 18-chloro-2,17-bidecarboxy-betanin | 519 | 497 | 335 |
| 7 | neobetanin | 466 | 549 | 387; 343; 299 |
| Pigment and Fragmentation Ions a | [M + H]+ Molecular Formula | [M + H]+ Observed | [M + H]+ Predicted | Error [mDa] | Error [ppm] | MS2 Ions |
|---|---|---|---|---|---|---|
| 18-chloro-betanin (1a) | C24H26ClN2O13 | 585.1127 | 585.1118 | 0.9 | 1.54 | 423; 377; 341; 295 |
| nl: -Glc | C18H16ClN2O8 | 423.0599 | 423.0590 | 0.9 | 2.13 | 387; 377; 341; 331; 297; 295; 287; 253; 251; 150 |
| 18-chloro-17-decarboxy- -betanin (2a) | C23H26ClN2O11 | 541.1211 | 541.1220 | −0.9 | −1.66 | 379; 343; 333; 297; 253 |
| nl: -Glc | C17H16ClN2O6 | 379.0687 | 379.0691 | −0.4 | −1.06 | 343; 333; 299; 297; 287; 255; 253; 150; 132 |
| 18-chloro-15-decarboxy- -betanin (3a) | C23H26ClN2O11 | 541.1234 | 541.1220 | 1.4 | 2.59 | 379; 333; 297; 251 |
| nl: -Glc | C17H16ClN2O6 | 379.0680 | 379.0691 | −1.1 | −2.90 | 343; 333; 297; 287; 253; 251; 150; 132 |
| 18-chloro-2-decarboxy- -betanin (4a) | C23H26ClN2O11 | 541.1239 | 541.1220 | 1.9 | 3.51 | 379; 333; 297; 253 |
| nl: -Glc | C17H16ClN2O6 | 379.0697 | 379.0691 | 0.6 | 1.58 | 343; 333; 297; 253; 150; 132 |
| 18-chloro-2,17-bidecarboxy--betanin (6a) | C22H26ClN2O9 | 497.1339 | 497.1321 | 1.8 | 3.62 | 335; 299; 253 |
| nl: -Glc | C16H16ClN2O4 | 335.0785 | 335.0793 | −0.8 | −2.39 | 299; 289; 255; 253; 150 |
| 18-Chloro-Betanin (1a) | 18-Chloro-17-Decarboxy-Betanin (2a) | 18-Chloro-2,17-Bidecarboxy-Betanin (6a) | ||||
|---|---|---|---|---|---|---|
| No. | 1H NMR a | 13C NMR b,c | 1H NMR a | 13C NMR b,c | 1H NMR a | 13C NMR b,c |
| 2 or 2a/b | 4.90, dd, 2.1; 8.4 | 68.0 | 4.72, dd, 3.1, 10.5 | 67.8 | 4.04, bt | 53.1 |
| 3a/b | 3.60 (overlap) 3.19, dd, 2.5; 16.4 | 36.2 | 3.61 (overlap) 3.18, dd, 3.4, 16.9 | 35.6 | 3.09, bt | 29.5 |
| 4 | 7.12, s | 116.7 | 7.09, s | 115.9 | 6.97, s | 115.9 |
| 5 | 147.1 | 147.2 | 146.1 | |||
| 6 | 149.4 | 148.7 | 147.8 | |||
| 7 | 7.00, s | 102.9 | 7.02, s | 102.6 | 6.82, s | 102.5 |
| 8 | 140.2 | 139.7 | 138.6 | |||
| 9 | 127.3 | 126.8 | 128.5 | |||
| 10 | 178.4 | 177.9 | - | |||
| 11 | 8.29, d, 12.4 | 147.6 | 8.21, d, 12.5 | 146.9 | 8.02, d, 11.9 | 146.7 |
| 12 | 5.97, d, 12.4 | 105.7 | 5.83, d, 12.6 | 104.4 | 5.83, d, 12.0 | 104.7 |
| 13 | 163.8 | 156.8 | 156.1 | |||
| 14a/b | 3.37, bm | 30.8 | 3.22, bm, | 30.2 | 3.18, bm | 30.4 |
| 15a/b | 4.48, t, 7.5 | 55.7 | 4.22, t, 8.5 | 56.3 | 4.22, t, 7.1 | 56.7 |
| 17 | 158.9 | 7.73, s | 156.3 | 7.65, s | 156.2 | |
| 18 | 105.6 | 108.2 | 109.0 | |||
| 19 | 176.7 | 177.1 | 177.8 | |||
| 20 | 169.7 d | - | - | |||
| 1’ | 5.05, d, 6.8 | 104.5 | 5.04, d, 7.2 | 104.2 | 5.05, d, 6.9 | 103.9 |
| 2’ | 3.59 (overlap) | 79.2 | 3.56 (overlap) | 79.1 | 3.58 (overlap) | 79.2 |
| 3’ | 3.52 | 72.3 | 3.49 | 72.2 | 3.51 | 72.6 |
| 4’ | 3.61 (overlap) | 75.8 | 3.58 (overlap) | 75.9 | 3.55 (overlap) | 75.6 |
| 5’ | 3.61 (overlap) | 78.6 | 3.59 (overlap) | 78.4 | 3.60 (overlap) | 78.4 |
| 6’a/b | 3.92, dd, 2.1, 12.5 3.77, dd, 5.4, 12.5 | 63.4 | 3.92, dd, 2.2, 12.5 3.76, dd, 5.4, 12.6 | 63.3 | 3.94, dd, 2,6; 12.3 3.76, dd, 5.3; 12.3 | 63.6 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kumorkiewicz-Jamro, A.; Starzak, K.; Sutor, K.; Nemzer, B.; Pietrzkowski, Z.; Popenda, Ł.; Wybraniec, S. Structural Study on Hypochlorous Acid-Mediated Chlorination of Betanin and Its Decarboxylated Derivatives from an Anti-Inflammatory Beta vulgaris L. Extract. Molecules 2020, 25, 378. https://doi.org/10.3390/molecules25020378
Kumorkiewicz-Jamro A, Starzak K, Sutor K, Nemzer B, Pietrzkowski Z, Popenda Ł, Wybraniec S. Structural Study on Hypochlorous Acid-Mediated Chlorination of Betanin and Its Decarboxylated Derivatives from an Anti-Inflammatory Beta vulgaris L. Extract. Molecules. 2020; 25(2):378. https://doi.org/10.3390/molecules25020378
Chicago/Turabian StyleKumorkiewicz-Jamro, Agnieszka, Karolina Starzak, Katarzyna Sutor, Boris Nemzer, Zbigniew Pietrzkowski, Łukasz Popenda, and Sławomir Wybraniec. 2020. "Structural Study on Hypochlorous Acid-Mediated Chlorination of Betanin and Its Decarboxylated Derivatives from an Anti-Inflammatory Beta vulgaris L. Extract" Molecules 25, no. 2: 378. https://doi.org/10.3390/molecules25020378
APA StyleKumorkiewicz-Jamro, A., Starzak, K., Sutor, K., Nemzer, B., Pietrzkowski, Z., Popenda, Ł., & Wybraniec, S. (2020). Structural Study on Hypochlorous Acid-Mediated Chlorination of Betanin and Its Decarboxylated Derivatives from an Anti-Inflammatory Beta vulgaris L. Extract. Molecules, 25(2), 378. https://doi.org/10.3390/molecules25020378